Abstract
The natural diatomite was treated with NaOH to obtain alkali-activated diatomite. The materials were systematically characterized by X-ray powder diffraction, X-ray fluorescence, Fourier transform infrared spectroscopic, scanning electron microscopy, and N2 adsorption–desorption. Meanwhile, the potential use of alkali-activated diatomite as adsorbent for the removal of basic fuchsin from aqueous solution was assessed by batch experiment. Results indicated that the structure and textural properties of diatomite were obviously changed via alkali activation, and then affecting its adsorption performance. The adsorption capacity of alkali-activated diatomite for basic fuchsin was higher than that of natural diatomite. In the case of alkali-activated diatomite, its adsorption capacity was increased with increasing the activation temperature, and the diatomite activated at 115°C (alkali-activated diatomite-115) exhibited the maximum adsorption capacity. The pseudo-first-order kinetics and the Sips isotherm model were preferable to describe the adsorption process of basic fuchsin on alkali-activated diatomite-115 and the thermodynamic parameters indicated that the adsorption process was endothermic and spontaneous.
Introduction
With the rapid development of the textile industry, the discharge of printing and dyeing wastewater is increasing year by year. The dyes composed of complex organic compounds with azo bonds and polyaromatic rings are toxic and difficult to degrade. If they are directly discharged into the river, they will have a great negative impact on the environment due to hindering the self-purification of the water body and poisoning the microorganisms and fish. Therefore, it is essential to remove dyes from wastewater before discharging in order to avoid pollution environment. The methods of dyes removal mainly include biological treatment, chemical treatment, membrane filtration, flocculation, oxidation, and adsorption (Cai et al., 2015; Gao et al., 2019; Hakimelahi et al., 2012; Huang et al., 2012; Ilha et al., 2009; Moura et al., 2018; Sójka-Ledakowicz et al., 1998; Tanyildizi, 2011). Among them, adsorption has been widespread application due to its simple operation, low operating costs, and high efficiency. Carbon material is a traditional adsorbent material due to its high adsorption capacity, in which activated carbon is the most widely used as adsorbent (Abbas and Harrache, 2019; Huang et al., 2012; Lafi et al., 2019; Streit et al., 2019; Wu et al., 2019). However, most of these carbon materials are microporous structures and more expensive, which limits their application in polymer dyes removal. Therefore, finding an adsorbent with high adsorption performance, abundance and cheapness is a hot topic in the field of printing and dyeing wastewater treatment. Generally, in order to assess and select an adsorbent, in addition to its adsorption capacity, other factors, such as production and regeneration cost, availability, environmental compatibility, should also be considered (Aivalioti et al., 2010).
Diatomite, a porous biogenic silicolite available in abundance deposits around the world, mainly consists of silicon (SiO2·nH2O) derived from aquatic, unicellular alga varying in shape and size of diatom with small amounts of Al2O3, Fe2O3, CaO, MgO, K2O, Na2O, P2O5 and organic matter (Li et al., 2017; Ma et al., 2015; Sharipova et al., 2017). Diatomite has the characteristic of high porosity, high permeability, large surface area, low density, excellent thermal, and mechanical stability (Deng et al., 2019). These properties make diatomite a potential adsorbent for removing aqueous pollutants, such as heavy metal ions (Dong and Zhang, 2013; Ma et al., 2015), dyes (Abukhadra et al., 2019; Li et al., 2017), triclosan (Sharipova et al., 2017), and benzene (Deng et al., 2019). In order to improve the adsorption ability of diatomite, the diatomite would be modified by physical and chemical methods. Al-Ghouti et al. (2009) used manganese oxides-modified diatomite to remove basic and reactive dyes from aqueous solution. Al-Qodah et al. (2007) used the acid treated diatomite to adsorb methylene blue from aqueous solutions. Aivalioti et al. (2010) used thermally modified diatomite to adsorb certain common petroleum contaminants. All these methods mentioned above can enhance the adsorption capacity of diatomite due to adjusting its structure.
