Abstract
This study was carried out to investigate the comparative effectiveness of ferrous sulfate (FeSO4) and magnesium chloride (MgCl2) in removing color from synthetic C.I. Reactive Black 5 dye wastewater. After chemical coagulation, treated wastewater was reused in the wash-off process. Using 400 ppm of MgCl2, a color removal efficacy of 97% was achieved at pH 12 for synthetic Reactive Black 5 dye wastewater. Coagulant FeSO4 yielded 85% color removal efficiency under the same conditions. The fabric washed with coagulant-treated wastewater had a total color difference (ΔEcmc) value of <1.0, which is commercially acceptable. The colorfastness of the fabrics washed with coagulant-treated wastewater was comparable to those washed off with fresh water.
Introduction
The textile industry is one of the most prominent and substantial industrial sectors in terms of water consumption, discharging unused dyes, and generation of chemically-contaminated effluent. 1 Dyehouses are the principal users of dyes in the textile industry. 2 Approximately 10% to 15% of reactive dyes are lost after the dyeing process and become a part of the effluent. 3 The presence of very small amounts of dye, even less than 1 ppm, in effluent is discernable and aesthetically displeasing, as well as hindering the growth of aquatic biota. 4 Also, many of these dyes released toxic byproducts such as naphthalene, aryl amines, and other aromatic complexes, which are carcinogenic or mutagenic to human beings. 5 Therefore, dye containing effluents require essential and adequate treatment before being discharged into the environment. 6
Worldwide textile effluent treatment methods for dye decolorization use biological, physicochemical, and advanced oxidation processes (AOPs). Biological treatment is generally simple, inexpensive, and eco-friendly but is ineffectual for decolorization due to the resistance of some dyes to biological degradation.7,8 AOPs are effective for removal of some dyes from wastewater. The major drawbacks of these two types of treatments include inefficiency, higher cost of operation, and requirement for large land areas.9,10
Harmful intermediates and toxic byproducts are not formed when a coagulation process was used because it is not based on incomplete decomposition of dye molecules. 11 Coagulation and flocculation processes are similar to the Fenton's oxidation (FO) process for color removal and chemical oxidation demand (COD) reduction. 12 The coagulation method has been used for primary and secondary treatment of textile dyeing wastewater decolorization due to its low operational cost.13,14 Therefore, the coagulation method is the most common option for the treatment of dye containing effluents.15,16Various research studies have been done on conventional coagulants such as lime, alum, iron-based salts (Fe+2 and Fe+3) and its combinations for textile effluent decolorization.17–20
Magnesium salts, especially MgCl2, are novel and attractive coagulants for removal of reactive dyes from textile effluents under alkaline conditions. Some research studies are available for textile dye wastewater decolorization.21,22 The most important feature of chemical treatment with magnesium chloride is that magnesium ions (Mg+2) are converted to precipitable magnesium hydroxides at alkaline pH. Also, magnesium hydroxide precipitates provide a large adsorptive surface area and a positive electrostatic superficial charge, which attract negatively charged particles (contaminants) from the effluent.23,24
Few studies are available on the effectiveness of iron- and magnesium-based coagulants, along with lime, for decolorization of textile wastewater. Therefore, the present study evaluates the comparative effectiveness of coagulants MgCl2, FeSO4, and their combination with alkalis (lime and NaOH) for color removal. The effects of dosage and pH of coagulant on color removal were evaluated for each combination at an optimal pH. Coagulant-treated wastewater was reused in the wash-off process after dyeing to reduce water consumption. Color difference and colorfastness properties of fabrics washed with coagulant-treated wastewater were also assessed.
