Abstract
Enhancement of the physicomechanical properties of rubber can be achieved by the incorporation of reinforcing fillers, such as clay. Cation-exchanged zeolites (CEZes) were prepared by exchanging the native Na+ ions with single inorganic cations (magnesium (Mg), zinc (Zn), or strontium) and/or double cations (Zn-Mg), then they were processed as reinforcing fillers in the two type of synthetic rubber, acrylonitrile-butadiene rubber (NBR) and styrene-butadiene rubber (SBR). Characterization of modified zeolite (Ze) was implemented using infrared spectroscopy, X-ray diffraction, and scanning electron microscopy. The aim of this article is to study the influence of Ze and CEZe loadings on the rheometrical and physicomechanical properties of both NBR and SBR composites, also the thermogravimetric analysis was examined and the results were discussed. The results showed that the composites exhibited remarkable improvements in tensile strength, elongation at break, and hardness in the presence of CEZe and also an increase in thermal stability was reported.
Introduction
Rubber possesses unique properties in terms of high elasticity, durability over multiple deformations, chemical and physical stability as well as relative ease of manufacturing, which renders it as recommended and irreplaceable material in a wide range of applications. 1 Meanwhile, it has undesirable properties like low tensile strength (TS) and more plastic and sticky properties on heating in organic solvent.
Reinforcement with filler is a promising technique that improves its undesirable properties besides the improvement of the mechanical, thermal, and physical properties of the formed polymer composites. 2,3 Carbon black is considered as active filler to enhance and reinforce rubber properties, but it is the cause of its black coloration.
On the other hand, for producing light-colored rubbers, carbon-black is replaced by light-colored fillers such as calcium carbonate, clay, or silica. 4 –6
Sodium aluminum silicate hydrate zeolite (sodium zeolites (Na-Zes)) is crystalline, hydrated aluminosilicate compound that has three-dimensional structures arising from a framework of tetrahedral units of SiO
Zeolites (Zes) are nontoxic materials that are used worldwide and are known for their remarkable thermal and chemical stability, versatility as well as uniform porosity. 10,11 It enhances the properties of rubber by increasing the TS hardness, elongation at break, and density. 12
Zes possess many advantages; they are cost savings in the production process, they can adsorb undesirable substances, and they can improve the properties of finished products. 9
The present study aims to utilize single cation-exchanged zeolites (CEZes) of magnesium (Mg), zinc (Zn) or strontium (Sr) and double cations of Zn-Mg as reinforcing fillers in acrylonitrile-butadiene rubber (NBR) and styrene-butadiene rubber (SBR) composites. The results revealed the high improvement of physicomechanical properties beside the thermal barrier against mass loss than those containing Na-Ze.
Materials
NBR is produced by Bayer AG, Germany with molecular weight MW of 63,000 g mol−1, Germany. Its acrylonitrile content is 34 ± 1%, density 1.17 g cm−3, and Moony viscosity ML (1 + 4) is 65 7 at 100°C. SBR copolymer has styrene content 23.5% with molecular weight MW of 140,000 g mol−1, specific gravity of 0.945 ± 0.005, Moony viscosity ML (1 + 4) 52 ± 3 at 100°C, and glass transition Tg = −60°C, and it was obtained from Transport and Engineering Company (TRENCO) Alexandria. Na-Ze molecular sieve was purchased from Aldrich, Germany. It is fine powder with white color having the following specifications: chemical formula: NaAlSi2O6·H2O, average particle size (Fisher) <10.0 micron, loss on ignition: 6.3% max. N-Cyclohexyl-2-benzothiazole sulphenamide (CBS) is used as accelerator of trade name: Rhenogran® CBS-80 and Vulkacit Zinc oxide and stearic acid are used as activators with specific gravity 5.55–5.61 and 0.9–0.97, respectively at 15°C. They were supplied by Aldrich Company, Germany. Elementary sulfur is used as a vulcanizing agent. It is a fine powder with pale yellow color; its density is 2.04–2.06 g cm−3 at room temperature (25°C ± 1) and it was supplied by Aldrich Company, Germany. Magnesium nitrate hexahydrate (Mg(NO3)2·6H2O) with MW: 256.41 g mol−1 and 99% purity and zinc nitrate hexahydrate (Zn(NO3)2·6H2O) with MW: 297.47 g mol−1 and 99% purity were supplied by WINLAB, UK. Strontium nitrate Sr(NO3)2 with MW: 211.63 g mol−1, and 99% purity, was produced by Oxford, India.
Experimental techniques
Preparation of cation-exchanged zeolites (CEZes)
Five grams Na-Ze were soaked in 0.1 N Mg2+, Zn2
+, Sr2+ to prepare single cations or in a soluble solution containing 1:1 of Zn-Mg in the case of preparing the double cations as shown in Figure 1. The solution was stirred for 1 h at ambient temperature, then the formed paste was filtered. Steps 1 and 2 were repeated several times to make sure that no more cation exchange can take place.
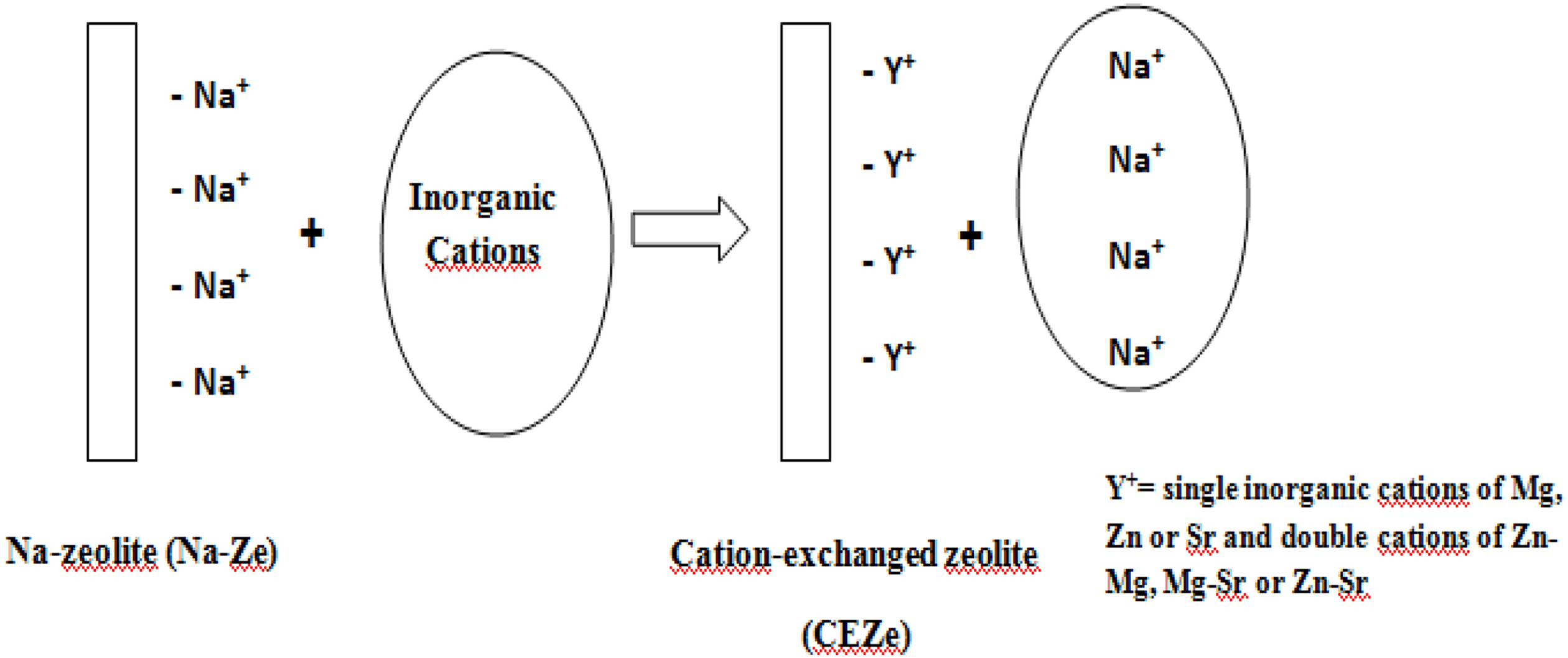
Preparation of CEZe.
Preparation of rubber samples
The cure characteristics of the rubber composites were determined using Monsanto Rheometer (model 100) (USA) at 152 ± 1°C according to ASTM D2084. The mechanical properties such as TS and elongation at break (%) of the rubber compounds were determined according to ASTM D412.
The formulations of NBR or SBR loaded with 6 and 12 phr of Na-Ze and CEZe are represented in Tables 1 and 2. The vulcanized sheets prepared for mechanical tests were cut into five individual dumb-bell-shaped specimens by steel molder of constant width (4 mm). The thickness of the tested specimen was determined using a gauge calibrated in hundredths of millimeter. A working part of size 15 mm was chosen for each test specimen.
Formulations of NBR loaded with different concentrations of zeolite and cation-exchanged zeolite.a

NBR: acrylonitrile-butadiene rubber; Na: sodium; Mg: magnesium; Zn: zinc; Sr: strontium; Ze: zeolite.
a Base recipe in phr (is part per hundred parts of rubber): NBR (acrylonitrile butadiene rubber) 100; stearic acid 2; zinc oxide 5; CBS (N-cyclohexyl-2-benzothiazole sulfenamide) 1; DOP (dioctyl phthalate) 1; TMQ (polymerized 2, 2, 4-trimethyl-1, 2-dihydroquinoline) 1; sulfur 2.
Formulations of SBR loaded with different concentrations of the zeolite and cation-exchanged zeolite.a
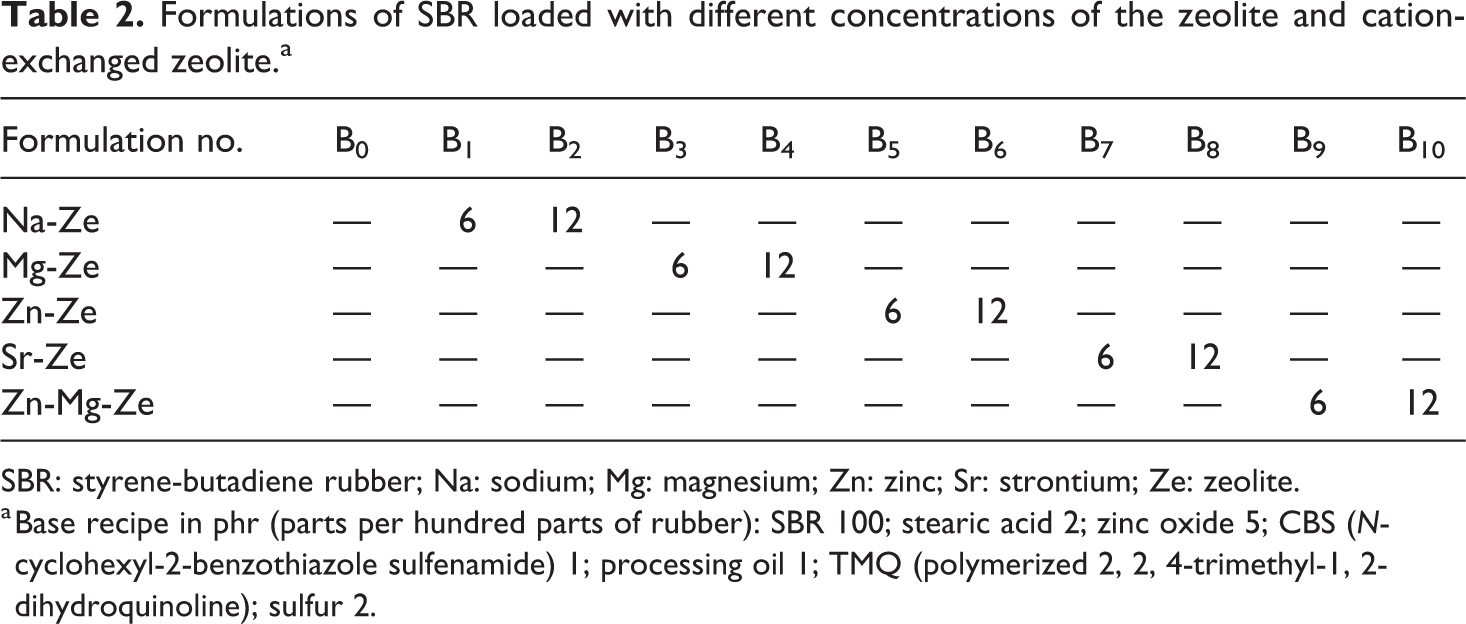
SBR: styrene-butadiene rubber; Na: sodium; Mg: magnesium; Zn: zinc; Sr: strontium; Ze: zeolite.
a Base recipe in phr (parts per hundred parts of rubber): SBR 100; stearic acid 2; zinc oxide 5; CBS (N-cyclohexyl-2-benzothiazole sulfenamide) 1; processing oil 1; TMQ (polymerized 2, 2, 4-trimethyl-1, 2-dihydroquinoline); sulfur 2.
