Abstract
In the present article, the optimization of removal of Cu2+, Ni2+, and Pb2+ using sugarcane-based activated carbon was studied. The activated carbon synthesized from low-cost sugarcane bagasse via ZnCl2 activation process was found to possess surprisingly high surface area (>1500 m2/g). In adsorption study, the individual and interactive effects of three critical parameters including initial concentration, pH of solution, and activated carbon dosage on removal efficiency of Cu2+, Ni2+, and Pb2+ were assessed by applying the response surface methodology combined with central composite design. The established second-order polynomial regression models provided an excellent interpretation of experimental data with acceptable coefficients of determination (
Keywords
Introduction
Heavy metals have been demonstrated to be harmful for both human and environmental health owing to their acute toxicity and persistence in nature. Although the presence of trace amounts of several metals is beneficial for biological transportation in micro-organisms, excessive intakes of heavy metals, such as copper, nickel, and lead, may cause serious health issues such as neurological damage, paralysis, blindness, and chromosome breakage (Wasi et al., 2013). According to the World Health Organization (WHO), the permitted concentration limits of copper, nickel, and lead in drinking water are 1.0, 0.02, and 0.05 mg/L, respectively (De Zuane, 2007). Thus, the removal of toxic metal ions from contaminated water attracts particular concern from the scientists and governmentalists all over the world.
Many methods have been attempted to eliminate heavy metals from aqueous solution, such as coagulation (Charerntanyarak, 1999), filtration with coagulation (Al-Abri et al., 2010), precipitation (Matlock et al., 2002), ozonation (Prieto-Rodríguez et al., 2013), adsorption, ion exchange (Da¸browski et al., 2004), reverse osmosis (Cath et al., 2006), and advanced oxidation (Barakat, 2011). Among those, the adsorption of heavy metals using activated carbon (AC) holds a number of important advantages, such as high adsorption capacity and efficient recovery of heavy metals, good selectivity, sludge free operation, and cost effectiveness (Charerntanyarak, 1999; Kadirvelu et al., 2001; Park et al., 2007). However, the fabrication of commercial activated from costly and non-renewable precursors such as petroleum residues, wood, coal, peat, and lignite carbon has restricted its applications (Marsh and Rodríguez-Reinoso, 2006). In recent years, the use of abundant agricultural wastes as a renewable and inexpensive alternative source for the synthesis of ACs has been developed (Ioannidou and Zabaniotou, 2007). The sugarcane (saccharum officinarum) is one of the most popular tropical species widely cultivated in tropical countries (Brazil, India, etc.) and makes up a large proportion of the sugar industries in the world. The extraction of each ton of milled sugarcane releases 180–280 kg of sugarcane bagasse (Farahani et al., 2011). Similar to other agricultural wastes, the low-cost sugarcane bagasse mainly consists of carbonaceous constituents, i.e. 42% cellulose, 25% hemicellulose, and 20% lignin, thus being a great resource for fabrication of ACs (Amin, 2008; Farahani et al., 2011). In recent years, there have been a number of reports on the fabrication of cheap and efficient powder and granular AC from sugarcane bagasse and molasse via different chemical activation methods. The resulting ACs possessed high surface area (>1000 m2/g) with well nanoporous structure as promising for many important applications ranging from wastewater treatment, water purification to electrical technology (G da C Gonçalves et al., 2016; SPC Gonçalves et al., 2016; Jain and Tripathi, 2015; Sreńscek-Nazzal et al., 2013; Tao et al., 2015).
