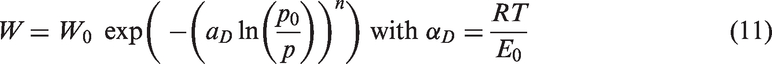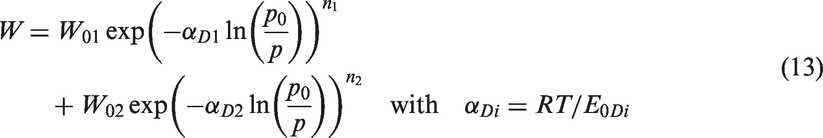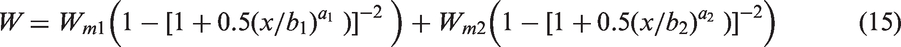Abstract
We show how the Dubinin isotherm and its extensions can be related to the isotherms derived from the general Brouers–Sotolongo isotherm. We compare them using benzene vapor adsorbed on activated carbon data from one of the original Dubinin’s paper. We use the same procedure to analyze data from the thesis of Francois Marquez-Montesino on the adsorption on activated carbons prepared from Pinus caribaea saw dust. We conclude by proposing a simple methodology to determine the macroscopic information given by genuine statistical isotherms.
Keywords
Introduction
Adsorption isotherm is a curve giving the functional relationship between adsorbate and adsorbent in a constant-temperature adsorption process. Since the discussion is quite general and can be applied to adsorption (a physical process), chemi-sorption, and bio-sorption, in the following we will use, when we do not mention explicitly adsorption, the more general term “sorption.” One measures the uptake (gas volume or sorbed quantity) as a function of the gas pressure or the solution concentration. Isotherms have been used for decades in the gaseous or aqueous phase with two main objectives, first to obtain information on the nature of the sorbent surface of porous materials (sorption energy, type of porosity, and type of heterogeneity) in order to use them, taking advantage of their large specific surface, and second, to prepare and characterize specific sorbent to eliminate particular molecules in the treatment of water and more generally for the purpose of physical, chemical, and biological decontamination. One usual practice is to fit the data to empirical or semi-empirical models in order to elect the best one, and then to use other empirical rules to have access to microscopic and mesoscopic information (type of porosity, sorption energy distribution, etc.) and thermodynamic quantities. This is not an easy task because, as discussed theoretically (Brouers and Sotolongo-Costa, 2005; Brouers et al., 2005; Jurlewicz and Weron, 1999; Stanislavsky and Weron, 2013), macroscopic measurements are generated by the extreme value distribution of microscopic quantities. They can give general scaling tendencies, but cannot yield valid detailed micro or meso information without establishing correlations with independent measurements and direct micrographic analysis.
The long history of sorption is dominated by three names, Freundlich (1906), Langmuir (1918), and Dubinin (1989). Freundlich (1906) showed that the Henry law which predicts a linear behavior between uptake and volume or concentration for initial sorption, and which results from classical thermodynamic, was rarely obeyed and introduced in the law an exponent which he related to the heterogeneity of the surface. This is one of the first apparitions, in that field, of the concept of fractality used nowadays in chemical and physico-chemical reactions (Brouers, 2014a; Brouers and Sotolongo-Costa, 2003, 2005, 2006; Brouers et al., 2005; Jurlewicz and Weron, 1999; Kopelman, 1988; Savageau, 1995; Stanislavsky and Weron, 2013). Langmuir (1918) using the law of mass action derived his famous isotherm for surface supposed to have a unique sorption energy. Since this is a situation rarely encountered, a number of generalizations, most of them purely empirical, were introduced in the literature. In the case of ultra-porous activated carbon, Dubinin introduced a semi-empirical isotherm which has various variants and, using some correlations with geometric observations and statistical hypothesis, related this isotherm to the pore distribution.
