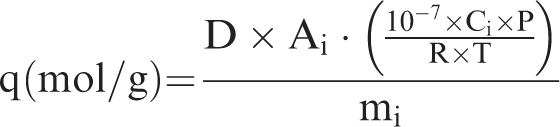Abstract
Study of preferential adsorption sites of H2O on NaX and BaX zeolites was carried out after adsorption/desorption of carbon dioxide (CO2). The profiles of CO2 desorption revealed two distinct zones. The first one, at low temperature (T < 533 K), corresponding to physisorbed CO2 where BaX zeolite has an energy of adsorption higher than that of NaX as well as strong interactions of adsorbent/adsorbate. The second one (533–623 K) corresponds to the chemisorbed CO2. The presence of water influences CO2 adsorption. The effect is more important for NaX than for BaX. For NaX, water occupies the adsorption sites of CO2 between 373 and 483 K. In contrast, BaX preserves a majority of adsorption sites of physisorbed CO2 in the presence of water, in particular the adsorption sites located in the interval of temperature 423–533 K.
Introduction
The increase of the emissions of CO2 in atmosphere is due to human activities such as combustion processes, fluid catalytic cracking, and other processes producing gas flows with low CO2 concentrations (Macario et al., 2005). However, CO2 can be removed from flue gas and other gas streams by various methods (Olajire, 2010; Zhao et al., 2013) such as membrane separation (Hasegawa et al., 2017), absorption with solvent (Aboudi and Vafaeezadeh, 2015; Adeyemi et al., 2017), and adsorption using molecular sieves (Khelifa et al., 2004). These methods, however, are costly and energy consuming. One technique for CO2 sequestration from gas stream can be based on the adsorption/desorption using three-dimensional porous basic compounds such as natural or synthetic faujasite-type zeolites. Zeolites are widely used as materials for gas separation and purification, ion exchange, and catalysis (Walton et al., 2006).
The faujasite X-type zeolites appear to be better adsorbents for CO2 compared to others zeolites such as faujasite Y-type zeolites, MFI, and LTA zeolites (Bonenfant et al., 2008; Choi et al., 2009; Montanari et al., 2011). In recent years, several studies have been carried out on CO2 adsorption using X-type zeolites exchanged with monovalent or bivalent cations (Bonenfant et al., 2008; Davis, 2003; Khelifa et al., 2004; Walton et al., 2006) to determine the effects of the crystal structure and the nature of the ion on the adsorption equilibrium. Other studies on infrared spectroscopy by Fourier transformation infrared (FTIR) (Di Cosimo et al., 1998; Doskocil and Davis, 1999; Hadjiivanov and Knözinger, 2009; Lavalley, 1996; Martra et al., 2002; Rege and Yang, 2001) and CO2 temperature-programmed desorption (TPD) (Doskocil and Davis, 1999) on X-type zeolites have shown two types of adsorption onto the surface: a physisorption corresponding to adsorbed CO2 linearly linked onto cationic sites and a chemisorption corresponding to adsorbed CO2 as bicarbonates (HO–CO2− or HCO3−), and unidentate or bidentate carbonates species (CO32−). Studies realized by Doskocil and Davis (1999) by FTIR and CO2-TPD on X-type zeolites that are exchanged by monovalent cations have shown that on KX and CsX zeolites, the adsorption bands corresponding to bicarbonates (Rege and Yang, 2001) disappeared at 473 K and those of the unidentate and bidentate carbonates species disappeared at 573 K.
In the presence of water, the adsorption capacities of CO2 decrease in the X-type zeolites (Yong and Alirio, 2002). Oliveira et al. (2008) made these observations in their work and according to them, these losses are due to a reduction of the electronic field strength of the zeolite caused by a strong adsorption of water on cationic sites. The basic properties of faujasite X zeolites come from cations and so the cationic sites of type X faujasite are sites of adsorption of carbon dioxide (CO2) and the more basic the cation, the more CO2 will be strongly adsorbed (Martra et al., 2002).
