Abstract

This graphical abstract describes in general the treatment processes of dye-polluted water using the prepared samples. Activated carbon undergoes both adsorption and catalytic oxidation processes, further the photocatalytic efficiency of each catalyst supported by activated carbon enhanced the removal of dye completely in a short time in addition to adsorption process in the early irradiation time.
Activated carbon (AC) prepared from activation of ground peach stones with phosphoric acid under flowing air was used as a support for CeO2 and ZnO nanoparticles through hydrothermal method at 200℃ for 120 min and followed by the calcination at 400℃ for 180 min. The produced samples were named as AC, CeO2–AC, and ZnO–AC, respectively. The characterization of samples was made by means of scanning electron microscope, energy dispersive spectroscopy, X-ray diffraction, Fourier transform infrared spectroscopy, and Brunauer–Emmett–Teller surface area. A remarkable impact on the surface, structural, and textural properties of AC support was obtained as a result of loading for CeO2 or ZnO. Results of X-ray diffraction showed that nanocrystallines CeO2 and ZnO phases on the AC support were formed. Removal efficiency of the prepared samples was investigated through adsorption, photodegradation, and wet peroxide oxidation processes using Maxilon Red basic dye (MR). Accordingly, the prepared samples exhibited different removal behaviors towards MR dye depending on the total surface area and type of catalyst; for example, the maximum removal of MR dye was found in the following orders: AC > ZnO-AC > CeO2-AC, CeO2-AC > ZnO-AC > AC, and AC > CeO2-AC > ZnO-AC via adsorption, photodegradation, and catalytic oxidation with H2O2, respectively. It was found that photodegradation process was the best when compared with the other methods. The photocatalytic activity of CeO2-AC catalyst was the highest. The reusability tests showed that CeO2-AC exhibited superior photocatalytic performance and stability till the fourth run as compared to photocatalytic activity of ZnO-AC which decreased from 90% to 78% in the fourth run. Therefore, the prepared CeO2-AC catalyst is more effective than ZnO-AC as confirmed by reusability tests.
Introduction
As one of the most reactive rare earth oxides, ceria or cerium dioxide (CeO2) is a ubiquitous catalyst used in various catalytic reactions such as redox reactions, water–gas shift reaction, thermo-chemical water splitting, wet oxidation of organic pollutants, and photocatalysis (Channei et al., 2014; Ji et al., 2009; Khan et al., 2014; Li et al., 2012; Neri et al., 2002; Rao and Mishra, 2003; Sun et al., 2012). In photocatalysis, CeO2 has attracted much interest due to its wide band gap (Eg = 3.2 − 3.4 eV) (Channei et al., 2014; Ji et al., 2009), and therefore, photogeneration of charge carriers can be obtained under UV light. Khan et al. (2014) reported that the modified CeO2 exhibited superior photocatalytic activities rather than the pristine CeO2 in the degradation of 4-nitrophenol and methylene blue using a visible light (λ > 400 nm).
Zinc oxide (ZnO), as an admirable semiconductor material, also has attracted great attention in numerous applications, such as piezoelectric devices, sensors, transparent electronics, optics, optoelectronics, and actuators (Arya et al., 2012; Dastkhoon et al., 2015; Farag et al., 2016; Mahmouda and Fouad, 2015). Moreover, in photocatalytic degradation of different dyestuff, ZnO exhibited better photocatalytic efficiency than TiO2, although the band gap energy of ZnO is the same as that of TiO2 (∼3.2 eV) (Arya et al., 2012; Mahmouda and Fouad, 2015).
In general, the supported porous materials are used in large scale during heterogeneous catalysis processes. The role of the support is to disperse the metal particles and maintain them from sintering. The dispersion of the metallic components on the support can influence the electronic and catalytic properties of the supported metal particles by electron transfer or chemical bond formation (Rao and Mishra, 2003). Activated carbon (AC) is one of the most important catalyst supports (Erhan et al., 2001; Pastor-Pérez et al., 2014; Pulido et al., 2009; Rodríguez-Reinoso, 1998; Roosta et al., 2015). It has unique characteristics to be used as a catalyst support, such as thermostable and acid/base-stable properties, easily recovering supported precious metals on it, and the possibility of tailoring its pore structure and surface chemical properties according to the targeted aims of the catalyst producers (Erhan et al., 2001). Additionally, not only the supporting material requires high-specific surface area, suitable pore structure, regular bead size, and sufficient abrasion resistance, but also the catalyst metal should have a high activity, dispersity, and good stability on its support (Erhan et al., 2001).