ACs are typically fabricated from carbonaceous materials by oxidative reaction of carbon atoms using the pyrolysis and/or chemical activation (Ioannidou and Zabaniotou, 2007). Structurally, adsorptive properties of ACs are determined not only by inherent features of material sources but also by the production methods. Two major methods are frequently utilized to synthesize the ACs consisting of physical and chemical activation. Physical activation is conducted in the presence of CO2 or steam followed by gasification at high temperatures (700–900℃) (Sun and Jiang, 2010). In chemical activation, initial precursor or its char residue is soaked with strong dehydrating reagents, such as KOH and ZnCl2, which is subsequently pyrolysed to facilitate the formation of the porous ACs (Hayashi et al., 2000). Although dehydrating reagent ZnCl2 is found to be environmentally less advantageous than the other chemical activators, ZnCl2 is still used as a very common chemical in the preparation of ACs (Caturla et al., 1991; Olivares-Marín et al., 2006). The activation using ZnCl2 has been reported to offer the ACs with distinct characteristics like very high surface area (possibly up to 3000 m2/g) and enrichment of surface functional groups (Caturla et al., 1991). Owing to such advantages, the excellent performance of ZnCl2-ACs in the elimination of toxic pollutants such as dyes, heavy metal ions have been found (Baccar et al., 2009; Caturla et al., 1991; Hsu and Teng, 2000; Lo et al., 2012; Olivares-Marín et al., 2006).
The present study focuses on the developing of potentially cost-effective and efficient sugarcane bagasse-derived AC for the removal of heavy metal ions (Cu2+, Ni2+, and Pb2+) using response surface methodology (RSM) and central composite design (CCD) as a strong mathematical tool for optimization of the adsorption process. In the fabrication process, the chemical activator ZnCl2 was applied to form porous carbon structure as well as create and diverse functional groups on the surface. The removal tests were performed with three common heavy metals including Cu2+, Ni2+, and Pb2+ ions. The quadratic polynomial regression models involving three input variables, i.e. concentration of heavy metal ions, adsorbent dosage, pH, and taking adsorption capacity of the synthesize-AC as the main response were successfully developed. The goodness of fit of the model and the significance of model terms were assessed via the analysis of variance (ANOVA) and coefficient of regression (R2).
Experimental procedure
Fabrication of AC from sugarcane bagasse
Vietnamese sugarcane bagasse was collected and washed with distilled water for several times to remove all dust and residual sugar. Then, the precursor was dried under the sunshine and ground to the diameters of approximately 1.0 mm. The dried materials were soaked with a ZnCl2 solution (ZnCl2: precursor = 1.5:1 by wt.%) for 24 h. The ZnCl2-impregnated bagasse was heated under nitrogen at 500℃ for 1 h. Finally, the receiving ZnCl2-AC was repeatedly washed with deionized water until approaching a neutral solution and dried at 105℃ for 24 h.
Determination of pH point of zero charges (pHzpc)
To determine pHzpc of sugarcane bagasse-derived AC, the pH value of KCl (0.1 mM.L−1) solution was adjusted (pH = 2–12) using NaOH and HCl 1 M. First, 0.25 g of AC was added to flasks containing 50 mL of 0.1 ml.L−1 KCl. The solutions were kept stably and their final pH was measured by using a pH meter. The curve was plotted via final pH against the initial pH. The pH point of zero charges was calculated at which the curve crosses pH initial = pH final.
Batch adsorption experiments
Adsorption experiments were carried out in the Erlenmeyer flasks containing 50 mL of aqueous solution of 

