Abstract
Thorium is a promising nuclear fuel; however, even a small amount in nuclear wastewater can cause significant radioactive pollution. Therefore, it is urgent to develop effective adsorbents for the efficient removal of thorium to solve this problem. Herein, the phytic acid modification of carboxylated Zirconium-based metal-organic framework materials (UiO-66-COOH-PA) has been obtained by rapid high-temperature carbonization, which exhibits excellent performance in removing thorium (IV) in acidic aqueous solutions. The maximum adsorption capacity of UiO-66-COOH-PA-2 (with 40% mass ratio of phytic acid) in solution reaches 315.5 mg/g at a pH of 4, which is more than twice that of the unmodified Zr-based metal organic framework (MOF) material. The adsorption kinetics of UiO-66-COOH-PA on thorium has been analyzed using two different isothermal adsorption models, the Langmuir and Freundlich isotherm adsorption models. It turns out that the Freundlich model is more suitable for the data. The thorium adsorption process reaches dynamic equilibrium very quickly, while the adsorption dynamics of UiO-66-COOH-PA on thorium follows the pseudo-second-order adsorption kinetic model. The excellent adsorption performance of UiO-66-COOH-PA-2 at low pH values indicates that adsorption material containing phosphorus is suitable for the removal of thorium especially in acidic systems. It also exhibits excellent selectivity (up to 90.2%) for Th(IV) adsorption in the presence of several coexisting rare-earth ions. Overall, the integration of phosphorus-modified in Zr-MOFs represents a significant advancement in thorium adsorption technology, offering a pathway toward more effective management of radioactive acidic waste liquid.
By introducing functional groups such as phosphoric or phosphonic acid groups, the adsorption capacity of the material for thorium ions is greatly enhanced, demonstrating its broad potential for nuclear waste treatment and radioactive wastewater purification. The research indicates that phosphorus functionalization not only optimizes the adsorption kinetics and enhances selectivity but also improves the material's chemical stability in complex environments, especially under acidic conditions. In general, this study provides a foundational basis for future exploration into optimizing these materials for real-world applications, addressing challenges related to scalability and long-term performance.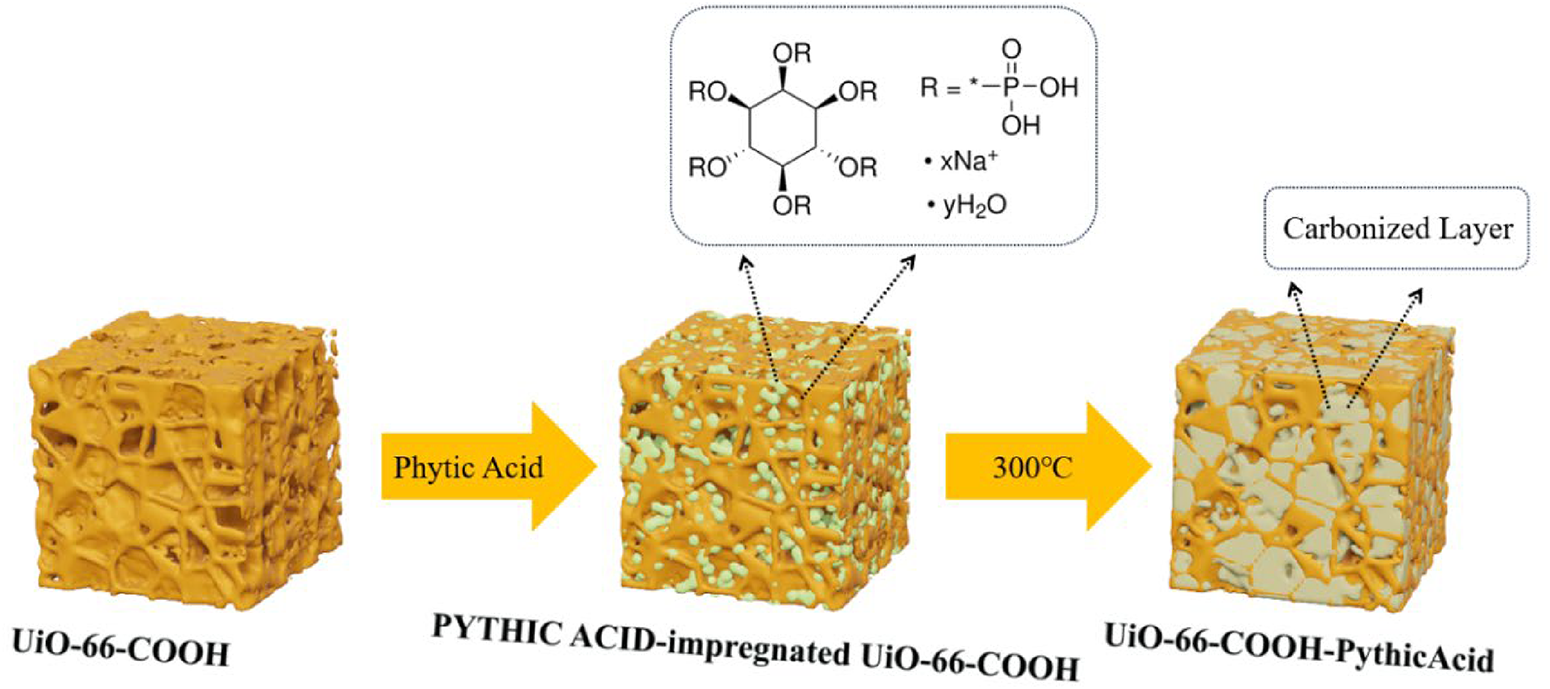
Introduction
As society advances and progresses, human society is confronted with enormous challenges in sustainable energy development (Luo et al., 2022). Traditional fossil fuels such as oil, coal, and natural gas are considered as nonrenewable sources (Liu et al., 2022). In contrast, nuclear energy is often considered as a clean energy source due to its ability to efficiently generate large amounts of electricity while avoiding the production of greenhouse gases (Zhang et al., 2017). Therefore, nuclear energy, as an efficient and clean energy source, has received much attention in recent years (Degueldre et al., 2011). Thorium has an abundance more than twice that of uranium, contributing to higher material reserves compared to uranium (Ashley et al., 2012). Furthermore, thorium (IV) can effectively reduce the production of highly radioactive fission by-products in the actinide series, making it a promising alternative to nuclear fuel (Arnold et al., 2014). This renders thorium one of the most promising nuclear fuels. However, it is no deny that using nuclear energy leads to generating radioactive pollution. The ensuing challenges of contamination in the environment and the treatment of nuclear wastewater present significant obstacles for humanity (Zhao et al., 2021). For instance, prolonged exposure to radioactive environments leads to an increase in the risk of cancer and poses potential harm to the kidneys. Persistent ingestion can lead to the accumulation of internal irradiation and chemical toxicity in the body (Yang et al., 2019). Hence, there is an urgent need to explore novel materials to remove radioactive thorium from the environment.
