Abstract
In this study, the adsorption capacities of peanut shell activated carbon samples prepared using three types of peanut shell as raw material were compared. The effects of activation state, carbonization temperature, carbonization time, adsorption time during decolorization, and dosage on the performance of the peanut shell activated carbon samples were investigated. The performance of the modified peanut shell (activated carbon) on the decolorization of reactive brilliant blue X-BR and the adsorption kinetics were evaluated systematically. Among the three types of peanut shell activated carbon, the activated carbon that was first activated by phosphoric acid and then carbonized at 450°C for 3 h displayed the best performance, with an optimum dosage of 4 g l−1 and an optimum adsorption time of 2 h. The pseudo-second-order kinetics equation and the intraparticle diffusion equation could well describe the adsorption behavior of the activated carbon prepared by phosphoric acid activation. Intraparticle diffusion was not the only factor affecting the adsorption rate of the activated carbon on reactive brilliant blue X-BR.
Introduction
Wastewater discharge is an important source of water pollution in China, where the textile dyeing and printing industry is large. Some of these pollutants even cause mutagenic effects. Azo dyes, anthraquinone dyes, and dyes of three phenyl methane are frequently used in the printing and dyeing industry. The advanced treatment technology of textile printing and dyeing wastewater involves several processes, including flocculation, adsorption, chlorine dioxide oxidation, Fenton oxidation, electrochemical oxidation, and membrane separation (Gulkaya et al., 2006; Raghu and Ahmed Basha, 2007; Wijetunga et al., 2010; Xu and Song, 2015). Adsorption is a highly efficient, renewable, and environment-friendly method, and its efficiency is mainly determined by the high specific surface area and the physical and chemical adsorption capacity of internal molecules (Ghaedi et al., 2012; Okoniewska et al., 2008; Wang et al., 2010). After adsorption, activated carbon can still be reused because it still possesses an adsorption capacity that confers regeneration benefits (Dong et al., 2014). Activated carbon is a high-quality and efficient adsorbent that has been widely used in many fields, such as environmental protection, chemical industry, and food industry.
All types of biological wastes, such as palm shell (Lim et al., 2015), peanut shell (Malik et al., 2007), coconut shell (Couto et al., 2015), castor shell (Thamilarasu and Karunakaran, 2013) and coffee residue (Lamine et al., 2014), can be used as low-cost precursors for the preparation of activated carbon. China is the world’s largest peanut producer. Peanut shell resources, which are abundant but have a low utilization rate, are only often used as animal feed. The phenolic hydroxyl groups and carboxyl groups contained in the peanut shell exhibit strong adsorption effects after chemical modification, making the performance of the activated carbon ideal. Thus, peanut shell can be used to prepare activated carbon for wastewater treatment. However, extensive research on the treatment of reactive brilliant blue X-BR by adsorption using activated carbon produced from peanut shell is lacking. Thus, the present work aimed to fill this research gap. The pore structure of activated carbon and the type and quantity of surface functional groups hardly meet the requirements for adsorbing various pollutants in wastewater. Therefore, modification of activated carbon is necessary (Bhatnagar et al., 2013). The physical and chemical properties, pore structure, surface acidity, or types and quantities of functional groups in the surface structure of activated carbon need to be modified using physical and chemical methods to improve the selectivity of activated carbon during adsorption, expand the scope of its application, and meet the special needs in engineering (Qiao et al., 2005). Modification by phosphoric acid can increase the polar functional groups on the surface of activated carbon, as well as change the chemical properties and enhance the hydrophilicity of its surface. This modification improves the adsorption capacity of polar organic matter and phosphate, thereby allowing the simultaneous removal of organic matter and phosphorus in water, given that dyes cannot be completely removed by physical adsorption alone. It can be used to evaluate the adsorption effect of the modified activated carbon (Zhou et al., 2015).
This research compared the efficiency of peanut shell activated carbon (PSAC) samples prepared using peanut shell as raw material in the removal of reactive brilliant blue X-BR from aqueous solutions. The influence of different carbonization temperatures, carbonization times, adsorption times, and dosages on the adsorption properties of PSAC was investigated. The modified activated carbon was characterized by high-resolution scanning electron microscopy (SEM) and adsorption curve analysis of reactive brilliant blue X-BR. The parameters in the application of activated carbon in the treatment of textile printing and dyeing wastewater were also optimized.
