Abstract
A ultaviolet–visible light responded photocatalytic nanocomposite, silver/silver bromide/titanium dioxide, supported on graphene oxide (GO; silver/silver bromide/titanium dioxide/GO) was fabricated via a layer intercalation method using n-butylamine, cetyltrimethyl ammonium bromide, titanium dioxide and silver/silver bromide-intercalated GO successively. The resultant silver/silver bromide/titanium dioxide/GO exhibited much stronger visible light absorption and enhanced photocatalytic efficiency than titanium dioxide/GO and titanium dioxide. Furthermore, the degradation efficiency of silver/silver bromide/titanium dioxide/GO was improved when irradiated under light without the ultaviolet cut filter. The apparent degradation rate constants,
Introduction
Chlorophenol is an important organochloride intermediate that has been widely used as antiseptics, herbicides and pesticides. 1 –3 Although most chlorophenols are moderate toxicity, they are easy to accumulate in the environment and human body, hard to degradate and have a strong medicinal taste and smell, leading serious risks to the environment and human health, and be classified as persistent organic pollutants (POPs). Therefore, the treatment of chlorophenols has become a challenging task. Photocatalytic degradation technology is an alternative solution to solve this problem, especially in wastewater treatment, due to its advantages of strong oxidation ability, simple operation and low energy consumption.
As a typical semiconductor photocatalyst, titanium dioxide (TiO2) has been widely studied for air and water purification applications, due to its unique properties such as high photocatalytic activity, low cost and non-toxicity. 4 –6 However, the wide band gap (approximately 3.2 eV) of TiO2 limits its application to ultaviolet (UV) environments, which means it utilizes no more than 5% of the total solar energy. Efforts on improving the photocatalytic efficiency of TiO2 have been extensively investigated in recent studies, 4 –7 mainly focus on lowering the band gap by doping or coupling TiO2 with noble metals, semiconductors, transition and non-metal anions, and thereby increasing its photocatalytic activity to visible light range.
Recently, silver/silver halide (Ag/AgX) plasmon resonance photocatalysts have triggered considerable attentions due to their excellent photocatalytic activity in the visible light range. 8 –10 Generally, TiO2 exhibits high photocatalytic efficiency by short-time UV light irradiation, whereas most visible light photocatalysts require much longer visible light irradiation time to achieve the same photocatalytic efficiency. Therefore, in order to obtain high photocatalytic efficiency in the broad UV–visible (UV-Vis) spectral range, TiO2 and Ag/silver bromide(AgBr) were composited on graphene oxide (GO) sheets to get Ag/AgBr/TiO2/GO photocatalytic nanocomposite in this work. As we know, GO is an outstanding photocatalyst carrier, due to its large surface area, high adsorption ability and good solubility and intercalation properties in aqueous solution. 11 –13 Besides, GO can increase the charge separation and decrease the recombination of photogenerated electron–hole pairs (e−–h+) and surface-adsorbed amount of organic molecules through π–π interactions. In this work, the Ag/AgBr/TiO2/GO nanophotocatalyst was synthesized using n-butylamine (n-BA), cetyltrimethyl ammonium bromide (CTAB), TiO2 and Ag/AgBr-intercalated GO successively. The resultant Ag/AgBr/TiO2/GO exhibited high UV-Vis light photocatalytic activity for the degradation of 4-chlorophenol (4-CP) solution and kept stable even after five consecutive photodegradation cycles. The photocatalytic mechanism of Ag/AgBr/TiO2/GO under light irradiation with/without UV cut filter was discussed respectively.
Experimental
Materials
Silver nitrate (AgNO3), TiO2 powder, CTAB, n-BA and 4-CP were all analytical grade and purchased from Sinopharm Chemical Reagent Co., Ltd. Natural graphite powder (44 μm) was provided by Qingdao Zhongtian Company. The chemicals were used without further purification.
Preparation of Ag/AgBr/TiO2/GO photocatalyst
As descripted by Figure 1, Ag/AgBr/TiO2/GO nanocomposite was fabricated by intercalation of GO as followed with n-BA, CTAB, TiO2 and Ag/AgBr. GO was prepared from purified natural graphite using a modified Hummers’ method 14 ; 50 mg of the obtained GO powder and 2.5 mL of n-BA were dispersed in 50 mL of water by sonication, forming a stable GO–n-BA colloid. After 30 min, 12 mL of CTAB (0.05 mol·L–1) aqueous solution was added and then sonicated for 30 min to form a GO–CTAB colloid; 0.5 g of TiO2 powder that pre-sonicated in 35 mL of water was added slowly into the above GO–CTAB colloid under sonication. After the suspension was stirred for additional 2 h, TiO2/GO was obtained. At last, the solution mixed with 0.21 g of AgNO3, 2.3 mL of NH3·H2O and 5 mL of water was quickly added into the suspension of TiO2/GO and then kept stirring for 12 h. The Ag/AgBr/TiO2/GO sample can be obtained by centrifugation (4400 r min−1, 8 min) of the mixture and washed with isopropyl alcohol, alcohol and water, respectively, and then dried at 60°C under vacuum for 12 h.