Thorium, as a representative element in the actinide series, is often found coexisting with transition metals such as lanthanides in rocks and soil (Gadd and White, 1989; Maes et al., 2017). During the mining and extraction process of thorium ore, a large amount of radioactive wastewater containing thorium will usually inevitably be generated. Although existing research has demonstrated the excellent performance of phosphorus-functionalized Zr-based metal organic frameworks (MOFs) in uranium adsorption (Yan et al., 2022; Tripathi et al., 2020), establishing a foundation for their application in the removal of radioactive metal ions (Pooley et al., 2018), systematic studies on thorium have not received adequate attention. In comparison to uranium studies, thorium possesses unique chemical properties and migration behaviors in aquatic systems, which is more prone to complex with hydroxide ions, which poses greater challenges for thorium removal in low-concentration and weakly acidic environments (Zahakifar and Khanramaki, 2024). This research gap may stem from a limited understanding of thorium's environmental behavior and toxicological characteristics on the condition of its relatively low concentrations and occurrence forms, resulting in a lower prioritization in water treatment research.
At present, the methods for treating radioactive wastewater mainly include precipitation, solvent extraction, ion exchange, membrane adsorption (Li et al., 2024), chemical adsorption (Sun et al., 2022), and other techniques. Adsorption method has become one of the most prominent methods for separating radioactive nuclides due to its unique features such as strong practicality, easy operation, low cost, and good removal effect from the wastewater. With the rapid development of materials science, more and more adsorption materials are currently employed to enrich radioactive elements in aquatic environments (Wang et al., 2022; Alotaibi and Ismail, 2022; Peng et al., 2015). Common adsorption materials include activated carbon (Pavithra et al., 2024), graphene oxide (Wang et al., 2016; Yang et al., 2019), graphitic carbon nitride (g-C3N4) (Dong et al., 2022), ferric oxide (Fe3O4) nanoparticles (Zhou et al., 2009), two-dimensional nanosheets (Li et al., 2018), polyelectrolyte nano-thin film (Aslani et al., 2017), and MOFs (Jia et al., 2021). Among them, MOFs are composed of metal ions/clusters and organic linkers, which can form highly flexible frameworks with adjustable pore size, pore volume, stiffness, and other modifiable functions (Zulkifli et al., 2022). Over the past few decades, MOFs have been demonstrated to exhibit outstanding performance in adsorption (Lee et al., 2014), catalysis (Liu et al., 2021), sensors (Yue et al., 2021), and other various fields. To date, a number of adsorbents have been applied in wastewater treatment.
Zirconium-based (Zr-based) MOF materials have garnered significant attention due to their high chemical stability (Ding et al., 2023) and excellent acid stability (Morris et al., 2017). Through long-term water immersion experiments, it has been verified to maintain its structural characteristics in water environments for a long time (Ahmadijokani et al., 2020). Structural defects allow functional groups on the surface and the bulk to be utilized for functional modification (Yan et al., 2022). Considering the robust adsorption affinity of phosphorus-functionalized groups toward actinide elements (Yang et al., 2017), phosphorus-functionalized groups can be introduced into the pores of MOFs to enhance their adsorption performance. The inherent high specific surface area of MOFs can be modified with functional groups containing active sites to improve the overall selectivity and adsorption capacity of the material (Lou et al., 2021; Zhang et al., 2021; Zhu et al., 2018).
Phytic acid (PA) is a natural substance primarily found in plant seeds. Due to the abundant phosphate groups in PA, it can easily chelate with metal ions in aqueous solutions through the surface complexation and electrostatic adsorption (Sfameni et al., 2022). On the one hand, PA has been demonstrated as an effective functionalizing moiety in the study of uranium adsorption experimental research (Bi et al., 2021). On the other hand, thorium in the natural environment typically exists in the tetravalent state. The phosphate groups in the PA molecule, which serve as Lewis acid, strongly attract thorium ions (Peng et al., 2021). Hence, we can use the physicochemical properties of the PA to construct phosphorus-modified MOF adsorbents, providing abundant active sites for cationic nuclides (Liu et al., 2015).
In this study, a phosphorus-modified MOF material was constructed using a post-synthesis modification method for investigating the adsorption behavior of thorium in weakly acidic environments. It has been proven to be an efficient modification method in improving the adsorption capacity of MOF materials to be highly efficient for enhancing the adsorption capacity of MOF materials (Khan and Jhung, 2019; Kim and Cohen, 2012; Li et al., 2016; Liu et al., 2018). Subsequently, key factors affecting the adsorption behavior of materials were explored, the modification dosage, pH value, temperature, contact time, and initial concentration. Additionally, detailed investigations into the physicochemical properties of the adsorbents were carried out using characterization methods such as scanning electron microscopy (SEM), transmission electron microscopy (TEM) with mapping EDS, Fourier transform infrared spectroscopy (FT-IR), X-ray diffraction (XRD), and X-ray photoelectron spectroscopy (XPS), respectively. A comparative analysis was conducted on the adsorption performance of PA modified MOF (UiO-66-COOH-PA) and unmodified Zr-based MOF material (UiO-66-COOH) for the efficient recovery of thorium in weakly acidic water environments. The adsorption kinetics, isotherm models, and thermodynamics were also evaluated to understand the adsorption process. Finally, the recycling regeneration of adsorbents was analyzed through the experiments of adsorption/desorption cycle regeneration. Finally, through adsorption/desorption cycle regeneration experiments, the cyclic regeneration ability of the synthesized adsorption material has been analyzed in detail, thus exploring its potential application for Th(IV) removal in acidic radioactive wastewater environment.
Experimental
Materials
The reagents used in the experiment were of analytical grade, including zirconium tetrachloride (ZrCl4, >98.5%), PA (70%), and gadolinium (III) nitrate hexahydrate obtained from Adamas. Terephthalic acid (TPA, 98%) and isophthalic acid (IPA, 98%) obtained from Aladdin. N,N-dimethylformamide (DMF) was acquired from a general reagent supplier. Acetic acid was obtained from HUSHI. Thorium (IV) nitrate hydrate, samarium (III) nitrate hexahydrate, lanthanum (III) nitrate hydrate, ytterbium (III) nitrate pentahydrate, and neodymium nitrate hexahydrate (III) were obtained from Macklin. Deionized water (H2O), anhydrous ethanol, concentrated hydrochloric acid (HCl), and solid sodium hydroxide were also employed. The thorium (IV) solution was prepared by dissolving a suitable quantity of thorium nitrate (Th(NO3)4·H2O) in deionized water and adjusting the dilution accordingly.
