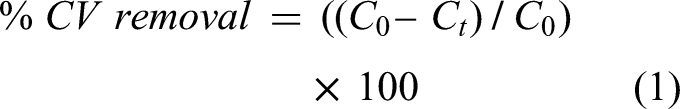Abstract
The increase in the world's population and the decrease in natural resources, including water, have caused mankind to look for ways to treat the wastes from various industries and reuse them. In this study, the effectiveness of the adsorbent prepared from oak tree fruit waste (activated carbon (AC)) has been investigated in order to remove crystal violet (CV) industrial dye from aqueous solution. The prepared adsorbent was characterized using the diffuse reflectance infrared Fourier transform (DRIFT), scanning electron microscopy (SEM), BET surface area measurement, energy dispersive X-ray (EDX) and powder X-ray diffraction (PXRD) analysis and applied as an effective adsorbent for removal of CV dye as an organic dye from an aqueous solution. Optimum conditions were obtained using 1.4 g adsorbent, 10 mg L−1 CV dye, 38 min contact time and pH 7.5 with 98% dye removal. Equilibrium data were evaluated using Langmuir, Freundlich and Tempkin isotherm. The Langmuir model best described the absorption of CV dye, indicating that it is monolayer and homogeneous (R2 = 0.996). The maximum absorption capacity under optimal conditions was 23.86 mg/g. Kinetic data were analyzed using pseudo-first-order, pseudo-second-order, and elovich equations, and the pseudo-second-order model showed the best fit for kinetic studies (R2 = 0.993).
Introduction
Nowadays, the rapid growth of industries has led to serious problems in the natural ecosystem. The effluents of many industries such as paint industries, metal plating, food industries, pharmaceutical industries, and battery manufacturing, that contain heavy metal ions, dyes, and organic materials, are discharged directly into water resources. According to the surveys, every year, about 300–500 million tons of solvents, heavy metals, toxic sludge, and other industrial wastes are released into water sources (Foroutan et al., 2022). Dyes and heavy metal ions, even at low concentration, can cause deleterious effects on human health (Mohammadzadeh Pakdel et al., 2018a, 2018b). In humans, this can cause skin irritation, gene change, device irritation, digestion, cancer, drowsiness, dysfunction of the liver, kidneys, brain, nervous and reproductive systems (Peighambardoust et al., 2022). In addition, the infiltration of different dyes into surface and underground water causes water sources to become colored. Dyes have a complex chemical structure and high molecular weight, which enter the environment due to various processes such as dyeing (Gamal and El-Sayed, 2011; McMullan et al., 2001). These dyes include disperse dyes, acid dyes, azo dyes, alkaline dyes, direct dyes, sulfur dyes, and reactive dyes. Crystal violet (C25N3H30Cl; Figure 1) is considered as one of the synthetic cationic dyes belonging to the triphenylmethane group, which causes purple color in aqueous solutions and easily enters the surface of negatively charged cell membrane (Siboni et al., 2014; Singh et al., 2011). It is used in textile industry, ink making and also as a biological dye in warm dyeing. This dye is non-biodegradable and stable in the environment and causes cancer, increased heart rate, severe eye irritation, vomiting, cyanosis, jaundice and tissue necrosis in humans (Saeed et al., 2010; Saha et al., 2012; Shengfang, 2010). Until now, various technologies such as absorption, chemical regeneration, precipitation, coagulation, microbial decomposition, ozonation, reverse osmosis, ion exchange and membrane processes have been used to remove dyes. Among all the effective methods in dyes removal, surface absorption and the use of inexpensive absorbents such as agricultural and vegetable waste such as oak tree fruit waste, rice and wheat straw, rice husk, almond husk, banana peel and sunflower stem are the most common methods proposed (Mohammadzadeh Pakdel et al., 2018b; Demirbas, 2009; Thinakaran et al., 2008). Surface adsorption is a process during which the concentration of molecules of the adsorbed substance decreases due to contact with the absorbent surface in the solution, which is due to the gravitational force on its solid surface. Various physical and chemical factors are effective in the process of surface absorption and its amount depends on the nature of the absorbent and the adsorbed substance (Gong et al., 2005; Mahir et al., 2005; Armagan et al., 2004; Dogan and Alkan, 2003; Ozcan Gong et al., 2005; Magdy and Daifullah 1998). The purpose of the current research is to prepare activated carbon from oak tree fruit waste to absorb crystal violet dye and to investigate effective parameters in the absorption process, including; contact time, adsorbent dose, initial concentration of dye in the solution and efficiency of the prepared adsorbent in different temperature conditions. The prepared activated carbon has unique properties such as porous structure, small particle size, high specific area and surface area, availability of precursor material, and high mechanical strength. Also, kinetic, isothermal models and thermodynamic equilibrium studies were investigated for the removal of crystal violet dye.

