Abstract
The main purpose of this study was to evaluate the ultraviolet protective factor (UPF) of fabrics coated with titanium-based metal-organic framework (MOFs) (NH2-MIL-125(Ti)) made using an In-situ synthesis method and more accurately assess the intrinsic properties of the fabric. The cotton-polyester twill fabric (70-30%) (246.67 g/m2) was coated. In-situ coating conducted in 4 steps washing the fabrics, preparation of MOFs, injecting the MOFs to fabrics, and drying the fabric after coating. The scanning electron microscope (SEM), FTIR spectrometer, dynamic light scattering (DLS) and UV-Vis spectrophotometer were used to analyses the data of coating and UPF results. Also, four standards such as ASTM D737, ISIRI 8332, ISIRI 4199, and ISIRI 567 were used for analyzing the intrinsic properties of the fabric. The results of SEM and DLS altogether confirmed the in-situ formation of MOFs onto the fabric fibers. Moreover, the UPF value of the uncoated and coated fabrics was 3.67 and 315.73, respectively. It was showed that the in-situ deposition of NH2-MIL-125(Ti) on fabric can provide adequate protection against UVR. Also, the results of analyzing the intrinsic properties of the fabric showed that there was no significant difference in the intrinsic properties between the coated and uncoated fabrics. Based on the results, it can be concluded that the UV protective property of workwear fabrics can be improved by coating NH2-MIL-125(Ti) on them without any effect on the cooling effect of perspiration evaporation.
Introduction
In addition to the biological effects of ultraviolet radiation (metabolism activation, blood circulation improvement, and protection against pathogens), it has harmful health effects including premature aging of the skin, allergies, sunburn, and skin cancer.1,2 The skin is the largest organ of the human body (accounting for 12–15% of our body weight) exposed to ultraviolet radiation from sunlight. Thus, experts suggest several methods of UV protection such as using sunscreens, avoiding prolonged exposure to the sun at its highest intensity, and wearing clothing that effectively covers the skin surface. Also, umbrellas and parasols can offer sun protection. 3
Outdoor workers such as farm-workers, mine workers, bricklayers, construction workers, etc., are exposed to UV radiation more frequently and for longer periods than people working indoor. The clothing worn by them is considered the most important personal protective equipment. Wearing protective clothing is the cheapest and easiest way to limit UV exposure.1,4 However, intrinsic properties of fabrics such as physical structure and chemical composition, low absorption, and UV– induced vulnerability of fabrics can affect their UV-blocking properties.
Therefore, the UV-blocking properties of the fabric should be optimized. In other words, improvement of the physical and chemical properties of fabrics cause changes in the Ultraviolet Protection Factor (UPF). 3 Fabrics generally have a UPF rating of 15–50, allowing only 2.5% of the sun’s UV rays to pass through and any fabric that allows less than 2% UV transmission is labeled UPF 50+. UPF values of the fabrics such as cotton, polyester, and silk have been reported to be around 2–4, 30, and 10, respectively. Hence, researchers have used different coatings as UV protectors for producing fabric fibers.5,6 The physical structure and chemical composition of fabrics affect the UPF. 7 Chemical additives such as chemical additives and nanoparticles used as UV protectors can become toxic and degraded by sunlight.8,9 The use of nanomaterials for the development of protective textiles has increased rapidly due to the numerous limitations of conventional textiles for protection against various environmental hazards.10,11 Some specific applications of nanomaterials for protective textiles include TiO2 nanomaterials for UV protective textiles, silver nanoparticles for antioxidant textiles, titanium silver nanoparticles for chemical protective textiles. 12 New materials such as metallic-organic frameworks (MOFs) can impart UV blocking properties to fabrics. 13
Metal-organic frameworks, formed by metal ions or clusters and organic linkers, possess unique physical and chemical properties, including large surface area, controllable pore size, homogeneous active sites, and diverse functions.14,15 Owing to their structural tunability, metallic-organic frameworks can transport the anti-UV properties to the assembled MOFs materials. Thus, MOFs are often used as modifiers to improve the performance of fabrics, polymers, and porous metals. Since 2017, a few studies have investigated the role of MOFs in improving the UV-blocking properties of fabrics.16–18 MOFs have been used to enhance water absorbency, antimicrobial, and permeability properties. A few studies have been published on the toxicity of MOFs in which no toxic effect on the human body has been reported. 16
Given the vast numbers of outdoor workers and their prolonged exposure to the sunlight, developing UV-protective textiles seems to be necessary. The intrinsic properties of fabrics such as physical structure and chemical composition, low absorption, and UV– induced vulnerability of fabrics can affect their UV-blocking properties. All the problems mentioned have been corrected in the past studies by coating textiles with some nanoparticles and chemicals, but some cases have not been highly effective. So far, very few studies have used the MOFs to improve UPF. Therefore, the present study aimed to evaluate the UPF (ultraviolet protective factor) of fabrics coated with in-situ synthesis of organic-metal framework and more accurately assess the intrinsic properties of the fabric (such as air permeability, abrasion resistance, tensile strength, and surface wetting resistance).