In this study, we carried out sodium cation exchange (Na+) by barium (Ba2+) to increase the strength of the basic sites and thus the interactions between CO2 and the cation. Then, we have investigated the influence of barium and sodium on the adsorption sites of CO2 in the presence of water. To do this, we have compared the adsorption and desorption of CO2 onto NaX and BaX zeolites in order to locate the preferential adsorption sites of the water in a precise temperature range. The use of the method of adsorption in fixed bed dynamics allowed us to compare the adsorption capacities of CO2 obtained in microgravimetry and to have information on the efficiency of the adsorbents bed to trap carbon dioxide.
Materials
The starting material is a NaX zeolite supplied by Axens. The replacement of the sodium cations by barium cations has been obtained by cationic exchange, using barium nitrate Ba(NO3)2 from Sigma-Aldrich (purity > 99%). Cationic exchange was carried out at 343 K by stirring 3 g of zeolites powder in 250 ml of aqueous solution containing [Ba2+] = 0.02 mol/l−1 of the metal nitrate for 24 h. The sample was then filtered, washed with ultrapure water (3 × 40 ml) to eliminate the free nitrate ions into the solids. It was then dried in a furnace during 24 h at 373 K. Exchanged BaX zeolite was finally sieved to obtain a particle size between 0.2 and 0.4 mm.
Characterizations
Elemental analysis of M-X zeolites was performed by ICP to quantify the amounts of ion-exchanged cation species.
The thermal stability (mass loss) was performed on an SDT-Q-600 TA instrument with a stream of dry argon (100 ml min−1) in a temperature range between 298 and 1073 K, using a ramp of 5 K min−1.
Measurements of surface area and pore volume were carried out using a Micromeritics TRISTAR, 3000 instrument. About 100–150 mg of the zeolite sample was pretreated during 1 h at 363 K and then 10 h at 623 K. The adsorption/desorption isotherm of nitrogen was carried out at 77 K. The specific surface areas of samples were evaluated by means of the Brunauer, Emmett, and Teller (BET) theory (Rouquerol and Rouguerol, 1999). Microporous and mesoporous volumes were determined by t-plot method of De Boer and Dubinin–Radushkevich equation, respectively (Rouquerol and Rouguerol, 1999).
X-ray diffraction (XRD) patterns were recorded at room temperature on a Siemens D5005 diffractometer using Cu Kα monochromatic radiation with 0.154050 nm wavelength. The samples were scanned in the 3–70° (2θ) range with a 0.01°/s step.
Adsorption equilibrium data on each adsorbent are measured thermogravimetrically at 296 K using a SETARAM microbalance. About 50–60 mg of sample is placed directly in the balance and then activated for 10 h under primary vacuum at 623 K. The temperature is kept constant during analysis by placing the tubes in a water bath (room temperature). The isotherm is then obtained by adding small amounts of CO2 up to atmospheric pressure.
For the experiments of adsorption in fixed bed dynamics, the total gas flow at the entry and in exit of the reactor is fixed at 150 ml min−1. The carbon dioxide concentration is of 900 ppm. The CO2 flow is of 14 ml min−1 in order to obtain a concentration applied in entry of the reactor of 84 ppm. When the experiment is carried out in the presence of steam water, in order to approach the industrial conditions, the relative humidity is fixed at 50%. This corresponds to an air flow crossing the saturator of 75 ml min−1. To obtain the fixed total flow (150 ml min−1), any difference is completed by a reconstituted air flow.
Adsorption experiments were carried out at room temperature (295 K) with a bed height of 2.9 cm (550–650 mg of adsorbents). Desorption experiments were carried out on the same bed of adsorbent, once this one was saturated. The principle is based on the sending of air reconstituted in entry of the reactor (flow 80 ml min−1) with a simultaneous heating of the sample following a ramp of 24 K min−1 until 623 K. The desorbed compounds are analyzed online using the chromatograph in gas phase and the gaseous composition in exit of the reactor.