Materials and methods
Main materials and reagents
Peanut shells were obtained from a local market in Nanjing City, China, and prepared for use. The shells were dried in the oven after washing with distilled water and then cooled before crushing and sieving in a 20-mesh screen for further use. This material was used as a precursor to prepare activated carbon.
Reactive brilliant blue X-BR and phosphoric acid (purchased from Nanjing Chemical Reagent Co., Ltd, China; 99.7% of purity) were used as the reagents in this study.
Experimental scheme and content
Preparation of simulated wastewater
Reactive brilliant blue X-BR is a type of anthraquinone dye that is widely used as a low-temperature reactive dye with excellent light fastness and weather fastness. Reactive brilliant blue X-BR is mainly used in cotton, silk, and other fibers to dye bright-colored products. A certain amount of reactive brilliant blue X-BR was placed in the oven at a constant temperature and then dried. Then, 0.1 g of the dye was dissolved in 1 l of distilled water to prepare simulated dye solution with a concentration of 1 g l−1. This solution was sealed in the reagent bottle and stored at room temperature
Preparation of PSAC
Three types of PSAC samples prepared using different methods and under different temperatures were compared to determine the optimal activation method and temperature.
First category (only carbonized): A certain amount of peanut shell powder was placed in a crucible in the muffle furnace, carbonized at 400 and 500°C for 3 h, cooled, and then stored in a sealed bag. PSAC samples carbonized at 400 and 500°C were denoted as C-PSAC-400 and C-PSAC-500, respectively.
Second category (acidification after carbonization): Certain amounts of C-PSAC-400 and C-PSAC-500 were partially weighed and mixed evenly at the ratio of 1 g of the activated carbon and 3 ml of phosphoric acid. The precursors were placed in a water bath at 80°C for 3 h, washed with 10% hydrochloric acid solution and deionized water to modify the pH between 6 and 7 after leaving at room temperature for 24 h, and then dried in a blast drying oven at 100–110°C. The samples were cooled for further use. The PSAC samples that acidified after carbonization at 400 and 500°C were denoted as CA-PSAC-400 and CA-PSAC-500, respectively.
Third category (carbonization after acidification): A certain amount of peanut shell powder was weighed and mixed evenly at the ratio of 1 g of the activated carbon and 3 ml of phosphoric acid (mass fraction was 50%) and then placed at room temperature for 24 h. The precursors were placed in a crucible in the muffle furnace; carbonized at 300, 350, 400, 450, 500, 550, 600, and 700°C for 3 h; washed with deionized water to modify the pH between 6 and 7 after cooling and crushing with a high-speed pulverizer; and then dried in a blast drying oven at 100–110°C to obtain the PSAC. The PSAC samples carbonized at 300, 350, 400, 450, 500, 550, 600, and 700°C after acidification were denoted as AC-PSAC-300, AC-PSAC-350, AC-PSAC-400, AC-PSAC-450, AC-PSAC-500, AC-PSAC-550, AC-PSAC-600, and AC-PSAC-700, respectively.
Adsorption kinetics experiment
A 1 l aliquot of the reactive brilliant blue X-BR solution with the concentration of 0.1 g l−1 was mixed with different types and dosages of the different PSAC samples. The mixed solution was shaken in a thermostatic oscillator for a certain time for adsorption and filtered by funnels. The supernatant was collected, and its absorbance was measured by a spectrophotometer (Spectrum lab 752 s UV VIS, Shanghai Lengguang Technology Co., Ltd, China).
Analysis method
Reactive brilliant blue X-BR solution was scanned in the wavelength range of 400–700 nm by a spectrophotometer (Spectrum lab 752 s UV VIS, Shanghai Lengguang Technology Co., Ltd, China). The maximum absorption wavelength of the reactive brilliant blue X-BR was determined to be 601 nm.
