Abstract
The removal mechanism of a microbial flocculant produced using a novel Paenibacillus sp. strain A9 (MBFA9) for two heavy metal ions, lead and zinc, from an aqueous solution is studied and compared. The removal rate of Pb(II) or Zn(II) by MBFA9 increased with an increase in the pH when the pH was less than 6.0. The removal rates were 91.26% for Pb(II) and 76.28% for Zn(II). The optimum condition was found at a pH of 5.5 and dosage of MBFA9 of 5% (v/v, 150 mg/L). When the initial concentration of metal ions was 100 mg/L, the removal capacity was 453.70 mg/g for Pb(II) and 398.48 mg/g for Zn(II). The capture process of a single metal ion of Pb(II) or Zn(II) by MBFA9 agreed with the Langmuir adsorption model with fitting coefficients of 0.9907 and 0.9408, respectively. Both the adsorption kinetics and adsorption equilibrium, usually described as adsorption isotherms, showed that this process of Pb(II) or Zn(II) capture by MBFA9 involved simple adsorption of a single molecular layer. The maximum reaction rate constant, K2, of MBFA9 in the captured Pb(II) decreased from 0.0497 to 0.0095, with an increase in the Zn(II) concentration under different Zn(II) concentrations (0, 20, and 40 mg/L). This indicated that the presence of Zn(II) competed with PB(II) for the binding sites of MBFA9 and led to a decrease in the removal rate. The process was controlled by chemical adsorption.
Introduction
The problem of heavy metal pollution due to increased industrial economic activity has aroused widespread societal concern in recent years. Human activities, such as paper, textile, plastic, ceramic, and cement manufacturing, mining, and electronic plating, produce a large amount of wastewater, thereby increasing the heavy metal concentration in natural water and deteriorating environmental quality (Shahid et al., 2015a, 2015b). Heavy metal ions have caused great harm to environmental and human health due to their high mobility in water and soil, as well as their ability to easily migrate and accumulate in the food chain (Akar et al., 2012; Bulut and Tez, 2007). The accumulation of lead in vital organs can cause poisoning, brain and kidney damage, anemia, and cancer (Ayranci and Duman, 2004; Duran et al., 2013). Acute zinc poisoning caused by water pollution can lead to gastrointestinal dysfunction. Because more than one heavy metal ion exists in wastewater, it is more important to remove two or more heavy metal ions simultaneously. The neutralization precipitation method is an approach that has been commonly used to treat heavy metal wastewater. This method is a simple low-cost process; however, it has serious disadvantages, such as low treatment efficiency and the production of a large amount of slag by-product (Gupta et al., 2006; Santos et al., 2015). The ferrite and vulcanization methods have also been used due to their treatment efficiencies, which are higher than those of the neutralization method. However, these two methods suffer from many drawbacks. For example, the operation of these methods is complex, and the vulcanization process easily produces hydrogen sulfide and other toxic gases. Furthermore, the ferrite reaction conditions require a large amount of control, and the processing efficiency of these methods typically does not meet the requirements of high treatment efficiency (Cheng et al., 2007). In addition, the application of biochemical and phytoremediation is poor and is limited by water quality and a longer treatment cycle. The sulfurization and evaporation concentration method produces a low volume of slag, has a low external drainage capacity, and can generate sulfuric acid as a by-product. However, the method is expensive, and it involves a long and complex process. The ion exchange method and the extraction method also have high operating costs, generate secondary pollution, and suffer from certain industrial limitations (Abdolali et al., 2014; Fu and Wang, 2011; Maturana et al., 2011; Montazer-Rahmati et al., 2011). Compared with the aforementioned methods, the removal of heavy metal ions using a biological flocculant has the advantages of low cost, simple operation, strong selectivity, and good treatment effect. Nharingo et al. found that Cactus Opuntia ficus indica (OFIP) produced high Pb(II) removal ratios from the Mukuvisi River. Under optimum conditions, the removal ratios of Pb, Zn, Cd, and Cu were 100%, 5.74%, 84.16%, and 93.02%, respectively (Nharingo et al., 2015). Diana et al. (2017) reported that “nopal” pectin was functional in coagulation–flocculation treatment to remove heavy metals under experimental conditions. Therefore, “nopal” pectin has been favored by researchers in recent years. The development of an inexpensive flocculant for wastewater treatment is an important topic in environmental science (Dundar et al., 2008).