Theoretical aspects
Sips (1948) equation model is used to refine the carbon dioxide adsorption isotherms. This model is similar to Langmuir equation but includes a parameter characterizing the heterogeneity of system
The composition of the gas mixture outflow of the adsorbents bed is analyzed every 4 min. It is thus possible to plot the curve of drilling corresponding to each gas. These curves translate the efficiency of the bed to trap or adsorb a gas. The saturation of bed is reached when the concentrations at exit of the reactor (C) are equal to the values of the concentrations of the “blank” (C0), that is the ratio C/C0 equal to the unit. We obtain surface areas in minute from integration by trapeze method
C0 and Ci are the concentrations of gas at the entry and outflow of the reactor at time ti, respectively. In addition, the quantity of gas adsorbed (q) in the bed is given by relation
Results and discussion
Ion exchange
The ionic radius of Na+ and Ba2+, the ion exchange rate, and the global Si/Al molar ratio are reported in Table 1.
Alkali-earth ionic radius and the cationic exchange for BaX.

The exchange rate is close to 100% and is the same order of magnitude as those obtained for Chabazite, Beta, X and Y in the preparation of solids for the elimination of CO2 (Seung-TaeJun and Wha-Seung, 2010; Zhang et al., 2008). Cations are considered as centers of adsorption for CO2 and that is why their numbers and distribution will have a direct influence on their performances as explained by Sips (1948). However, the replacement of monovalent cations by divalent cations results in a decrease in the number of cations and of the electronic density in the cationic supercage (Khelifa et al., 2004, 2001). This would cause a reduction of the adsorption capacities of CO2 onto zeolites modified with the divalent cations.
XRD analysis
XRD diffractograms obtained for the NaX and BaX zeolites are shown in Figure 1. No amorphous phase was observed on these diffractograms. The crystallographic structure of the parent NaX after cationic exchange does not seem to be affected since the significant XRD peaks of NaX remain visible for the XRD spectrum of the BaX sample. Moreover, analysis of these XRD spectra indicates the absence of parasite phase and/or impurities within the detection limit of the apparatus (∼2%) for BaX. The diffractogram for BaX shows some peaks more intense in the region between 10° and 45° probably due to new arrangement of cationic sites within of the X zeolite structure.

XRD patterns of NaX and BaX zeolites (Cu Kα = 0.154050 nm).
DTA–TGA
The thermal stability of samples was investigated by using DTA–TGA method (Figure 2).

Thermogravimetric curves of the NaX and BaX zeolites (heating rate of 5 K min−1).
For the two samples, most of the weight is lost between 300 and 600 K attributed to the loss of water adsorbed into the pores of the solids (Moı¨se et al., 2001). Beyond 600 and 1023 K, the weight loss is very much lower. The ratio between the total mass lost in 623 and 1073 K was calculated (Table 2). This ratio is very high as it is between 92 and 96%. This result is in line with those of Moı¨se et al. (2001) who showed that the amount of water adsorbed on MgX zeolite was higher than that adsorbed on NaX zeolite. The author attributed this behavior to the radius of the cation Mg2+ which is higher than that of Na+ resulting in a radius of hydration higher for Mg2+.
Nitrogen adsorption and ATD–ATG results for NaX and BaX zeolites.

ATD:Analysis Thermique Differential; ATG: Analysis Thermogravimetric; BET: Brunauer, Emmett, and Teller.
Nitrogen adsorption
The adsorption isotherms of nitrogen are reported in Figure 3. According to IUPAC classification, the shape of the isotherms is of Type I for all the samples indicating that they can be considered as microporous solids. A strong adsorption is observed at low p/psaturation (less than 0.2) followed by a plateau at higher pressure indicating that no more adsorption occurs. From these isotherms, BET surface areas and microporous and total porous volumes have been calculated (Table 2).
