Abstract
This study investigates the removal effectiveness and characteristics of phenanthrene and naphthalene using low-cost steel slag with batch experiments. The adsorption characteristics of steel slag were measured and analysed using X-ray fluorescence, X-ray diffraction, and Fourier transform infrared spectroscopy. The batch experiments investigated the effect of the time gradient, pH, and steel slag dosage gradient on the adsorption of the steel slag. The results show that with time and dosage of steel slag increased, the adsorption capacity of phenanthrene and naphthalene increased and gradually became balanced, but pH had no obvious effect on the adsorption of phenanthrene and naphthalene. The Langmuir isotherm model best describes the phenanthrene and naphthalene removal by the steel slag, which shows the adsorption occurring in a monolayer. The maximum adsorption capacity of the steel slag to phenanthrene and naphthalene is 0.043 and 0.041 mg/g, respectively. A pseudo-first-order kinetic model can better represent the adsorption of phenanthrene and naphthalene by steel slag. The research demonstrates that the steel slag has a certain adsorption capacity for phenanthrene and naphthalene.
Introduction
Polycyclic aromatic hydrocarbons (PAHs), aromatic compounds with more than two benzene rings, are difficult to degrade and harmful to organisms (Wilcke, 2000). PAHs are man-made contributions to the environment, being derived primarily from incomplete combustion, volatilization, and oil leakage (Jeffrey and Edward, 2014). The degradation process of PAHs is slow and complex, with transport and distribution occurring with certain regularity in the atmosphere, water, sediment, soil, and biology. Since the PAHs are hydrophobic, they are easily absorbed in the non-aqueous phase by hydrophobic organic matter; therefore, PAHs accumulate easily in organisms (Villholth, 1999). PAHs are highly teratogenic and carcinogenic mutagenic compounds, harmful to human health and the living environment. Therefore, it is very important to remove PAHs from the environment.
In recent years, several researchers have reported extensive work on the removal of PAHs. The adsorption method is widely considered for this application because of its advantages of simple operation, low cost, and reusable adsorbent (Jiang, 2001). Researchers removed PAHs from the environment with various natural adsorbents or modified adsorbents, such as activated carbon, natural porous zeolites, kaolinite, diatomaceous earth, and fly ash (Angove et al., 2002; Douce et al., 1997; Zhou et al., 2008). To reduce the cost of wastewater treatment, application of some cheap industrial and agricultural wastes with good adsorption characteristics has gradually attracted attention, and adsorption is broadly applied for large areas of wastewater treatment (Hegazi, 2013).
Basic oxygen furnace (BOF) steelmaking is the major technology of steel production in the world. During the process, it will generate steel slag while the limestone (or dolomite) as a flux is added into the hot metal. Steel slag contains many alkaline oxides, porous characteristics, large surface area, high density, and easy solid–liquid separation; therefore, steel slag can remove various pollutants from wastewater by adsorption and chemical reaction (Kim et al., 2008). Steel slag can also be effective for the adsorption of organic matter from the wastewater. Zheng et al. (2010) measured the removal rate of organic matter from water which can reach 51% using slag in the cylinder test. Huang et al. (2011) showed that the steel slag as a wetland substrate was feasible to decrease the chemical oxygen demand (COD) of 120.93–653.33 mg/l faecal sewage by building a vertical subsurface flow constructed wetland system, and the average removal rate of COD was more than 70%. The removal of COD from wastewater by steel slag is based mainly on the oxide minerals released from the steel slag, which can adsorb and precipitate organic matter (Hibino et al., 2016). However, COD is a comprehensive index to reflect a variety of organic pollutants in the water. Therefore, using steel slag to remove COD in various experiments cannot determine the effect of the steel slag on the removal of each type of organic matter and the mechanism of removal. COD always contains highly toxic PAHs, such as the PAHs resulting from road surface run-off (Beasley and Knealem, 2002; Hoffman et al., 1984). However, at present, the effect and the mechanism of removal of PAHs by steel slag has not been determined.
