Abstract
A novel three-dimensional porous metal organic framework Zn(BTC)4 (BTC = benzene-1,3,5-tricarboxylic acid) is synthesized by the solvothermal method. This structure is characterized by single-crystal X-ray diffraction, scanning electron microscopy, and thermogravimetric analysis. This metal organic framework crystallizes in a monoclinic (
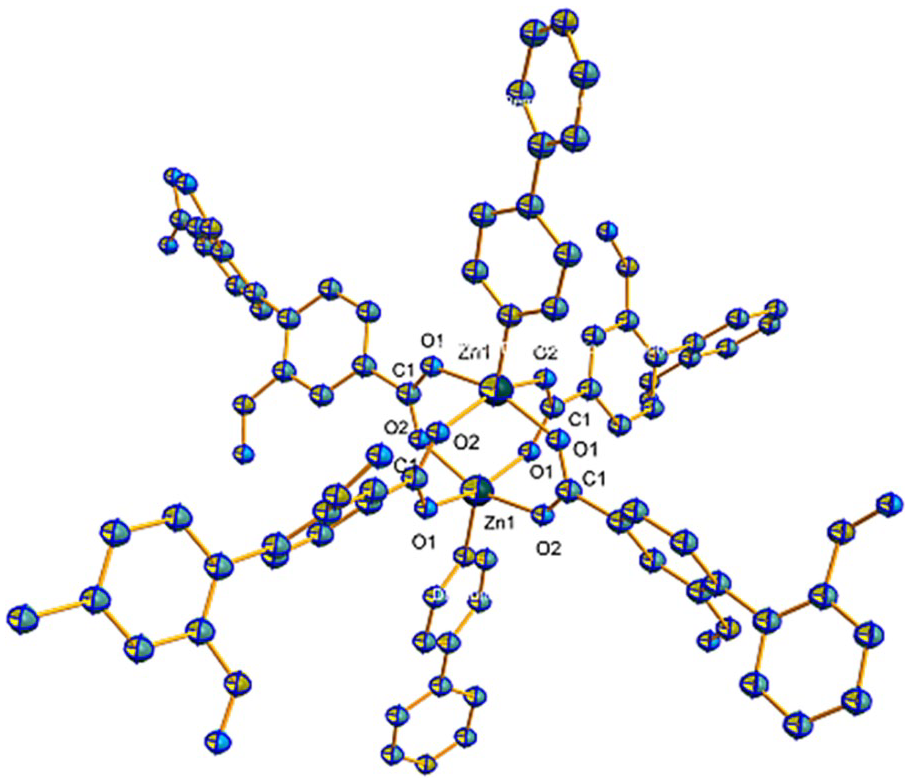
MOF Zn(BTC)4
Introduction
Metal organic frameworks (MOFs) are a type of important hybrid inorganic–organic material, 1 and due to their unique topological structure, their applications have attracted more and more attention. 2 Most MOFs have high porosity and good stability, 3 controllable pore structures, and large specific surface areas. 4 Therefore, MOFs have extensive application prospects compared with other porous materials, for example, in chiral catalysis, 5 adsorption separation, 6 adsorption storage, 7 magnetic materials, 8 and optical materials. 9 As an important part of the framework, organic ligands play a decisive role in the pore properties of these compounds, so we can design and adjust the ligands to selectively adsorb gas. 10
Today, in the face of energy shortages, seeking new energy sources is a solution to aid the energy crisis. 11 Among new energy sources, the calorific value of gas energy, such as from hydrogen, is relatively high. 12 Hydrogen is also clean, green, and is available from many rich sources. It is also expected to help alleviate the oil crisis. 13 However, the storage and transportation of hydrogen is a significant obstacle to industrial utilization. 14 On the contrary, the greenhouse effect is more and more destructive to the environment. Methods to effectively store and reuse greenhouse gases such as carbon dioxide and methane without emissions into the atmosphere is a key factor in solving this problem. 15 Among all the various gas storage technologies, adsorption technology has the characteristics of being fast and flexible, so the preparation of efficient adsorption materials has remained an important topic. 16
Results and discussion
The MOF Zn(BTC)4 was prepared from benzene-1,3,5-tricarboxylic acid and Zn(NO3)2•6H2O in H2O at 80 °C in an autoclave.
Crystal structures
The single-crystal diffraction data show that this compound belongs to the monoclinic system and has space group

Secondary building units (SBU) image of compound

The 3D framework of compound
Scanning electron microscopy characterization of compound 1
The morphology of compound