Ten years ago (Brouers et al., 2005), in order to give a more fundamental basis to the isotherm models and following the work of Zeldovitch (1934), we show that the Freundlich exponent was related to the value and the distribution of energies contributing to the sorption, that the power law of the Freundlich isotherm could be obtained by assuming that the scale T-dependent b coefficient of the Langmuir isotherm (equation (4)) was distributed as a heavy-tail Lévy distribution. In that way, the sorption mechanism was viewed as a birth–death (sorption–desorption) mechanism dominated by the highest values of the random sorption energies. We showed that the exponent of the Freundlich law which appears in generalizations of the Langmuir (under the generic name “Freundlich–Langmuir isotherms”) was related to the average sorption energy and the width of its distribution. We proposed, as a consequence of the discussion, to use a Weibull function as a realistic isotherm which since has been named Brouers–Sotolongo (BS) isotherm and has been used with some success, for example, in porous activated carbon and carbon nanotubes (Altenor et al., 2009; Deliyanni and Lazaridis, 2014; Hamissa et al., 2007; Kyzas et al., 2014; Ncibi and Sillanpää, 2015; Njoku et al., 2014), biosorption (Brouers and Al-Musawi, 2015; Figaro et al., 2009), water treatment (Figaro et al., 2009; Podder and Majumder, 2015; Sobhanardakani and Zandipak, 2015), bacteriology (Naidoo et al., 2012; Singh et al., 2013), and geology studies (Masjkur, 2012).
Recently, extending the scope of that paper (Brouers, 2014b), we demonstrated that some of the commonly used isotherms belong to the family of a generalized Brouers–Sotolongo (GBS) isotherm obtained by replacing the exponential in the Weibull function by a deformed exponential, a function now extensively used in econometry, ecology, hydrology, and many other complex systems. This function is known in the literature as BurrXII–Singh–Madalla (Burr, 1942; Singh and Maddala, 1976) or q-Weibull. It is a natural extension to natural and physico-chemical systems of the classical concept of exponential and appears naturally in attempts to generalize the classical thermodynamic to complex systems (Brouers, 2013, 2015; Tsallis, 1988). In this paper, we want to demonstrate that the Dubinin isotherms belong asymptotically to the same family and analyze the various approximations using data on benzene taken from one of the original Dubinin papers and analyzing in the same manner the data published in the PhD thesis of one of us.
The generalized Brouers–Sotolongo isotherm
In Brouers (2014b), based on statistical and mathematical considerations, we introduce the GBS isotherm:
As shown in detail in Brouers (2014b), some of the most popular empirical and semi-empirical isotherms can be derived simply from the GBS isotherm, and others are purely empirical such as the Freundlich (1906), the Redlich–Peterson (1959), and the Generalized Freundlich isotherms (Sips, 1950). They are not correct asymptotically for large values of x, and the last two have a Henry behavior which is not always observed. In our opinion, these isotherms should be used with care, especially when the data are partial and do not reach saturation.
For c = 0, one gets the BS isotherm:
For c = 0 and a = 1, one gets the Jovanović isotherm (Jovanović, 1969).
This isotherm has arisen from a discussion of the effect of a Lévy distribution of the Langmuir scaling parameter due to an assumed exponential adsorption energy distribution in a heterogeneous model (Brouers et al., 2005). It is similar to the Freundlich–Jovanović isotherm Quiñones and Guiochon, 1996, which has been obtained by assuming that the rate of decrease of the fraction of the surface unoccupied by the adsorbate molecules is proportional to a certain power of the partial pressure of the adsorbate (Quiñones and Guiochon, 1996).
For c = 1, one gets the Sips–Hill (SH) isotherm:
For c = 1, a = 1, one recovers the Langmuir isotherm:
As it is well known, the three isotherms (equations (1) to (3)), when
where W and a are empirical parameters.
The Dubinin isotherm is a modified form of the Freundlich isotherm
In a previous paper (Brouers et al., 2005), we have shown, assuming a Lévy distribution for the scale parameter b of the Langmuir isotherm resulting from the energy distribution of a heterogeneous Langmuir model, that the Freundlich exponent a (1/nF) has a physical meaning and is related to the distribution of adsorption energies. These results allow us to write the Freundlich isotherm as
We can transform equation (6) as follows:
If
The Dubinin equation has been generalized to
Equation (11) is known as Dubinin–Astakhov isotherm (Stoeckli et al., 1989).
When n = 2,
Equation (12) is called Dubinin–Radushkevich isotherm. It is the one used by Dubinin (1989) for the case of benzene and in the Marquez thesis.
