Abstract
Agricultural by-products have long hinder farmers, and subsequently, the food supply chain. Making use of their natural by-products will both reduce waste and increase industrial production. In particular, pineapple leaf fibers (PALF) can be extensively studied. Here, the curing kinetics of chemically modified PALF/epoxy resin crosslinked by an anhydride hardener was investigated by non-isothermal and isothermal methods with the differential scanning calorimetry technique. In this study, the Kissinger-Akahira-Sunose and Flynn-Wall-Ozawa methods, as well as Kamal's model, were employed to analyze the curing behavior of epoxy in non-isothermal and isothermal processes, respectively. The highest activation energies for pure epoxy and PALF/epoxy composite calculated differ when using the methods. Additionally, a decreasing trend in the activation energy values during the late stages of epoxy curing was observed. The results from Kamal's model indicate that the k1 values of the PALF/epoxy composite are only greater than those of pure epoxy at 100°C and 110°C. However, all the k2 values of PALF/epoxy are greater than those of pure epoxy. Additionally, the m value of the PALF/epoxy composite is lower than that of pure epoxy only at 100°C, while the n and m+n values of the PALF/epoxy composite are all greater than those of pure epoxy. Moreover, the results reveal that the Cure Index of the PALF/epoxy composite was larger than ΔH* and smaller than ΔT*. With PALF, it was found that the epoxy resin’s curing rate was increased and the activation energy was reduced. Meanwhile, the degree of crosslinks was less than that of the virgin resin. It is speculated that the hydroxyl groups on the plant fibers and the amine groups on the coupling agent-modified fibers can promote the cross-linking reaction. However, the curing reaction of the composite is affected by steric obstacles and high viscosity resulting from the addition of PALFs.
Introduction
The global population has exceeded eight billion, raising concerns about issues such as natural resource depletion, water scarcity, and food insufficiency. Furthermore, the substantial volume of waste generated on a global scale demands urgent attention. 1 Towards the global goal of sustainability, industries and agriculture have been striving to build upon reasonable economic growth, innovation, limited consumption and production. This originates from the increasing awareness toward global warming from industrial processes. Recognizing the inherent nature of sustainable environmental issues associated with waste and acknowledging the potential of natural plant resources for producing natural fibers, the research on natural fiber-reinforced composites has received significant attention. 2
As such, the global initiatives of using natural resources more effectively have increased dramatically. Natural fibers as reinforcement in plastics are nowadays an eco-friendly and economical substitute for synthetic fibers. The advantages of natural fibers over man-made fibers include low density, low cost, recyclability and biodegradability. Many researchers have investigated the strengthening effects of the natural fiber reinforced epoxy and various polymer composites.3,4 A variety of fruits such as bananas, watermelons, papayas, mangoes, and pineapples are greatly valued for their taste and nutritional benefits. However, over 40% of the fruit's total mass, including the peel, leaves, and seeds, is inedible and predominantly contributes to substantial waste production. Many of these discarded materials contain a notable amount of cellulose fibers. 5 Natural fibers are emerging as suitable alternative materials to glass fibers for reinforcing plastic polymers in various applications. Their ease of processing, which requires minimal energy consumption, combined with the growing demand for energy-efficient and lightweight solutions, gives natural fibers a distinct advantage over synthetic fibers. Various kinds of natural fibers, such as groundnut shell powder, 6 pineapple leaf fibers (PALF), sisal fibers, 7 and kenaf fibers, 8 have been employed in the fabrication of polymer composites.
Among the types, fibers from pineapple leaves have gathered extensive studies. Pineapple (
The fibers can be applied into matrices such as epoxy resins. Epoxy resins are thermosetting polymers with low volume shrinkage, dimensional stability, good adhesion, high strength, corrosion resistance, electrical insulation and other excellent properties. It has been widely used in industrial fields. Among the different types of epoxy resins, bisphenol-F epoxy resin has the advantages of low viscosity and good fluidity. Therefore, it can be used without addition of a reactive diluent or solvent, and its thermal stability and chemical resistance will not be affected. 12
It should be noted that the poor compatibility of natural fiber with polymer matrix was an issue for its application. The effect is due to the hydrophilic nature of the fibers. To overcome this, a surface treatment of such fibers is an essential step to improve their interfacial adhesion with polymer matrices. Such chemical processes have been applied to improve the properties of natural fibers. Among them, alkali and silane treatments are largely employed due to their effective ability to improve the interfacial compatibility.13,14 Overcoming the issue has led to the success of PALF composite fabrication. Continuous studies have focused on the improvements in strengths and the dynamics of the polymer matrices. Some have found as much as 58% improvement in tensile strength, depending on the percentage composition. 15
While PALF promises sufficient strength improvement of the products, it is necessary to investigate how the industrial process of fabricating such polymer is changed. There are, however, few literatures that discussed the effect of plant fiber on the reaction kinetics of epoxy resin.16,17 Bessa et al. investigated the effects of raw and treated Arundo donax L. cellulosic fibers on the curing kinetics of bisphenol A-based benzoxazine. The investigation of the curing kinetics of composites was conducted using the differential scanning calorimetry (DSC) technique under non-isothermal conditions and isoconversional integral kinetic methods. A decrease in the heat of curing, as well as the activation energy (Ea), was observed. The Avrami-Erofeev autocatalytic kinetic model was found to be the most suitable for describing the curing behavior of the composites. 16 The study by Libera et al. assessed the impact of in nature, KOH or NaOH treated curauá fibers on the cure kinetics using the non-isothermal method. They discovered that the activation energy was higher for treated fibers than in nature. 17
In this study, it is of interest to investigate the effect of PALF on the curing process. The study here begins with raw PALF. The material is then alkali- and silane-treated before incorporating into a bisphenol-F epoxy resin as a fabrication of a PALF reinforced epoxy composite. The treated PALF content was 20 wt%, exhibiting excellent mechanical and thermal properties. 18 In order to provide a basis of curing process theory for applications of the PALF/epoxy resin composite, we investigate the curing kinetics of this system. DSC is a widely used and effective method to study the behavior of epoxy curing, including isothermal and non-isothermal methods. The recommendations for thermal polymerization processes have already been developed by the Kinetics Committee of the International Confederation for Thermal Analysis and Calorimetry (ICTAC). Among these recommendations, the Kissinger-Akahira-Sunose (KAS) and Flynn-Wall-Ozawa (FWO) methods are commonly used for isoconversional analysis. In addition, the Kamal model was particularly recommended because it can represent the mechanism of competition between a regular and autocatalytic reaction. 19 This type of model can be used to simulate curing kinetic curves of much more diverse shapes. Indeed, it can describe a wide range of curing systems, including epoxy-amine and epoxy-anhydride systems, among others. Therefore, in this study, for the non-isothermal analysis, FWO 20 and KAS 21 methods were used to predict the curing behavior of the epoxy resin and its composite. For isothermal method, the Kamal’s model 22 was applied to analyze the curing kinetics of the system. The changes induced by the PALF on the curing kinetics are discussed.
Materials and methods
The following outlines the preparation of the composite. The setup to understand the effect of the fibers on the curing of the resin under thermal analysis is also presented.
Materials
This study proceeds to the fabrication of composite samples for their calorimetric measurements of the kinetic parameters. The PALF were prepared and added to the epoxy resin. Here, the epoxy resin (Bisphenol F-epoxy resin, ML3564) with epoxy equivalent of 165–175
Modification of pineapple leaf fibers and preparation of composite
The raw PALF obtained were first treated in an alkaline solution. 15 They were then washed with distilled water and dried in an oven at 60°C to preserve their compositions. Fibers were obtained by having the leaves chopped and screened to obtain an average length between 2 and 4 mm, and were subsequently treated by 3-aminopropyltri-ethoxysilane to obtain the modified PALF (MAF). The epoxy, the curing agent, and the accelerator were mixed with a ratio of 100:90:2. The MAF (20 wt%) were mixed to obtain the modified fiber-reinforced epoxy composites (MAF/EP).