Chemical structure of CV.
Experimental
Instruments and reagents
UV-Visible spectrophotometer model V-530 (Jasco, Japan) at 590 nm wavelength was used to measure the concentration of CV dye. pH meter model-686 thermometer Metrohm was used to measure the pH of solutions (Metrohm, Switzerland, Swiss). The diffuse reflectance infrared Fourier transform (DRIFT) spectra were determined using a Brucker-Vector 22 (Brucker Analytik, Karlsrhur, Germany). In order to investigate the morphology of the prepared absorbent surface, a KYKY-EM3200 scanning electron microscope (SEM) was used (China). BET analysis (SA 3100, Beckman Coulter) was used to determine properties such as specific surface area, volume and diameter of active carbon cavities. Energy dispersive X-ray (EDX) was performed using a Seron AIS 2300 instrument (Korea) for the elemental analysis of the absorbent. Powder X-ray diffraction (PXRD) was obtained using a Bruker D8 ADVANCE diffractometer (Germany). All chemicals including CV dye (MW :407.98 g mol−1, dye content ≥95%, λmax = 590 nm) NaOH, HCl and EtOH with the highest purity available were purchased from Merck (Darmstadt, Germany).
Measurements of dye uptake
The dye concentrations in the aqueous solution were estimated quantitatively using the linear regression equations obtained at different CV concentrations. The efficiency of CV removal was determined for 10 mg L−1 of initial CV concentration with 1.4 g of AC at 38 min of contact time and pH 7.5. The experiments were also performed in the initial CV concentrations range of 10–40 mg L−1 to obtain adsorption isotherms. The CV removal percentage was calculated using the following equation (Ghaedi et al., 2011; Araújo et al., 2020; Elhamifar et al., 2016):
The adsorbed CV amount (qe (mg g−1)) was calculated by the following mass balance relationship:
Preparation of adsorbent from oak
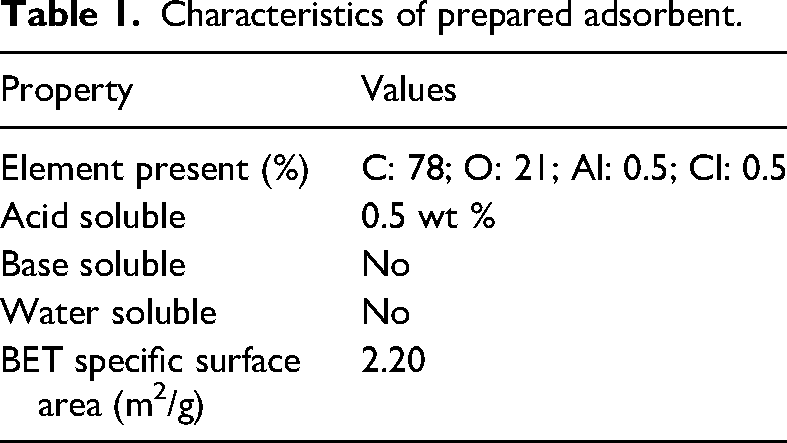
In order to prepare active carbon, firstly, oak tree fruit waste was ground as a precursor and placed in an oven at a temperature of 80°C to dry completely, then it was carbonized in a pyrolysis furnace at a temperature of 650°C in the presence of argon gas for 2 h. The carbonized sample was removed from the reactor after cooling the furnace to room temperature. Then it was mixed with potassium hydroxide at a ratio of 3:1. After this step, the sample was placed in an oven with a temperature of 100°C to dry completely. Next, the sample was placed in an oven with a temperature of 800 °C for 2 h in the presence of argon gas. Finally, the sample was cooled to room temperature in the presence of argon gas flow and in order to remove potassium and ash, the carbon was washed using 0.01 normal hydrochloric acid solution and distilled water. Then it was placed in the oven at 100°C for 24 h to dry completely (Chandrasekhar and Pramada, 2006; Hamdaoui, 2006). The characteristics of the prepared adsorbent are given in Table 1.
Characteristics of prepared adsorbent.
