Abstract
The adsorption removal of quinoline from model diesel by using alumina, diatomite, silica gel, and allochroic silica gel as adsorbents was investigated. The experiment results indicated that the adsorption denitrogenation performance of allochroic silica gel was more superior to other three adsorbents. The silica gel and allochroic silica gel were confirmed by characterization with X-ray diffraction, nitrogen adsorption–desorption, and ammonia temperature programmed desoption (NH3-TPD). X-ray diffraction results indicated that both the samples were amorphous structures. The average pore diameters of silica gel and allochroic silica gel were 18.46 and 1.80 nm, the Brunauer–Emmett–Teller surface areas were 437.86 and 623.39 m2/g, and the pore volumes were 0.9724 and 0.3442 cm3/g, respectively. The results of TPD showed that the acidity of allochroic silica gel was far stronger than that of silica gel which greatly enhanced its adsorption denitrogenation performance. The adsorption denitrogenation performance of allochroic silica gel for quinoline, aniline, and pyridine from model diesel was as follows: aniline, pyridine, and quinoline. Adsorption temperature, particle size, and arenes added in model diesel had little impact on the removal of aniline and pyridine except quinoline. The adsorbent-to-oil ratio had significant effects on adsorption denitrogenation, especially for quinoline. The N–Co bond between Co in allochroic silica gel and N atom in the basic nitrogen compounds molecule played a significant role. Furthermore, the allochroic silica gel could be easily regenerated by its adsorption denitrogenation performance for quinoline and pyridine by using calcination once or several times, except aniline. The adsorption isotherm results revealed that the adsorption of pyridine and aniline belonged to the Langmuir–Freundlich binding model, but the adsorption of quinoline belonged to Freundlich model.
Keywords
Introduction
With the increasing concern about environmental protection, regulation of emissions control for vehicles is more and more stringent. The development of the refinery enterprises will focus on clean refinery production and the production of cleaner fuels, especially diesel oil. The removal of nitrogen compounds from diesel oil has become a more and more important research subject. It is well known that the nitrogen compounds in diesel oil can be sorted into nonbasic nitrogen compounds (such as pyrroles, indoles, and carbazoles) and basic nitrogen compounds (such as anilines, pyridines, and quinolines) (Cheng et al., 2011; Laredo et al., 2002) which are the major harmful nitrogen compounds (Glaucia et al., 2017). The latter basic nitrogen compounds can also seriously affect deep removal of sulfur compounds by using hydrodesulfurization (García-Gutiérrez et al., 2014; Polo, 2015). Main techniques for the denitrogenation of diesel oil include hydrogenation refining which needs strict operation conditions and huge instrument investment and nonhydrogenation refining such as liquid–liquid phase partitioning, nitrogen removal methods by solid adsorbents, and so on. These adsorbents for removing the nitrogen from diesel oil mainly include activated carbon (Schmitt et al., 2017; Wen et al., 2017), silica gel (Gaudino et al., 2016; Mushrush et al., 2011), metallic oxide (Silva et al., 2014; Silveira et al., 2015), metal–organic framework crystalline materials (Imteaz et al., 2013; Imteaz and Sung, 2014; Nazmul et al., 2017), modified NaY (Hong and Tang, 2015a; Liu et al., 2008), mesoporous materials SBA-15 (Shahriar et al., 2012; Zhu and Shen, 2012) and MCM-41 (Tang and Hong, 2016; Zhang and Song, 2012) and clays (Baia et al., 2017; Mambrini et al., 2013). Presently, there are some studies on adsorption denitrogenation over silica gel; Mushrush et al. (2011) used silica gel to remove nitrogen from 20 kinds of diesel fuels from different parts of the world and almost all organic nitrogen could be removed. Pretreatment of light cycle oil by silica gel (Yang et al., 2004) before hydrotreating was able to reduce nitrogen content from 635 to 16 µg/g while sulfur and aromatic content remained unchanged. Another pretreatment method with silica gel to remove nitrogen-containing compounds was studied (Lee et al., 2003) before deeper desulfurization. The nitrogen removal ratio could be 82% for straight run gas oil and 86% for coker gas oil. But the reports about the adsorption denitrogenation over allochroic silica gel are hardly found. There are some researches about the adsorption removal of quinoline which typical represents basic nitrogen compounds in fuel (Imteaz et al., 2013; Imteaz and Sung, 2014; Mushrush et al., 2011; Wen et al., 2017), but the researches about the adsorption removal of anilines and pyridines are not enough. In this study, first, the adsorption removal of quinoline in model diesel over alumina, diatomite, silica gel, and allochroic silica gel was investigated. Then the allochroic silica gel was used to remove the basic nitrogen compounds such as quinoline, anilines, or pyridines from model diesel. The geometry optimization of quinoline, aniline, and pyridine was optimized by DMol3 program and these optimization results were linked to the experiment results which contribute to explore the adsorption mechanism. The adsorption isotherm and kinetics of the basic nitrogen compounds on the allochroic silica gel were also investigated.
