Abstract
The metal–organic frameworks material functionalized by grafting amino group of tetraethylenepentamine on the coordinative unsaturated Cr (III) centers is described. The obtained tetraethylenepentamine-functionalized adsorbents with different mass ratios of tetraethylenepentamine have been characterized by scanning electron microscopy, Fourier-transform infrared, X-ray powder diffraction, and N2 adsorption and desorption isotherms. Significantly, MIL-101-tetraethylenepentamine 60% exhibited high adsorption capacity (350 mg/g) for removal of uranium (VI) from water at pH 4.5. At uranium concentration <20 mg/L, the isothermal plot was best represented by Freundlich model. At U(VI) concentration approximately >30 mg/L, the isotherm was best described by Langmuir model.
Introduction
With the rapid development of industry, more and more waste is polluting the environment and the natural resources. Therefore with the development of low carbon economy policy, nuclear power is paid much attention to nowadays. In the course of generation of nuclear power large amount of nuclear waste is produced, which can contaminate waste water with uranium. Uranium is an important nuclear fuel and its wide application has important significance to the development of nuclear industry (Chen et al., 2011). But in the development of nuclear energy, it also produces large amount of radioactive substances containing uranium in the waste water. If the waste water without treatment is directly discharged, it will seriously pollute the environment and also lead to serious threat to the human health. So purification of industrial waste water such as nuclear mill effluents is becoming the focusing issue (Gao and Gao, 2014; Xia et al., 2008). In order to increase the utilization rate of resources and reduce the harm of radioactive waste water, uranium separation, and enrichment should be carried out as an important research project. Moreover, thus extraction of uranium from uranium bearing waste water can also improve the utilization of uranium resources and reduce the gap between supply and demand.
Various technology have been developed to extract uranium from aqueous solutions, such as chemical coprecipitation (Bertrand et al., 2012; Mehta et al., 2014), membrane filtration (Hoyer et al., 2014; Zhou et al., 2014), ion exchange (Manos and Kanatzidis, 2012), solvent extraction (Bartosiewicz et al., 2015), magnetic separation and adsorption (Gao et al., 2014; Liu et al., 2013; Wang et al., 2012), involved in the treatment for low concentration uranium or sea water (Tang et al., 2003; Wei and Xu, 2007; Yusan and Erenturk, 2010; Zhang et al., 2003). It is noteworthy that adsorption is widely considered as not only an effective and convenient method but also an environment-friendly technology for removal of uranium from waste water containing low concentration of uranium.
A number of studies have indicated that bacteria, fungi, algae, polymeric resins, activated carbon, clay, zeolites, silica gel, and inorganic adsorption materials have certain enrichment effects on uranium or heavy metal ions (Liu and Zhou, 2010; Naushad et al., 2016, 2017; Samin et al., 2015; Sharma et al., 2017a, 2017b; Zou et al., 2010). Therefore, the achievement of high adsorption capacity, and good selectivity will be the focus of the research. It is necessary to explore some adsorbents with great potential for quick purification, less pollution, easy recycling, and recovery of uranium. Natural polymers possess structure with a variety of functional groups such as amino and carboxyl (Raize et al., 2004). Carbon materials and composite materials have the advantages of large specific surface area, abundant porosity, good thermal stability, and good chemical stability (Choi and Jang, 2008; Deng and Bai, 2004; Lam et al., 2006; Saha et al., 2008). In particular, polyamines have been used to functionalize silica (Yoshitake et al., 2005), cellulose (Qu et al., 2005), polystyrene (Torres et al., 2006), polyacrylonitrile fiber (Deng et al., 2003), and polymers (Atia et al., 2007; Liu et al., 2008) to prepare adsorbents for removing heavy metal ions from aqueous solutions. These above materials used as adsorbents, catalyst carriers, and composite matrixes, can also be used to deal with radioactive waste water. Among the various studies, amine-functionalized adsorbents are perhaps the most common, arising from the strong chelation properties of amine to transition metals and actinides. In particular, adsorbents with nitrogen-containing functional groups have been widely explored because these functional groups, such as ethylenediamine (EDA), diethylenetriamine (DETA), triethylenetetramine (TETA), and tetraethylenepentamine (TEPA) have been found to be most effective for removal by adsorption and other ions (Liu et al., 2010).