Differential scanning calorimetry
Once the composite was prepared, the curing behavior was analyzed by a DSC (TA Instruments Q20). The thermoanalytical equipment is used to measure the specific heat of the sample as a function of temperature. The equipment model has an accuracy of ±0.1°C and a precision of ±0.05°C. To measure the non-isothermal kinetics, a sample mass of 5∼10 mg was sealed in an aluminum pan. It was then subjected to a heating program from 30°C to 250°C at different heating rates of 5, 10, 15 and 20°C/min under a constant nitrogen atmosphere. The isothermal curing was conducted at 100, 110, 120, and 130°C, and for 120 min each. A sample of pure epoxy resin, and a sample of MAF/EP were prepared for the DSC for comparison.
Kinetic analysis
The study analyses the samples via both the isothermal and non-isothermal methods. A related derivation is presented, and is related to the degree of curing reaction (or conversion), of which is measured by α. It is calculated by using the change in heat of the reaction.
Non-isothermal cure kinetics
Extensive non-isothermal kinetics analysis has a complexity of derivations and models.
21
Therefore, a simplifying assumption involves the description of the reaction rate as a product of two functions, and is expressed as:
Expressing equation (2) in integral form gives the total reaction over time, i.e.,
Where
Kissinger method
The investigation calls upon the Kissinger method. The method is a simple way to calculate activation energy that is based on the assumption of constant conversion (independent heating rates) when the reaction rate reaches maximum. It therefore involves the measurement of the peak temperature (Tp) at various heating rates (β). The integral form of the Kissinger equation is shown below.
21
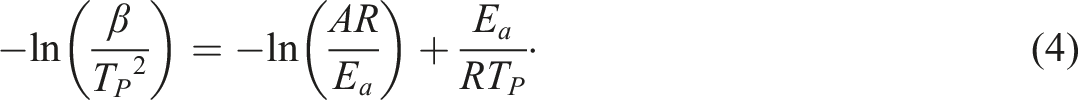
Ozawa method
Meanwhile, Ozawa’s formulation
21
found that the activation energy of a reaction is related to the heating rate, β, and the peak temperature of the reaction, 
The activation energy (
The KAS and the FWO methods
In this study, we also determine the kinetic parameters for various conversion using the KAS and the FWO methods. The equation for fitting the KAS model is provided below:
The equation for fitting the FWO model is as follows:
The kinetic study was conducted using equations (6) and (7) from the KAS and FWO methods, respectively. 23 To calculate the activation energy of pure epoxy and MAF/EP composite using the KAS method, a plot was generated between ln (β/T2) and 1/T, and the slope was determined for various conversions (α). Activation energies were computed for each specific conversion (α), ranging from 0.2 to 0.9, based on the obtained linear segments. Similarly, for the FWO method, a graph was plotted between ln (β) and (1/T). Linear segments were achieved for each conversion, and by utilizing the slope of these linear segments, activation energies were computed.
Isothermal cure kinetics
For the cure kinetics based on the conversion and not 
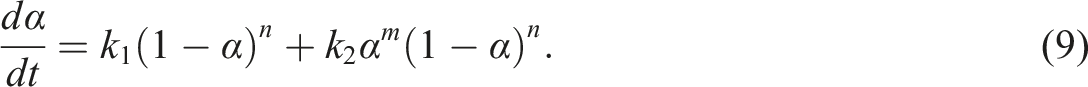
The first term on the right side of equation (9) is the nth-order reaction model and the second one is the autocatalytic reaction model. According to the nth-order kinetics model, the maximum reaction rate will be observed at t = 0, and, according to the autocatalytic model, the reaction rate is zero initially and attains a maximum value at some intermediate conversion. It is worth noting that, equation (9) enables the superposition of the two models. And for this property, when natural logarithm is applied to both sides of equation (8), the following can be derived: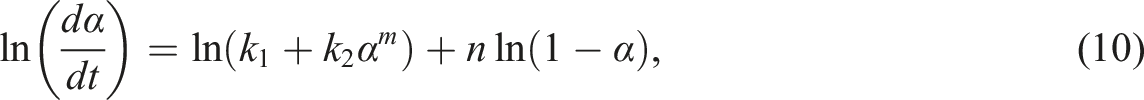
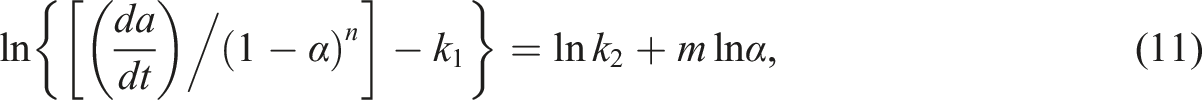
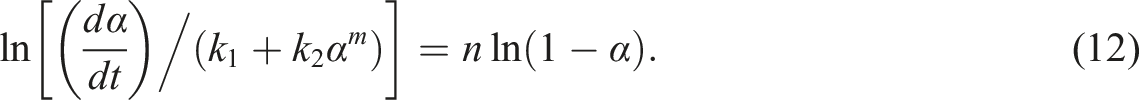
Cure index
With an understanding of the curing behavior and kinetics of thermoset composites, Jouyandeh et al.
24
introduced the dimensionless “Cure Index” (CT) based on nonisothermal DSC experimental data as follows:



In order to prevent the impact of filler aggregation on crosslinking reactions, an extremely low filler concentration can be employed, typically less than 0.2 wt% relative to the total weight of the resin/hardener mixture. Consequently, Jouyandeh et al. can straightforwardly classify the curing state of a thermoset composite compared to the reference sample into three possible cases indicated by the CI, as outlined below: I. CI >ΔH* II. ΔT*< CI <ΔH* III. CI <ΔT*
For this case: III (a): CI < ΔT* for Tp* < 1 III (b): CI < ΔT* for Tp* > 1

In equation (17), the variables Tp,c and Tp,ref represent the exothermic peak temperatures of the polymer with the additive and the pristine polymer, respectively. These values can be easily extracted from their respective DSC thermograms.