Naphthalene and phenanthrene are PAHs with a higher solubility in water, which therefore produces a greater influence on natural systems and human health. However, the high octanol–water partition coefficient of phenanthrene and naphthalene leads to hydrophobicity, and the compounds are easily assigned to the non-aqueous phase, so this situation provides an ideal opportunity to use a solid adsorbent to remove phenanthrene and naphthalene. In this study, the removal effectiveness for phenanthrene and naphthalene was analysed by batch experiments using steel slag as an adsorbent, and the removal characteristics were further investigated.
Materials and methods
Materials
The steel slag was obtained from Nanjing Meishan Iron and Steel Plant, Nanjing, China. The steel slag was broken with a jaw crusher to a size below 3 cm. The steel slag was subsequently placed in a planetary ball mill to grind to a size under 150 µm and sent to the vacuum oven for drying. The ground slag was then dried (100°C for 24 h) and stored in a desiccator.
Phenanthrene solid powder (200 mg, Shanghai Aladdin Biochemical Technology Co., Ltd, Shanghai) was weighed on an electronic balance and dissolved in methanol to form 100 ml of solution. The standard solution was set at a concentration of 2000 mg/l and placed in a freezer (2°C). The method of preparing the naphthalene (Shanghai Aladdin Biochemical Technology Co., Ltd, Shanghai) solution is same as the method for preparation of the phenanthrene. The solutions were stable for a period of three months.
Methods
Batch experiment
In this study, the optimum conditions for the adsorption of phenanthrene and naphthalene on steel slag were determined using single factor batch experiments. The time gradient experiment set the concentration of phenanthrene and naphthalene at 1 and 5 mg/l; the pH value at 6.07; and the time gradients were set for 1, 2, 4, 8, 12, 24, and 48 h, respectively. The steel slag dosage gradient experiment set the phenanthrene concentration at 1 mg/l, and the amount of the slag gradient was set, respectively, at 100, 200, 500, 800, 1000 mg at a pH of 6.07. The pH gradient test set the pH values, respectively, at 3.07, 5.67, 6.07, 8.04, 9.25, and 11.09. The isothermal adsorption experiment set the concentration gradient of phenanthrene at 0.05, 0.1, 0.2, 0.4, 0.5, 0.8, and 1 mg/l, and the concentration gradient of naphthalene was 3, 4, 5, 6, 7, 8, and 10 mg/l. The pH value was 6.07, and the solutions were shook for 24 h at a constant temperature with no light.
The main process involved in the experiment was as follows: seal the prepared solution and the steel slag into the centrifuge after mixing them, and the centrifuge is set to 200 r/min. The temperature is set at 25°C with no light oscillation. Then, timing is started. A sample of 30 ml of the oscillation solution is taken from the solution and put into a 50 ml centrifuge tube after the set time.
Chemical analysis
Solid-phase extraction (SPE)
SPE is a technology that utilizes the characteristic of physical adsorption of organic matter on a SPE column rather than water. The sample matrix and interferences are separated, followed by eluting the SPE column to separate the organic matter and the SPE column. SPE technology is widely used in many fields, and it is a technology with a mature extraction effect. SPE is involved in food safety monitoring and life sciences (Zhang and Yang, 2000). The extraction procedure is improved by Niu et al. (2006), as follows:
First, take the samples out of the thermostated shaker after shaking, then remove 30 ml of the solution from a glass centrifuge tube to centrifuge the solution at 4000 r/min for 5 min, and finally remove 4 ml of the supernatant for extraction. Activation: according to the experimental sample number, number the SPE column and then use 2.5 ml of methanol to flow through the SPE column to activate it, followed by transflux with 5 ml of distilled water. The purpose is to remove impurities from the SPE column and to create a solvent environment and stabilize the recovery rate. Add sample: transfer the sample into the SPE column with the transfer liquid gun. It is significant to ensure that the surface of the column is covered with liquid to avoid contact with air when the sample is filtered. Washing: after filtering, utilize 2 ml of distilled water to flow through the column to remove the interferents. Drying: after elution, put the column into a vacuum suction device and open the vacuum after sealing for 10 min to remove water. Elution: use 4 ml n-hexane to elute the column. The eluent flows into a 4 ml sample bottle and is then stored in a sealed container in the refrigerator until analysis.
Instrumental analysis
Gas chromatography/mass spectrometry
In the present study, the instrument used for the quantitative determination of phenanthrene and naphthalene in the samples was a gas chromatograph coupled with a mass spectrometer GC–MS-QP2010PLUS (GC/MS) (SHIMDZU, Japan).
The GC/MS detection parameters are shown in Tables 1 to 3.
GC parameter settings.