Materials and experiment methods
Materials
Quinoline, naphthalene, toluene, and benzene (all these reagents are analytically pure) are supplied by Sinopharm Chemical Reagent Co., Ltd. Anilines (analytically pure) and pyridines (analytically pure) are supplied by Tianjin Guangfu Fine Chemical Research Institute. Allochroic silica gel (CoCl2 content 2%) and silica gel are supplied by Tianjin Yongda Chemical Reagent Co., Ltd. Alumina is supplied by Henan Qingquan Environmental Technology Co., Ltd. Dodecane (98%) is supplied by Fortuneibo-tech Co., Ltd. Diatomite is supplied by Jilin Yuantong Mining Co., Ltd.
Characterization methods
The analysis methods of X-ray diffraction (XRD), NH3-TPD, and nitrogen adsorption–desorption are shown as follows:
The XRD spectra were acquired on a German Bruker D8 Advance X ray diffractometer with monochromatic CuKα radiation scanned in the 2θ range of 0°–80°; power supply voltage: 40 kV; tube current: 40 mA; scan rates: 4°/min. NH3-TPD measurements were carried out on an American Micromeritics Instrument Corp 2920 apparatus with a TCD detector. The supported samples (about 50 mg) were added into a U-utilizes quartz tube reactor, blown out under 30 ml/min helium at 10°C/min heating rate until to 550°C, treated in the helium stream at 550°C for 2 h, and cooled to 120°C in the same atmosphere; NH3–Ar (10% NH3) was adsorbed at 120°C for 0.5 h and then swept with 30 ml/min helium as carrier gas until the TCD was stable. Desorption was carried out at a heating rate of 10°C/min. Nitrogen adsorption–desorption isotherms were measured at −196°C with a Japanese BEL Corp BelSorp-Max physisorption apparatus. The pretreatment conditions of samples were degassed at 300°C for 12 h. Brunauer–Emmett–Teller (BET) and Barrett–Joyner–Halenda (BJH) methods were used to investigate the specific surface area and pore size distribution, respectively. The nitrogen content of the treated model diesel was measured by using SH/T 0162-92 of China.
Adsorption denitrogenation method
In the experiment, model diesel containing known amounts of quinoline, aniline, or pyridine as basic nitrogen compounds in dodecane with a total nitrogen concentration of 960.56 µg(N)/g was prepared (the molar fraction of every basic nitrogen compounds was 0.0514 mol/l). The compositions of the model diesels are summarized in Table 1. The adsorption denitrogenation experiments were referred to Tang and Hong (2016). The specific method was as follows: the model diesel and adsorbent were mixed in a flask in certain proportions with a magnetic stirrer by using static stirring method. After stirring for a certain time, the adsorbents were separated by centrifugation separation of 15 min at about 4000 r/min. The basic nitrogen content of the treated model diesel was measured by using SH/T 0162-92.
Compositions of model diesels.

Model system and calculation method
Geometry optimization of quinoline, aniline, and pyridine was optimized by DMol3 program in the software of Materials Studio from American Accelrys Software House, using double numerical with polarization basis sets and generalized gradient approximation exchange-correlation functionals like Becke–Lee–Yang–Parr. The accuracy of self-consistent field was set at “fine.” The convergence of total energy, gradient, and displacement was 0.00001 Ha, 0.002 Ha/Å, and 0.005 Å, respectively.
Results and discussion
Adsorption removing basic nitrogen compounds over alumina, diatomite, silica gel, and allochroic silica gel
The nitrogen removal ratio and adsorption capacity of quinoline in model diesel over different adsorbents are shown in Figure 1. The nitrogen removal ratio and adsorption capacity of silica gel and allochroic silica gel, significantly higher than that of alumina and diatomite, were 36.47%, 12.10 mg(N)/g adsorbent and 66.88%, 23.56 mg(N)/g adsorbent, respectively. The allochroic silica gel, supported CoCl2 on the silica gel, had much better adsorption denitrogenation performance than silica gel, implying that Co might improve its denitrogenation performance greatly. In our recent studies (Hong and Tang, 2015b), we found that the Co in the channel wall of Co–MCM-41 could enhance the acid amount of samples and the N–M(Co) coordination between the N atom in basic nitrogen compounds molecule and adsorption sites played the important role on the adsorption denitrogenation. Since the Co2+ atom allochroic silica gel has 1s22s2p63s2p6d7 electron configuration and 4s empty orbits, implying that the quinoline, aniline, or pyridine could coordinate directly with the Co metal in allochroic silica gel through N–M(Co) coordination interaction more than π complexation interactions, as Ma et al. (2002) stated in his paper which has investigated a new approach to deep desulfurization of gasoline, diesel fuel, and jet fuel by selective adsorption.

Nitrogen removal ratio and adsorption capacity over different adsorbents for quinoline in model diesel 1 (experiment conditions: diatomite and 140–160 mesh alumina, silica gel, and allochroic silica gel; adsorption temperature: room temperature; adsorbent-to-oil ratio (mass)= 1:35; adsorption time = 0.5 h).
