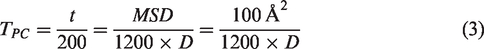Abstract
The diffusion coefficient of C3–C8 normal paraffins in 5A zeolite channels from the simulation ranged from 4.22 × 10−10 cm2/s to 4.68 × 10−14 cm2/s, indicating a slow mass transfer process. In addition, the diffusion rate becomes apparently slower with the increasing carbon numbers of the examined paraffins. In order to improve the adsorption separation efficiency of for n-paraffins, 5A zeolites with mesoporous structures were synthesized by hydrothermal method with the dimethyl octadecyl [3-(trimethoxysilyl)propyl] ammonium chloride as the template agent. Porosity and pore size distribution analysis demonstrated that micropores with the diameter of 0.5 nm were connected with 7–13 nm mesopores in the synthesized samples. The formation of hierarchical pore structure and change of surface morphology of zeolite particles could be controlled by the concentration of [3-(trimethoxysilyl)propyl] ammonium chloride used in the synthesis. The normal paraffin/cyclohexane solutions were used to investigate the adsorption kinetics of pentane, hexane, and heptane on the synthesized 5A zeolites. When the [3-(trimethoxysilyl)propyl] ammonium chloride concentration is 0.01%, the liquid phase diffusion coefficients of n-paraffins in the hierarchical 5A zeolite are 4–48 times larger than those in the microporous 5A zeolite. Our work demonstrates that the diffusion behavior and adsorption kinetics could be greatly improved by introducing mesoporosity into the pore structure of 5A zeolites.
Introduction
Adsorptive separation of hydrocarbons has been widely explored in the petrochemical industry for isolation of normal alkanes, separation of xylene isomers, recovery of aromatics, and molecular management of refining components to improve fuel quality (Azhin et al., 2008; Eldridge, 1993; Faiz and Li, 2012; Herm et al., 2014). Depending on adsorbent properties such as pore size, porosity, and chemical functionalities, and adsorbate properties such as molecular size and chemical nature, three different mechanisms are employed in adsorption processes for separation applications. First, equilibrium-based separation relies on the different adsorption capacities of individual components in a mixture. Second, kinetic-based separation relies on the dissolution and diffusion rate of molecules, in which the component with a slower diffusion rate will be enriched at the effluent of adsorption bed. Third, steric-based separation relies on the difference of molecular size so that smaller molecules can pass through a certain pore size, while larger ones will be excluded (Do, 1998; Ruthven, 1984; Yang, 1997).
Among different porous materials, microporous LTA zeolites, particularly 5A molecular sieve, has been extensively used to separate n-paraffin from naphtha mixtures due to its suitable pore size of 4.4 Å, which allows the intracrystalline adsorption and diffusion of linear alkanes with a cross-sectional diameter of only 4.2 Å, but blocks the larger branched alkanes (Ahmed and Hussein, 2015; Cao and Shen, 2009; Silva et al., 2000; Vavlitis et al., 1981). Thus, many adsorption equilibrium data of different hydrocarbons on 5A zeolites have been collected. F. Miano reported that the saturation adsorption capacity of linear alkanes including C6, C8, C10, C12, C13, and C16 paraffin is from 0.2 to 0.37 mmol/g in the temperature range of 593–653 K (Miano, 1996). Peterson and Redlich estimated that the capacity of n-butane and n-heptane is about 1.36 and 0.95 mmol/g at 333K, respectively (Peterson and Redlich, 1962). Li et al. group reported the adsorption capacity of n-butane and n-heptane, which is from 0.69 to 1.2 mmol/g for butane and 1.1 mmol/g for heptane at 533–653 K, respectively (Li et al., 1998). A much higher capacity of n-butane with 4.32 mmol/g at 308 K was reported by Nitta et al. (1984), which was used later by Silva et al. in a pressure swing adsorption (PSA) modeling for n/iso-paraffin separation (Silva et al., 2000). Besides the equilibrium capacity, kinetic studies have also been performed to understand the diffusion behavior of hydrocarbons on 5A zeolite. Roberts and York reported that the diffusion rate constant of n-hexane on 5A zeolite was 1.94 × 10−4 s−1, while the diffusivity is approximately 2 × 10−12 cm2/s at 298 K (Roberts and York, 1967). The corrected diffusivity of n-pentane, n-octane, and n-decane at 568K was 96 × 10−11, 6.3 × 10−11, and 8.3 × 10−11 cm2/s, respectively, reported by Vavlitis et al. (1981).