In view of diatomite mainly composed of SiO2, in this work, the diatomite was modified by alkali activation method due to silicon dissolved by alkali, and a series of modified diatomite with different structure were obtained by varying the alkali activation condition, meanwhile it was used as adsorbent to adsorb basic fuchsin (BF) from aqueous solution. Meanwhile, the adsorption performance of modified diatomite to BF was evaluated by batch adsorption experiments. The isotherms, kinetics, and thermodynamics of the adsorption process were determined. It will lay the foundation for the construction of diatomite-based adsorbent with high-performance.
Materials and methods
Modification of diatomite
An appropriate amount of natural diatomite (Nat-Dia) was added into 6 wt.% NaOH solution to keep the concentration of diatomite suspension 4 wt.%. The mixture was vigorously stirred using magnetic stirrer (DF-101S) for 2 h at different temperatures (60°C, 85°C, 115°C, respectively), and then centrifuged at 4025 g-force for 6 min using high speed tabletop centrifuge (TG16-WS), washed with distilled water several times to neutral state, subsequently, dried in an oven (DHG-9101A) at 105°C for 12 h, and obtaining the modified diatomite was abbreviated to Alk-Dia-t, where t was treatment temperature.
Characterization techniques
X-ray powder diffraction (XRD) patterns of the samples were carried out on a Bruker D8 Advance X-ray diffractometer using monochromatized Cu/Kα radiation (40 kV, 40 mA). The samples were scanned with a rate of 2° min−1 in the range of 5° to 70°.
Fourier transform infrared spectroscopic (FT-IR) analysis was performed on a Bruker Tensor 27 spectrometer. The samples were scanned in the transmittance mode with a resolution of 4 cm−1 in the range of 400–4000 cm−1 using the KBr pressed disk technique.
The N2 adsorption–desorption isotherms of samples were recorded at −196°C using BelSorp-Max (Bel Japan Inc.). Before analysis, the sample (ca. 1000 mg) was degassed at 350°C for 5 h. The surface area was calculated using the Brunauer–Emmett–Teller (BET) equation.
Scanning electron microscopy (SEM) images were obtained on a Philips-FEI model Quanta 200. The samples were held with carbon adhesive tape on aluminum sample holder, and then coated with a gold layer using a sputter coater (Bal-Tec, SCD-050 model, Balzers, Liechtenstein), subsequently, examined with an operating voltage of 20 kV and a current of 30 mA.
X-ray fluorescence (XRF) spectra were recorded on a Bruker S2 Ranger X-ray fluorescence spectrometer equipped with a Pd X-ray tube. About 5 g of finely ground sample was pressed into a pellet of 40 mm in diameter using a 40-ton press. The pellet was placed within a round holder, inserted in a shielded chamber and then measured.
Adsorption experiment
Batch adsorption experiments were performed in a group of 100 mL stoppered conical flasks with 50 mL BF solution; BF (molecular structure illustrated in Figure 1) belongs to triphenylmethane dyes with molecular formula C20H20ClN3 and molecular mass 337.85 Da. Firstly, a certain amount of adsorbent was added into BF solution. And then the mixture was agitated at 150 r/min on a constant temperature oscillator. After the adsorption, the solution was filtered by a 0.45 µm polypropylene syringe filter, the residual concentration of BF was measured using spectrophotometer at 542 nm. The adsorption capacity was calculated by equation (1)