Materials and Methods
Materials
All chemicals in this study, including magnesium chloride (MgCl2·6H2O), ferrous sulfate (FeSO4·7H2O), sodium hydroxide (NaOH), and calcium hydroxide (Ca(OH)2) of commercial grade (Merck), were used without any further purification. C.I. Reactive Black 5 dye was supplied by KISCO Dyes–Korea. The chemical structure of Reactive Black 5 dye is shown in Fig. 1. This dye is widely used in textile dyeing; therefore, it was chosen as the representative refractory chemical. 25 A bleached, 100% cotton knitted fabric sample (5 g) was used for the wash-off process at a 1:10 liquor ratio (LR) throughout the study. Distilled deionized water (DDW) was used for preparing synthetic aqueous solutions at 100 ppm dye concentration without addition of any dyeing auxiliary chemicals. All experiments were carried out at ambient temperature (24 ± 1 °C). 1.0 N H2SO4, NaOH, and Ca(OH)2 were used to adjust the pH of the synthetic dye solution.
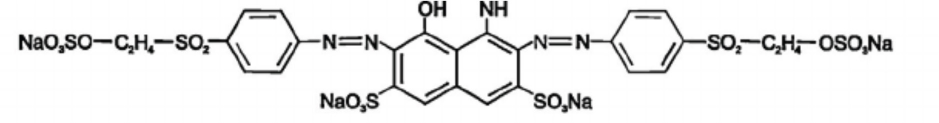
Chemical structure of C.I. Reactive Black 5 dye.
Chemical Coagulation Methodology
In all chemical coagulation experiments, 500-mL sample volumes were used for each run. MgCl2 and FeSO4 doses from 100 to 600 ppm were introduced into the synthetic dye wastewater sample to determine the optimal dose that yielded the maximum color removal. Each experiment consisted of rapid stirring (3 min at 120 rpm), followed by slow stirring (15 min at 50 rpm), and then by a 30 min settling time. After this, the supernatant was taken and filtered through 45-mm pore size Whatman filter paper. The coagulant treated and filtered wastewater was used in the fabric wash-off process. All experimental conditions were the same for both fabric samples, either washed of with fresh water or with coagulant-treated wastewater.
Dyeing and Wash-Off Procedure
Fabric dyeing took place using fresh water in an IR laboratory dyeing machine (Datacolor) at at a 1:10 LR, in the presence of 80 g/L of sodium sulfate and 20 g/L of sodium carbonate. All fabric dyeing runs were carried out at 60 °C for 45 min under isothermal conditions. A standard isothermal dyeing process is shown in Fig. 2. At the end of fabric dyeing, one fabric sample was washed-off with fresh water. This fabric sample was considered as the reference sample. The remaining fabric sample wash-offs were washed with coagulant-treated wastewater. Typical washing steps used in these experiments are shown in Table I. Coagulant-treated wastewater wash-off samples were compared with the reference samples and a comparison was made of color difference and colorfastness.
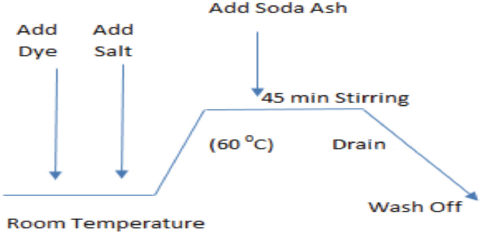
Standard isothermal dyeing process for cotton.
Typical Wash-Off Procedure a
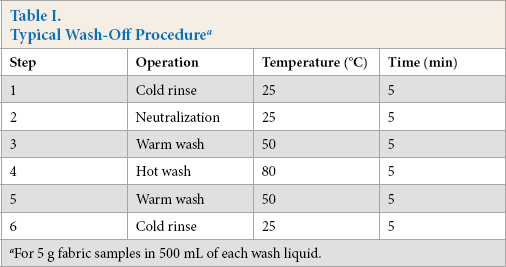
For 5 g fabric samples in 500 mL of each wash liquid.
Color Removal Determination
Sample dye concentrations were determined by absorbance measurements using UV-Visible spectrophotometer DR/4000 (Hach-USA). Percentage color removal was obtained using Eq. 1.
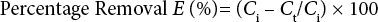
E (%) is removal efficiency, Ci is the concentration of the untreated sample, and Ct is the concentration of the coagulant-treated sample.