Results and discussion
Surface characteristics of activated carbon
Figure 1 shows the SEM images of the sample of the peanut shell (Figure 1(a)), the PSAC (Figure 1(b)), and the PSAC that was activated by phosphoric acid (Figure 1(c)). These images were used to verify the possible changes in morphological features of the samples before and after the activation process. Figure 1(a) shows that the surface of the untreated peanut shell was irregular and nonporous with a dense fibrous structure. After carbonization (Figure 1(b)), the surface became relatively rough, of which the original fibrous structure was interrupted to form a certain pore structure. After modification with phosphoric acid (Figure 1(c)), the surface of PSAC presented particles with an uneven size distribution and complex pore structure. As listed in Table 1, the specific surface area and micropore volume of AC-PSAC-450 were 1138 m2 g−1 and 0.4061 cm3 g−1, respectively, which were almost two times higher than those of C-PSAC-500. These results indicate that the PSAC that was activated by phosphoric acid can improve the physical characteristics of peanut shell. These characteristics can improve the accessibility of active sites and the diffusion of large dye molecules during adsorption. Zhang et al. (2018) applied activated carbon from bamboo as an electrode material for supercapacitors and found that activation treatment can dramatically increase the specific surface area and total pore volume and decrease the average pore size of this material. This finding is similar to our results that high surface area and micropore volume provided more active sites.

SEM images of peanut shell (a), PSAC (b), PSAC that is activated by phosphoric acid (c).
Characterization results of the activated carbons.
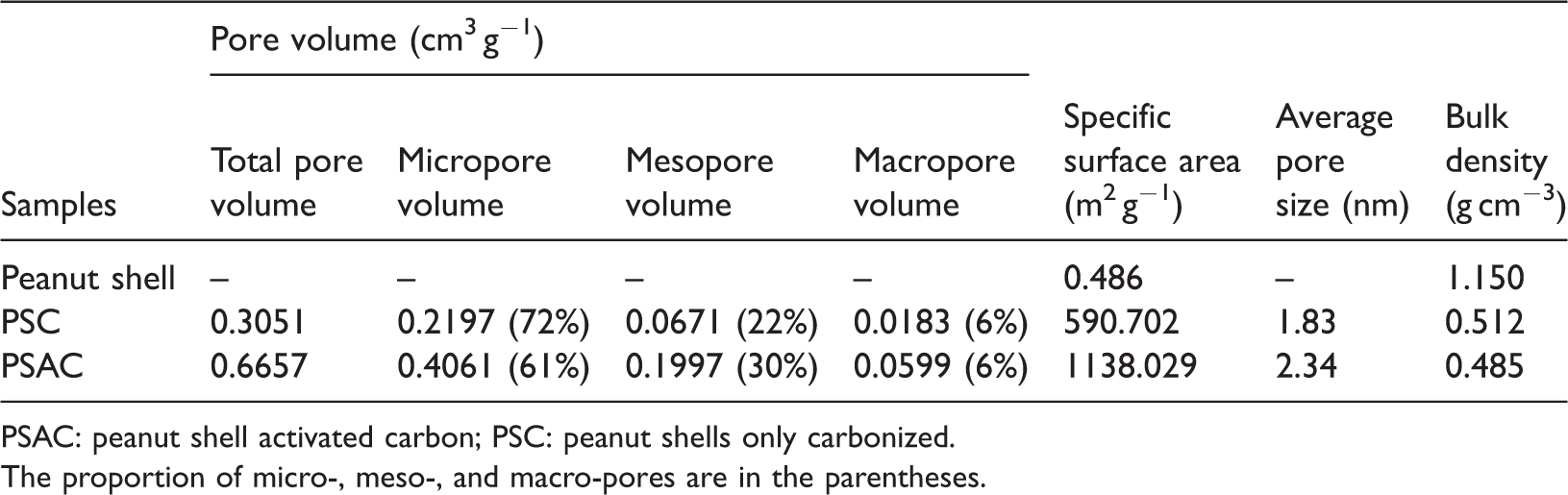
PSAC: peanut shell activated carbon; PSC: peanut shells only carbonized.
The proportion of micro-, meso-, and macro-pores are in the parentheses.
Adsorption experiment
Effect of preparation methods on the decolorization ability of activated carbon
Peanut shells carbonized at 400 and 500°C were used to prepare PSAC by using the three preparation methods mentioned above. At 25°C, different dosages of these three types of PSAC were added to 1 l of reactive brilliant blue X-BR solution with the concentration of 0.1 g l−1 and shaken in the thermostatic oscillator at the speed of 100 r min−1 for 2 h. Figure 2 shows the effect of activation states and dosages on the removal rate of active brilliant blue X-BR.