Metal–organic frameworks (MOFs) are a novel class of hybrid inorganic–organic materials and a chromium terephthalate-based solid with unusually large pore volumes and surface area has been synthesized named MIL-101(Cr) (Ferey et al. 2005). The porous structure and specific surface area of the ambassador have unique properties. MOFs possess porous adjustable structure, large free volume, high surface areas, and large unsaturated metal ions inside. Capture of uranium from aqueous solution using MOFs was first reported with MOF-76 (Yang et al., 2013) and UiO-68 (Carboni et al., 2013). During the past few years, various MOF materials have been developed as an adsorbent for uranium (Carboni et al., 2013; Falaise et al., 2017; Feng et al., 2013; Ferey et al., 2005; Liu et al., 2010; Yang et al., 2013) or other highly toxic metal ions (Alqadami et al., 2017; Chen et al., 2017; Liu et al., 2017; Zheng et al., 2017; Li et al., 2017; Zhu et al., 2017; Sheng et al., 2017). Amino groups have a strong coordination effect with metal ions and exhibit good adsorption effects for uranium (Sert and Eral, 2010). Introducing of amino groups into the MOF for uranium adsorption has been reported and studied in details. NH2, Ethanediamine (ED), and DETA were grafted into MIL-101 by post-synthetic method (Bai et al., 2015) and show efficient uranium adsorption displaying with the percent adsorption increasing with increase pH value. The adsorption capacity of these MOFs for uranium adsorption follows with the order of MIL-101-DETA > MIL-101-ED > MIL-101-NH2 > MIL-101. Specially, when the grafted ED/Cr ratio is 0.68, the MIL-101-ED exhibits the maximum adsorption capacity at pH of 4.5 (Zhang et al., 2015). However, uranium adsorption can be enhanced by introduction of amino group into organic ligand, probably due to the lower activity of aromatic amines, decrease of surface area and formation of intermolecular hydrogen bonds (Luo et al., 2016). Acylamide and carboxyl functionalized MOF have also been reported (Wang et al., 2015). As pored wall functionalized by both carboxyl groups and acylamide groups show good adsorption properties for uranium from waste water, it has been succeeded in introducing TEPA with an amino group has been introduced into MIL-101(Cr) and was first synthesized by G Ferey (Hwang et al., 2008). MIL-101(Cr) MOF functionalized with DETA (DETA-MIL-101) possessed the highest adsorption capacity of 350 mg⋅g−1 at pH of 5.5 for U (VI) adsorption (Bai et al., 2015). While MIL-101(Cr) MOF functionalized with TEPA (TEPA-MIL-101) has been reported for CO2 capture (Huang et al., 2016) and as a highly selective adsorbent for CO2 over CO (Wang et al., 2012), but in this work we used TEPA-MIL-101 as an effective adsorbent for low concentration of uranium. The polyporous MIL-101(Cr) has very large surface areas and numerous potential unsaturated chromium sites (up to 3.0 mmol g−1). After vacuum treatment at 423 K for 12 h, terminal water molecules were removed from the framework, thus providing the coordinatively unsaturated metal centers (CUS) as anchoring points for the chosen amine ligands in the structure usable for the surface functionalization (Hwang et al., 2008).
In the present investigation, TEPA is introduced into this MOF as amino group has strong adsorption property to uranyl ions. Hence the adsorption capacity of the material can be greatly improved when a large amount of amino has been introduced. The chemical properties and morphology of the TEPA-MIL-101s are analyzed by scanning electron microscopy, Fourier transform infrared (FT-IR), powder X-ray diffraction (PXRD), BET surface areas (BET), and N2 adsorption and desorption isothermal, respectively. Batch adsorption experiments are carried out to study the effect on adsorption of uranium of various experimental parameters, such as solution pH, contact time, temperature, and initial uranium concentration. The adsorption kinetics, isotherm models, and thermodynamics are also evaluated to understand the adsorption process.
Experimental
Reagents
Terephthalic acid (H2BDC), chromium nitrate, anhydrous sodium acetate, N,N′-dimethylformamide (DMF), anhydrous ethanol, and anhydrous toluene were from Beijing chemical plant. TEPA was from Sinopharm chemical reagent. All the chemical reagents used in this study were of analytical grade, and without further purification. U (VI) stock solution (Hangzhou Daji Photoelectric Instrument Co. Ltd.) was prepared by dissolving appropriate amounts of uranyl nitrate hexahydrate [UO2 (NO3)2·6H2O] in deionized water. Freshly deionized and distilled water was used as the dispersion medium.
Instrumentations
Surface morphological images were characterized by SEM (HITACHI Hitachi S-4800, Japan) after gold plating at an accelerating voltage of 10 kV. The complexation among adsorbents was confirmed by FTIR (VERTEX70, Bruker company). TEPA-MIL-101 and unfunctionalized MIL-101 were pelletized using KBr prior to FTIR measurement. Surface area of sorbent was determined from N2 adsorption isotherms at 77 K with BET method using a Micromeritics ASAP 2010 volumetric analyzer. Prior to the surface area measurement, TEPA-MIL-101s and unfunctionalized MIL-101 were outgassed at 150°C or 120°C for 2 h under a reduced pressure of 10−5 Torr, respectively. XRD analysis was carried out on a D8 Advance instrument (Bruker company) with Cu Kα radiation source (λ = 1.5406 Å). The operating conditions were at scan rate of 3°/min from 1° to 10° to obtain the XRD pattern. The uranyl ion concentration was determined by trace uranium analyzer (WGJ-III, Institute of comparative geology of nuclear industry).
Synthesis
Synthesis of MIL-101
MIL-101 MOF was synthesized according to a procedure reported previously (Guo et al., 2012). Specifically, 3.2 g of Cr(NO3)3·9H2O, 1.33 g of 1,4-benzenedicarboxylic acid (H2BDC), 0.164 g sodium acetate anhydrous were dissolved in 40 mL H2O. They were mixed by ultrasonification and transferred to a teflon lined stainless steel autoclave and then heated to 473 K in an oven. After 12 h, the mixture was cooled down naturally, the crude MIL-101 was separated by centrifugation (10,000 r/min) and washed with adequate water. The solid sample was dried at 353 K. Then the green powder was soaked in DMF and ultrasonicated for 30 min at 40% power to remove the unreacted H2BDC. The filter was dried finally overnight in an oven at 353 K.
Synthesis of TEPA-MIL-101
MIL-101 was outgassed at 423 K under high vacuum for 12 h to remove water and generate the coordinatively unsaturated sites. 0.5 g dehydrated MIL-101 was soaked in 50 mL anhydrous toluene solution. To this suspension, an appropriate amounts of TEPA solution (≥98%) (50 μL, 150 μL, and 300 μL, respectively) were added respectively and the mixture was stirred with heating to reflux at 393 K for 12 h to obtain the samples as MIL-101-TEPA 10%, MIL-101-TEPA 30%, and MIL-101-TEPA 60%. The product was recovered by filtration and washed with ethanol, and then dried at 343 K for 12 h.