Material and methods
Materials
The Chemical substances including 2-Aminoterephthalic acid (166.13 g/mol): H2NC6H3-1,4-(CO2H)2: Sigma-Aldrich Co. No. 381071, Titanium isopropoxide (TTIP) (284.22 g/mol, C₁₂H₂₈O₄Ti): Merck No. 821895, ethanol 99.99%, MERCK 106009 Methanol, and Merck 103053 N, N-Dimethylformamide: C3H7NO (73.09 g/mol) were purchased from Shimi Parsian Co Tehran, Iran. Also, the cotton-polyester twill fabric (blend ratio: 70/30%) (246.67 g/m2) was provided by Yazd Baf Co, Yazd, Iran. To remove the interference effects of dying on the UPF value, the fabric was purchased from the factory before being dyed. The cotton-polyester twill fabric (blend ratio – 70/30%) was used since it is the most common workwear fabric in Iran based on standard No. ISIRI 6914. 19
Synthesis of MOFs
In-situ synthesis of a titanium-based metal-organic framework (NH2-MIL-125(Ti)) on the fabric surface was used in the present study as described below:
The fabric samples in cuts of 4 cm × 4 cm were washed with ethanol inside a shaker and then again rinsed in distilled water and dried in an oven at 80°C. Next, the solutions A and B were prepared as follows to make the MOFs. Solution A was prepared by mixing 30 mL of methanol and 0.4 mL of TTIP in a beaker, and the mixture was stirred for 90 min at room temperature by a stirrer. Solution B was prepared by mixing 12 mL of DMF and 0.4 g 2-Aminoterephthalic acid in a beaker, and the mixture was stirred for 90 min at room temperature by a stirrer. In the meantime, the fabric was soaked in solution A so that the solution could penetrate the fabric. Solution B was then added dropwise to solution A which was stirring at room temperature. The new solution was stirred for 90 min at room temperature by a stirrer. The final solution containing the fabric has been incubated for 48 h at room temperature. The fabric has been then removed from the final solution and dried in an oven at 80°C for 3 h. Finally, it was washed three times in distilled water and completely dried at room temperature.
Structural properties
The morphological structure of the fabrics was investigated using a scanning electron microscope (SEM) and energy-dispersive X-ray spectroscopy system (EDX) (XL30, supplied by Philips Healthcare, the Netherlands). The samples were placed on the SEM stub and coated with gold. An FTIR spectrometer (Tensor 27, Bruker, Ettlingen, Germany) was used to analyze the type and structure of the synthesized metal-organic framework and metal oxide particles and the amount of metal loaded. Moreover, a dynamic light scattering analyzer (Microtrac MRB’s NANOTRAC Wave, USA) was used to measure the average size and size distribution of metal-organic frameworks and metal oxide particles. The energy-dispersive X-ray spectroscopy system, attached to the same microscope, was also used for elemental analysis.
UV protection
The UV transmittance spectra (280–400 nm) of the fabric were measured at a distance of 5 nm using a spectrophotometer (Varian Cary 100, Australia).
