Abstract
Gelatin and activated carbon materials have been combined together to obtain a gelatin/activated carbon composite bead form which is ecofriendly, nontoxic, biocompatible, and inexpensive material. In this paper, gelatin/activated carbon adsorption for Pb2+ ions from aqueous solutions was studied experimentally under various conditions. The experimental conditions such as contact time, solution pH, and gelatin/activated carbon dosage were examined and evaluated by using batch adsorption experiments. The maximum adsorption capacity of gelatin/activated carbon for Pb2+ ions was obtained to be 370.37 mg g−1. This maximum capacity was comparable with that of commercial ion exchange resins and it was much higher than those of natural zeolites. The uptake process for Pb2+ ions was found to be relatively fast with 92.15% of the adsorption completed in about 5 min in batch conditions. The adsorption capacity was also strongly solution pH dependent. Adsorption was observed at pH value as low as 2.0 and maximum adsorption was achieved at a pH of approximately 5. The results indicated that the gelatin/activated carbon was effective to be used as an adsorbent for Pb2+ ions removal in wastewater treatment.
Introduction
The excess of Pb2+ ions released in environment from many activities such as agriculture and chemical industry, mining, and metal processing is very harmful to human and animal health. A low lead concentration of 0.006 mg l−1 in blood can damage fetal brain causing diseases of kidney, circulatory system, and nervous system (Barakat, 2011). Therefore, it is necessary to treat lead-containing wastewater prior to its discharge to the environment.
There are various methods for heavy metal removal in industrial wastewater treatment such as ultrafiltration (Majewska-Nowak, 1989), coagulation–flocculation (Prashant, 2012), adsorption (Bansode et al., 2003; Chuenchom, 2004; Dogan et al., 2007; Jiang et al., 2014; Ma et al., 2016; Suna et al., 2010), precipitation (Yüzera et al., 2008), electroplating (Fabíola et al., 2016), evaporation (Zeng et al., 2008), ion exchange (Li et al., 2014), and membrane processes (Lina et al., 2007). Many of these processes when applied to dilute systems with heavy metal ion concentrations of less than 100 mg l−1 are less effective or cost restrictive (Jose and Qiming, 1999). Thus, adsorption technology for the removal of trace amounts of heavy metals from dilute aqueous solutions attends to a growing interest.
Adsorption of heavy metal ions by activated carbon (AC) has been widely utilized as AC is one of the most effective adsorbent because AC has a porous structure with very high surface area, high capacity, and high rate of adsorption (Sirichote et al., 2008) for organic substances (Geçgel et al., 2016) and heavy metal ions (Gupta and Maheshwari, 2014). There have been several reports that AC can be used to adsorb Pb2+ ions. These include AC prepared from sugarcane bagasse (Tran et al., 2016), apricot stone (Kobya et al., 2005), date stones (Jamal et al., 2013), coconut shell (Jyotsna et al., 2004), and walnut wood (Ghaedi et al., 2015). However, AC in fine powder form limits its usage (Ninan et al., 2014). Therefore, studies on the adsorption of Pb2+ ions by granular AC have also been reported. Granular ACs prepared from various low-cost materials such as walnut shell (Kima et al., 2001), pecan shell (Bansode et al., 2003), peanut shell (Wilson et al., 2006), coconut shell (Jyotsna et al., 2005), red mud (Zhua et al., 2007), and cherry stones (Jaramillo et al., 2009) have been studied for heavy metal ion adsorption. However, generally the maximum adsorption capacities of granular AC are lower than those of powdered AC.
The combination of gelatin (GE) and powdered AC in the form of GE/AC bead for the adsorption of pollutants from aqueous solutions as well as actual wastewater samples has been reported (Hayeeye et al., 2015). GE is a biodegradable polymer derived from collagen from various animal by-products. It is an excellent material for the formation of bead forms of powdered materials. Recently, a commercially available GE made from peptone primagen of animal tissue was used to prepare adsorbent bead of GE and powdered AC (2 wt%). It was used for the adsorption of a cationic dye (Rhodamine B) with a maximum adsorption capacity of 13.12 mg g−1 (Hayeeye et al., 2015).
