Abstract
The discharges enriched with heavy metals, particularly Cr(VI), are a serious environmental problem. In this work, it is presented the use of biosorbents from eggshells membranes of ostrich, quail, duck and chicken; the membranes were easily prepared and studied for the removal of Cr(VI) from aqueous solutions. Maximum uptake of 1.41 mmol g−1 membrane permitted the identification of ostrich membrane as the material with the highest Cr(VI) uptake. N2 adsorption experiments permitted to classify ostrich membranes as mesoporous materials, since they had pore size of 14.8 nm. This represented an advantage on the nature of these membranes to be used as biosorbents with respect to other membranes. The adsorption isotherm models such as Langmuir and Freundlich were verified using experimental data. Langmuir model described the adsorption process satisfactorily at pH 1 and 2 whereas Freundlich model fitted data better at pH 3 and 5. The adsorption kinetic data were adjusted to a pseudo-second order kinetic model. TGA results permitted demonstrating a low thermal stability of ostrich membrane, since it starts decomposing at around 50°C due to the fact that membranes have about 60% protein. Through SEM microscopy was possible to observe the morphology of the membrane surfaces before and after Cr(VI) uptake which reflects the textural characteristics of the biosorbent. FTIR and XPS analyses suggested that Cr was adsorbed on ostrich membranes through the amine groups of proteins conforming the fibers of membranes. Despite the oxygen shows higher electronegativity than N, Cr shows more preference toward N due to fact that pH conditions favor the positive charges on the surface of the membrane. Our results make evident that this waste material is a novel and efficient option for the removal of Cr(VI) from aqueous solutions.
Introduction
The removal of heavy metals from aqueous media is an important aspect to reverse the pollution indexes that degrade the natural media. Due to industrial development and urban discharges, high amounts of heavy metals such as cadmium, mercury, chromium and lead are discharged into the environment and must be removed from water streams. Particularly chromium which is widely used in industrial processes such as: metallurgy (Karamchedu et al., 2015; Wang et al., 2018), tannery (Moretto, 2015), textile industry (Sanyal et al., 2015), plating (Kwon et al., 2004), wood conservation (Ohgami et al.,2015) and in chromate compounds preparation. Chromium exists in oxidation states +3 (III) and +6 (VI) and both are present in the environment (Oliveira, 2012). Toxic effects of Cr(VI) in humans are lung cancer (Abreu et al., 2018; Gibb et al., 2000), dermatitis (Roto et al., 1996; Yoshinaga et al., 2018), skin and mouth ulcers (Sun et al., 2015), perforation of nasal septum (Aiyer and Kumar, 2003; Yoshinaga et al., 2018), and all these at concentrations as low as 0.025 mg L−1. The presence of this metal ion in aquatic media is undesirable and has to be removed. The World Health Organization (WHO) sets a maximum permissible limit in drinking waters of chromium at 0.05 mg L−1 (WHO Library Cataloguing-in-Publication Data, 2011).
During the last decades, several research works focused on the search of non-conventional adsorbents for the removal of organic and inorganic pollutants. Thus, it has been reported the use of adsorbents such as activated carbon from papaya shell (Abbaszadeh et al., 2016; Rangabhashiyam et al., 2018), almond shell (Bouaziz et al., 2015; Maaloul et al., 2017), biomass (Gautam et al., 2014), chitosan (Wang and Chen, 2014), fruit remains (Romero-Cano et al., 2016), rice shell (Song et al., 2013), and sawdust (Basso et al., 2002), among others, were reported for the removal of heavy metal ions from aqueous solutions. The use of waste materials as biosorbents resulted in a good option from the environmental point of view, since in addition to being efficient these adsorbents can be obtained relatively easy. The study of eggshells for this purpose, has increased in the last few years, since the eggshells demonstrated to be capable of removing metal ions from solution (Guru and Dash, 2014). Although not only the eggshells have been used for this aim, eggshell membranes have also demonstrated to be good biosorbents (Baláž, 2014). However, the reported materials had the drawback of using non-ambient conditions for the removal of heavy metal ions.
Chicken Eggshell Membranes have been used in the area of biotechnology for mineralization studies as adsorbents for the removal of heavy metals (Pettinato et al., 2015), organic substances as dyes (Pramanpol and Nitayapat, 2006), and as biosensors (Zheng et al., 2010). Studies conducted by Rajendran and Mansiya (2011) and by Daraei et al. (2014) reported the use of hen eggshell membrane in the uptake of Cr(VI) considering effects such as temperature, contact time, concentration, pH and amount of adsorbent. The high adsorption this material showed on the metal uptake was due to its physical properties and to its composition, and it was influenced by the pH. However, to the best of our knowledge, there are no up to date studies dealing with eggshell membrane wastes of birds such as ostrich, quail and duck used for the removal of heavy metal ions from aqueous solutions, with the exception of chicken.