GC: gas chromatography.
Parameter settings of column temperature.

MS parameter settings.

MS: mass spectrometer; SIM: Selected ion monitoring.
The GC/MS plot of phenanthrene and naphthalene peaks is shown in Figure 1. The graticule of phenanthrene and naphthalene is shown in Figure 2.

GC/MS plots of (a) phenanthrene and (b) naphthalene.

Standard curves of (a) phenanthrene and (b) naphthalene.
2. Chemical composition analysis of steel slag
Scanning electron microscopy (SEM) S-4800 (Hitachi, Japan) was used to perform qualitative analysis of the surface characteristics of the steel slag. The physical adsorption instrument ASAP2020 (Micromeritics Instrument (Shanghai) Ltd, China) was used for quantitative detection of slag surface area which ranged from 0.0005 m2/g (Kr measurement) to no upper limit. The aperture analysis ranged from 0.35 to 500 nm (nitrogen adsorption), and the minimum pore volume for the detection was 0.0001 cc/g. An X-ray fluorescence (XRF) spectrometer XRF-1800 (Shimadzu Corporation, Japan) was used to test the chemical composition of the steel slag, and an X-ray diffractometer XRD-D8 ADVANCE (X-ray diffraction (XRD)) (Bruker, German) was also used. The functional groups on the surface of the steel slag were determined using Fourier transform infrared spectroscopy (FTIR) Nicolet iS5FT-IR spectrometer (Thermo Scientific, USA).
Adjustment of data
Recovery rate: 1 mg/l (C3) aqueous solution of phenanthrene was prepared, 4 ml of the aqueous solution was used to carry out the SPE, and 4 ml of phenanthrene extraction solution (C2) was obtained. Distilled water (4 ml) was used as the blank control, and the same SPE was carried out to obtain 4 ml of phenanthrene extraction solution (C1).
By the formula of recovery rate

Adsorption model
Adsorption kinetic model
The time and the data of the pollutant concentration were recorded. Next, the record was used to fit with the adsorption kinetic model. Adsorption kinetic models primarily include the following:
Pseudo-first-order kinetic model
The expression of the pseudo-first-order kinetic model is given by equation (1) (Bogan et al., 2003; Park et al., 2010)

Pseudo-second-order kinetic model
The pseudo-second-order kinetic equation was obtained from Ho and McKay (1998, 1999) and the expression is shown as equation (2)

Isothermal adsorption model
The adsorption isotherm models are mainly the Langmuir isotherm equation and Freundlich isotherm equation.
The linear expression of the Langmuir isotherm equation is presented as equation (3) (Ho et al., 2002)

RL in the Langmuir equation can predict whether the reaction is favourable equation (4)

When 0 < RL<1, the reaction is favourable for adsorption; when RL = 0, irreversible adsorption is observed; when RL=1, linear adsorption occurs; RL>1 is unfavourable to adsorption (Fu and Sheng, 1990).
The linear expression of the Freundlich isotherm equation is shown in equation (5)

Results and discussion
Properties of steel slag
Chemical composition of steel slag.