Although different macroscopic and microscopic measurement techniques often deliver different kinetic results in terms of diffusion coefficient, 5A zeolites synthesized by conventional methods typically possess only micropores, which have shown many shortcomings in terms of diffusion behavior and separation performance. First, the microporous 5A zeolites have shown a low diffusion rate and long diffusion path length of adsorbates, which causes difficult access to the active sites in the center of zeolite particles, thus a low efficiency for large particles. Second, both rates of adsorption and desorption are limited, resulting in a longer operation time per cycle of PSA. Third, microporous channels are apt to coke formation and pore blockage, causing a lower efficacy and shorter lifetime of adsorbents in industrial conditions (Cao et al., 2014). In this work, the diffusion of different normal paraffins in microporous 5A zeolites was calculated using Transition State Theory. The self-diffusivity of C3–C8 normal paraffins in 5A zeolites was 4.22 × 10−10 cm2/s to 4.68 × 10−14 cm2/s at 450 K indicating a strong need to add mesoporosity to microporous 5A zeolite particles so that mass transfer rate can be improved without the molecular sieving effects being compromised. We report synthesis and characterization of hierarchical 5A zeolites, and compare the adsorption equilibrium and kinetic behavior between hierarchical and microporous 5A zeolites. The effective diffusivities are also reported.
Materials and methods
Chemicals
The chemicals were purchased from commercial vendors and used without further purification, which include sodium aluminate (98.5%), sodium metasilicate nonahydrate (98.5%), sodium hydroxide (99%), [3-(trimethoxysilyl)propyl]octadecyldimethyl ammonium chloride (TPOAC) (60% in methanol), calcium chloride (98.5%), polyethylene glycol 10000, hexadecyl-trimethyl-ammonium bromide (99%), hexadecyltrimethoxysilane (95%), poly(ethylene glycol)-block-poly(propylene glycol)-block-poly(ethylene glycol) (98%), nitric acid (65%), methanol (99.5%). The hydrocarbons used include pentane (99.5%), hexane (98.5%), heptane (99.5%), and cyclohexane (99.5%).
Synthesis of hierarchical 5A zeolites
The synthesis procedure of hierarchical 5A zeolites includes steps of sol–gel formation, crystallization, washing, template removal, ion exchange, and activation (Sun et al., 2008). In a typical synthesis, a mixture of TPOAC, sodium metasilicate, and sodium hydroxide was dissolved in 100 mL water as silicon source, and sodium aluminate was dissolved in 50 mL water as aluminum source. The solution of aluminum source was added to the solution of silicon source dropwise at 40°C, and a vigorous agitation was carried out for 10 minutes. The mixed solution was transferred to a hydrothermal reactor and heated up and kept at 105°C for 6 hours. The obtained crystals were filtered out and washed by methanol and deionized water multiple times until the pH value of the solution is about 8.5. The crystal powders were transferred to an oven and dried for 6 hours at 100°C, and baked at 550°C for 4 hours. The ion exchange was carried out with a 1:20 ratio of solid to liquid in a 0.5 M calcium chloride solution at 55°C for 1 hour. The ion-exchanged solids were activated at 450°C for 3 hours to obtain the mesoporous 5A zeolites.
Characterization
Powder X-ray diffraction (XRD) of zeolites was performed on Rigaku D/max 2550VB/PC diffractometer (Cu Kα radiation, 40 kV, 250 mA). Scanning electron microscopy (SEM) images were collected using JOEL JSM-6360LV Scanning Electron Microscope. Surface area and porosity analysis were performed on Micromeritics ASAP 2020. The zeolite samples were degassed at 300°C for 6 hours before the test, and N2 adsorption isotherms at −196°C were collected at the pressure range of p/p0 = 10−6 ∼ 0.95. The BET method was used to estimate the surface area, and a density functional theory (DFT) method was employed to analyze the pore size and distribution.