Results and discussion
Characterization of adsorbent
The DRIFT spectrum of AC is illustrated in Figure 2(a). According to it, the bands observed at about 2850–2925 cm−1 are attributed to the aliphatic C-H groups. Also, the O-H band is located at about 3450 cm−1. The observed peaks in wave numbers 1724 cm−1 and 1610 cm−1 can be related to the carbonyl stretch of carboxyl and C-O stretching, respectively. According to the IR spectrum, the presence of important surface functional groups was confirmed (Ghaedi et al., 2011; Araújo et al., 2020; Elhamifar et al., 2016). The DRIFT spectrum of AC after adsorption of CV showed the same bands with changes in some areas (Figure 2(b)). As shown, the intensity of the bands corresponding to aliphatic C-H vibrations and C = C vibrations, is little increased, confirming successful adsorption of CV dye on AC (Ghaedi et al., 2011; Araújo et al., 2020; Elhamifar et al., 2016). SEM analysis was performed to evaluate the morphology of the prepared activated carbon (Figure 3(a) and (b)). The SEM image of the AC (Figure 3(a)) revealed the presence of regular and side-by-side holes with uniform morphology. The SEM image of the AC after adsorption of CV was also investigated (Figure 3(b)). This image demonstrated a different morphology in comparison with its AC parent. This observation may be attributed to adsorption of the CV molecules onto/into the AC. BET analysis was used to determine properties such as the specific surface area of activated carbon. The specific surface area of the AC prepared from oak based on BET surface area analyzer was 2.2 m2/g. Figure 4 shows the pore size distribution curve of the proposed adsorbent based on the nitrogen equilibrium adsorption isotherm at 77 K. According to the shape, the prepared adsorbent shows the distribution of particles in both the macropore and mesoporous ranges. About 60% of the pores were in the diameter range below 40 nm with a total pore volume of 11.60 × 10−3 mL/g nm, indicating a high mesopore volume. The second part of the pores appeared in the range of 40 to 100 nm. In this range, the pore volume was 0.41 × 10−3 mL/g nm, which includes about 40% of the total pore volume and indicated the presence of macropores. As it can be seen, that the size of the holes in the prepared activated carbon mainly includes microporous structures. The chemical composition of the AC was determined by energy-dispersive X-ray (EDX) spectroscopy. Table 1 shows the elemental composition of the AC. Also, amorphous features are observed in PXRD patterns (Figure 5). This may be due to the lack of minerals in lignocellulosic biomass. The wide range of the spectrum indicates the irregular structure of activated carbon, which leads to the placement of more free carbon on the surface and basically increases the activity energy and reactivity of the surface. As a result, the surface interactions between the adsorbent and the adsorbed will increase (Ghaedi et al., 2011; Araújo et al., 2020; Elhamifar et al., 2016).

Diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) of AC (a) before and (b) after adsorption of CV dye.

(a) Scanning electron microscopy (SEM) image of AC before the adsorption of CV (b) after the adsorption of CV.

Pore size distribution of adsorbent prepared from oak.

Powder X-ray diffraction (PXRD) pattern of the AC.
Effect of pH
The pH level is one of the most important properties of wastewater and aqueous solutions, which can affect the surface characteristics of the absorbent and the absorption capacity by changing the degree of ionization (Chandrasekhar and Pramada, 2006; Hamdaoui, 2006). The effect of initial pH on the CV removal was studied in the pH range of 2–8.5 with CV concentration of 10 mg L−1 and adsorbent dose of 1.4 g. The experiments were conducted for 38 min of contact time at room temperature and the respective results are presented in Figure 6(a). As can be seen, the maximum removal of the CV is obtained at pH 7.5. Therefore, all subsequent studies were carried out at pH = 7.5 as optimum pH. Furthermore, the point of zero charge (pHPZC) of an adsorbent is a very important characteristic that determines the pH at which the adsorbent surface has net electrical neutrality. To find out pHpzc, the experiments were conducted at different pH ranges from 2 to 10 under other optimal conditions. The pHPZC of AC was determined to be 6.8. At the solution pH > pHPZC, the adsorbent surface negatively charged and favors uptake of cationic dyes due to increased electrostatic force of attraction. At lower pH (pH < pHPZC), the surface of AC may get positively charged as a result of being surrounded by H3O+ ions and thus the competitive effects of H3O+ ions as well as the electrostatic repulsion between the dye molecules and the positively charged active adsorption sites on the surface of the AC lead to a decrease in the uptake of dye molecules (Chandrasekhar and Pramada, 2006; Hamdaoui, 2006). Another important factor for this adsorption process is H-bonding interactions between N-sites of dye and OH sites of adsorbent.