Chicken eggshell membranes are different from the ostrich eggshell membrane in that the second one contains lectin-type C proteins called struthiocalcin-1 (SCA-1) and struthiocalcin-2 (SCA-2) as the main components, whose amino acid sequence is 132 and 136 long, respectively, and show only one sequence identity of 41% with respect to the protein from hen eggshell in the case of SCA-1 and only 36% in the case of SCA-2. Ostrich eggshell membrane contains a seventh additional cysteine located near the extreme of C-terminal from SCA-1 by which ostrich eggshell membrane contains sections of amino acids negatively charged absent in the ovocleidin 17 (OC-17) of Chicken Eggshell Membranes (Mann and Siedler, 2004), and which can be beneficial for metal uptake. These features make the ostrich membranes unique and profitable for heavy metal ions removal.
The main objective of this work was to study the use of waste biosorbents from eggshell membranes of different birds in the removal of Cr(VI) from aqueous solutions, and to select the one with the best Cr(VI) uptake for further characterization as well as to describe the efficiency and adsorption mechanism on the biosorbents. Moreover, specific surface area, pore volume and average pore diameter of these membranes were analyzed by Nitrogen adsorption measurements as well as surface morphology by Scanning Electron Microscopy (SEM). Thermal stability of these membranes as biomaterials was tested using Thermogravimetric Analysis (TGA). Elemental Analysis (EA) and FTIR spectroscopy were performed to determine the chemical composition and functional group changes of the membranes before and after the Cr(VI) treatment. Finally, X-ray photoelectron spectroscopy (XPS) was conducted to determine the presence of Cr onto the membranes and which functional groups could be involved in the adsorption process.
Experimental
Eggshell membranes from different birds
Ostrich waste eggshells were collected from a local restaurant devoted to the growth and sale of ostrich products. Quail and duck waste eggshells were obtained from a local market and chicken eggshells from a local bakery. Membranes were separated by hand from the eggshells, washed with deionized water several times, and dried at 313.15 K in an electric oven for 4 h. Then, the membranes were crushed in liquid nitrogen and sieved through a No. 18 ASTM sieve; after this process they were stored in plastic bags at room temperature.
Aqueous solutions of Cr(VI) and their concentrations
A stock solution of Cr(VI) (1.41 g L−1) was prepared by dissolving dry K2Cr2O7 in deionized water with a resistivity of 18 MΩ (Barnstead,Chicago, Ill). All diluted solutions used were prepared by dilution of the stock solution and adjusted to different pH values with 0.01 mol L−1 NaOH or metals-free HNO3.
Cr(VI) concentration in aqueous solutions
The real concentration of Cr(VI) was determined by using a colorimetric method by reaction of Cr(VI) with diphenylcarbazide in acid medium; this develops a pink color complex whose photometric intensity at 540 nm is directly proportional to the concentration of Cr(VI) in solution. A calibration curve was prepared (0.2–1 mgL−1) to determine the final solution concentration by using a Thermo Scientific spectrophotometer model Nicolet iS5 iD5 at 540 nm.
Maximum Cr(VI) uptake
The maximum uptake measurement was performed with the aim of selecting the egg shell membrane that showed the highest Cr(VI) uptake. For this measurement the membranes of quail (QM), chicken (Chicken White Membrane, CWM, and Chicken Red Membrane, CRM), duck (DM), and ostrich (OM) were selected and contacted with a Cr(VI) solution of known concentration by dilution from a stock solution of 5000 mgL−1 Cr (VI) prepared from K2Cr2O7 (99.8%, Fermont); the pH of this solution was adjusted to 5.1 with 0.10 NaOH. About 0.1 g of each membrane were put in an Erlenmeyer flask and added 20 mL of the 5000 mgL−1 solution and stirred in a Thermoscientific thermo agitator model 3597 operated at 120 rpm and 298 K for 3 h. After this, the suspension was filtered and the pH and Cr(VI) concentrations of both initial and final solutions were measured.
Adsorption equilibrium data
Adsorption equilibria measurements were conducted on sample OM since this sample showed the highest Cr(VI) uptake. Samples were previously treated as mentioned in ‘Eggshell membranes from different birds’ section. Several solutions were prepared at different Cr(VI) concentrations (100–5000 mgL−1) and the pH adjusted to 1,2,3 and 5. About 0.1 g of the membrane OM were contacted with 20 cm3 of each solution in Erlenmeyer flasks and stirred at 298 K. Preliminary experiments showed that 5 days were enough to reach equilibrium. After this, the contacting solutions were filtered and the pH of both initial and final solutions measured with a Thermo Scientific Orion Star A211 instrument. The final concentrations of Cr(VI) were also measured. The amount of Cr(VI) adsorbed in equilibrium was determined by the following mass balance (Ballav et al., 2018):
Where q is the mass of Cr(VI) adsorbed by mass of adsorbent (mg g−1), v is the initial volume of Cr(VI) solution added to the adsorbent, L, C0 is the concentration of initial Cr(VI) solution (mg L−1), C is the concentration of Cr(VI) at equilibrium (mg L−1), and m is the mass of ostrich membrane used.
Adsorption kinetics of Cr(VI) uptake
The kinetic study was performed by using 0.1 g of adsorbent and contacted with 20 cm3 of a 100 mg L−1 Cr(VI) solution at pH 1. The flasks were placed in a thermoagitator Thermoscientific model 3597 operated at 120 rpm and 298 K. Then, solution samples were taken at different time intervals and filtered to analyze Cr(VI) concentration. The amount of Cr (VI) adsorbed was determined by equation (1).