The SEM image of compound
Thermoanalysis
In order to examine the thermal stability of compound

The TG curve of compound
Solid-state luminescence
The luminescence properties of compound

Luminescence properties of compound
Gas adsorption properties and pore size analysis
Figure 6 shows the hydrogen storage curve of this material. At 298 K, it was activated under vacuum for 48 h and then the hydrogen storage performance of this material was tested at 77 K. The experimental results show that the hydrogen storage capacity of the sample is 2.01 wt% at pressure of 10 bar.
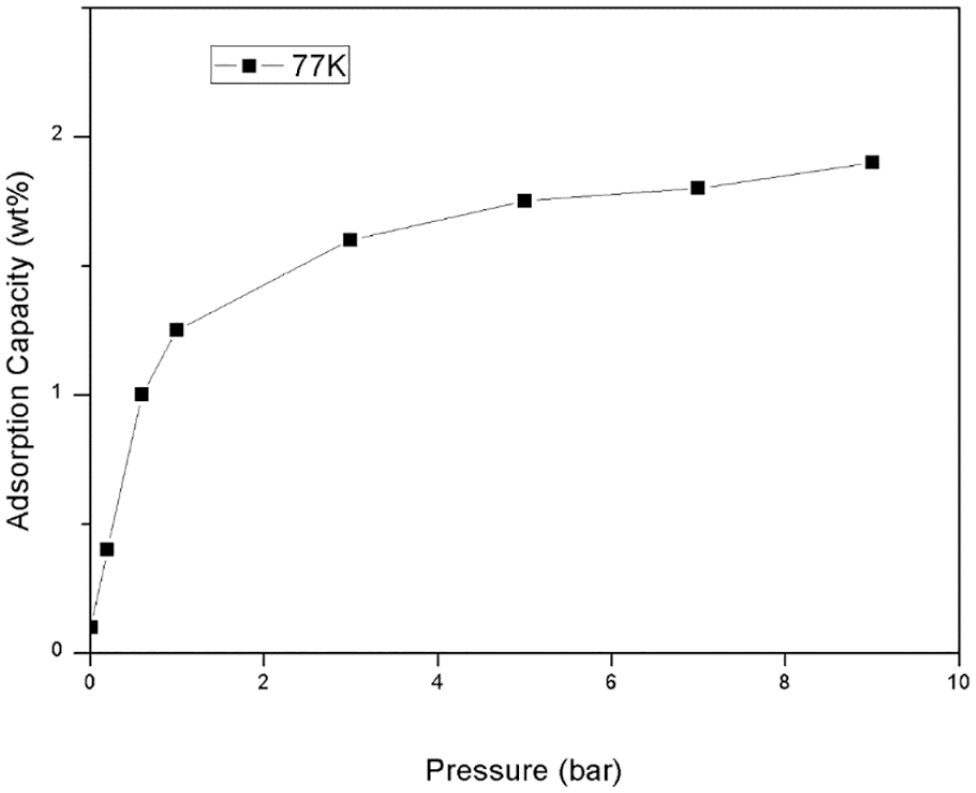
Hydrogen storage curve of compound
Figure 7 shows the adsorption curves of carbon dioxide at 298 and 323 K. It can be seen from Figure 6 that the adsorption capacity of carbon dioxide increases with an increase of pressure. When the pressure was increased to 10 bar, the material adsorbed 4.17 mmol/g carbon dioxide at 298 K. Moreover, the adsorption capacity for carbon dioxide decreases on increasing the temperature. When the temperature was increased to 323 K, only 3.76 mmol/g of carbon dioxide was adsorbed at 10 bar. The pore structure parameters are listed in Table 1.
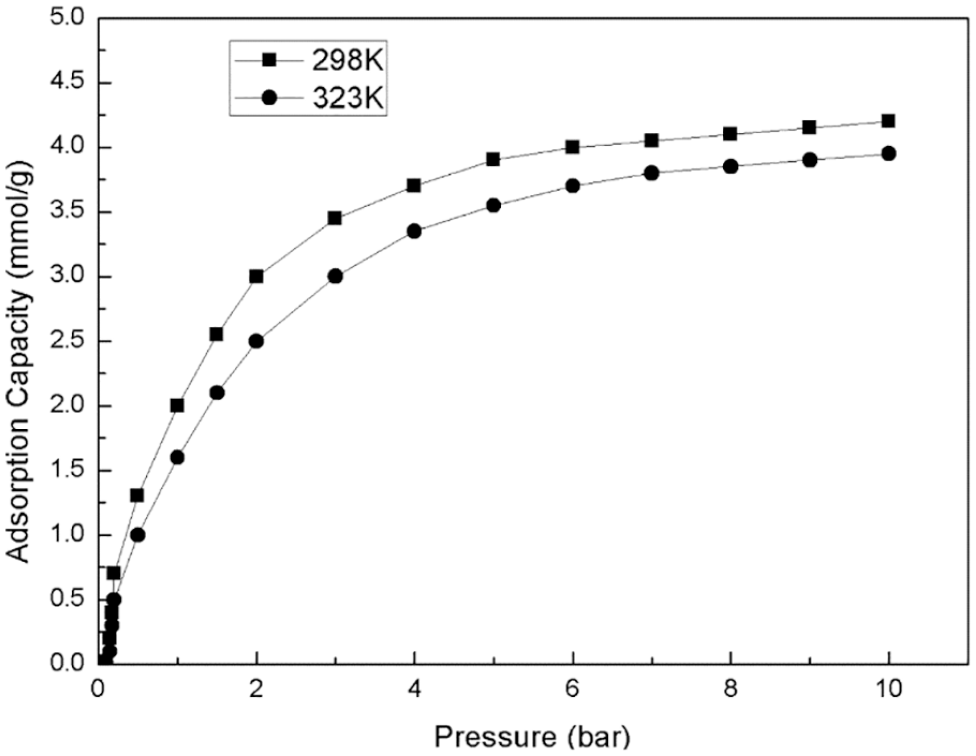
Carbon dioxide adsorption curve of compound
Pore structure parameters of compound