Hardness (shore A) of the vulcanized samples was measured using the shore A durometer according to ASTM D2240 by pressing circular sheet with radius 12 mm and thickness 6 mm, in a hot press at temperature 152 ± 1°C with respect to the investigated optimum cure time.
The swelling test was carried out according to ASTM D3616.
In this test, the specimens were cut from the sheets and weighed using an electric balance, followed by immersion in toluene for 24 h at room temperature to extract any uncross-linked component and low molecular weight materials from the samples, and then they were dried until constant weight. These samples (3–5 per test) were first weighed to give the initial weight, and then immersed in toluene till reaching the equilibrium swelling. The interaction between rubber and filler in rubber composites has been investigated using the equation of Lorenz and Parks 15

where Qf and Qg are the equilibrium swelling in the presence of filler and gum rubber, respectively, z is the weight fraction of filler in the vulcanizate, while a and b are constants characteristic of the system.
The equilibrium swelling (Q%) can be calculated using equation (2)
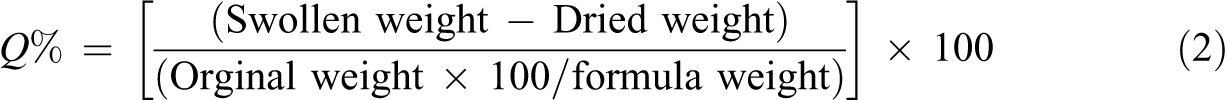
The cross-link density was measured by applying the Flory Rehner equation 16,17 as given in equations (3) to (5)
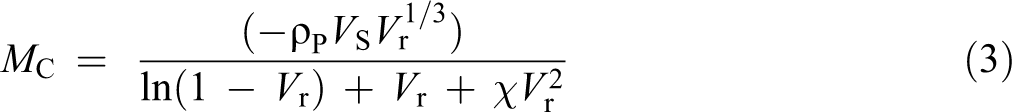


where Mc is the molecular weight between cross-links, ρP is the density of rubber (for NBR = 1.17 and for SBR = 0.913), Vs is the molar volume of toluene (106.35 cm3 mol−1), Vr is the volume fraction of swollen rubber, Qm is the swelling mass of rubber filled with CEZe in toluene, while χ is the interaction coefficient between rubber network and toluene (for NBR = 0.39 and SBR = 0.446), and Vc is the degree of cross-link density.
X-ray fluorescence
Chemical composition of Na-Ze and CEZes were determined using Axios, sequential WD-X-ray fluorescence (XRF) spectrometer, PANalytical 2005, USA.
Infrared spectroscopy’
Fourier transform infrared (FTIR) spectra were performed using a JASCO FTIR-6100E FTIR spectrometer (Japan) operated in the absorption mode, in the wave number range 4000–400cm−1 after mixing with potassium bromide and pressed in the form of discs. The spectra were collected at a resolution of 4 cm−1.
X-ray diffraction
X-ray powder diffraction patterns were obtained at room temperature using a Philips diffractometer (type PW1390) (Japan) by employing nickel-filtered CuKα radiation source (k = 1.5404 Å), Japan. The diffraction angle, 2θ, was scanned at a rate of 5° min−1.
Scanning electron microscopy
Scanning electron microscopy (SEM) was performed using JEOL (JXA-840A) electron probe microanalyzer, Japan. This technique is used to determine the shape and the distribution of filler particles in the rubber matrix. Rubber specimen was broken in liquid nitrogen and its cross section was covered with a thin layer of gold.
Results and discussion
Characterization of CEZes
XRF analysis
It is clear from Table 3 that Na-Ze is mainly composed of silica and aluminum in the following elemental composition silicon dioxide 38.72% and aluminum oxide 33.5%. The cation exchange capacity (CEC) of cations present in Zes can be determined using XRF. It can be concluded that the CEC of sodium cation by Mg was about 73%. This may be due to that Mg2+ ions have higher ionic radius than Na
XRF results of zeolite and cations-exchanged zeolites.
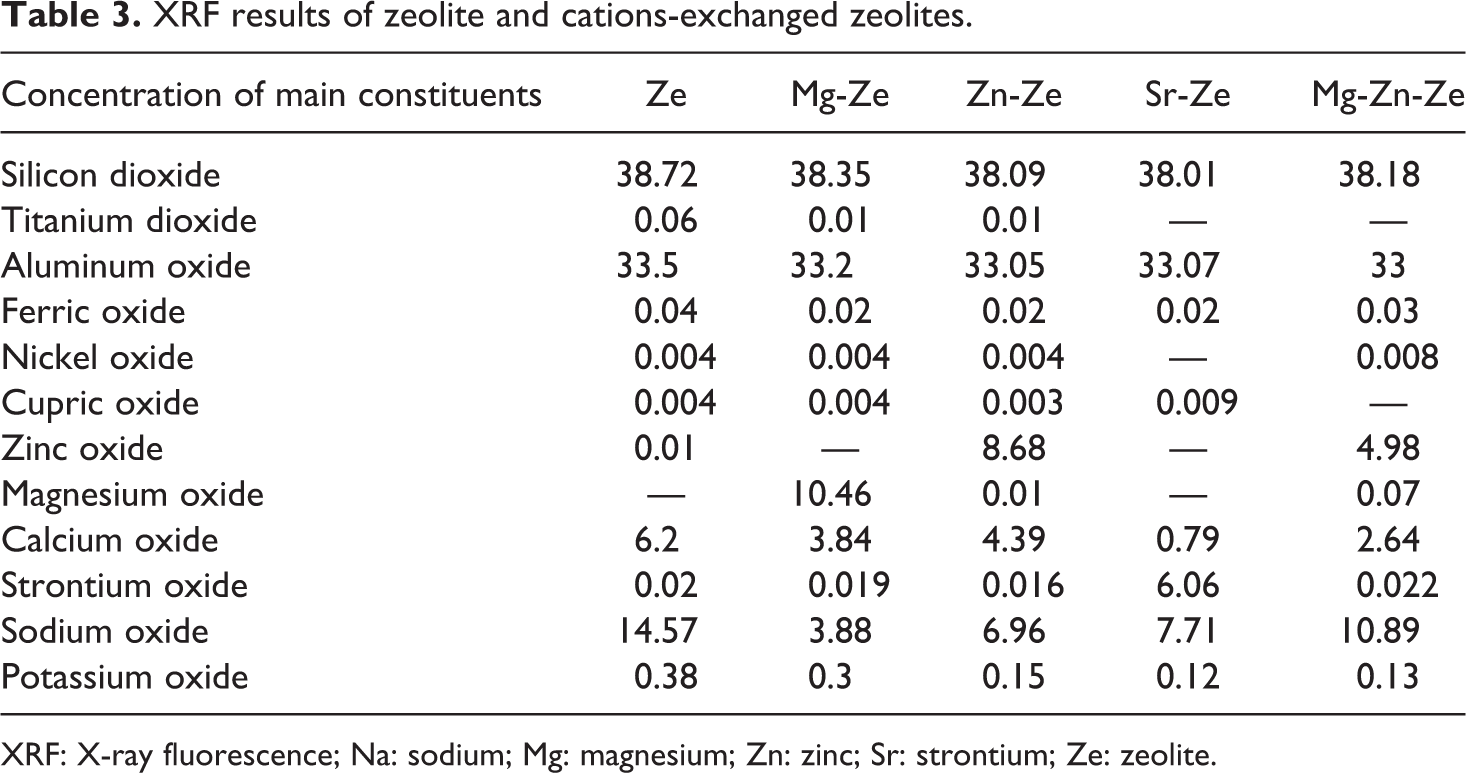
XRF: X-ray fluorescence; Na: sodium; Mg: magnesium; Zn: zinc; Sr: strontium; Ze: zeolite.
In the case of single cation:
The CEC of sodium cation by Zn and Sr was about 52% and 47%, respectively.
In the case of double cations: The CEC of sodium cation by Zn and Mg was about to 34% and 21%, respectively.
The ionic radius of the cation beside the affinity of Ze toward this cation is the most important factors affecting the concentration of the hosted cation. 14
FTIR spectroscopy
The IR spectra of Ze in Figure 2 revealed several absorption bands; bands in the region of 3438–2930 cm−1 are assignable to Si–OH, Si–OH–Al, and –OH hydroxyl groups species.
18
On the other hand, the absorption band in the region of 1658 cm−1 corresponds to O–H bending vibration, while bands in the region 900–1200 cm
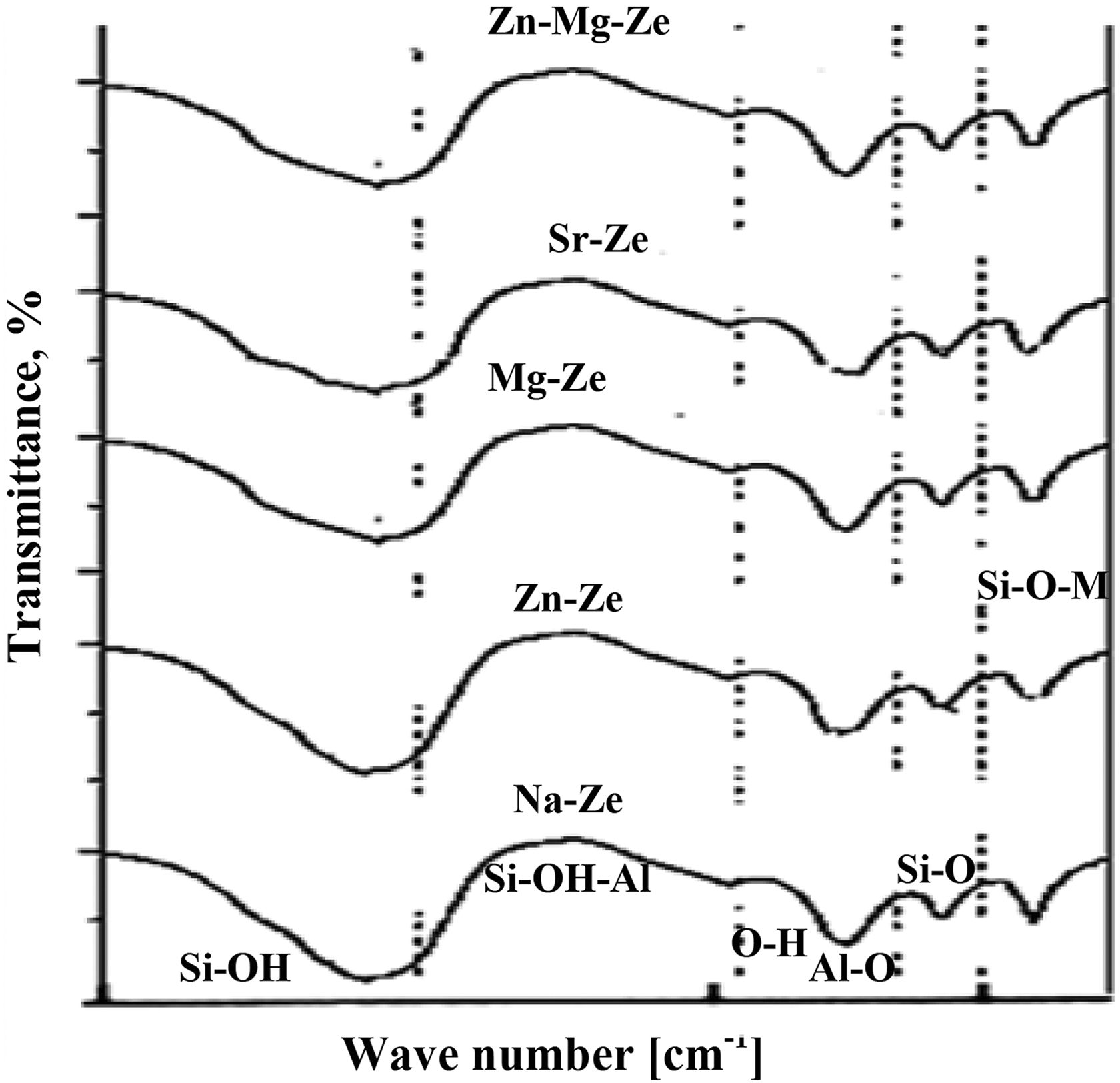
FTIR spectra of Na-Ze and cations exchange zeolite (Mg-Ze, Zn-Ze, Sr-Ze, and Zn-Mg-Ze).