Experimental design with RSM


Independent variables matrix and their encoded levels.

Instruments and techniques
The diffraction spectra were recorded with a scan rate of 0.02°/s. The angle range (2
Results and discussion
Characterization of the AC
The AC was fabricated from the sugarcane bagasse using ZnCl2 as the activating reagent following the protocol developed in our previous study. The detailed investigations about the effect of fabrication conditions as well as surface and structure properties have been already reported (Tran et al., 2016). Accordingly, the activation temperature of 500℃ was selected since it was able to provide the AC with high surface area and preferable surface chemistry. The typical characteristics of the synthesized AC are listed in Table 1. By measuring N2 adsorption/desorption isotherm at −196℃, the BET surface area of the AC was calculated to be 1502.0 m2/g with the average pore radius at 0.85 nm and the micro–pore volume of 0.886 cm3/g (Tran et al., 2016). In comparison with other studies (Kalderis et al., 2008; Suescún-Mathieu et al., 2014; Tsai et al., 2001), the surprisingly high surface area of 1500 m2/g, which was reported to be only attainable at the temperature as high as 850℃, can be obtained at the moderate activation temperature (500℃) using this fabrication protocol. Moreover, the average pore size (0.85 nm) appeared lower than those in other studies (1.70–2.03 nm) (Kalderis et al., 2008; Suescún-Mathieu et al., 2014; Tsai et al., 2001), proposing that high micropore volume and narrow micropores of this AC could possibly support high adsorption capacities (Caturla et al., 1991; Olivares-Marín et al., 2006; Tongpoothorn et al., 2011).
The SEM photograph in Figure 1 shows that the AC exposes a rough texture surface, where a variety of pore sizes are randomly distributed. The formation of porous and defect structure carbon can be explained due to the release of non-carbon elements such as hydrogen, oxygen, and nitrogen from the surface of char during pyrolysis process to form rigid carbon skeleton with rudimentary pore structure. The pH point of zero charge of the AC was determined to be 6.6 according to the results shown in Figure 2 and Table 2.
SEM micrograph of the AC. Measurement of pHzpc: the initial versus final pH plot. Properties of the AC.


Assessment of experimental results with Design–Expert

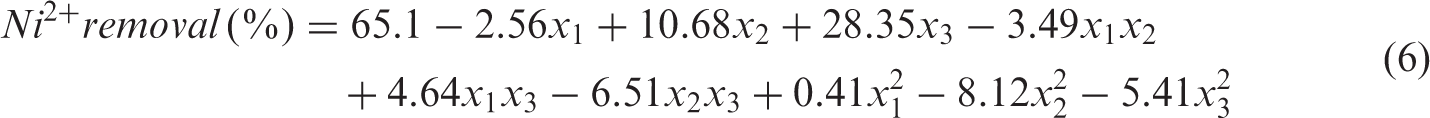

Matrix of observed and predicted values.

ANOVA for response surface quadratic models.

Note:
Significant at
Insignificant at

Experimental versus predicted plot of regression models for the removal of Cu2+ (a), Ni2+ (b), and Pb2+ (c).
Model confirmation.

Optimization of the removal efficiency of Cu2+, Ni2+, and Pb2+ using the RSM
The surface responses of the quadratic polynomial models were described using three-dimensional (3D) curve plots, in which one variable was maintained at the zero levels while the others fluctuated in the examined ranges. The effects of input variables (initial concentration, adsorbent dosage, and pH of solution) on the removal efficiency of metal ions (Cu2+, Ni2+, and Pb2+) can be observed in Figures 4–6 and Table 5. In detail, Figure 4 shows the obtained removal percentages while varying the two factors, initial concentration (892 mg/L) and adsorbent dosage (0.8–9.2 mg/L), at the constant value of pH 4.0. According to the observation in Figure 4(a) and (b), the effect of initial concentration on the removal efficiency of Cu2+ and Ni2+ was unremarkable while the adsorbent dosage plays a more controlling role. For example, the removal of metal ions increased to the maximum percentage removal of approximately 80% for Ni2+ and 50% for Cu2+ when increasing the AC dosage from 0.8 to 7.5 g/L and from 0.8 to 5 g/L, respectively. Further addition of adsorbent dosage resulted in drops of removal efficiencies. Both two variables including initial concentration and AC dosage affected strongly the removal of Pb2+ from aqueous solution. On the other hand, based on the results shown in Figure 6(c), up to 100% removal was obtained in the low initial concentration range (8–25 mg/L) with high values of adsorbent dosage (75–92 g/L).The former enhancement in adsorption can be explained due to the decrease of metal density in the solution while the latter likely resulted from the increased number of active sites as the adsorbent dosage increased.
3D plots of the removal of Cu2+ (a), Ni2+ (b), and Pb2+ (c): Effect of initial concentration and AC dosage at pH 4.
The effects of initial concentration (8–92 mg/L) and pH of the solution (0.6–7.4) on the removal efficiency for Cu2+, Ni2+, and Pb2+ ions at the adsorbent dosage of 5.0 g/L are illustrated in Figure 5. The removal of metal ions hardly occurred in strongly acidic solution (pH < 2) regardless of initial concentration whereas a neutral environment (pH = 5.5–7.4) facilitated the adsorption of Cu2+, Ni2+, and Pb2+ on the AC surface. Accordingly, the optimal pH values were found at 6.0, 6.8, and 6.5 for Cu2+, Ni2+, and Pb2+, respectively. The adsorption of metal ions tents to increase with increasing pH as resulted from a decrease in electrostatic repulsion between the cations and the positively charged surface of the AC. As pH decreased, the more competition between H+ ions in solution and metal ions at the surface adsorption sites caused a decrease in adsorption of metal ions. In this study, the optimal pH-values for the three metals were reasonably close to the pHzpc value (6.6).
3D plots of the removal of Cu2+ (a), Ni2+ (b), and Pb2+ (c): Effect of initial concentration, pH of the solution, and at the AC dosage of 5 g/L. 3D plots of the removal of Cu2+ (a), Ni2+ (b), and Pb2+ (c): Effect of AC dosage, pH of the solution, and at the initial concentration of 50 mg/L.