Molecular structure of BF.jpg.
Results and discussion
Characteristics of samples
As shown in Figure 2, for Nat-Dia, there are four strong diffraction peaks at 2θ = 21.9°, 28.4°, 31.5°, and 36.1°, and they are assigned to the (101), (111), (102), and (200) planes of crystalline silica form (JCPDS No: 39-1425), which indicates that the main composition of diatomite is SiO2. In the case of Alk-Dia, these diffraction peaks still exist; however, their intensities, especially the (101) plane are obviously decreased in the order of Alk-Dia-60 > Alk-Dia-85 > Alk-Dia-115. These observations indicate that the structure of diatomite may be partially destroyed as a result of the desilication in the alkali solution. Furthermore, the destruction of diatomite becomes severer with increasing the activation temperature.

XRD patterns of Nat-Dia (a), Alk-Dia-60 (b), Alk-Dia-85 (c), and Alk-Dia-115 (d) .jpg.
In order to further testify the desilication of diatomite did occur in the alkali solution, XRF studies are performed and the results are shown in Table 1. The main ingredient of natural diatomite is SiO2 with a hint of Al2O3, Na2O, Fe2O3, K2O, and CaO. And the content of SiO2 for Alk-Dia is gradually decreased with increasing the temperature of the alkali activation. This indicates the silicon of diatomite is leached during the alkali activation, and the content of which is strongly dependent on the activation temperature. This is agreeable with XRD results.
The composition of natural and alkali-activated diatomite (wt. %).

To further assess the effect of alkali treatment on the chemical and structural properties of diatomite, FT-IR studies are performed and the results are shown in Figure 3. For the Nat-Dia, the characteristic absorption bands of Si–O in-plane stretching vibration (1090 cm−1), Si–O symmetric stretching vibration (794 cm−1), Si–O–Si bending vibration (472 cm−1) are clearly observed (Deng et al., 2019; Khraisheh et al., 2005; Yuan et al., 2013). In addition, the band at 625 cm−1 may be ascribed to the perpendicular vibration of Al–O (Zhao et al., 2015), which is impurity in diatomite. However, compared with spectrum of Nat-Dia, all of the Alk-Dia samples exhibit two new bands at about 3460 and 1645 cm−1, which are attributed to the –OH stretching and bending vibrations of molecular water (Zhao et al., 2015), furthermore, the intensities of them are increased with increasing the activation temperature, reflecting that the amount of water adsorbed by the Alk-Dia is gradually increased. This indicates that the surface properties of diatomite have changed during the alkali activation. In the case of Alk-Dia-115, the characteristic absorption band of Si–O in-plane stretching vibration (1090 cm−1) becomes wider, which demonstrates its structure is damaged in the 115°C alkali solution.