In this work, we developed adsorbent beads by increasing the amount of AC with GE ratio 10% w/w (GE/AC) to study adsorption of Pb2+ ions, which was selected as a representative of the heavy metal ions in effluents from wastewater. Batch equilibrium and kinetics experiments were carried out under various conditions to evaluate and optimize the adsorption efficiencies. Moreover, the adsorption behavior of the GE/AC for the removal of Pb2+ ions from aqueous solutions was investigated.
Materials and methods
Materials and analytical methods
The GE/AC composite bead was prepared by the facile method as follows: a homogeneous solution of GE of 30 wt% was prepared by dissolving GE powder in distilled water. An amount of powdered AC of 10 wt% was added in the GE solution and stirred at room temperature for 30 min. The mixture was then added dropwise into cold water by using a syringe to form the bead form. The GE/AC beads were filtrated out and immersed in 1 wt% of glutaraldehyde solution for overnight. The GE/AC beads were then dried in an oven at 60 ℃ for 6 h. Further details of the preparation methods have been reported elsewhere (Hayeeye et al., 2015). The chemical structure of the GE used is given in Figure 1 (Martin, 1975).
Chemical structure of gelatin.
Compound Pb(NO3)2 was procured from UNILAB, UK and used to prepare aqueous solutions of Pb2+ ions. The concentrations of Pb2+ ions were determined by using an atomic adsorption spectrophotometer (GBC SDS-270) (Martin, 1975).
Characterization of GE/AC
The specific surface area measurements for GE/AC were obtained from nitrogen adsorption isotherms at 77 K using a Quanta Chrome Autosorb automated gas sorption test, at the Institute for Scientific and Technological Research and Services, King Mongkut's University of Technology Thonburi.
The point of zero charge (pHpzc) was determined by using the pH drift method (Jia et al., 1998, 2002). In brief, a series of 50 ml of 0.1 M NaCl solution was adjusted to desired pH from 2 to 12, by using 0.01 M HCl or NaOH. GE/AC of 0.15 g was added into each of this series and then sealed and kept overnight at ambient temperature. The relation of final pH (pHf) to initial pH (pH0) was used to determine the pHpzc value of the adsorbent beads. At this point the initial and final pH values do not differ, and the surface of GE/AC is neutral (Hayeeye et al., 2014).
The exterior surface of GE/AC was obtained by scanning electron microscopy (SEM). The distribution of elemental component for GE/AC can be analyzed using the mapping analysis of scanning electron microscopy with energy dispersive X-ray spectroscopy (SEM/EDX) using a SEM, Quanta 400 at the Scientific Equipment Center, Prince of Songkla University, to characterize the morphology and the elemental components in GE/AC, respectively.
Batch kinetic experiments
Kinetic experiments were conducted at different initial Pb2+ concentrations of 50, 300, and 500 mg l−1. The initial pH of the lead solution was adjusted to 5.5 by using 0.1 M HCl and NaOH solutions. An amount of 0.15 g of GE/AC was added to a continuously shaken container containing 50 ml of Pb2+ ions solution. Samples of 1 ml solution were withdrawn at predetermined time intervals by using a syringe and analyzed for Pb2+ concentration by atomic adsorption spectrophotometry. The amount of adsorption for Pb2+ onto the adsorbent was calculated by using equation (1) as follows (Hayeeye et al., 2014)
All experiments were carried out in triplicates and mean values including standard deviations were reported.