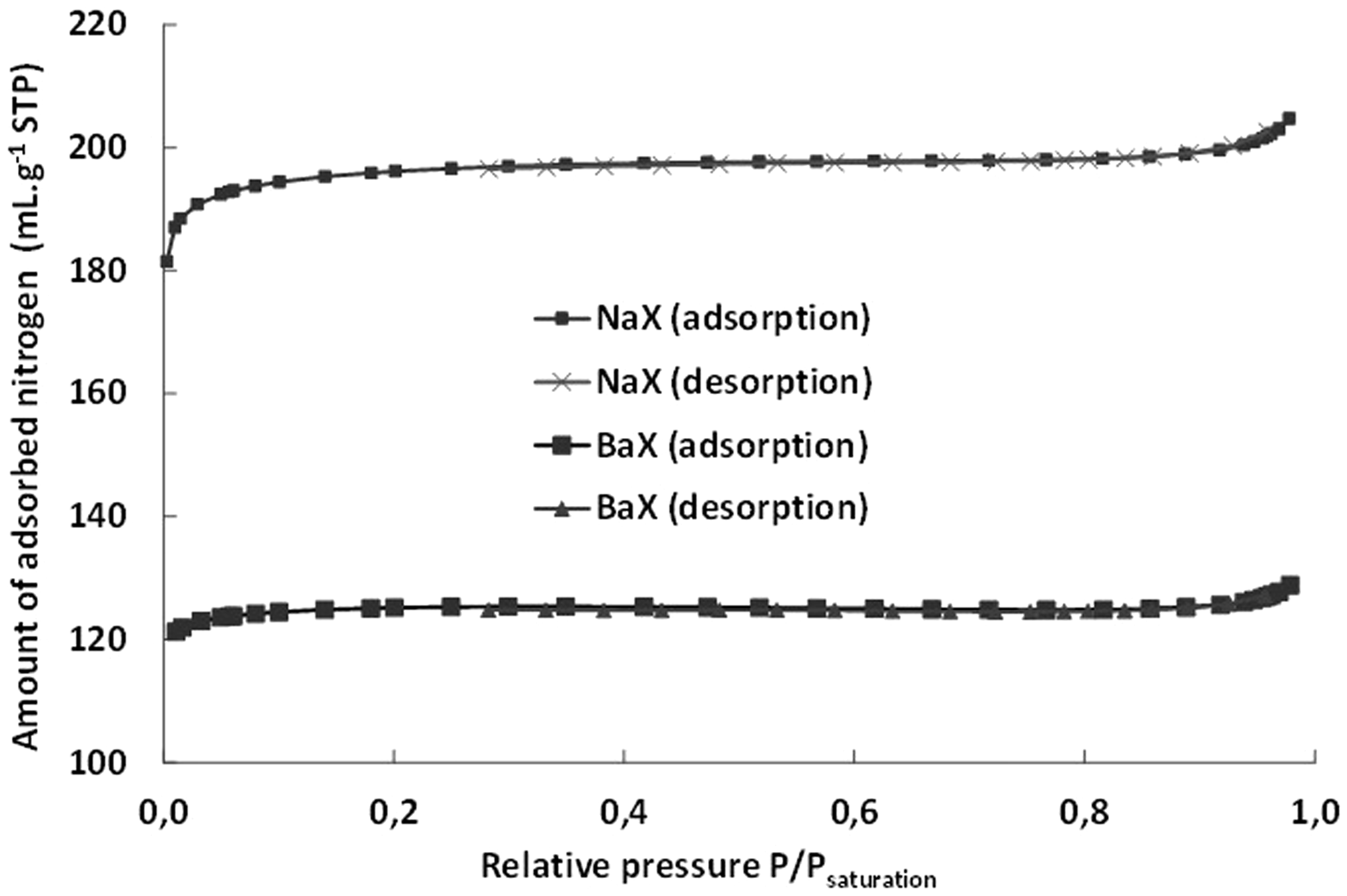
Isotherms of N2 adsorption/desorption obtained at 77 K on NaX and BaX zeolites.