Results and discussion
Non-isothermal kinetics analysis
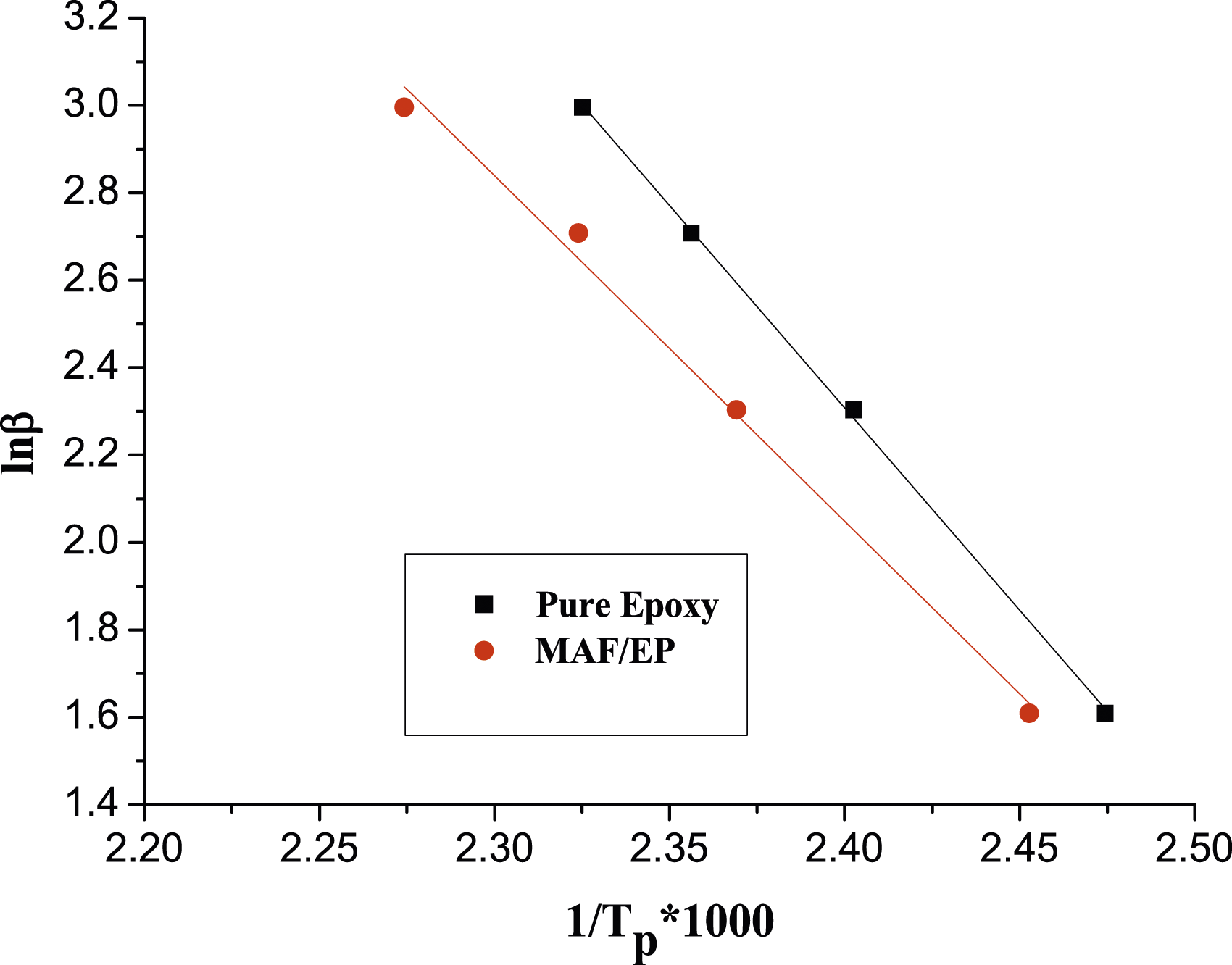
The DSC thermograms of pure epoxy and the MAF/EP composite at various heating rates are illustrated as Figures 1 and 2. The results from the DSC were recorded for both the pure epoxy sample, and for the MAF/EP. Different heating rates were used to calculate the reaction activation energies of pure epoxy resin and MAF/EP composite through Kissinger and Ozawa methods. The analysis diagrams of pure epoxy resin and MAF/EP composite drawn in Kissinger and Ozawa models are shown in Figures 3 and 4, respectively. It can be seen that the curves are shifted almost uniformly, indicating that the effect of PALF was little related to DSC thermogram of pure epoxy. DSC thermogram of MAF/EP composite. Kissinger model of MAF/EP composite compared with pure epoxy. Ozawa model of MAF/EP composite compared with pure epoxy.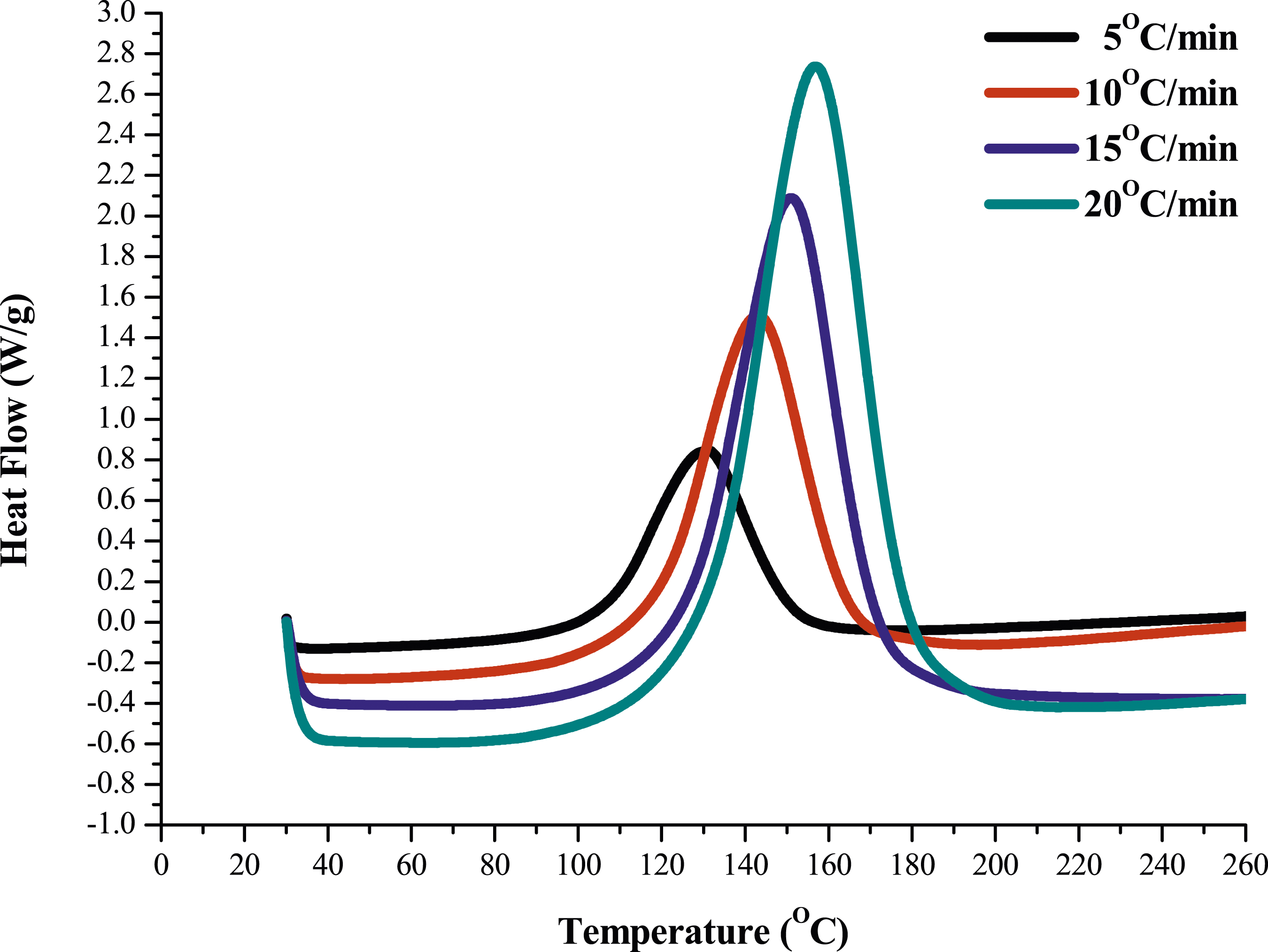