Adsorption of hydrocarbons in liquid phase
The study of adsorption kinetics was carried out in a setup as shown in Figure 1. In a typical test, 10 g of zeolites was put into the sample holder of the autoclave. At time zero, 100 g of the mixture solution including 5% examined hydrocarbon (pentane, hexane, or heptane) and cyclohexane was pumped into the autoclave to start the adsorption process. The solution was sampled and analyzed using gas chromatography, and the adsorbed amount of hydrocarbons can be calculated by the equation
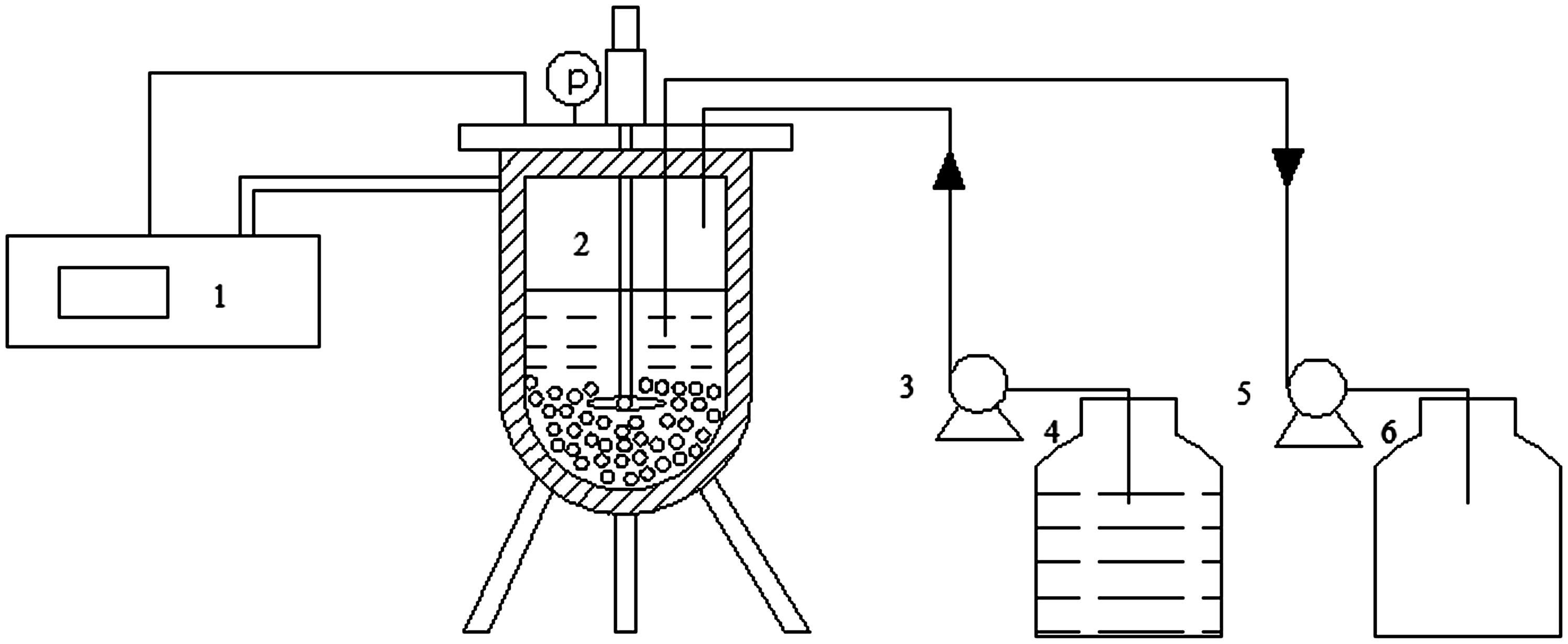
Setup for liquid adsorption experiments: (1) Temperature control system, (2) autoclave, (3) feed pump, (4) feed tank, (5) sample pump, and (6) sample tank.
