Abstract
This research investigates the simultaneous removal of lead ion-Pb(II), cadmium ion-Cd(II), and cobalt ion-Co(II) from aqueous solution and real samples using activated carbon (AC-C) synthesized from cypress fruit. The investigation meticulously examines the prompt of several adsorption parameters (pH, adsorbent dosage, temperature, initial concentration, and agitation time) on the effectiveness of heavy metal removal by AC-C. FTIR, SEM, and EDS analyses provide convincing evidence for the effective adsorption of metal ions onto the AC-C surface. Pb(II) performed best at a concentration of 40 mg.L−1 in an acidic solution (pH 5) with 70 mg of adsorbent and 100 min of agitation time. Co(II) and Cd(II) achieved their highest removal rates at a concentration of 90 mg.L−1 in basic solutions (pH 10) with 70 mg of adsorbent and 90 min of agitation time. Under these optimal conditions, AC-C demonstrates remarkable removal efficiencies of 98.68% for Pb(II), 95.60% for Co(II), and 76.45% for Cd(II). Kinetic studies reveal that the adsorption process fits a second-order model for the simultaneous removal of the three metal ions. Additionally, equilibrium data are best characterized by the Langmuir isotherm model, indicating monolayer adsorption of the heavy metal ions mixture. Thermodynamic analysis provides further insights into the adsorption process. The positive enthalpy (ΔH°) confirms the endothermic adsorption of the three ions, and the positive entropy (ΔS°) predicts an unpredictable nature at the solid-solution interface. This study establishes AC-C derived from cypress fruit as a highly effective and promising candidate for the simultaneous removal of Pb(II), Cd(II), and Co(II) from aqueous environments. Its remarkable performance, coupled with the readily available natural resource, paves the way for wider application of AC-C in environmental remediation strategies.
Keywords
Introduction
The contamination of natural water sources by heavy metal ions presents acute environmental trouble with far-reaching implications for human health and ecological integrity (Zhang, 2023). Heavy metals, such as lead, arsenic, cadmium, nickel, chromium, and mercury, are persistent pollutants that find their way into water bodies through industrial discharges, agricultural runoff, and improper waste disposal. Once released into the environment, they can accumulate in aquatic ecosystems, posing a severe threat to aquatic life and potentially entering the human food chain (Swaroop et al., 2022). Lead for example, often originating from industrial processes, plumbing systems, and certain paints, is notorious for its harmful effects, particularly on the nervous system, cognitive function, and the maturity of children (Sanders, 2009). Cobalt, commonly present in industrial effluents and electronic waste, can adversely affect various organs, including the heart and kidneys, while also raising concerns about its potential carcinogenicity. Cadmium, arising from industrial discharges and agricultural practices, poses severe health risks due to its association with kidney damage, respiratory issues, and an increased risk of cancer (Saikat Mitra, 2022). As these heavy metals realize their way into water bodies, their potential to accumulate in living organisms and the subsequent biomagnification within food chains emphasize the insistence of addressing water contamination. Considering the toxicity of metal ions such as lead, cobalt, Mercury, chromium, and cadmium in water is pivotal for developing effective strategies to monitor, mitigate, and prevent their harmful effects on human health and the environment (Madiha Zaynab, 2022). This necessitates comprehensive research into the sources, pathways, and health consequences of exposure to these heavy metals, laying the foundation for informed regulatory measures and water quality management. The need for effective and sustainable methods for removing heavy metal ions from water has never been more pressing. Adsorption on activated carbon stands out as a versatile and powerful method. By capitalizing on the remarkable surface area and complex pore structure of activated carbon, this approach converts it into a “molecular magnet” capable of attracting and entrapping a wide variety of contaminants (Moosavi, 2020). From harmful chemicals and heavy metals to unpleasant odors, activated carbon effectively removes them from water as it flows through. This adaptability makes it a crucial player in various water purification applications, from large-scale municipal drinking water treatment facilities to industrial wastewater processing plants. Whether it is eliminating chlorine and organic compounds, filtering out harmful metals, or tackling taste and odor issues, activated carbon adsorption offers a reliable, efficient, and cost-effective solution. Through its remarkable adsorption prowess, it plays a vital role in the continuous pursuit of clean and safe water for all (Wang et al., 2023). Activated carbon, derived from various sources, including cypress wood, has emerged as a promising adsorbent for heavy metal removal due to its high surface area, porous structure, and chemical reactivity (Wang, Adsorption of heavy metal onto biomass-derived activated carbon: review. RSC advances, 2023). The utilization of activated carbon, in particular, has gained attention for its potential to simultaneously remove multiple heavy metal ions from contaminated water sources (Yantus and Neolaka, 2023).
This research aims to delve into the kinetics, thermodynamics, and isotherms governing the simultaneous removal of heavy metal ions by activated carbon derived from cypress fruit. Understanding the fundamental mechanisms and behavior of this adsorption process is essential for optimizing the design of water treatment systems and achieving efficient removal of heavy metal contaminants. Moreover, the exploration of cypress-derived activated carbon as a sustainable and eco-friendly adsorbent showcases a commitment to environmental stewardship and resource conservation.