The objectives of the current work were (i) preparation of CeO2 or ZnO nanoparticles supported on porous AC via hydrothermal method and then followed by calcination at 400℃ for 180 min; the AC used was derived previously from chemical activation of peach stones with H3PO4 and air at 500℃ for 120 min (Girgis et al., 2007), and (ii) study the adsorption and catalytic properties of the prepared catalysts in relation to their physicochemical characteristics of the prepared samples through three different processes such as adsorption, visible light irradiation, and wet oxidation with H2O2 for removing Maxilon Red 3GL basic dye from the aqueous solutions. The influence of supported metal oxides on surface, structural, textural, and catalytic properties of AC was further explored using different techniques such as scanning electron microscope–energy dispersive spectroscopy (SEM-EDS), X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), and Brunauer–Emmett–Teller surface area (BET).
Experimental
Chemicals
Ortho phosphoric acid (H3PO4, 85 wt.%, Mr = 98.00 g/ mol) was supplied from Rasayan (Turkey). Nitric acid (HNO3, 65–68%, Mr = 63.00 g/mol) was purchased from Alpha-Chemika. Sulfuric acid (H2SO4, 95–98 wt.%, Mr = 98.08 g/mol) and sodium hydroxide (NaOH, 98 wt.%, Mr = 40.00 g/mol) were obtained from Panreac. Zinc acetate hydrate [Zn(O2CCH3)2(H2O)2] and ceria (CeO2) powders were purchased from Sigma-Aldrich. Hydrogen peroxide solution (H2O2, 30 w/v%, Mr = 34.01 g mol−1) was obtained from Aldwic. Maxilon Red 3 GL dye (Yoracryl Red RL C.I. basic red 60, CAS#61847-54-9, BESTCHEM company, Germany). All solutions were prepared with distilled water.
Preparation of AC as a substrate
AC was obtained from chemical activation of peach stone shells impregnated with 50 v/v% H3PO4 at 500℃ under flowing air for 120 min as described previously (Girgis et al., 2007). The obtained AC was oxidized with 5 M solution of HNO3 acid following the procedure described earlier by Pereira et al. (2003). The treated AC sample was separated from nitric solution and washed thoroughly with hot water and then distilled water till pH of filtrate becomes 6.0. Afterward, the sample was dried at 100℃ overnight and denoted as “AC”.
Preparation of CeO2 and ZnO supported on AC
Approximately 10 wt.% of each metal oxides was loaded separately onto the surface of AC substrate via hydrothermal process as follows. Typically, a definite amount of a commercial CeO2 powder was dispersed in 10 mL isopropanol and 20 mL distilled H2O under heating at 50℃ with vigorous magnetic stirring for 60 min. After that, powdered AC was added to the suspension of cerium dioxide under stirring for another 60 min. The mixture was transferred to Teflon-autoclave tube and treated hydrothermally at 200℃ for 120 min and then followed by filtration, washing, and drying at 100℃ overnight. Then, the dried sample was calcined at 400℃ for 180 min and the final yield of CeO2-AC catalyst was found to be 33.8%.
In ZnO-AC preparation, an amount of zinc acetate was dissolved in 20 mL distilled water and then powdered AC was added and stirred for 60 min at 50℃. This mixture was treated hydrothermally in Teflon-autoclave tube and heated at 200℃ for 120 min. Then the resulting solid was separated by filtration to wash and dry at 100℃ overnight. Finally, the dried sample was thermally treated at 400℃ for 180 min and the final yield of ZnO-AC catalyst was found to be 38.1%.