20
The UV protection factor (UPF), UV-A (315–400) protection factor, and the UV-B (280–315) protection factor were evaluated using AS/NZ 4399: 1996 and equation (1). Three replicate measurements were carried out for each sample and an average value was reported. According to the AS/NZ 4399: 1996 standard, the spectral intensity of radiation proportional to that of the solar spectrum (Sλ) in the spectrophotometer was proportional to any wavelength at 290 nm (4–10 × 767/0 W.m−2.nm−1) to 400 nm (180/1 W.m−2.nm−1).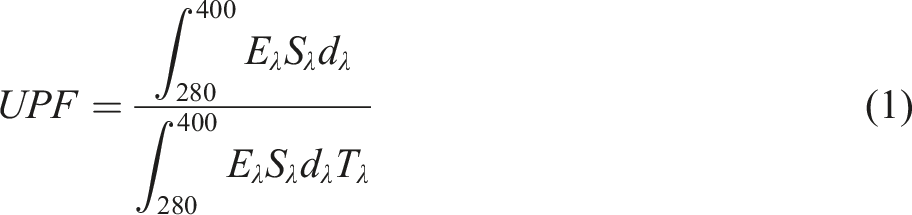
Eλ relative efficiency of the radiation source
Sλ initial spectrum of the radiation source (W.m−2. nm−1)
dλ bandwidth (nm)
Tλ the amount of light transmitted
Properties of the fabrics
The intrinsic properties of fabrics contribute to their performance, resistance, and heat transfer. Thus, the effect of the coating materials on these properties needed to be investigated. The following properties were examined in the present study: 1- Air permeability: the rate of airflow passed through the pores. This property which indicates the resistance of the fabric to sweating-induced evaporative cooling was tested via the ASTM D737 method using the TF164E Air Permeability Apparatus.
21
In this test, the volume of air passing through the fabric in terms of cubic decimeters per minute (dm3/min). Then air permeability (R) in terms of dm3/h/m2 was calculated in each of the studied fabrics under laboratory conditions under a pressure difference of 100 Pascal based on the following equation (Equation (2)). 2- Abrasion resistance: the ability to resist wearing and indicates the cut resistance. This property was estimated using the ISIRI 8332 standard.
22
In this test, the fabric is subjected to rotary rubbing under controlled conditions of pressure and wear. One of the wheels wears the fabric from the outside to the periphery, and the other from the inside to the center. As a result of wear, a series of crossed arcs with an approximate area of 30 square centimeters is created. 3- Tensile strength: resistance to tensile force and indicates the strength of the fabric against tearing. This property was evaluated using the ISIRI 4199.
23
To perform this test, a device that basically consists of two vertical jaws on fixed bases. A set consisting of at least 5 fabrics in the direction of the warp and 5 fabrics in the direction of the weft of the fabric was prepared using a cutting template. The test has been performed in an environment with relative humidity of 65% and temperature of 20°C, and finally, according to the used capacity, the number placed in front of the screen was noted,and the sliding force was estimated in Newton (N). 4- Surface wetting resistance: penetration of water through the fabric or the resistance to wetting. It was measured using the ISIRI 567.
24
Based on this standard, initial weight (M1) of 5 fabric samples from each of the studied fabrics were measured. Then, the fabric samples were placed horizontally in a container containing distilled water with a temperature of 20°C. In such a way that the depth of the water level to the fabric was about 2 cm. Again, the secondary weight of the soaked fabric (M2) was recorded. Finally, the percentage of absorbed water in the studied fabric was calculated using the following equation (Equation (3)).


Results and discussion
Properties of the fabric coated with MOFs
Coating thickness was determined by gravimetric analysis. The results showed that the weight of the fabric was 0.4297 g before coating and 0.4756 g after coating, so 10.68% of MOFs had been coated on the surface of the fabric. EDX analysis, as well as SEM images, confirmed the in-situ formation of MOFs onto the fabric fibers (Figure 1). Comparing the SEM images before and after the MOFs coating shows the presence of the particles. Ti signals observed in EDX analysis patterns of the fabric modified with NH2-MIL-125(Ti) were recorded. The diameter of MOF particles on the fabric fibers was measured using the DLS method. The average diameter of NH2-MIL-125(Ti) determined by DLS was found to be 621.5 nm (Figure 2). Dense MOFs were observed on the fabric, indicating the effect of the chemical composition of the fabric on the nature of the prepared MOFs.25,26 EDX and scanning electron microscope analysis of the coating of MOFs (10.68%) onto the fabrics (d = 1cm): a and b) Imaging of the uncoated fabric with image sizes of 20 μm and 10 μm. c, d, and f) Imaging of MOFs-coated fabric with image sizes of 50 μm, 5 μm, and 2 μm e) EDX spectra of the agents coated onto the fabric surface. EDX = X-ray spectroscopy system; MOFs = metal-organic frameworks. Size distribution of NH2-MIL-125(Ti) obtained by dynamic light scattering.

The chemical composition of the fabric was investigated using FTIR before and after direct in-situ synthesis of MOFs (Figure 3). In the cotton/polyester uncoated fabric, the absorbance bands were observed at 2903 and 3338 cm−1 due to C-H stretch and aliphatic asymmetric C-H stretch, respectively.27,28 The absorption bands at 1711 cm−1 and 1410 cm−1 were associated with C≡C and CH2 groups, respectively. Surface modification of MOFs led to the alteration of the absorption bands, besides the alteration of the visible absorption spectrum. The peaks associated with OH, CO, and CH2 groups shifted to 2902 and 3285 cm−1, 1710 cm−1, and 1244 cm−1, respectively. These results confirmed the interaction between the MOFs and the fabric functional groups.