Effects of preparation methods and dosages on the adsorption ability of the activated carbon to the reactive brilliant blue X-BR.
As shown in Figure 2, the PSAC samples prepared using the three methods differed in decolorization ability. The adsorption performance of the PSAC prepared by carbonization after acidification was much better than those of the PSAC prepared by the two other methods. Under the condition of carbonization at 400°C, the removal rate of the active brilliant blue X-BR in 1 l of the solution could reach 55.4% by adding 1 g of AC-PSAC-400. When the dosage increased to 6 g l−1, the removal rate reached 93.95%, which was much higher than that of the other two types of the activated carbon at the same dosage. This result can be attributed to the phosphate activator, which can rapidly penetrate the cellulose molecules of the peanut shell, expand the pore structure, and promote the formation of carbon structure to enhance the adsorption properties of the PSAC. Therefore, the activated carbon obtained by carbonization after acidification exerted the best effects on the removal of reactive brilliant blue X-BR.
The removal rate of active brilliant blue X-BR in the solution also gradually increased with increasing dosage. Many micropores were present on the surface of the activated carbon. Thus, the adsorption point on the surface also increased with increasing dosage, which consequently improved the adsorption efficiency of the dye solution. For the activated carbon prepared by carbonization after acidification, the trend of the removal rate curve became stable when the dosage was greater than 4 g l−1. The chroma in the solution was removed by the activated carbon, and the effect of increasing dosage on the removal rate of active brilliant blue X-BR was negligible.
Effect of carbonization temperature on the decolorization ability of activated carbon
Peanut shells were acidified and then carbonized at different temperatures (300, 350, 400, 450, 500, 550, 600, and 700°C) to prepare PSAC. At 25°C, different dosages of PSAC were added to 1 l of the active brilliant blue X-BR solution with the concentration of 0.1 g l−1 and then shaken in the thermostatic oscillator at the speed of 100 r min−1 for 2 h. Figure 3 shows the effects of carbonization temperature on the removal rate of active brilliant blue X-BR.

Effect of carbonization temperature on removal rate of the reactive brilliant blue X-BR solution.
The peanut shells were not completely carbonized at very low carbonization temperatures because this condition was not conducive for the formation of the pore structure of the activated carbon and greatly reduced the adsorption effect. Results showed that the adsorption effect of the activated carbon and the removal rate of reactive brilliant blue X-BR solution increased gradually as the carbonization temperature was increased from 300°C. The maximum dye removal efficiency of the activated carbon was obtained at 450°C, and the removal rate was close to 100% when 14 g of AC-PSAC-450 was added to 1 l of the reactive brilliant blue X-BR solution. As a result, high dosages of AC-PSAC-450 would lead to low economic benefits to its application in practical engineering.
However, the adsorption effect of activated carbon slightly declined with increasing carbonization temperature. As shown in Figure 3, the active brilliant blue X-BR removal rate of AC-PSAC-300 was 61.40% when the dosage was 2 g l−1. The active brilliant blue X-BR removal rate of AC-PSAC-450 was up to 71% when the dosage was 1 g l−1, and the removal rate was raised to 93.64% when the dosage was 2 g l−1. However, as the temperature was increased to 600 and 700°C, the removal rate decreased to 53.40 and 56.85%, respectively. This result can be attributed to the activator phosphoric acid, which greatly reduced the required temperature for carbonization (Kwiatkowski et al., 2017) and thus facilitated the preparation of activated carbon with a developed pore structure at a lower temperature (Gao et al., 2016). During carbonization, the micropore structure of the materials was formed at 150°C. The mesoporous structure was mainly formed at 200–450°C and still formed at temperatures higher than 450°C (Demiral and Güngör, 2016; Üner and Bayrak, 2018). However, when the peanut shell was carbonized at extremely high temperatures, its cellulose became carbonized and nodular, which was not conducive for the formation of the pore structure of the activated carbon (Fu et al., 2017; Pallarés et al., 2018).
As illustrated in Figure 3, the best preparation temperature of the activated carbon was 450°C. The adsorption efficiency was stable when the dosage was more than 4 g in every 1 l of dye solution. Therefore, the optimum dosage of AC-PSAC-450 was 4 g l−1.