Characterizations of silica gel and allochroic silica gel
The wide-angle XRD patterns of silica gel and allochroic silica gel are shown in Figure 2. Both samples were amorphous in structure and very similar to each other with no significant changes. Therefore, the CoCl2 was well dispersed both in the channels and on the surfaces of the silica gel and did not change the crystallinity nature of silica gel.

Wide-angle XRD patterns of silica gel and allochroic silica gel.
As shown in Figure 3, the nitrogen adsorption–desorption isotherms of silica gel and allochroic silica gel were obviously different and the former could be classified as type IV isotherm and the increasing step appeared at p/p0=0.7–0.9, indicating that silica gel had a large pore diameter of 18.46 nm and wide pore distribution (see BJH pore size distribution in Figure 3) and its surface areas and total pore volumes were 437.86 m2/g and 0.9724 cm3/g; for allochroic silica gel, 1.80 nm, 623.39 m2/g, and 0.3442 cm3/g. The adsorption–desorption isotherm of allochroic silica gel could be classified as type I isotherm and typical for microporous materials and narrow pore distribution. Its desorption isotherm almost coincided with adsorption isotherm except at p/p0=0.32–0.78—a less obvious hysteresis hoop (as shown in Figure 3 magnified section) which indicated that the framework of allochroic silica gel had very small amounts of mesoporous structures. The molecule size of quinoline after geometry optimization was 0.7116 × 0.5002 nm (see Table 2), implying that quinoline molecule could easily access to the pore of silica gel and allochroic silica gel. Although the surface area of allochroic silica gel was only 1.42 times more than that of silica gel, its adsorption capacity (23.56 mg(N)/g, see Figure 1) was almost double that of silica gel (12.10 mg(N)/g). It was correct to infer that the higher quinoline adsorption observed for allochroic silica gel was not due to its higher surface area. Cobalt atoms in allochroic silica gel had an important role in quinoline adsorption.
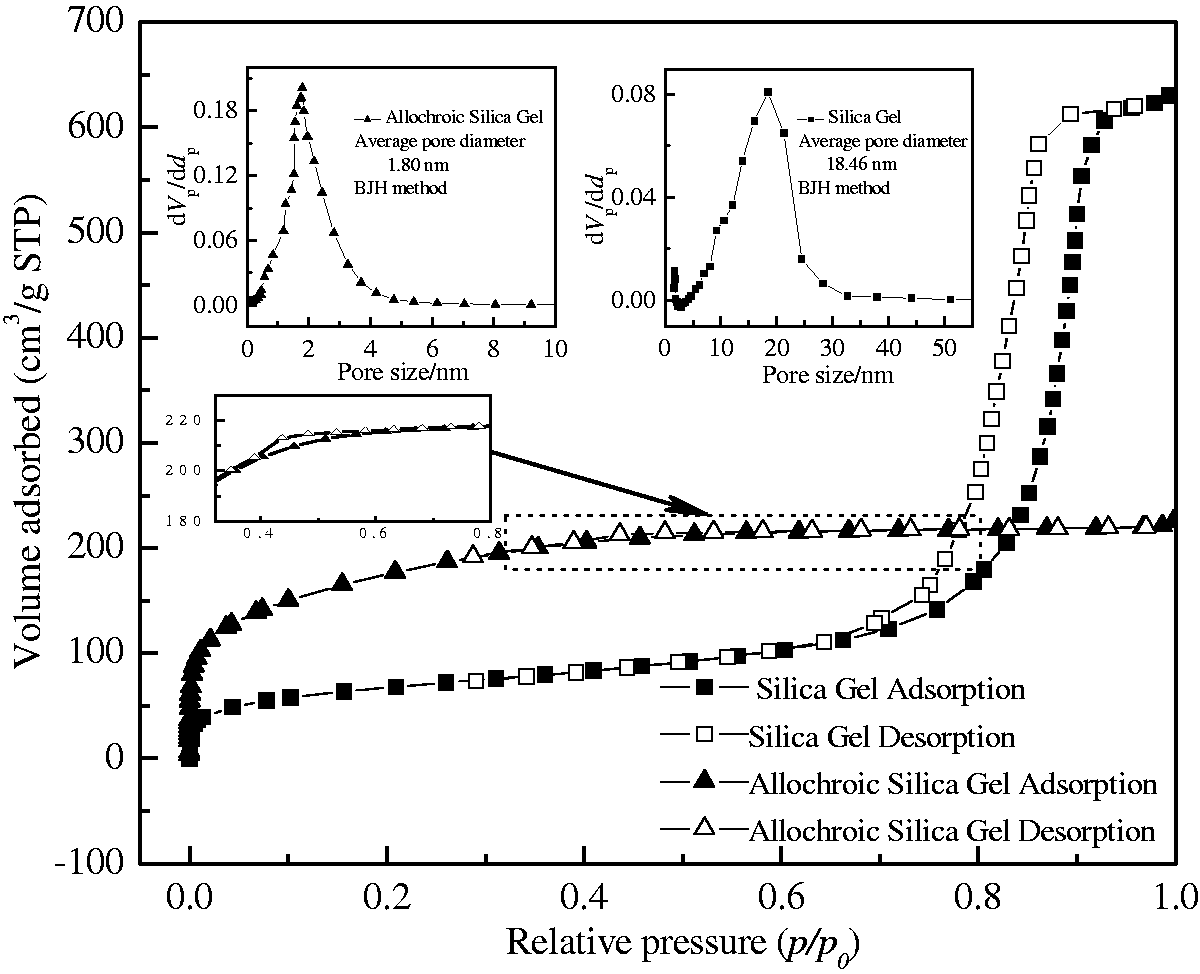
Nitrogen adsorption–desorption isotherms and pore size distribution of silica gel and allochroic silica gel.