Note that effect of initial concentration in the optimal pH range was quite divergent. For instance, the highest removal percentage of Cu2+ (approximately 100%) was achieved at the concentration as high as 92 mg/L while that appeared at low concentrations (<24.8 mg/L) and in a wide intermediate concentration range (25–75 mg/L) for Ni2+ and Pb2+, respectively.
The effects of adsorbent dosage (0.8–9.2 g/L) and pH of the solution (0.6–7.4) on the removal efficiency of Cu2+, Ni2+, and Pb2+ ions at the constant initial concentration of 50 mg/L are shown in Figure 6. Again, the inefficient removal of metal ions at the low pH values (0.8–2.0) was observed in spite of the increase in AC dosage. The optimal pH value appeared to depend on the range. However, in the higher pH range (5.0–7.0), the change of pH value has more significant effect in the presence of low AC dosage; the larger amount of adsorbent led to better removal efficiency of metal ions at a higher value of pH (5.0–7.0).
According to the constructed model, the predicted maximum removal efficiencies follow the order: Ni2+ (65%) < Cu2+ (90%) < Pb2+ (99.9%). In verification experiments, the values obtained at optimum conditions were 66.4%, 90.0%, and 99.9% for Ni2+, Cu2+, and Pb2+, respectively, which approached the predicted values, indicating the suitability of the suggested models. In comparison, the order of adsorption capacity of the sugarcane bagasse-derived AC for the three metals: Ni2+ (2.99 mg/g) < Cu2+ (13.24 mg/g) < Pb2+ (19.3 mg/g) is a good agreement with the previous publications (Faur-Brasquet et al., 2002; Kadirvelu et al., 2000; Kongsuwan et al., 2009).
Conclusion
The results of this study show that the AC produced from sugarcane bagasse by the suggested ZnCl2-activation protocol is an efficient adsorbent for Cu2+, Ni2+, and Pb2+ ions. By applying RSM involving CCD, the influence of three independent variables, including metal ion concentration, pH, and AC dosage on the removal efficiency of Cu2+, Ni2+, and Pb2+ was interpreted via the quadratic regression equations with sufficient statistical significance. Accordingly, the maximum adsorption capacities and removal efficiencies for Cu2+, Ni2+, and Pb2+ were obtained, respectively, at a concentration of 75.0 mg/L, 22.5 mg/L, and 65.7 mg/L; AC dosage of 5.1 g/L, 5.0 g/L, and 3.4 g/L; and pH values of 6.0, 6.8, and 6.5. Based on the experimental and modeling data obtained in the present study, the potentially cost-effective sugarcane bagasse-derived ZnCl2-AC holds a great promise for application in the elimination of hazardous metal ions from industrial effluents.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by Foundation for Science and Technology Development Nguyen Tat Thanh University, Ho Chi Minh City, Vietnam.