FT-IR spectra of Nat-Dia (a), Alk-Dia-60 (b), Alk-Dia-85 (c), and Alk-Dia-115 (d) .jpg.
To explore the impact of alkali activation on the morphologic of diatomite, the natural and alkali-activated diatomite are subjected to SEM observations, and the results are shown in Figure 4. The SEM image of natural diatomite in Figure 4(a) presents that the structure of diatomite is integral and likes a disc-shaped with relative smooth surface, and there are some impurities and debris surrounding it. For Alk-Dia-60, there is no obvious change in morphology; however, the surrounding impurities are significantly reduced. With further increasing the activation temperature, i.e., Alk-Dia-85, although, the general appearance of the disc-shaped structure is preserved, clumps of agglomerated particles and some cracks attached on disc-shaped diatomite are clearly observable, and its surface is extra rough, reflecting the partial destruction of the diatomite structure. For Alk-Dia-115, it looks to be melted to such an extent that the original disc-shaped of the natural diatomite can hardly be seen, and clumps of amorphousness-like agglomerates is appeared, which indicates the structure of diatomite is further destroyed. Thus, the effect of alkali activation on the diatomite structure can be clearly observed from the SEM images, which is consistent with the results of XRD and FT-IR.
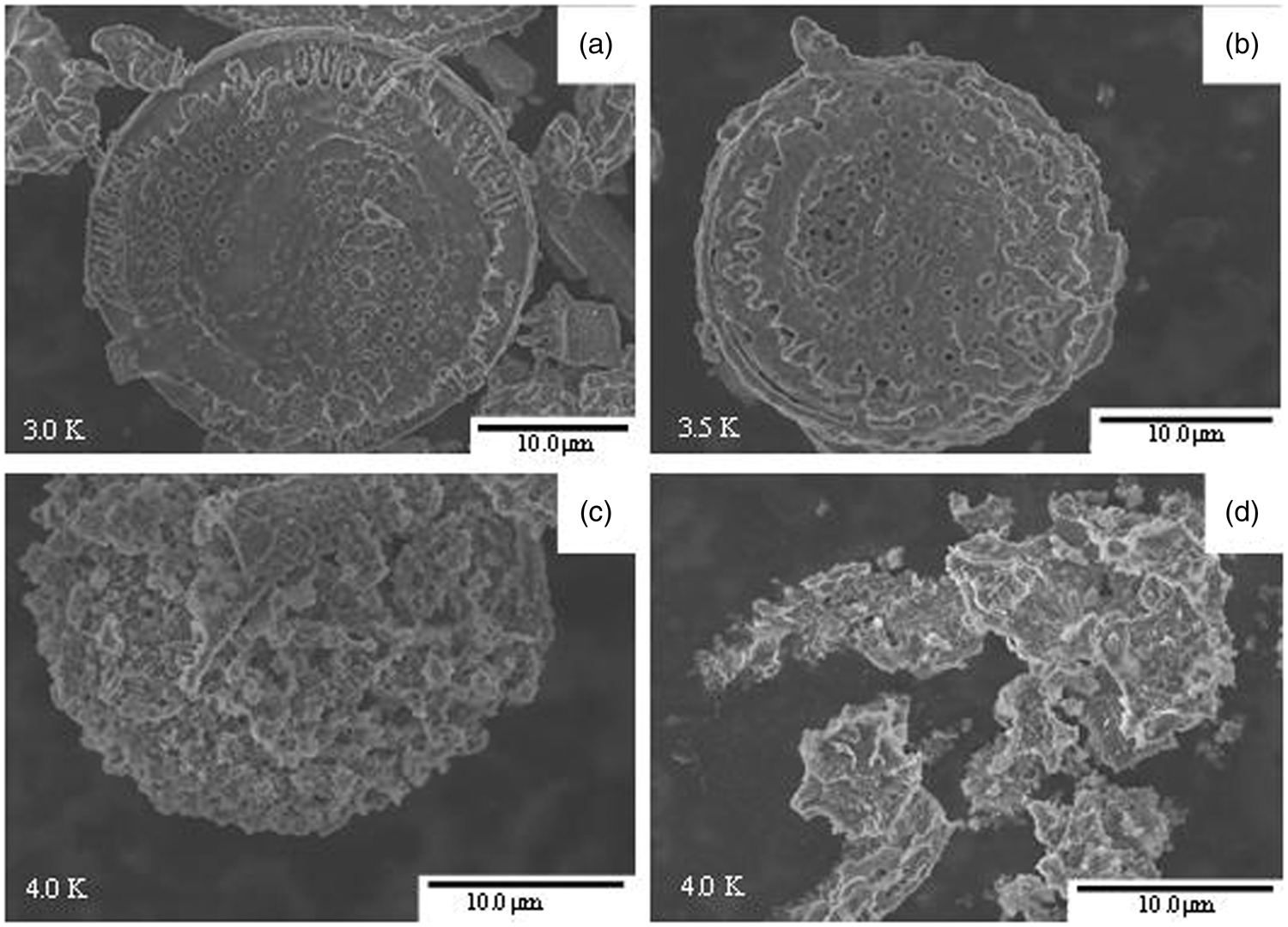
SEM images of Nat-Dia (a), Alk-Dia-60 (b), Alk-Dia-85 (c), and Alk-Dia-115 (d) .jpg.
The N2 adsorption–desorption isotherms of the Nat-Dia and Alk-Dia are presented in Figure 5. As indicated from the isotherm, for the Nat-Dia, the N2 uptake is very low, indicating its surface area is smaller. After diatomite is activated by alkali solution under different conditions, the distinctly enhanced N2 uptake is exhibited, especially in the higher-pressure region (p/p0 > 0.7), and the extent of which is strongly dependent on the activation temperature. This indicates the generation of a large number of pores due to silicon leaching in diatomite during alkali treatment. The content of silicon leaching is increased with temperature (Table 1), and then leading to increasing amount of pore. To quantitatively compare the porous properties of the Alk-Dia, the BET surface area and pore volume are calculated. As can be observed from the data shown in Table 2, the Nat-Dia presents very low BET surface area (1.9 m2⋅g−1) and small pore volume (0.006 m3⋅g−1). Compared with Nat-Dia, the BET surface area and pore volume of all the Alk-Dia are sharply increased; moreover, they are clearly increased with increasing the activation temperature. The Alk-Dia-115 exhibits the highest BET surface area (24.0 m2⋅g−1) and pore volume (0.181 m3⋅g−1). This can be reasonably ascribed to a large number of pores created during alkali treatment.