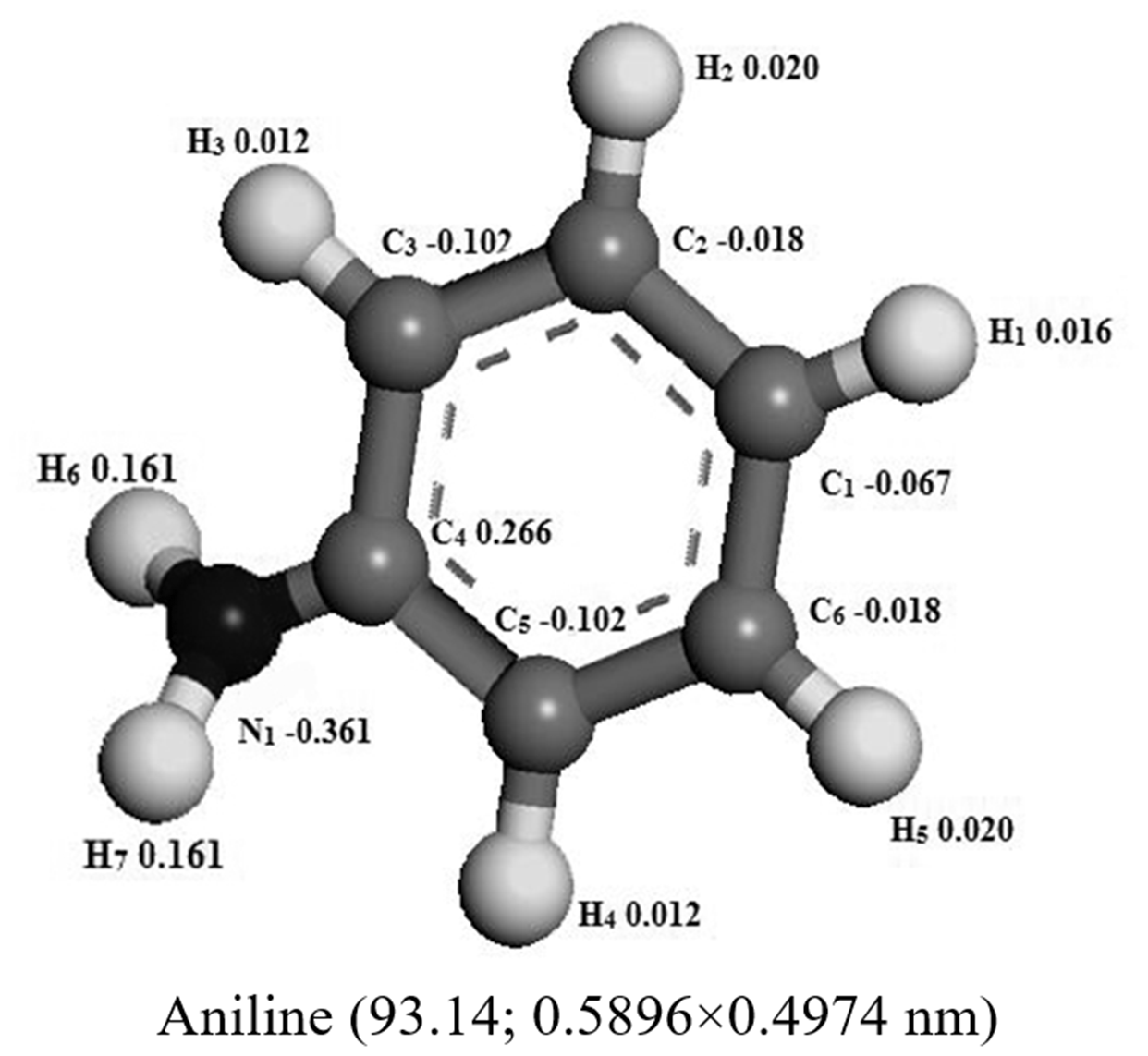
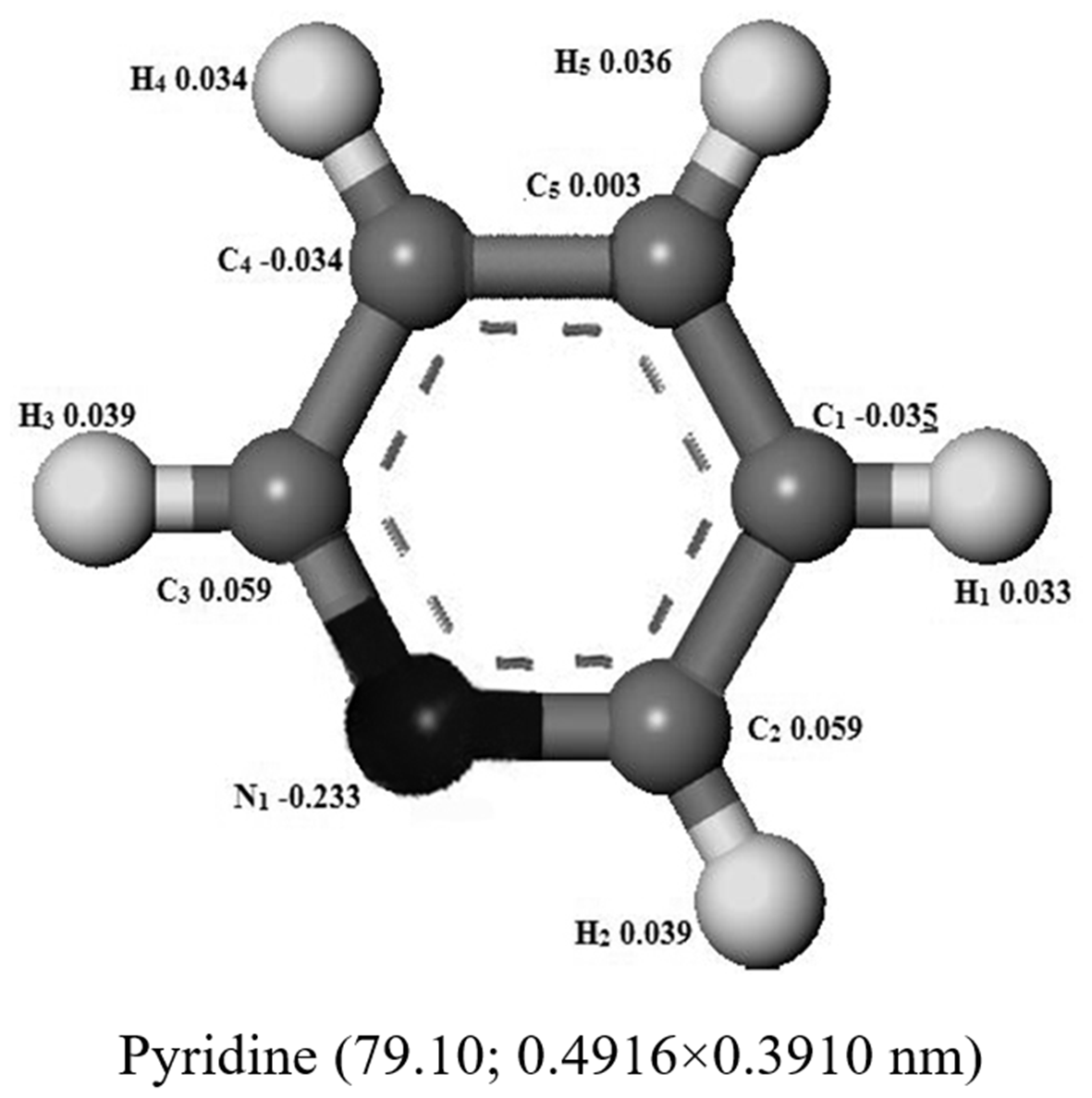
Optimized structures of quinoline, aniline, and pyridine molecules and arenes in the model diesels.

The NH3-TPD profiles of silica gel and allochroic silica gel are shown in Figure 4. It was obvious that the area under desorption profile of allochroic silica gel was greater than silica gel and the amount of ammonia desorbed from the former (0.728 mmol/g) was much more than those from the latter (0.302 mmol/g). Two samples had almost exactly the same desorption peak at about 160°C, which was due to weak acid sites perhaps deriving from acid hydroxyls. The silica gel had only another weak desorption peak at 300°C indicating that it exhibited some medium strong acid. But the allochroic silica gel had two strong peaks at 350 and 480°C, respectively, which could be attributed to the desorption of ammonia from the strong acidic sites. Hence it was concluded that the allochroic silica gel had the more acidic sites with a higher acidity strength which could be attributed to the incorporated Co only. The acidity of silica gel and allochroic silica gel greatly influenced their capacity of adsorption of quinoline, since quinoline was the typical representation of basic nitrogen compounds in fuel.

NH3-TPD patterns of silica gel and allochroic silica gel. TCD: Thermal Conductivity Detector.