Materials and methods
Chemicals
Lead (II) nitrate, cadmium (II) nitrate, and cobalt (II) nitrate with a purity of > 99% were purchased from Sigma Aldrich (Germany). Double distilled water was used for the preparation of all solutions.
Preparation of solutions
To prepare the stock solutions, 0.16 g of Pb(NO3)2, 0.5 g Co(NO3)2, and 0.617 g of Ni(NO3)2 were dissolved in double distilled water in 100-mL volumetric flasks. The stock solutions were then diluted with distilled water to produce 5–100 ppm solutions of the ions. A 1.0 M HCl and NaOH solutions were employed to adjust the pH of the solutions.
Adsorbent preparation and characterization
Cypress fruit, sourced from the university campus in Jordan, embarked on a remarkable journey to become an eco-friendly remediation tool. The collected cypress fruit was thoroughly washed and dried at 100 °C for 24 hours. Next, it was crushed and impregnated with concentrated phosphoric acid, followed by heating at 450 °C. The prepared activated carbon was subsequently washed with a 1.0 M NaOH solution for neutralization (Thotsaporn Somsiripan, 2023). This process yielded activated carbon (AC-C), which was then ground and sieved to obtain particles of 180 μm size. To elucidate the surface properties of the AC-C, Fourier transform infrared (FTIR, TENSOR model from BRUKER. Germany) and scanning electron microscopy (SEM, Apreo 2 S LoVac, USA) techniques were employed.
Adsorption experiment
The aqueous solution containing several concentrations of Pb(II), Cd(II), and Co(II) ions was prepared and agitated with a fixed amount of activated carbon (AC-C) for a definite time. An atomic absorption spectrometer was used to analyze the lasting concentration of each heavy metal ion. The study investigated the effect of several parameters on the adsorption process, including the AC-C dosage, initial metal ion concentration, solution pH, agitation time, and temperature. The amount of each adsorbed metal ion was measured using a ContrAA 800 Atomic Absorption Spectrometer from Analytik Jena, Germany. Additionally, equations 1 and 2 (Table 1) were used to calculate both the adsorbed quantities and the percentage removal of the metal ion mixture.
Equations and mathematical relationships.

Results and discussion
Characterization of AC-C adsorbent
FTIR
FTIR analysis revealed a wealth of functional groups present on both the pristine AC-C adsorbent and the AC-C-Mix, which was obtained after being covered with the metal ion mixture. The detailed FTIR analysis is presented in Table 2. Notably, distinct shifts in absorbance and intensity were observed, providing concrete evidence for the efficacy of AC-C in the simultaneous removal of the heavy metal mixture. These crucial results provide valuable insights into the mechanisms underlying the adsorption process and underline the potential of AC-C as a promising material for environmental remediation applications. Functional groups, including aromatic groups, aldehydes, and carboxylic acids, were identified within the frequency ranges of 3400–2400, 1725–1700, and 2830–2695 cm−1, respectively. Metal often attaches itself to carbon through ligand synthesis and ion exchange. As shown in Scheme 1, all three ions in our investigation formed ligands with functional groups by substituting metal ions for H+, resulting in the formation of an organometallic complex on the adsorbent surface (Soumya Banerjee, 2016).

Adsorption mechanism of the metal ion with carboxylic acids and phenolic compounds on the surface of AC-C.
FTIR results of AC-C and AC-C-mix.

SEM and EDS analysis
SEM analysis was employed to investigate the characteristics of the AC-C surface before and after the adhering of the metal ions mixture. As depicted in Figure 1, the observed changes in surface structure specify fascinating evidence for the effective removal of the metal ion mixture by AC-C. Notably, the images reveal how the pores within the AC-C structure were effectively occupied by the adsorbed metal ions mixture, visually confirming the adsorption process. This critical analysis reinforces the recognizing of AC-C's capabilities in tackling environmental contaminants and shows its potential as a adaptable tool for the removal of heavy metal.

SEM analysis of (a) AC-C and (b) AC-C-Mix.