Batch equilibrium experiments
Batch equilibrium experiments were carried out to obtain the equilibrium isotherms and study the effects of contact time (5 min–24 h), initial concentration of Pb2+ ions (50, 300, and 500 mg l−1), dosage of GE/AC (0.03–0.21 g), and solution pH (Jyotsna et al., 2005). Amounts of 0.15 g of GE/AC were added into 50 ml Pb2+ ions solutions and the mixtures were placed in a rotary shaker for 24 h. Then the supernatant solutions were analyzed for Pb2+ ions concentrations. All the experiments were carried out in triplicates and mean values were reported. For studying the effect of pH, both the initial pH and the final pH of the solutions were adjusted to a value between 1.5 and 5.5 by using 0.1 M HCl and NaOH solutions. Pb2+ ions concentrations were analyzed by atomic adsorption spectrophotometry. The amount of adsorbed Pb2+ ions was calculated by using equation (1).
Mathematical models for data analysis
The adsorption kinetics of Pb2+ ions was analyzed by the use of pseudo-first-order and pseudo-second-order kinetic models. The pseudo-first-order kinetic model can be written as shown in equation (2) (Lagergren, 1898)
The pseudo second-order kinetic model can be written as in equation (4) (Ho and McKay, 1999; Umar et al., 2015)
For the evaluation of the equilibrium data, the commonly used Langmuir and Freundlich isotherm equations were used. The Langmuir equation (Langmuir, 1916, 1918) is often used to describe the adsorption on homogeneous surfaces based on the assumptions of monolayer adsorption. The Langmuir equation and its linearized form can be expressed as equations (6) and (7)
The Freundlich isotherm (Freundlich, 1907) is used for nonideal adsorption, which involves systems with heterogeneous surface energy. It assumes that the adsorption occurs at sites of different energy. The Freundlich equation and its linearized form are given in equations (8) and (9) as follows
Results and discussion
Characterization of GE/AC
By BET surface area measurement, the narrow pore size distribution and its average pore diameter of 3.25 nm were found for GE/AC bead. This result indicated that this bead adsorbed was in mesoporous scale (2–50 nm). The specific surface area for GE/AC is 34.75 m2 g−1. The point of zero charge of GE/AC is 4.5 indicated that the surface nature of GE/AC is acidic.
The SEM photographs in Figure 2(a) and (b) were taken at 20 × and 200 × magnification to observe the shape and surface morphology, respectively. SEM image presented the spherical bead with diameter about 4 mm (Figure 2(a)). At higher magnification, the rough surface showing many pores was also observed.
The SEM photographs of (a) bead size and (b) external surface of GE/AC. GE/AC: gelatin/activated carbon; SEM: scanning electron microscopy.
The elemental components for GE/AC before and after adsorption of Pb2+ ions were detected by SEM/EDX using a standardless qualitative EDX analytical technique. The peak heights in the EDX spectra are proportional to the elemental concentrations. The qualitative spectra for GE/AC before and after Pb2+ ions adsorbed are shown in Figure 3(a) and (b), respectively, and indicated that C, N, and O are the main constituents. In addition, the EDX spectrum of GEAC after Pb2+ ions adsorbed was illustrated in Figure 3(b). The presence of Pb2+ ion in the spectrum revealed that Pb2+ ions were adsorbed on GE/AC.
EDX spectrums of GE/AC (a) before and (b) after adsorption of Pb2+ ions. EDX: energy dispersive X-ray spectroscopy; GE/AC: gelatin/activated carbon.
Figure 4 shows EDX dot mapping of GE/AC (a) before and (b) after Pb2+ ions adsorbed. It can provide indication of the elemental distribution mapping. The blue bright points represented the Pb2+ ions adsorbed on GE/AC which are clearly shown in Figure 4(b). The high density of blue dot distributed on the surface of GE/AC indicated the abundant Pb2+ ions. These results confirmed that Pb2+ ions of 7.7 wt% adsorbed on GE/AC surface.
EDX dot mapping of GE/AC (a) before and (b) after adsorption of Pb2+ ions. EDX: energy dispersive X-ray spectroscopy; GE/AC: gelatin/activated carbon.