Batch adsorption
Experiments with TEPA functionalized MIL-101(Cr) were carried out such as variation of pH, initial concentration, contact time, temperature, and their effect on U(VI) adsorption was studied in detail.
The solution pH was adjusted from 2 to 5 by using dilute HCl or NaOH. It was performed in 10 mL centrifuge tubes with a working volume of 1 mL, and 5 mg of the adsorbents were placed in centrifuge tubes and mixed with 9 mL of 100 ppm of uranyl solution. The effect of initial uranyl ion concentration was performed using 9 mL of different concentrations from 5 ppm to 300 ppm and 5 mg of adsorbents at the pH value of 4.5. The reaction time and temperature of those two adsorption experiments were at 298 K for 2 h. The effect of temperature and contact time was conducted according to the following procedure. Five milligrams of the MIL-101-TEPA 60% was placed in centrifuge tube and mixed with 9 mL of 100 ppm of uranyl solution using the pH value of 4.5. This experiment contact time ranged from 5 min to 120 min at three different temperatures following with 298 K, 308 K, and 318 K, respectively. Then the adsorbents were separated by centrifugation at 3000 r/min for 5 min. The uranyl ion concentration in the supernatant was determined by trace uranium analyzer.
The adsorptive capacity (Qe) and the removal percentage (E) were calculated as follows in equations (1) and (2).
Results and discussion
Characterization
The adsorbents are analyzed by scanning electron microscopy, as shown in Figure 1. The morphology of the MIL-101(Cr) MOF in Figure 1(a) shows the granular crystal with a particle size of about 200 nm, which differs from previously reported MIL-101 crystals synthesized by Bai et al. (2015) using hydrofluoric acid (HF) as mineralizer. The experiment uses sodium acetate as the mineralizer for the synthesis of MIL-101 in order to promote the solubility of terephthalic acid and to improve the yield and also simplify the subsequent purification procedures. The SEM images of MIL-101-TEPA 60% still kept its granular crystal structure (Figure 1(b)). The addition of TEPA did not affect crystal morphology, and the size of the granular crystal was still at about 200 nm. Because the size of the synthesized crystal is small and the crystals are agglomerated, it is difficult to see the obvious difference. But as the amount of TEPA increases, the color of the sample decreases from dark green to light green.

SEM micrograph of MIL-101 and MIL-101-TEPA 60%.
It shows the XRD patterns of those four samples in the range of 2–25° in Figure 2(a). According to the XRD, the characteristic peaks of MIL-101 can be clearly observed, indicates the successful synthesis of the material. Although the addition of TEPA has some effect on the symmetry of the crystal, it has not destroyed the framework of the MIL-101. With the increase of the amount of TEPA added, the position of characteristic peaks is not changed, but with some slight decreases of peak intensities and color changes from green to gray as shown in Figure 2(b). The XRD patterns of MIL-101-TEPA60% before and after uranium adsorption indicate that the structure was maintained in this process, but the intensity of the peaks become weaker. These results may have resulted from U(VI) ions occupation of the windows of MIL-101-TEPA60%.

(a) XRD of MIL-101, TEPA-MIL-101s, and U(VI)-adsorbed MIL-101-TEPA60%. (b) Pictures of MIL-101 and TEPA-MIL-101s.
The FT-IR spectra of the samples are shown in Figure 3. For all the TEPA-grafted MIL-101, the NH2 stretching vibration appears in the range of 3200–3500 cm−1 (Bernt et al., 2011; Huang et al., 2008, 2016), which confirms the presence of amino groups. The band at 1400 cm−1 and 1500 cm−1 implies respectively the presence of dicarboxylate within the MIL-101. It can be seen the band at 657 cm−1 and 1612 cm−1 is attributed to N–H (Wang et al., 2012), and the stretching vibration of C–N group can be seen at the band of 1027cm−1. The absorption band of –COOH in MIL-101 can be seen at 1712 cm−1, which is due to C=O in the free –COOH. While MIL-101-TEPA 10%, MIL-101-TEPA 30%, and MIL-101-TEPA 60% do not have this characteristic absorption band because the introduced amino groups made it neutralized. With the addition of TEPA, the peak intensity increases at about 1600 cm−1 which is attributed to N–H. While the absorption peak of –COOH at around 1720 cm−1 (Aydincak et al., 2012; Ryu et al., 2010) decreases, indicating that the reaction occurred between the acid and TEPA.