Characterization of the prepared catalysts
The surface structure and chemical composition analyses of the prepared catalyst samples were estimated by field-emission scanning electron microscope (FE-SEM) combined with energy-dispersive spectroscopy and electron backscatter diffraction (FE-SEM, FEI Quanta FEG-250, EDS). The crystalline phases in the obtained samples were determined by XRD analysis using a Bruker diffractometer (Bruker D8 advance, Germany). The patterns were run with CuKα1 target with monochromator at 40 kV, 40 mA. The fundamental surface functional groups of the prepared samples were determined by FTIR, using KBr pellets (JASCO, FT/IR-460 plus). The textural properties such as BET (SBET, m2/g), total pore volume (VP, cm3/g), and average pore radius (RP, nm) were determined using nitrogen adsorption analysis at −196℃ (BEL-Sorp, Microtrac Bel Crop, Japan). Moreover, slurry pH values of the prepared samples (pHslurry) were measured according to the following method: 0.05 g of the sample was mixed with 25 ml of distilled water and allowed to boil for 30 min in stoppered glass bottle. After cooling, the slurry pH of the samples was determined using a digital pH meter (HANNA, Model HI1110), allowing 5 min for the pH probe to equilibrate and then the pH was recorded in order to identify the nature of surface groups (i.e., acidic, basic or neutral).
Determination of adsorption and catalytic properties of the prepared catalysts
In this study, the adsorption and catalytic performances of the prepared catalysts were studied as a function of removal of Maxilon Red dye (MR) through batch adsorption, photodegradation, and catalytic wet peroxide oxidation (CWPO) methods. Throughout the experiments the pH value was adjusted using 0.1 mol/L of NaOH or H2SO4 solution. The adsorption efficiency of the prepared catalysts (AC, CeO2-AC and ZnO-AC) was studied with different initial concentrations of Maxilon Red 3GL dye (20–100 mg/L) under batch working parameters: catalyst dose (1 g/L), pH of dye solution with catalyst (6), and contact time (180 min). The amount of MR basic dye taken by the obtained samples was calculated by the mass–balance equation
To calculate the maximum adsorption capacity (mg/g), Langmuir isotherm model was used in a linear form
Also, the influence of the isotherm shape on whether the adsorption is “favorable” or “unfavorable” at highest initial concentration of the dye, C0, can be described by a term “RL” a dimensionless constant separation factor in the equation
The calculated value for RL indicates the nature of adsorption process as given: irreversible (RL = 0), favorable (0 < RL < 1), linear (RL = 1), and favorable (RL > 1). The values of RL were found to be lower than unity, suggesting that the adsorption system is fulfilled using the prepared samples.
The photocatalytic behavior of the prepared catalysts was demonstrated using visible light under different pH values and initial concentrations of dye in the range from 4.0 to 10 and from 10 mg/L to 30 mg/L, respectively, keeping temperature and catalyst dose constant. The visible light irradiation was carried out in a photo-reactor using house hold fluorescent lamp (44 W Philips, λ > 420 nm). This lamp irradiates at a distance of 10 cm from the surface of dye solution.
CWPO process was performed to decompose the MR in the presence of H2O2 using the prepared catalysts under fixed operating conditions as follows: catalyst dose (40 mg), initial concentration of MR dye (100 mg/ L), volume of dye solution (40 mL), temperature (T = 50℃), and pH (6). The decolorization degree of the dye solution at different time intervals was measured by the change in the absorbance maxima of MR dye at 520 nm using UV–vis spectrophotometer (Shimadzu-PC2401). In order to assess the reproducibility and error of the applied experiments the analyses were carried out in triplicate, and the results showed that the relative errors were lower than ±5%.
Stability and reusability tests
In the first run, about 50 mg of CeO2-AC or ZnO-AC photocatalyst was conducted with 50 mL dye solution (C0 = 50 mg/L) and kept under stirring in the dark for 15 min to saturate the adsorption of MR and then exposed to a visible light. After 30 min, the sample was separated from the treated dye solution by filtering, washing, and drying. After that, it was added again to fresh MR dye solution with the same initial concentration, being again kept in the dark for about 15 min, and then exposed to visible light (i.e. for three runs consecutively).