Kinetics of adsorption
The adsorption kinetics were studied at three different initial concentrations of 50, 300, and 500 mg l−1 and the respective kinetic profiles obtained are shown in Figure 5. The results indicated that the rate of Pb2+ ions uptake was rather fast with 90% of the total uptake occurred in about 5 min. However, after that, there was a slower process of uptake to about 1 h, after which no significant further adsorption was observed. Thus, the equilibrium time for Pb2+ ions adsorption was determined to be around 60 min at 23 ± 2℃, pH 3–5, and dosage of GE/AC of 0.15 g. Moreover, the results show that different initial concentrations were not effect to equilibrium time.
Kinetic profiles of Pb2+ ions adsorption onto GE/AC at different initial Pb2+ concentrations ((a) 50 mg l−1, (b) 300 mg l−1, and (c) 500 mg l−1). GE/AC dose 0.15 g/50 ml, temperature 23 ± 2 ℃ and pH = 5. GE/AC: gelatin/activated carbon.
The evaluations of the kinetic profiles with the use of linearized forms of the pseudo kinetic models are shown in Figure 6 and the associated parameter values listed in Table 1. The values of kinetic parameter k1 and k2 were obtained from the slopes of the linear plots and those for q
e
from the intercepts.
Linearized plots of the pseudo-first-order model (a) and pseudo-second order model (b) for Pb2+ ions adsorption onto GE/AC. GE/AC dose 0.15 g/50 ml, temperature 23 ± 2 ℃, and pH = 5. GE/AC: gelatin/activated carbon. Kinetic parameters of pseudo-first-order and pseudo-second-order model at different initial concentrations.

The plots in Figure 6 show that both the first- and the second-order pseudo kinetic models can fit the concentration profiles, but a better fit with a higher correlation coefficient (R2 > 0.99). The q e values obtained from the model were also in close agreement with those obtained experimentally. The pseudo-second-order rate constants were in the range of 4.19 × 10−3 to 36.85 × 10−3 g mg−1 min−1.
Effect of pH on Pb2+ ions uptake by GE/AC adsorbent
Solution pH affects the ionic states of functional groups on the adsorbent surface, such it is expected the adsorption is solution pH dependent. This effect was studied by evaluating the adsorption capacities at different solution pH in the range of 1.5–5 and the results are shown in Figure 7. Experiments beyond an initial pH value 5.5 were not conducted to avoid Pb2+ ions precipitation. It can be seen from Figure 4 that the adsorption capacity (q
e
) of the GE/AC adsorbent increased with the pH of solution. The sharpest increase in q
e
was observed between pH 2 and 3, while around pH 4 a plateau was reached. These results showed the suitability of the GE/AC adsorbent for the treatment of acidic wastewater. At a low pH range, the concentration of H+ is high which promotes the protonation of the functional groups of the adsorbent, resulting it to be more positively charged, which in turn leads to the electrostatic repulsion of the positive charged Pb2+ ions. Moreover, at low pH values, the H+ ions compete with Pb2+ ions for the adsorption sites (Kobya et al., 2005).