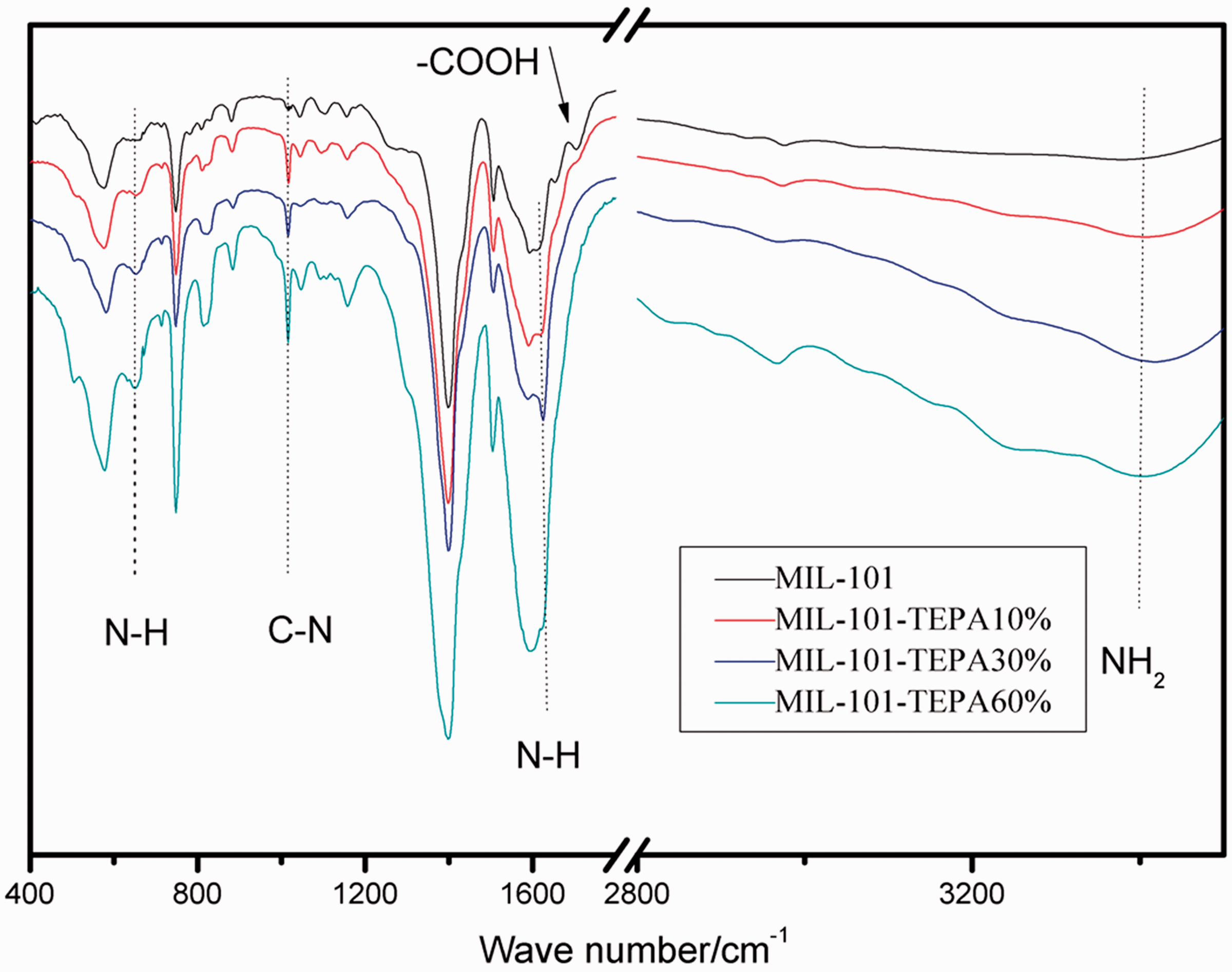
FT-IR spectra of MIL-101 and TEPA-MIL-101s.
The BET of unmodified MIL-101 was measured as 2310 m2/g, and the pore volume was 2.467 cm3/g. The BET and pore volume of TEPA-MIL-101 decreased with the amino grafting into MIL-101, as shown in Table 1, because the introduction of amino group took up the pores of the framework and hinder the diffusion and adsorption of N2.
Adsorption model
A schematic diagram showing the MIL-101-TEPA structure and formation prior to the metal ion adsorption would be contributed to understanding the capture process of U(VI) into the TEPA functionalized MIL-101. The evolution of coordinatively unsaturated sites was from chromium trimers in mesoporous cages of MIL-101 after vacuum treatment at 423 K as shown in Scheme 1. The adsorption sites were changed from the original coordinatively unsaturated chromium trimers in mesoporous cages of MIL-101 into bonded TEPA sites, which lead to more U(VI) ions adsorption in MIL-101-TEPA.

Schematic diagram for capture of U(VI) into the TEPA functionalized MIL-101. (A and B) Evolution of coordinatively unsaturated sites from chromium trimers in mesoporous cages of MIL-101 after vacuum treatment at 423K. (C) MIL-101 functionalized with TEPA. (D and E): U(VI) adsorption model.
Effect of pH on U (VI) adsorption
The pH value of the solution is very important to the metal ions adsorption ability. Firstly, it can change the charge of adsorbent surface. Secondly, it influences the existence form of metal ions. UO22+ was the mainly species in acidic pH from 2.0 to 5.0, with the increase of pH it might be transformed into multi-nuclear hydroxide complexes. As shown in Figure 4, U (VI) absorption test was performed at pH 2–5. The adsorption capacity of MIL-101-TEPA 10%, MIL-101-TEPA 30%, and MIL-101-TEPA 60% increased with increased pH values, while the amino unmodified MIL-101 did not change. Because a large amount of H+ is present in the solution at low pH values, which will occupy the lone pair electrons on N and compete the adsorption site with uranyl ions. In high pH value conditions, UO22+ will hydrolyze to produce polynuclear hydroxides, and the Ksp of precipitation UO2(OH)2 is 1.1 × 10−22, so the pH was chosen to be 4.5 in the next experiment. What needs to be pointed out here is that unfunctionalized MIL-101 adsorption property shows slightly higher than the amino functionalized at the very low pH value, because the introduction of TEPA into MIL-101 can reduce the surface area and pore volume of the materials.

Effect of pH on U(VI) adsorption of MIL-101 and TEPA-MIL-101s. (Adsorption dosage 5 mg, C0 = 100 mg/L, agitation time t = 2 h, T = 25°C and pH = 2–5).