Effects of particle size of allochroic silica gel on the adsorption denitrogenation
There are some researches about the adsorption removal of quinoline from model diesel. But so far, the researches on the adsorption removal of aniline and pyridine are not enough. In this experiment, effects of the particle size of allochroic silica gel on nitrogen removal ratio and adsorption capacity are shown in Figure 5. The adsorption denitrogenation performance for quinoline, aniline, and pyridine of allochroic silica gel was improved with decreasing particle size (the mesh increased) and the final nitrogen removal ratio and adsorption capacity for any particle size were still the model diesel 2 (aniline)>3 (pyridine)>1 (quinoline). The particle size had little impact on the adsorption denitrogenation performance for aniline, pyridine, and the nitrogen removal ratio of aniline and pyridine only increased from 88.62 to 91.59% and 75.27 to 81.9%, respectively. But the particle size had a significant impact on the adsorption denitrogenation performance for quinoline and the nitrogen removal ratio increased from 40.58 to 69.72%. The access of basic nitrogen compounds in model diesel to the pores depends on the molecule dimension compared to the pore dimension, and the channel length of sample became smaller when the particle size (the mesh increased) decreased. The molecule sizes of quinoline, aniline, and pyridine were 0.7116 × 0.5002, 0.5896 × 0.4974, and 0.4916 × 0.3910 nm (see Table 2), respectively, so that the particle size had a big effect on the adsorption of quinoline since its molecule size was significantly larger. The particle size had a negligible effect on the adsorption denitrogenation when the mesh size is more than 140–160.

Effect of the particle size of allochroic silica gel on nitrogen removal ratio and adsorption capacity (experiment conditions: adsorption temperature: room temperature; adsorbent-to-oil ratio (mass)=1:35; adsorption time = 0.5 h).
Effects of adsorption temperature on the adsorption denitrogenation over allochroic silica gel
As seen in Figure 6, adsorption temperature had little impact on the adsorption denitrogenation performance for aniline and pyridine and a significant impact on the adsorption denitrogenation performance for quinoline. The nitrogen removal ratio could be reached by maximizing at 50°C, but slightly lower when the temperature was above 50°C. On the one hand, it was well known that the physical adsorption more easily desorbed than the chemical adsorption at higher temperature. On the other hand, the rate at which the molecules move depended upon the energy they had which was determined by temperature. The temperature greatly affected the thermal motion of quinoline since its molecule size was significantly much larger than other molecules which contributed to the adsorption for quinoline. But at higher temperature, the adsorbate molecules were easily desorbed since the energy of molecules was higher, so the nitrogen removal ratio showed slight declines when temperature was above 50°C.

Effect of adsorption temperature on nitrogen removal ratio and adsorption capacity over allochroic silica gel (experiment conditions: adsorbent-to-oil ratio (mass)=1:35; adsorption time = 0.5 h).
Effects of adsorbent-to-oil ratio on the adsorption denitrogenation over allochroic silica gel
The nitrogen removal ratio for quinoline, aniline, and pyridine over allochroic silica gel was increasing dramatically with the increase of adsorbent-to-oil ratio until 1:15 (see Figure 7). And the nitrogen removal ratio was almost the aniline>pyridine>quinoline at any adsorbent-to-oil ratio. The basic nitrogen compounds in model diesel could not be completely removed and even allochroic silica gel reached saturation when the adsorbent-to-oil ratio was small (i.e. less allochroic silica gel). But the adsorption denitrogenation results had been improved with the increase of adsorbent-to-oil ratio until the adsorbent could remove almost all basic nitrogen compounds from the model diesel (i.e. the nitrogen removal ratios were almost 100%) when adsorbent-to-oil ratio was more than 1:15 and the adsorption capacity had fallen since the total nitrogen content in model diesel was a constant value. The adsorption time (10–60 min) had less influence on the adsorption denitrogenation in the research (not shown).
Effects of arenes on the adsorption denitrogenation over allochroic silica gel
The effects of arenes on the adsorption denitrogenation over allochroic silica gel were investigated and the results are shown in Figure 8. These arenes, added in model diesel (the compositions of these model diesels are summarized in Table 1), had the similar molecular structure containing π bonds with the basic nitrogen compounds (see Table 2). Here arenes were used as a model compound for aromatic compounds in transportation fuel that might compete for adsorption sites in allochroic silica gel (by π complexation) against the basic nitrogen compounds. The nitrogen removal ratio decreased a little except the model diesels containing quinoline, and whatever none or any arenes were added into the model diesel, the final nitrogen removal ratio and adsorption capacity were still the model diesel 2 (aniline)>3 (pyridine)>1 (quinoline).
In the experiment, geometry optimization of quinoline, aniline, and pyridine was shown in Table 2. It is well known that the N atom in quinoline and pyridine molecule has a long pair of electrons which would lead to their strongly alkaline. The lone pair electrons of N atom in aniline molecule formed the delocalized conjugated system with benzene, so the aniline molecule had less electron density and weakly alkaline. In the N atom in aniline molecule, presented in the amidogen group, steric hindrance is obviously less than that of quinoline and pyridine in that the latter N atom is on the benzene ring. On the other hand, the charges on N atom of quinoline, aniline, and pyridine were −0.252, −0.361, and −0.233 (see Table 2), implying that the N–M(Co) coordination between aniline molecule and the adsorption sites is much stronger than that of pyridine or quinolone (Tang and Hong, 2016). But the steric hindrance of N atom in quinoline molecule is obviously greater than that of pyridine since the former has two benzene rings and is larger molecule. So the final sequence of nitrogen removing was the aniline>pyridine>quinoline. In accordance with the above analysis, the N–M(Co) coordination between the N atom in molecule and adsorption sites played the important role on the adsorption denitrogenation of quinoline, aniline, and pyridine which could be attributed to the nitrogen–metal(Co) interaction rather than π complexation interactions.