N2 adsorption–desorption isotherms of Nat-Dia (a), Alk-Dia-60 (b), Alk-Dia-85 (c), and Alk-Dia-115 (d) .jpg.
Summary of the textural properties of natural and alkali-activated diatomite.

BET: Brunauer–Emmett–Teller.
Adsorption performance of natural and alkali-activated diatomite
The adsorption performance of BF on Nat-Dia and Alk-Dia is presented in Figure 6. In comparison with Nat-Dia, the amount of adsorbed BF onto Alk-Dia is significantly increased. The adsorption capacity of Alk-Dia is increased with increasing the activation temperature. The Alk-Dia-115 exhibits the highest adsorption capacity (2.1 mg⋅g−1), which is much higher than that of Nat-Dia (0.6 mg⋅g−1). It is found that the adsorption capacity of Nat-Dia and Alk-Dia is obviously positive correlation with their surface by comparing Table 2 and Figure 5, which further proves that the larger surface area of adsorbents, the higher adsorption capacity. Therefore, the Alk-Dia, especially Alk-Dia-115 seems to be a more efficient material than natural diatomite for the BF removal.

The adsorption capacity of natural and alkali-activated diatomite.jpg.
Effect of contact time and adsorption kinetics
As shown in Figure 7, the adsorption capacity increases rapidly in initial adsorption stage before 20 min. As the contact time extends, it increases slowly. In the final adsorption stage after 30 min, it almost does not change with time. This implies that adsorption processes have reached steady state. At the beginning of the adsorption, all active sites on the diatomite are vacant and BF concentration is high, therefore, the adsorption driving force which originates from the concentration gradient of BF molecules is higher, then resulting in a fast adsorption rate. However, with the adsorption proceeding, the active sites on the diatomite surface are gradually consumed, furthermore, the BF concentration would be decreased, thus, the adsorption driving force is decreased, then resulting in a slight decrease in uptake rate. Finally, BF distribution of solid–liquid phase reaches equilibrium, and adsorption capacity is not increased.

Fit of adsorption kinetics of BF adsorption onto Alk-Dia-115.jpg.
Adsorption kinetics studies are very important to explain the adsorption mechanism and adsorption characteristic. In attempting to describe the adsorption kinetics of BF onto Alk-Dia-115, the adsorption capacity (qt) at various time (t) is fitted by two conventional kinetic models, i.e., pseudo-first-order (equation (2); Lagergren, 1898) and pseudo-second-order model (equation (3); Blanchard et al., 1984)
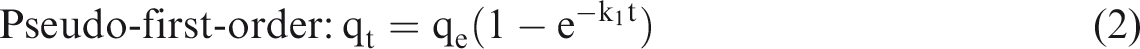

The kinetics parameters for the adsorption of BF on Alk-Dia-115 estimated via non-linear fitting analysis of the experiment data (Figure 7) using different kinetics models (i.e., equations (2) and (3)) with software Origin 8 are shown in Table 3. It is evident from this figure that the experimental points for BF adsorption by Alk-Dia-115 are preferably described by the pseudo-first-order model. This fact is further confirmed by the determination coefficient (R2) shown in Table 3, the R2 of the pseudo-first-order model is close to unity (0.9948), which is higher than that of the pseudo-second-order (0.8922). In addition, by the contrast analysis between the data in Table 3 and Figure 7, it can be found that the equilibrium adsorption capacity predicted by the pseudo-first-order model is closer to the experimental value, which further confirms that the adsorption process would meet pseudo-first-order kinetics.
Kinetic parameters and determination coefficients of BF adsorption onto Alk-Dia-115.