Results and discussion
Structural and textural properties of the prepared catalysts
Figure 1(a–c) shows different SEM images for the prepared samples indicating significant changes in the surface of AC after modification with CeO2 and ZnO nanoparticles. It can be concluded that both metal oxides exhibited a strong impact on the surface properties of the AC substrate. Irregular porous structure is observed through the surface of AC. In CeO2-AC sample, agglomerated pellets or layers with wavy shapes (white color) of CeO2 nanoparticles are covered the surface of AC substrate with presence of some pores. On the other hand, the surface of ZnO-AC sample is homogeneously covered with layers of ZnO particles which accompanied with formation of wide pores also. Accordingly, the total surface area and total pore volume are decreased significantly as seen in Table 1. Whereas the pore diameter is increased apparently from 2.02 nm to 6.27 and 8.21 nm when AC sample was loaded with either CeO2 or ZnO nanoparticles, respectively. This finding indicates that the loading of AC using CeO2 or ZnO particles led to a widening in the micropores and consequently a substantial diminishing of surface area is obtained. Probably the obtained surface areas of CeO2-AC and ZnO-AC samples are attributed to the covered surface area by CeO2 or ZnO nanoparticles on the AC surface as shown in their SEM photographs. EDS analysis was used to analyze the chemical composition of a material under SEM. EDS of the prepared samples is given in Table 1, which confirms the existence of main elements such as carbon, oxygen, phosphorous with the addition of cerium in CeO2-AC and zinc in ZnO-AC in atomic percentage. It can be seen that the atomic percent of carbon was decreased significantly from 74.7% to 35.3% and 41.9% in CeO2-AC and ZnO-AC, respectively, which maybe attributed to the annealing process of catalyst which happened on surface of AC substrate.
SEM photographs of the prepared samples: AC (a), CeO2-AC (b), and ZnO-AC (c). The surface and adsorption properties of the investigated catalysts.

The crystalline phases of the prepared samples were characterized by XRD analysis. Figure 2 displays the XRD patterns of the prepared samples. Considerable structural changes in the amorphous nature of AC support are detected after loading with ceria and ZnO. The XRD results showed that the CeO2-AC and ZnO-AC samples primarily consists of nanocrystallines CeO2 and ZnO phases on the AC support. For AC sample, two broad XRD peaks at 2θ = 25o and 42.8o are observed which currently associated with diffraction from the 002 and 100/101 set of planes in graphite, respectively. A high fragmentation in microstructure of AC is obtained when CeO2 nanoparticles loaded on the surface of AC, referring to formation of an organized structure in the lattice of AC substrate. In XRD profile of CeO2-AC, many peaks at 2θ = 28.6o, 33.3o, 47.6o, 56.4o, 59.1o, 69.7o, 76.7o, and 78.8o are observed. Such peaks reveal the well-developed cerium oxide which matches with (ICDD PDF No. 81-0792) (Li et al., 2012) average crystallite sizes in the range between 19.6 nm and 39 nm. Also, it can be shown clearly that CeO2 increased the gasification of AC surface to form crystalline structure as detected by XRD patterns and EDS analysis. On the other hand, ZnO loading on AC showed partially organized structure by forming three sharp XRD peaks at 2θ = 24.8o, 29.6o and 47.5o within the broadening XRD peaks which are related to graphite structure in AC. The first two peaks indicated the interaction of ZnO with AC surface and the second one indicated the presence of free ZnO. The average crystallite sizes are 39.3 nm, 78.5 nm, and 39.2 nm, respectively. Hence, nanosized particles of CeO2 or ZnO can be synthesized over AC surface during the employed procedure. In conclusion, the loading of ceria or ZnO onto the surface of AC under heat treatment at 400℃ led to significant structural alterations in lattice of AC including an increase in crystallinity with destruction in basic graphitic structure of AC and thus the physico-chemical, adsorption, and catalytic properties of the AC support could be affected.
XRD profiles of the prepared samples.