Simulation: Molecular dynamics and transition-state theory
Molecular dynamics (MD) has been used as a common method to estimate the theoretical diffusivity of molecules. In this work, MD simulation was carried out using the Forcite module of materials Studio 5.5 (Accelrys Software Inc.), with the Compass27 force field and the NVT ensemble. The parameters of Lennard–Jones potentials could be found from our previous publications (Liu et al., 2017). However, a big challenge with MD simulation is their high demand in computing time. In particular, for diffusion of strongly interacting molecules inside a porous crystal, MD simulation is often too slow to be a feasible approach because the molecule spends most of its time executing fast local motions trapped within favorable spots of the crystal framework, and only infrequently jumps from one such spot to another by overcoming a barrier in the energy associated with its translational motion. Thus, when the diffusion coefficients are very small (<10−8 cm2/s), it will take a much longer time to finish the simulation causing the well-known “MD time scale problem” (Dubbeldam et al, 2005). Based on the computing power, we used for this work, it took 1 hour to simulate a 200 ps molecular dynamic process for a single molecule in a one-dimensional channel of 100 Å in length. For paraffins with diffusion coefficients around 10−6 to 10−15 cm2/s, the effective diffusion time, which requires the moving distance of paraffin molecules larger than 10 Å, namely mean square displacement (MSD) > 100 Å2, could be calculated using the following equation
The real computing time (Tpc) is estimated using the following equation, and the results are listed in Table 1
The correlation between computing time and diffusion coefficients.

As shown in Table 1, the computing time will increase rapidly with the decrease of the diffusion coefficients, which makes the direct MD simulation of paraffin diffusion in zeolites almost infeasible.
Transition-state theory (TST) has been proposed and successfully applied to simulate the diffusion in zeolites, in which cage-to-cage jumps were found to be the rate-determining step of the diffusion process. The examined system normally includes two stable states (cages), A and B, with a dividing free energy barrier between them (Figure 2). In this case, diffusion can be regarded as an activated process, in which the adsorbate molecules jump from one cage to the next, and the actual crossing time is negligible compared to the time a molecule spends inside the cage. The diffusion coordinate is denoted by q, indicating the progress of the diffusion from cage A to cage B, while q* is the location of the dividing surface. Two characteristic functions nA and nB are defined using the following equation to measure whether the system is in state A or B



Typical snapshot of methane diffusion in 5A zeolites.
By assuming that the molecular velocities follow a Maxwell–Boltzmann distribution, the rate constant for transition of molecules over the energy barrier is given using the Bennett–Chandler approach (Abouelnasr and Smit, 2012)
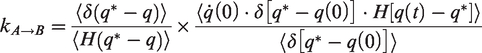

Under the condition of infinite dilution, intermolecular correlations are negligible so that a random walk process can be used to model the molecular crossing through the cages. The self-diffusivity is given by

Results and discussion
Effects of TPOAC concentration on porosity and morphology of 5A zeolite particles
A series of 5A zeolites were synthesized with the molar ratio of SiO2 : Na2O : Al2O3 : H2O :TPOAC = 100 : 333 : 67 : 20000 : n to obtain T-n samples. For example, T-0 is the microporous 5A zeolites without using TPOAC template, and T-0.001 is the 5A zeolites synthesized with the concentration of 0.001% in respect to SiO2. The powder XRD patterns of 5A zeolites synthesized using different TPOAC concentrations (0.001–0.4%) are given in Figure 3, in which all samples showed the same pattern as seen in the microporous 5A zeolites indicating they all have the exact same crystal lattices.