(a) XRD of steel slag (RO is Mg, Mn, Al, and other metal oxides) and (b) FTIR analysis of steel slag.
The SEM results (Figure 4) show that the surface of the steel slag is porous and has a large specific surface area. The specific surface area of steel slag is 2.854 m2/g, and the total pore volume is 0.01253 cm3/g (Table 5).

Scanning electron micrograph of steel slag.
Specific surface area and porosity of steel slag.

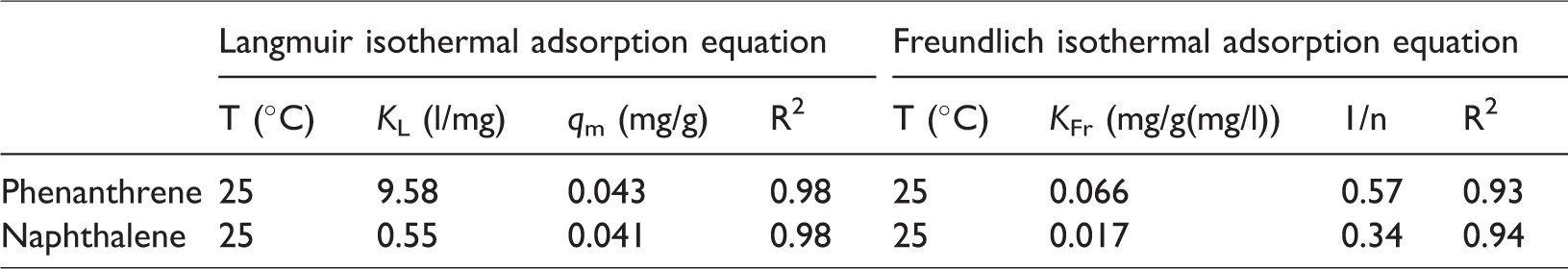
Fitting parameters of the Langmuir and Freundlich isotherm adsorption of phenanthrene and naphthalene.
The XRD analysis showed that steel slag contains Ca silicates and free lime which can react with water as shown in equations (6) and (7). The release of Ca2+ and H2SiO42− to solution can lead to oversaturation with respect to calcium silicate hydrates (Ca–Si–H) equation (8) (Hobson et al., 2017). Through flocculation, calcium silicate hydrates gel will combine with some hydrophobic organics to achieve the purpose of removing the organics (Fu et al., 2012). The analysis of the specific surface area of the steel slag shows that the steel slag clearly has a large specific surface area and is porous with a better ability to adsorbing (Xue et al., 2009)