The investigation of functional groups present on the surface of prepared samples was confirmed by the FTIR analysis. Figure 3 displays the FTIR spectra of the following samples: AC and both CeO2-AC and ZnO-AC catalysts before and after photocatalysis process. The FTIR spectra of CeO2-AC and ZnO-AC catalysts before photodegradation of dye show most the absorption bands which appeared at the spectrum of AC, however, their intensities are shifted significantly to low transmittance. Most of these peaks are located approximately at the following regions; broad band at 3430 cm−1, small bands at 2930 cm−1 and 2860 cm−1, shoulder and sharp bands 1625 cm−1 and 1457 cm−1, broad band between 1300 cm−1 and 1000 cm−1, and small bands between 870 cm−1 and 450 cm−1, respectively. In that order, the resulting surface functional groups relating to these regions are found to be hydroxyl group of water molecules adsorbed; C–H symmetric and asymmetric stretching of residual methylene groups on the surface; –COOH carboxylic group or NH2–C = O– amide groups and conjugated with C–OH alcohol group in the aromatic rings at 1457 cm−1; the absorption bands of phosphorous containing groups (e.g. P = O, P–O–P, P = OOH, P–O–C) and C–O groups are located between 1300 and 1000 cm−1 (Bandosz and Ania, 2006; Puziy et al., 2002), consecutively. It is observed that a shoulder peak at 450 cm−1 is obtained in FTIR spectrum of ZnO-AC indicating the presence of few free ZnO (Farag et al., 2016; Pulido et al., 2009). Additionally CeO2 and ZnO catalysts expanded the absorption bands at 3430 cm−1 (–OH), 1625 cm−1 (COOH), and 1082 cm−1 (C–O), confirming the interaction of ceria and ZnO with these functional groups. These findings are obtained also with significant effect when CeO2 was loaded on the surface of AC. Finally, the last region is usually associated to the residues of aliphatic groups and the out-of-plane deformation mode of C–H in substituted benzene rings, resulting in carbonization of lignocellulosic materials (Puziy et al., 2002). On the other hand, the changes in functional groups over the surface of catalysts after photodegradation the MR dye can be assessed in the FTIR spectra of CeO2-AC-dye and ZnO-AC-dye. It can be seen that all peaks shifted to very low-absorption intensity with two new bands appearing at 1383 cm−1 (C–O) and 487 cm−1 (C–H) as a result of the photodegradation of the dye onto the surface of both catalysts.
FTIR spectra of the prepared samples (1) AC (2)CeO2-AC, (3) ZnO-AC, (4) CeO2-AC-dye and (5) ZnO-AC-dye.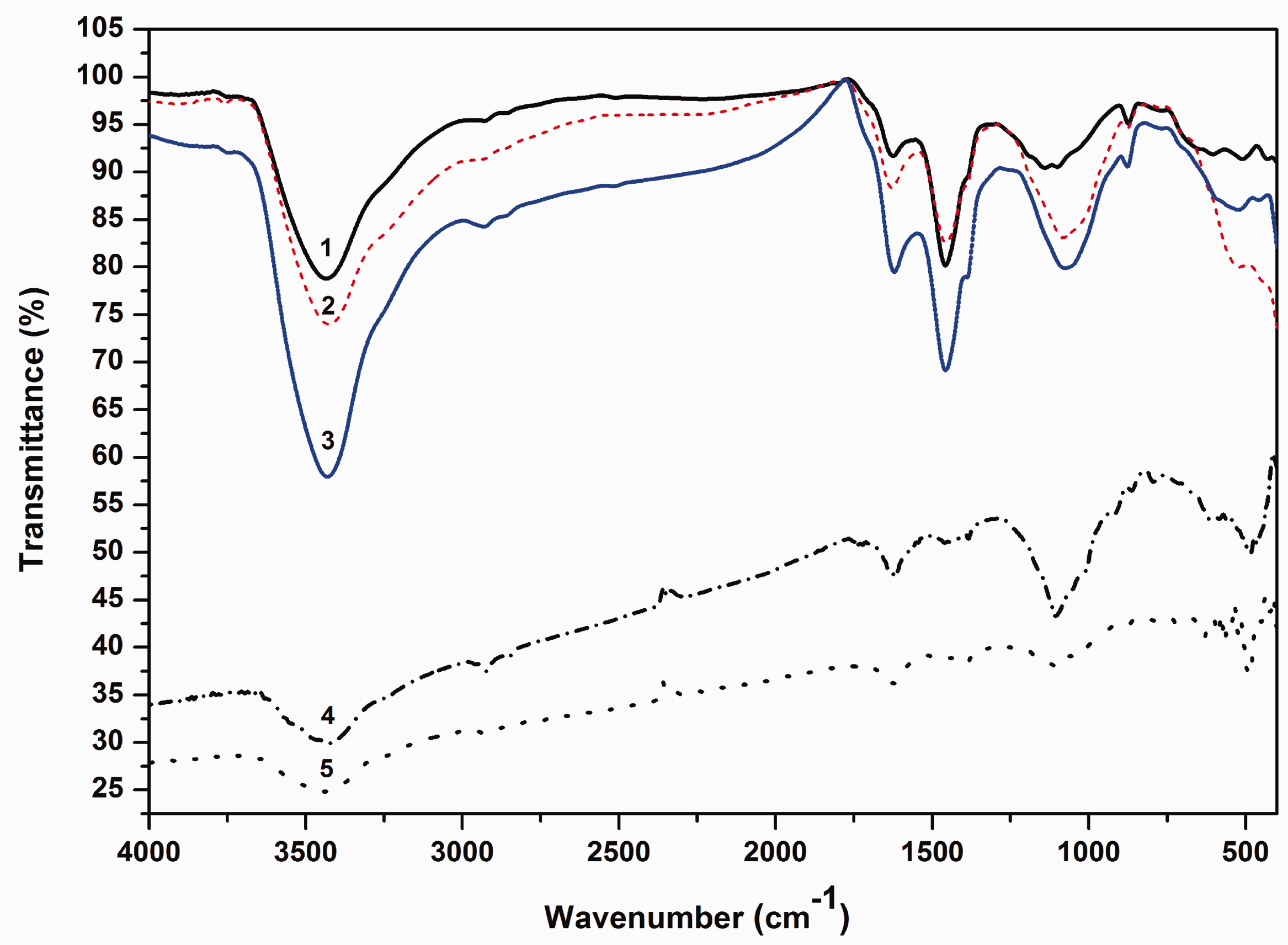