Characterization analysis
Scanning electron microscopy and TEM were used to observe the surface morphology and elemental distribution of adsorption materials. Different functional groups in the materials were analyzed and identified through Infrared Spectrometer (Vertex70, Bruker Company). The crystallinity of the material was evaluated by XRD at a scanning rate of 5°/min and a spectrum detection range of 10–80° (Bruker Company). X-ray photoelectron spectroscopy (AXIS SUPRA+) was employed to analyze the elemental composition and content of the materials. The specific surface area of the materials was measured through N2 adsorption–desorption isotherms using ASAP 2460 instrument (MicroActive, America). The influence of temperature variation on the material mass was recorded using a Thermal Analysis Instrument. Zeta potential information for the synthesized adsorption materials was recorded at room temperature using a Zetasizer Nano ZS instrument (Malvern, England). The changes of Th(IV) ion concentration in the solution before and after adsorption were detected by using Inductively Coupled Plasma Optical Emission Spectrometry (ICP-OES).
Synthesis of materials
Zr-based MOF material
The carboxylated zirconium-based MOF material UiO-66-COOH was synthesized according to the literature (Liu et al., 2017). In summary, zirconium chloride (2.33 g), TPA (1.22 g), and IPA (0.44 g) were dissolved in 60 mL of DMF. After 15 min of ultrasonic treatment, 2 mL of acetic acid was added as a modulator. The solution was stirred to ensure uniform mixing, then transferred into a stainless steel high-pressure vessel lined with polytetrafluoroethylene and heated at 120°C for 12 h. After cooling to room temperature, the obtained mixture was filtered under reduced pressure, and then followed by repeated washing with DMF and deionized water to obtain a white precipitate (UiO-66-COOH). The obtained material was subjected to vacuum drying at 80°C for 12 h.
Preparation of phosphorus-modified Zr-based MOF materials
Based on the literature (Liu et al., 2018), we rely on rapid high-temperature carbonization to achieve the phosphorus functionalization of the material. An appropriate amount of UiO-66-COOH was dispersed in 50 mL of deionized water. After ultrasonic dispersion, a specific amount of PA was added to achieve mass ratios of 10% and 40% in the mixture, respectively. Phytic acid was liquid, and it also appeared liquid after immersing in MOF. For the convenience of subsequent adsorption experiments, the PA impregnated MOF material was placed and heated in a muffle furnace for 1 min. This treatment could maintain the structure of MOF materials. Unless mentioned, all samples used were treated at 300 °C for 1 min. It was immediately removed when the color of the material changed from white to pale yellow. To control variables, the unmodified UiO-66-COOH underwent the same heat treatment at the same temperature. The obtained solid with deionized water was purified for multiple times, then transferred it to a vacuum-drying oven and heated at 80°C for 12 h. The prepared materials were named as UiO-66-COOH-PA-1 and UiO-66-COOH-PA-2, respectively.
Adsorption experiments
Thorium nitrate solution was prepared by dissolving solid thorium nitrate in deionized water. In the static adsorption experiment, the ratio of the adsorption material mass to thorium nitrate solution volume was 0.22 /L. In batch adsorption experiments, the effects of initial pH (2.5–4.0), initial thorium concentration (5–300 mg/L), different contact times (0–180 min), and various temperatures (298K–318K) on thorium adsorption behavior were investigated. The supernatant was obtained by centrifuging the polyethylene tube at 11,000 r/min for 5 min, and the residual Th(IV) ion concentrations in the supernatant were detected using ICP-OES. Langmuir and Freundlich isotherm models, as well as pseudo-first-order and pseudo-second-order adsorption kinetic models, were employed to fit the experimental data. Further analysis of the thorium adsorption behavior of UiO-66-COOH, UiO-66-COOH-PA-1, and UiO-66-COOH-PA-2 was conducted. The adsorption capacity of the prepared materials for metal ions was calculated using the following formula (1):
Thorium-selectivity (STh) reflected the level of adsorption selectivity of prepared material toward Th(IV), which was calculated using the following formula (2):
Results and discussion
Characterization
The phosphorus-modified MOF materials (UiO-66-COOH-PA) have been synthesized by rapid high-temperature carbonization and is illustrated in detail. By the method of immersion, the PA molecules enter the pores of the MOF. The green particles in step 2 represented the PA molecules, while the dark yellow sheet-like structures in step 3 represents the carbonized layer formed from the PA by rapid high-temperature carbonization, rendering the MOF surface roughened.
In order to investigate the morphology and microstructure of the materials, SEM analysis has been conducted to characterize the synthesized adsorption material. It shows in Figure 1 that UiO-66-COOH exhibits a smooth and irregular spherical structure. Compared to UiO-66-COOH, the morphology and particle size of UiO-66-COOH-PA-1 and UiO-66-COOH-PA-2 modified by PA have not undergone significant changes and maintain an intact structure even after short-term high-temperature treatment. UiO-66-COOH-PA-1 (Figure 1(b)) and UiO-66-COOH-PA-2 (Figure 1(c)) present rough edges, indicating that the PA molecules doped into the UiO-66-COOH channels have successfully formed a carbonized layer after short-term high-temperature treatment. To further explore the spatial elemental distribution of UiO-66-COOH-PA-2, TEM has been conducted for in-depth characterization analysis (Figure 1(d)–(f)). It can be clearly seen from the image that zirconium (Figure 1(e) and (g)) and phosphorus (Figure 1(f) and (g)) elements are uniformly distributed on the surface of UiO-66-COOH-PA-2, suggesting that PA has successfully modified UiO-66COOH nanoparticles.