Effect of initial uranium concentration on U (VI) adsorption
As can be seen from Figure 5, the adsorption capacity of U(VI) in TEPA functionalized MIL-101 increases with the initial uranium concentration. When the initial U(VI) concentration is low, more amino groups can be coordinated with the uranyl ions, and up to saturation point as the concentration increases gradually. Moreover, at low concentration, the adsorption rate of U(VI) in amino functionalized adsorbent is extremely high up to 99%, and the adsorption rate decreases with the increase of uranium concentration. For the MIL-101-TEPA 60%, the adsorption capacity of U(VI) during the equilibrium state is as high as 350 mg/g because of a large number of amino groups be introduced into MIL-101. As unsaturated Cr (III) center is limited, no more amino groups could be introduced into MIL-101, as shown in Table 5.
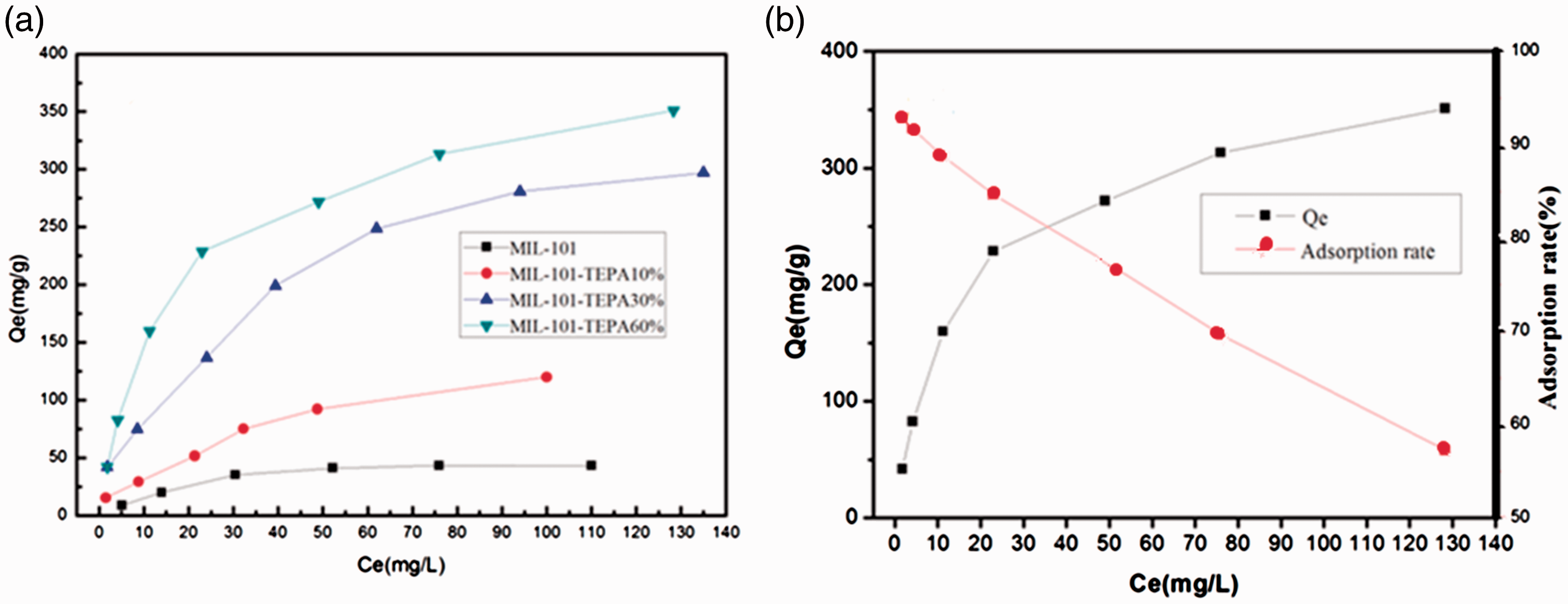
(a) Isotherms of U(VI) adsorption of MIL-101 and TEPA-MIL-101s (adsorption dosage 5 mg, agitation time t = 2 h, T = 25°C, and pH = 4.5). (b) Effect of initial U(VI) adsorption of MIL-101-TEPA60% (adsorption dosage 5 mg, agitation time t = 2 h, T = 25°C, and pH = 4.5).
Effect of temperature and adsorption time on U (VI) adsorption
Figure 6 is the adsorption curve of the MIL-101-TEPA 60% at the temperature of 298 K, 308 K, 318 K from 2 minutes to 120 minutes. As shown in the diagram, MIL-101-TEPA 60% reaches adsorption equilibrium in a relatively short time about 30 minutes. As temperature has a certain effect on ion diffusion, the adsorption capacity of the material is affected by the temperature. The adsorption capacity increases with the raising of the temperatures, which is due to the quick facile diffusion in the pores of the metal ions with the TEPA introduced at high temperature.