Characterization of biosorbents
Studies about surface morphology of ostrich eggshell membranes were conducted by Scanning Electron Microscopy (SEM) using a Hitachi model TM1000 microscope for the stereoscopic observation. Textural properties of membranes such as specific surface area and pore size were studied by N2 adsorption measurements using an ASAP 2020 Micromeritics instrument (Nocross, GA, USA) at 77 K. Approximately 0.3 g of sample was heated at 50 °C for 8 h under vacuum to remove all impurities such as H2O vapor and CO2 previously adsorbed onto the surface of membranes that otherwise will cause an error in the surface area measurement. The BET surface area (SBET) was calculated using the BET equation. Structural parameters such as pore size and pore volume were calculated using the BJH method (Thommes et al., 2015). Percentage compositions of carbon, nitrogen, sulfur, and oxygen on samples of the different membranes were determined by elemental analysis using a LECO TruSpec CHNS instrument. About 2 mg of the dried sample was used for the analysis. Sample analysis was conducted in triplicate and averaged values are reported. The TGA analysis was performed using a TA Instruments equipment Discovery model. The analyses were performed by using dried and sieved samples. About 10 mg of sample was placed in a platinum pan; a temperature scanning in the range of 25–500°C was used; the heating rate was 20 °C/min under a nitrogen atmosphere at 60 mL min−1. FTIR spectra were obtained using an FT-IR Thermo Scientific spectrophotometer model Nicolet iS5 iD5 instrument; the wavenumber range was 400–4000 cm−1 with a spectral resolution of 4 cm−1 at 298 K. XPS measurements were performed with a SPECS XPS spectrometer. The base pressure in the analyzer was less than 1.2 × 10−9 Torr. XPS spectra were taken using an aluminum anode X-ray source, operating the Al anode at 3.9 kW (13 kV and 300 mA). High-resolution scans were performed over the range of 580–602 eV (Cr 2p) with pass energy adjusted to 25 eV. After Shirley base line subtraction, curve fitting was performed using the nonlinear least squares algorithm and assuming a Gaussian variable proportion peak shape.
Aqueous phase chemical speciation of chromium
In order to have a better understanding about the adosprtion of Cr(VI) on the membranes, Cr species in solution were identified by using the software MINEQL+ to solve the aqueous phase chemical equilibria at different pH values, ionic strength and temperature (Schecher and McAvoy, 2003). This program solves chemical equilibria by considering corrections on activities and ion strength on the equilibrium constants of solution by using the Debye–Hückel equation as well as on the enthalpy on the stability constants; the concept of solubility product is included in the program; the program also makes simulations of addition of acids and bases. The system considered was (K2Cr2O7 – H2O). The chemical species considered are shown in Table 1 and chemical reactions are shown in Table 2. Chemical species distribution diagram for Cr(VI) as a function of water pH is shown in Figure 1 for a concentration of 1.92 x 10−2 mol L−1 (1000 mg L−1) and at 25 C. The diagram shows that at pH < 6.0 chromium is present in anionic form as both acidic chromate HCrO4- and dichromate Cr2O7−2. CrO42- species is about 90% of the total concentration at pH > 7. These results indicate that at pH < 5 ostrich eggshell membrane surface protonation is favoured, thus allowing charge interactions between the surface and Cr ionic species in solution.
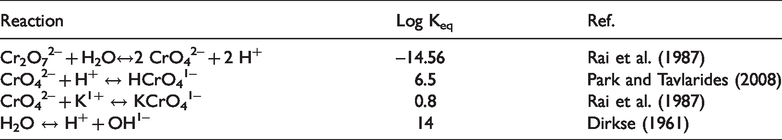
Chemical species considered for the system K2Cr2O7 – H2O.

Equilibrium chemical reactions in aqueous phase for Cr(VI).

Cr(VI) species distribution as a function of solution pH. Cr(VI)TOT = 1.92 × 10−2 mol L−1; T = 298.15 K.
Results and discussion
Nitrogen adsorption results
Specific surface area, pore volume and average pore diameter of sample OM were determined with values of 1.95 ± 0.08 m2g−1, 0.004 cm3g−1, and 148.4 Å, respectively. The highest specific surface area corresponds to OM, and the lowest is presented by DM, while CRM and QM present similar values in surface areas (Table 3); then, OM would have greater possibilities for Cr removal. Regarding the pore size, the lowest value corresponds to CRM, suggesting that this material has a greater possibility of adsorption; however, its specific surface area is lower than the corresponding to OM sample. In order to define which of these two factors is determinant in the Cr(VI) uptake process, the maximum Cr(VI) uptake was evaluated, as described later. On the other hand, regarding the type of pore and according to IUPAC classification (Thommes et al., 2015), OM corresponds to a mesoporous material, since it has a pore size of 148.4 Å. This represents an advantage on the nature of OM to be used as biosorbent with respect to the other membranes. A type II isotherm is obtained (Figure 2(a)) and it represents unrestricted monolayer-multilayer adsorption. The beginning of the almost linear middle section of the isotherm is often taken to indicate the stage at which monolayer coverage is complete and multilayer adsorption about to begin. A very slight hysteresis loop of the H3 type is obtained, which according to IUPAC classification, does not exhibit any limiting adsorption at high relative pressures and is observed with aggregates of plate-like particles giving rise to slit-shaped pores. In order to validate the viability of BET equation in isotherms, the CBET parameter was included in Table 3 for all membrane samples. Parameter CBET gives an idea of the half life time of adsorbate on the surface of adsorbent. A high value of C (∼100) is associated with a sharp knee in the isotherm, thus making it possible to obtain by visual inspection the uptake of the point at which the beginning of the almost linear middle section of the isotherm. It is often taken to indicate the stage at which monolayer coverage is complete and multilayer adsorption is about to begin. On the other hand, if C is low (<20) this point cannot be identified as a single point on the isotherm (IUPAC, 1982). In other words, if CBET is high, the residence time of adsorbate is high, indicating a high superficial interaction potential, and consequently an increased heterogeneity. In this way, CBET parameter gives an idea of the heterogeneity of the surface. The results for CBET are relatively low for all membrane samples (23.82–105.37), indicating a medium to high heterogeneity of the surfaces.