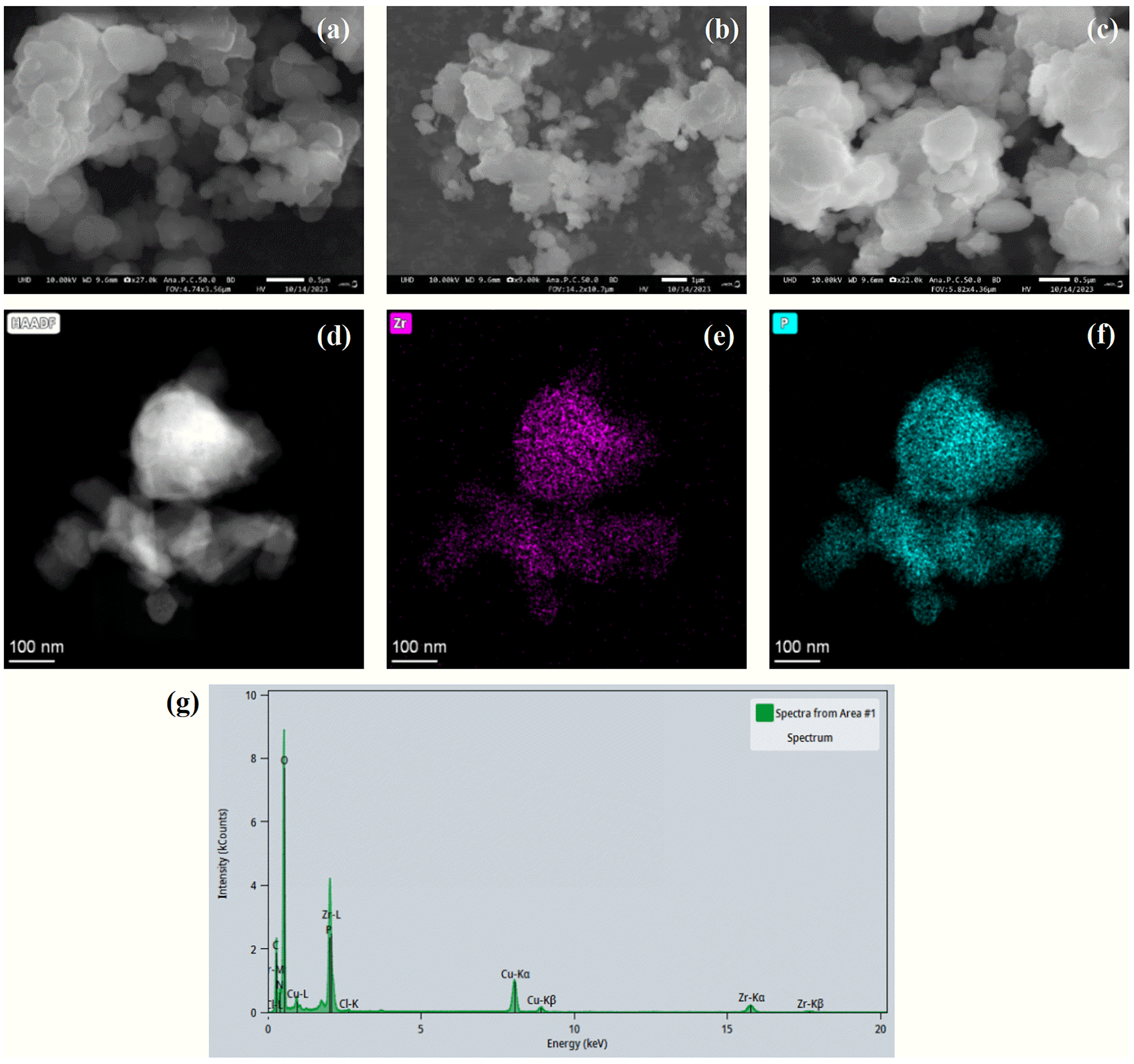
Scanning electron microscopy (SEM) images of the materials for (a) UiO-66-COOH, (b) UiO-66-COOH-PA-1, (c) UiO-66-COOH-PA-2, as well as transmission electron microscopy (TEM) images of (d) UiO-66-COOH-PA-2 corresponding elemental mapping of (e) Zr, (f) P, and (g) EDS images of UiO-66-COOH-PA-2.
The XRD spectra in the range of 5–80° for UiO-66-COOH, UiO-66-COOH-PA-1, and UiO-66-COOH-PA-2 are presented in Figure 2. In the XRD pattern of UiO-66-COOH, typical peaks at 7.5° indicate the successful synthesis of the crystalline configuration, which is consistent with the reported patterns for zirconium-based MOFs in the literature (Song et al., 2016). Following PA modification, the diffraction peaks near 7.5° in UiO-66-COOH-PA-1 and UiO-66-COOH-PA-2 exhibit a noticeable decrease in intensity and broadening. This suggests that the process of impregnation and high-temperature treatment lead to a decrease in the overall crystallinity of the material and thus merging several peaks into a broader one. However, XRD data indicate that they can still maintain the UiO-66-COOH structure. With an increase in the PA loading in the synthesized material, the weaker diffraction peaks at 17.3°, 25.4°, and 27.9° are enhanced, respectively, which should be related to the phosphorus from the added PA and have a good matching relationship with phosphorus oxide PDF # 50–1494.

X-ray diffraction (XRD) patterns of the materials for UiO-66-COOH, UiO-66-COOH-PA-1, UiO-66-COOH-PA-2, and phosphorus oxide PDF#5-1494.
The main functional groups in the sample are identified using FT-IR, verifying the modification of PA molecules in the series of synthetic materials of UiO-66-COOH. As shown in Figure 3, a typical infrared absorption peak near 1699 cm−1 is observed for UiO-66-COOH (Zhang et al., 2017), attributed to the stretching vibration of the free carboxyl groups in the material, indicating the successful synthesis of carboxylated UiO-66-COOH. In FT-IR of UiO-66-COOH-PA-1 and UiO-66-COOH-PA-2, bands at 955, 1080, and 1120 cm−1 are associated with the vibrational modes of the phosphonic acid groups and the stretching vibration of P=O (Dong et al., 2016). Several characteristic peaks of the phosphonic acid groups in the PA molecule collectively form a broader peak in the range of 955∼1120 cm−1 (Sun et al., 2021), and the intensity of the peak increases with an elevated loading of PA in the adsorbent. Compared with PA, the spectral bands associated with phosphonic acid groups in functionalized MOFs shift toward higher wavenumbers, which may be due to the interaction between PA and zirconium metal.