Microbial flocculants (MBFs), such as natural biopolymer flocculants, have been widely used in sewage treatment, medicine, and food processing industry in recent years because they are highly efficient, non-toxic, and biodegradable. In addition, they generate no secondary pollution and come from a wide variety of sources (Gomma, 2012; Li et al., 2013; Liu et al., 2015). MBFs exhibit the ability to bind with heavy metal ions depending on their chemical composition and structure. Therefore, it is important to investigate possible applications of MBFs to remove heavy metal ions from wastewater. Li et al. (2013) found that MBFGA1 had a good removal effect on Pb(II), with the main mechanism being electrical neutralization and adsorption bridging. CaO and MBFGA1 were used to remove Ni(II) in water. The initial concentration of Ni(II) was 100 mg/L, and MBFGA1 and CaO at concentrations of 6.59 × 10−3% (w/w) and 1.3 × 10−2% (w/w), respectively, were added. The stirring time was 61.97 min. The optimization result of the response surface method resulted in a maximum adsorption efficiency and capacity of 99.35% and 225.16 mg/L, respectively (Zhou et al., 2017). Martínez-Quiroz et al. (2018) used the zeta potential of ChD and ChDA derivatives to evaluate the influence of pH on the surface charge. In particular, it was used to determine the amine protonation capacity. The 1:1 stoichiometry found for the complex using zeta potential measurements reinforced the proposal of the formation of supramolecular aggregates (Martínez-Quiroz M et al., 2018). Guo et al. (2018) reported that the zeta potential of PRGO-Fe3O4 was approximately five, so when the pH ranged from 5 to 6, PRGO-Fe3O4 would be negatively charged. Pb(II) is the dominant species of Pb(II), and the electrostatic attraction between the two facilitates adsorption, resulting in a dramatic enhancement in adsorption.
It is assumed that metal biosorption involves a physicochemical interaction between the metal ions and functional groups on the cell surface that is based on physical adsorption, ion exchange, complexation, and precipitation. Moreover, metal biosorption performance depends on external factors, such as pH, the other ions in the solution (which may be in competition), the organic material in the solution, and the temperature (Esparzasoto and Westerhoff, 2003; Ozdemir et al., 2005).
Strain A9 (Paenibacillus shenyangensis sp. nov.), which has a high-flocculating ability, was isolated from soil located under a peach tree located in Shenyang, PR China (Jiang et al., 2015). The primary composition of the microbial flocculant, A9 (MBFA9), produced by strain A9 is a polysaccharide. Information regarding MBFA9’s optimization of culture conditions, characteristic analysis, mechanism of flocculation, application in the wastewater industry (printing and dyeing, starch wastewater), and the removal mechanism for Pb(II) ions has been previously reported by this research team. Compared with other flocculants, MBFA9 has the greatest advantage due to the ability to use less dosage, a higher flocculation rate, and no need to add other coagulant aids, such as CaO and PFS (Deng et al., 1999a, 1999b, 2001a, 2001b).
To study the removal effect and mechanism of mixed ions in water using MBFA9 thoroughly, MBFA9 is prepared in a clean manner and is used to remove Pb(II) and Zn(II) from an aqueous solution. The thermodynamics of MBFA9 to capture Pb(II) and Zn(II) are analyzed using different pH levels and different kinetics to capture Pb(II) and Zn(II), in addition to using different dosages of MBFA9 and initial concentrations of metal ions. A kinetic and thermodynamic analysis of the Pb(II)-Zn(II) binary system is conducted to determine the competitive adsorption dynamics between Pb(II) and Zn(II). In addition, the mechanism that MBFA9 uses to capture Pb(II) and Zn(II) is investigated using field emission scanning electron microscopy (FSEM) (Zeiss ULTRA PLUS).
Materials and methods
Materials
The microbial flocculant-producing bacteria, Paenibacillus sp. strain A9, was isolated from peach tree cultivation soil located in the Liaoning Province, China. It was identified as a new strain of Paenibacillus sp., named Paenibacillus shenyangensis A9T (Jiang et al., 2015), and preserved in the Preservation Center of the Institute of Microbiology with Preservation No. CGMCC2040. The flocculability of the fermentation broth of A9 to a kaolin solution can reach 96% (Deng et al., 1999a).
The molecular weight of the polysaccharide flocculant, MBFA9, generated from A9 was 2.594 × 106 Da (Deng et al., 2001a). The MBFA9 used in this study was the fermentation broth of A9 (FBA9, polysaccharide content 3 g/L) cultured in a liquid fermentation medium for 48 h and then centrifuged at 8000 r/min to remove the bacteria. The culture medium formula was as follows: glucose 20.0 g/L, potassium dihydrogen phosphate 2.0 g/L, hydrogen phosphate two potassium 5.0 g/L, seven water magnesium sulfate 0.2 g/L, sodium chloride 0.1 g/L, urea 0.5 g/L, yeast extract 0.5 g/L, and pH = 7.0–7.5. The culture medium was then sterilized at high pressure at 115°C for 20 min. After inoculation, the temperature was 30°C, and the shaking speed was 150 r/min. The supernatant of the A9 fermentation broth was screened using liquid chromatography-mass spectrometry (LC/MS) (6530-UHD, Agilent, USA). The same volume of supernatant containing the same fermentation products was ensured by maintaining the same optical density (OD) (refer to Figure AM1 in Supplementary material for the polysaccharide content of A9 at different fermentation times).
Lead nitrate (Pb(NO3)2) and zinc sulfate (Zn(SO4)2) salts were used for the preparation of stock solutions. Other solutions were prepared by dilution using deionized water.