Moreover, the slurry pH of prepared samples (pHslurry) may give a good indication about the surface oxygen complexes and the electronic surface charges of them. For example, when the solution pH is higher than the pHslurry, the surface of the solid is negatively charged, favoring the adsorption of cationic species, while pH of the solution is lower than the pHslurry, the surface of solid is positively charged and would favor the adsorption of anionic species (Radovic and Rodríguez-Reinoso, 1997). The surface of prepared samples is slightly acidic because pHslurry is between 5.15 and 5.65, and thus when the prepared samples contacted with MR basic dye of pH ∼ 6.25, forming surfaces with negative charges and then the adsorption of MR basic dye from aqueous solution could be suited.
Adsorption of the prepared samples
Figure 4 shows the adsorption isotherms of MR basic dye on the prepared samples. It was found that the MR basic dye adsorbed substantially more on AC than on CeO2-AC and ZnO-AC, with monolayer adsorption capacity higher three times than that of both catalysts as listed in Table 1. Also it is observed that most of adsorption occurred on AC sample within 180 min as shown in Figure 4. These adsorption isotherms assumed a conventional Langmuir isotherm type. The values of R2 in Table 1 confirm that Langmuir isotherm is well-described in the adsorption process of MR basic dye solution onto the prepared samples. Also the adsorption data in Table 1 show that the sample with the highest internal porosity (BET surface area, total pore volume and pore diameter) has the highest adsorption capacity for MR dye as shown in the subsequent order: AC > ZnO-AC > CeO2-AC.
Absorption isotherms of the MR basic dye onto the prepared samples at 25℃.
Effect of pH on photoactivity of the prepared catalysts
The pH of dye solution is a vital parameter to determine the optimal photoactivity of the prepared samples. It affects the decolorization process of dyes on the surface of catalysts. Figure 5 shows the variation in the removal of dye using AC, CeO2-AC, and ZnO-AC as a function of pH of MR dye solution. By fixing the initial dye concentration (20 mg/L), catalyst dose (1 g/L), and temperature (25℃) the photodegradation of MR basic dye tests under changing pH from 4 to 10 was employed. The results showed that the decolorization efficiency is significantly pH dependent. The maximum removal efficiency of the MR basic dye using all prepared samples was obtained at pH 10. The high removal efficiency at pH 10 may be attributed to the high concentration of hydroxyl anions. Such hydroxyl anions can be converted into oxidizing radicals such as •OH and •−O2 species which can accelerate the photodegradation process. As shown in Figure 5, AC sample attained 76% of removal efficiency at 150 min; CeO2-AC sample photodegraded the dye completely in 60 min whereas ZnO-AC sample achieved full degradation in 120 min. Thus the prepared ceria-AC catalyst exhibited superior photocatalytic activity than that of ZnO-AC catalyst. Probably this result seems to be due to its high wide band gap energy and redox potential of the Ce4+/Ce3+ couple as well as its strong light absorption in the UV region which enhanced to absorb visible light when combined with other materials (Ji et al., 2009; Li et al., 2012; Sun et al., 2012).