Conclusion
To sum up, the MOF, Zn(BTC)4, can be synthesized by a solvothermal method using benzene-1,3,5-tricarboxylic acid as the ligand. The TG analysis shows that the material is stable up to 350 °C, then the skeleton collapses between 350~510 °C. The luminescence test shows that the material gives out strong emissions at 384 and 462 nm. The maximum hydrogen and carbon dioxide storage capacity of this MOF is 2.01 wt% (77 K, 10 bar) and 4.17 mmol/g (298 K, 10 bar) respectively, which shows that this MOF material may act as a potential material for gas adsorption. Further studies in this area toward the exploration of new structures and functions of MOFs are underway.
Experimental
Reagents and instruments
The chemical reagents used in this experiment are all commercial reagents and were used directly.
The crystal structure was solved using a Bruker SMART 1000 CCD diffractometer.
A HITACHI s4800 scanning electron microscope was employed to study the morphology.
Infrared (IR) spectra were recorded using a Bruker Tensor 27 Fourier-transform infrared spectroscopy (FTIR) spectrometer in the range of 4000–400 cm−1 (KBr pellets).
The luminescent properties were studied using an F-4500 fluorescence spectrometer at room temperature.
Synthesis
The main raw materials of compound
Structural characterization
Single crystals with regular shape and of appropriate size were selected to collect the diffraction data. Using Mo-Kαradiation (λ = 0.071073 nm) monochromated by a graphite monochromator (T = 298(2)K), the diffraction points were collected in the range of 2.17° < θ < 25.01° in the ω/2θ scanning mode. Data are reduced (SAINT) for absorption corrections (SADAB) after diffraction. The crystal structure was solved by direct method using SHELXL-2014 program. 17 The coordinates of all n-hydrogen atoms and their anisotropic thermal parameters were corrected by the full matrix least squares method (SHELXL-2014). Crystallographic data are shown in Table 2, and bond lengths and bond angles are shown in Table 3; CCDC: 1539458.
Crystal data of compound

Selected bond lengths (Å) and angles (°) for compound

Gas adsorption and pore size characterization
An intelligent gravimetric analyzer (IGA-001) was used to adsorb hydrogen and carbon dioxide under high pressure. Before the test, a 50-mg sample was activated by drying in a vacuum for several hours. The activated samples were used to adsorb carbon dioxide at 298 and 323 K, respectively. Hydrogen was adsorbed on the sample at 77 K.
Footnotes
Acknowledgements
1. National Natural Science Foundation of China. No. 12064050.
2. National Natural Science Foundation of China. No. 21965038.
3. Applied Basic Research Program of Yunnan Province. No. 2019FB024.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Natural Science Foundation of China/Regional Science Foundation Project. No. 21965038 and Applied Basic Research Program of Yunnan Province/General Projects of Basic Research. No. 2019FB024.