Effect of carbonization time on the decolorization ability of AC-PSAC-450
AC-PSAC-450 was carbonized for different times (2, 3, 4, 5, 6, and 7 h) to prepare PSAC. At 25°C, different dosages of PSAC were, respectively, added to 1 l of the active brilliant blue X-BR solution with the concentration of 0.1 g l−1 and shaken in the thermostatic oscillator at the speed of 100 r·min−1 for 2 h. Peanut shells that were acidified and then carbonized at 450°C for 2, 3, 4, 5, 6, and 7 h were denoted as AC-PSAC-450-2, AC-PSAC-450-3, AC-PSAC-450-4, AC-PSAC-450-5, AC-PSAC-450-6, and AC-PSAC-450-7, respectively. Figure 4 shows the effect of carbonization time on the active brilliant blue X-BR removal rate.

Effect of carbonization time on the removal rate of the reactive brilliant blue X-BR solution.
As shown in Figure 4, the removal rate of the reactive brilliant blue X-BR solution increased substantially when the carbonization time was extended from 2 to 3 h. However, the removal rate decreased when the carbonization time exceeded 3 h. The results showed that the pore size changed with prolonged carbonization time. Thus, the activated carbon with changed pore size was no longer suitable to adsorb materials with the same size of X-BR, leading to the decrease in the removal rate. Figure 4 shows that the removal rate of AC-PSAC-450-3 reached the maximum regardless of the change in dosage, indicating that most of the carbohydrates and noncarbon components in the peanut shell were removed and a developed microporous structure for efficient adsorption was formed (Dizbay-Onat et al., 2017; You et al., 2018). Thus, the optimum carbonization time was 3 h.
Among the PSAC samples, AC-PSAC-450-3 showed the best performance, with a reactive brilliant blue X-BR removal rate of 98.74% at the dosage of 4 g l−1. Su et al. (2008) quantum-sized ZnO to degrade reactive brilliant blue X-BR with a removal rate exceeding 90%. Chu et al. (2014) prepared activated carbon from Thalia dealbata and obtained a removal efficiency of 90% at an initial X-BR concentration of 100 mg l−1. Compared with the adsorbents mentioned above, AC-PSAC-450-3 can achieve efficient removal of the reactive brilliant blue solution.
Adsorption kinetics
Analysis of adsorption kinetics curve
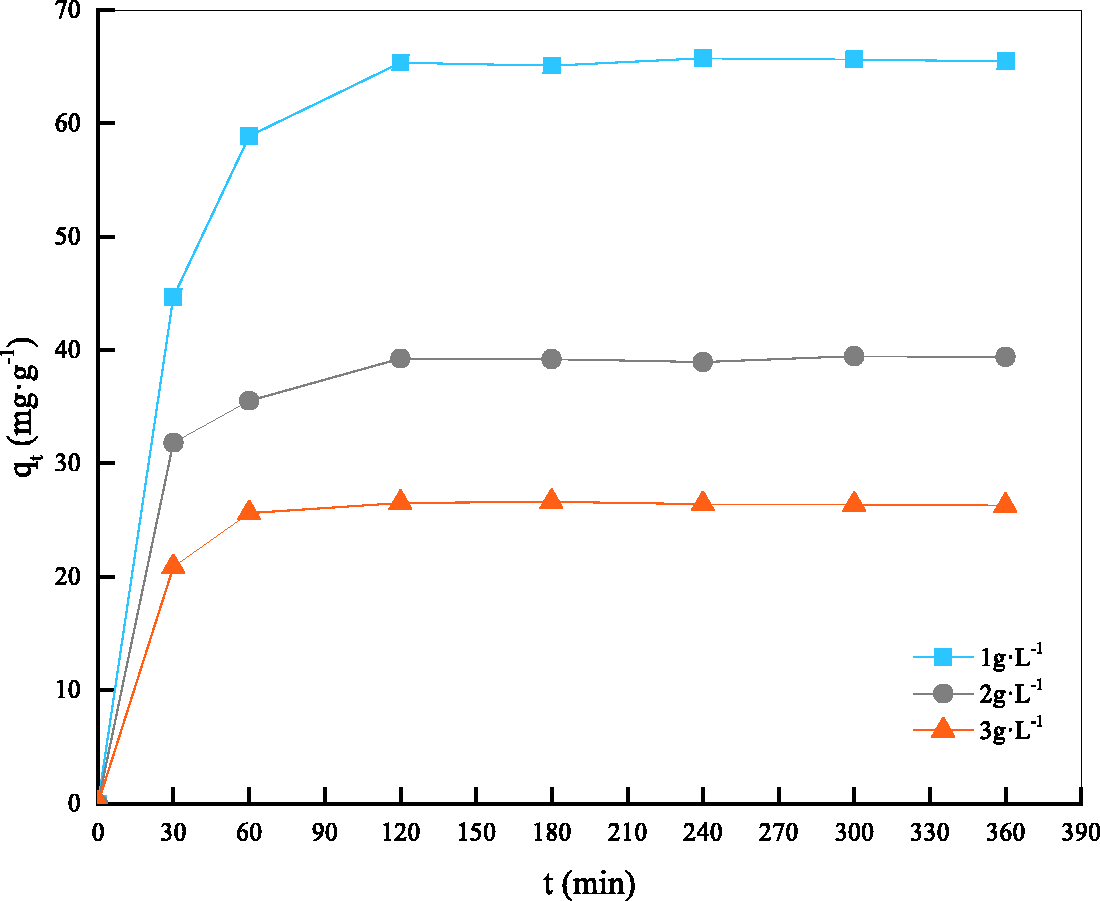
Figures 5 and 6 show the changes in the adsorption amount of reactive brilliant blue X-BR on AC-PSAC-450-3 over time (at 25°C and with pH 7 ± 0.1). The adsorption kinetics of AC-PSAC-450-3 to treat the reactive brilliant blue X-BR solution with different initial concentrations (35, 60, 85, and 100 mg l−1) are presented in Figure 5, and the adsorption kinetics of the PSAC with different dosages (1, 2, and 3 g l−1) are presented in Figure 6.