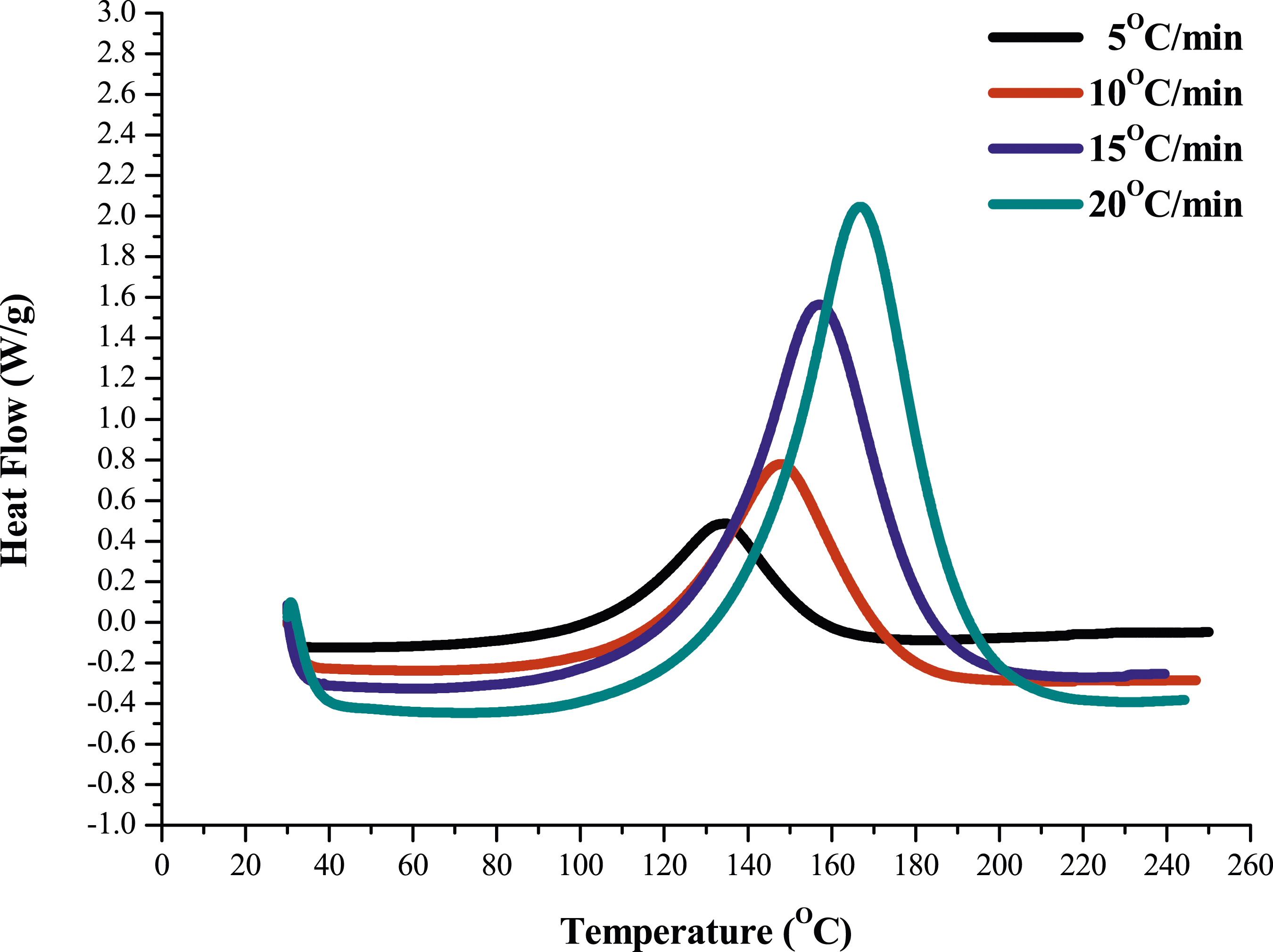
Moreover, the activation energy of pure epoxy and MAF/EP composite calculated using the KAS method, a plot of ln (β/T2) versus 1/T, for various conversions (α) is depicted in Figure 5(a) and (b). Activation energies were computed for each specific conversion (α), ranging from 0.2 to 0.9, based on the obtained linear segments. The calculated activation energies are presented in Table 1. Similarly, for the FWO method, a graph was plotted between ln (β) and (1/T) as illustrated in Figure 6(a) and (b) and activation energies were computed and outlined in Table 1. Plot of ln (β/T2) versus 1/T of (a) Pure epoxy (b) MAF/EP composite for various conversions (α) using the KAS method. Slope and activation energies