As discussed in Dubinin (1989), the relation between the argument of the Dubinin exponential and the characteristic adsorption energy is based on the Eucken–Polanyi potential theory.
With this formula, Dubinin has established a series of relations and correlations with the microscopic geometric and energetic distribution of the micro-pores and meso-pores of the sorbent.
The comparison between equations (8) and (11) shows that the empirical Freundlich isotherm (for partially soluble adsorbate) can be viewed as a special case of the Dubinin–Astakhov isotherm. Therefore, they can be related to the family of GBS isotherms (equation (1)).
Comparison of isotherms in the case of benzene
In the vast literature on sorption, two groups of theories have emerged. One mostly influenced by Soviet scientists’ works using the Dubinin formalism essentially for activated carbons, and the other using Langmuir theory and its extensions. Since nowadays efficient nonlinear regression numerical programs exist in the market, a comparison of these theories, which, as this has been demonstrated in the previous sections, are not unrelated, is in order. We present now such comparison for original data of Dubinin (Table 4 of Dubinin, 1989) on the adsorption of vapor of benzene onto an activated carbon presenting two types of porosities ultra-micro and micro porosity.
In that paper, Dubinin extended his original formula to treat simultaneously the two types of porosity (D2) (micropores and ultramicropores):
The values chosen to fit the experimental data were (D1989):
W01 = 0.2 cm3/g, W02 = 0.3 cm3/g, E0D1 = 25 kJ/mol, E0D2 = 12.5 kJ/mol, corresponding to αD1 = 0.1, αD2 = 0.2.
We will use the new capabilities of nonlinear fitting methods and we will consider the parameters of equation (13) as free parameters (D2) which will be determined by a nonlinear regression method in order to compare the two families of isotherms. For comparison, we have used extensions of the SH and BS isotherms to the double porosity Dubinin–Radushkevich model.
The results of the numerical calculations show that in this instance as in many others, due to the difficulty to determine with precision the coefficient c for sorption data, which are unique sets with a limited number of experimental points, there is practically no difference between SH (c = 1) and BS (c = 0) isotherms. In that case, the recommended practice (Brouers and Al-Musawi, 2015) is to use an intermediate expression, the so-called Brouers–Gaspard (BG) isotherm (c = 0.5).
It has all the properties of a genuine statistical function with the right asymptotic properties. The choice of this kinetics is useful when it is not possible to discriminate between BS and SH isotherms. We have found that it is the case here and we have extended equation (14) to the two-porosity model (BG2).