Characterization of MBFA9
The fermentation broth of A9 (FBA9) was diluted three times with distilled water and centrifuged for 15 min at a speed of 8000 r/min. After evaporation and concentration using a rotary evaporator, ethanol was precipitated three times, stored at 4°C for 24 h, and washed with 95% ethanol, and then MBFA9 was collected for Fourier transform infrared spectroscopy (FTIR) (Nicolet 380 Thermo Scientific), field-emission scanning electron microscope (FESEM) (Zeiss ULTRA PLUS), and energy dispersive spectrometer (EDS) microscope detection to observe the composition.
The zeta potentials of MBFA9, MBFA9-Pb(II), and MBFA9-Zn(II) were performed to evaluate the pH influence on the surface charge. The experiments were done for MBFA9, MBFA9-Pb(II), and MBFA9-Zn(II) within a pH range of 3–6 using 0.1 M NaOH and 0.1 M HCl to adjust the desired pH value.
Methodology of the adsorption experiments
To investigate the adsorption behavior in the solutions with different pH values, 5% (v/v) MBFA9 was added to 100 mL of a solution that contained 50 mg/L Pb(II) or Zn(II) and shaken for 30 min with 150 r/min at 30°C. The 1.0 mol/L HCl and NaOH solutions were used to adjust the desired solution pH values from 3.0 to 6.0. Finally, the supernatant liquid was used for the Pb(II) or Zn(II) concentration determination by using inductively coupled plasma atomic emission spectroscopy (ICP-AES) (ProdigyXP ICP, Leeman-Labs, USA). The effect of the MBFA9 dosage on the Pb(II) or Zn(II) capture ratio was studied by using MBFA9 with dosages ranging from 0.5% to 10.0% (V/V) in a 50 mg/L Pb(II) or Zn(II) solution (100 mL) with a pH value of 5.5 shaken at 150 r/min at 30°C for 30 min. The effect of the initial metal ions on the capture ratio was studied by using 5%(v/v) MBFA9 in 10, 20, 30, 50, 75, and 100 mg/L Pb(II) or Zn(II) solutions (100 mL) with pH values of 5.5 shaken at 150 r/min at 30°C for 30 min. The removal efficiency (Q%) and removal capacity (q, mg/g) were calculated as follows
Isotherms of adsorption
Isotherms of adsorption were performed at pH = 5.5 and 6.0 for Pb(II) and at pH = 5.5 for Zn(II). A given concentration of MBFA9(5%, v/v) was dropped into 50 mL of the metal solution at the appropriate pH at 30°C. The Pb(II) or Zn(II) concentration was varied between 10 and 100 mg/L. After 30 min contact time at a speed of 150 r/min in a shaking incubator, the solution was centrifuged for 10 min, and the residual metal concentration in the suspension was analyzed using ICP-AES (ProdigyXP ICP, Leeman-Labs, USA). A similar procedure was used to investigate metal adsorption in binary solutions.
Kinetics of adsorption
Kinetics of adsorption was performed at different MBFA9 concentrations (0.5%, 1.0%, 3.0%, 5.0%, and 10.0% (v/v)) with different initial metal concentrations ranging from 10 to 100 mg/L at 30°C. A given concentration of MBFA9 was dropped into a 50 mL metal solution at the appropriate pH. Samples were collected at different contact times (5, 10, 15, 20, 25, 30, and 35 min) and analyzed using ICP-AES (ProdigyXP ICP, Leeman-Labs, USA). The influence of different Zn(II)concentrations on the process of MBFA9 capture of Pb(II) in binary solutions was also determined.
Results and discussion
Characteristics of MBFA9
MBFA9 is stable in acid at high temperatures. According to the infrared spectrum analysis in Figure AM6(a) in Supplementary material, the main component of the flocculant is a macromolecular polysaccharide that consists of an hydroxyl (−OH), carboxyl (−COO−), and amine group (−NHCOCH3). This makes the flocculant have thermal stability, and the flocculation mechanism primarily consists of adsorption bridging (Li, 2016). The result of the zeta potential test indicated that the surface of MBFA9 belongs to the anionic flocculant, which has a negatively charged group (the zeta potential of MBFA9 was −9.84 mV at a pH of 5.5. Refer to Table AM1 in Supplementary material). The supernatant and the cell sample were screened and analyzed using the LC/MS method. The results indicated that the main metabolites in the process of cell growth and metabolism included many types of organic acids (leucinic acid, citric acid, oxoglutaric acid, succinic acid, malic acid, and others), various amino acids, carbohydrate substances, and other carbohydrates (Pang, 2016).
Effect of pH
pH affects the ionic states of functional groups on an adsorbent surface; therefore, it is expected that adsorption is pH dependent. Excessively high or low pH has an adverse impact on the metal ion removal ratio (Guo, 2015; Hayeeye et al., 2018). pH may affect adsorption in the following aspects: (1) the effect on the metal form, solubility, and chemical properties of metal ions, (2) biomass degradation, and (3) the activity of the active group, protonation, and deprotonation. Thus, the appropriate pH is an important preliminary step in the study of metal removal (Yipmantin et al., 2011). At various pH levels, metal ions exist in different forms. For example, the solution equilibrium and the complexation equilibrium of Pb(II) in a solution are as follows: Pb(II)+2(OH)−→Pb(OH)2(s)→Pb(II)+2(OH)−; Pb(II)+(OH)− = Pb(OH)+; Pb(II)+2(OH)− = Pb(OH)2; Pb(II)+3(OH)− = Pb(OH)3+. Therefore, under an alkaline condition, metal ions will precipitate, thus affecting the removal ratio (Guo and Yu, 2014). As a result, the range of pH for Pb ion removal is 3.0–6.0.