calculated using KAS and FWO methods for pure epoxy and MAF/EP composite. Plot of ln(β) versus 1/T of (a) Pure epoxy (b) MAF/EP composite for various conversions (α) using the FWO method.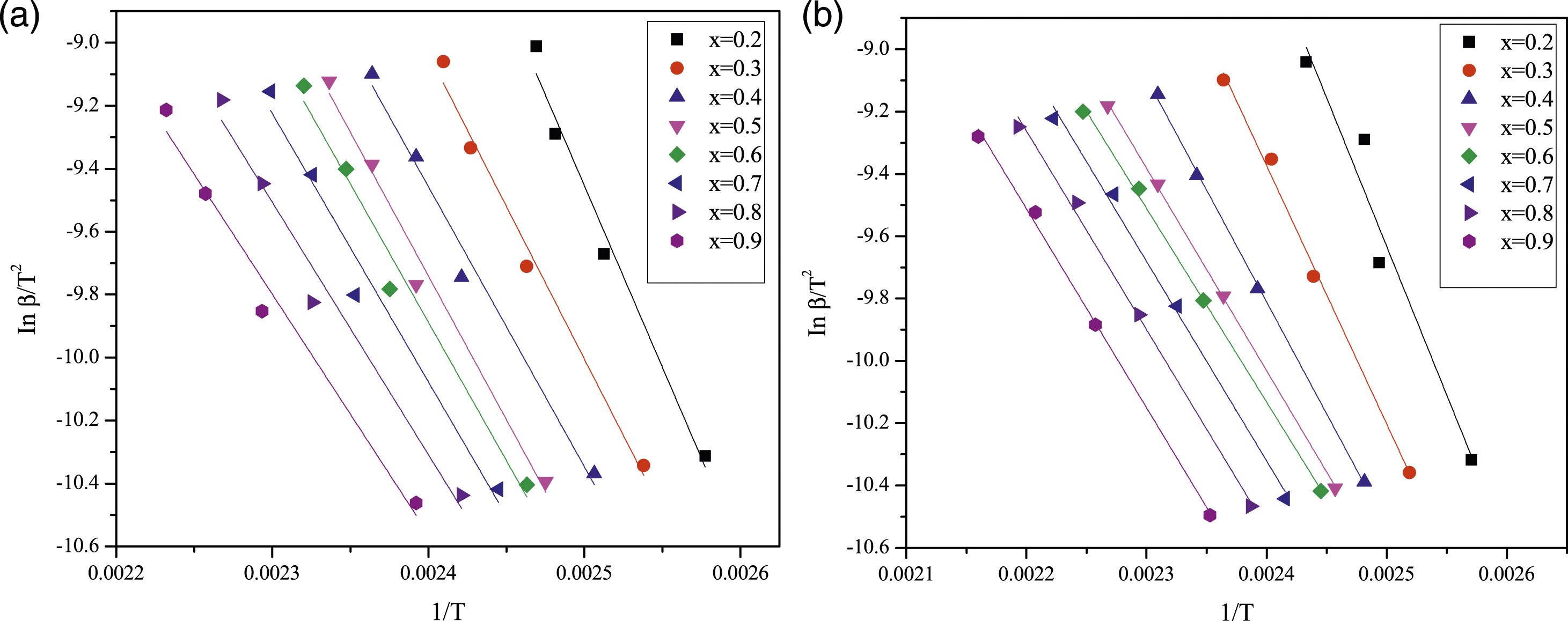
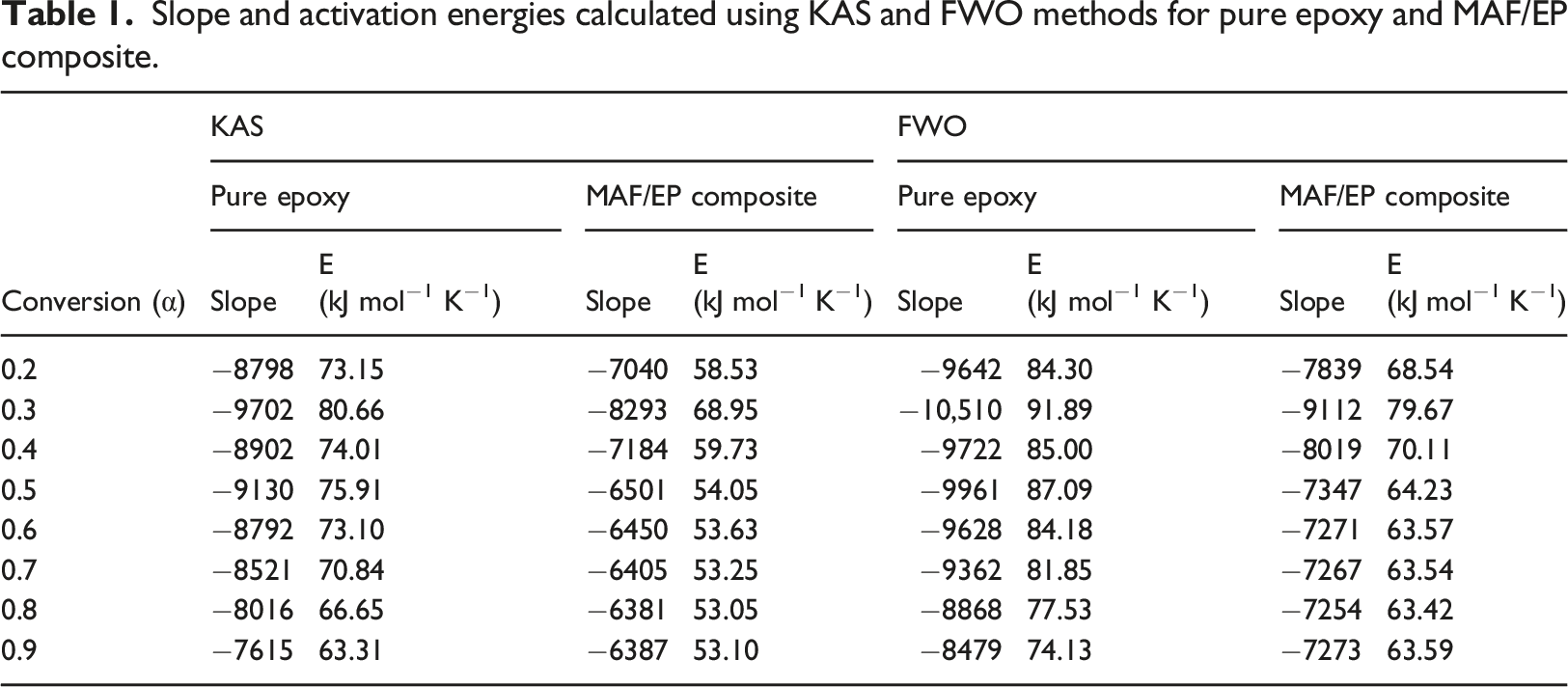
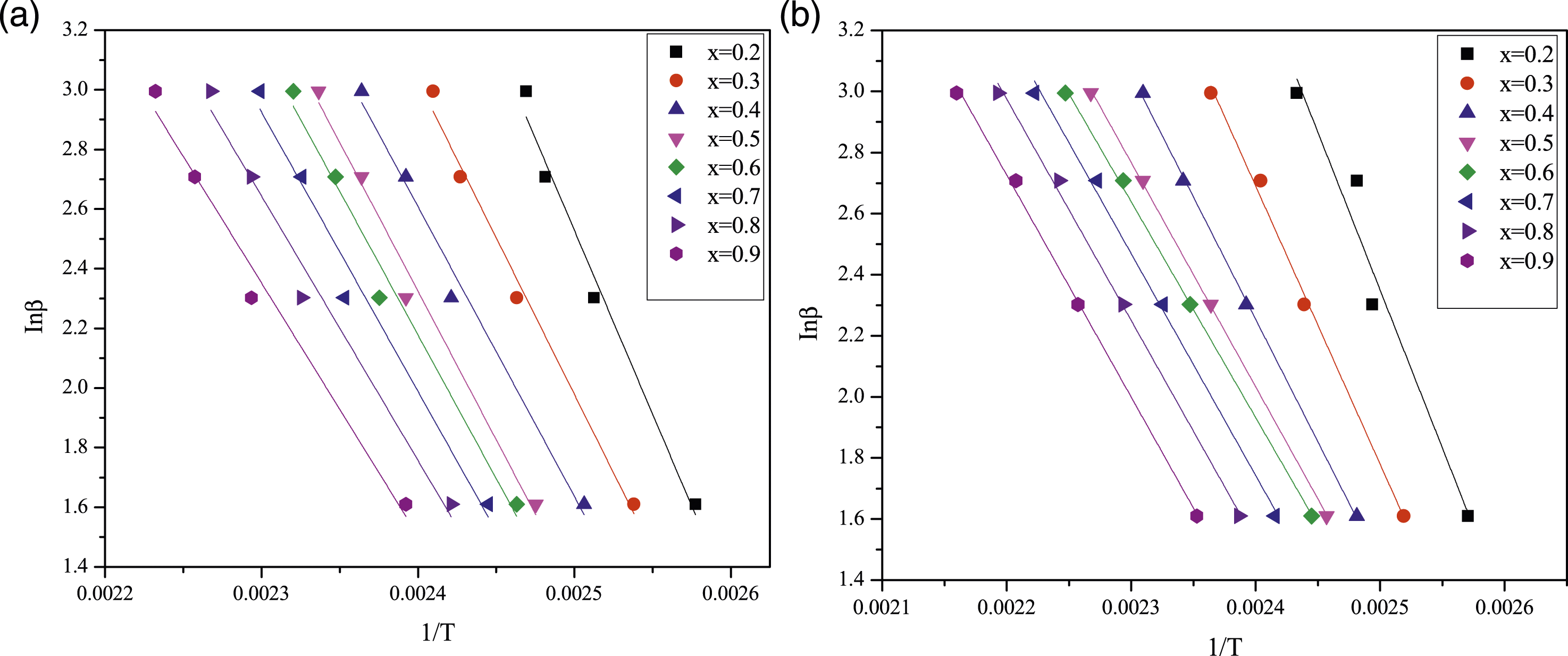
Also noted in Table 1, the activation energies calculated for each conversion factor using the FWO method were slightly higher than those obtained through the KAS method. The highest activation energy for pure epoxy at different conversion levels was determined to be 80.66 kJ mol−1 K−1 using the KAS method and 91.89 kJ mol−1 K−1 using the FWO method. Likewise, for the MAF/EP composite, the maximum activation energy at various conversion levels was identified as 68.95 kJ mol−1 K−1 using the KAS method and 79.67 kJ mol−1 K−1 using the FWO method.