Regeneration of allochroic silica gel
Regeneration of the used allochroic silica gel was achieved by calcining at 550°C in air for 5 h since its top tolerance temperature was 600°C. As shown in Figure 9, its adsorption denitrogenation performance for quinoline and pyridine was almost not changed after regeneration. After calcination, the quinoline and pyridine in channels of allochroic silica gel would burn off since their ignition points were 480 and 482°C, then active adsorption sites could reactivate. But the allochroic silica gel had a poor regenerability for aniline in that the ignition point of aniline was 615°C and its nitrogen removal ratio was lowered from 95.58 to 42.78%, adsorption capacity from 13.78 to 6.16 mg(N)/g.
Adsorption isotherm models of allochroic silica gel
The adsorption isotherm researches of model diesel containing quinoline, aniline, or pyridine under 50°C were performed to determine the maximum adsorption capacity of basic nitrogen compounds on allochroic silica gel. The experiment results are shown in Figure 10(a) and the adsorption isotherm data were applied to the Langmuir, Freundlich, and Langmuir–Freundlich models (see Figure 10(b) to (d)). The correlation coefficients are summarized in Table 3.
Langmuir, Freundlich, and Langmuir–Freundlich fitting coefficients for adsorption removal of basic nitrogen compounds—quinoline, aniline, and pyridine.

The sorption amount of basic nitrogen compounds at equilibrium, 

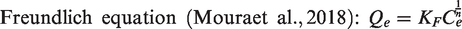

The values of correlation coefficients (R2) obtained for Langmuir–Freundlich models about pyridine and aniline (see Table 3 and Figure 10(c) and (d)) were greater than that of other models which illustrated that the Langmuir–Freundlich binding model provided a better fit than other model when pyridine or aniline in model diesel was removed over allochroic silica gel. But the value of R2 obtained for Freundlich model was greater than that of other models, implying that the Freundlich model provided a better fit than other models for quinoline adsorption (see Table 3 and Figure 10(a)). The value of qm obtained for Langmuir model about aniline and pyridine was greater than that of quinoline which illustrated that the allochroic silica gel had more higher adsorption capacity for aniline and pyridine which was in agreement with the previous paper final sequence in Figures 5 to 7 at any experimental conditions. The value of KL for quinoline was maximum in three regression equations, implying that the allochroic silica gel could adsorb quinoline well at low concentrations (see Table 3 and Figure 10).

Effect of adsorbent-to-oil ratio on nitrogen removal ratio and adsorption capacity over allochroic silica gel (experiment conditions: 140–160 mesh allochroic silica gel; adsorption temperature: 50°C; adsorption time = 0.5 h).

Effects of arenes on the removal of basic nitrogen compounds over allochroic silica gel (experiment conditions: 140–160 mesh allochroic silica gel; adsorption temperature: 50°C; adsorbent-to-oil ratio (mass)=1:35; adsorption time = 0.5 h).

Effects of regeneration times on the removal of basic nitrogen over allochroic silica gel (experiment conditions: 140–160 mesh allochroic silica gel; adsorption temperature: 50°C; adsorbent-to-oil ratio (mass)= 1:15; adsorption time = 0.5 h).