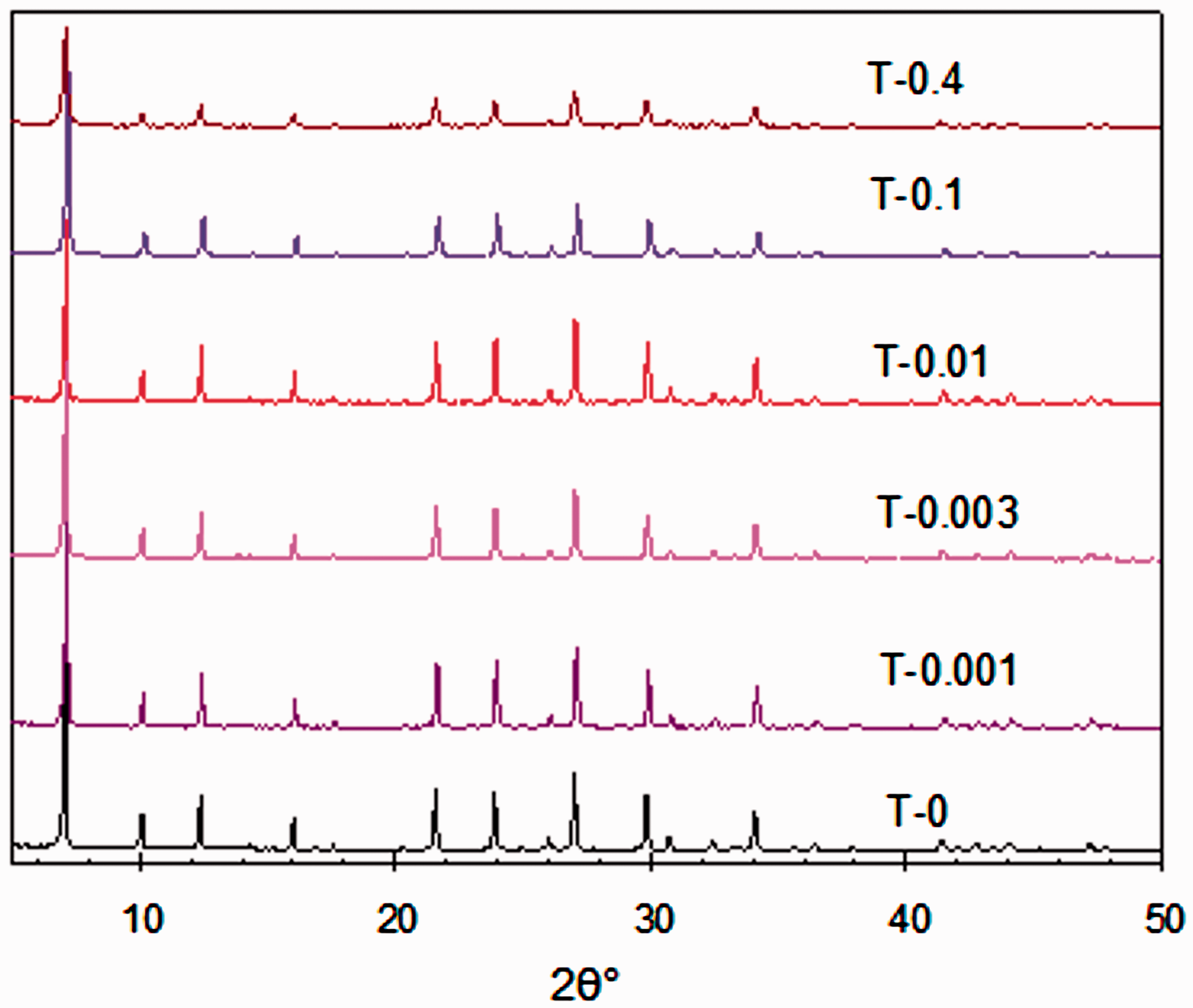
Powder XRD patterns of zeolites synthesized with different amounts of TPOAC show that all synthesized samples have the same crystal structure of 5A zeolites.
The effects of TPOAC concentration on surface morphology and particle size of synthesized zeolites are revealed using SEM images. As shown in Figure 4, the particle of microporous 5A zeolites (T-0) is cubic with truncated edges (Archimedean truncated cube). The particle size is about 2–3 µm with a smooth surface. The particle of T-0.001 sample shows the same Archimedean truncated cube morphology with some small pores appeared at the surface. With the increase of the TPOAC concentration used in the synthesis, more pores were created at the surface and the surface roughness increased. Moreover, the whole particle lost the neat surface and became rounded. However, the average particle size is the same as the microporous zeolites as shown in the last two SEM images in Figure 4.

SEM images of 5A zeolites synthesized with different amounts of TPOAC demonstrate the surface morphology changing with TPOAC concentration.
N2 isotherms at 77 K revealed the porosity and pore size change of synthesized samples with the varying TPOAC concentration. As shown in Figure 5, T-0 sample showed a Type I isotherm, in which the adsorption mainly happens at the relative lower pressure (P/P0 < 0.01). After the first stage of adsorption, the isotherm curve is nearly stable with a slight increase with the pressure until all micropores are saturated (P/P0 = 1.0), and desorption branch shows a totally reversible process, which is a typical behavior of microporous zeolite materials. When TPOAC was used in the synthesis to create mesopore in the particle, the isotherms showed a combination of Type I and IV curves. At the lower pressure, micropore filling still played a major role so that the adsorbed amount quickly increased with the pressure. However, with the increasing pressure, a hysteresis loop is observed between adsorption and desorption branches indicating that capillary condensation occurred in the mesopores and the level of hysteresis indicates the amount of mesopores in the particle. From the isotherms, we can tell that more mesopores exist at higher concentration of TPOAC indicating that hierarchical porous structures were successfully created in 5A zeolites.