Batch experiments for adsorption
Effect of time on adsorption
During the adsorption of phenanthrene, the adsorption speed in the beginning 1–8 h is rapid. As time elapses, the adsorption rate of phenanthrene gradually slows down and eventually reaches equilibrium (Figure 5(a)). The process of adsorption of naphthalene is similar to the process of adsorption of phenanthrene (Figure 5(b)). The adsorption amount of phenanthrene and naphthalene by steel slag changes over time. The adsorption rate was fast because the adsorption sites on the surface of the steel slag had not yet reached saturation at initial adsorption. The adsorption sites gradually become saturated, and the adsorption rate becomes slow and eventually tends to balance with time. According to the experimental results, we determined that 24 h was the adsorption equilibrium time.
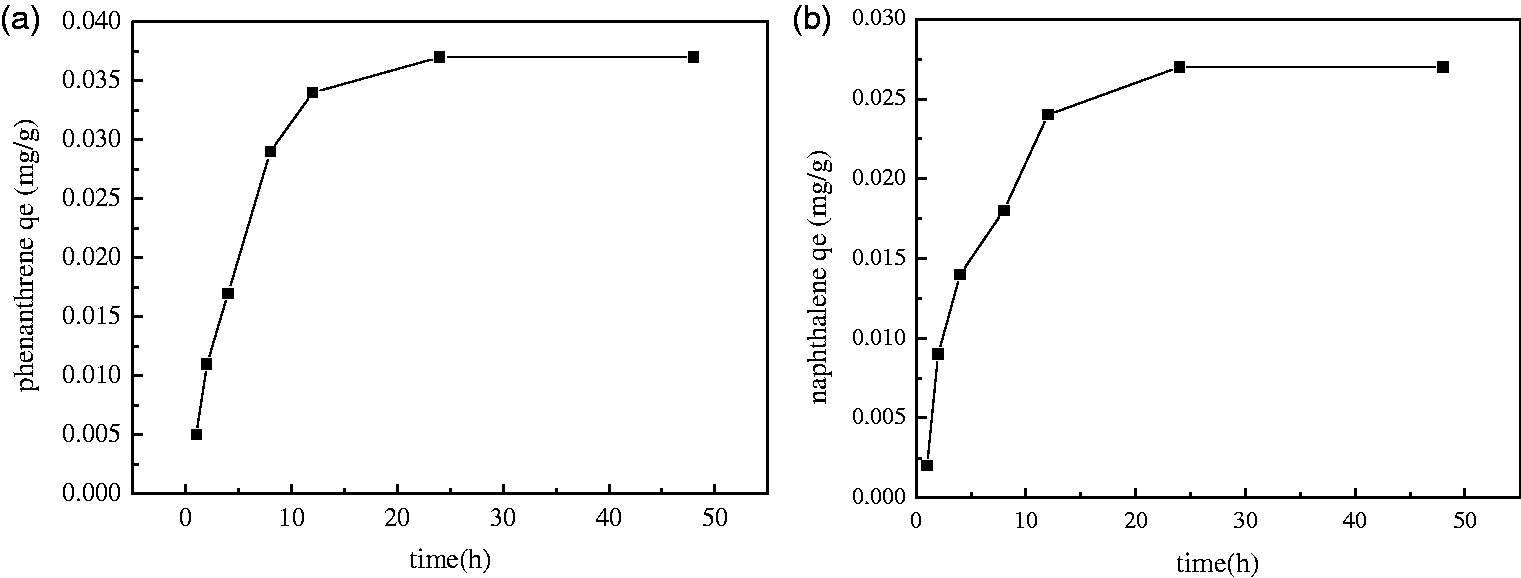
Effect of reaction time on qe of phenanthrene (a) and naphthalene (b) by steel slag (500 mg steel slag, pH 6.07; the initial concentration of phenanthrene was 1 mg/l, the initial concentration of naphthalene was 5 mg/l).
Effect of pH on adsorption
At different pH values, the adsorption capacity of the steel slag for phenanthrene is fluctuating slightly, and there is no obvious effect of pH on the adsorption of phenanthrene by the steel slag (Figure 6). Other studies also showed that pH had no effect on phenanthrene adsorption (Chen, 2003). The concentration of ions in aqueous solution changed when the pH changes, but phenanthrene is a non-polar and stable substance that exists in the water in the form of a molecule, meaning that it is possible neither to ionize phenanthrene nor for phenanthrene to exist in the polarized state. Therefore, pH has little influence on the adsorption of phenanthrene by steel slag. The chemical properties of naphthalene and phenanthrene are similar, so the adsorption of naphthalene on steel slag is not affected by the pH value. The initial pH of the experiment was 6.07; therefore, the pH of the follow-up experiment was set to 6.07.

Effect of pH on the adsorption of phenanthrene (500 mg steel slag, the initial concentration of phenanthrene was 1 mg/l).
Effect of steel slag dosage on adsorption
With the increase of the dosage of the steel slag, the equilibrium adsorption efficiency of phenanthrene by the steel slag decreased gradually (Figure 7(a)), but the total adsorption amount of phenanthrene (Figure 7(b)) increased gradually. Due to the increase in the dosage of slag, the adsorption sites increased in the aqueous solution. However, the amount of phenanthrene in the aqueous solution was constant, and the greater the dosage of the steel slag, the less the adsorption efficiency of the steel slag per unit mass was. Because of the similar properties of phenanthrene and naphthalene, the adsorption of naphthalene was similar to phenanthrene when the dosage of steel slag increased.