For the parent NaX zeolite, the BET surface area and the microporous volume are, respectively, equal to 590 m2 g−1 and 0.317 cm3 g−1. These values are fully consistent with those reported by Hammoudi et al. (2008), namely 556 m2 g−1 and 0.315 cm3 g−1. For BaX, a decrease in the microporous volume per mass is observed (0.200 cm3 g−1 instead of 0.317 cm3 g−1). Because the Si/Al ratio is of 1.2, the formula of a unit cell of NaX is Na86Al86Si106O384 leading to a molar weight of 13,412 g unit cell−1. Since 2Na+ are replaced by 1Ba2+, and since the Si/Al ratio has not changed (Table 1), the formula of a unit cell of BaX is Ba43Al86Si106O384 leading to a molar weight of 17,338 g unit cell−1. The microporous volume of a unit cell of NaX is then equal to 4252 cm3 unit cell−1 and that of BaX is equal to 3468 cm3 unit cell−1. A loss of 17% in the microporous volume is then calculated. If now, we calculate the volume of 2Na+ and 1Ba2+ by the use of ionic radius (Table 1), the volume of 86Na+ is 230 cm3 unit cell−1 and that of 43Ba2+ is 267 cm3 unit cell−1. Thus, the loss in the microporous volume cannot be attributed to the replacement of 86Na+ by 43Ba2+. To do these calculations, it was assumed that NaX and BaX are free of water. To explain our results, it should be assumed that the cations are not free of water in spite of the severity of the pretreatment before the nitrogen adsorption. To help in further discussion, the total pore volume corresponds approximately to 122 and 100 mole of N2 per mole of unit cell (about 15 and 12 molecules of N2 per supercage) for NaX and BaX, respectively.
CO2 adsorption in microgravimetry
The CO2 adsorption capacity as a function of pressure at 296 K for the NaX and BaX zeolite is shown in Figure 4. The adsorption capacity is higher for NaX than for BaX. The operating conditions did not allow to reach the adsorption plateau which is, usually, observable only for the relative pressure (p/psaturation) higher than 0.2 (see Figure 3 for nitrogen adsorption on NaX and BaX). In fact, the equipment used to perform the adsorption isotherms of CO2 does not operate above a pressure of 1 bar. At a temperature of 296 K, the vapor pressure of CO2 is close to 63 bar. This implies that, in the case of our experiments conducted at 296 K, the relative pressure p/psat is close to 1/63 = 0.016. To calculate the maximum adsorption capacity, the Sips model was used and the maximum adsorption capacity, the adsorbate/adsorbent affinity A, and the n parameter are reported in Table 3.