N2 isotherms of synthesized 5A zeolites at 77 K indicating that more mesopores were created with the increasing amount of TPOAC used in the synthesis.
Analysis of pore size distribution using the nonlocal density functional theory (NLDFT) method based on cylindrical pore model further confirmed the existence of mesopores in the 5A zeolites synthesized using TPOAC. As shown in Figure 6, T-0 samples possess only micropores 0.5 nm in diameter, while samples synthesized using TPOAC demonstrated the existence of mesopores near 7–13 nm in diameter. Moreover, the pore volume of mesopore increases with the concentration of TPOAC. The pore structure parameters are listed in Table 2. The changing microporous surface area shows a maximum value with the increasing concentration of TPOAC; however, most microporous structures have been reserved in the different samples. With the increasing amount of TPOAC used in the synthesis, mesoporous surface area and mesopore volume increased. At the highest concentration (T-0.4), the ratio between mesopore and micropore volume is around 0.3.
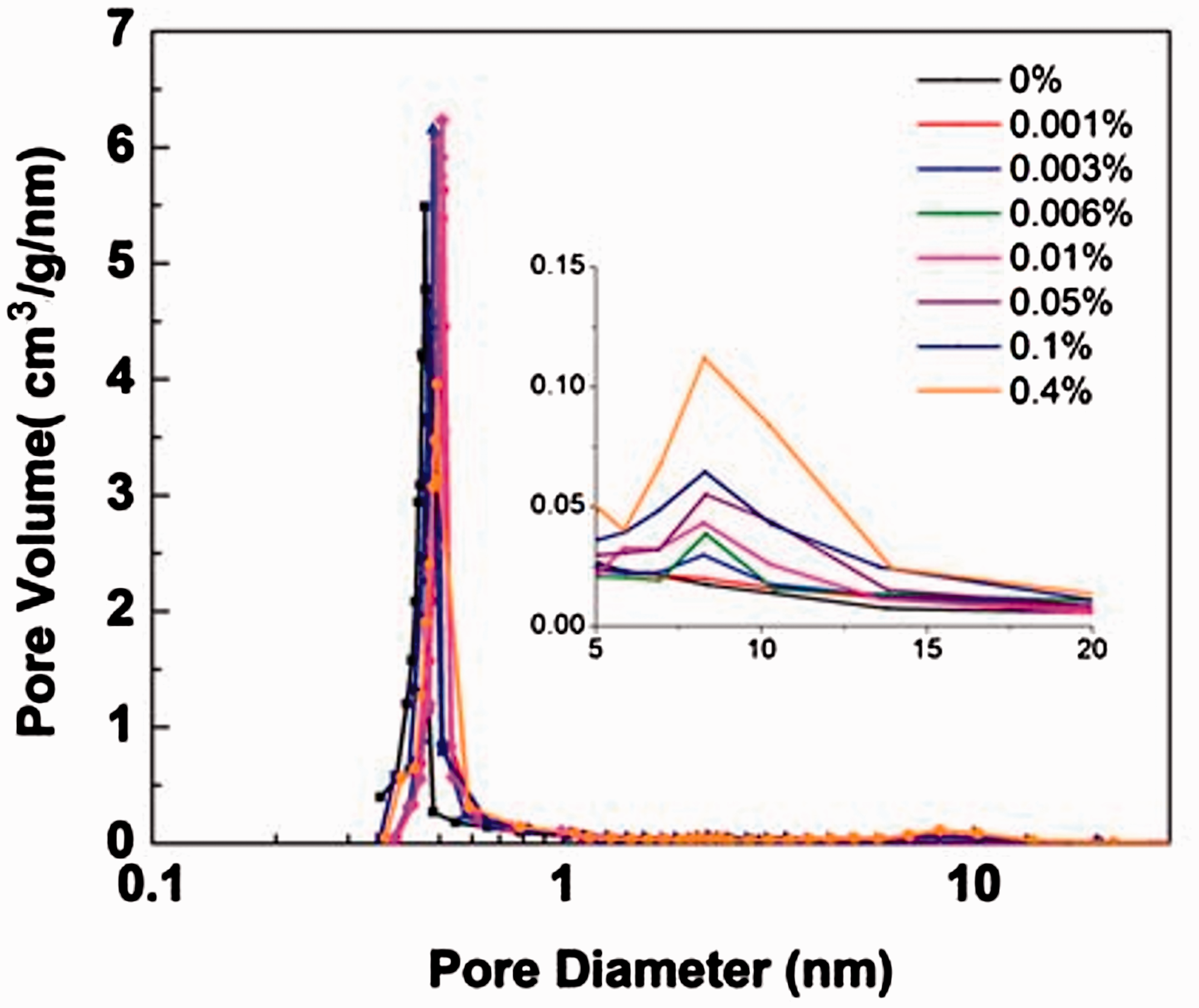
Pore size distribution of 5A zeolites synthesized with different amounts of TPOAC demonstrates the pore volume of mesopores increases with the concentration of TPOAC used in synthesis.