Summary of properties of adsorbents.

QM: quail membrane; DM: duck membrane; CWM: chicken white membrane; CRM: chicken red membrane; OM: ostrich membrane.
Errors are the standard deviation of three independent measurements.

(a) Nitrogen adsorption isotherm on OM sample at 77 K, (b) Maximum Cr(VI) uptake on different eggshell membranes from different birds.
Elemental analysis results
The percentages of N, C, H, S and O in this sequence were 14.33%, 45.08%, 7.57% 2.52 and 30.5% for CRM; 14.73% 43.51% 7.28%, 3.15% and 31.33% for CWM; 14.51%, 41.13%, 7.05% 3.09 and 34.22% for DM; 14.75%, 44.77%, 7.29%, 2.66 and 30.53% for QM; and finally, 14.03%, 40.46%, 7.27%, 3.57% and 34.67% for OM The sulfur percentage of OM (3.57%) was slightly greater than the obtained for CWM (3.15%), which can be associated to the presence of additional cysteine (Mann and Siedler, 2004) that has OM in its terminal chain and which can result beneficial for Cr (VI) uptake.
Maximum Cr(VI) uptake
The maximum Cr(VI) uptake is shown in Figure 2(b)) and corresponds to OM sample with 73.4 mg Cr (VI)g−1 (1.41 mmol g−1 membrane) in 3 h. Furthermore, this membrane showed the highest surface area (1.95 m2g−1), therefore, OM is the material with higher probability to remove more Cr(VI) ions. Consequently, OM was used for the study and characterization of Cr(VI) removal from aqueous solutions.
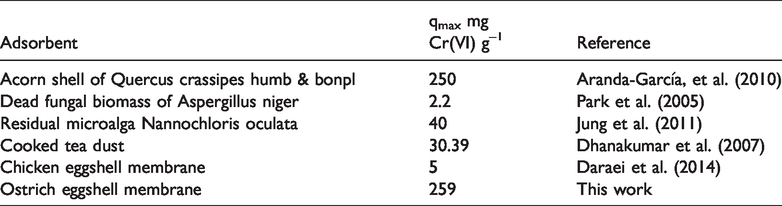
With the objective of comparing the properties of biosorbents studied in this work with other natural materials used for removal of Cr(VI), in Table 4 are shown the adsorbent capacities employed by other authors from the literature. It can be observed that biosorbent OM of our work results to be a good option for the removal of Chromium ions (259 mg Cr(VI) g−1 of OM) since it can compete with other natural adsorbents under acidic conditions and having clear advantage on the chicken membrane (5 mg g−1) and slightly overcoming the acorn shell from Quercus crassipes humb & Bonpl (250 mg g−1) studied by Aranda-García et al. (2010).
Comparison of Cr(VI) uptake with different materials.
Kinetics and adsorption equilibrium results
Adsorption kinetics demonstrated that Cr(VI) concentration of the contacting solution affects directly the amount of Cr(VI) adsorbed and that the higher the concentration the higher the adsorption of metal (Figure 3(a)). On the other hand, the equilibrium time can be observed to be more defined from 20 h for a concentration of 100 ppm, while in the case of 500 ppm it starts having no significant changes from 80 h. Finally, the time corresponding to 1000 ppm shows that it reaches equilibrium after 100 h. It can be inferred that there are mass transfer resistances on the adsorbent and therefore it takes longer in reaching the equilibrium despite the fact that uptake is comparable with other natural materials. The interaction of Cr(VI) with OM agrees with our FTIR findings where the bands of functional groups C = O were more affected after metal uptake, indicating that Cr(VI) could be interacting with these groups due to the acidic conditions promoting amino acid protonation which attract the ionic forms of chromium in solution. This fact can be observed in table S1 of the Supplementary information where correlation coefficients are close to unity and the values of rate constant increase as concentration decreases.

(a) Adsorption kinetics of ostrich eggshell membranes (OM) using 20 cm3 of Cr(VI) solution at pH 1 at different C(VI) concentrations and 0.1 g of OM at 298.15 K, (b) Plot of pseudo-second order kinetics for adsorption of Cr(VI) on OM at different concentrations, (c) Adsorption isotherms for Cr(VI) uptake on sample OM at three pH values. Experimental conditions: solution volume = 25 cm3, weight of OM = 0.1 g, T = 298.15 K, (d) Adsorption isotherm for Cr(VI) uptake on OM sample at pH 1. Experimental conditions: solution volume = 25 cm3, weight of OM = 0.1 g T = 298.15 K.