Figure 1(a) shows that with an increase in pH, the removal ratio of metal ions by MBFA9 increases. With a pH between 3.0 and 4.0, the removal ratio of Pb(II) or Zn(II) was low at 50–75%. Above pH 5, the removal ratio tended to stabilize at 85–90% and 70–78% for Pb(II) and Zn(II), respectively. These phenomena can be explained as follows. When the pH value is between 3.0 and 4.0, there are a large number of hydrogen ions in solution. Because MBFA9 is anionic flocculant, the neutralization of the electrical interaction of hydrated hydrogen ions competes with metal ions for the active sites of MBFA9, decreasing the chance of metal ions to bind to MBFA9 active sites, thereby reducing the removal ratio. With an increase in the pH value, the concentration of hydrated hydrogen decreases and the surface negative charge increases. Thus, it is not easy to combine with MBFA9 surface active sites, leading to greatly reduced competition between metal ions and active sites. Concurrently, the activity of the MBFA9 surface active group is enhanced, thereby promoting the adsorption of metal ions. As a result, the removal ratio increases gradually with increasing pH (Gupta and Bhattacharyya, 2006).

Removal ratios of metal ions and zeta by MBFA9 under different pHs. (a) Removal ratios of metal ions (b) Zeta potential under different pHs.
It is also important to evaluate the impact of adsorption on pH variation. In fact, the acidity and alkalinity of adsorbents often cause proton binding or proton release, resulting in a substantial change in the pH value. pH may vary with the concentration of metal ions, the number of adsorbents, and the ionic strength of the solution. In the process of adsorption, substantial changes in pH will greatly affect the comparison of adsorption properties, the interpretation of the adsorption mechanism, and the modeling of experimental data. Changes in the pH value of the MBFA9 solution before and after the capture of Pb(II) or Zn(II) are shown in Figure AM2 (Supplementary material).
The pH of the Pb(II) solution after adsorption was close to the first bisection line, and the dispersion of pH values was not more than 0.1 units. Compared with Pb(II), the pH change in the Zn(II) solution after adsorption was relatively significant, with an increase of approximately 0.2 units. In particular, when pH was greater than 5.5, the change was more obvious. Therefore, to investigate the kinetics and thermodynamics of MBFA9-captured metal ions, the pH was set to 5.5.
Zeta potential is an interfacial parameter related to the low solubility condition of dissolved metals when the isoelectric point is reached. There are inclusive precipitating agents that assist the formation of metal hydroxides by the zeta potential reduction of residual water (López-Maldonado et al., 2014). If particles have many negative or positive charges, meaning that the absolute value of the zeta potential is high, they will repel each other, and the whole system has high stability. In contrast, if the absolute value of the zeta potential is very low, they will attract each other, and the whole system will be unstable and easily produce precipitates. When the absolute value of the zeta potential is greater than 30 mV, the solution system is considered stable, and no precipitation occurs (Lam et al., 2016). When the zeta potential is 0 mV, the solution pH value is the isoelectric point of the colloidal particles, and the system is mostly unstable and easily produces precipitates.
The zeta potential was explored at different pH levels in the range of 3.0–6.0 in 50 mg/L of Pb(II) or Zn(II) (see Figure 1(b)). In Figure 1, the zeta potential increased with an increase in the pH up to 5.5. The point of zero charge occurred at pH = 5.5 (−0.23 mV), where the graph crosses the x-axis. Therefore, when the pH was 5.5, the system became nearly completely charge neutralized, and pH = 5.5 was identified as the optimum pH for the removal of Pb(II) and Zn(II) by MBFA9 from an aqueous solution.
Effect of the initial concentration of metal ions
Figure 2 shows that when the concentration of Pb(II) was in the range of 10–50 mg/L, with an increase in the initial concentration of Pb(II), the removal ratios increased from 55% to 92%. When the initial concentration of Pb(II) was higher than 50 mg/L, the removal ratios tended to stabilize at approximately 90%, whereas the adsorption capacity still gradually increased. When the initial concentration of Pb(II) was 100 mg/L, the adsorption capacity was as high as 453.70 mg/g. This behavior may have occurred because the binding site of MBFA9 and Pb(II) had not yet reached saturation, and there was a fixed binding site in the case of no saturation (Song, 2014).

Effects of different initial concentrations on the removal ratios and adsorption capacity. (a) Pb(II) (b) Zn(II).