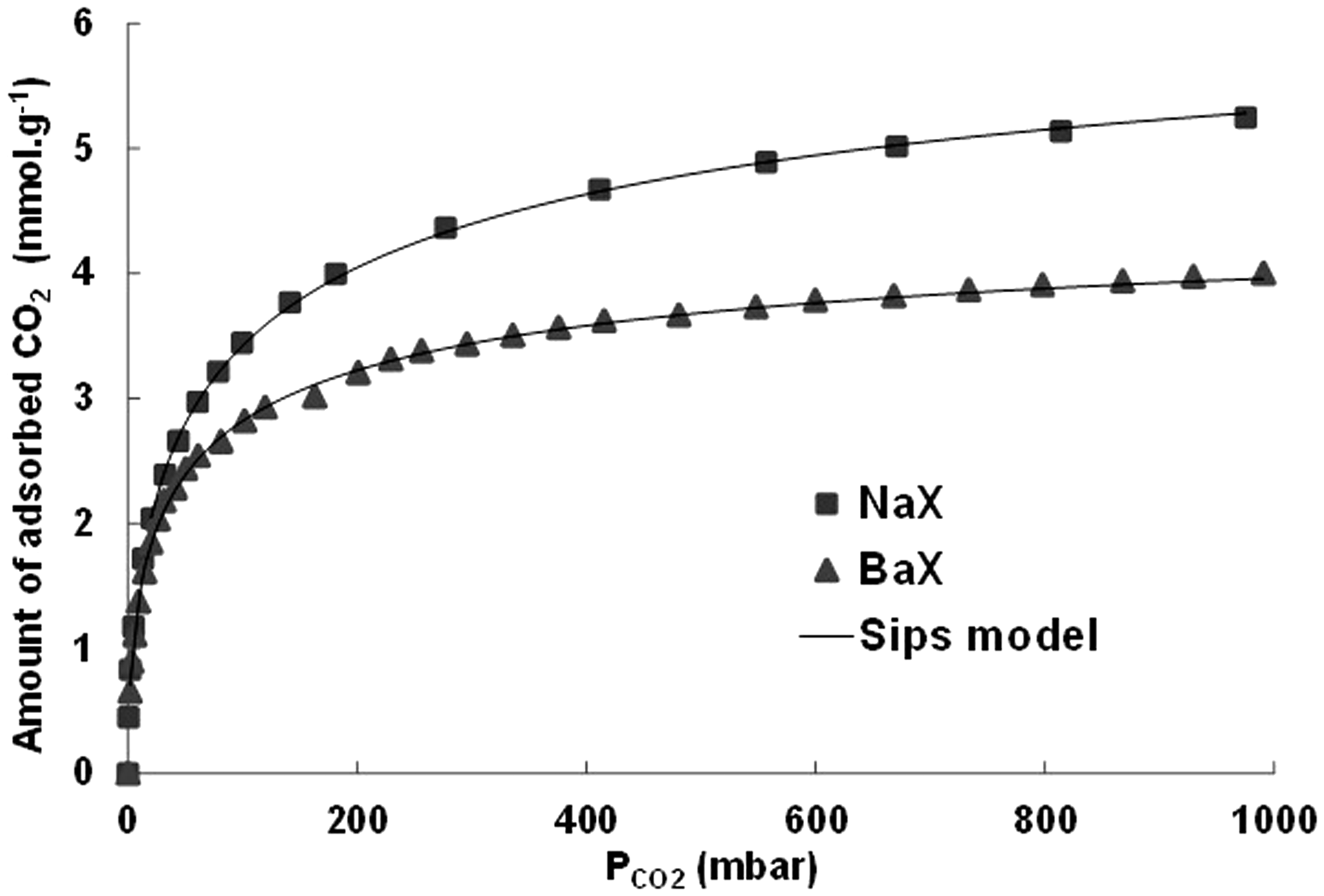
Isotherms of CO2 adsorption at 296 K on the NaX and BaX zeolites.
Parameters of isothermic adsorption of CO2 at 296 K using Sips model.

As observed in the case of adsorption of nitrogen, the maximum amount adsorbed per weight is higher for NaX than for BaX. By repeating the same calculations done in the case of nitrogen adsorption, a maximum amount of 87 and 83 mole of CO2 per mole of unit cell (about 11 and 10 molecules of CO2 per supercage) are obtained, respectively, for NaX and BaX. So, the maximum amount of adsorbed CO2 is independent of the nature of the compensating cation. It is also independent of the number of cations since a cell of NaX contains 86Na+ cations and a cell of BaX contains 43Ba2+ cations. These two observations suggest that CO2 occupies the entire available pore volume. One can thus calculate the density of the CO2 within the zeolite at the maximum filling. For NaX, a maximum capacity of 6.5 mmol g−1 corresponds to a specific capacity of 0.286 g g−1. Knowing the microporous volume (0.300 ml g−1), a density of 0.95 g ml−1 is calculated. A similar calculation leads to a density of 1.09 g ml−1 for BaX. These two high values suggest the existence of a significant effect of confinement of CO2 within the lattice of these two zeolites at the maximum filling that can be attributed to the porous structure, the nature, and the number of cations.
The constant A is related to the adsorbate/adsorbent affinity. The higher the value, the stronger the adsorbate/adsorbent interactions. This parameter is proportional to the energy of interaction. The values of A are of the same order of magnitude for NaX and BaX. The values of n are close from NaX and BaX. They are far higher from to 1 indicating extensive heterogeneity of the two solids. This suggests that the adsorption is located on cationic sites and/or is of a significant effect of containment.
CO2 adsorption/desorption in fixed bed dynamics
CO2 adsorption
The drilling curves obtained at 295 K on NaX and BaX zeolites are shown in Figure 5. The bed composed of NaX zeolite seems more effective than that composed of BaX zeolite. Indeed, the beginning of drilling for zeolite NaX (163 min) occurs later compared to zeolite BaX (86 min). Moreover, the capacities at saturation also confirm this evolution (Table 4). This confirms the results obtained on the evolution of the adsorption capacities of carbon dioxide obtained in microgravimetry on these two samples.