In order to study the adsorption kinetics of Cr(VI), the pseudo-first and pseudo-second order kinetic models were considered. The first model assumes that the adsorption process from a liquid to a solid phase is a reversible reaction with the equilibrium established between the phases; this kinetics assumes that adsorbate accumulates on the surface of adsorbent as time elapses. Equation (2) represents the pseudo-first order rate (Chen and Wang, 2009):

Where qe and qt represent the amount adsorbed at equilibrium at any time and k1 is the pseudo-first order rate constant. k1 was obtained from the slope when plotting log qe and qt vs time. On the other hand, the pseudo-second order model is based on the equilibrium capacity of adsorption of the system and indicates that the adsorption capacity is proportional to the number of active sites occupied on the surface. Equation (3) represents the pseudo-second order rate (Ho and McKay, 1999):

The values of Kad and qe can be described from the slope when plotting t/qt vs. t, respectively. When comparing the R2 values for the pseudo-first and pseudo-second order kinetic models it can be concluded that a pseudo-second order kinetics is followed. Determination of kinetic parameters in the adsorption of Cr(VI) from liquid phase to solid phase can be considered a second order reaction where the adsorption rate constant Kad was obtained by assuming that desorption rate is small compared to adsorption rate. Data shown in Figure 3(b) follow the second order equation proposed, and shown as integrated rate is:

Where Co is the initial solution concentration and C is the concentration of the aqueous phase at time t.
Langmuir and Freundlich isotherms were applied here to obtain a better understanding of the adsorption process of chromium on the OM sample. Langmuir isotherm works perfectly in the processes of monolayer adsorption where it is considered that at any point on the surface of adsorbent there is the same activity of adsorption of each molecule on the surface. Freundlich isotherm assumes a heterogeneous surface where the magnitude of adsorption heat varies exponentially with the covering of the surface and expresses a multimolecular adsorption. Langmuir and Freundlich isotherms are given by:


Where qmax is the maximum amount of Cr(VI) adsorbed (mg g−1), K is the equilibrium constant (L mg−1), C is the equilibrium Cr(VI) concentration (mg L−1), a is a constant that represents the adsorption capacity (mg1−1/ n L1/ n g−1), and 1/n is Freundlich constant and indicates the intensity of adsorption. Table 5 shows the regressed values of Langmuir and Freundlich isotherms and the experimental values of initial and final metal concentrations of the adsorption of Cr(VI) on OM and indicate that the Langmuir model describes the adsorption process satisfactorily at pH 1 and 2 while Freundlich model fits better at pH 3 and 5. In general, the correlation coefficients are close to 1, and the difference between the two models can be explained by the fact that at values of pH < 4 despite the surface is positively charged, there was a dissolution reaction of fat of the adsorbent (at pH >4) which promoted that the amount of Cr(VI) adsorbed decreased noticeably. Figure 3(c) depicts the adsorption isotherms of Cr(VI) on ostrich eggshell membranes at pH 2, 3 and 5. It can be observed from this figure that pH has a significant effect on metal uptake, since the highest uptake (63.25 mg g−1) is shown at low pH values. This behavior is due to the fact that when pH decreases the protonation increases, which makes that Cr(VI) shows preference on the protonated active sites, whereas when pH increases, electrostatic repulsion forces increase causing retarded diffusion and therefore low Cr(VI) uptake. On the other hand, Figure 3(d) shows adsorption isotherm at pH 1 showing the maximum Cr(VI) uptake (285.9 mg g−1) on the material and due to scaling issues, it has to be shown independently. It can be observed from this plot that the isotherm remains unchanged from a 2000 mgL−1 concentration thus considering that under these conditions equilibrium is reached, indicating that the ostrich membrane starts saturating when approximately 259 mg Cr (VI) g−1 have been adsorbed. Furthermore, after complete saturation, the membranes were easily regenerated with 50 cm3 of 1.0 M HNO3 (or HCl) solution with 82% of the chromium recovered for the biosorbents. Then, the material can remove higher amounts of Cr(VI) when pH 1, being evident that Cr(VI) species present in the solution will tend to equilibrate with the membrane active sites.
Linear regression values of Langmuir and Freundlich isotherms and initial and final values of experimental concentrations.

aFreundlich equation isotherm.