Color Difference Determination
Reflectance measurements of the reference and coagulant-treated wastewater fabric samples were measured using a Spectrafash 600 PLUS-CT (Datacolor) spectrophotometer. Color values (lightness L*, red/green axis a*, yellow/blue b*, chroma c*, and hue h) were calculated and total color differences (color difference ΔE*, lightness difference ΔL*, chroma difference Δc*, and hue difference ΔH*) were determined using the CMC (2:1) equation. 26
Colorfastness Determination
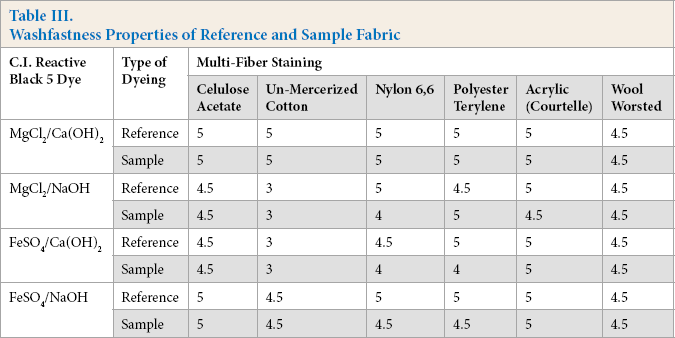
Colorfastness properties of all dyed samples were measured using the ISO 105-C06 wash test. 27 A piece of the dyed sample was attached to an adjacent multi-fiber fabric strip containing six different types of fibers (cellulose acetate, unmercerized cotton, nylon 6,6, polyester terylene, acrylic, and worsted wool). The degree of staining on the adjacent multi-fiber fabric strip were measured after drying. The rating scale for colorfastness ranged between 1 (poor) to 5 (excellent). The results of samples and reference were then compared.
Results and Discussion
pH Variation
The color removal rate of Reactive Black 5 dye by magnesium- and iron-based salts is governed by the pH of the synthetic dye solution. The pH variation controls the charge on hydrolysis products as well as precipitation of metal hydroxides. Therefore, the effluent pH must be controlled in the chemical coagulation process. It is evident from Fig. 3 that the color removal efficacy increased by increasing the pH from 3 to 12 when MgCl2 and FeSO4 were used as coagulants. The color removal efficacy by MgCl2/Ca(OH)2 was almost 97% when the pH value was 12. Several researchers have also reported pH 12 as an optimal pH for coagulant MgCl2.1,28 At alkaline pH (12), the magnesium ions actively react with impurities and formed magnesium hydroxide precipitates. MgCl2 together with Ca(OH)2 at pH 12 removes the dyes through a two-fold coagulating mechanism (i.e., charge neutralization, as well as adsorption). 24
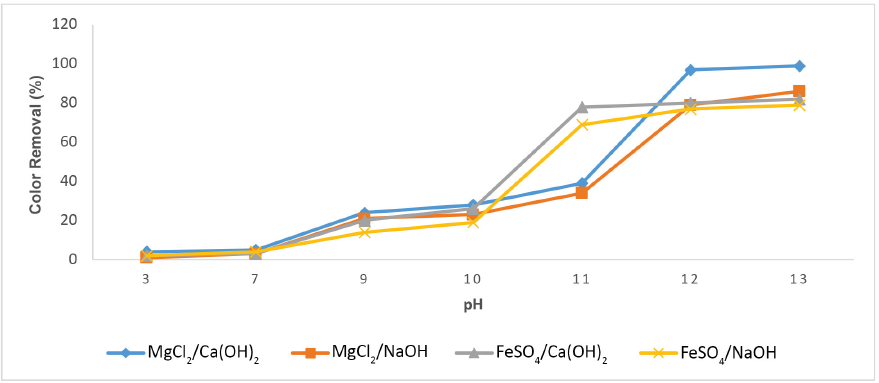
Color removal of C.I. Reactive Black 5 dye by MgCl2 and FeSO4, with coagulant at different pH values.