Also, absorption bands of fundamental vibrations around 1008 cm
X-ray diffraction
Ze is identified by X-ray diffraction peaks which confirm high purity and good crystallinity as presented in Figure 3. The type of Ze is zeolite A, and its main peaks appeared at d Å12.278, 3.2803, 2.614, and 1.817. All CEZes possessed the same structure as Na-Ze, that is, no collapse or destruction has appeared in the different Zes, only some decrease in the intensity of the patterns was detected which means that a decrease in the degree of crystallinity has occurred. 14
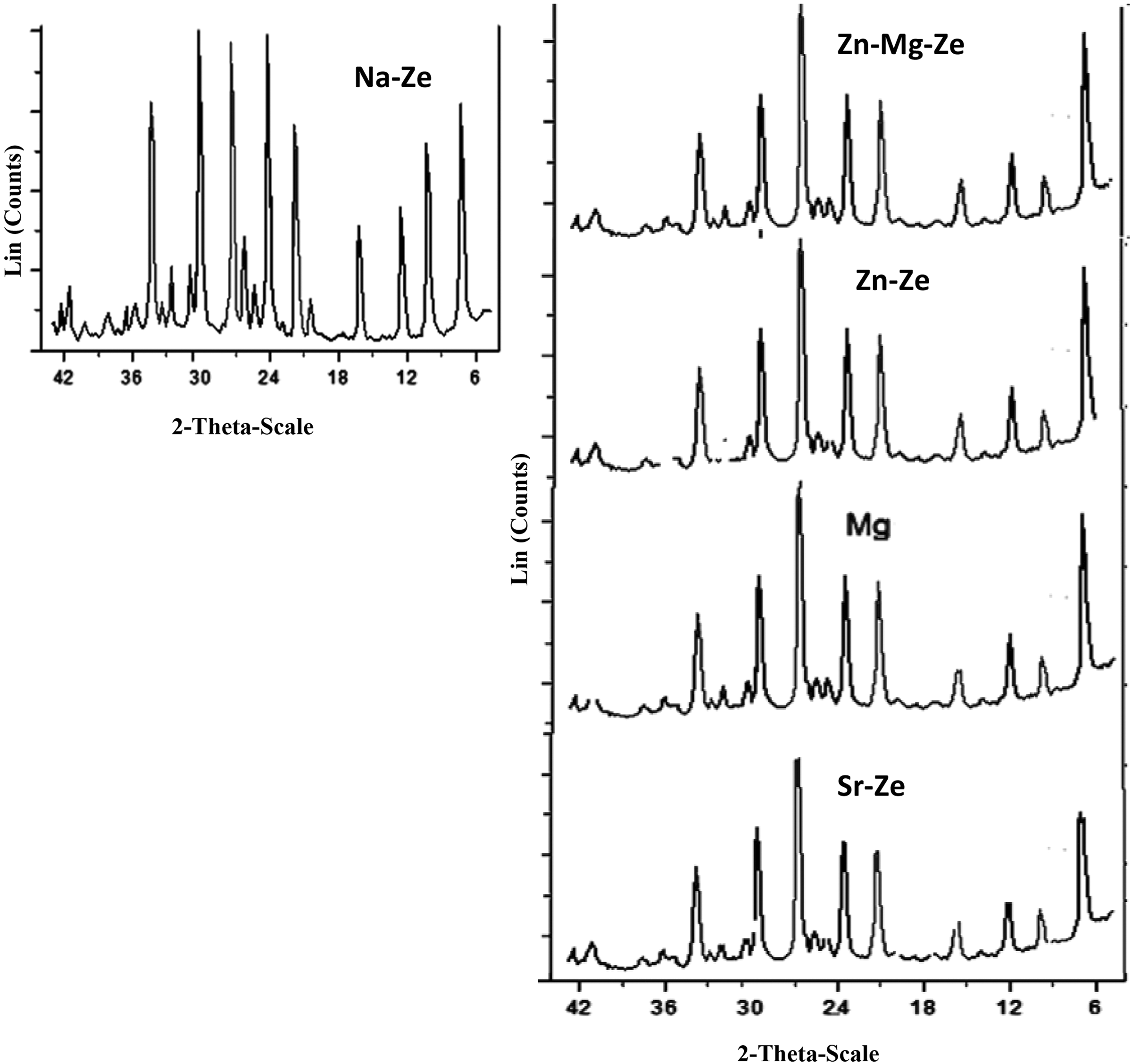
XRD chart of Na-Ze and cations exchange zeolite (Mg-Ze, Zn-Ze, Sr-Ze, and Zn-Mg-Ze).
Scanning electron microscopy
SEM morphologies of Na-Ze and CEZe are shown in Figure 4. In one step, the cations exchanged with single and double cations must produce Zes without any change in structure and consequently no detectable change in SEM images was found. 20 It can be seen that the shapes of both Na-Ze and CEZe were regular cubic crystals with sharp edges, showing slippery between the layers that occur at different levels, leading to higher mechanical properties. These cubic structures showed that the Ze has not changed by the presence of the different cations. 21

SEM images of Na-Ze and CEZe at magnification 104×. (a) Na-Ze, (b) Mg-Ze, (c) Zn-Ze, (d) Sr-Ze, and (e) Mg-Zn-Ze.
Characterization of rubber composites
Curing properties
Table 4 revealed that the minimum torque ML, which reflects minimum viscosity of the mixes and is a measure of filler–filler interaggregate formation, slowly increases by adding 12 phr of Sr-Ze and Zn-Mg-Ze. This increase is related to the presence of these cations in NBR matrix which reduced the movability of the macromolecular chains of these composites, while the maximum torque MH increases after adding Mg-Ze and Zn-Mg-Ze.
Rheological characteristics of NBR composites loaded with Na-Ze and cation-exchanged zeolite.
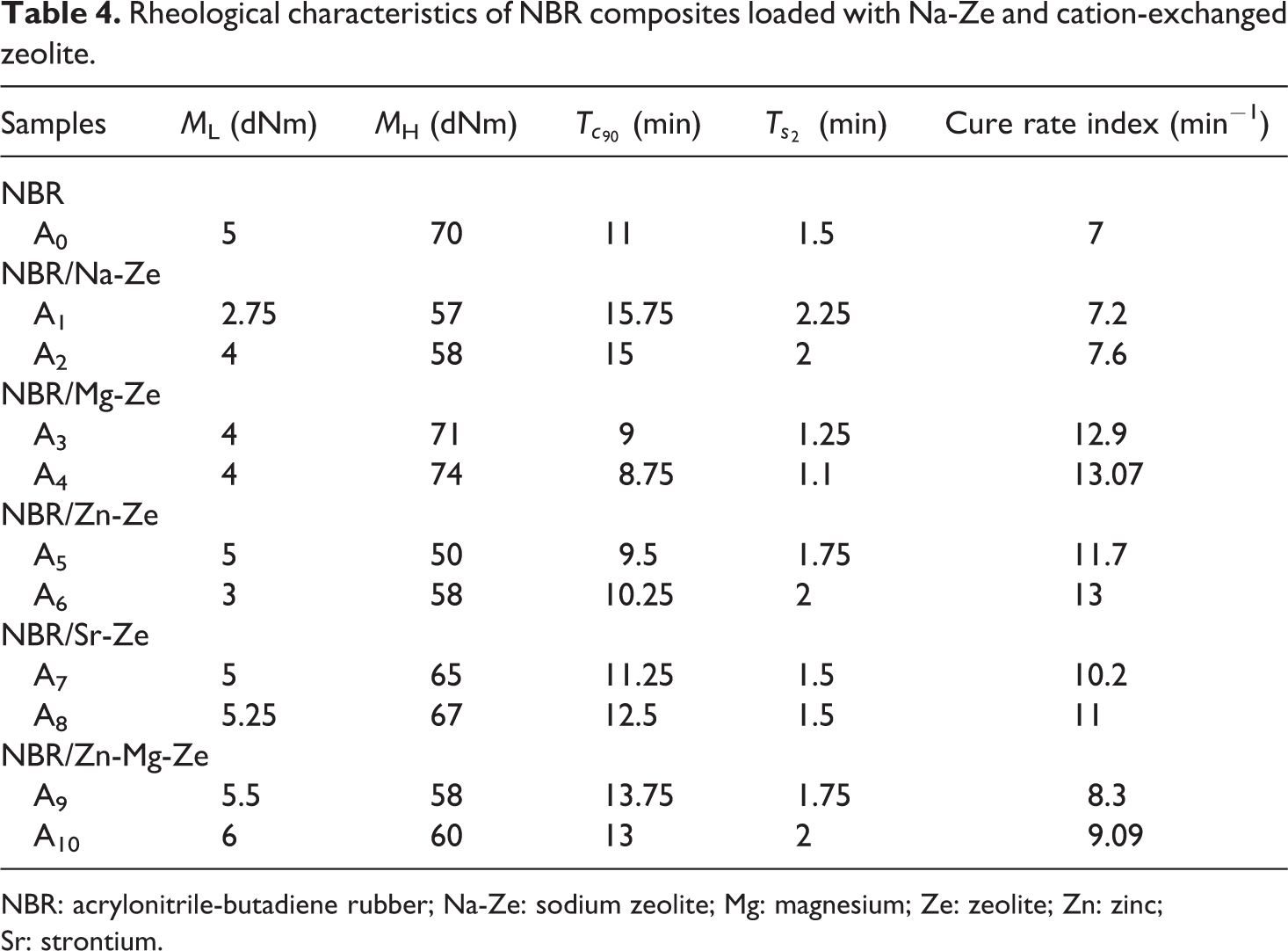
NBR: acrylonitrile-butadiene rubber; Na-Ze: sodium zeolite; Mg: magnesium; Ze: zeolite; Zn: zinc; Sr: strontium.
Scorch time (

It is seen that CRI increases with the addition of CEZe and it supports the activation of the cure reaction especially at 12 phr Mg-Ze and Zn-Ze and these cations increased the number of reactive sites on the rubber molecules which require more time for cross-linking in rubber composites. At loading of Na-Ze and Zn-Mg-Ze, a deactivation of the cure process was observed due to the increase of the optimum cure time,
For SBR composites, the cure characteristics are given in Table 5. It can be deduced that the incorporation of Na-Ze and CEZe in SBR matrix increases the MH and ML values, indicating that increasing the filler loading rendered the composites of more difficult processability. The decrease of torque value at 6 phr Mg-Ze may also be attributed to the reaction between aluminum and silica in Ze with the curative agent, which resulted in slight increase in the cross-link density and decrease in the torque. On the other hand, the optimum cure time (
Rheological characteristics of SBR composites loaded with Na-Ze and cation-exchanged zeolite.
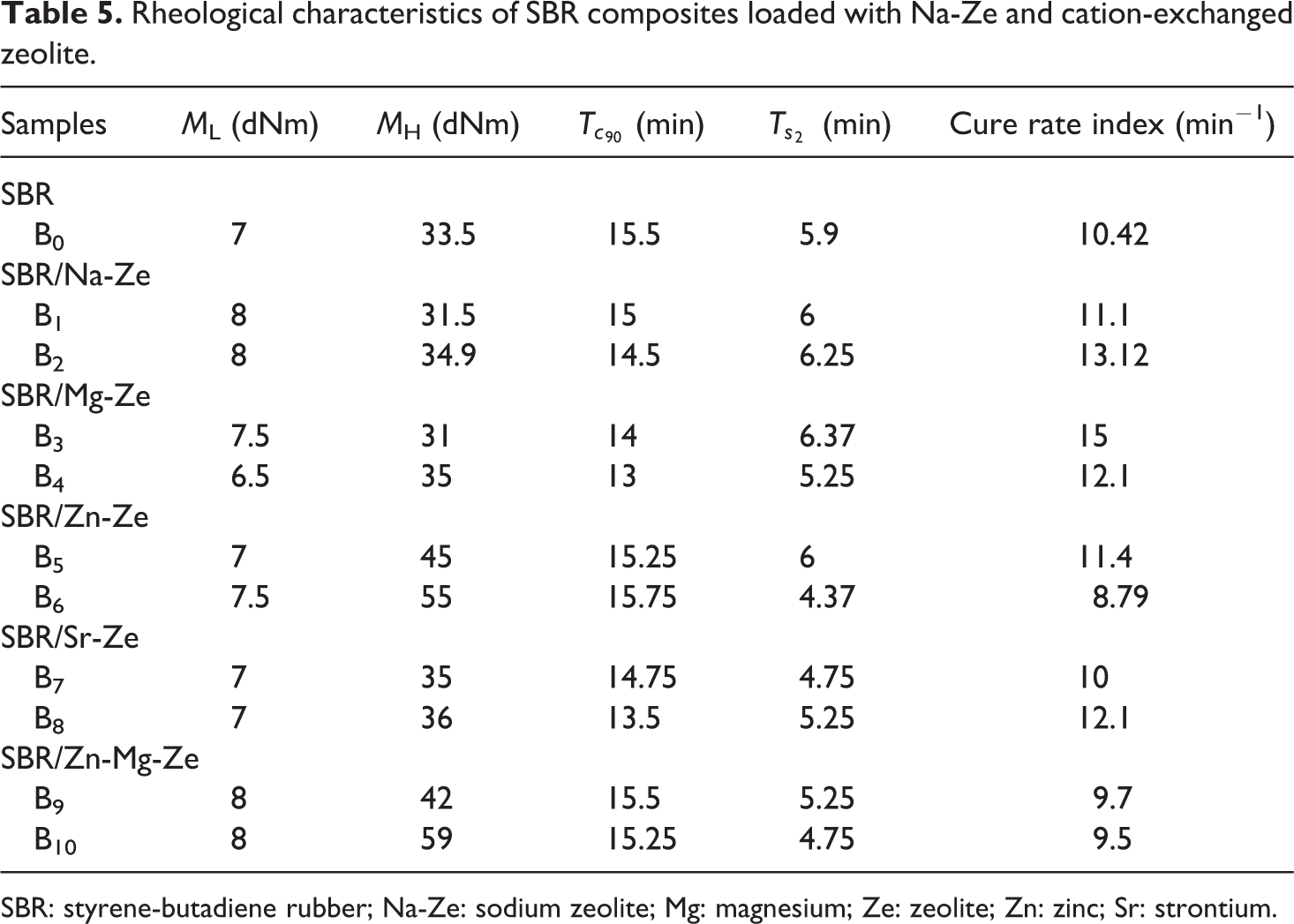
SBR: styrene-butadiene rubber; Na-Ze: sodium zeolite; Mg: magnesium; Ze: zeolite; Zn: zinc; Sr: strontium.