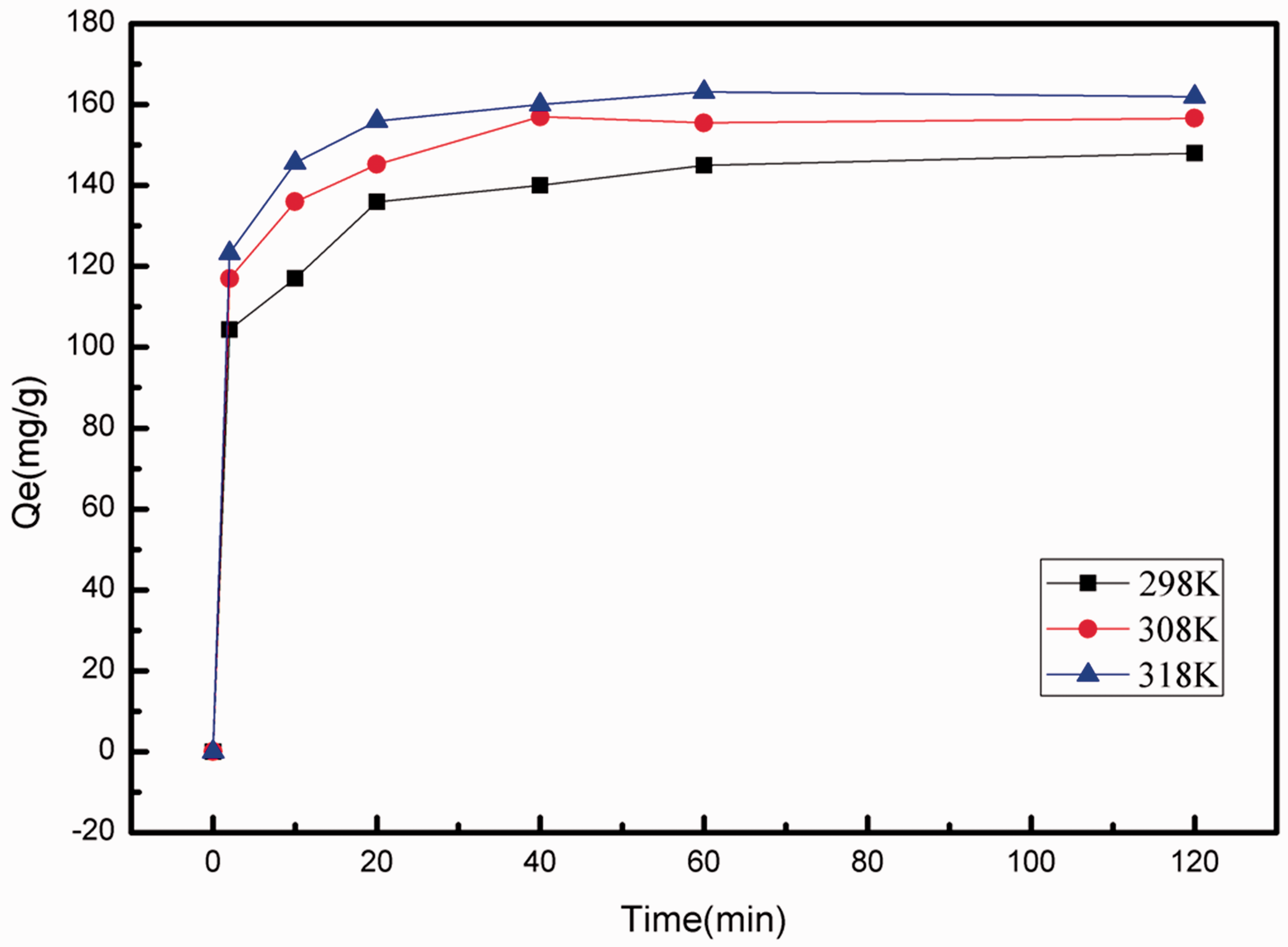
Effect of contact time for U(VI) adsorption of MIL-101-TEPA60% at different temperature (adsorption dosage 5 mg, C0 = 100 mg/L, agitation time t = 0–2 h and pH = 4.5).
Desorption experiment
The desorption rate of the MIL-101-TEPA 60% in different desorption solutions in Figure 7. The desorption rate of the material is very low in water, which is about 10%. While in the 0.1 mol hydrochloric acid and nitric acid, the desorption rate is up to 70% as the material is not dissolved in acidic solution. Sodium carbonate as a type of desorption solution, also displays of a high desorption level, because U(VI) form soluble anionic carbonate species like UO2(CO3)22−, UO2(CO3)34− etc.

Elution efficiency of different elution solutions for MIL-101-TEPA60%.
Adsorption isotherms and adsorption kinetics
As shown in Figure 8 is the kinetic adsorption of U(VI) by MIL-101-TEPA 60%. Adsorption kinetics is very important to evaluate the adsorption efficiency of an adsorbent. The pseudo-second kinetic model (Kuang et al., 2013; Ho, 2006) could be represented by equation (3), and the related data is shown in Table 2.

Pseudo-second order plot for the removal of U(VI) by MIL-101-TEPA60%.
BET and pore volume of MIL-101 and TEPA-MIL-101s.

Kinetic parameters for adsorption of U (VI) by the MIL-101-TEPA 60% (adsorption dosage 5mg, C0 = 100 mg/L and pH = 4.5).
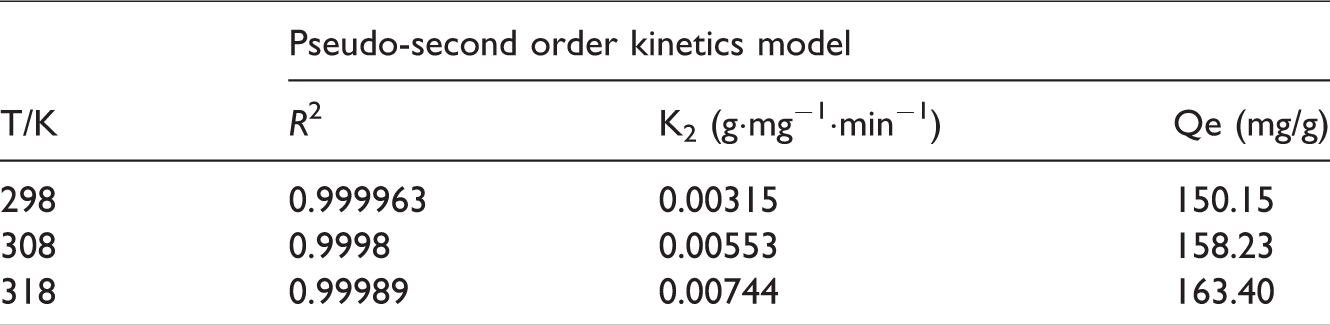
Qe and Qt are the amount of adsorbent at equilibrium and time respectively. k2 is the rate constant for the pseudo-second-order kinetics. According to Table 2, it can be seen that with the increase in temperature, K2 and Qe values simultaneously increase hence the temperature affects the reaction rate and maximum U(VI) adsorption capacity.