SEM results
With the SEM technique it was possible to observe the morphological surface of the membrane before and after contacting with aqueous solutions of Cr(VI) which reflects the textural characteristics of the biosorbent. The profile of pristine OM is shown in Figure 4(a) noticing that the proteins of membranes are cross-linked. OM presents an easily distinguishable arrangement due to the orientation (Mann and Siedler, 2004). It is noticeable that the outer membrane fibers are of higher diameter compared to the ones from internal diameter, which makes larger spaces to be observed between the fibers of outer membranes. Therefore, it is evident from micrograph in Figure 4(a) that OM before metal uptake shows only cross-linkage of proteic fibers. However, in the case of material contacted with metal (Figure 4(b), pH 1 and 5000 mg L−1 Cr(VI)) it can be observed surficial differences such as the enlarging of the fibers of membrane where the metal remains adsorbed. This enlargement makes that the surface of proteins and, consequently, the functional groups, to be more exposed. In micrograph of Figure 4(b), it is also possible to observe that in the surface of fibers, small cumulus of solids are present corresponding to the interaction of chemical functional groups of proteins, which act as nucleation centers where metal ions keep accumulating. Despite the fact that characterization from N2 adsorption measurements showed that the membranes were mesoporous, a Cr(VI) uptake above 70 mg g−1 was obtained, thus demonstrating that the responsible of metal uptake are the superficial interactions between chemical functional groups. Furthermore, these results demonstrate the way in which Cr(VI) is caught by the membrane fibers in agreement with the results provided from the nitrogen adsorption measurements and the equilibrium adsorption isotherms. Finally, these results suggest that Cr(VI) has influence on the morphology of the surface after metal uptake.

(a) Profile of pristine OM, (b) OM membrane after Cr(VI) uptake.
TGA results
Thermograms obtained are shown in Figure 5 as mass % loss and its first derivative as a function of heating temperature T for samples OM (Figure 5(a)) and CWM (Figure 5(b)) for comparison purposes only. The thermal stability of both materials OM and CWM is very low, since they start decomposing at around 50°C due to the fact that membranes have about 60% protein of their composition (collagen, glucosamine, chondroitin, and hyaluronic acid) (Mittal et al., 2016).These components are sensitive to elevated changes of temperature, mainly collagen whose tridimensional structure can be altered in temperature changes, pH, the presence of salts or nonpolar solvents (Feduchi et al., 2012). Furthermore, the results show that thermal decomposition of the sample OM is carried out in several stages. In the first stage, when heating of collagen starts, its denaturalization is caused and whose first consequence is the shortening of its fibers, which in this case is made at around 50 ˚C and agrees with the study of Bozec et al. (Bozec and Odlyha, 2011), in which the authors explained the denaturalization of collagen at different temperatures and it served as a reference for understanding of the degradation process of membranes. In the first stage occurs the main transition of gelatinization process under a hydrated environment, caused by the breakage of cross-linked fibers that shorten the fibers. The loss of mass of both samples is evident in this first stage where OM keeps losing mass constantly as temperature increases, whereas CWM is maintained without continuous changes and which can be attributed to the presence of hydroxyproline contained in each sample (Feduchi et al., 2012). The second stage occurs from 250°C for both samples where the loss of mass is about 40% and it can be associated to the changes of the triple helix of collagen. Finally, at temperatures higher than 336°C the loss of mass is higher than 50% in both samples indicating a constant degradation of collagen dried fibers as well as inorganic components such as calcium, magnesium, silica, zinc, etc (Baláž, 2014). It can be concluded that due to the composition of membranes, their thermal stability is very low. However, it can be noted that decomposition stages very slightly between the OM and CWM, mainly during the first stage where it can observed that the loss of mass is more notorious from OM within the first 50°C and that probably it can be related with the type of quaternary structure of collagen composing each of these membranes.

Thermograms of (a) Ostrich eggshell membrane (OM) and, (b) Chicken White Membrane (CWM).
FTIR results
Complexation between acidic chromate ion (HCrO4−) and chelating groups contained in the samples of OM was evaluated. In Figure 6 are shown the FTIR spectra of the samples of OM without chromium uptake (Figure 6(a)) and with chromium treatment at pH 1 (Figure 6(b)), pH 3 (Figure 6(c)) and pH 5 (Figure 6(d)). Due to the fact that OM is composed principally of proteins, repetition units of polypeptides and proteins conformation are given place to 9 absorption bands named amides A, B, I-VII (Kong and Yu, 2007). From these groups, infrared signals of amides I and II located below 1700 cm−1 are the most prominent and therefore, the most sensitive region to evaluate changes in the secondary structural components of the proteins. The OM spectrum (Figure 6(a)) shows characteristic absorption bands pattern for proteins where the broader signal at 3280 cm−1 corresponds to stretching vibrations of O-H and N-H groups. Stretching vibrations of C-H (sp2) and C-H (sp3) of the hydro carbonated system are observed at 3060 and 2924 cm−1, respectively. Furthermore, the most intense bands are associated to C = O stretching vibration of amide I at 1639 cm−1 and at 1533 cm−1 to the amide II (N-H bend, C-N stretching). Also, the signal at 1236 cm−1 corresponds to N-H and C-N of amide III associated to glycoproteins. Small signals located at 1450 and 1420 cm−1 can be attributed to C-H bending of the aliphatic carbons in the protein corroborating the band at 2924 cm−1. The bands at 1073 and 620 cm−1 correspond to stretching modes of C-O and C-S (Baláž, 2014). On the other hand, changes in the pattern of OM sample used to adsorb HCrO4- at different pH´s (Figure 6(b) to (d)) are associated principally to the amide moieties. In general, signals of the amide I and II decrease in intensity as a consequence of the presence of the HCrO4- ion which can be interacting with protonated N-H groups (ammonium ions) promoted by acid media and affecting the carbonyl group vibration mode. Some other signals of aliphatic C-H, amide III and C-O are also affected (1450 to 1000 cm−1) principally at pH 1 and 2. Here, by comparison with the original OM sample, the C-H aliphatic region is modified as a broad signal composed of several bands. The intensity of the amide III and C-O bands decrease by the presence of the Cr(VI) ion in the samples. This can be originated by the perturbation of second structure arrangement (Mallamace et al., 2015) either for the metal ion association or by the acid pH used in the treatments. Although changes in the intensity of the amide I and II bands are similarly observed in the spectra of sample at pH 5, the C-H, amide III and C-O bands region was less affected. It can be explained as the pH 1 and 3 media promote protein hydrolysis in the OM sample and that is supported by the broad signal composed of the several bands observed between 1500 to 1200 cm−1 in spectra b and c. In contrast, the presence of the chromium ion in the samples is also shown by a shift of frequency in the C-S band (from 597 to 625 cm−1). Finally, the presence of Cr(IV) is confirmed with the weak signals near to 890 and 950 cm−1 observed from the spectra b and c. They are attributed to Cr-O vibration bond of the HCrO4−1 anion (Holman et al., 1999).