Impact of BF concentration and adsorption isotherms
The effects of initial BF concentration on the adsorption performance of Alk-Dia-115 are illustrated in Figure 8. The adsorption capacity of Alk-Dia-115 increases with increasing initial BF concentration. It is well known that the concentration gradient of BF molecules will determine the driving force for the adsorption of BF onto Alk-Dia-115. With increasing BF concentration in the aqueous phase, the increasing difference of concentration between the solid–liquid phases would enhance the interaction of BF with the Alk-Dia-115. Therefore, the amounts of BF absorbed by Alk-Dia-115 increase with increasing initial BF concentration. However, due to the limited adsorption sites of Alk-Dia-115, once it reaches saturation, no more BF molecules will be adsorbed, even under larger adsorption driving force. Therefore, the increase of adsorption capacity is slowed down at higher initial BF concentration, with further increasing the BF concentration, the absorption capacity changes very slowly due to nearly reaching adsorption saturation. This is similar to the results reported by Liu et al. (2019).

The effect of initial BF concentration on the adsorption capacity.jpg.
In order to understand the interaction between the adsorbate and adsorbent, the equilibrium adsorption data of BF (qe) at various residual BF concentrations (Ce) is fitted by the two-parameter Freundlich (equation (4); Freundlich, 1906) and the three-parameter Sips isotherm models (equation (5); Sips, 1948) with software Origin 8, and the fitting curves of the two isothermal models are presented in Figure 9. The isothermal adsorption parameters and R2 values for the adsorption of BF estimated via non-linear fitting analysis of the experiment data (Figure 9) are given in Table 4



The adsorption isotherms fit of BF adsorption onto Alk-Dia-115.jpg.
Parameters of Freundlich and Sips isotherms of BF adsorption onto Alk-Dia-115.

The results show that the adsorption equilibrium data are well fitted by the Sips isotherm model due to R2 value (0.9414) which is higher than that of Freundlich isotherm model (0.8129). In fact, the Sips isotherm model is a combination of the Freundlich and Langmuir isotherm models. In the case of ns = 1, the Sips isotherm model, i.e., equation (5) transforms into a Langmuir type equation. Alternatively, as either Ce or KS approaching 0, this isotherm becomes the Freundlich isotherm (Belhachemi and Addoun, 2011). As revealed from ns (0.506) in Table 4, it can be considered that the adsorption is intermediate between Langmuir and Freundlich isotherm. In additional, the Sips model exponent ns indicates surface heterogeneity, in the case of a highly heterogeneous system, the deviation of ns value from unity will be higher (Anirudhan et al., 2009). The value of ns (0.506) indicates that the Alk-Dia-115 surface is heterogeneous in nature and this surface heterogeneity would be another factor benefiting BF adsorption on Alk-Dia-115. The Sips model provided qmax (9.84 mg⋅g−1) is very similar to the experimental results.
Impact of adsorption temperature and adsorption thermodynamics
The influence of temperature on BF adsorption is carried out by varying the temperature in the range of 20–60°C under identical adsorption conditions, and the results are shown in Figure 10. The amount of adsorbed BF increases with temperature, which ascribes to the rapid rate of diffusion of molecules in high temperature and a large number of molecules to gain enough energy to engage an interaction with active sites on the surface of adsorbent (Dogan and Alkan, 2003).

The effect of temperature on the adsorption capacity.jpg.
To investigate whether the adsorption process of BF on Alk-Dia-115 is endothermic or exothermic, spontaneous or nonspontaneous, the thermodynamic analysis for BF adsorption is performed. The free energy change (△G0), enthalpy change (△H0), and entropy change (△S0) are calculated by equations (6) and (7) (Labied et al., 2018)



Linear plot of ln kd versus 1/T for the adsorption of BF on Alk-Dia-115 is presented in Figure 11. The values of ΔH0 and ΔS0 are obtained from slope and intercept of this plot, as shown in Table 5.

Fit of adsorption thermodynamics of BF onto Alk-Dia-115.jpg.
Parameters of thermodynamics for BF adsorption by Alk-Dia-115.