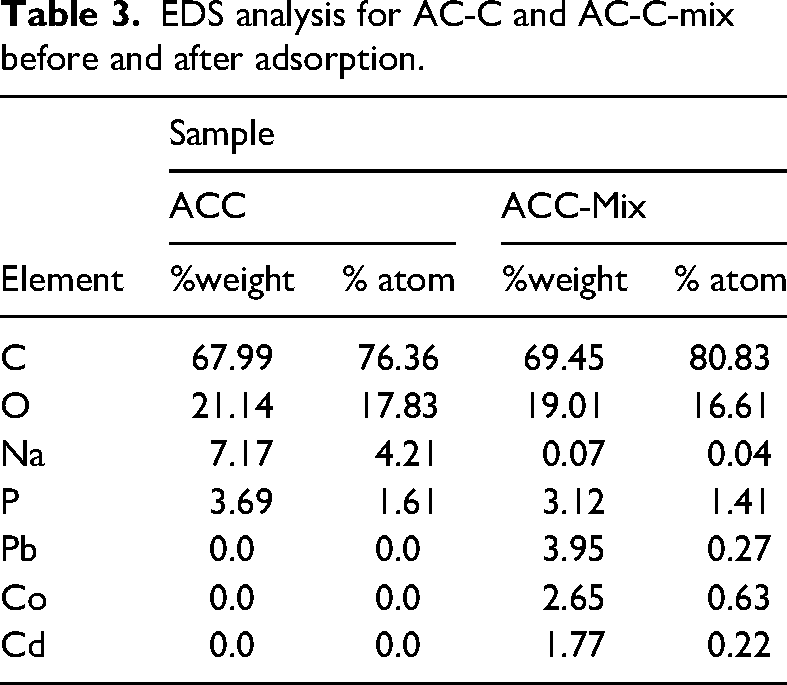
Figure 2 represents the results of the EDS analysis, specifying irrefutable evidence for the simultaneous removal of the metal ions mixture by the AC-C. As specified in Table 3, the concentrations of Pb(II), Co(II), and Cd(II) adsorbed onto the AC-C matrix were determined to be 3.95%, 2.65%, and 1.77%, respectively. These results unambiguously demonstrate the AC-C’s capability to remove a diverse range of heavy metal ions simultaneously. Furthermore, the results show a superior affinity for Pb(II) compared to the other two metal ions. EDS analysis provides definitive confirmation of the AC-C's effectiveness in simultaneously removing Pb(II), Co(II), and Cd(II). Table 3 investigates the weight and atom percentages for elements in the samples.

EDS analysis of AC-C and AC-C-Mix.
EDS analysis for AC-C and AC-C-mix before and after adsorption.
Batch adsorption
Influence of adsorbent dose
The percentage of heavy metal ions adsorbed by AC-C is directly influenced by the amount of the adsorbent used. In this study, the AC-C dosage varied from 20 to 100 mg, while the temperature was held constant at 25.0 ± 1 °C. The results in Figure 3 expose that 70 mg of AC-C ascends as the optimal dosage for attaining the highest removal efficiency for all three metal ions. This result can be ascribed to the existence of eagerly available active sites on the AC-C surface, which simplify the initial adsorption of the metal ions. However, as the system approaches equilibrium, the rate of adsorption decreases. This observation underlines the importance of optimizing the AC-C dosage to amplify its effectiveness in removing heavy metals from aqueous environments.

Influence of AC-C mass on the simultaneous uptake of metal ions mixture of Pb(II), Co(II), and Cd(II), AC-C mass = 20–100 mg, Ci = 50 mg. L−1; pH = 7.0, agitation time = 60 min, and T = 298 K.
Influence of ion initial concentration
This study investigates the relationship between initial metal ion concentration and the simultaneous adsorption of Pb(II), Co(II), and Cd(II) onto AC-C. The investigation explored a range of concentrations in the range of 30–120 mg L−1 at a constant temperature of 25 ± 1 °C. The results, depicted in Figure 4, exposed optimal adsorption percentages of 98.24% for Pb(II), 83.97% for Co(II), and 84.40% for Cd(II). Notably, the optimal concentrations were determined to be 60 mg L−1 for Pb(II) and 90 mg L−1 for both Co(II) and Cd(II). As the initial metal ion concentration increased, a consistent increase in occupied sites on the surface of the AC-C was observed. This phenomenon can be attributed to the enhanced availability of metal ions for binding to the AC-C surface. However, with further increase in concentration, the occupation of available sites reached saturation, leading to a decrease in the equilibrium adsorption rate.

Influence of metal ion initial concentration on the simultaneous uptake of metal on AC-C. mass = 70 mg, Ci = 30–120 mg. L−1; pH = 7.0, agitation time = 60 min, and T = 298 K.
Influence of agitation time
The batch adsorption experiments involved agitating 70 mg of AC-C with a 50 ml mixture containing 60 mg L−1 of Pb(II) and 90 mg L−1 each of Co(II) and Cd(II) at a constant temperature of 25 ± 1 °C were conducted. The agitation time was varied from 30 to 120 min to investigate its influence on the adsorption process. Figure 5 vividly illustrates the differences in the adsorption of each metal ion onto AC-C as the agitation time increased. Notably, the optimal agitation time for maximizing the removal of all three metal ions was determined to be 90 min. These results suggest that sufficient time is necessary for the metal ions to effectively interact with the available active sites on the AC-C surface and establish strong bonds. Beyond 90 min, the rate of adsorption reaches a plateau, indicating that the majority of metal ions have been removed. This information is important for optimizing the adsorption process and diminishing needless agitation time, resulting in a more efficient and cost-effective approach to heavy metal removal.