Drilling curves of CO2 (84 ppm) obtained at 295 K on NaX and BaX zeolites. The lines are present to facilitate the reading.
Adsorption capacities of CO2 obtained at 295 K in fixed bed dynamics on NaX and BaX zeolites.

Otherwise, the slopes of the drilling curves of NaX and BaX zeolites are different (Figure 5). For NaX, this drilling can translate weak adsorbate/adsorbent interactions and/or a very fast diffusion of CO2 in zeolitic porosity. On BaX, CO2 is adsorbed slowly until saturation of the bed. In this case, it seems that the adsorbate/adsorbent interactions are strong and/or that the diffusion of carbon dioxide is slow in the pores. The value of the affinity of BaX zeolite determined by Sips model (Table 3) is in agreement with the first hypothesis. These characteristics are to be closer to the porous specific surface and the volume porous measurements. At identical masses, BaX sample has lowest porosity.
CO2 desorption
The evolution of the percentage of CO2 desorbed as a function of the temperature of NaX and BaX zeolites is represented in Figure 6. Desorption profile obtained on NaX shows a maximum peak of desorption (295–373 K) as well as a shoulder (373–483 K). In addition, the total desorption of the physisorbed species continues until 483 K. Indeed, we showed in a previous study in infrared that the NaX zeolite had adsorption sites of physisorbed CO2 until 473 K (Belin et al., 2013). Between 573 and 623 K, NaX zeolite shows also the presence of hydrogenocarbonates (Belin et al., 2013).

Desorption curves of CO2 obtained at 24 K min−1 in fixed bed dynamics on NaX and BaX zeolites.
On the other hand, desorption profile of the BaX zeolite is completely different from that of NaX. BaX zeolite shows two fields of desorption (Figure 6). The first (295–373 K) corresponds to a zone where the desorption of the physisorbed species is mainly observed and the second (423–533 K), a zone where the percentage of the desorbed species is weak. The temperature of total desorption of the physisorbed species on BaX (528 K) is higher than that of NaX (Figure 6). BaX zeolite seems to have adsorption sites of physisorbed CO2 above 473 K.
In fixed bed dynamics, the BaX exchanged zeolite has adsorption sites (between 473 and 533 K) of energy values higher than those of the NaX zeolite as well as strong interactions of adsorbent/adsorbate.
CO2 adsorption in the presence of humidity
Table 5 recapitulates the adsorption capacities of carbon dioxide obtained in the presence of humidity on two adsorbents before drilling and at saturation.
Adsorption capacities of CO2 obtained at 295 K in fixed bed dynamics in the presence of humidity (relative humidity = 50%) on the NaX and BaX zeolites.

The capacities of NaX before drilling and at saturation are, respectively, 0.038 and 0.004 mmol g−1. The quantities of CO2 adsorbed on NaX zeolite in the presence of water are comparable with those obtained by Rege and Yang (2001). A loss of capacity on adsorption is observed on NaX in the presence of water. Brandani and Ruthven (2004) already made these observations in their work. The losses of capacities at saturation observed on NaX zeolite are estimated 91%. Moreover, the ratio of the quantities of CO2 adsorbed only on the quantities of CO2 adsorbed in the presence of water shows that the losses of capacities of this sample due to water are of 98%. It thus appears that during the trapping of carbon dioxide in fixed bed dynamics, the presence of water enormously disturbs the storage of CO2 in NaX zeolite.
The quantities trapped before drilling and to saturation on BaX zeolite (0.039 and 0.007 mmol g−1, respectively) are comparable with those obtained on NaX zeolite (Table 5). In addition, the losses of capacities to saturation estimated on BaX (82%) are lower than those of NaX (91%), just as the losses in the adsorbed quantities due to the presence of water (93%). These results let us think that NaX zeolite is more hydrophilic than BaX. This assumption is in agreement with the losses of mass obtained in thermogravimetry (Table 2). Water indeed influences the trapping of carbon dioxide in zeolites of the type X. However, this influence seems less on exchanged BaX zeolite.