The adsorption of U (VI) is determined as a function of equilibrium U (VI) concentration (Ce). With the increase of initial concentration of U (VI), the adsorption capacity of U (VI) is also increased. At low concentration of U (VI), the increase is extremely fast as the material presents a lot of adsorption sits inside (Freundlich, 1906; Langmuir, 1918).


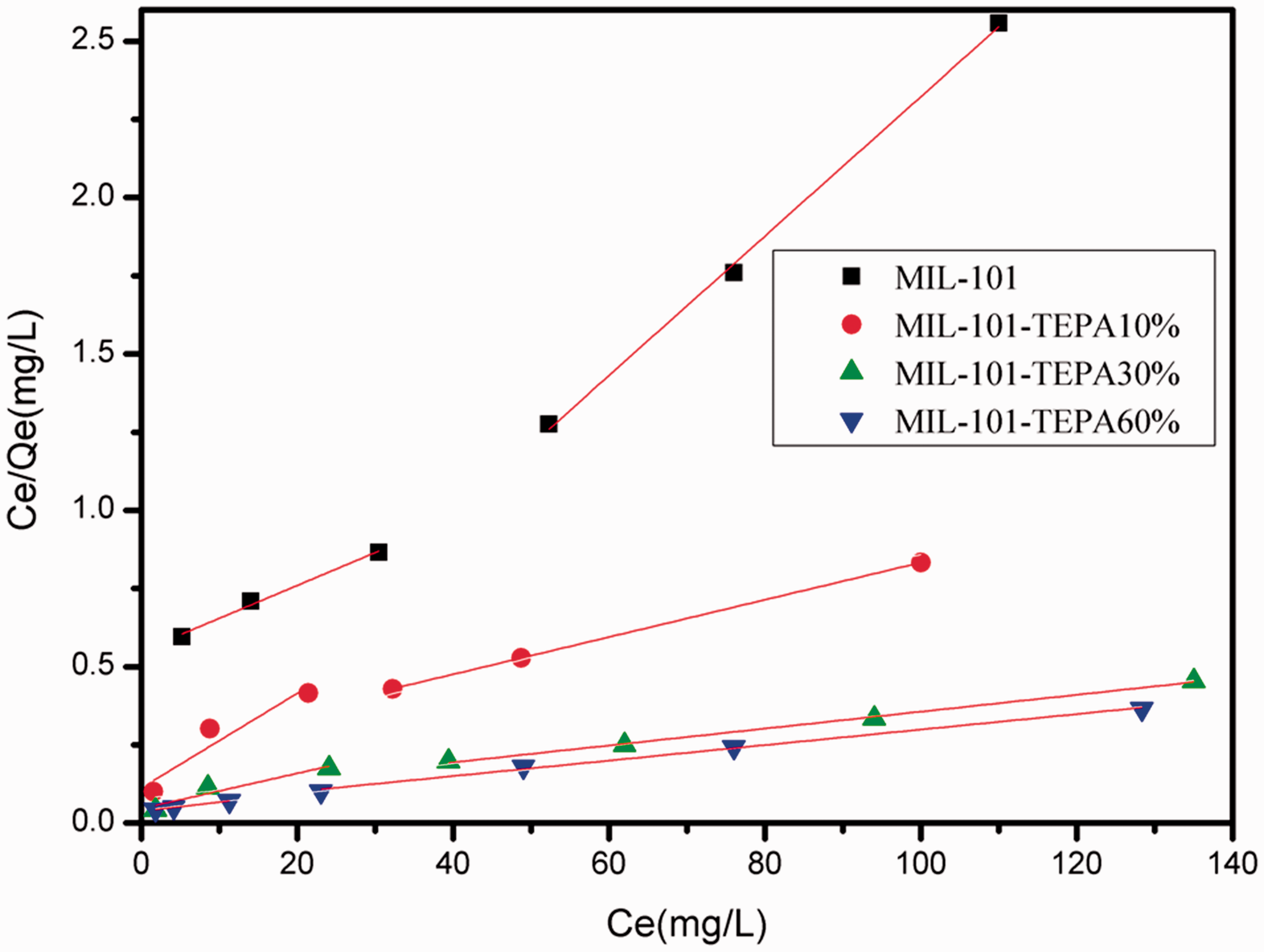
Langmuir plots for the removal of U(VI) by MIL-101 and TEPA-MIL-101s.