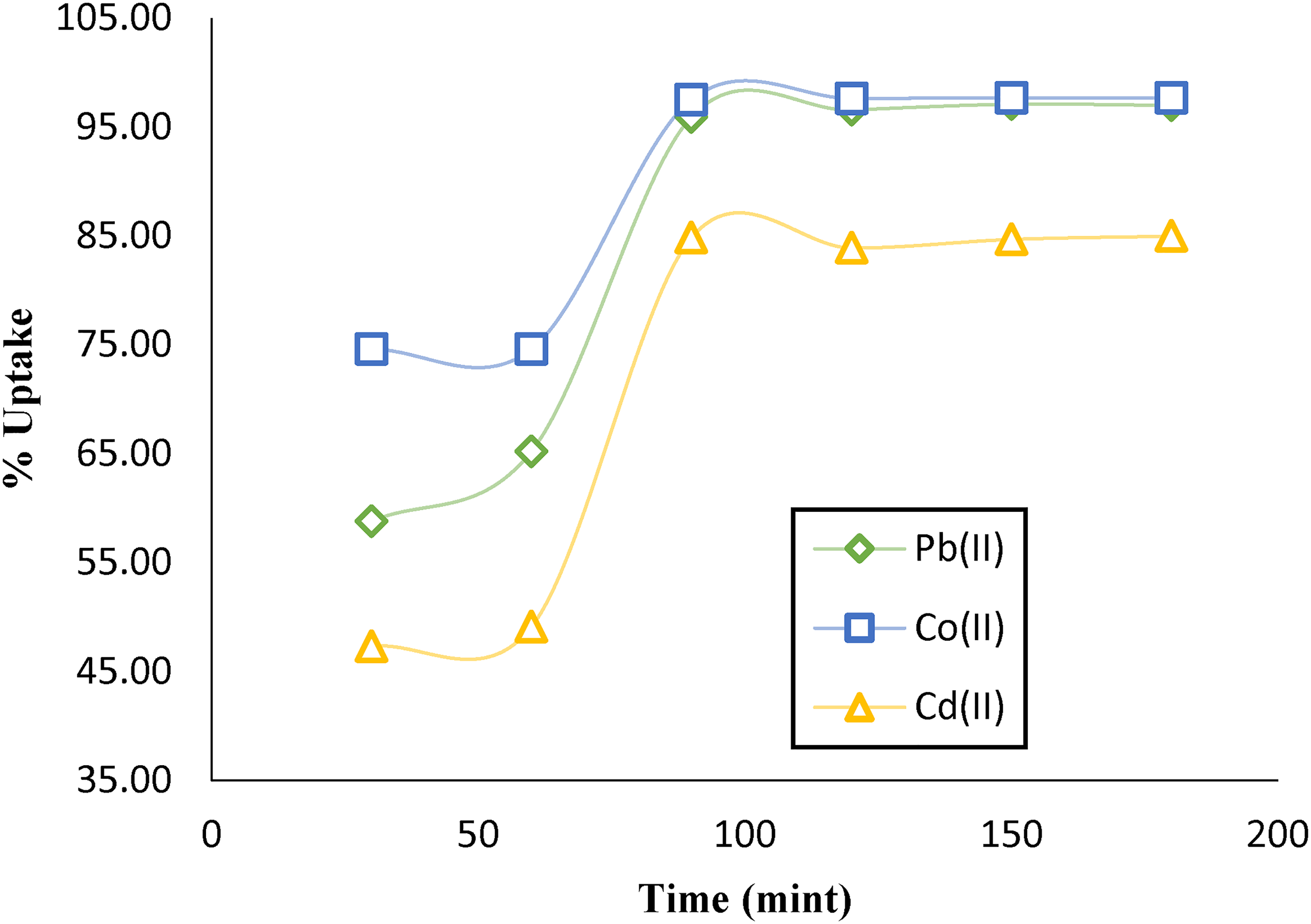
Influence of agitation time on the simultaneous uptake of metal ions on AC-C. mass = 70 mg, Ci = 40 mg. L−1 pb(II), and 90 mg. L−1 for Cd(II) &Co(II); pH = 7.0, and T = 298 K.
Influence of pH
Marking the point of zero charge (pHpzc), the pH at which the surface of adsorbent material exhibits neutrality is important for identification of the adsorption process. The pHpzc of AC-C was determined by shaking 0.15 g of adsorbent material with 50.0 ml of 0.1 M NaOH solution at several pH values (2–12) for approximately 24 h (Sun et al., 2016). The analysis revealed that the AC-C surface converts neutral at a pH of 7.02. According to this result, the study investigated the influence of pH on the efficiency of the adsorption process. Samples of 0.07 g AC-C were agitated with 50 ml of a metal ion mixture solution containing 60.0 mg L−1 Pb(II) and 90 mg L−1 each of Co(II) and Cd(II) at a temperature of 25 ± 1 °C for one hour. The pH varied from 3 to 11. As illustrated in Figure 6, the results demonstrate a significant increase in the removal of metal ions by AC-C with increasing pH. This remark can be ascribed to the electrostatic interactions that occur between the negatively charged AC-C surface and the positively charged metal ions. At higher pH values, a bigger number of negative charges exist on the AC-C surface, simplifying stronger electrostatic attraction and subsequent adsorption of the metal ions.