The reason for lower cure time values in the case of SBR containing 12 phr of Mg-Ze may be due to the ability of the magnesium oxide (MgO) particles to accelerate the vulcanization reaction in SBR, especially at higher loadings, it can also act as activator due to its large surface area, which offers greater sites for interactions with the curing agents during the vulcanization mechanism. 23,24
On the other hand, the values of CRI of SBR composites containing Mg-Ze were higher than those loaded with Na-Ze and other cations, which can be declared by the presence of inorganic cations that can act as effective vulcanizing agents. 23,25
Curing time is lower in NBR matrix than in SBR due to its strong interaction with aluminum silicate which is the main component of Ze.
Phyiscomechanical properties
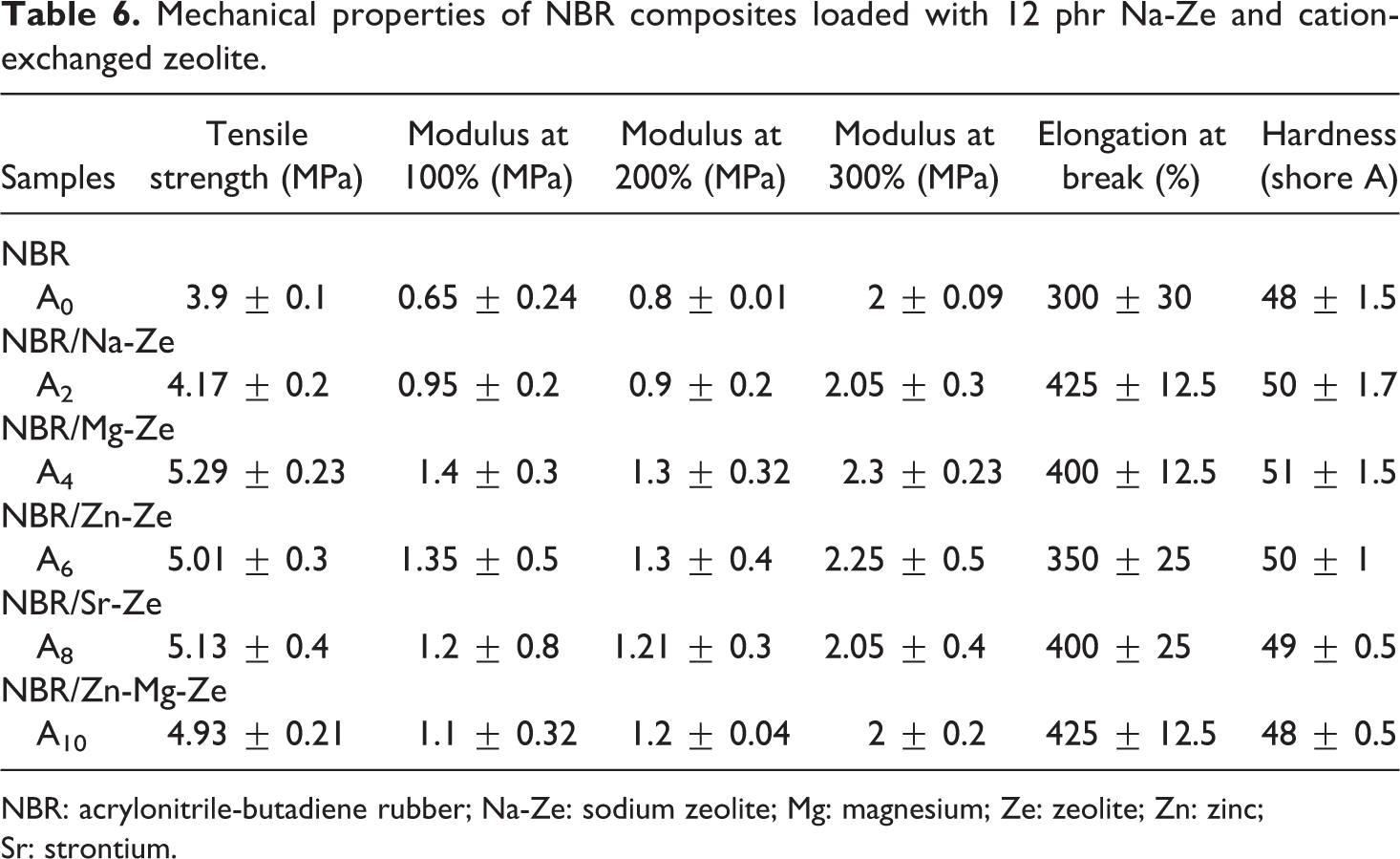
Figure 5(a) and Table 6 illustrate the TS of NBR composites filled with Na-Ze and the different CEZes. It was found that at the same filler loading, NBR composites filled with CEZe showed better values of TS, especially those containing Mg-Ze which exhibited higher TS than the other cations, this may be due to the good interactions between the MgO particles with NBR compounds. 26
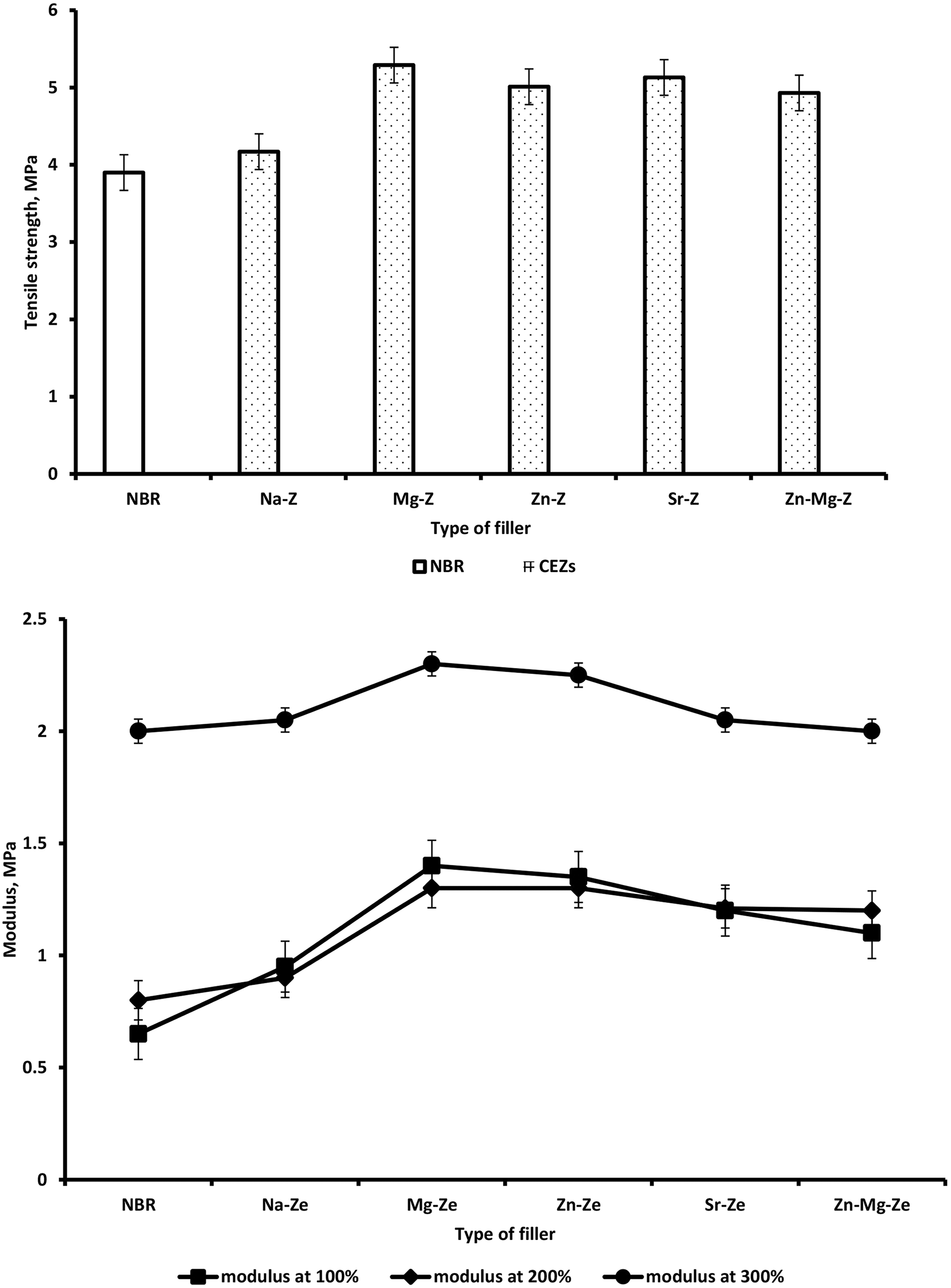
Variation of (a) tensile strength and (b) modulus at 100%, 200%, and 300% elongation for NBR and NBR composites loaded with 12 phr Na-Ze and CEZe.
Mechanical properties of NBR composites loaded with 12 phr Na-Ze and cation-exchanged zeolite.
NBR: acrylonitrile-butadiene rubber; Na-Ze: sodium zeolite; Mg: magnesium; Ze: zeolite; Zn: zinc; Sr: strontium.