qt–t curves of AC-PSAC-450-3 adsorption on the reactive brilliant blue X-BR with different initial concentrations.

qt–t curves of AC-PSAC-450-3 adsorption on the reactive brilliant blue X-BR with different dosages.
As shown in Figure 5, the changes in the adsorption kinetics curves of AC-PSAC-450-3 with different initial concentrations (35, 60, 85, and 100 mg l−1) were the same at the same temperature. At any time, the adsorption amount of reactive brilliant blue X-BR increased as the initial concentration of the solution was increased, and the highest adsorption capacity was reached when the initial concentration was 100 mg l−1. This result can be ascribed to the fact that the high initial concentration of reactive brilliant blue X-BR can produce a large mass transfer force, which can allow the maximum application of the adsorption point on the inner and outer surfaces of the activated carbon (Magdy and Altaher, 2018; Miyah et al., 2018; Zhang et al., 2018). In addition, two adsorption stages were observed. The adsorption capacity increased with the adsorption time at the initial stage, and the adsorption reached equilibrium after a certain time. The adsorption capacity of AC-PSAC-450-3 increased rapidly within 0–1 h, which indicated that the adsorption reaction was strong in this period. With prolonged adsorption time, the adsorption state became stable. The adsorption kinetics curve was approximately a horizontal line after 2 h, indicating that the adsorption of AC-PSAC-450-3 on reactive brilliant blue X-BR reached equilibrium at about 2 h. Magdy and Altaher observed the same stages in the adsorption of acid red onto cement kiln dust. They observed a large number of vacant adsorption sites at the initial stage and that a high concentration of the adsorbate provides a high driving force for adsorption. The number of available sites decreased with time and the adsorbate molecules started competing for the remaining sites, creating repulsive forces during the later stage. This process was slow.
Figure 6 describes the adsorption curves of AC-PSAC-450-3 with different dosages (1, 2, and 3 g l−1). Comparison of Figures 5 and 6 showed that the curves of the two figures were the same, but the adsorption capacity decreased as the dosage of AC-PSAC-450-3 was increased. The addition of AC-PSAC-450-3 was equivalent to the decrease in the equilibrium concentration, and the equilibrium adsorption capacity decreased gradually.