Influence of pH on the simultaneous uptake of metal ions on AC-C. mass = 70 mg, Ci = 30 g. L−1; agitation time = 100 min, and T = 298 K.
Kinetic and mechanism
To investigate the particulars of how AC-C removes a mixture of heavy metal ions, kinetics studies were conducted using various linear models, including pseudo-first-order, pseudo-second-order, and intra-particle diffusion. Equations 3–5 employed for these models are presented in Table 1 (Kumar et al., 2018). A comprehensive analysis of the results, depicted in Figure 7, explores a convincing result of pseudo-second-order model provides the best fit for describing the kinetics of simultaneous metal ion removal by AC-C. This implies that the rate-limiting step of the adsorption process involves chemisorption, where chemical bonding occurs between the metal ions and the AC-C surface.
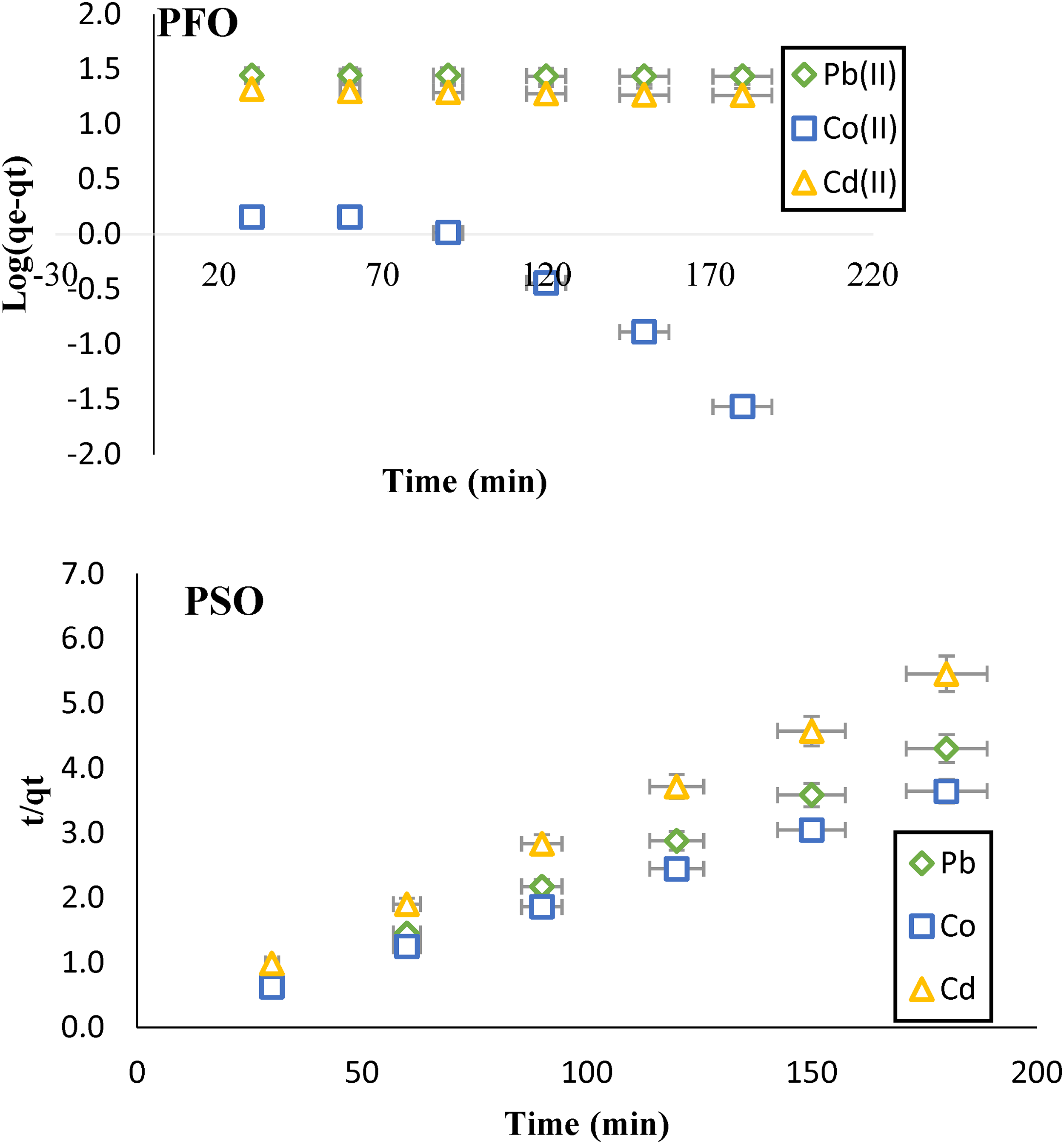
Linear kinetics of (a) PFO and (b) PSO, the simultaneous removal of the heavy metal ions mixture [Pb(II), Co(II), Cd(II)] on AC-C.