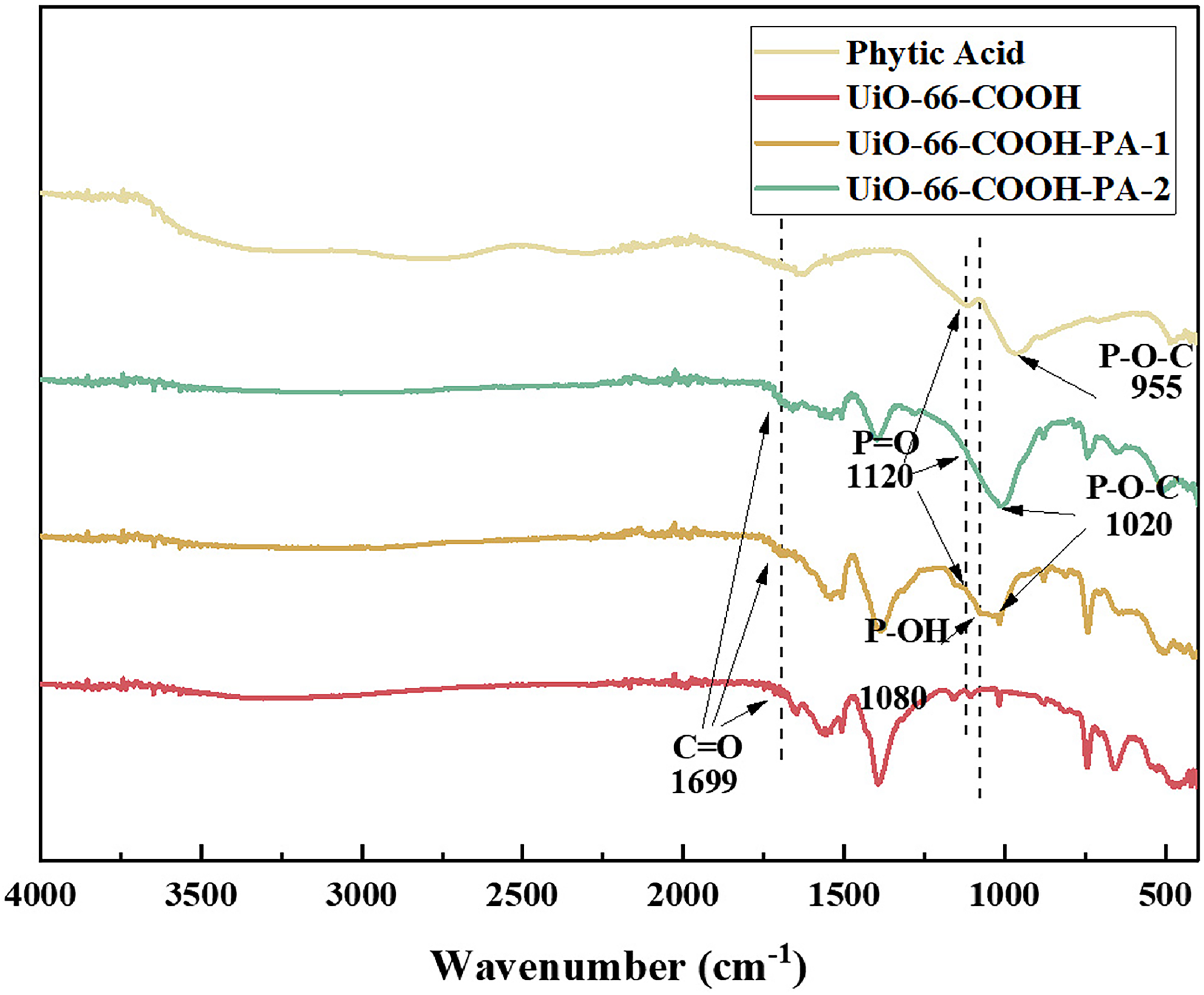
Comparison of Fourier transform infrared spectroscopy (FTIR) spectra of the materials for phytic acid, UiO-66-COOH, UiO-66-COOH-PA-1, and UiO-66-COOH-PA-2.
To explore the optimal reaction temperature for loading PA during high-temperature treatment and to verify the thermal stability of the adsorbent before and after phosphorus modification, thermogravimetric analysis (TGA) has been performed on UiO-66-COOH, UiO-66-COOH-PA-1, and UiO-66COOH-PA-2 at a heating rate of 10°C/min under a nitrogen atmosphere within the temperature range of 40–800°C. As shown in Figure 4, all the three prepared materials exhibit significant quality changes in the range of 40–200°C, which is speculated to be caused by the loss of organic solvents in the pores of the materials. In the range of 325–500°C, the higher PA content in UiO-66-COOH-PA-2 led to a more pronounced mass loss compared to UiO-66-COOH and UiO-66-COOH-PA-1, attributes to the thermal decomposition effect of PA molecules. When temperature is beyond 600°C, the framework structures of all three materials tend to stabilize. The results of thermogravimetric analysis indicate the temperature for high-temperature carbonization treatment should not exceed 300 °C to preserve the porous material's framework structure as much as possible.
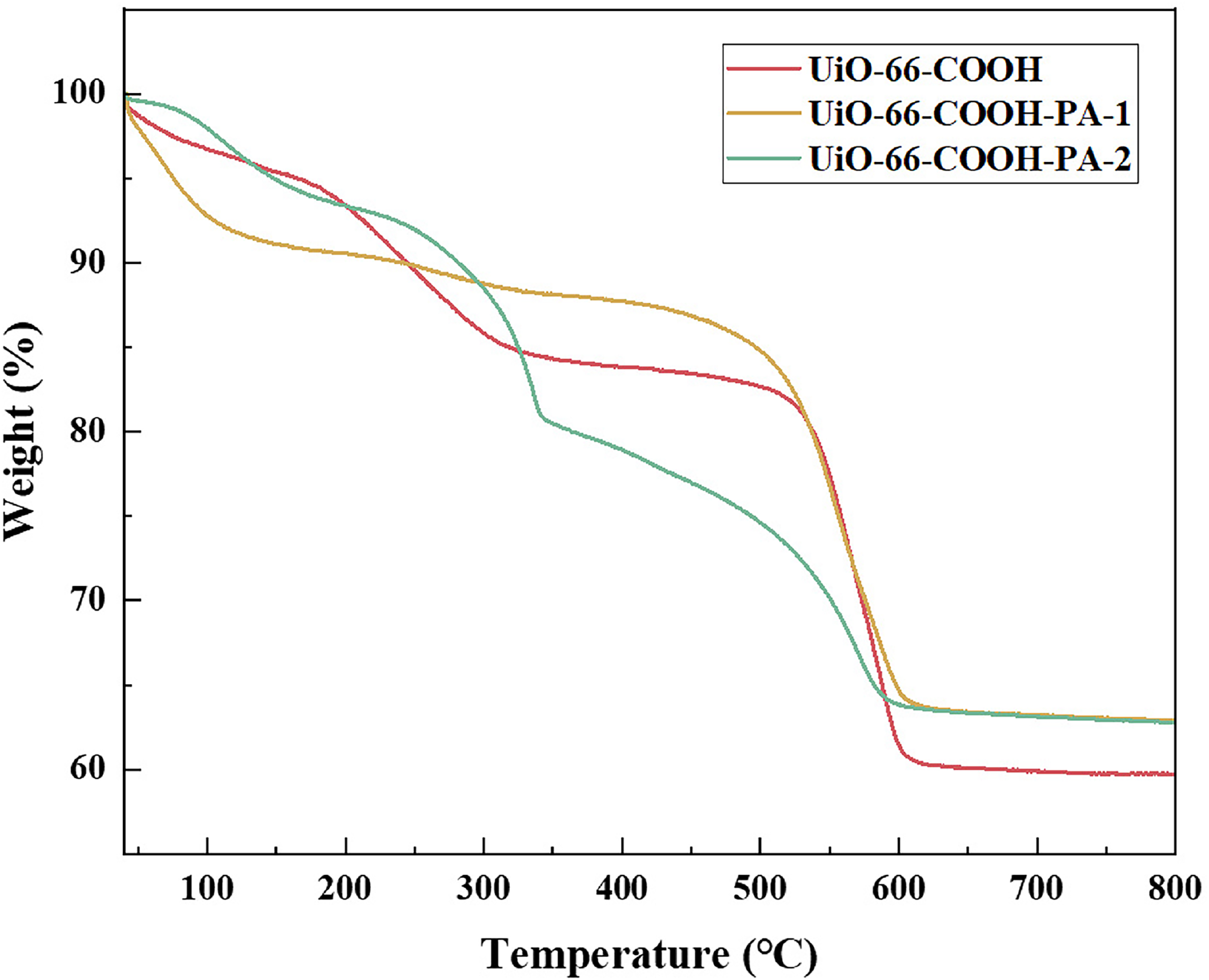
Thermogravimetric analysis of the material for UiO-66-COOH, UiO-66-COOH-PA-1, and UiO-66-COOH-PA-2.