29
FTIR spectra of uncoated fabric compared with fabric coated with MOFs: a: Uncoated fabric, b: fabric coated with MOFs. MOFs = metal-organic frameworks.
Figure 4 shows the UV blocking capacity of the uncoated and coated fabric measured using the UV transmittance spectra. The UPF value of the uncoated fabrics calculated via equation (1) was 3.67 and the UPF value of fabrics coated with MOFs was 315.73, indicating the strong UV blocking ability of the coated fabrics. It was confirmed that the in-situ synthesis of Nano-sized MIL-MOFs on fabrics can provide adequate protection against UVR. According to the Australian/New Zealand Standard and the British standard,
20
uncoated fabrics with UPF < 20 have insufficient UV protection. After direct in-situ modification, the fabrics reached UPF > 50 and could be classified in the excellent UV protection category according to the mentioned standard. Other studies have also revealed that NH2-MIL-125(Ti) coated on fabrics can provide adequate protection against UVR.7,15 UVA/UVB transmittance property of the uncoated fabrics and the fabrics coated with metal-organic framework according to the AS/NZ 4399:1996. UVA: Ultraviolet (315-400 nm); UVB: Ultraviolet (280-315 nm).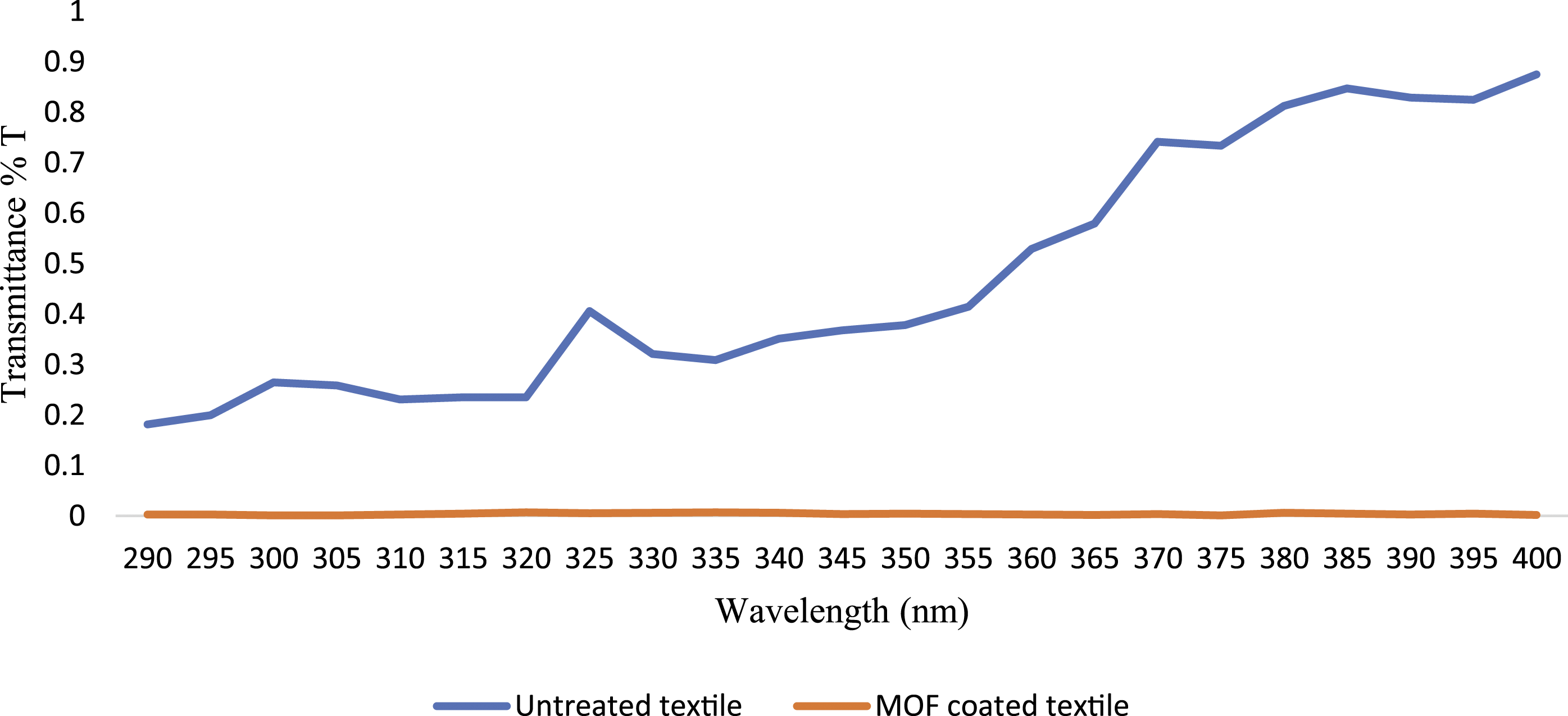
The authors know of two studies that have investigated the UV-blocking capacity of the MOFs which have shown similar results. Previous studies have also shown that the in-situ synthesis of MIL-MOFs on the fabric surface can lead to UV protection capacity, providing good to excellent UV protection. There are three main reasons for these results. First, the incorporation of MOFs in the fabrics enabled the formation of MIL-MOFs inside fabric pores and easy UV absorption. Second, the in-situ synthesis of MOFs resulted in higher MIL-MOFs content of the modified fabrics and improved UV protective property. In addition, the fabrics contained multiple binding sites acting as chelates during the formation of MIL-MOFs from 2-aminotrephthalic acid.7,15,17 Emam and et al. used MIL (Ti, In)-MOF. The results showed that in cotton fabrics containing both types of MOFs, the UPF increased from 3.5 to more than 100. 7 Lu et al. aimed to improve the UPF of cotton fabric using ZnBDC MOF. The results showed that ZnBDC-impregnated cotton fabric has excellent UPF, more than 50. 18
Also, the MOFs absorb ultraviolet light in such a way that the organic part in them usually absorbs radiation, and then they are transferred to the metal node through the process of ligand-to-metal charge transfer (LMCT). 30 Bonded ligands, rich in conjugated electrons with photosensitive function with extensive light absorption, are often chosen as the organic part in the construction of titanium-based frameworks. When the monoamine BDC-NH2 ligand is reduced in MIL-125, resulting from N2p electron donation to the aromatic rings. Also, due to their small size, controlled porosity, large surface area, and meanwhile, organic linking ligands or metal ions from the framework can create UV inhibitory and blocking properties. 31 Based on what was stated in the previous studies, the metal part in the MOF has a more prominent role for absorbing UV.