To elucidate the mechanism underlying the simultaneous removal of the metal ion mixture by AC-C, the intra-particle diffusion model was employed. This model is represented by equation 5 in Table 1 (Lafi et al., 2018). The results depicted in Figure 8 explore that the intra-particle diffusion process governs the simultaneous removal of the metal ions by AC-C, with the surface adsorption step playing a dominant role (C > 0). This signifies that the initial adsorption of metal ion onto the AC-C surface is the rate-limiting step, followed by intra-particle diffusion of the adsorbed ions within the AC-C pores.

Linear intra-particle model for the simultaneous adsorption of the heavy metal ions mixture [Pb(II), Co(II), Cd(II)] on AC-C.
The values of R2 and constants for all kinetics models were summarized in Table 4.
Results of kinetics studies for the simultaneous removal of a mixture of Pb(II), Co(II), and Cd(II) by AC-C.

Adsorption isotherms
In this study, four models were utilized to unravel the adsorption mechanism of Pb(II), Co(II), and Cd(II) onto activated carbon (AC-C); Langmuir (equation 6), Freundlich (equation 7), Dubinin-Radushkevich (D-R) (equation 9), and Temkin (equation 8) (Table 1). The linear forms of these models are visually presented in Figure 9 (a)–(d), while Table 5 provides a detailed breakdown of the associated constants, plot-based correlation coefficients (R2), and their dimensional components. The AC-C surface boasts remarkable uniformity, with a consistent distribution of homogeneous adsorption sites. This convincing evidence points towards the Langmuir isotherm's ability to accurately portray the removal of metal ions by AC-C. The D-R isotherm calculations yielded insightful energy values: 35.36 kJ mol−1 for Pb(II), 8.84 kJ mol−1 for Co(II), and 10.66 kJ mol−1 for Cd(II). These values reveal that Co(II) undergoes physical adsorption, while Pb(II) and Cd(II) are chemically removed by the AC-C surface. Temkin constant provides clues about interaction strength. The negative Temkin constant values (B) detected for each metal ion offer valuable perceptions into the strength of interaction between the metal ions and the AC-C adsorbent. The results explore AC-C's notable affinity for these metal ions, prominence its effectiveness in removing them through miscellaneous mechanisms, both physical and chemical.

The linear isotherms of (a) Langmuir, (b) Freundlich, (c) Temkin, and (d) D-R of Pb(II), Co(II), & Cd(II) removal by AC-C.
Isotherms model constants.

Thermodynamics
To attain a deep recognizing of the energetic scene directing metal ion removal by AC-C, a rise of accurately controlled thermodynamic investigations were assumed. The experiments were conducted at three distinct temperature plateaus—25 °C (±1.0 °C), 35 °C (±1.0 °C), and 45 °C (±1.0 °C)—with a constant agitation time of 1.0 h. This accurate protocol confirmed precise data acquisition and facilitated the ordering of the underlying energy dynamics. From the accurately collected data, the enthalpy change (ΔH°) and entropy change (ΔS°) associated with metal ion removal were precisely calculated. This involved leveraging Equation 13 (Table 1), which investigates the linear affiliation between the natural logarithm of the equilibrium constant (ln KL) and the reciprocal of the temperature (1/T). This rigorous approach unveiled the role of both enthalpy and entropy in governing the efficiency of metal ion removal by ACC. Furthermore, the conventional Gibbs free energy (ΔG°) was estimated using Equation 12 (Table 1), supplying valuable insights into the spontaneity of the process. The findings of the thermodynamic analysis are presented in Table 6.
Thermodynamic parameters of metal ion removal by AC-C.

The negative ΔG° for Pb(II) adsorption onto AC-C signifies that the process is spontaneous, indicating a natural tendency for Pb(II) to be removed from the aqueous solution by AC-C. Conversely, the positive ΔG° obtained for both Co(II) and Cd(II) adsorption propose a non-spontaneous adsorption. The positive ΔH° for the three metal ions designates an endothermic adsorption onto AC-C. The positive ΔS° value propose a strong affinity between the AC-C surface and the metal ions, leading to a more ordered and stable arrangement of the adsorbed species compared to the bulk solution.
Evaluating AC-C for heavy metal removal from polluted water
To validate the applicability of AC-C, a real sample from polluted water was used to examine the efficiency of AC-C for the removal of heavy metal ions. A 50 ml of polluted water sample was agitated with 0.07 g of AC-C under the optimized conditions recognized for the metal ions in this study. The results obtained in Figure 10 provide convincing evidence for AC-C efficacy in simultaneously adsorbing and removing a diverse range of contaminants from aqueous environments.