As shown in Figure 5, the nitrogen adsorption–desorption isotherms of UiO-66-COOH, UiO-66-COOH-PA-1, and UiO-66-COOH-PA-2 exhibit type I isotherms. The detailed parameters of the adsorption–desorption isotherms for the three adsorbents are presented in Table 1. The pore sizes are 2.064 nm, 2.299 nm, and 2.203 nm, respectively, falling within the typical mesoporous range of 2–50 nm. Compared to UiO-66-COOH, the specific surface areas of UiO-66-COOH-PA-1 and UiO-66-COOH-PA-2 have decreased from 553.49 m2/g to 422.50 m2/g and 94.65 m2/g, respectively. The reduction in surface area is positively correlated with the amount of PA doping. Additionally, due to the formation of the carbonized layer after short-term high-temperature treatment in UiO-66-COOH-PA-1 and UiO-66-COOH-PA-2, the pore volume of the materials has decreased in some extent compared to UiO-66-COOH, reducing from 0.256 cm3/g to 0.192 cm3/g and 0.044 cm3/g, respectively. However, the pore sizes have increased after doping. These observations collectively indicate the successful modification of UiO-66-COOH with PA molecules.

Nitrogen adsorption–desorption isotherms of the materials for UiO-66-COOH, UiO-66-COOH-PA-1, and UiO-66-COOH-PA-2.
BET surface areas, pore volumes, and pore sizes of the adsorption materials for UiO-66-COOH, UiO-66-COOH-PA-1, and UiO-66-COOH-PA-2.

To establish the association between PA and UiO-66-COOH, we conducted XPS analysis to examine the elemental composition in the adsorption materials. As illustrated in Figure 6, the peaks correspond to O 1s, C 1s, and Zr 3d, respectively. Upon the introduction of PA into UiO-66-COOH, a new peak emerges in the P 2p region in Figure 6(b) and (c), which indicates the successful modification of the phosphate group into UiO-66-COOH. The results reveal that the P 2p peak area of UiO-66-COOH-PA-2 surpasses that of UiO-66-COOH-PA-1 due to the higher PA content in MOFs. In comparison to UiO-66-COOH, the intensity of the O1s peak is elevated in UiO-66-COOH-PA-1 and UiO-66-COOH-PA-2. The XPS survey spectrum corroborates the presence of P and Zr, aligning well with the EDS findings.

X-ray photoelectron spectroscopy (XPS) spectra of the materials for (a) UiO-66-COOH, (b) UiO-66-COOH-PA-1, and (c) UiO-66-COOH-PA-2.
Effect of initial pH
In acidic media, thorium typically exists in positively charged ionic forms such as Th4+, Th(OH)3+, and Th(OH)22+ (Chen and Wang, 2007). Around pH 4, thorium ions are commonly present in the form of hydroxides. Using HCl and sodium hydroxide (NaOH) solutions with concentrations ranging from 0.01M to 1M to control the pH value of the adsorption solution within the range of 2.5–4.0, can avoid significant precipitation of Th(IV) itself. The Zeta potential curves of three materials versus pH values are depicted in Figure 7(a). With the increasing of pH, there is a noticeable decrease in surface charge on the adsorbent modified with PA. At times, UiO-66-COOH shows no significant changes. Figure 7(b) illustrates the significant influence of pH on the adsorption capacities of the three adsorption materials. The adsorption capacities for thorium follow the order as UiO-66-COOH-PA-2 > UiO-66-COOH-PA-1 > UiO-66-COOH, and the adsorption capacity is positively correlated with the loading of PA. Especially, the adsorption capacity of UiO-66-COOH-PA-2 is almost 1.8 times higher than that of UiO-66-COOH.

(a) the Zeta potentials as a function of pH. (b) Effect of pH on Th(IV) adsorption. (c) Effect of treatment temperature on Th(IV) adsorption. (d) Adsorption capacity per unit specific surface area of the three adsorption materials for UiO-66-COOH, UiO-66-COOH-PA-1, and UiO-66-COOH-PA-2 (C0 = 100 mg/L, m/V = 0.22 g/L, and T = 298 K).
Protonated UiO-66-COOH with positive surface charge exhibits lower adsorption capacity at low pH values due to electrostatic repulsion with positively charged thorium ions (Zhu et al., 2021). In the meanwhile, it can be seen obviously that the adsorption capacity of UiO-66-COOH at low pH value (pH ≤ 3.0) is almost zero while that of UiO-66-COOH-PA-2 is upto 90 mg/g. The enhanced adsorption ability suggests a strong chelating effect of PA on thorium ions. With increasing pH, the complexation of thorium by PA, combined with electrostatic effects, makes the adsorption capacity of thorium much more pronounced. Additionally, to investigate the influence of different treatment temperatures (300°C, 500°C, 700°C) on the adsorption capacity of the adsorption material, the adsorption capacity for Thorium (IV) is tested at pH of 4. As illustrated in Figure 7(c), the adsorption capacities of UiO-66-COOH-PA-1 and UiO-66-COOH-PA-2 heated at 300°C are markedly higher than others. Considering the practical utility, the adsorption materials used in the subsequent batch adsorption experiments are all subjected to rapid high-temperature carbonization at 300°C. Due to the significant reduction in the specific surface area of the adsorption material caused by the doping of PA, the unit area adsorption capacity of UiO-66-COOH-PA-2 is shown in Figure 7(d), which is much higher than that of UiO-66COOH, further indicating the adsorption effect of the doped phosphorus on thorium.
Effects of initial concentration
To assess the adsorption capacity of the synthetic materials for thorium ions, adsorption isotherms have been determined by varying the initial concentration (5∼300 mg/L) systematically. As shown in Figure 8(a) and (b), the adsorption of thorium ions significantly increased. UiO-66-COOH reaches saturation in adsorption in a 150 mg/L initial concentration solution, with a maximum equilibrium adsorption capacity of 151.2 mg/g at pH of 4. While UiO-66-COOH-PA-1 and UiO-66-COOH-PA-2 exhibit a slow growth rate in adsorption capacity at concentrations of 150 mg/L to 300 mg/L, with maximum equilibrium adsorption capacities of 244.4 mg/L and 315.5 mg/L, respectively. This suggests that PA plays a crucial role in capturing thorium ions acting as an ion trap.

(a) Adsorption isotherms, (b) Freundlich and Langmuir isotherm models fit, (c-d) Freundlich and Langmuir isotherm linearized plots for Th(IV) adsorption of the three materials for UiO-66-COOH-PA, UiO-66-COOH-PA-1, and UiO-66-COOH-PA-2, (e) Th(IV) adsorption kinetics of UiO-66-COOH-PA-2 under various temperature, and (f) The pseudo-second-order kinetic linearized plots for Th(IV) adsorption on UiO-COOH-PA-2 at different temperature.