Analysis of adsorption kinetics model
The adsorption of reactive brilliant blue X-BR versus time data was tested against various available kinetic models. The aim of the kinetic studies was to explore the possible mechanism of sorption of reactive brilliant blue X-BR onto AC-PSAC-450-3. Well-established kinetic models, such as pseudo-first-order, modified pseudo-first-order, pseudo-second-order models, and intraparticle diffusion model, were tested in this study. The conformity between the predicted model values and the experimental data was expressed by the regression coefficient (R2). The model with the highest regression coefficient with the available kinetic experimental data was deemed applicable.
The pseudo-first-order kinetic model is generally expressed by the following equation (1)

The integrated form of the above equation is expressed as the following equation (2)

The pseudo-second-order kinetic model is expressed by the following equation (3)

The modified pseudo-first-order model is expressed by the following equation (4)

The intraparticle diffusion model is first proposed by Weber–Morris and the formula is expressed as the following equation (5)

The experimental data of AC-PSAC-450-3 adsorption at different initial concentrations in Figure 5 were subjected to the least squares method to fit the data and obtain the fitting curves of the four kinetic equations. The fitting results are shown in Figure 7, and the related kinetic parameters are shown in Table 2.
Four types of kinetic equation fitting curves: (a) pseudo-first-order kinetic equation fitting curves of AC-PSAC-450-3 adsorption on the reactive brilliant blue X-BR, (b) modified pseudo-first-order kinetic equation fitting curves of AC-PSAC-450-3 adsorption on the reactive brilliant blue X-BR, (c) pseudo-second-order kinetic equation fitting curves of AC-PSAC-450-3 adsorption on the reactive brilliant blue X-BR, and (d) intraparticle diffusion equation fitting curves of AC-PSAC-450-3 adsorption on the reactive brilliant blue X-BR.
Kinetic parameters of the adsorption of the reactive brilliant blue X-BR with different initial concentrations.
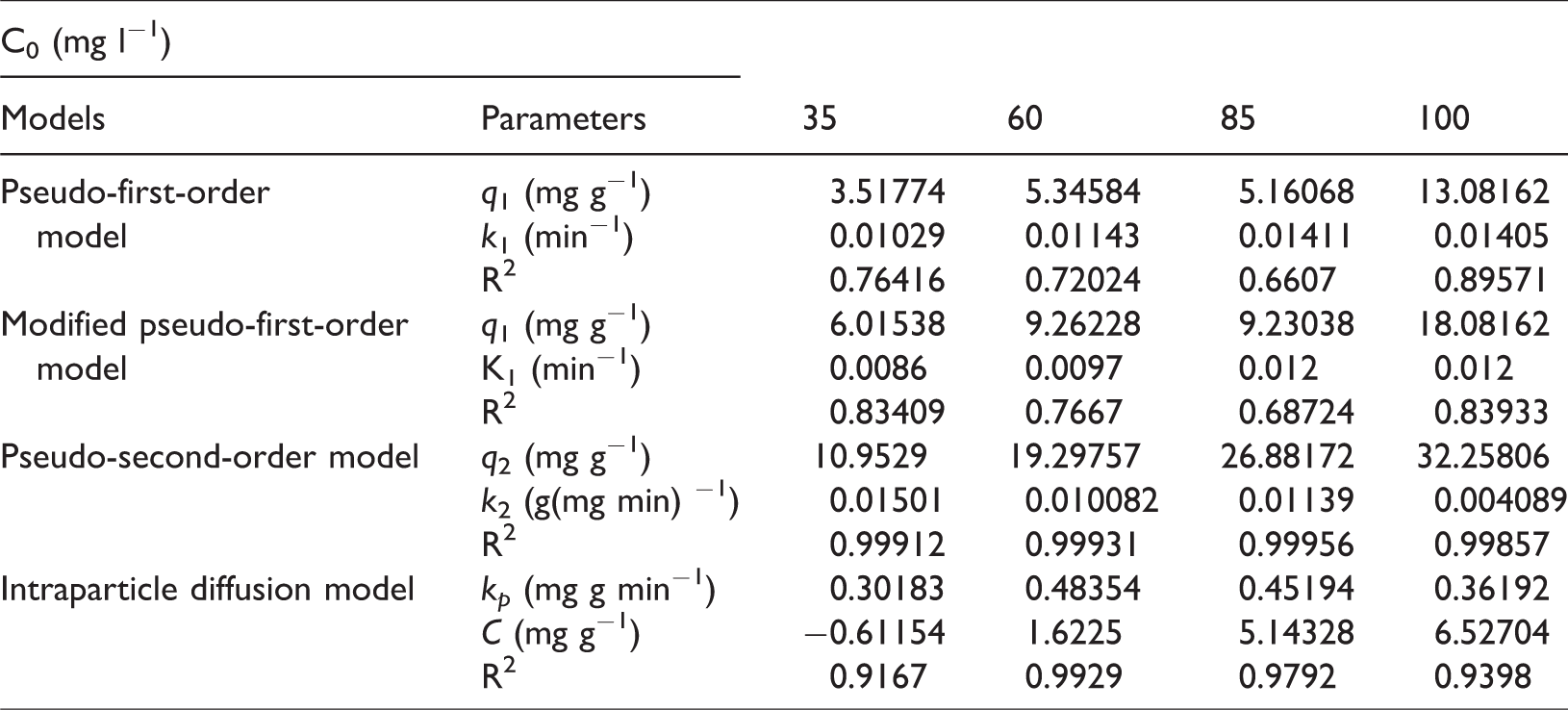
Based on the R2 values, the fitting degree of the pseudo-first-order kinetic equation and modified pseudo-first-order kinetic equation was the lowest under different concentrations (Table 2). The reason can be ascribed to the fact that the pseudo-first-order kinetic model is widely used, but it can only describe the adsorption kinetics of the initial stage and cannot accurately describe the whole process of adsorption.