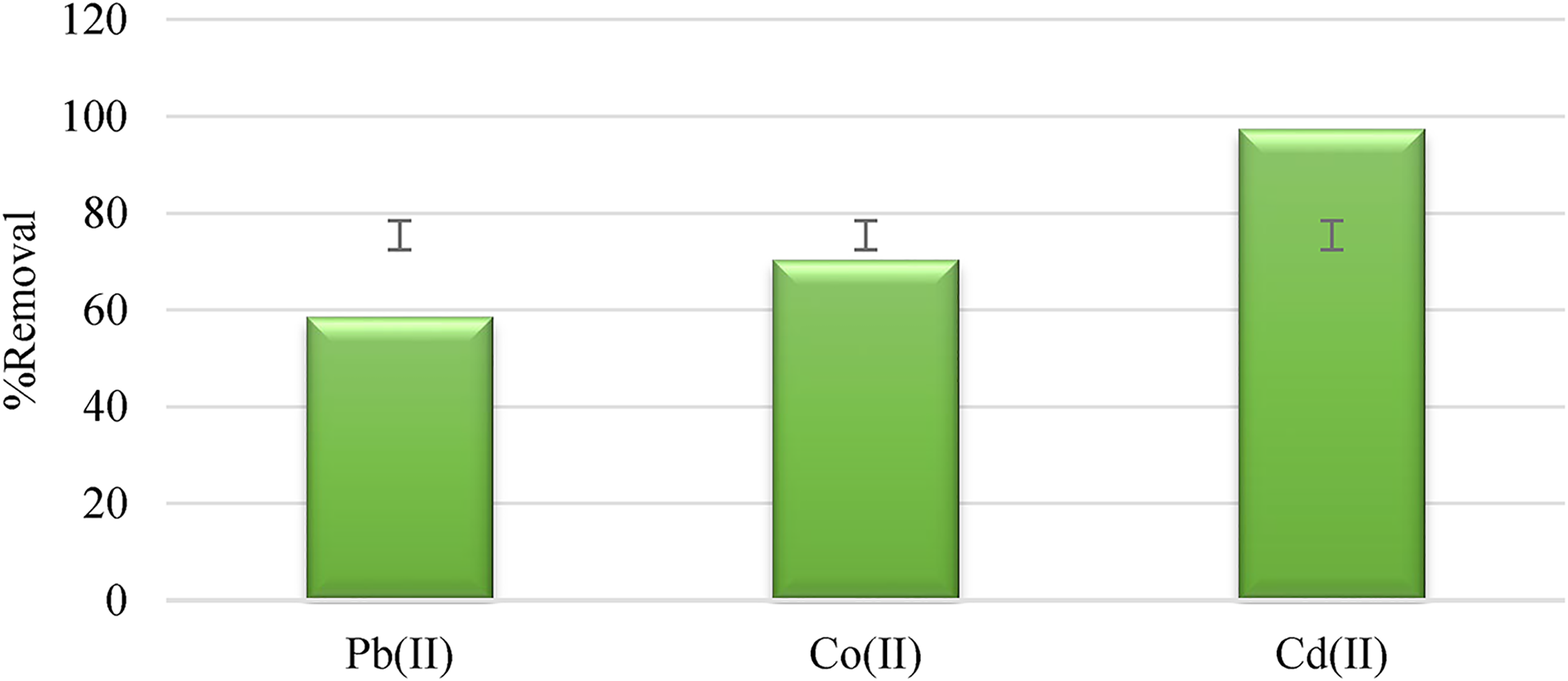
The simultaneous adsorption of metal ions Pb(II), Co(II), and Cd(II) on AC-C for a real sample.
Regeneration of AC-C
Hydrochloric acid (HCl) was credited as the optimal solution for eluting different metal ions from various adsorbent materials. In the current study, a mixture of metal ions of Pb(II), Co(II), and Cd(II) adhered to the AC-C surface was effectively eluted using an HCl solution and consequently analyzed using an atomic absorption spectrometer. The recovered percentage of metal ions was calculated using equation 14 (Table 1) (Nidheesh et al., 2012), offering a valuable estimation of the efficiency of the adsorption–desorption process. This important step allows for the quantification of metal ions removed from the solution and ultimately contributes to the assessments of AC-C performance as an environmental remediation tool. Figure 11 offers convincing evidence for AC-C's remarkable reusability. After undergoing five adsorption–desorption cycles, the recovery of metal ions revealed a minor decrease, with Pb(II) dropping from 89.29% to 73.14%, Co(II) dropping from 66.49% to 43.77%, and Cd(II) experiencing a reduction from 64.94% to 51.36%. The progressive waning of AC-C indicates its capacity to be reused repeatedly in an aqueous environment to remove heavy metals in an efficient manner.