Drilling curves obtained on NaX and BaX zeolites are represented in Figure 7. Both curves show all the same profile with an adsorption field (C/C0 = 0), followed by a strong desorption of CO2 (C/C0 ≫ 1) and then a return to equilibrium (C/C0 = 1).

Drilling curves of CO2 obtained at 295 K on NaX zeolite and on exchanged zeolite BaX in the presence of 50% relative humidity.
The peak of desorption observed on the adsorbents represents a competitive adsorption between carbon dioxide and water. The most polar molecule (H2O) is adsorbed preferentially on some sites of adsorption (Bonenfant et al., 2008) and thus desorbs a part of carbon dioxide.
CO2 desorption: Preferentially adsorption sites of H2O
Desorption profiles of CO2 obtained after saturation of NaX and BaX zeolites in CO2 in the presence of water are shown in Figure 8. For NaX, between 373 and 483 K, no desorption of CO2 was observed while a large peak of desorption of CO2 was observed in the absence of water as it can be seen in Figure 6. This observation suggests that water occupies the adsorption sites of CO2 in the range 373–483 K. However, the adsorption sites of CO2 are not occupied by water between 573 and 623 K corresponding to the chemisorption of CO2.

Desorption curves obtained (24 K min−1) after saturation of NaX and BaX zeolites in CO2 in the presence of water (relative humidity = 50%).
For BaX zeolite, the two peaks observed between 295–323 and 323–423 K are lower than those observed in the absence of water (Figure 6). On the other hand, desorbed species at higher temperature (423–533 K) are always present and in greater proportion (Figure 8). We can thus suppose that these species correspond to hydrogenocarbonates species slightly adsorbed on this BaX zeolite (Siriwardane et al., 2003).
In conclusion, contrary to NaX zeolite, BaX preserves a majority of adsorption sites of the physisorbed carbon dioxide in the presence of water, in particular those at high temperatures (423–533 K).
Conclusion
We carried out the study of preferential adsorption sites of H2O on NaX zeolite and on exchanged zeolite BaX after adsorption/desorption of CO2 in fixed bed dynamics in the presence of humidity.
The results of characterizations of the two samples indicate that the CO2 adsorption takes place into the whole pore volume available for these two zeolites. The adsorption capacity is lower for BaX (4.8 mmol g−1) compared to NaX (6.5 mmol g−1) due to the higher atomic volume of Ba2+ but no pore blocking occurs. These results are confirmed in dynamic CO2 adsorption. Indeed, the drilling beginning for zeolite NaX (163 min) occurs later compared to zeolite BaX (86 min). However in adsorption of CO2 alone, contrary to NaX, on BaX zeolite CO2 is adsorbed slowly. This indicates a slow diffusion of carbon dioxide in the pores of BaX zeolite.
The profiles of CO2 desorption alone on these zeolites revealed two distinct zones. The first, at low temperature (295–533 K), corresponding to adsorption sites of the CO2 molecules which are physisorbed, and the second, to adsorption sites of chemisorbed CO2. At low temperatures, BaX zeolite has adsorption sites (473–533 K) of energy values higher than those of the zeolite of NaX as well as strong interactions of adsorbent/adsorbate.
In dynamic CO2 adsorption in the presence of humidity (relative humidity 50%), shows that water influences the adsorption of carbon dioxide in both NaX and BaX zeolites. However, this influence seems less on exchanged zeolite BaX. The desorption profile of CO2 obtained in the experiment on NaX zeolite shows that water occupies all the adsorption sites between 373 and 483 K. BaX zeolite shows a loss of the species present between 295 and 323 K and a decrease of quantity of species between 323 and 423 K. In this temperature range, water also occupies the preferential adsorption sites on BaX zeolites. Contrary to NaX zeolite, BaX preserves a majority of adsorption sites of physisorbed CO2 in the presence of water, in particular the adsorption sites located in the interval of temperature 423–533 K.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.