It is found that both the FWO and KAS methods indicate a reduction in the value of activation energy at late stages of epoxy curing. Generally, the physical state of the curing reaction within the system shifts from a liquid state in the early stages to a cross-linked solid state as the curing reaction advances. As the curing reaction progresses, the molecular weight of the system increases, resulting in an elevated viscosity due to reduced molecular mobility of the curing components. Consequently, the curing kinetics transition from a chemical-controlled mechanism to a diffusion-controlled mechanism. This change leads to a reduction in the activation energy due to the diffusion of small curing components associated with the vitrification phenomenon. 25
Isothermal kinetics analysis
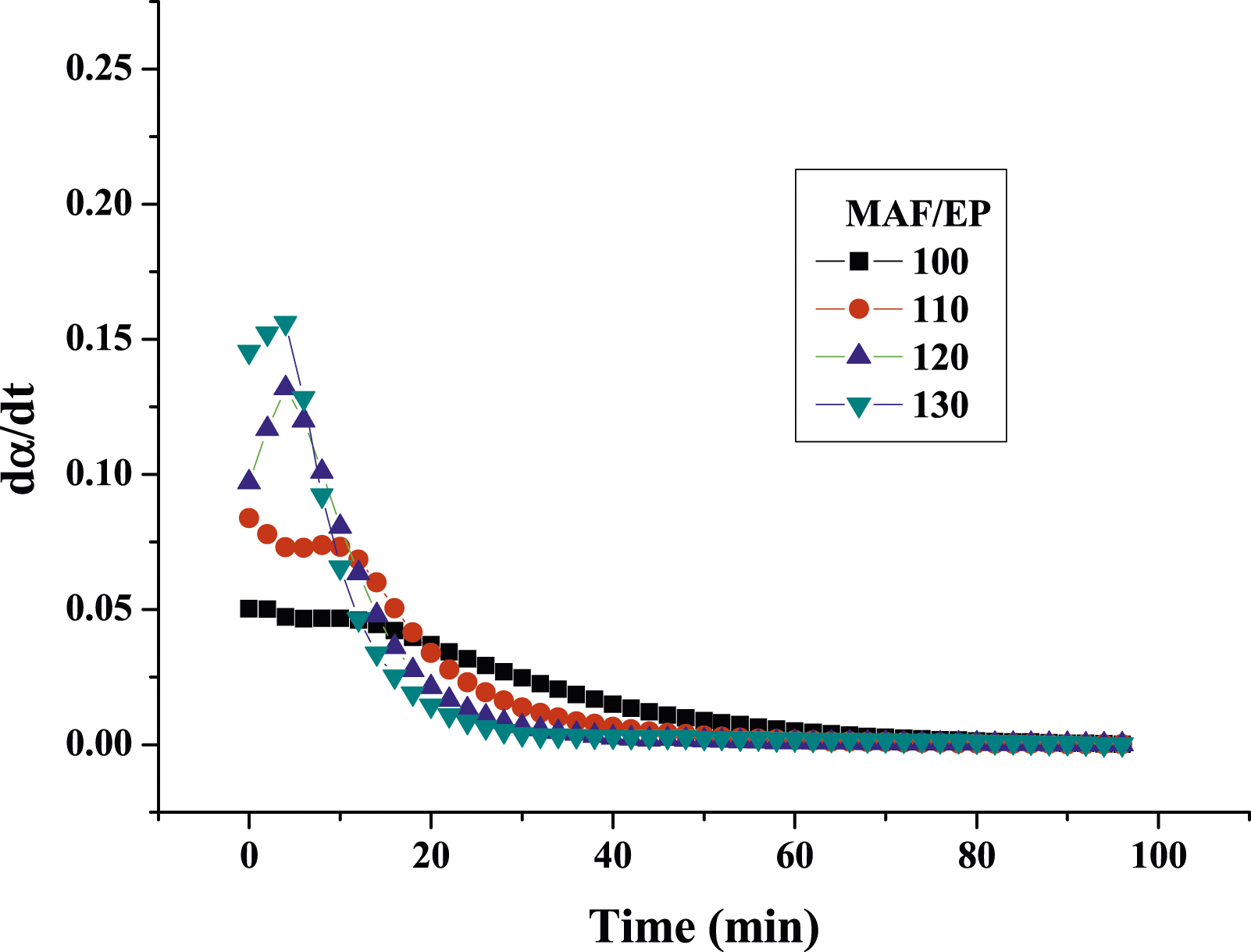
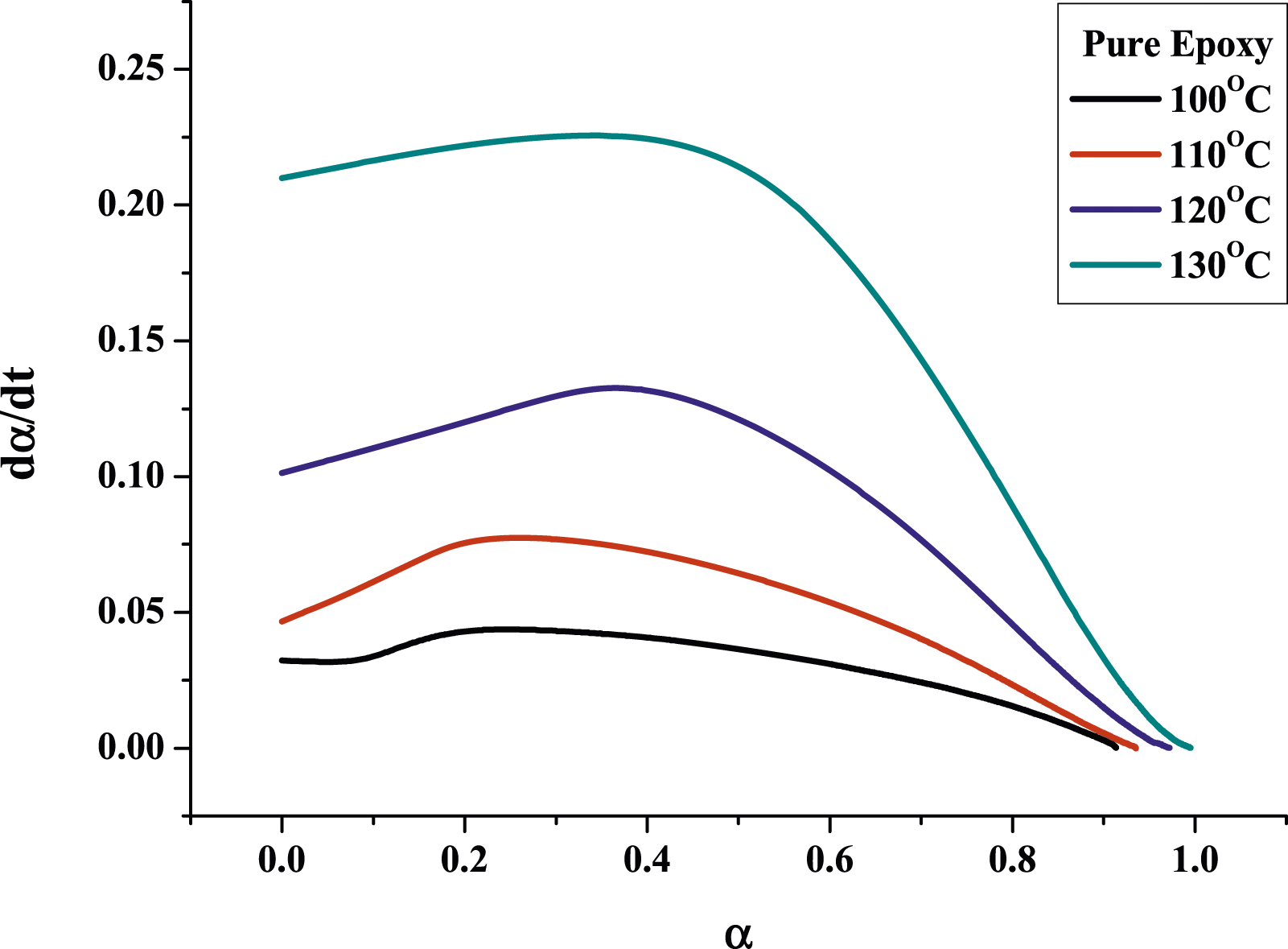
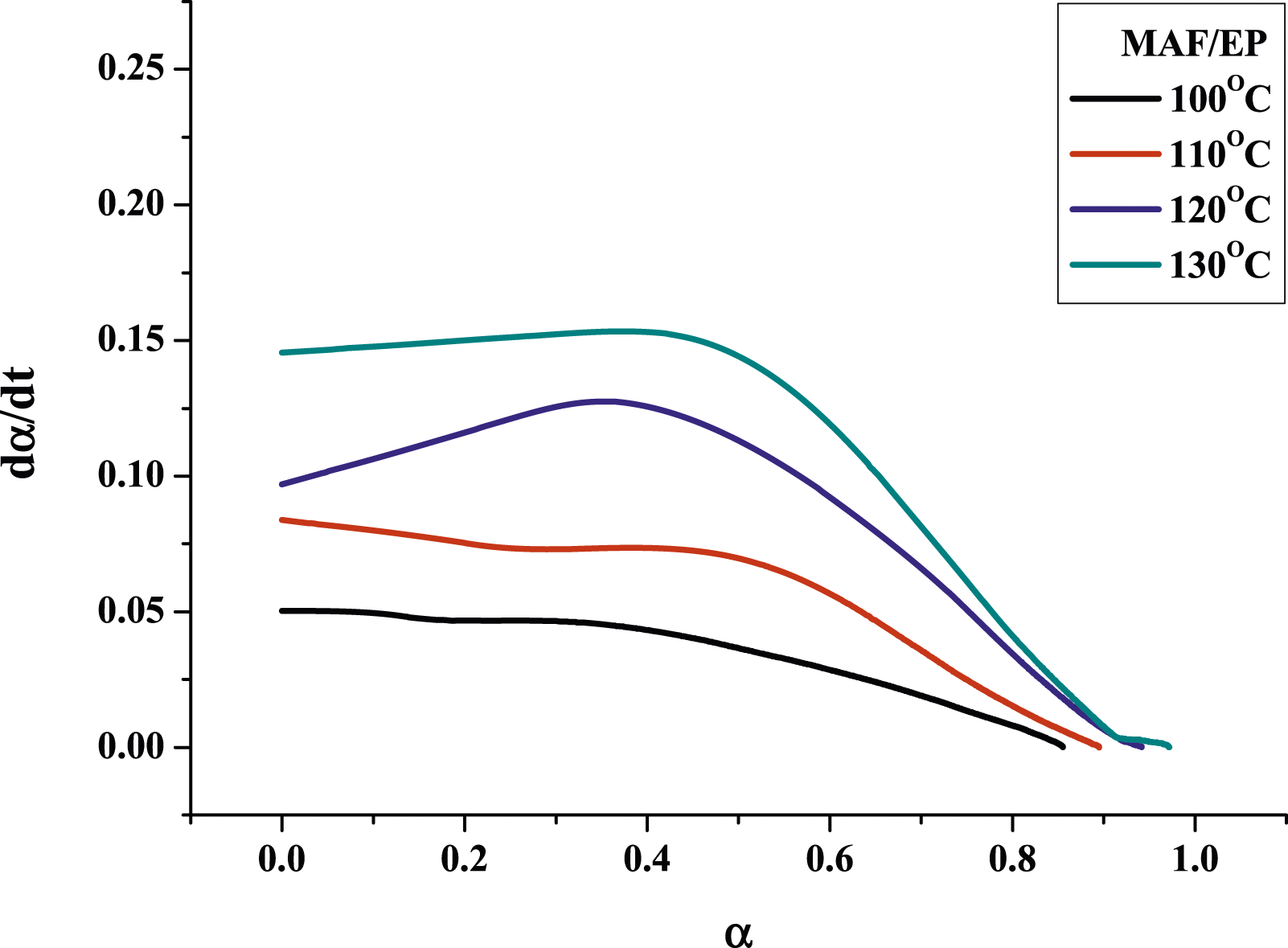
Figures 7 and 8 show the plots of the curing reaction rate (dα/dt) versus time t at different isothermal temperatures. For pure epoxy resin, the reaction rate increases as the scanning time increases. When it reaches a maximum value, the reaction rate will gradually slow down to zero, and the peak of the reaction rate became higher and shifted to a shorter time with increasing temperature. This reflects the type of epoxy resin conversion process containing autocatalytic reaction. However, for MAF/EP composite, the curves of the temperature at 100°C and 110°C do not fully conform to the autocatalytic reaction mechanism, while the temperature at 120°C and 130°C do. It is possible that the increased viscosity and steric obstacles of fibers will affect the progress of the curing reaction. However, at the increased temperatures of 120°C and 130°C, the impact of the steric hindrance produced by the pineapple leaf fiber will become less significant. The reaction rate (dα/dt) plotting against the conversion (α) is shown as Figures 9 and 10. In addition, Table 2 shows that pure epoxy has a higher degree of cross-linking than MAF/EP composite. It can be seen that the steric barrier and high viscosity of the MAF-containing composite may affect the mechanism of the cross-linking reaction. Curing reaction rate versus time for pure epoxy at different curing temperatures. Curing reaction rate versus time for MAF/EP composite at different curing temperatures. Reaction rate against conversion for pure epoxy at different curing temperatures. Reaction rate against conversion for MAF/EP composite at different curing temperatures. Dynamic parameters calculated by the kamal model for epoxy and MAF/EP composites.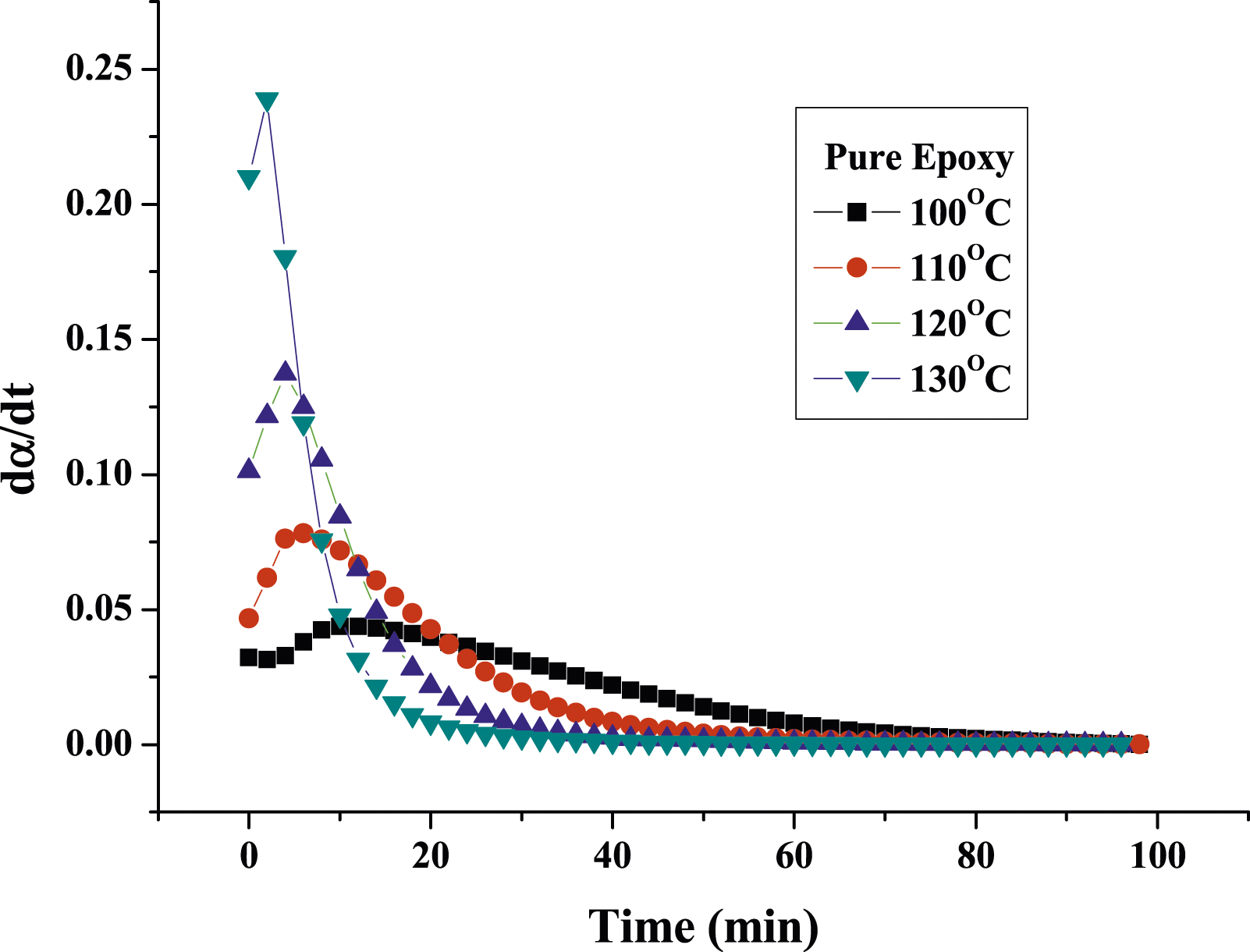
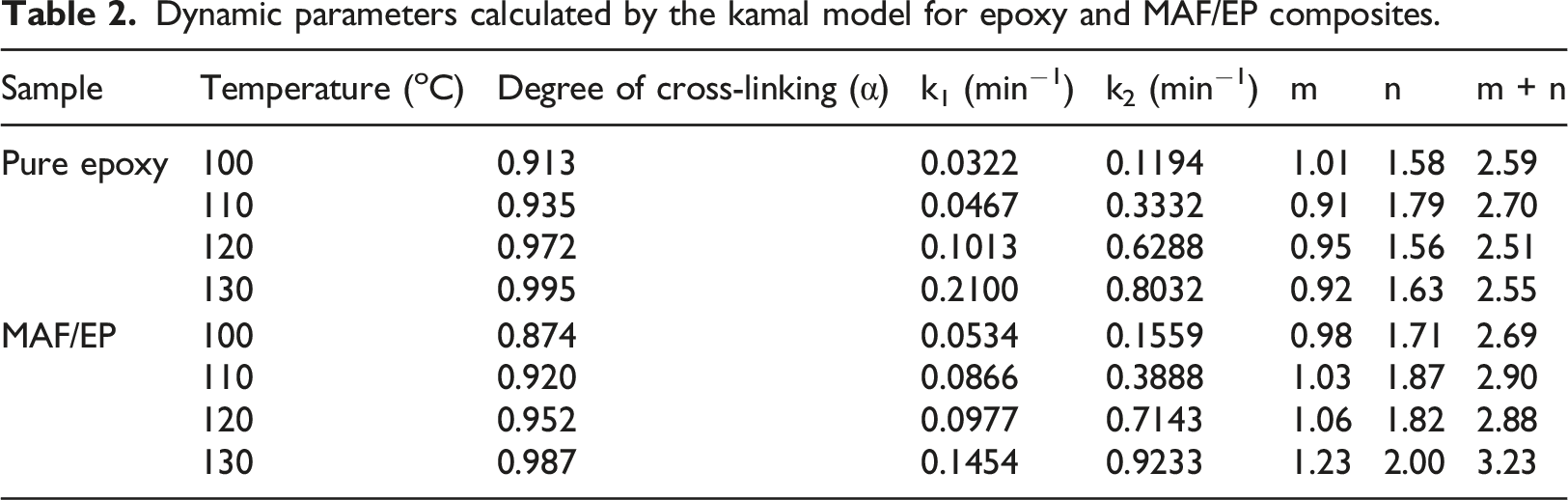
The kinetic parameters obtained from Kamal’s model are shown in Table 1. It can be seen that both
Cure index
Cure characteristics of MAF/EP composites as a function of heating rate.