The substitution of sodium cations by inorganic cations (single and double) showed more stress and exhibited higher TS because of the high flexibility of the cation linkages which render them capable of withstanding high stresses. It can be seen that TS decreases with higher Na-Ze loading (12 phr) resulting in weak filler–rubber interaction, due to the formation of agglomerations. It was also clearly seen that in Figure 5(b) and Table 6 that modulus at 100, 200, and 300% elongation increases slightly for NBR composites containing Na-Ze and CEZe. Hence, the effect of the interaggregate association should disappear, and the interaction between rubber and filler may now play an important role in modulus. Figure 6 and Table 6 illustrate that the elongation at break of NBR composites gradually increases depending on the type of CEZe. 27
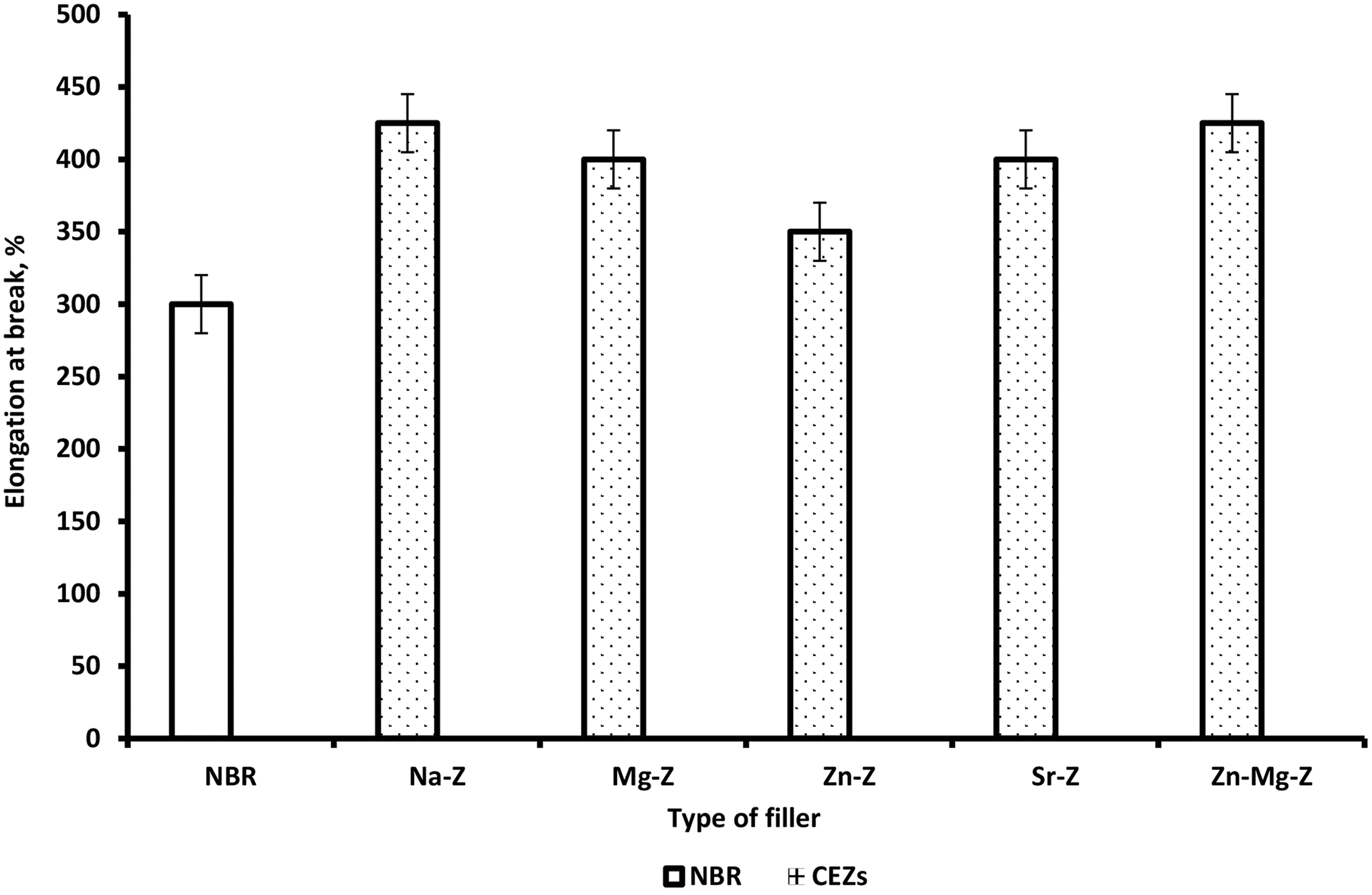
Variation of elongation at break for NBR and NBR composites loaded with 12 phr Na-Ze and CEZe.
Hardness values were improved for rubber composites filled with Na-Ze and CEZe compared to the respective neat rubber. NBR chains trapped between the CEZe particles forming the bonded rubber possessed the rigidity to the composites. Therefore, the presence of CEZe in the NBR composite could carry much more stress and slightly increased the hardness than neat NBR as shown in Figure 7 and Table 6.
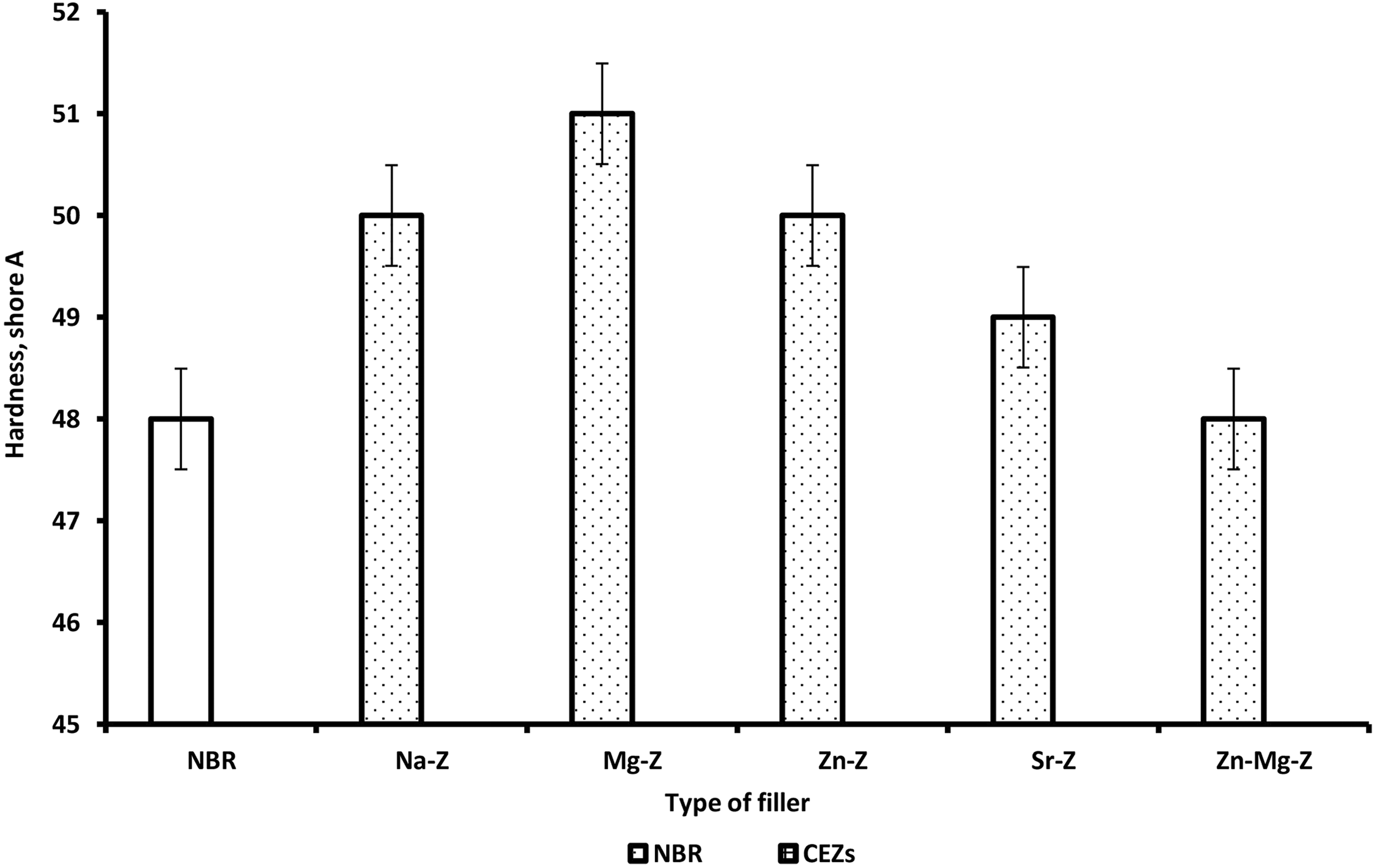
Variation of hardness for NBR and NBR composites loaded with 12 phr Na-Ze and CEZe.
The effect of Na-Ze and CEZe on TS of SBR composites is presented in Figure 8(a) and Table 7 which indicate that the TS was improved for composites loaded with Na-Ze and CEZe. This can be possibly explained by the fact that, Na-Ze is mainly composed of silica and aluminum which improves the mechanical properties of the vulcanized rubber. On the other hand, Figures 8 and 9 showed a significant increase in TS and elongation at break upon adding CEZe, indicating a clear reinforcing effect. Moreover, the elongation at break values of SBR loaded with Na-Ze and CEZe where in the same.
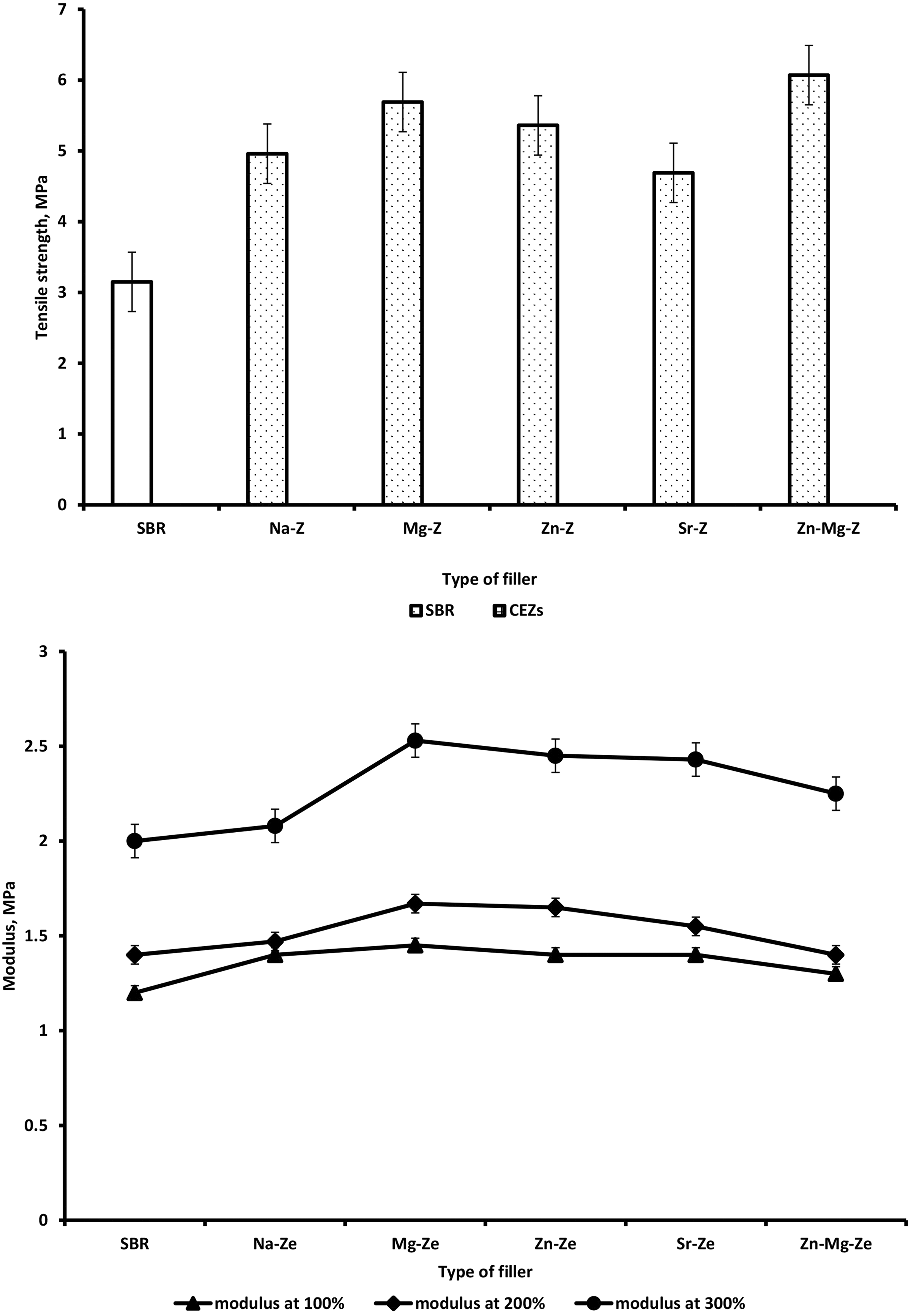
Variation of (a) tensile strength and (b) modulus at 100% elongation for SBR and SBR composites loaded with 12 phr Na-Ze and CEZe.
Mechanical properties of SBR composites loaded with 12 phr Na-Ze and cation-exchanged zeolite.