The adsorption isotherm is a key criterion for evaluating the adsorption mechanism of adsorption materials. Linear fitting of adsorption data has been performed using Langmuir and Freundlich isotherm models to further evaluate adsorption behavior. The Langmuir isotherm equation is widely used to describe single-layer adsorption processes with constant adsorption energy, assuming that each active adsorption site on the adsorbent surface corresponds to a solute molecule. In addition, all sites on the surface of the adsorption materials are considered to be equivalent. The linear form of the Langmuir isotherm equation can be expressed as follows (equation (3)):
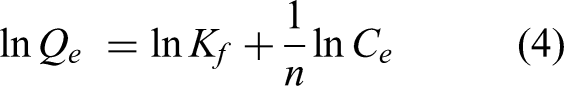
The adsorption isotherm linear fitting parameters and correlation for the three types of materials are shown in Figure 8(c) and (d) and Table 2. Th(IV) adsorption kinetics of UiO-66-COOH-PA-2 under various temperature and the pseudo-second-order kinetic linearized plots for Th(IV) adsorption on UiO-COOH-PA-2 at different temperature are displayed in Figure 8(e) and (f) (Please find the fitting of adsorption isotherm in Supplementary Information).
Isotherm parameters for Th(IV) adsorption of the adsorption materials for UiO-66-COOH, UiO-66-COOH-PA-1, and UiO-66-COOH-PA-2.

As shown in Table 2, the linear fitting results indicate that the Freundlich model compared to the Langmuir model is more suitable for describing the adsorption process. The related adsorption data are considered to be inadequately fitted to the Langmuir model. The Freundlich R2 values for UiO-66-COOH, UiO-66-COOH-PA-1, and UiO-66-COOH-PA-2 are all exceeding 0.95. This suggests that the adsorption of thorium ions involves a multipoint adsorption process on the nonuniform surface of the adsorbent. This adsorption process may be the result of the combined action of free carboxyl groups and phosphate groups in the adsorbent.
Effect of temperature and adsorption time on Th(IV) adsorption
Related experiments have been conducted at different temperatures (298K, 308K, 318K), the adsorption time are ranged from 5–150 min. As shown in Figure 8(e), the adsorption capacity of UiO-66-COOH-PA-2 increases rapidly within the first 5 min, then the adsorption rate just slow down, reaching equilibrium within 20 min. This rapid adsorption phenomenon is attributed to the coordination effect between thorium ions and the phosphate groups in PA. Additionally, an increase in temperature positively affects the adsorption performance of UiO-66-COOH-PA-2 for thorium ions, with a noticeable enhancement in adsorption capacity as the temperature increased.
In order to analyze the adsorption processes of adsorption material, particularly for describing chemical adsorption phenomena, we use the pseudo-first-order model, pseudo-second-order model and Elovich adsorption kinetic models to fit the experimental data (Wang et al., 2015). As illustrated in Figure 8(f), it anticipates the adsorption behavior of thorium ions by UiO-66-COOH-PA-2 at different temperatures.
The pseudo-second-order kinetic model is commonly employed to analyze the adsorption processes of adsorption material, particularly for describing chemical adsorption phenomena. As illustrated in Figure 8(f), it anticipates the adsorption behavior of thorium ions by UiO-66-COOH-PA-2 at different temperatures. The pseudo-second-order kinetic model is expressed by equation (5) as follows: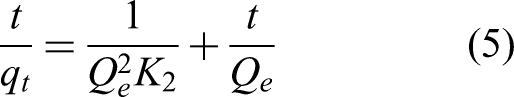
As shown in Table 3, according to the calculation of the pseudo-second-order dynamic model, both Qe and K2 values increase simultaneously with the increase in temperature. This indicates that an increase in temperature has a positive impact on the adsorption process of UiO-66-COOH-PA-2, leading to a significant enhancement in adsorption capacity. The adsorption of Th(IV) by UiO-66-COOH-PA-2 is an endothermic reaction. The linear fitting of the adsorption data at different temperatures results in ideal correlation coefficients using the pseudo-second-order kinetic model (R2 > 0.99), (Figure 8(f)) providing clear insight into a well-defined chemical adsorption process (Elwakeel et al., 2014) (Please find the thermodynamic study in Supplementary Information).
Kinetic parameters for Th(IV) adsorption of the materials for UiO-66-COOH-PA-2.

Effect of coexisting ions
In view of thorium is commonly coexist with various rare earth minerals, such as monazite mineral and thorite (Berry et al., 2017), the separation and recovery of thorium from such minerals hold significant importance for the sustainable development of the nuclear industry. In order to further analyze the adsorption selectivity of UiO-66-COOH-PA-2 for thorium, the adsorption capacity of the synthesized adsorption material is measured in detail in a multi cation rare earth ion solution with pH of 4. As shown in Figure 9, the selectivity of UiO-66-COOH-PA-2 for Th(IV) is 90.2%, which is much higher than that of other coexisting ions (La3+, Nd3+, Gd3+, Yb3+, Sm3+). It indicates the excellent affinity of the phosphonate groups for Th(IV) ions.