Schematic illustration of the preparation of Ag/AgBr/TiO2/GO. AgBr: silver bromide; Ag: silver; TiO2: titanium dioxide; GO: graphene oxide.
Characterization
X-ray diffraction (XRD) patterns of samples were scanned on a D8 Advanced X-ray diffractometer (Bruker, Karlsruhe, Germany) using Cu
Evaluation of photocatalytic performance
The photocatalytic activity of Ag/AgBr/TiO2/GO composite was examined through photodegradation of 4-CP. Typically, 100 mg of Ag/AgBr/TiO2/GO was dispersed into a 100 mL aqueous solution of 4-CP (20 mg·L−1). The pH value of 4-CP solution was adjusted to 4. Prior to irradiation, the dispersion was kept in the dark under magnetic stirring for 1 h to establish an adsorption–desorption equilibrium state. The irradiation light was provided by a Xenon arc lamp (500 W), operated on a GHX-2 photochemical reactor. The temperature of the reaction was kept at about 25°C by recycled water. Air was bubbled through the reaction solution from the bottom with an air flow of 250 mL·min−1 to ensure effective dispersion. To monitor the photodegradation, 3 mL of dispersion was taken out from the reaction system at certain time intervals and separated through centrifugation and then monitored by UV-Vis spectroscopy (Shimadzu UV-2450) through recording the absorbance changes. For comparison, the photocatalytic performances of TiO2 and TiO2/GO were investigated under the same condition.
Results and discussion
Characterization of the as-synthesized nanocomposite
The powder XRD peak of GO was observed at around 2
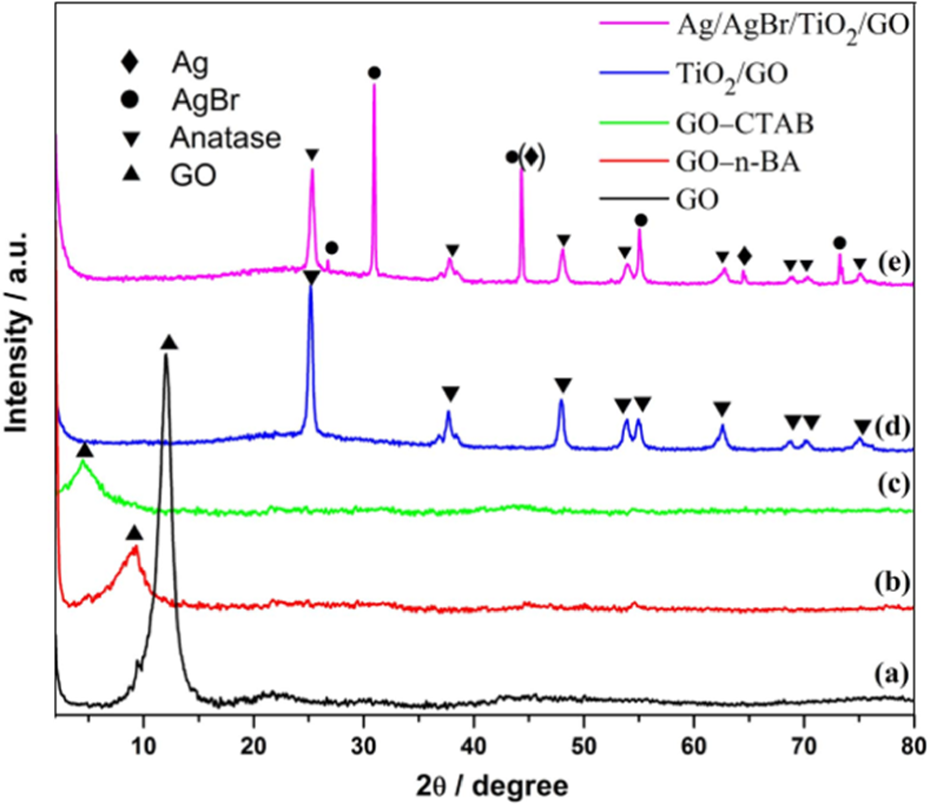
XRD patterns of the resultant. (a) GO, (b) GO–n-BA, (c) GO–CTAB, (d) TiO2/GO, and (e) Ag/AgBr/TiO2/GO. AgBr: silver bromide; XRD: X-ray diffraction; GO: graphene oxide; n-BA: n-butylamine; CTAB: cetyltrimethyl ammonium bromide; Ag: silver; TiO2: titanium dioxide.
The microscopic morphologies and structures of Ag/AgBr/TiO2/GO were observed via TEM and SEM. Figure 3 shows that the almost transparent and crumpled silk-like GO sheets were decorated by two kinds of nanoparticles: TiO2 nanoparticles with an average diameter of approximately 40 nm and the large AgBr nanoparticles with an average diameter of approximately 350 nm. It is known from recent works 8 –10 and our previous work 17 that the small nanoparticles (approximately 30 nm) adhered onto the surface of AgBr were Ag0 decomposed from AgBr. The calculated mole ratio of Ag–Br–Ti of Ag/AgBr/TiO2/GO composite from Energy-dispersive X-ray spectroscopy (EDS) image was 10:9:81 (Online Supplementary Figure S1), indicating that a small proportion (10%) of AgBr is decomposed into metallic Ag0, which agrees well with the XRD spectrum.