Effect of pH on GE/AC for Pb2+ ions adsorption. GE/AC dose 0.15 g/50 ml, temperature 23 ± 2 ℃, Pb2+ concentration of (a) 50 mg l−1 and (b) 200 mg l−1. GE/AC: gelatin/activated carbon.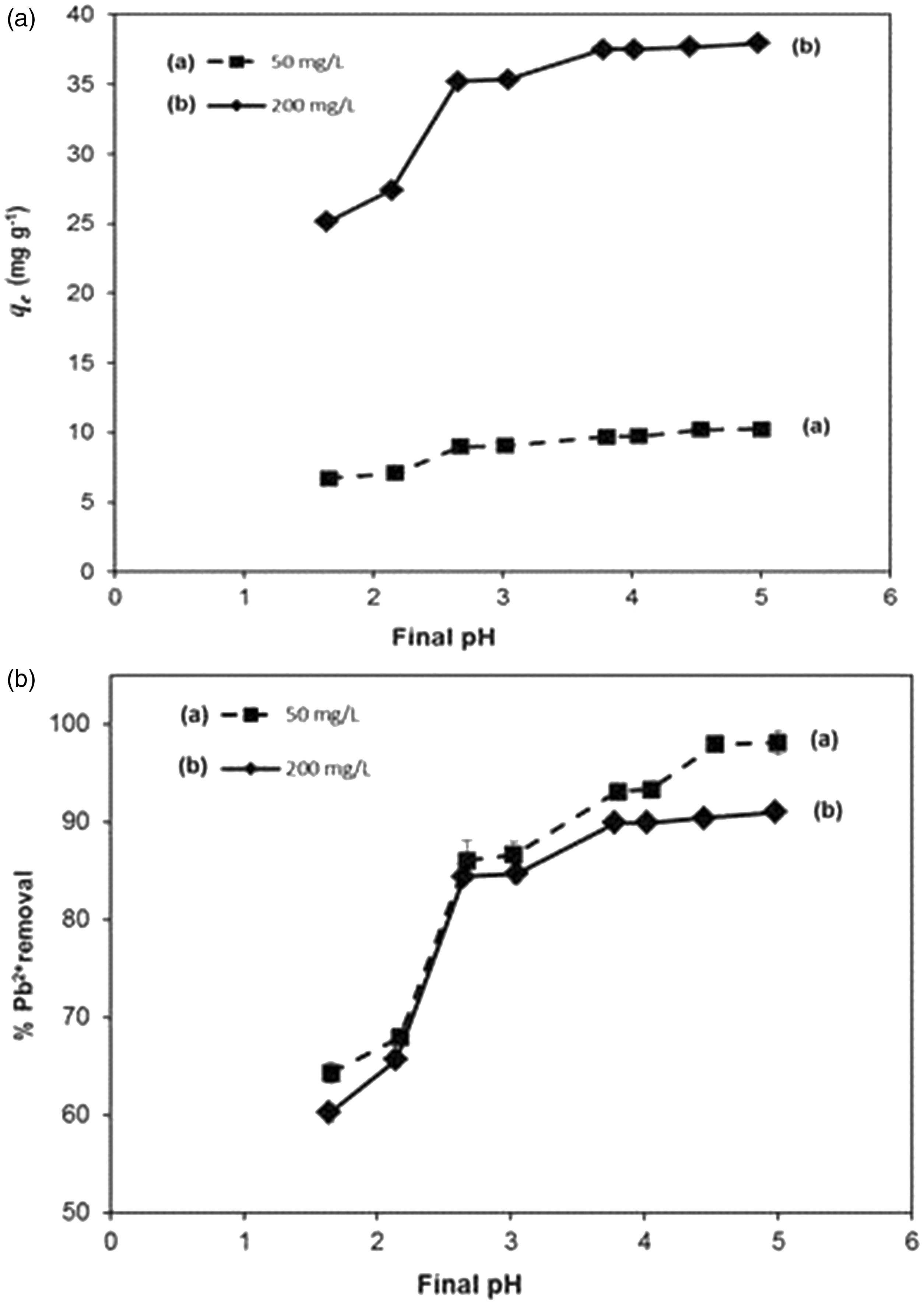
Adsorption equilibrium isotherms
The adsorption isotherms for GE/AC adsorbent for Pb2+ ions presented in the aqueous phase at the solution pH range of 3–5 are shown in Figure 8. The adsorption isotherm has a typical shape of the L-2 type isotherms (Limousin et al., 2005). This indicated that there was a reduction in the number of active sites on the adsorbent surface at a high Pb2+ concentration in the solution phase.
Adsorption equilibrium isotherms of Pb2+ ions onto GE/AC, GE/AC dose 0.15 g/50 ml, temperature 23 ± 2 ℃, pH (3, 4, and 5), and equilibrium time 24 h. GE/AC: gelatin/activated carbon.
The equilibrium isotherms in Figure 8 were fitted with the linearized forms of the both the Langmuir and the Freundlich equations as shown in Figure 9. The isotherm equation parameters are given in Table 2.