Figure 1 and Table 1 summarize the results of the computation using the data of Table 4 of Dubinin (1989).
Note: The difference between calculated values and experimental data is for both less than 1%, except for the first and last point of BG2 which is 4% and the last point of D2 which is 2%. The computation confirms the approximate values of m and n chosen by Dubinin.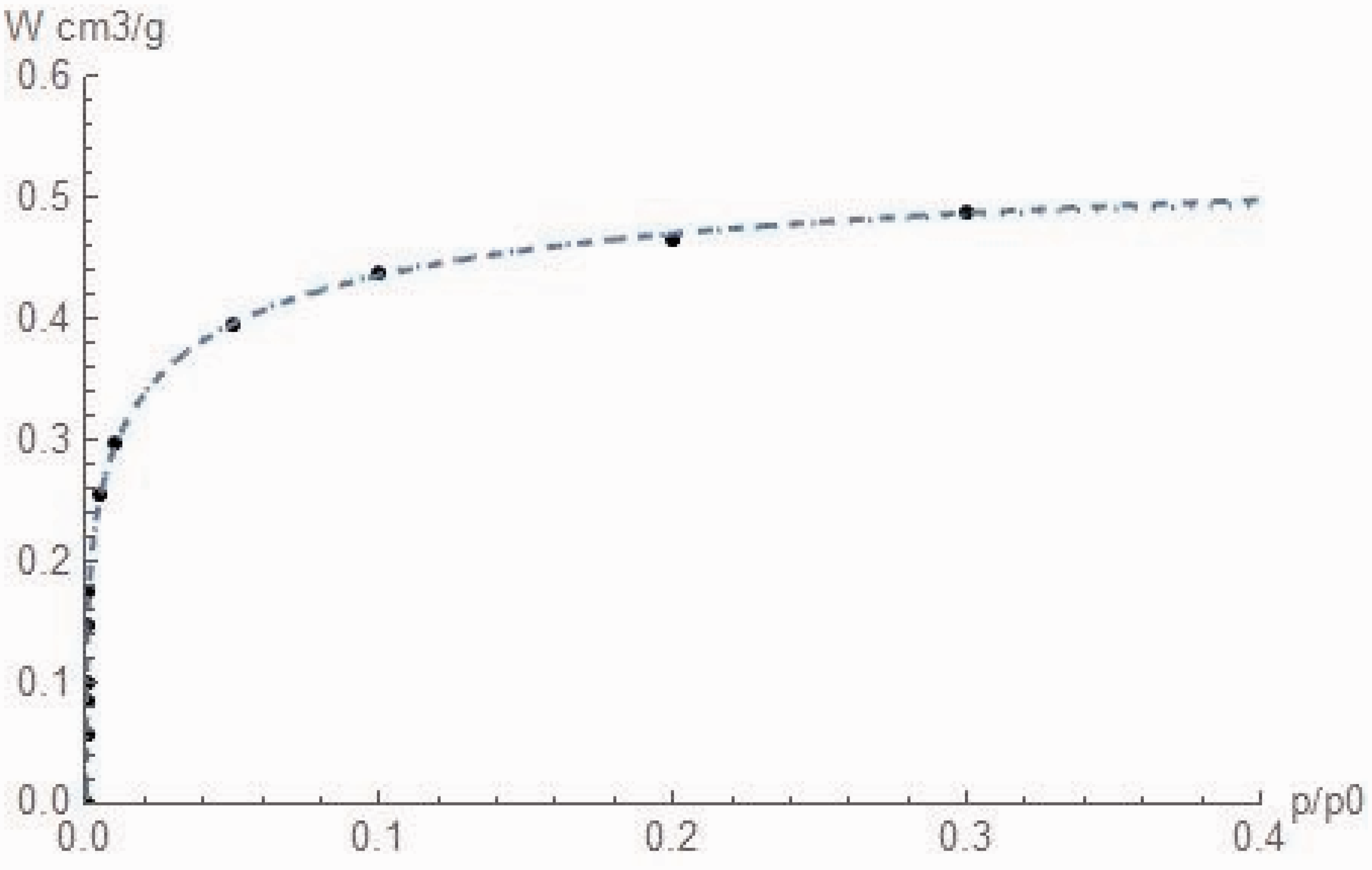
In Figure 1, we present the double porosity Dubinin–Radushkevich isotherm (equation (13)) (dotted) and double porosity BG (equation (15)) isotherm (dashed). They cannot be distinguished and both perfectly fit the data.
In Table 1, we report the results for the two-porosity models. As this can be observed, there is no significant difference between the two approaches (B2 and BG2) and the Dubinin original (D1989) assumed values of the coefficients m and n.
The energies obtained are different: in the Dubinin formalism, it is the characteristic adsorption energy. In the BG formalism, the energy is related to the averaged sorption energy depending on the adsorption energy distribution (Brouers et al., 2005).
Analysis of the Marquez thesis data
We will now analyze with isotherms (equations (2) to (5) and (11)) and discuss the complete set of data from one of us (Marquez-Montesino, 2000; Marquez-Montesino et al., 2001) on powered activated carbon from Pinus caribea saw dust with the use of
Here again, it is difficult to make a distinction between BS and SH isotherms (some are better fitted with BS, others with SH) and to construct the table and observe the correlation between the isotherms’ coefficients and physical characteristics we have used in the intermediate (c = 0.5) BG isotherm (equation (14)) as discussed in the section “Comparison of isotherms in the case of benzene”. The results obtained with that isotherm are compared with the physical and isotherm data reported in the work of Marquez-Montesino (2000) who had used the Dubinin–Radushkevich isotherm. The results reported here show that there is practically no difference between the two methods. The only difference arises from the different interpretation of the obtained energy: the adsorption energy in Dubinin’s approach and an estimation of the average adsorption energy in the second approach. The results are summarized in Figure 2 and Tables 2 to 4. In order to find some useful correlations, the isotherms’ parameters are presented with the corresponding physical data (degree of activation, BET surface, porosity) taken directly from Marquez-Montesino (2000) and Marquez-Montesino et al. (2001). To illustrate the results graphically, we have chosen the isotherm with the lowest coefficient of determination R2 (Figure 2).