(a) TEM image of the resultant Ag/AgBr/TiO2/GO. (b) SEM image of the resultant Ag/AgBr/TiO2/GO. AgBr: silver bromide; TEM: transmission electron microscope; Ag: silver; TiO2: titanium dioxide; GO: graphene oxide; SEM: scanning electron microscope.
UV–Vis DRS spectra of TiO2, TiO2/GO and Ag/AgBr/TiO2/GO photocatalysts are compared in Figure 4. TiO2/GO exhibited stronger visible light absorption and a red shift to longer wavelength range compared to TiO2 due to the incorporation of GO. For Ag/AgBr/TiO2/GO, the surface plasmon resonance (SPR) of Ag nanoparticles
18
and visible light responded AgBr nanoparticles
19
can strongly absorb visible light. Therefore, Ag/AgBr/TiO2/GO showed significant enhancement of absorption in the visible light range compared to TiO2 and TiO2/GO. The band gap energy (

(a) UV-Vis DRS spectra of TiO2, TiO2/GO and Ag/AgBr/TiO2/GO and (b) Tauc plots of these nanocomposites. AgBr: silver bromide; UV-Vis: ultaviolet–visible. UV-Vis DRS: UV-Vis diffuse reflectance spectroscopy; TiO2: titanium dioxide; GO: graphene oxide; Ag: silver.
Photocatalytic performance for degradation of 4-CP
To evaluate the photocatalytic activity of the resultant Ag/AgBr/TiO2/GO, firstly, the photodegradations of 4-CP were carried out under visible light irradiation provided by a Xenon arc lamp with a UV cut filter (