In this study, ΔT* was greater than one for the MAF/EP composite, especially at higher heating rates of 15 and 20°C/min. The study conducted by Jouyandeh et al. also found that the SiO2 modified by polyethylenimine notably amplified ΔT, especially with elevated heating rates. Remarkably, the substantial nitrogen-rich macromolecules affixed to the surface of SiO2 nanoparticles exert a profound influence on diffusion-controlled crosslinking during advanced stages. This influence arises from the active participation of nitrogen groups, which continue to engage in the ring-opening reaction of epoxy groups even after vitrification. 26
The impact of fillers on the epoxy resin curing process can be explained by considering the competition between two opposing factors: the acceleration effect of functional groups available or attached to the particle surfaces, and the retarding effect caused by the steric hindrance of nanoparticles. These conflicting effects play a crucial role in controlling the crosslinking process. From a molecular perspective, the addition of reactive fillers to the epoxy system restricts the mobility of reactive species, resulting in increased system viscosity. Consequently, one can anticipate that the cure reaction of epoxy may be hindered by the introduction of PALF, especially when the PALFs content is increased. 27
Furthermore, the effects can be related to the CI theory that is based on an extremely low filler concentration, typically less than 0.2 wt% relative to the total weight of the resin/hardener mixture. Nonetheless, in this study, the PALF content reached as high as 20 wt%, making it essential to consider the impact of filler aggregation and the resulting high viscosity on crosslinking reactions.
It has been observed that CI values are higher at higher heating rates (15 and 20°C/min) compared to those at lower heating rates (5 or 10°C/min). This suggests that the influence of filler aggregation and high viscosity on the curing process may be mitigated by the rapid heat supply associated with higher heating rates. This observation is consistent with the findings of the kinetic study mentioned above.
Conclusion
The application of PALF is hope to increase as it is an naturally available fiber, and the use of which reduces considerable wastes. With the structural benefits that the PALF provides, an investigation was performed to assess its effect in the curing of the epoxy of its composite. Namely, the curing kinetics of the composites were analyzed via the isothermal and non-isothermal models. Thermal data was obtained by the use of the DSC. The curing performance of PALF was compared with pure epoxy resin, respectively. According to the recommendations for thermal polymerization processes developed by ICTAC. The KAS and FWO methods were used for the non-isothermal analysis, and Kamal model was used for the isothermal analysis. The highest activation energies for pure epoxy and MAF/EP composite at different conversion levels calculated using the KAS method were 80.66 kJ mol−1 K−1 and 68.95 kJ mol−1 K−1, respectively. On the other hand, the highest activation energies for pure epoxy and MAF/EP composite at different conversion levels calculated using the FWO method were 91.89 kJ mol−1 K−1 and 79.67 kJ mol−1 K−1, respectively. Moreover, it reveals