Fit with the Dubinin isotherm (black), the BG isotherm (dashed), and the Freunclich isotherm (gray) corresponding CA S72547 (R2 = 0.99997). They practically coinicide with the data. All other fittings have a higher coefficient of determination R2 and are even better. BG: Brouers–Gaspard. Results of BG and Dubinin–Radushkevich (as in Brouers and AL-Musawi (2015) isotherms analysis of carbon activated with water vapor). Source: Data from Marquez-Montesino (2000) and Marquez-Montesino et al. (2001) and parameters from equation (14). BG: Brouers–Gaspard; C.A.: Activated Carbon.

The fitting in this paper which are performed with the “mathematica” nonlinear fitting program gives the following complete set of results.
Results of BGand Dubinin–Radushkevich isotherm analysis of carbon activated with CO2.
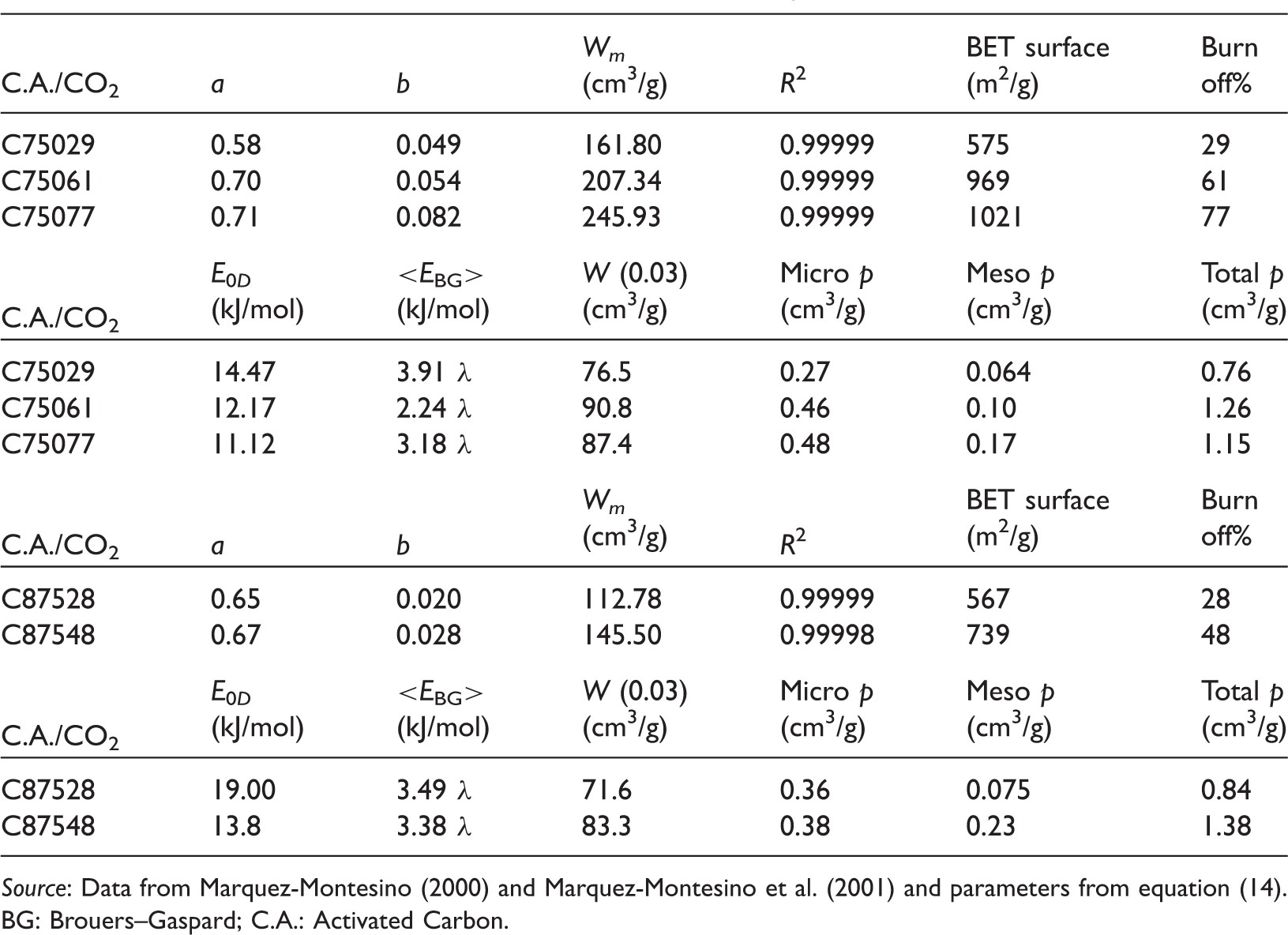
Source: Data from Marquez-Montesino (2000) and Marquez-Montesino et al. (2001) and parameters from equation (14).