Photocatalytic activities of the resultant TiO2, TiO2/GO and Ag/AgBr/TiO2/GO for photodegradation of 4-CP molecules under light irradiation (a) with and (b) without the UV cut filter
Secondly, the photodegradations of 4-CP were also carried out with the same Xenon lamp but without the UV cut filter. All of the three samples showed enhanced photocatalytic activities in this case (Figure 5(b) and (c)). As we know, the light provided by a Xenon arc lamp is very similar to natural sunlight in the UV and visible light ranges. The output percentages of UV, visible and infrared light are 10.9, 33.9 and 55.1%, respectively. TiO2 could be excited by UV light and generate electron–hole pairs to exhibit efficient photocatalytic activity for degrading 4-CP. Due to TiO2 was excited, the time for degradation of 4-CP was reduced, and the degradation efficiencies of these TiO2 nanospecies were improved. After irradiated for 5 h, the removal of 4-CP reached to 91.5% in the presence of Ag/AgBr/TiO2/GO, while 65.5 and 26.5% removal for TiO2/GO and TiO2, respectively.
At low concentration of 4-CP, the pseudo-first-order kinetics model, a simplified form of Langmuir–Hinshelwood model,
25
was roughly applied for analysis of the photocatalytic degradation kinetics of 4-CP in our system


where
As shown in Figure 5(d), the calculated values of apparent rate constants
The stability of Ag/AgBr/TiO2/GO was further investigated by recycling it for 4-CP degradation as shown in Figure 6. In the photodegradations cycles, Ag/AgBr/TiO2/GO was easily recycled by simple centrifugation without any other treatment. As shown in Figure 6, the high photocatalytic activity was still achieved without significant decrease after five consecutive cycles. The previous research of our group 17 has shown that Ag/AgBr could keep stable in GO-based nanocomposites under light irradiation. It is found that the Ag/AgX photocatalysts, especially Ag/AgX deposited on a conducting support, are quite stable and high efficient. The stability of the Ag/AgX photocatalysts under light irradiation arises from the clusters of Ag atoms around AgX surface which restrict the decomposition of AgX, in fact most likely from that a photon is absorbed by the Ag nanoparticles, and an electron separated from an absorbed photon remains in the nanoparticles rather than being transferred to the Ag+ ions of the AgX lattice. In this article, the morphology of Ag/AgBr/TiO2/GO did not display significant changes after five cycling photodegradations in Online Supplementary Figure S2. Based on the above results and discussion, Ag/AgBr/TiO2/GO could keep structure stable and exhibit good reusability and photochemical stability during the photocatalytic process.

Five consecutive cycling photodegradation curves of 4-CP over Ag/AgBr/TiO2/GO under light irradiation without the UV cut filter. AgBr: silver bromide; UV: ultaviolet; 4-CP: 4-chlorophenol; Ag: silver; TiO2: titanium dioxide; GO: graphene oxide.
Proposed photocatalytic mechanism
Armed with the above results, a possible photocatalytic mechanism for degradation of 4-CP by Ag/AgBr/TiO2/GO under visible light irradiation with/without the UV cut filter was proposed in Figure 7. When irradiated with light of

Proposed photocatalytic mechanism of Ag/AgBr/TiO2/GO for photodegradation of 4-CP under irradiation (a) with and (b) without the UV cut filter. AgBr: silver bromide; Ag: silver; TiO2: titanium dioxide; GO: graphene oxide; 4-CP: 4-chlorophenol; UV: ultaviolet.
When irradiated under light without the UV cut filter, all of the three components (TiO2, Ag and AgBr) could generate photoelectrons. Besides the photoexcited AgBr and plasmonexcited Ag, TiO2 could be excited by the 10.9% output UV light from a Xenon arc lamp. Although the proportion of UV light is small, the superior UV photocatalytic efficiency of TiO2 made a valuable contribution to the total degradation as illustrated in Figure 5(b) and (c).
Conclusion
In order to obtain high photocatalytic efficiency in the broad UV-Vis spectral range, Ag/AgBr/TiO2/GO photocatalytic nanocomposite was synthesized using n-BA, CTAB, TiO2 and Ag/AgBr-intercalated GO successively. The resultant Ag/AgBr/TiO2/GO nanocomposite exhibited enhanced photocatalytic efficiency for the degradation of the POPs molecules 4-CP under light irradiation without the UV cut filter. The apparent degradation rate constants
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (51302113) and Natural Science Foundation of Jiangsu Province (BK20130512). We are also indebted to Jiangsu Training Programs for Innovation and Entrepreneurship for Undergraduate (201613986010Y).
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