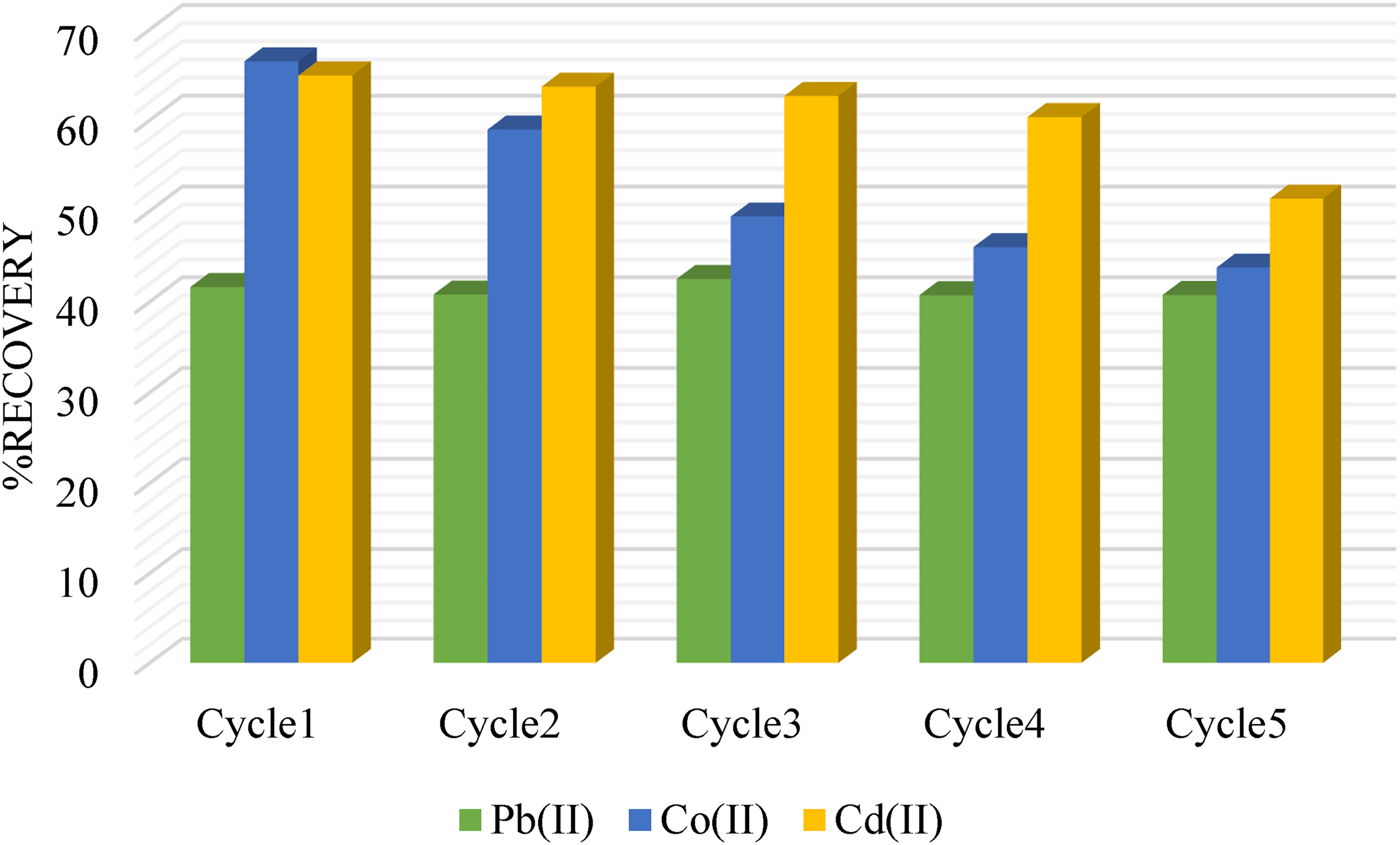
Regeneration of AC-C adsorbent.
Conclusion
The current study validates the effectiveness of the simultaneous removal of Pb(II), Co(II), and Cd(II) from aqueous solutions and synthetic samples by utilizing activated carbon prepared from cypress fruit (AC-C). The findings of the experiments have definitively demonstrated the optimal parameters that are most suitable for attaining the highest possible simultaneous removal of Pb(II), Co(II), Cd(II) by the use of AC-C. The experimental results clearly show that 0.07 g of AC-C and 90 min of agitation are the optimum conditions for AC-C to remove the three metal ions simultaneously. The results also demonstrated that acidic and neutral solutions are effective for removing Pb(II), whereas basic solutions perform better for Co(II) and Cd(II). The kinetic and isothermal calculations have shown that the adsorption of the heavy metal ion mixture by AC-C closely approximates the pseudo-second-order kinetic model and the Langmuir isothermal model, respectively. In addition, the thermodynamic analysis (+ve enthalpy and entropy) demonstrated that AC-C removes a mixture of heavy metal ions from the environment in a non-spontaneous, endothermic manner. After five cycles, an AC-C would become less effective, but it would still work.
Footnotes
Acknowledgment
The authors would like to express their appreciation to Isra University Innovation Center (IUIC) for the vital role in testing the samples. The state-of-the-art infrastructure at IUIC significantly contributed to the accuracy of our findings, and we are grateful for their commitment and professionalism.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
We confirm that this work is original and has not been published elsewhere nor is it currently under consideration for publication elsewhere.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Isra University with grant number of [8-38/2020/2021].