The coagulant FeSO4 yielded 85% color removal efficiency under the same conditions of pH and coagulant dose. FeSO4/NaOH and MgCl2/NaOH exhibited less than 80% color removal efficiency. An increase in pH above 12 gave a negligible increase in color removal percentage that was observed for all coagulants. At a pH less than 12, the results showed poorer color removal efficiency. This is because the magnesium (Mg2+) ions precipitated.29,30 At the optimal pH value, MgCl2/Ca(OH)2 was found to be a superior coagulant in color reduction as compared to FeSO4/Ca(OH)2, FeSO4/NaOH, and MgCl2/NaOH.
Coagulant Dosage
For coagulation processes to be competitive with other treatment methods, it is crucial that there be good control of coagulant dosage. Coagulant dosages varied from 100 to 600 ppm to get the optimal dose that achieved maximum color reduction. The optimal dosage of coagulant was determined by varying the coagulant doses and adjusting the optimal pH either with lime (Ca(OH)2) or with NaOH. Fig. 4 shows that with increasing coagulant dose, the color removal efficiency also increased in almost all combinations. About 97% color removal efficiency was observed using 400 ppm of MgCl2 for the wastewater containing Reactive Black 5 dye when lime was used to maintain the optimal solution pH. Coagulant combinations such as FeSO4/Ca(OH)2 gave 85% color removal efficiency and FeSO4/NaOH was also found to be effective which gave more than 80% color removal efficiency at a 400 ppm coagulant dosage. However, a considerable decrease in color removal efficiency of 78% was observed for MgCl2 at a 400 ppm coagulant dose, when NaOH was used to maintain the optimal pH. Adjustment of optimal pH with lime with NaOH showed an improvement in color removal efficiency. Lime works as coagulant aid and is capable of giving a certain degree of color reduction. 23 At a dose of 600 ppm, MgCl2/NaOH and FeSO4/NaOH gave >90% color removal efficacy. At <400 ppm coagulant doses, less effectiveness for color removal was found for all combinations.
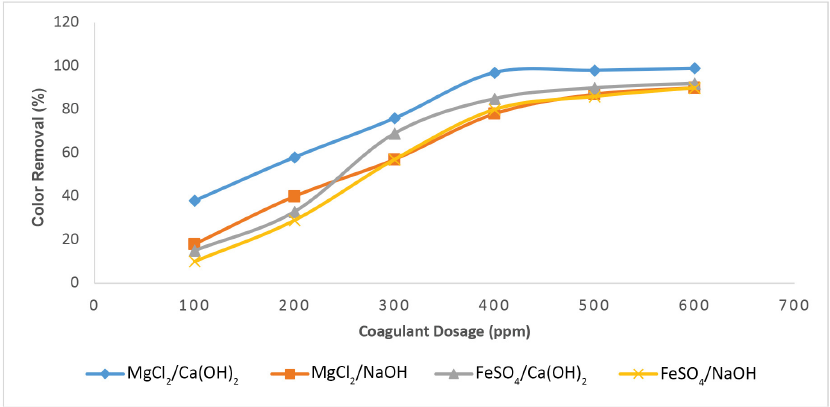
Color removal of C.I. Reactive Black 5 dye by MgCl2 and FeSO4, with coagulant at varying dosages.
Dye Concentration
Fig. 5 shows the effect of dye concentration on decolorization at the optimal pH using coagulant doses of 25 to 200 ppm. At 100 ppm concentration, color removal was good for both coagulants. The color removal efficiency vigorously decreased for dye concentrations >100 ppm. At dye concentrations of 100 ppm, the removal efficiency for MgCl2/Ca(OH)2 coagulant was >90%; at 200 ppm, the color removal efficiency was 60%. A substantial decrease in color removal was observed at the 250 ppm dye concentration for all coagulant combinations. Approximately 12%, 11%, and 9% color removal efficiencies were achieved for the coagulants FeSO4/Ca(OH)2, MgCl2/NaOH, and FeSO4/NaOH, respectively. Greater dye concentrations required greater coagulant doses. The effect of dye concentration on the process efficacy provided important information for operations at industrial wastewater treatment plants.