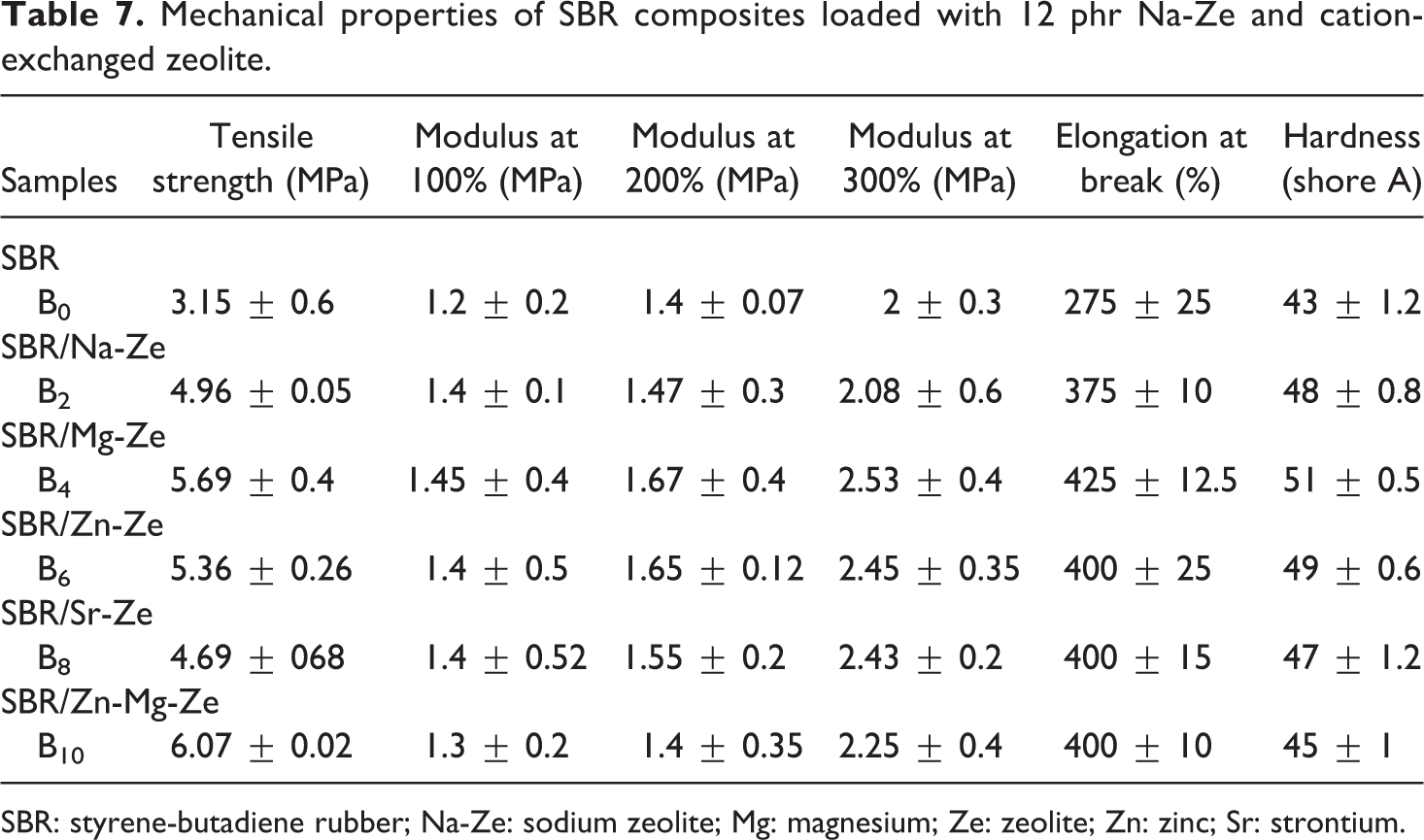
SBR: styrene-butadiene rubber; Na-Ze: sodium zeolite; Mg: magnesium; Ze: zeolite; Zn: zinc; Sr: strontium.
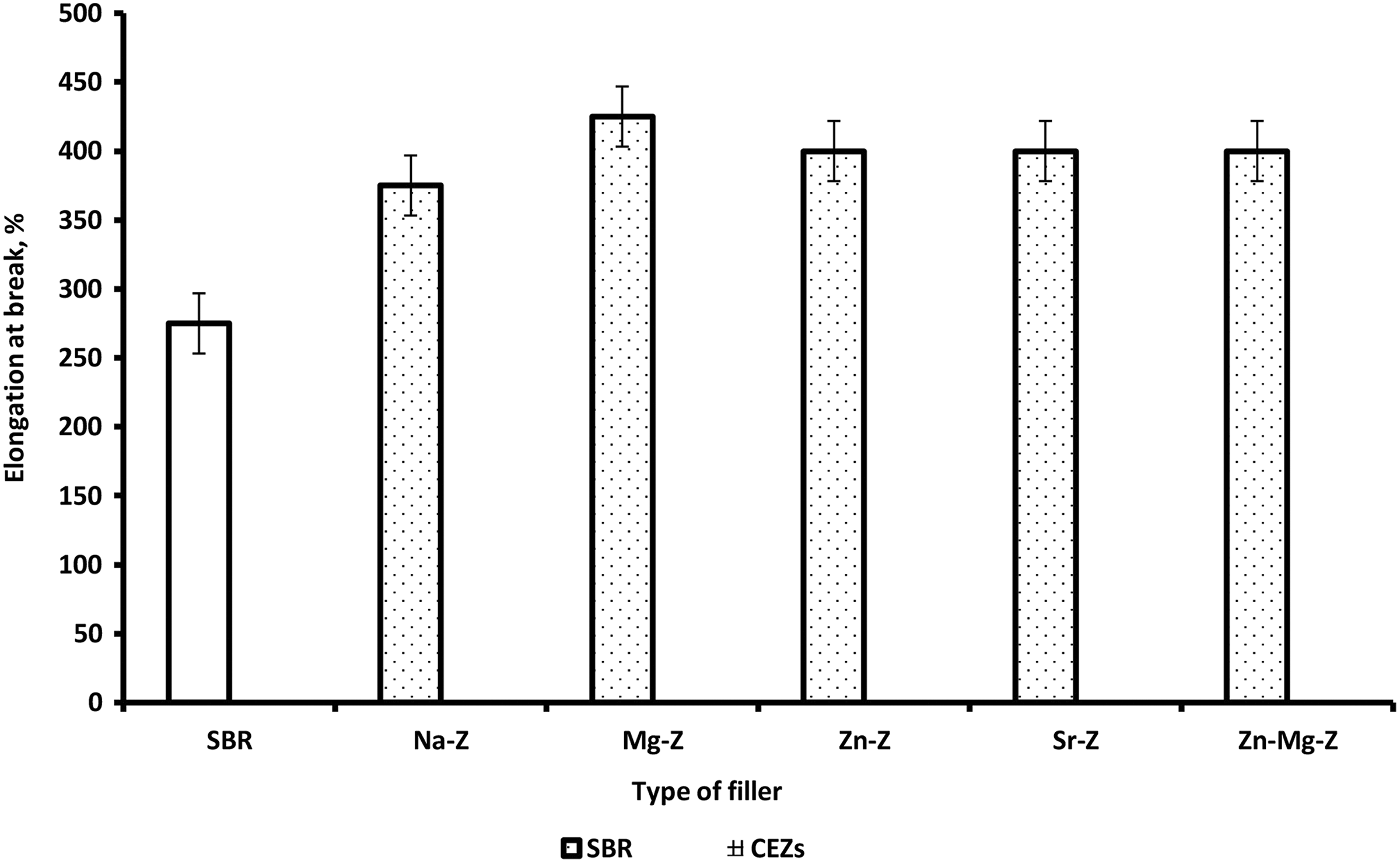
Variation of elongation at break for SBR and SBR composites loaded with 12 phr Na-Ze and CEZe.
In the case of SBR/Mg-Ze and SBR/Zn-Mg-Ze composites, high TS values coupled with high elongation were determined due to the good dispersion of these cations leading to improving the interactions between CEZe and SBR, and allowing the reorientation of the platelets along the stress direction. Also Figure 8(b) and Table 7 showed that improvement in the modulus at 100, 200, and 300% elongation after adding CEZe into SBR. This can be attributed to the introduction of CEZe into SBR matrix led to an increase in the cross-linking between polymer molecules reducing their mobility, and thus increasing the modulus and at the same time improving the hardness and other mechanical parameters. 28 –30
The hardness of SBR composites slightly increased with loading of Na-Ze and CEZe as the plasticity of the rubber chain is increased (Figure 10), resulting in rigidity on incorporating more filler in the rubber matrix. 31
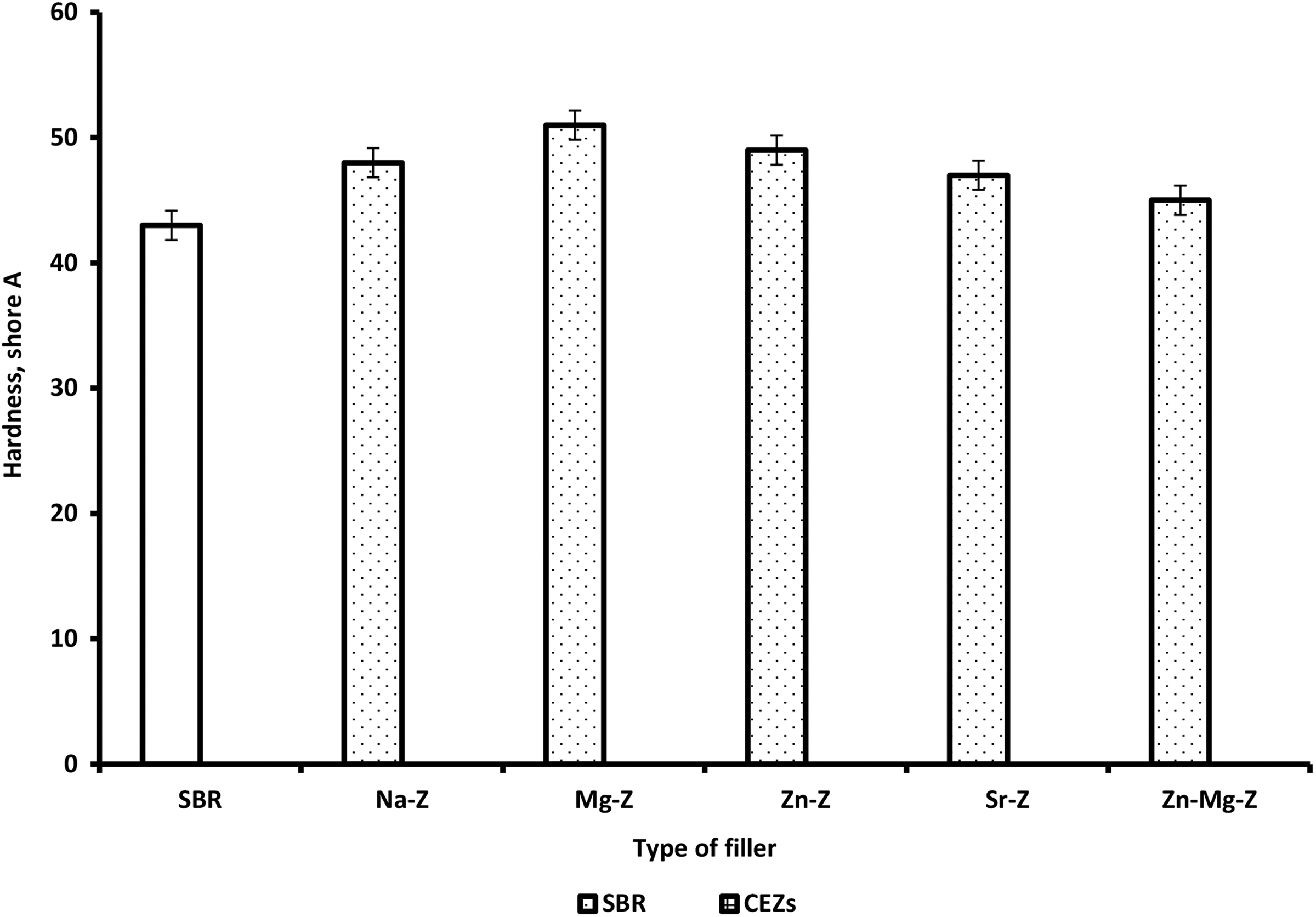
Variation of hardness for SBR and SBR composites loaded with 12 phr Na-Ze and CEZe.