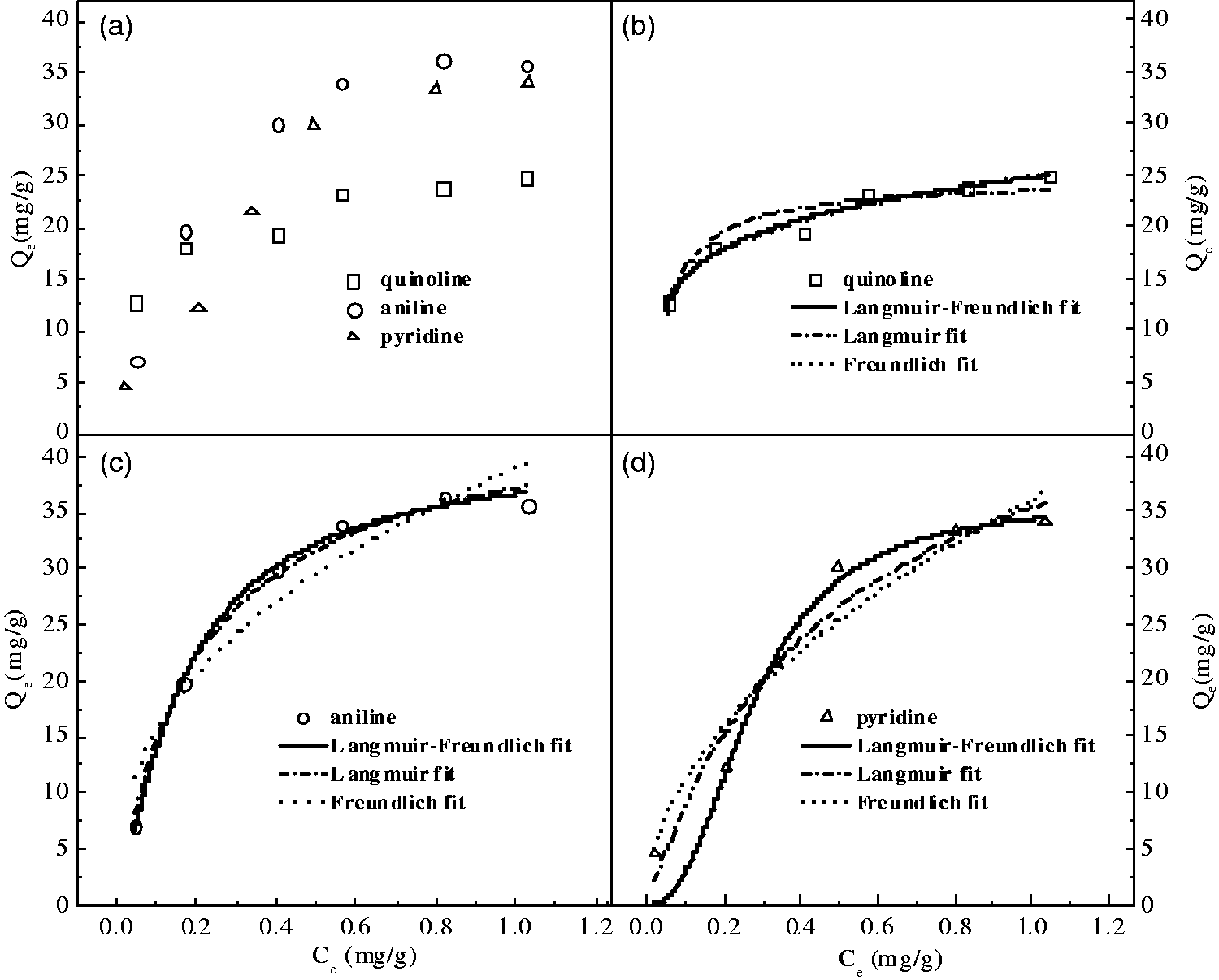
Adsorption isotherms of quinoline, aniline, and pyridine over allochroic silica gel (a) (experiment conditions: 140–160 mesh allochroic silica gel; adsorption temperature: 50°C; adsorbent-to-oil ratio (mass)=1:35; adsorption time = 0.5 h) and Langmuir, Freundlich, and Langmuir–Freundlich of adsorption isotherm fitting from adsorption data ((b) to (d)).
Conclusion
The nitrogen removal ratio and adsorption capacity of silica gel and allochroic silica gel, significantly higher than that of alumina and diatomite, were 36.47%, 12.10 mg(N)/g adsorbent and 66.88%, 23.56 mg(N)/g adsorbent, respectively. The adsorption denitrogenation performance of allochroic silica gel was much better than that of other adsorbents.
Characterization results indicated that both silica gel and allochroic silica gel were amorphous structures. The average pore diameters of them were 18.46 and 1.80 nm, the BET surface areas were 437.86 and 623.39 m2/g, and the pore volumes were 0.9724 and 0.3442 cm3/g, respectively. The results of TPD showed that the acidity of allochroic silica gel was far stronger than that of silica gel which greatly enhanced its adsorption denitrogenation performance.
The adsorption denitrogenation performance of allochroic silica gel for different model diesel was as follows: aniline, pyridine, and quinoline. Adsorption time had almost no influence on the removal ratio of three basic nitrogen compounds. Adsorption temperature, particle size, and arenes in model diesel had little impact on the removal of aniline and pyridine, but evident influences on the removal of quinoline. The adsorbent-to-oil ratio had significant effects on adsorption denitrogenation, especially for quinoline. The N–Co bond between Co in allochroic silica gel and N atom in the basic nitrogen compounds played a significant role which could be attributed to the nitrogen–metal(Co) interaction rather than π complexation interactions. Furthermore, the allochroic silica gel could be easily regenerated by its adsorption denitrogenation performance for quinoline and pyridine by using calcination once or several times, except aniline.
The adsorption of pyridine and aniline over allochroic silica gel belonged to the Langmuir–Freundlich binding model, but the adsorption of quinoline over allochroic silica gel belonged to Freundlich model.
Footnotes
Acknowledgements
We are grateful to the Key Laboratory of Petrochemical Catalytic Science and Technology of Liaoning Shihua University for the molecules structure optimization of quinoline, aniline, and pyridine.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Liaoning Provincial Natural Science Foundation of China (201602380) and opening project of Guanxi College and Universities Key Laboratory of Beibu Gulf Oil and Natural Gas Resource Effective Utilization (2016KLOG04).