FTIR spectra of OM before (a) and after Cr(VI) uptake: (b) at pH 1, (c) at pH 3 and (d) at pH 5.
XPS results
In the spectrum of C 1 s of OM (Figure 7(a)) there are two components, one at 283.4 eV associated to adventitious carbon, commonly found by using the XPS technique (Barr et al., 1995), the second component at 285.5 eV is associated to carbonyls and carboxylates (Beamson et al., 1991). After Cr adsorption (Figure 7(b)) this last component decreases in intensity compared to the one of adventitious carbon. Therefore, the carbonyl group also participates in Cr adsorption. This result is in agreement with the decrease of intensity of the C = O band from the FTIR spectra. In both spectra there is a third component at 282.2 and 281.9 eV (Figure 7(a) and (b)) whose assignation is uncertain since this is not recorded in the literature. Despite not knowing the assignation of this carbon species, its participation in complexation was ruled out due to the fact that intensity of the band remains high. On the other hand, the transition window of N 1 s (Figure 7(c) to (d)) shows that there are three components before adsorption of Chromium ions in the case of OM sample (Figure 7(c)) at 394.2, 396.7 and 398.7 eV characteristic of N in amines and amino acids. However, after Cr adsorption (sample OM-Cr, Figure 7(d)) the peak at 394.2 is not present indicating that the amount of nitrogen decreased on the surface of OM, the nitrogen species with binding energy of 394 eV was affected by Cr adsorption and consequently was the one that participated in complexation. According to the database of the National Institute of Standards and Technology (NIST) with this binding energy are present the species N = N (Kudo et al., 1986). Nevertheles, it cannot be ruled out that amine groups from the membrane also participate in complexation, since the bands associated to amines from FTIR results were affected after Cr adsorption. This can be explained with the interaction R-NH-CrO4, in this way the first coordination sphere is not affected and the XPS component appears at the same position. From Figure 7(e) and (f) it is clearly seen that sulphur does not participate in Cr uptake. Its two components showed approximately the same binding energy and intensity without significant changes before adsorption (Figure 7(e)) and after adsorption (Figure 7(f)). These results show that for this element there is a small displacement of binding energy of both components which is not significant and indicates that there is an increase in electronic charge, leading us to propose that sulphur does not actively participate in the metal chelation process compared with the rest of elements analyzed. In this sense, nitrogen is taken as an example in which significant changes of active participation in the chelating process can be observed, as evident from FTIR measurements (at 3300, 1600 and 1350 cm−1) corresponding to N-H or C-N of amides, whereas in XPS it is shown the disappearance of peak at 394.2 eV. Finally, the characteristic peak for Cr 2p can be seen from spectrum in Figure 7(g) for OM-Cr with a binding energy of 584.209 eV for the peak 2p1/2 and 574.5 for 2p3/2, proving that Cr is adsorbed on sample OM. Base line data treatment was performed by considering the Shirley criteria (Spectroscopy and Phenomena, 2006). The split of spin is 9.7 eV according to reports from the literature; the position of peaks is related to an oxidation state of +6 suggesting that Cr is bonded either to oxygen or nitrogen (Moulder et al., 1979). As a result of XPS and FTIR analyses it is possible to propose a schematized structure (Scheme 1) depicting the possible surface complex formed on OM where the metal is bonded through N groups of amine forming the primary structure of proteins conforming the membrane.

XPS spectra of: (a) C 1 s OM, (b) C 1 s OM-Cr, (c) N 1 s OM, (d) N 1 s OM-Cr, (e) S 2p OM, (f) S 2p OM-Cr, and (g) Cr 2p OM-Cr.