The fitting degree of the pseudo-second-order dynamic equation and the intraparticle diffusion equation was relatively high. The R2 values of the fitting line of the pseudo-second-order dynamic equation were greater than 0.998, indicating that the pseudo-second-order equation could well describe the adsorption behavior of AC-PSAC-450-3 on reactive brilliant blue X-BR. The pseudo-second-order kinetics model was established on the basis of chemical adsorption, showing the whole process of liquid film diffusion, particle diffusion, and surface adsorption, which can highlight the adsorption mechanism of AC-PSAC-450-3 on reactive brilliant blue X-BR (Huang et al., 2017; Yuliani et al., 2017). The R2 values of the fitting line of the pseudo-second-order dynamic equation were better than those of the other types of kinetic models. These results indicate that the pseudo-second-order kinetic model is more accurate than the three other models for the adsorption behavior of reactive brilliant blue X-BR.
In addition, the R2 values of the fitting line of the intraparticle diffusion equation were between 0.9167 and 0.9929, which indicated that the adsorption process followed this model. According to Nassar (1999), if qt and t0.5 in the fitting line of intraparticle diffusion equation show a good linear relationship through the origin, then the diffusion process of the material in the internal particle is the only factor controlling the adsorption rate. Figure 7 shows that the lines obtained from fitting the data to the intraparticle diffusion do not pass by the origin. The results showed that the intraparticle diffusion was not the sole mechanism controlling the adsorption rate of AC-PSAC-450-3 for reactive brilliant blue X-BR. In addition, the adsorption rate was controlled by liquid film diffusion, surface adsorption, and other factors that were also related to the structural properties of reactive brilliant blue X-BR.
Conclusions
In this study, activated carbon samples were obtained from peanut shell with 50% phosphoric acid as an activator. These samples were characterized and applied to remove reactive brilliant blue X-BR.
The activated carbon prepared by carbonization after the activation of peanut shell powder with phosphoric acid had high specific surface areas and a large number of mesopores. The activated carbon obtained by the activation of peanut shell powder with 50% phosphoric acid for 24 h and the carbonization at 450°C for 3 h (AC-PSAC-450-3) was the best. When AC-PSAC-450-3 was used to decolorize the reactive brilliant blue X-BR solution with the concentration of 0.1 g l−1, the optimum dosage was 4 g l−1, and the optimum adsorption time was 2 h. The pseudo-second-order kinetics equation and the intraparticle diffusion equation could well describe the adsorption behavior of AC-PSAC-450-3. Intraparticle diffusion was the main factor controlling the adsorption rate of the activated carbon on the reactive brilliant blue X-BR solution.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Primary Research & Development Plan of Jiangsu Province (No. BE2016703) and the Natural Science Youth Fund of Jiangsu Province (No. BK20171017) and the National Natural Science Youth Fund of China (No. 51707093).