(a) Langmuir and (b) Freundlich adsorption isotherms for Pb2+ ions adsorption onto GE/AC. pH (3, 4, and 5), equilibrium time 24 h, temperature 23 ± 2 ℃, GE/AC dose 0.15 g. GE/AC: gelatin/activated carbon. Parameter values of Langmuir and Freundlich equations of Pb2+ ions sorption on GE/AC at different solution pH at 23 ± 2 ℃. GE/AC: gelatin/activated carbon. Comparison of the adsorption capacities (qm, mg g−1) of Pb2+ ions by powder, modified, and granular activated carbon reported in prior literature. GE/AC: gelatin/activated carbon. t
eq
is time at equilibrium.

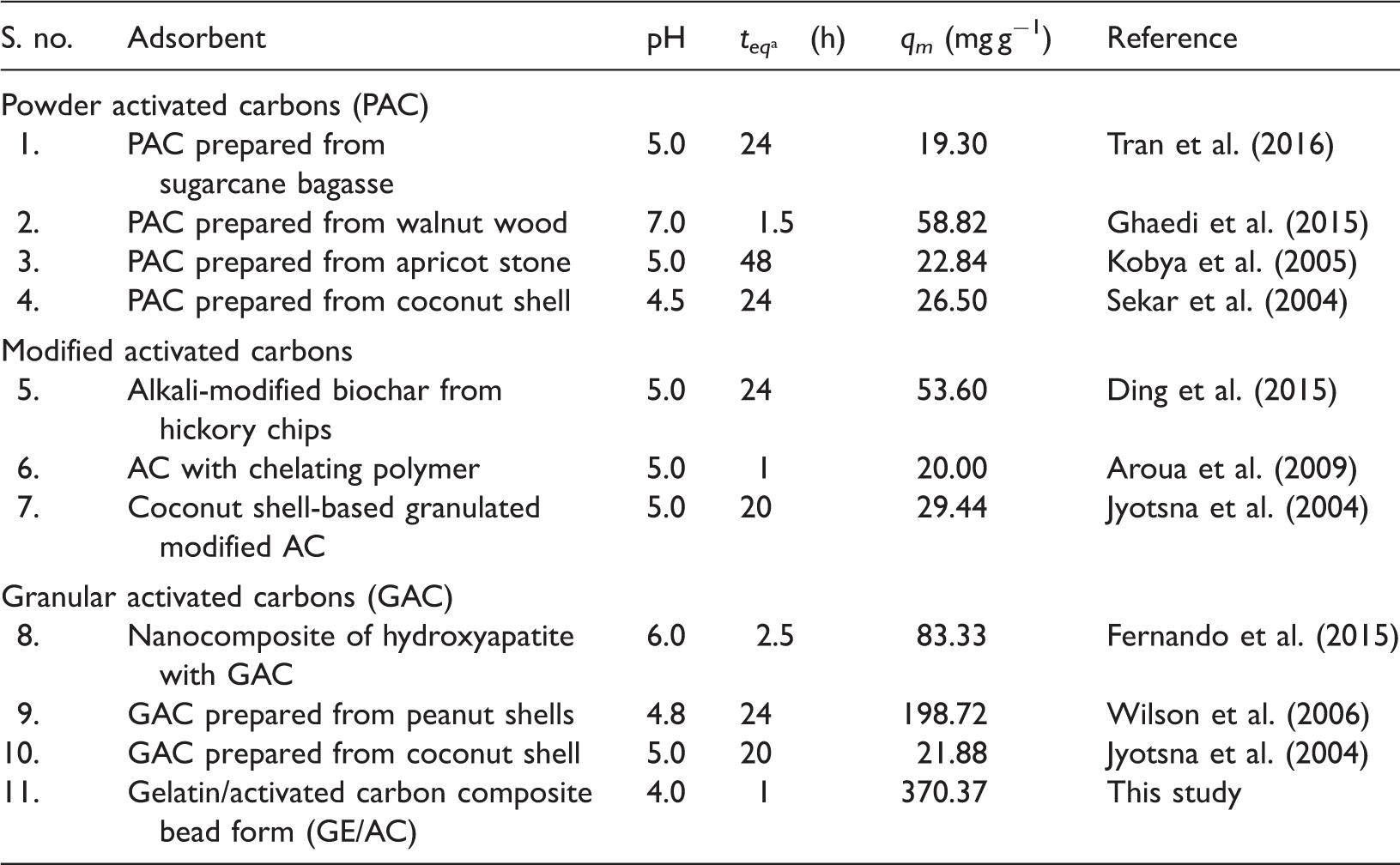
Figure 9 shows that the equilibrium isotherms are well correlated with the Langmuir model, with the R2 values > 0.99 observed. On the other hand, the correlation with the Freundlich model was less satisfactory, particularly in the higher concentration ranges. The highest maximum adsorption capacity (q m ) of GE/AC for Pb2+ ions determined from the Langmuir model as 370 mg g−1, which was higher than those of some granular AC. Table 3 show a comparison of the maximum adsorption capacities of Pb2+ ions (q m , mg g-1) by GE/AC to those of other adsorbents reported in the literature. The GE/AC bead was employed to increase a particular adsorption capacity of granular activated carbon. Moreover, the GE/AC bead adsorbent is easier to use for adsorption than activated carbon powder.
Effect of GE/AC dosage on Pb2+ ions removal
Experiments with the use of different dosages of GE/AC from 0.03 to 0.21 g per 50 ml of solutions were carried out to evaluate the effect of dose on Pb2+ ions removal at the initial concentrations of 100, 200, and 300 mg l−1. The results are shown in Figure 10. An increase in adsorbent dosage increased percentage of Pb2+ ions adsorbed at equilibrium due to the surface area and availability of adsorption sites (Namasivayam and Kadirvelu, 1999), thus increased the removal efficiency. On the other hand, the adsorption capacity decreased with the increasing of GE/AC. This result may be because after a certain dose of GE/AC, the maximum