Swelling properties
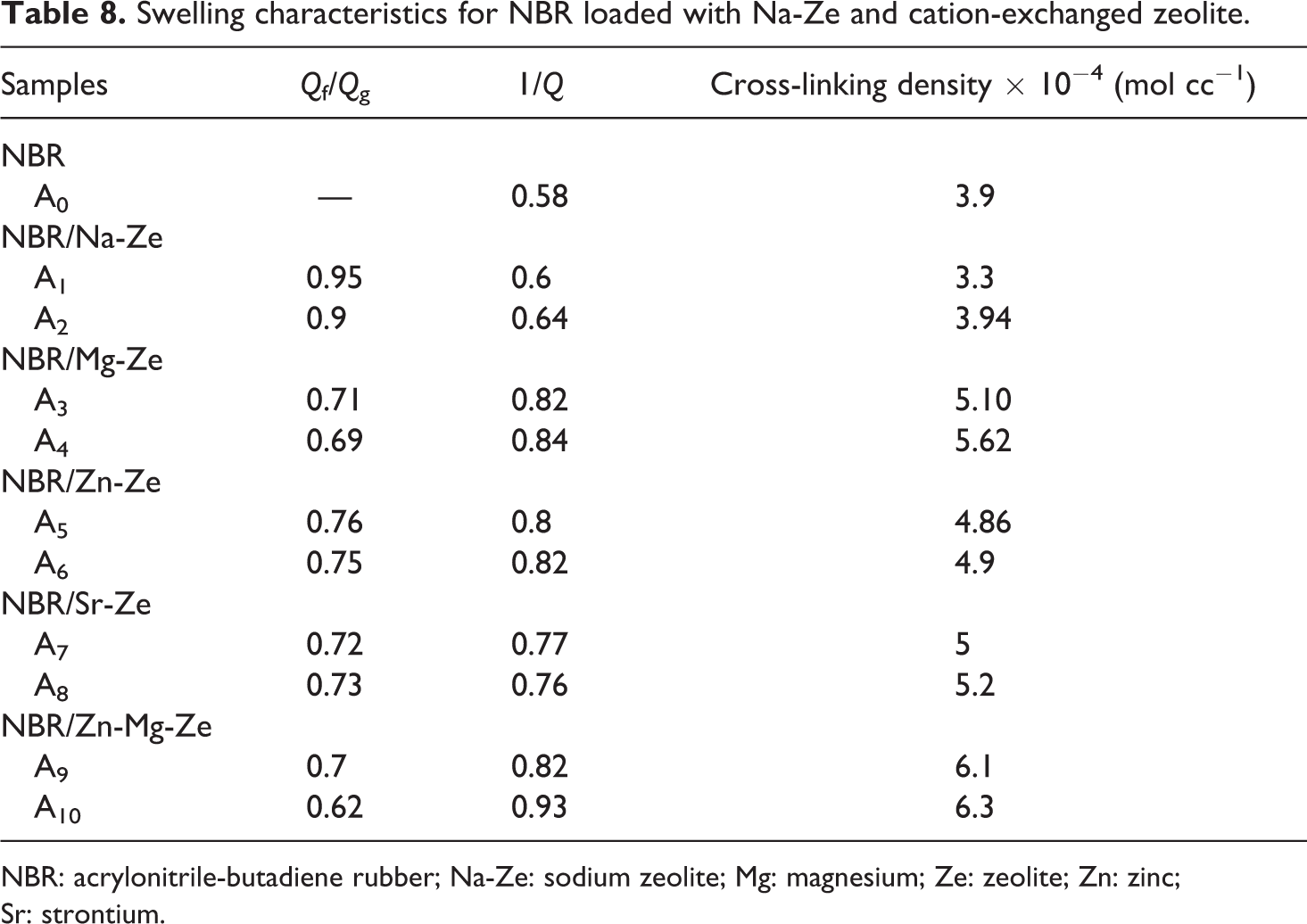
Table 8 shows the swelling properties in various NBR composites loaded with different loadings of Na-Ze and CEZe. Higher value of (Qf/Qg) can lead to lower interaction between the filler and the rubber matrix, and thus the swelling percentage was considerably decreased for composites containing Na-Ze and CEZe in comparison to neat NBR. Two main factors are responsible for this effect; dilution effect of rubber and the absorption of Ze. The low swelling of composites containing Mg-Ze (single cation) and Zn-Mg-Ze (double cations) in comparison to Na-Ze is probably due to higher cure degree indicated by the high torque values as explained before. Hence, the higher reinforcement of CEZe in the NBR matrix restricts the extensibility of the rubber chains induced by swelling. This makes it difficult for the solvent to penetrate into the gaps between the rubber molecules, and thus a decrease in the swelling percentage occurs. Lower percentage of swelling indicates lower interaction between polymer and toluene, while further increase in Na-Ze loading (i.e. 6 and 12 phr) leads to higher swelling percentages. This is confirmed by the low torque values as indicated in Table 4, which is probably due to the degradation of rubber composites.
Swelling characteristics for NBR loaded with Na-Ze and cation-exchanged zeolite.
NBR: acrylonitrile-butadiene rubber; Na-Ze: sodium zeolite; Mg: magnesium; Ze: zeolite; Zn: zinc; Sr: strontium.
It was found that the cross-linking density of NBR composites was increased by the addition of CEZe, and the higher increase was observed in samples containing Mg-Ze and Zn-Mg-Ze. The increase in cross-linking density leads to reduced molecular movement of the rubber, making it more difficult for toluene to penetrate through it as can be detected in Table 8. Also, this may be due to the interaction between NBR matrix and CEZe which increases the cross-linking rate. Further increase in the cross-linking density was observed on adding 12 phr of Mg-Ze and Zn-Mg-Ze (12 phr) to the rubber composites. 32
The equilibrium swelling of SBR vulcanizates containing Na-Ze and CEZe was evaluated to determine the extent of cross-linking densities. It can be noticed that Qf/Qg decreased with adding CEZe, especially composites containing Mg-Ze and Zn-Mg-Ze. This may be due to the sufficient adhesion between CEZe and SBR rubber, and it is clear that lower the values of Qf/Qg, the higher the extent of interaction between Ze and rubber matrix.
From Table 9, it can be observed that a decrease in the equilibrium swelling occurred as a result of the incorporation of Na-Ze and CEZe into SBR matrix, due to the presence of hydroxyl groups on Ze surface which are responsible for the lower interactions between the rubber and the solvent.
Swelling characteristics for SBR loaded with Na-Ze and cation-exchanged zeolite.a
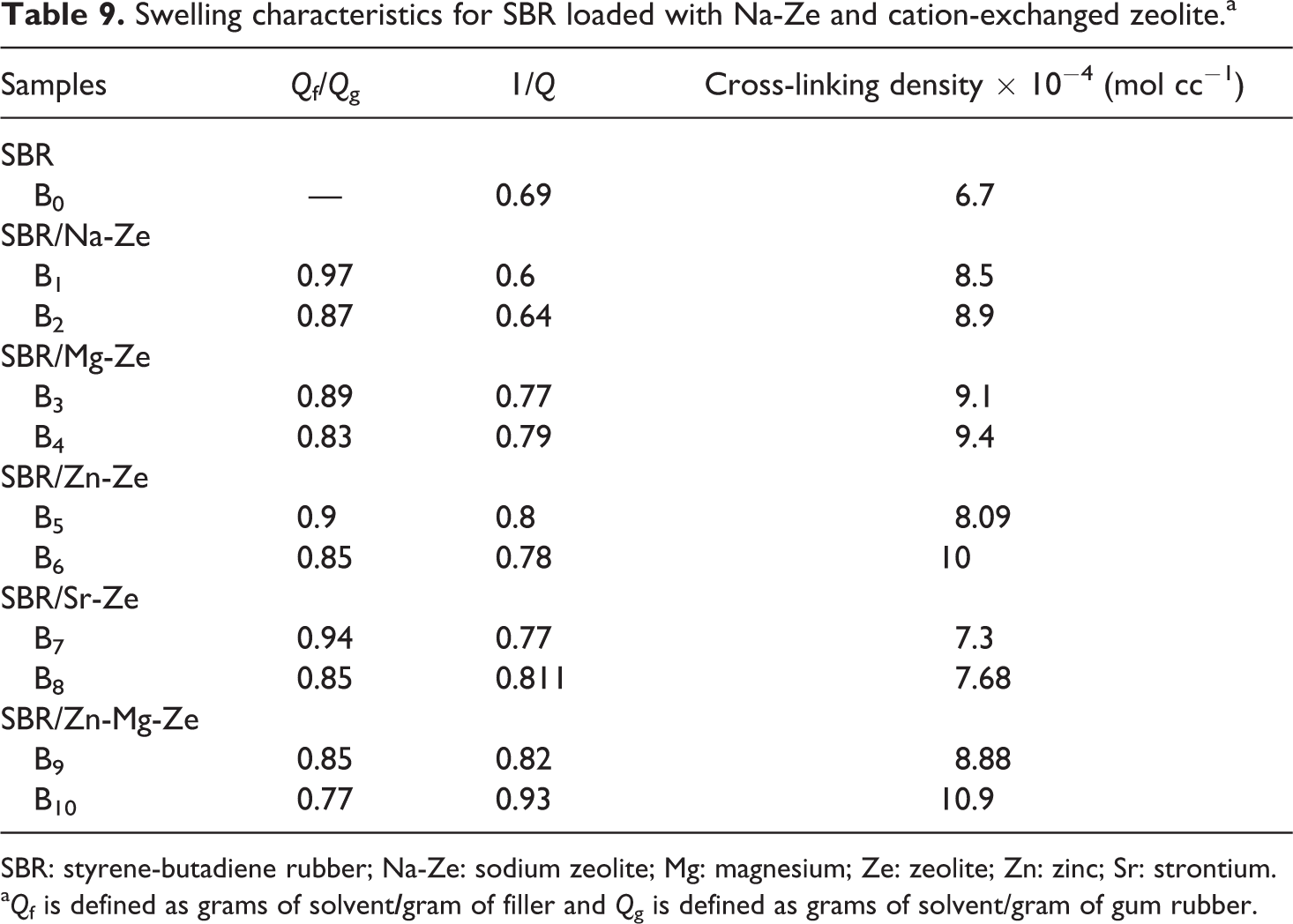
SBR: styrene-butadiene rubber; Na-Ze: sodium zeolite; Mg: magnesium; Ze: zeolite; Zn: zinc; Sr: strontium.
aQf is defined as grams of solvent
The values of cross-linking density are illustrated in Table 9, indicating that the cross-linking density of SBR composites increased by the addition of CEZe, which may be due to the interaction of Ze surface with the cure system as well as the compatibility between both phases that allow the formation of additional interaction. This ensures that the highest cross-linking density was obtained for SBR/Zn-Mg-Ze composites, which is mainly due to the increased interactions at the boundaries between rubber components. 33
Scanning electron microscopy
Figure 11 shows the SEM photos that determine the dispersion of Ze in the NBR composites containing Na-Ze and CEZs. The photos cleared that Na-Ze particles were not clustered and were uniformly dispersed in the NBR matrix. This phenomenon is caused by the reaggregation through polar silicates at the time of coagulation even in the case of Ze exfoliation during swelling, which is assumed to be the result of using CEZe that induces the cation exchange reaction with Na

SEM images for NBR and NBR loaded with 12 phr Na-Ze and CEZe at magnification 500×. (a) NBR, (b) NBR/Na-Ze, (c) NBR/Mg-Ze, (d) NBR/Zn-Ze, (e) NBR/Sr-Ze, and (f) NBR/Mg-Zn-Ze.
The less polar surface of silicates resulting in the reaction leads to lowering of the silicate–silicate interaction and improves their dispersion. Therefore, NBR/CEZe composites perform substantial influence on the dispersion, resulting in the clustering of the Ze which can be explained on the basis of agglomeration. It can be seen that loading of 12 phr of Mg-Ze and Zn-Mg-Ze in NBR composites shows high dispersion in the matrix and the tensile fracture surfaces exhibited many tear lines. 34
SEM images in Figure 12 clarified the dispersion of Na-Ze and CEZe in SBR composites. The dark lines in the figure represent the intersection of the silicate layers, while the white background corresponds to the SBR matrix. Photos of SBR/Na-Ze composite in Figure 12(a) showed unsatisfactory dispersion that is caused by the aggregation of silicates confirming that bulks of CEZe dispersion are in the intercalated state which ensures better dispersion in SBR. SBR/Mg-Ze composite shows homogeneous dispersion of Ze platelets throughout the SBR matrix. In these composites, CEZe was disorderly oriented and only few particles were stacked providing an evident of exfoliation and formation of agglomerates at some spots in the SBR matrix.

SEM images of SBR loaded with 12 phr CEZe at magnification 500×. (a) SBR, (b) SBR/Na-Ze, (c) SBR/Mg-Ze, (d) SBR/Zn-Ze, (e) SBR/Sr-Ze, and (f) SBR/Mg-Zn-Ze.
The previous results concluded that NBR showed stronger interaction with the Ze than SBR. This was confirmed by the morphology shown in the SEM image, and the high dispersion of Ze and CEZe that occurred due to the interfacial hydrogen bonding between aluminum silicate of Ze and NBR. 35
Thermogravimetric analysis
Thermogravimetric analysis is the most efficient technique used to study the influence of filler on the thermal properties of composites and the results are shown in Figure 13(a) and summarized in Table 10. Despite the onset thermal decomposition temperature was the same, the weight losses of the composition have differed according to the type of CEZe.