The same trend occurred in the trapping process of Zn(II) by MBFA9. When the initial concentration of Zn(II) was 100 mg/L, the adsorption capacity was 398.48 mg/L. However, the removal ratio of MBFA9 to Pb(II) was greater than that of Zn(II).
Effect of different MBFA9 addition amounts
When the dosage of MBFA9 was 5%, the adsorption capacity reached a maximum of approximately 226.85 and 191.40 mg/g for Pb(II) and Zn(II), respectively, and the removal ratio reached a maximum value of 91.85% and 82.34% for Pb(II) and Zn(II), respectively (see Figure 3). When the mass concentration of metal ions in the solution was constant, with an increase in the MBFA9 dosage, the binding sites of MBFA9 increased. Therefore, more metal ions were captured in the water, and the removal ratio increased. However, the removal ratio did not increase as the flocculant dosage increased. If the concentration of metal ions was constant and the dosage of MBFA9 was too high, a large number of flocculants would wrap in the particle surface, thereby inducing particles to disperse. As a result, the adsorption capacity and the removal ratio might both decrease.

Effects of different MBFA9 addition amounts on the removal ratios and the adsorption capacity. (a) Pb(II) (b) Zn(II).
Effect of different components of MBFA9
Li observed that the FESEM characterizations of the MBFA9 and A9 fermentation broths that captured Pb(II) were different (Li, 2016). It was speculated that other fermented products of A9 were also involved in the capture of Pb(II). The main metabolites in the process of bacterial growth and metabolism also include various organic acids, such as citric acid, ketoparic acid, succinic acid, and malic acid. Therefore, the addition of separate organic acids would be able to verify whether other metabolites participate in the reaction.
Table 1 shows that four types of organic acids have a certain ability to remove Pb(II). The highest removal ratio of malic acid to Pb(II) is 48.85%, followed by ketopamyl diacid, citric acid, and succinic acid. The removal ratios of the four organic acids to Zn(II) are low, and the highest is only 12.78% by ketopamyl acid. In a binary system, the removal ratios reduced. In the process of the capture of Pb(II) by MBFA9, the organic acids produced by A9 also precipitate metal ions in the reaction, thereby improving the capture capacity to some extent. The removal ratio of MBFA9 to Pb(II) is higher than that of Zn(II) because Pb(II) combines more easily with organic acids produced by A9.
Removal ratios of Pb(II) or Zn(II) by different components of MBFA9.

Parameter values of the Langmuir and Freundlich models.

Adsorption isotherms
Single system
The adsorption isotherms of the capture of Pb(II) and Zn(II) by MBFA9 are shown in Figure AM3 (Supplementary material). And the parameter values of the Langmuir and Freundlich models of the capture of Pb(II) and Zn(II) by MBFA9 are shown in Table 2. These isotherms fit well to both the Freundlich and Langmuir equations. Based on previous studies, the Freundlich isotherm was developed as an empirical equation that is usually used to describe adsorption on heterogeneous surfaces, while the Langmuir isotherms involve monolayer adsorption on a set of adsorption sites with uniform energies (Lei et al., 2018). A better fit with the Langmuir equations is an indication that the process of MBFA9 capture of Pb(II) or Zn(II) uses a simple adsorption of a single molecular layer. MBFA9 shows a greater affinity for Pb(II), with a monolayer adsorption capacity of 189.8856 mg/g of Pb(II) (Q, mg/g) being larger than the 142.8112 mg/g of Zn(II). The radius of the hydration ion of Zn(II) hydration ion is 4.30, which is greater than that of Pb(II) (4.01). This observation can be likely explained by the fact that the adsorption process is related to the size of the hydrous ion of metal ions, and this is negatively correlated (Liu, 2010).
The Pb(II) adsorption isotherms under different pHs are shown in Figure AM4 (Supplementary material). Two adsorption isotherms are superimposed, and the small changes in pH do not affect the maximum adsorption capacity. When the initial pH is 5.5, the pH range is 5.5–5.7 after MBFA9 captures Pb(II). When the initial pH is 6.0, the pH range is 5.9–6.2 after MBFA9 captures Pb(II). The stability of the pH in solution after the MBFA9 capture of Pb(II) shows that the adsorption isotherm is meaningful.
Binary system
To further prove that the affinity of MBFA9 to Pb(II) is higher than that of Zn(II), it was tested in a solution containing 100 mg/L of Pb(II) and different concentrations of Zn(II) simultaneously. In the presence of Zn(II), the initial slope, that is the affinity of metal ions with adsorbents (Yipmantin et al., 2011), and the maximum adsorption capacity decrease simultaneously with a decrease in the concentration of Zn(II) (see Figure 4). Although the two metal ions exist at the same time and they will compete for the same adsorption site, the maximum amount of adsorption of MBFA9 to Pb(II) is still high. The extended Langmuir model has a higher fitting degree to a binary system compared with the Langmuir–Freundlich model. In the binary system, the maximum monolayer saturation adsorption of the two metal ions declines to some extent compared with a single system (see Table 3). It can be further explained that in the binary system of Pb(II)-Zn(II), metal ions have competitive adsorption on MBFA9 surface binding sites. With an increase in the competitive ion concentration, the maximum adsorption capacity of the target ion will further decrease.