adsorption is reached due to both of amount of Pb2+ ions bound to the adsorbent and the amount of free ions remaining constant even with further addition of the dose of adsorbent (Charles and Odoemelam, 2010). A higher initial concentration of Pb2+ resulted in lower removal efficiency, as the number of active sites of GE/AC adsorbent is limited (Jamal et al., 2013).
Effect of dosage on GE/AC for Pb2+ ions adsorption. GE/AC dose 0.03–0.21 g/50 ml, temperature 23 ± 2 ℃, and pH = 5 and initial Pb2+ concentration of 100, 200, and 300 mg l−1. GE/AC: gelatin/activated carbon.
Conclusions
Adsorption results obtained in this study indicated that the adsorbent obtained from the combination of GE and powdered AC in bead form could be used as practical adsorbent for the recovery of Pb2+ ions from wastewater. The GE/AC was an efficient and eco-friendly adsorbent. Batch kinetic studies showed that the rate of lead ion uptake by GE/AC adsorbent was high even at low initial concentrations and the concentration profiles followed the pseudo-second-order kinetic model. The adsorption capacity was found to be solution pH dependent with a higher capacity obtained at the higher pH ranges. Batch equilibrium studies showed that the equilibrium isotherms were well described by the Langmuir isotherm model and the highest maximum adsorption capacity was found to be 370 mg g−1 at pH 5. The highest adsorption efficiency observed was about 90%. Therefore, the GE/AC appears to be well suited for removal of Pb2+ ions from wastewater streams.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the Strategic Scholarships Fellowships Frontier Research Networks (Specific for Southern Region), Griffith School of Engineering, Nathan Campus, Griffith University, Brisbane, Queensland, Australia, Graduate School, Prince of Songkla University, Department of Chemistry, Faculty of Science, Prince of Songkla University, Hat Yai campus and Department of Science, Faculty of Science and Technology, Prince of Songkla University, Pattani campus, in Thailand for financial support.