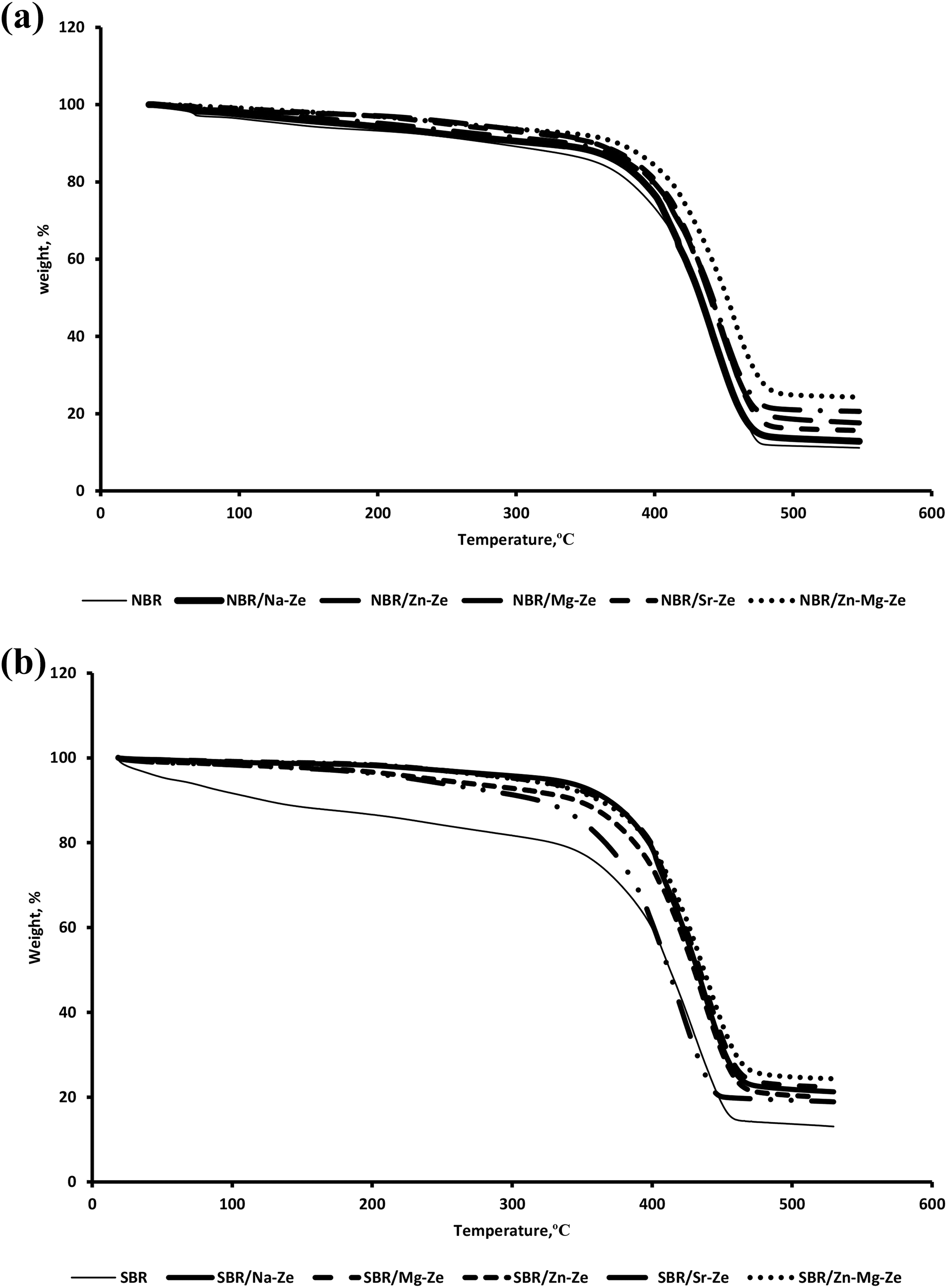
Thermogravimetric analysis curves of weight (a) NBR composites and (b) SBR composites loaded with 12 phr Na-Ze and CEZe.
Thermal degradation for NBR loaded with12 phr Na-Ze and cation-exchanged zeolite.a
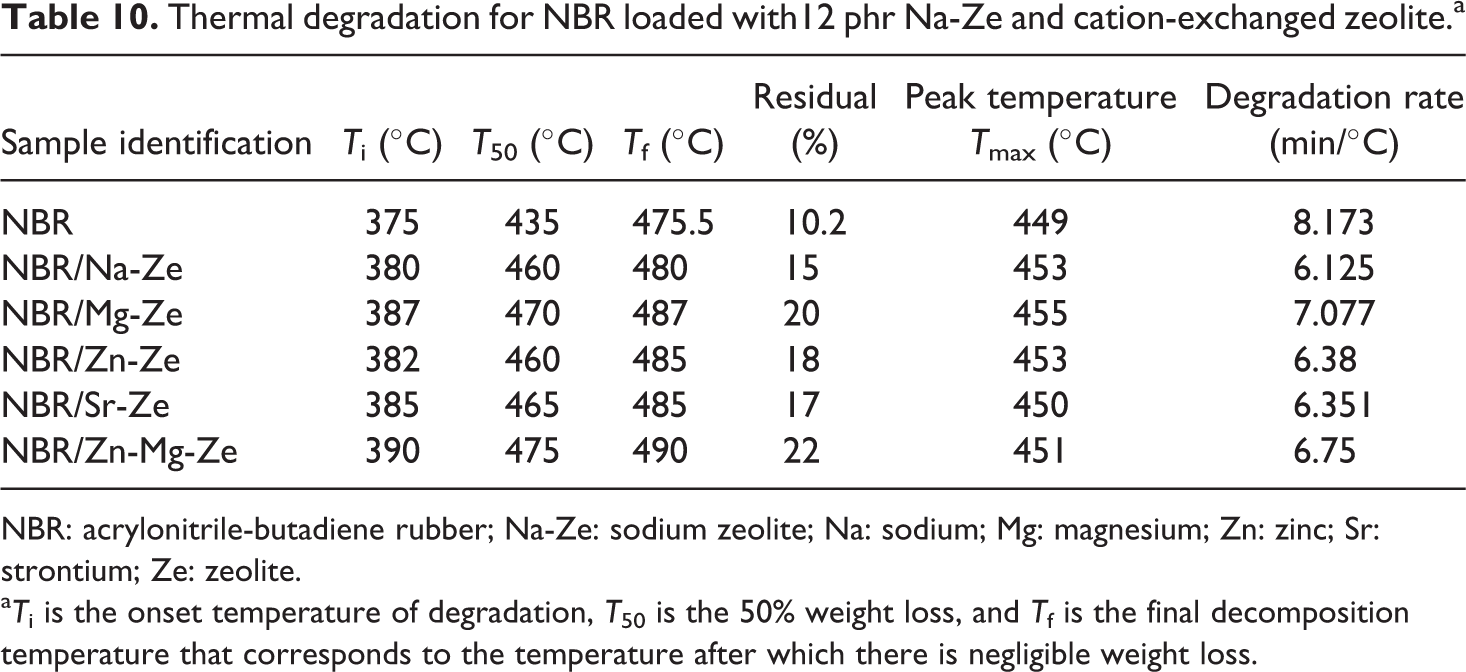
NBR: acrylonitrile-butadiene rubber; Na-Ze: sodium zeolite; Na: sodium; Mg: magnesium; Zn: zinc; Sr: strontium; Ze: zeolite.
aTi is the onset temperature of degradation, T50 is the 50% weight loss, and Tf is the final decomposition temperature that corresponds to the temperature after which there is negligible weight loss.
NBR composites started degradation at Ti 375°C, this temperature increased in the case of NBR containing Na-Ze and CEZe up to a maximum value at 390°C for NBR/Zn-Mg-Ze composites. The known stability of Ze increases the stability of composites (NBR/CEZe). This may be due to the release of water of crystallization of Ze at higher temperature. While NBR/Mg-Ze and NBR/Zn-Mg-Ze composites gave the highest residual weight percentage 20 and 22, respectively, indicating that these composites were more stable than other composites which can be attributed to the decrease in the diffusion of volatile gases in the polymer/filler matrix due to the homogeneous distribution of these fillers. 36 The thermal degradation peak temperatures (Tmax) of NBR composites are shifted to higher temperature with loadings of CEZe. This indicates that significant improvement in thermal stability is more likely attributed to the presence of Zes in NBR matrices, which offer a great barrier effect to hinder the formation of small molecules, resulting from thermal decomposition and simultaneously resisting their movement during desorption from the surface. 37
Table 11 and Figure 13(b) demonstrate the thermogravimetric degradation of SBR composites in the temperature range 360–395°C. The Ti value of SBR is 360°C, this Ti increases with the addition of Na-Ze and CEZe to SBR up to 395°C and Also, Tf for SBR composites were higher than that for neat SBR as can be seen in Table 9, where Tf was at 475 and 492°C for neat SBR and SBR composite containing Zn-Mg-Ze. SBR composites containing 12 phr of Mg-Ze and Zn-Mg-Ze gave the highest residual weight percentage and this indicated that these composites were more stable than neat SBR; this can be attributed to the fact that addition of Zes to SBR brought thermal stability to the composites. The thermal degradation peak temperatures (Tmax) of composites containing CEZe ranged between 425°C and 453°C, indicating their thermal stability, and revealing that the extent of curing reaction are nearly the same. The higher thermal stability of SBR composites (containing Na-Ze and CEZe) may be due to the high thermal stability of these fillers as well as the interaction between the Ze layers and the rubber matrix through intercalation/exfoliation. These intercalated SBR chains are covered by Ze layers which prevent the direct exposure of these chains to the thermal influence. 38
Thermal degradation for SBR loaded with12 phr Na-Ze and cation-exchanged zeolite.a
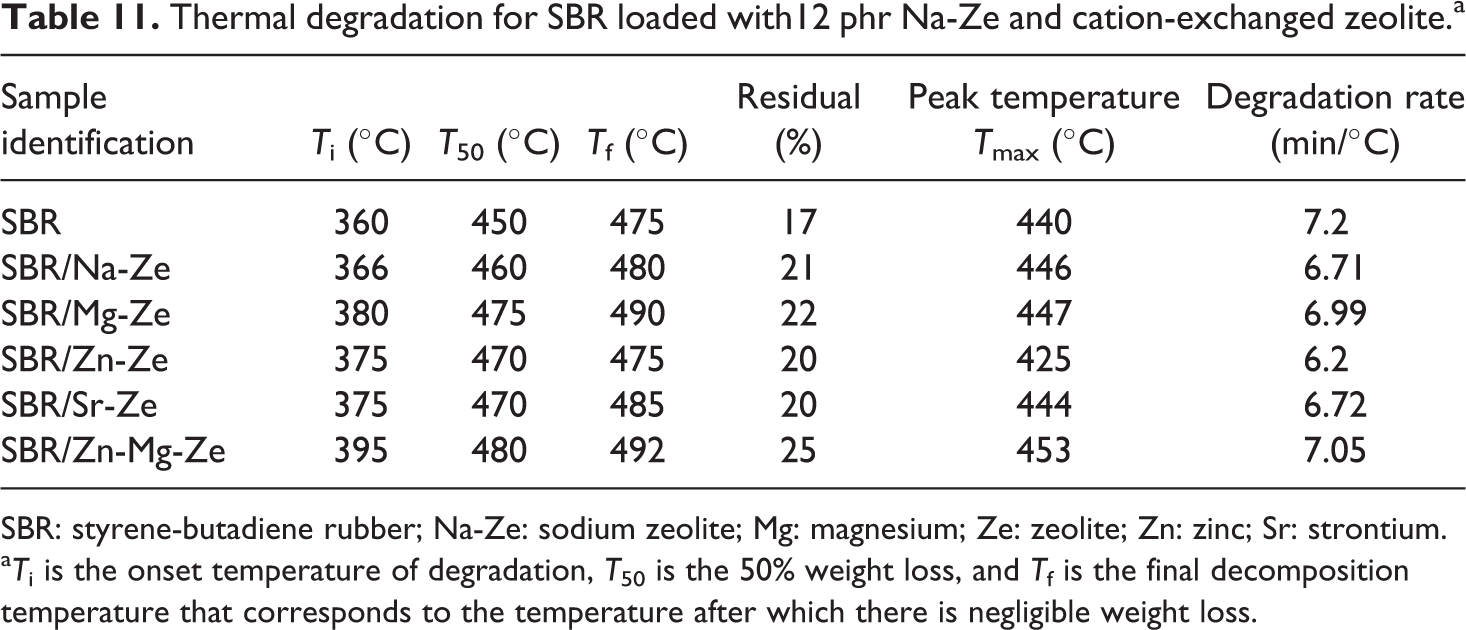
SBR: styrene-butadiene rubber; Na-Ze: sodium zeolite; Mg: magnesium; Ze: zeolite; Zn: zinc; Sr: strontium.
aTi is the onset temperature of degradation, T50 is the 50% weight loss, and Tf is the final decomposition temperature that corresponds to the temperature after which there is negligible weight loss.
Conclusions
Rubber composites containing Na-Ze and CEZe were introduced to be innovative products with improved properties to achieve a demand for advanced rubber industries. To evaluate their impact on the properties of NBR and SBR composites, the following findings were recorded. Incorporation of Na-Ze and CEZe in NBR and SBR matrices enhanced their curing characteristic. Mg-Ze (single cations) and Mg-Zn-Ze (double cations) showed high mechanical properties in both NBR and SBR matrices. The swelling percentage was considerably decreased for NBR and SBR composites containing Na-Ze and CEZe. NBR and SBR composites containing Mg-Ze and Zn-Mg-Ze were well dispersed in the matrix and their tensile fracture surfaces exhibited many tear lines. NBR and SBR composites filled with CEZe constituted enhanced thermal barrier against mass loss. Both Ti and Tf were increased with the addition of CEZe to the matrix, giving high residual wt%. Na-Ze and CEZe appeared to be more compatible in NBR matrix than in SBR, which can be attributed to the higher compatibility between the chemical nature of the acrylonitrile rubber and the inorganic cations that have replaced Na