Pore structure parameters of synthesized zeolites.

Cho synthesized LTA zeolites with mesopores using organosilane surfactant as templates. The typical zeolites had the mesoporous surface of 52 m2/g and the mesopore diameters of 6.8 nm (Cho et al., 2009). The mesoporous structures are similar to that of the synthesized zeolites in this paper. Tromp et al. synthesized Mordenite with mesopores by acid leaching, which had the mesoporous surface of 30 m2/g, the micropore volume of 0.19 cm3/g, and the mesopore volume of 0.04 cm3/g (Tromp et al., 2000). The mesopores generated using template method are similar to that from acid leaching method.
Diffusion coefficient of normal paraffins in microporous 5A zeolites
The self-diffusion coefficients of normal paraffins in 5A zeolite at 450 K from the TST simulation are listed in Table 3. The examined paraffins include methane (CH4), ethane (C2H6), propane (C3H8), pentane (C5H12), hexane (C6H14), and octane (C8H18). The calculated diffusion coefficients of methane and ethane are compared with the results from MD simulation, showing good agreements between both methods. The diffusion coefficient of C3–C8 normal paraffins ranges from 4.22 × 10−10 cm2/s to 4.68 × 10−14 cm2/s, indicating a slow mass transfer process. In addition, the diffusion rate becomes apparently slower with the increasing carbon numbers of the examined paraffins. The correlation between carbon numbers of paraffin and the diffusion coefficients is shown in Figure 7, in which the logarithmic scale is used and a straight line is drawn to give the fitting equation

The self-diffusivity of C1–C8 n-paraffins from transition-state theory (TST) and molecular dynamics (MD) simulations.


The correlation between carbon numbers of paraffin and the diffusion coefficients, in which the insert used a logarithmic scale and a linear line is drawn to give the fitting equation of y = 1.176 ×10−8·e(−1.331×).
Effects of mesopore structure of 5A zeolites on adsorption of normal parrafins
Different 5A zeolites synthesized in this work have a similar adsorption capacity (13 wt%) of n-paraffins. However, a slightly decreasing trend of capacity is observed with the increasing concentration of TPOAC used in the synthesis of zeolites because the appearance of mesopore may lower the total surface area of available micropores in the particles, which become apparently worse as observed in the case of T-0.4 samples as shown in Table 4.
The adsorption capacities of n-paraffins in different hierarchical 5A zeolites.

The kinetic adsorption curves of pentane, hexane, and heptane at 303 K on different samples demonstrated the effects of pore structure on adsorption rates. As shown in Figure 8, the adsorption of n-pentane takes 80 minutes to achieve the equilibrium on microporous 5A zeolites (T-0 sample), 20 minutes on T-0.001 samples, and 2 minutes on T-0.01 samples, respectively. The adsorption of n-hexane will take longer time to achieve the equilibrium, in which 100 minutes are needed on T-0 samples, 40 minutes on T-0.001 samples, and about 10 minutes on T-0.01 samples. The adsorption of n-heptane will take more than 200 minutes to achieve the equilibrium due to the longer hydrocarbon chain. The equilibrium adsorption time is shortened to 80 minutes on T-0.003 samples, and 20 minutes on T-0.01 samples. In all cases, the adsorption rate was apparently increased by creating mesopores in the particles, although the equilibrium time increases with the carbon numbers of paraffins.