(a) Influence of the dosage of steel slag on the amount of phenanthrene adsorption (b) and the influence of the total amount of steel slag on phenanthrene adsorption (pH = 6.07, phenanthrene initial concentration was 1 mg/l).
According to the results of the batch experiments, the equilibrium time for the adsorption of phenanthrene and naphthalene by steel slag was 24 h, the pH was 6.07, and the dosage of steel slag was approximately 500 mg.
Characteristics of adsorption
Isothermal adsorption models for phenanthrene and naphthalene
According to the fitting data from the Langmuir isotherm model and the Freundlich isothermal adsorption model, we can conclude that the isothermal adsorption of phenanthrene and naphthalene conforms more to the Langmuir model, and the R2 of phenanthrene and naphthalene is 0.98, respectively (Figure 8). The qm of phenanthrene calculated by the Langmuir isotherm model is larger than the same for naphthalene.
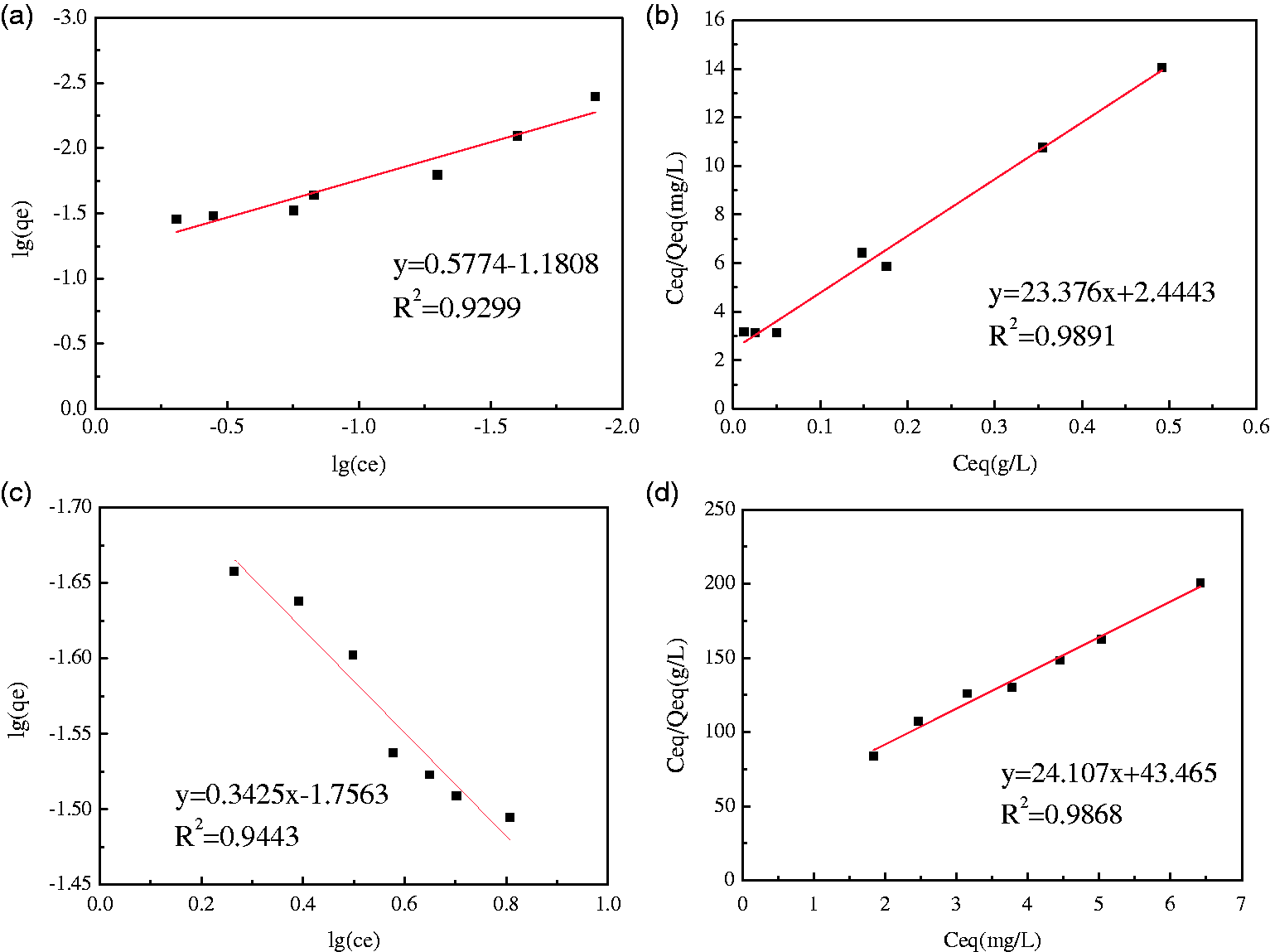
Isothermal model for adsorption of phenanthrene and naphthalene by slag (500 mg steel slag, pH 6.07, the initial concentration of phenanthrene was 1 mg/l and naphthalene concentration was 5 mg/l). (a) The Freundlich fitting chart for phenanthrene, (b) the Langmuir fitting chart for phenanthrene, (c) the Freundlich fitting chart for naphthalene, and (d) the Langmuir fitting chart for naphthalene.
Two assumptions are associated with the Langmuir model: one assumption is that the surface of the adsorbent is uniform, and there is no interaction between adsorbed molecules. The other assumption is that the adsorption process is a single molecular adsorption, and the adsorption sites can only combine with one molecule. The simulation