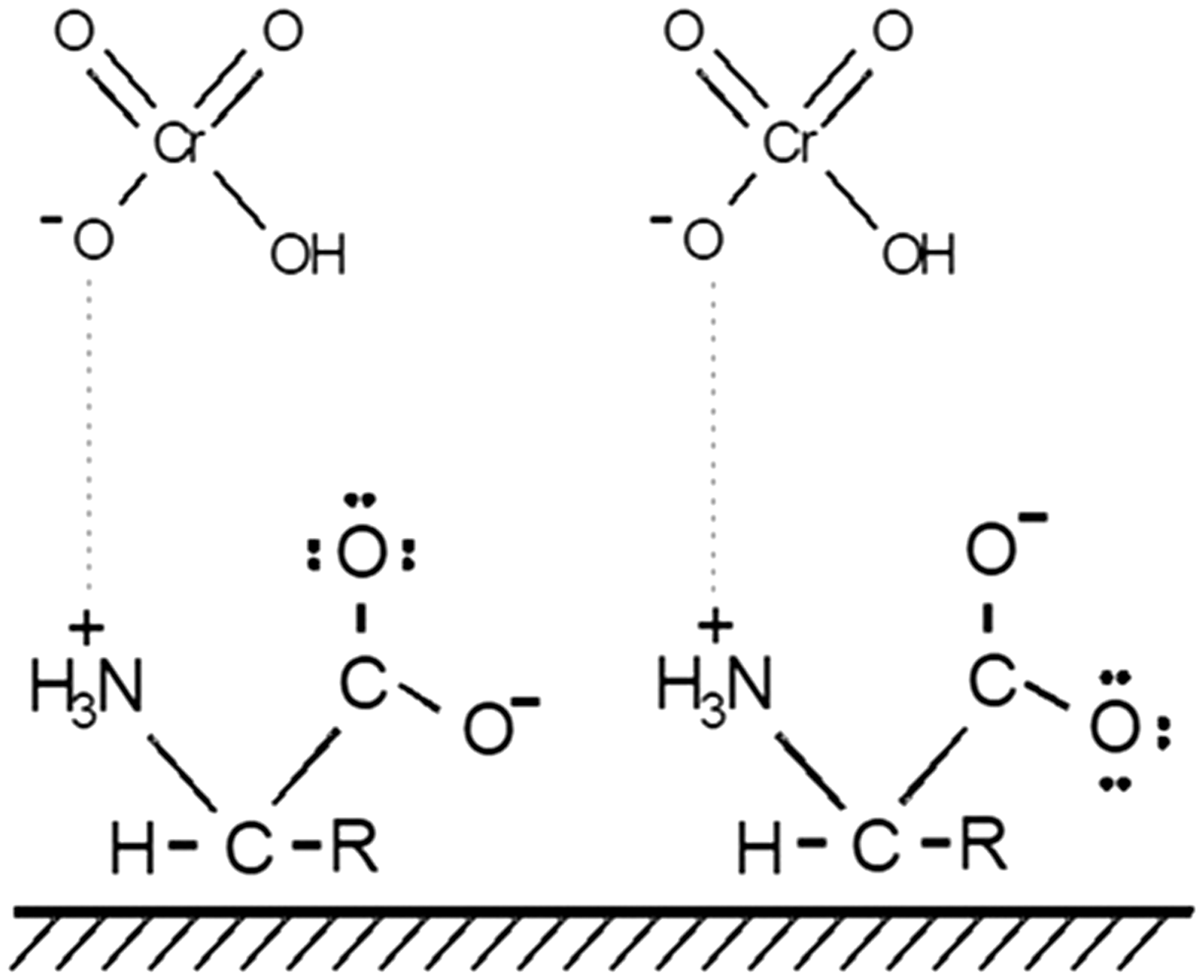
Proposed Cr complex formed on the surface of Ostrich Membrane, OM.
Conclusions
Eggshell membranes are waste materials generated in large amounts that can be reused in the removal of Cr(VI) from aqueous solutions. According to the results obtained ostrich membranes showed to be the materials with the highest Cr(VI) uptake at pH 1. The effect of solution pH on Cr(VI) uptake indicates that it can be applicable to relatively acidic solutions since membranes showed no dissolution effect. On the other hand, the thermal stability presented by ostrich eggshell membrane with respect to chicken eggshell membrane remains unchanged of loss of mass at 300 C. The adsorption process is explained better by a pseudo second order model, whereas equilibrium data could be adjusted to both Langmuir and Freundlich models depending upon adsorption pH of Cr(VI), this is due to the effect of dissolution of membranes at pH >3. The XPS analyses suggested that Cr was adsorbed on OM membranes through the N of amine groups of proteins conforming the proteic fibers of membranes, despite the oxygen shows higher electronegativity than N, Cr shows more preference toward N due to fact that the pH conditions favor the positive charges on the surface of the membrane.
Supplemental Material
sj-pdf-1-adt-10.1177_0263617420956893 - Supplemental material for Cr(VI) adsorption from aqueous streams on eggshell membranes of different birds used as biosorbents
Supplemental material, sj-pdf-1-adt-10.1177_0263617420956893 for Cr(VI) adsorption from aqueous streams on eggshell membranes of different birds used as biosorbents by Jessica Badillo-Camacho, Eulogio Orozco-Guareño, Gregorio G Carbajal-Arizaga, Ricardo Manríquez-Gonzalez, Icela D Barcelo-Quintal and Sergio Gomez-Salazar in Adsorption Science & Technology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Financial support of Mexicós Council of Science and Technology (CONACYT) is gratefully acknowledged for a scholarship granted to Jessica Badillo-Camacho No. 253704/214610. We thank to Dr. Victor Soto Garcia for his support with XPS analysis.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