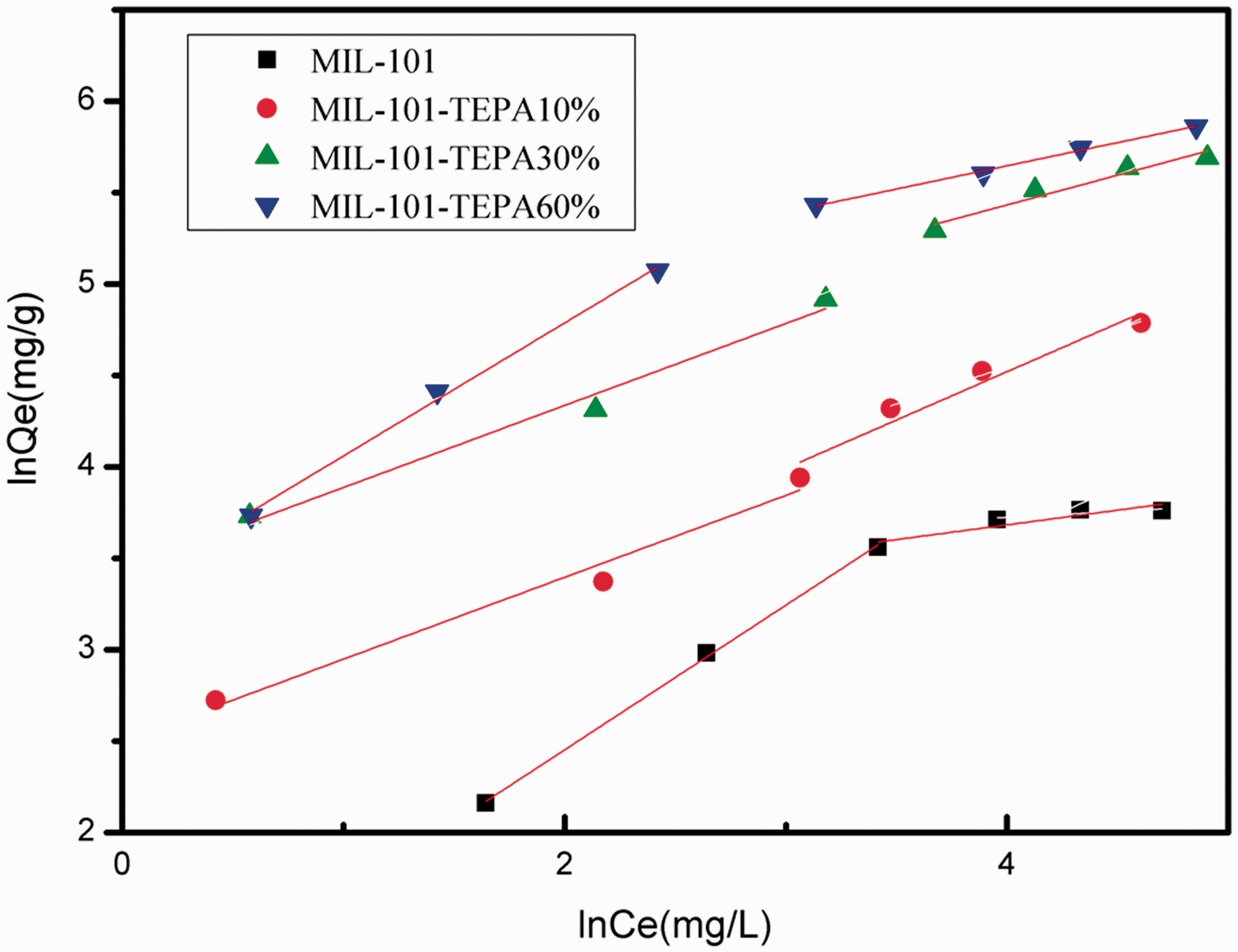
Freundlich plots for the removal of U (VI) by MIL-101 and TEPA-MIL-101s.
Adsorption constants for Langmuir isotherm by MIL-101 and TEPA-MIL-101s (adsorption dosage 5 mg, agitation time t = 2 h, T = 25°C and pH = 4.5).

Adsorption constants for Freundlich isotherm by MIL-101 and TEPA-MIL-101s (adsorption dosage 5 mg, agitation time t = 2 h, T = 25°C and pH = 4.5).

Comparison of the maximum adsorption capacity of uranium on different metal–organic frameworks (MOFs) adsorbent.

Conclusions
As amino group had strong adsorption property to uranyl ions, TEPA was successfully intruduced into MOF to obtain MIL-101 (Cr) MOF such as TEPA-MIL-101s and was thermally stable at 318 K. The adsorption capacity for U (VI) of this porous TEPA functionalized (TEPA-MIL-101s) could be greatly improved when a large amount of amino had been introduced. Compared with the three different amounts of TEPA, the mass ratio of 60 wt% adsorbent of MIL-101-TEPA 60% exhibited significantly high adsorption property with the maximum adsorption capacity of 350 mg/g for uranium (VI) adsorption from water at pH of 4.5.
Significantly, MIL-101-TEPA 60% exhibited high adsorption capacity (350 mg/g) for uranium (VI) from water at pH 4.5. The adsorption kinetics and adsorption isotherms studies of these TEPA-MIL-101s showed that, at uranium concentration <20 mg/L, the isothermal plot was best represented by Freundlich model. At U(VI) concentration approximately >30 mg/L, the isotherm was best described by Langmuir adsorption isotherm in the process of adsorption kinetics. In the meanwhile, the adsorbed U (VI) ions could be desorbed with HCl, HNO3, or Na2CO3, respectively. The study showed that grafting amines with proper mass ratio of TEPA on the open metal sites of MOFs would be an effective method of achieving high adsorption property for U (VI) and the MIL-101-TEPA 60% could be explored to be a promising material for enhancing U (VI) adsorption from waste water.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.