Structural properties of the fabric
Comparison of uncoated and coated fabric with metal-organic frameworks in terms of intrinsic properties of the fabric.

*Mann-Whitney Test
This lack of difference shows that coating the fabrics with MOFs did not affect their intrinsic properties, since the heat and air transfer capacities of the modified fabric were similar to those of the uncoated fabric. Also, the modified fabric had sufficient resistance to withstand work environments. Few studies have investigated the intrinsic properties of the fabric after coating it with MOFs and the majority of studies conducted in this field aimed to make fabrics waterproof. No studies have been conducted to design workwear fabrics with high UPF values without imposing changes to their intrinsic properties. However, previous studies have also shown that hydrophilicity is an important property that refers to absorbing moisture vapor and can add to the comfort properties of fabric.32,33 Li et al. showed the hydrophilicity of MOFs causing ready adsorption of water. They also investigated the abrasion resistance of the fabric coated with MOFs. 17 Consistent with the present study, they showed strong interactions between MOFs and substrates of the modified fabrics which exhibited great flexibility, so they can be adaptable for practical applications.
Conclusion
The present study aimed to evaluate the UPF of fabrics coated with MOFs made using an in-situ synthesis method and more accurately assess the intrinsic properties of the fabric. The results showed that the fabric coated with MOFs had greater UPF values than uncoated fabric, and also the intrinsic properties of the coated fabric did not change significantly. Based on the results, it can be concluded that the UV protective property of workwear fabrics can be improved by coating MOFs on them without any effect on the cooling effect of perspiration evaporation. However, production of workwear fabrics requires further research. Future studies can evaluate the cytotoxicity and antibacterial properties of MOFs to ensure that MOF coating of fabrics has no adverse effects on human skin.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was part of the research projects supported by Shahid Beheshti University of Medical Sciences (Grant no. 28590).
Ethics approval and consent to participate
Ethical approval for this study was obtained from School of Public Health and Neuroscience Research Center, Shahid Beheshti University of Medical Sciences (IR.SBMU.PHNS.REC.1400.024).