Adsorption kinetics of pentane (a), hexane (b), and heptane (c) at 303 K on different samples demonstrated the effects of pore structure on adsorption rates.
Effects of mesopore structure of 5A zeolites on effective diffusivity
Effective diffusion coefficients can be estimated using adsorption kinetic models. To model the adsorption kinetics in a batch system with diffusion equations, some approximations used include: (1) heat transfer is assumed to be sufficiently fast compared to the mass transfer in porous materials; (2) an equivalent sphere will be used to represent the real geometry of solid particles; (3) diffusivity is concentration independent over a small change in adsorbed phase concentration; (4) concentration of adsorbate is constant at the surface of adsorbent. The solution for the adsorption uptake with the time is given according to the approach of Crank (1975), and has been used for many different adsorption systems (Armstrong et al., 2017). The adsorbed amount 

By fitting the kinetic data into the model, the diffusion coefficients were obtained, and the results are listed in Table 5. The liquid diffusion coefficient of pentane, hexane, and heptane on T-0 samples is 5.37 × 10−12, 2.91 × 10−12, and 4.89 × 10−13 cm2/s, respectively. It is apparent that the diffusion coefficient decreases with the increase of carbon numbers of n-paraffins in microporous 5A zeolites. However, the mesopore created in the particle improved the diffusion coefficient to a great level. The diffusion coefficient of pentane, hexane, and heptane on T-0.001 samples increases to 159.21 × 10−13, 53.3 × 10−13, and 13.29 × 1013 cm2/s, respectively, which is about 1.8–3 times larger than those on T-0 samples. With the further increase of TPOAC used in the synthesis, diffusion coefficients of three examined paraffins increase until approaching the value of 2.89 × 1011 cm2/s. When the concentration of TPOAC was above 0.01%, the diffusion coefficient slightly increases and the value among different paraffins is approaching the same, which is different in the case of microporous 5A zeolites. It is because the mesopore diameter is around 7–13 nm, much larger than the chain length of the examined paraffins so that the diffusion behavior of paraffins are affected less by the pore size. Thus, the diffusion coefficient of paraffins is approaching the same value with the increasing number of mesopores in 5A zeolites.
The effective diffusivity of n-paraffins in different hierarchical 5A zeolites.

Conclusions
A series of 5A zeolites possessing micropores and mesopores have been synthesized using TPOAC as the template agent. With increasing amounts of TPOAC used in the synthesis, the surface area, and pore volume of mesopores increase so that a hierarchical porous structure with major 0.5 nm micropores connected by 7–13 nm mesopores have been created.
Compared to microporous 5A zeolites, the hierarchical porous structure provides faster diffusion for the examined normal paraffins. When the concentration of used TPOAC was 0.001% (T-0.001 sample), the diffusion coefficient of pentane, hexane, and heptane increases to 159.21 × 10−13, 53.3 × 10−13, and 13.29 × 1013 cm2/s, respectively, which is about 1.8–3 times larger than their respective microporous samples. Further increase of TPOAC will continue enhancing the mesoporosity of zeolites and therefore diffusion coefficients, which will achieve a diffusivity improvement of one order of magnitude. However, serious surface defects have been observed at higher concentration of TPOAC, as seen from SEM images. In addition, further increase of TPOAC concentration above 0.01% only slightly improved the diffusion coefficients by 11% by increasing TPOAC concentration to 0.4%, and the difference of diffusion coefficient among normal paraffins are diminished as we see that diffusion coefficients of three examined paraffins finally approach the same value of about 2.89 × 1011 cm2/s. By considering all factors, the samples using <0.01% TPOAC are regarded as the best samples for adsorption separation of examined normal paraffins due to the regular particle shape, great improvement of diffusion coefficient, and reasonable loss of adsorption capacity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support Joint Fund by the National Natural Science Foundation of China and PetroChina (Project U1862204) and the Training Program of the Major Research Plan of the National Natural Science Foundation of China (Grant 91634112) is gratefully acknowledged.