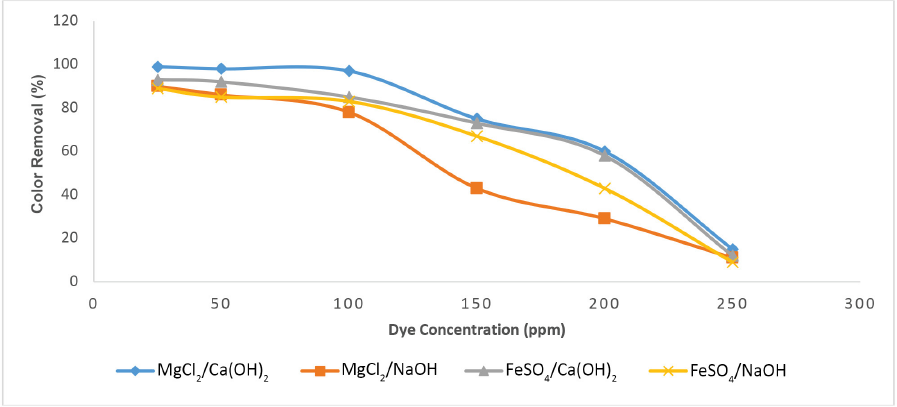
Color removal of C.I. Reactive Black 5 dye by MgCl2 and FeSO4, with coagulant at varying dye concentrations.
Coagulant-Treated Wastewater Reuse
The practicality of coagulant-treated wastewater reuse in the dyeing wash-off step was studied. A reference fabric wash of was performed in fresh water and the sample fabric wash of in coagulant-treated wastewater, followed by comparisons of their color difference and colorfastness properties.
Color Difference
Table II displays total color difference (ΔEcmc) values of fabric samples washed of using four different coagulant-treated wastewater systems as compared to samples washed of with fresh water. The color difference values for MgCl2/Ca(OH)2 and FeSO4/NaOH were <1.0, which was considered commercially acceptable. Coagulant-treated wastewater fabric samples treated with MgCl2/NaOH and FeSO4/Ca(OH)2 showed color differences of 1.14 and 1.75 respectively.
Sample Color Difference (ΔEcmc) after Coagulant Treatment

Colorfastness to Washing
Table III shows the colorfastness to washing properties for both reference and coagulant-treated wastewater samples. Almost identical colorfastness properties were observed for both the reference and coagulant-treated samples. The colorfastness results of fabric samples washed with coagulant-treated wastewater were in the range of 4.5 to 5.0, which are considered excellent in the textile industry. The good colorfastness values confirmed that coagulant-treated wastewater can effectively be reused in the wash-off step of reactive dyeing.
Washfastness Properties of Reference and Sample Fabric
Conclusion
The present work explores the effectiveness of the chemical coagulation process in color removal from a synthetic solution of C.I. Reactive Black 5 dye. The experimental results showed that maximum color removal efficiency occurred when the pH value of the dye solution was highly alkaline (12). Magnesium chloride (MgCl2) with lime (Ca(OH)2) was a more effective coagulant than iron sulfate (FeSO4). The color difference (ΔEcmc) results of fabrics washed with coagulant-treated wastewater when compared to fresh water wash-off were found to be good (∼1.0) and washfastness values were at an acceptable level for the textile industry.
The reuse of coagulant-treated wastewater in wash-off steps resulted in fresh water savings and also pollution reduction into natural water bodies.
This study concluded that the dye-containing wastewater can be used, after its treatment by magnesium chloride, in wash-off processes without affecting the final fabric quality. This method reduced the amount of generated wastewater, and consequently, the load on the wastewater treatment system, as well as fresh water consumption. The fresh water savings can help avert future water shortages. Further studies should be performed on treatment of real dyeing and finishing effluents, which contain considerable quantities of organic dyes, textile auxiliaries, salts, and surfactants.