results show that both phenanthrene and naphthalene can be adsorbed on the surface of steel slag, and the interaction between phenanthrene and naphthalene can be ignored. The RL values for phenanthrene and naphthalene were 0.70 and 0.81, respectively, and both of these values were less than 1, indicating that the adsorption of phenanthrene and naphthalene by steel slag could be carried out spontaneously.
According to the Langmuir model, the adsorption amount of phenanthrene and naphthalene by steel slag was 0.043 and 0.041 mg/g, respectively (Table 6), indicating that the amount of adsorption of steel slag to phenanthrene and naphthalene is similar, but the amount of phenanthrene is slightly higher than that of naphthalene. Phenanthrene and naphthalene are difficult to ionize; they are in the form of molecules in the solution. Phenanthrene is composed of three benzene rings, and naphthalene is composed of two benzene rings. In addition, with the increasing molecular weight of PAHs, the hydrophobicity of the PAHs increases, and the PAHs are more easily adsorbed by the steel slag. Since phenanthrene has more hydrophobicity, phenanthrene is more easily adsorbed by steel slag in solution, while naphthalene is more likely to dissolve in the water. The results show that the adsorption amount of organic solute in solid matter is related to the partition coefficient of octanol/water. The log KOW increases with the hydrophobicity of the organic solute increasing, and the more convenient it is to adsorb solid matter (Brigg, 1973).
Kinetic adsorption simulation models of phenanthrene and naphthalene
Pseudo-first-order and pseudo-second-order kinetic model fitting of phenanthrene and naphthalene.