Influence of pH on the photoactivity of prepared catalysts (C0 = 20 mg/L, catalysts dose: 1 g/L and temperature: 25℃).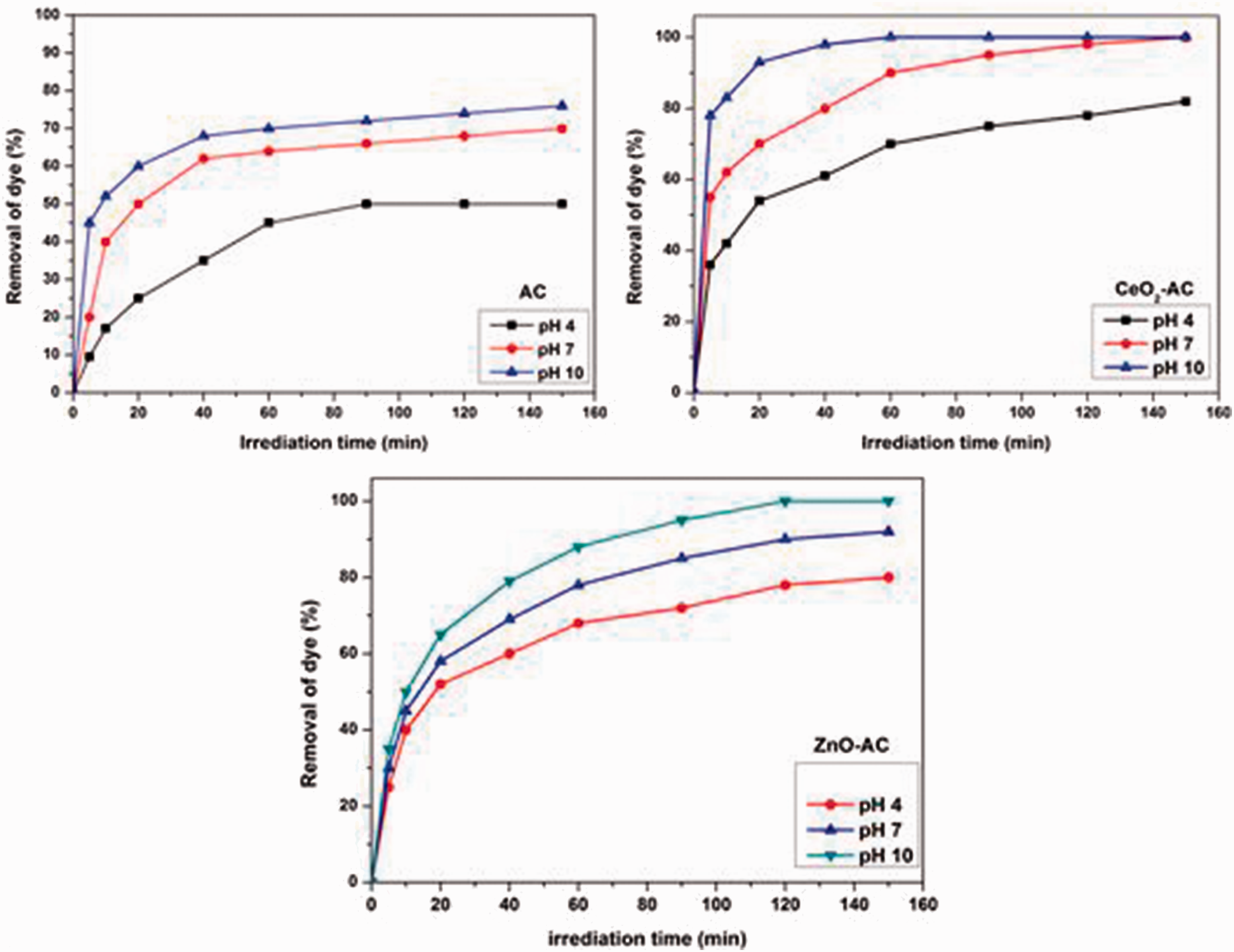
Effect of initial dye concentration on photoactivity of the prepared catalysts
The influence of initial dye concentration on the removal efficiency of the prepared catalysts, CeO2-AC and ZnO-AC, was studied. Figure 6 illustrates the change in removal efficiency when the MR dye concentration varied from 10 to 30 mg/L under fixing other parameters (e.g. catalyst dose: 1 g/L, pH 10 and temperature: 25℃). Generally, in the smaller initial concentration the higher removal efficiency is found. However, CeO2-AC showed a relative decrease in the removal efficiency at a low irradiation time when the initial concentration of dye increased from 10 to 20 mg/L while a considerable decrease further increased to 30 mg/L. In case of ZnO-AC, a remarkable decrease is found in the removal efficiency when the concentration of dye increased from 10 to 30 mg/L with the increase in irradiation time. This may be attributed strongly to the high photocatalytic property of CeO2 comparatively to ZnO which can accelerate the degradation of the dye at a low irradiation time (∼60 min). Overall, the decrease in removal efficiency with increase the initial concentration may be ascribed to the decrease in number of active sites required for enhancing the photodegradation of dye onto the surface of samples.