It is well known that the forward reaction is spontaneous when ΔG0 is negative, while the reverse reaction is spontaneous when ΔG0 is positive, and the system has reached an equilibrium state when ΔG0 is zero. For BF adsorption on Alk-Dia-115, the negative values of ΔG0 decrease from −1.39 to −3.40 kJ/mol with increasing temperature, which demonstrates the feasibility and spontaneity of adsorption process. Moreover, this phenomenon also suggests that the adsorption process is more favorable at higher temperature. The positive △H0 value suggests that the adsorption process is endothermic, and a proper enhancement of the temperature is helpful to the adsorption, which corresponds with the experimental data. In addition, the value of △H0 can determine whether adsorption is the physical adsorption or chemisorption. Generally, the △H0 value of absolute physical adsorption is less than 20 kJ mol−1, while that of chemisorption is in the range of 80–200 kJ mol−1 (Gu et al., 1994; Li et al., 2010). In this study, the value of △H0 is 16.81 kJ mol−1, implying that BF adsorption processes should be regarded as physical adsorption. The positive △S0 value demonstrates that the adsorption of BF on solid–liquid interface of Alk-Dia-115 is disordered and together with greater confusion.
Effect of initial pH on adsorption
The pH value of solution is an important parameter in adsorption process. The experiments for the effect of initial pH on the adsorption of BF are carried out and the results are shown in Figure 12. The amount of adsorbed BF increases with pH, and the higher adsorption capacity is obtained in alkaline condition. The change in solution pH would influence the surface charge of the adsorbent by protonation and deprotonation phenomenon (Pawar et al., 2018; Wu et al., 2009). At lower pH, the surface of adsorbent becomes positively charged due to protonation, therefore, the positive BF molecules and the adsorbent surface would cause repulsion, and then leading to poor adsorption of BF. Especially, when pH is lower pHPZC of the adsorbents (pHPZC= 5.5, which is determined according to the method in literature (Tran et al., 2016)), the adsorption capacity is dropped rapidly. Whereas, at higher pH, more negatively charged active sites at the adsorbent surface are presented due to deprotonate, and then resulting in the higher uptake of cationic BF. Moreover, at acidic pH, H+ ions would compete with cationic BF molecules for the adsorption onto the adsorbent surface. The suppression effect of H+ ions on the adsorption of BF would be decreased with increasing pH in solution, which also results in the amount of adsorbed BF increased with increasing pH.

Effect of initial pH on BF adsorption onto Alk-Dia-115.jpg.
Adsorption mechanism
The dye molecules would cover the surface and diffuse into the pores of Alk-Dia-115 by capillary force, which is reflected from decreasing the surface and pore size of spent Alk-Dia-115 (Table 2). Therefore, the Alk-Dia-115 with high specific surface area and pore volume would adsorb more dye molecules. The capillary force inside the mesopore facilitates the mass transfer of dyes, which enhances the total adsorption capacity. In additional, the electrostatic interaction existed between the BF cations and Si–O– from the dissociating of the Si–OH on the surface of Alk-Dia-115 would be also conducive to the adsorption. Thus, the BF adsorptions onto Alk-Dia-115 involve interplay between capillary force and electrostatic interaction.
Conclusion
The natural diatomite was treated with NaOH to prepare Alk-Dia with high specific surface and pore volume due to forming many pores derived from the destroyed disc shaped structure of diatomite during alkali activation. The Alk-Dia showed a higher adsorption potential of BF than natural diatomite. Furthermore, the adsorption capacity of Alk-Dia was increased with increasing the activation temperature, and the Alk-Dia-115 exhibited the maximum adsorption capacity. The adsorption kinetics of the BF on the Alk-Dia-115 can be well described by the pseudo-first-order model. Furthermore, the Sips isotherm model was used to fit the experimental data better than the Freundlich isotherm model for the adsorption behavior of BF on Alk-Dia-115. The thermodynamics indicated the adsorption process of BF on Alk-Dia-115 was an endothermic and spontaneous process. The BF adsorptions onto Alk-Dia-115 involved interplay between capillary force and electrostatic interaction. According to the results of this work, it demonstrated Alk-Dia derived from natural diatomite could be employed as cheap, efficient, and ecofriendly adsorbent and could be considered as an alternative to adsorbent for removing dyes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 21707009), the Doctor Startup Research Foundation of Liaoning Province (No. 20180540007), the Natural Science Foundation of Liaoning Province (No. 20180510057), the Doctor Startup Research Foundation of Bohai University (No. 0159bs009).