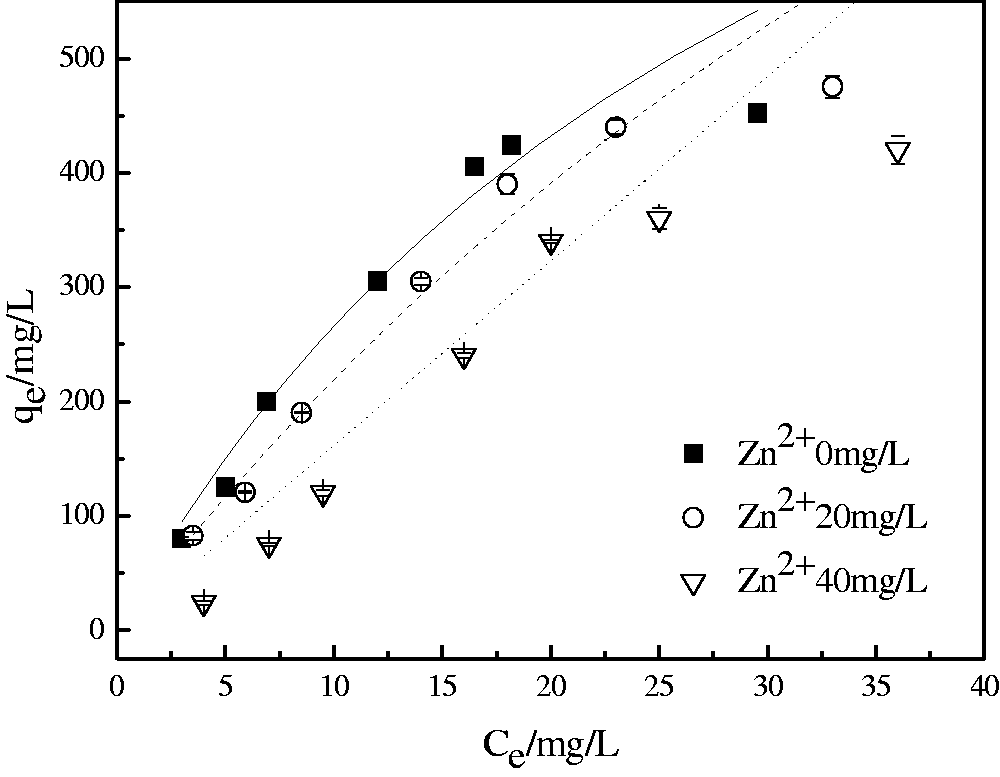
Adsorption isotherms of Pb(II) captured by MBFA9 under different concentrations of Zn(II).
Parameter values of the Langmuir models of Pb(II) captured by MBFA9 under different concentrations of Zn(II).

Uptake kinetics
Effect of the MBFA9 addition amount
A study of the adsorption kinetics is the basis for determining the selection and operation parameters of a device during the removal process, and it is the basis for the design of the adsorption process in large-scale practical industrial applications (Liu et al., 2009). Table 4 shows that the amount of MBFA9 added significantly affects the equilibrium concentration. The pseudo-first-order reaction rate equation determination coefficient of different dosages of MBFA9 capturing Pb(II) or Zn(II) is low. The determination coefficients of the pseudo-second-order kinetic equation are 0.9992–0.9999 and 0.9997–0.9998 for Pb(II) and Zn(II), respectively. In addition, the equilibrium adsorption capacity and test values are basically consistent, indicating that different dosages of MBFA9 capturing Pb(II) or Zn(II) are more consistent with the pseudo-second-order kinetic equation. In some cases, the variation in the adsorbent dosage influences the concentration gradient between the solution and the internal reactive groups. This influence may be caused by the strong adsorption of the solute on the surface of the external adsorbent, a decrease in the concentration gradient, and the effect of the driving force on the internal diffusion rate (Yipmantin et al., 2011). Ao reported that the adsorption kinetics of Pb(II)-IIP featured a particularly rapid initial step and then approached equilibrium within 16 min, and the experimental data fit well with the pseudo-second-order kinetic model. The saturation adsorption capacity (29.67 mg/g) is two times larger than Pb(II)-NIP, and the adsorption process obeys the Langmuir isotherm model (Ao and Guan, 2018).
Effect of the MBFA9 addition amount on the uptake kinetics data.

Effect of the initial metal concentration
Table 5 lists the parameters of the kinetic models pertaining to the MBFA9 capture of different initial concentration metal ions. According to the determination coefficient of the pseudo-second-order kinetic equation in Table 6, under different initial concentrations of the metal ions, the process of the MBFA9 capture of Pb(II) or Zn(II) is controlled by chemical adsorption. This result agrees with the results obtained from the thermodynamic analysis.
Effect of metal concentration on the uptake kinetics data using MBFA9.