that the activation energies calculated for each conversion factor using the FWO method were slightly higher than those obtained through the KAS method. In addition, a reduction trend in the value of activation energy at late stages of epoxy curing, indicating the curing kinetics transition from a chemical-controlled mechanism to a diffusion-controlled mechanism. The kinetic parameters obtained from Kamal's model indicate that the k1 values of the MAF/EP composite (0.0534 and 0.0866) are only greater than those of pure epoxy (0.0322 and 0.0467) at 100°C and 110°C. However, the k2 values of MAF/EP (0.1559, 0.3888, 0.7143, and 0.9233) are greater than those of pure epoxy (0.1194, 0.3332, 0.6288, and 0.8032) at all temperatures. In addition, the m value of the MAF/EP composite (0.98) is lower than that of pure epoxy (1.01) only at 100°C, while the n and m + n values of the MAF/EP composite (2.69, 2.90, 2.88, and 3.23) are greater than those of pure epoxy (2.59, 2.70, 2.51, and 2.55) at all temperatures. This is because the hydroxyl groups on the plant fibers and the amine groups on the coupling agent-modified fibers can promote the cross-linking reaction. However, the curing reaction of the composite is affected by steric obstacles and high viscosity resulting from the addition of PALFs. Therefore, the nth-order reaction becomes more significant than the autocatalytic reaction at lower temperatures for the composite. It is suspected that the catalytic reaction caused by the hydroxyl groups on the PALF and the amine groups on the coupling agent-modified fibers tends to be more significant at higher temperatures. Therefore, the effects of the steric obstacles of the pineapple leaf fiber and high viscosity become relatively small. Moreover, the results reveal that the CI of the MAF/EP composite was larger than ΔH* and smaller than ΔT*. Additionally, both Tp* > 1 and ΔT* > 1 were found. Notably, in this study, the PALF content reached as high as 20 wt%, necessitating consideration of the impact of filler aggregation and the resulting high viscosity on crosslinking reactions. The reaction is more likely to continue above a critical conversion due to the slight retardation of gelation and vitrification phenomena in the proximity of the diffusion-controlled region. Consequently, the crosslink density could decrease. Based on the above analyses, it can be observed that all the results consistently indicate that the addition of modified PALF can catalyze the cross-linking reaction of epoxy. However, the high viscosity of the system may lead to a lower degree of cross-linking. By industrial practices, different fiber types and their percentages are mixed with the epoxy resins to increase strengths. The findings in this investigation can be implemented in industrial fabrication to reduce the energy required for curing. This offers an additional merit of using PALF composites. The double-fold benefit of reducing consumption and improving production will abridge agricultural and industrial partnership towards sustainability.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