Kinetic model of the adsorption of phenanthrene and naphthalene by steel slag: (a) Pseudo-first-order kinetic curve of phenanthrene, (b) pseudo-second-order kinetic curve of phenanthrene, (c) pseudo-first-order kinetic curve of naphthalene, and (d) pseudo-second-order kinetic curve of naphthalene.
The adsorption of phenanthrene and naphthalene using steel slag can be represented by the pseudo-first-order kinetic model, which is attributed to the stage of organic adsorption onto BOF slag is dominated and controlled by the external surface adsorption (Xue et al., 2009). Because the steel slag contains abundant minerals such as calcium ferrite, calcium silicate, and calcium carbonate, the surface of the steel slag presents Ca2+ and Mg2+ equations (6), (7), and (9) (Hobson et al., 2017; Zhu et al., 2004). These cations interact with the π-electronic structure of phenanthrene and naphthalene in the water through electrostatic attraction, polarization, and other interactions to form cationic–π bonds to achieve adsorption (Tsuzuki et al., 2003; Zhu et al., 2003) (Figure 10(a))


Schematic diagram of the adsorption of phenanthrene and naphthalene by steel slag: (a) the cation–π bond, (b) the hydrogen bond, and (c) C–S–H gel flocculation. C–S–H: calcium silicate hydrate.
The steel surface with a negative charge in the water can combine with the water to form a layer of water film and form a surface microlayer of water that can generate a hydrogen bond with benzene (Ralph, 2012) (Figure 10(b)) and may also produce the adsorption of phenanthrene and naphthalene.
In addition to the external surface absorption, flocculation may also have some effect on the removal of phenanthrene and naphthalene by steel slag. Ca2SiO4 in steel slag can react with water to produce calcium silicate hydrate (C–S–H) (equations (6) to (8)). C–S–H is insoluble in water and immediately separates out in the form of colloidal particles and then gradually condenses into C–S–H gel. The gel plays a role in phenanthrene and naphthalene removal by flocculation (Fu et al., 2012) (Figure 10(c)).
Adsorption efficiency of phenanthrene and naphthalene
The adsorption amount of phenanthrene and naphthalene by steel slag was 0.043 and 0.041 mg/g, respectively, according to the Langmuir model. Compared with other adsorbent studies, the adsorption efficiencies were as follows: loess > kaolin > steel slag > quartz sand (Tables 8 and 9). The effect of adsorbent on the removal of organic matter was related to the organic matter content and specific surface area of the adsorbent. Because organic matter can change the distribution of hydrophobic organic pollutants in the adsorbent and water environment (Luo, 2005), the adsorption capacity of phenanthrene and naphthalene increased when the content of organic matter increased (Fan, 2011). Since the content of organic matter in loess is higher than the content of organic matter in kaolin, and the organic matter in steel slag and quartz sand can be ignored, the adsorption capacity of phenanthrene and naphthalene in loess is much better than other adsorbent. In addition, the specific surface area of steel slag was 2.854 m2/g, and the specific surface areas of quartz sand, kaolin, and loess were 0.173, 8.499, and 18.754 m2/g (Fan, 2011; Zhu et al., 2004), respectively. Then, the adsorption effect of phenanthrene and naphthalene increased as the specific surface area increased. According to the comparison, compared with the other inorganic material, quartz sand, the steel slag has a larger specific surface area, and the adsorption capacity of phenanthrene and naphthalene is superior.
Comparison of adsorption capacity of adsorbents to phenanthrene.

BET: Specific surface area.
Comparison of adsorption capacity of adsorbents to naphthalene.

Conclusions
In this study, the effect and characteristics of steel slag on phenanthrene and naphthalene adsorption were studied. The batch experiment obtained the best conditions for the phenanthrene and naphthalene adsorption by steel slag, pH is 6.07, equilibrium time is 24 h, and dosage of steel slag is 500 mg. Moreover, the results show that the adsorption kinetics model of the steel slag to phenanthrene and naphthalene is in accordance with the Langmuir isotherm adsorption model, which consists of monolayer adsorption. The RL of phenanthrene was 0.70, and the RL of naphthalene was 0.81, and both the RL values were less than 1, indicating that the adsorption was spontaneous. According to the Langmuir equation, the maximum adsorption capacities of the phenanthrene and naphthalene by steel slag were 0.043 and 0.041 mg/g, respectively. Based on the fitting results of the adsorption kinetic data of naphthalene and phenanthrene, the adsorption kinetics model of the steel slag to phenanthrene and naphthalene accords with the pseudo-first-order kinetic model.
Footnotes
Acknowledgements
We would like to thank editor and anonymous reviewers for their suggestions which significantly improved the quality of the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