Influence of initial dye concentration on the removal efficiency of the prepared samples (pH = 10, catalysts dose: 1 g/L and temperature: 25℃).
Efficiency of CWPO
The catalytic oxidation of prepared AC and supported ceria and ZnO was investigated in the presence of hydrogen peroxide solution. The CWPO runs were carried out under the following conditions: catalyst dose (1 g/L), concentration of MR dye (100 mg/L), concentration of H2O2 (0.1535 mol/L), pH (6), and temperature (50℃). Interesting results are shown in Figure 7, where the AC showed the highest catalytic oxidation of MR basic dye as compared to other catalysts. The amount of MR dye degraded at 150 min by AC, CeO2-AC, and ZnO-AC samples was found to be 100%, 87%, and 72%, respectively. In fact this can be due to the highest BET surface area and the available functional groups which increased the adsorption process over the oxidation process. However, the other samples exhibited good catalytic oxidation performance, although their BET surface areas are less than that of AC sample by ∼98%.
Efficiency of catalytic wet peroxide oxidation in removal of MR dye over the prepared sample (C0 = 100 mg/L, catalysts dose: 1 g/L, pH = 6 and temperature: 50℃).
Reusability studies
After the first photocatalytic degradation of 50 mL of MR dye (50 mg/L) using 50 mg of CeO2-AC or ZnO-AC photocatalyst, the catalyst was separated and reused for three further runs after washing with hot water and drying at 80℃. Figure 8 reveals clearly that the prepared CeO2-AC exhibited a higher photocatalytic performance and stability till the fourth run as compared to photocatalytic activity of ZnO-AC which decreased from 90% to 78% in the fourth run. Therefore, the prepared CeO2-AC catalyst is more effective than ZnO-AC when examined using reusability tests.
Four consequent runs in photodegradation of MR dye over CeO2-AC and ZnO-AC catalysts at 25℃ (C0 = 50 mg/L, catalysts dose: 1 g/L, pH = 10).
Conclusions
In summary, activated carbon (AC) was used as a support for preparing CeO2 and ZnO by means of hydrothermal method at 200℃ and heat treatment at 400℃, consequently. The effect of metal oxides loading on the surface, structural, and textural properties of AC was investigated by SEM-EDS, XRD, FTIR, and BET surface area measurements. It was found that loading of CeO2 or ZnO has remarkable impacts on the surface, structural, and textural properties of AC support. The adsorption and catalytic properties of the prepared samples were further determined after removal of Maxilon Red basic dye from aqueous solution using batch adsorption, photocatalysis, and wet peroxide oxidation process. During adsorption and CWPO processes, the prepared AC exhibited high adsorption capacity due to its large surface area and free functional oxygen groups on the surface as compared to other samples. In photocatalytic process under varying pH and initial concentration of MR dye solution, CeO2-AC sample showed superior photocatalytic degradation of dye in 60 min with fast removal rate initially owing to its wide band gap energy and redox reactions. The photocatalytic degradation of MR dye is significantly dependent on pH and initial dye concentration. Therefore, AC undergoes both adsorption and catalytic oxidation processes, further the photocatalytic efficiency of each catalyst supported on AC enhanced the removal of dye completely in short time, associating with adsorption process in the early irradiation time. Reusability tests showed that CeO2 exhibited a higher photocatalytic performance and stability till the fourth run as compared to photocatalytic activity of ZnO-AC which decreased from 90% to 78% in the fourth run. Finally, the presence of CeO2 or ZnO enhanced the photocatalytic properties of AC whereas it imposes negative impact on its physico-chemical characteristics which make AC as an active adsorbent in wastewater treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.