BG: Brouers–Gaspard; C.A.: Activated Carbon.
Comparison of the precision of the isotherms discussed in this paper for the data CA S72547.

BG: Brouers–Gaspard; SH: Sips–Hill.
In order to be complete and give a better picture of the performances of the isotherms discussed in this paper, in Table 4, we give the corresponding values of the coefficient of determination parameter R2 for the whole curve as well as in Figure 3 the fit for very low relative pressure (up to 0.005) for the case CA 572547.
Sips–Hill (dashed) and Dubinin–Astakhov (dot-dashed) isotherms and Langmuir (dotted) and Freundlich (full) isotherms for low relative pressure corresponding to data of Figure 2. The Sips–Hill (with exponent a = 0.68) and the Dubinin–Astakhov isotherms almost coincide lying between the traditional Freundlich (with exponent a = 0.48) and Langmuir (obeying Henry law) isotherms.
Conclusions
The results of our paper show that the Brouers-Sotolongo and the Sips-Hill isotherms which are among the many semi-empirical formulas used in the literature and which are the only ones having the good asymptotic behavior for small and high values of concentration or pressure and genuine statistical distribution properties (Brouers, 2014) give almost undistinguishable results. That important conclusion has been observed in many instances where the isotherm is measured until saturation. The suggestion has been made in practice to use an intermediate isotherm (BG isotherm introduced in Brouers and Al-Musawi, 2015) by giving the intermediate value half to the complexity parameter c in equation (1). It is with this isotherm that we have treated the set data of Dubinin (1989), Marquez-Montesino (2000), and Marquez-Montesino et al. (2001). We get very close results if we use both Dubinin and BS formalisms. The macroscopic parameters derived from these isotherms are complementary and can be used to characterize and compare the physical, physico-chemical, and morphological properties of porous activated carbons.
To conclude, we suggest that in usual practice, it would be sufficient to restrict oneself to the Dubinin–Astakhov (equation (11)) SH, or BS (equation (14)), the two extremes of the GBS isotherm (equation (1)). If in one series of data it is not possible to choose for comparison, between these two last isotherms, practically, as this is done in this paper, one can choose the intermediate BG isotherm. Nonlinear regression methods are a must. The comparison with a great number of empirical isotherms and the use of imprecise linearization methods has become obsolete. We must also remember that the isotherms can give only macroscopic information and the correlation of the isotherms parameters with the micro- or mesopore geometry requires, to be complete, independent methods like micrography and other physical methods. The statistics of isotherms are not very precise: generally, one makes a unique measurement for every choice of physical parameters (T, pH, concentration,…), and the measurements depend on the experimental conditions (Douven et al. 2015) and are the result of a multi-scale averages.
Footnotes
Acknowledgements
We are grateful to Professors Sara Gaspard (Guadeloupe, France), Mongi Seffen (Sousse, Tunisie), and Al-Musawi (Baghdad, Iraq) for useful discussions and to Prof JP Pirard (Liège, Belgique) for introducing us to this field. We thank warmly the anonymous referee for his careful reading of the manuscript and his numerous suggestions to improve the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interests with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.