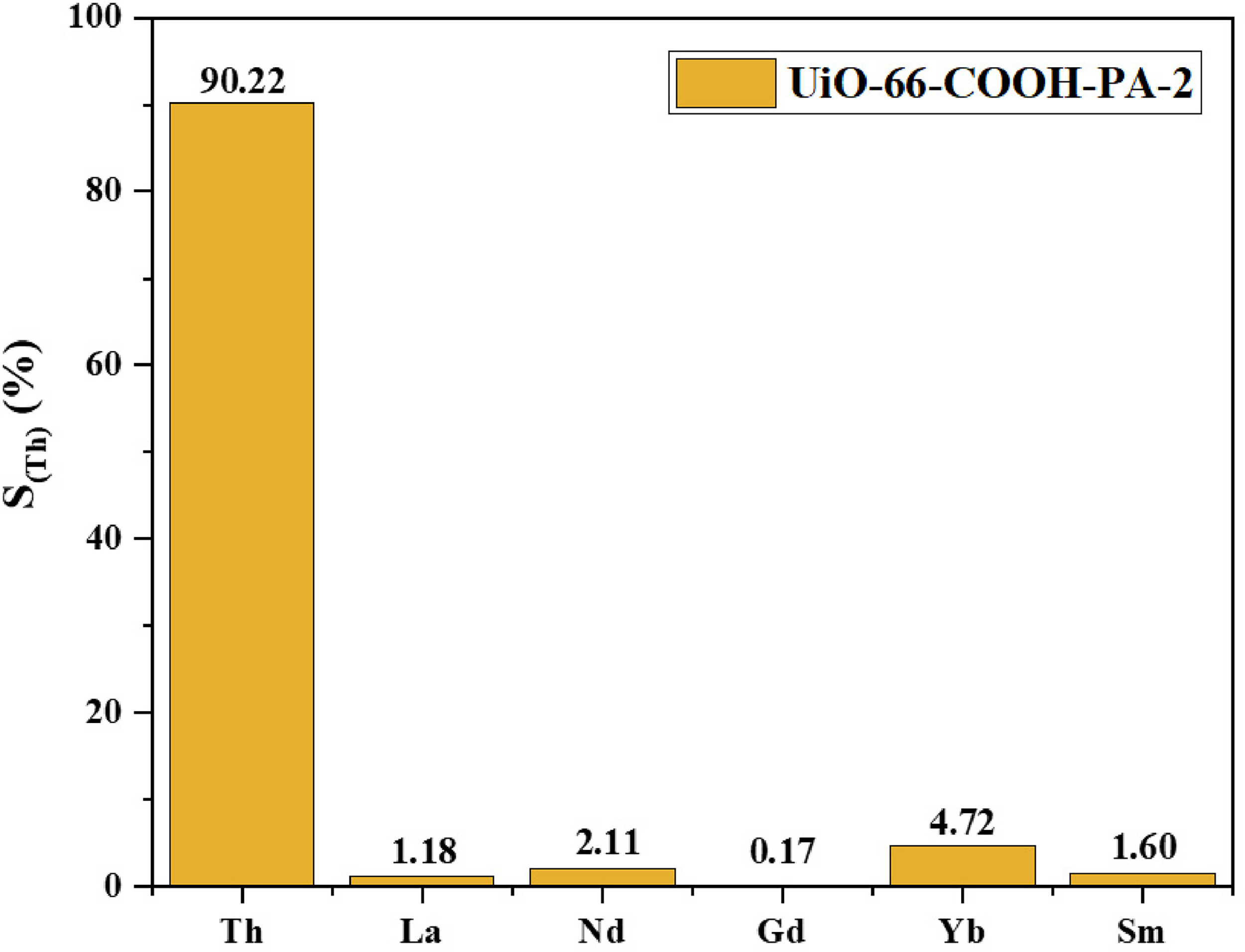
Effect of competitive ions on the selective adsorption of Th(IV) onto UiO-66-COOH-PA-2 (C0 = 100 mg/L, m/V = 0.22 g/L, and T = 298 K).
Cyclic regeneration experiments
In order to evaluate the reusability of the adsorption material for thorium ion adsorption, adsorption–desorption experiments have been conducted (Cheng et al., 2022). Considering the little adsorption capacity of the adsorption material for thorium at lower pH, different concentrations of HCl solutions (0.01M, 0.05 M, 0.1 M) have been chosen as the eluent for the desorption of thorium, and the reusability has also been evaluated by measuring the Th4+ ions concentration in the desorbed solutions. In each cycle, the supernatant has been taken to measure the amount of Th4+ released in HCl solutions, using different concentration of HCl solutions (0.01M, 0.05 M, 0.1 M) for the desorption of thorium ions from the adsorption material. Subsequently, deionized water has to be used to repeatedly wash the adsorption material to remove any remaining thorium ions and HCl, drying for the next adsorption cycle. As shown in Table 4, the desorption efficiency of Th(IV) using 0.01M HCl for UiO-66-COOH-PA-2 only reaches 64.4%, while the desorption efficiency of 0.1 M HCl solution for UiO-66-COOH, UiO-66-COOH-PA-1, and UiO-66-COOH-PA-2 all exceed 95%. Therefore, higher concentration of HCl solutions contribute to stronger desorption capacity for Th4+ ions, further confirming the strong chelating effect of the phosphate group on Th4+ ions.
Effect of different concentration of HCl onto the adsorption materials.

The regenerated adsorption experiments for UiO-66-COOH-PA-2 have been conducted under the same conditions, and the results are shown in Figure 10. Due to the high stability of Zr-based MOFs in acidic solutions, the reusability of the adsorption material does not significantly decrease after desorption in 0.1 M HCl solution. The adsorption capacity of reused UiO-66-COOH-PA-2 has no obvious change compared to the fresh one, and its regenerative adsorption capacity still exceeds 80%. These findings demonstrate that the adsorption material exhibits excellent stability and can serve as an economical and environmentally friendly material for the removal and pretreatment of thorium ions in water environments.

Reusability of Th(IV) by UiO-66-COOH-PA-2 after adsorption/desorption cycles (C0 = 100 mg/L, m/V = 0.22 g/L, and T = 298 K).
Phytic acid-functionalized Zr-MOFs typically exhibit higher adsorption capacities for heavy metals (such as uranium) in aqueous environments compared to their unmodified counterparts (Peng et al., 2021). Previous studies have shown that functionalization can enhance the hydrophilicity and selectivity of the material, thereby improving its affinity for specific ions (Bi et al., 2021). Research indicates that PA-functionalized Zr-MOFs possess excellent thermal and chemical stability, which is crucial for their long-term use in practical applications (Zhao et al., 2022). The conclusions of the aforementioned studies are substantiated by the results of this research.
Conclusions
In summary, PA has been introduced into UiO-66-COOH pores through a short-term high-temperature treatment, and a phosphorus-functionalized Zirconium-based metal-organic framework (UiO-66-COOH-PA) has been successfully created. A series of adsorption experiment results show that this material exhibits exceptional adsorption property as well as high selectivity in the condition of several rare-earth ions coexisting in acidic environments, and the equilibrium adsorption data of this adsorption material for thorium ions follows the Freundlich isotherm model. Furthermore, cyclic regeneration experiments demonstrated excellent stability, the regenerative adsorption capacity of UiO-66-COOH-PA-2 exceeds 80% after five times adsorption/desorption cycle compared to the fresh one. The excellent adsorption performance of UiO-66-COOH-PA-2 at low pH values indicates that adsorption material containing phosphorus are more suitable for the removal of thorium in acidic systems in the solution. These results open up new possibilities for designing phosphorus-enriched MOF materials and offer a promising approach for Th(IV) adsorption performance in acidic water environments. The material's radiation stability and potential structural degradation during prolonged use warrant further investigation and optimization. In our future study, the environmentally friendly and cost-effective absorption material would be further optimized for the application of thorium removal especially in much more acidic radio wastewater.
Supplemental Material
sj-docx-1-adt-10.1177_02636174241298776 - Supplemental material for Phosphorus-modified Zr-based metal-organic framework materials for their effective removal of thorium from aqueous solutions
Supplemental material, sj-docx-1-adt-10.1177_02636174241298776 for Phosphorus-modified Zr-based metal-organic framework materials for their effective removal of thorium from aqueous solutions by Yinghao Zhuang, Shuai Sun, Kai Jia, Lei Shi and Xiangshan Yang in Adsorption Science & Technology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Radioactivity analysis of environmental samples, (grant number /).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