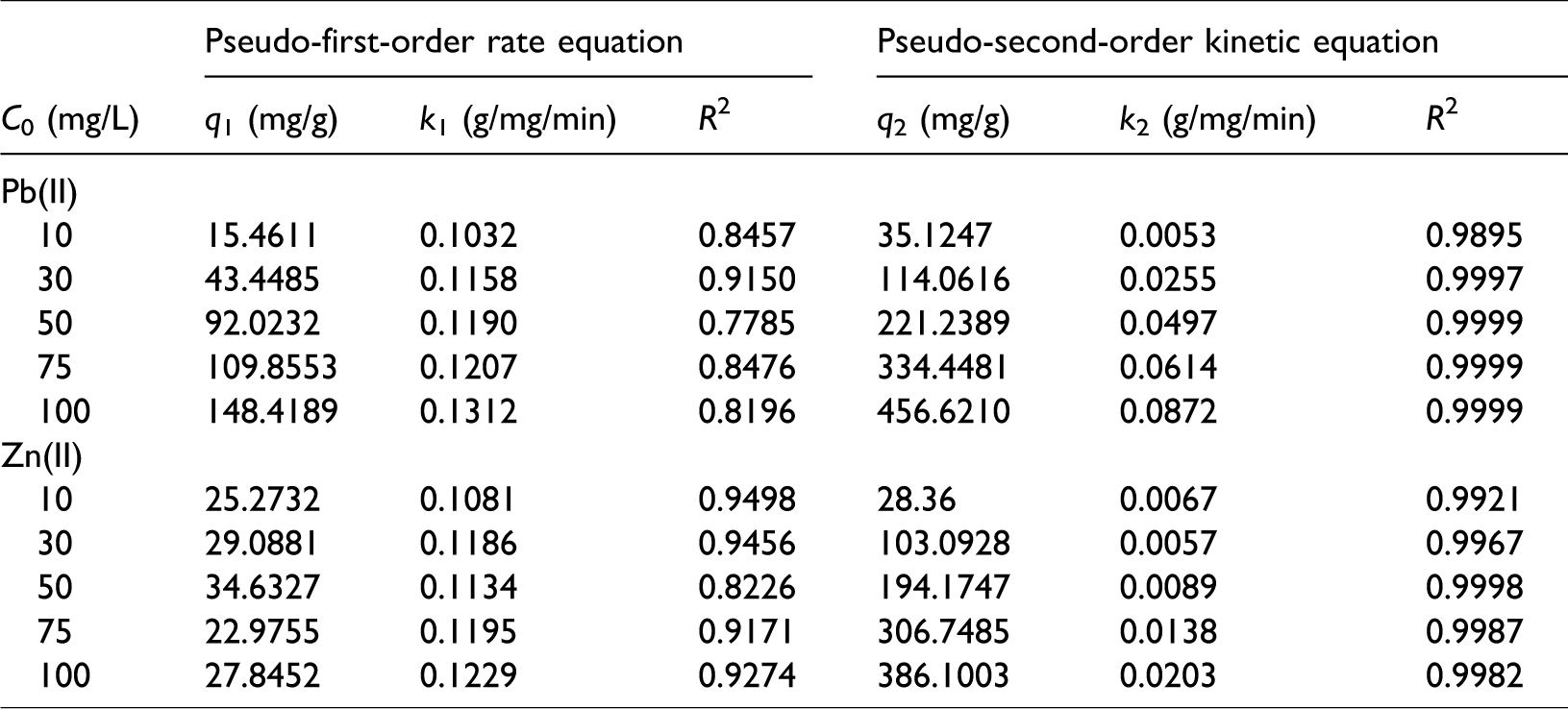
Effect of Zn(II) concentrations on the Pb(II) uptake kinetics data using MBFA9.

Effect of Zn(II) concentration
With an increase in the Zn(II) concentration, the value of k2 of the MBFA9 capture of Pb(II) decreases from 0.0497 to 0.0095. Because of ion exchange, Zn(II) competes for the binding sites of the MBFA9 with Pb(II), which can reduce the reaction rate (see Table 6). The MBFA9 capture of Pb(II) under different Zn(II) concentrations is more consistent with the pseudo-second-order kinetic equation, and the equilibrium adsorption amount is basically consistent with the experimental value. This result agrees with the results obtained from the thermodynamic analysis.
FESEM and EDS analysis
Three different morphologic structures were formed in the fermentation broth of the A9 flocculating bacteria. One was the crystallization of a large number of needle-like or rod-like particles after the capture of Pb(II). Another was a large number of massive crystals that formed after the capture of Zn(II), which could be speculated to be compounds formed by Pb(II) or Zn(II) and the metabolites (organic acids). The third petal or floc structures should be a chelate or complex formed by MBFA9 and Pb(II) or Zn(II) (see Figure 5(b) and (c)).

FESEM and EDS characterization of the capture of Pb(II) and Zn(II) by MBFA9. (a) Before the capture of Pb(II) and Zn(II); (b) After the capture of Pb(II); (c) After the capture of Zn(II).
Figure AM5 (Supplementary material) shows the FESEM characterization of the MBFA9 capture of Pb(II) and Zn(II) in the SingalA = AsB. Under this probe, the bright spot in Figure AM5(a) is Pb(II), whereas the MBFA9 capture of Zn(II) has no bright background in Figure AM5(b). It was speculated that Zn(II) was primarily in the form of oxide after the MBFA9 capture of Zn(II).
FTIR spectra analysis
To investigate the characteristics of the capture process and the mechanism, FTIR was employed to examine the interactions between Pb(II) and the functional groups of MBFA9. The characteristic IR absorption peaks of MBFA9 primarily occurred at 3423.81, 2933.34, 1652.78, 1529.60, 1396.64, 1238.10, and 1041.43 cm−1. After capturing Pb(II), that is MBFA9-Pb(II), many peaks, such as 3414.06, 2965.54, 1650.74, 1542.41, 1400.51, and 973.77 cm−1 obviously shifted with changes in wavenumber and intensity. This suggested that the aforementioned functional groups were primarily involved in the adsorption of Pb(II) onto MBFA9 (see Figure AM6 in Supplementary material). The chemical interactions between the Pb(II) and the O−H/N−H, C−H, C = O, C−N, and C−O groups occurred on the surface of MBFA9.
After MBFA9 captured Pb2+, the stretching vibration of O−H shifted 9.75 cm−1 to the right (from 3423.81 to 3414.06 cm−1), but the wave shape did not significantly change. This indicates that part of the hydroxyl groups participated in the capturing process, and this caused a partial breakup of the hydrogen bonds, resulting in a shift of the maximum peak of the stretching vibration of the hydroxyl groups. The stretching vibration of C−H from aliphatic CH2 shifted 32.2 cm−1 to the left (from 2933.34 to 2965.54 cm−1), that of C = O from COOH shifted 2.04 cm−1 (from 1652.78 to 1650.74 cm−1), and that of C−O shifted 67.66 cm1 to the right (from 1041.43 to 973.77 cm−1), with an obviously enhanced peak intensity. Such a shift could be attributed to the change of carboxyl anions in the counterbalancing cations. The stretching vibration of C−N shifted 3.87 cm−1 to the left (from 1396.64 to 1400.51 cm−1), that of N−H shifted 12.81 cm−1 to the left (from 1529.60 to 1542.41 cm−1), and the peak intensity weakened slightly.
There was a bridging effect among MBFA9 and Pb(II) or Zn (II), the functional groups of the MBFA9, and the involved hydroxyl (−OH), carboxyl (−COO−), and amine groups (−NHCOCH3). This agreed with what we reported in our previous studies (Jiang et al., 2014). There also was a coordination compound formed with Pb(II) or Zn (II) that played an important role in promoting the capture of metal ions. Moreover, there was a net scavenging effect between the flocs (Jiang et al., 2016).
Conclusion
In summary, MBFA9 was prepared with Paenibacillus shenyangensis A9T in a liquid fermentation medium, and the removal of Pb(II) was significantly higher than that of Zn(II) by MBFA9 from an aqueous solution. Moreover, Pb(II) not only formed more complexes with the flocculation polysaccharide MBFA9 but also displayed improved combination with the organic acids produced by the A9 flocculation bacteria. There was competitive adsorption between Pb(II) and Zn(II) in the Pb(II)-Zn(II) binary system. This process was more in agreement with the pseudo-second-order kinetic equation. Therefore, this process was controlled by chemical adsorption, which is consistent with the results obtained from the thermodynamic analysis. Based on the result of the FSEM and EDS characterizations, it can be concluded that MBFA9 had a great effect on the capture of Pb(II) or Zn(II) from aqueous solution. The mechanism primarily involved three aspects: metal ions and functional groups with negative charges on the surface of the MBFA9 can form coordination compounds, a bridging effect between MBFA9 and metal ions, and a net scavenging effect between the flocs.
Supplemental Material
Supplemental Material1 - Supplemental material for The effect of microflocculant MBFA9 and the mechanism of Pb(II) and Zn(II) removal from an aqueous solution
Supplemental material, Supplemental Material1 for The effect of microflocculant MBFA9 and the mechanism of Pb(II) and Zn(II) removal from an aqueous solution by Jiaqi Zhang, Binhui Jiang, Bo Zhang, Yu Li, Ping Fang and and Xiaomin Hu in Adsorption Science & Technology
Supplemental Material
Supplemental Material2 - Supplemental material for The effect of microflocculant MBFA9 and the mechanism of Pb(II) and Zn(II) removal from an aqueous solution
Supplemental material, Supplemental Material2 for The effect of microflocculant MBFA9 and the mechanism of Pb(II) and Zn(II) removal from an aqueous solution by Jiaqi Zhang, Binhui Jiang, Bo Zhang, Yu Li, Ping Fang and and Xiaomin Hu in Adsorption Science & Technology
Footnotes
Acknowledgment
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the National Key Research and Development Plan Subject for Monitoring, Early Warning and Prevention of Major Natural Disasters (2017YFC1503105) and the National Natural Science Foundation of China (51278090).
Supplemental Material
supplemental material is available online for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