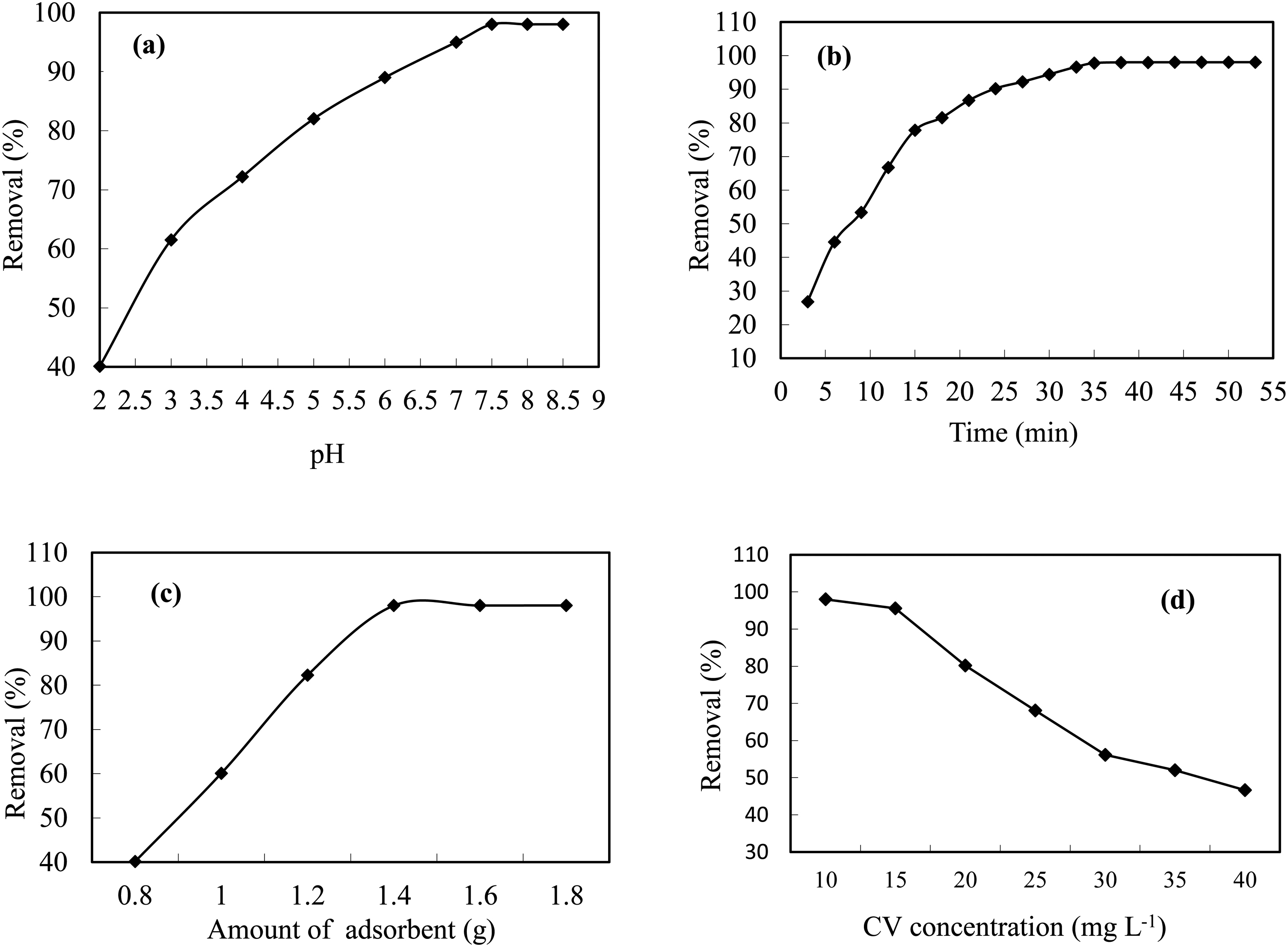
Effect of (a) ph; (b) contact time; (c) adsorbent dosage; (d) initial dye concentration on the removal of CV by AC.
Effect of contact time
After determining the optimal pH, in this step, the effect of contact time of the absorbent with the color solution on the dye removal efficiency by AC was investigated. The pH, concentration and dose of adsorbent were kept constant and the absorption efficiency was checked by changing the contact time from 3 to 55 min (Figure 6(b)). According to this figure, in the first minutes of adsorption, due to the high concentration of dye in solution, the number of effective interactions between the dye and the adsorption sites on the surface AC are increased. Moreover, with the progress of time, the concentration of the dye is decreased and the adsorption sites are mostly saturated (Asgari et al., 2013). The adsorption of dye onto AC reached equilibrium within 38 min. Also, the rapid attainment of equilibrium on the AC surface can be attributed to the abundance of surface adsorption sites, which are increased due to the high porosity of the adsorbent and suitable functional groups. Therefore, the availability of binding sites and the existence of relatively strong electrostatic interactions between these sites and CV dye molecules make it possible to reach the equilibrium state in a short time (Asgari et al., 2013; Ravanan et al., 2014).
Effect of adsorbent dosage
The amount of adsorbent is an important and influential parameter in the absorption process, so it determines the capacity of the adsorbent for a certain initial concentration of the adsorbate (Asgari et al., 2013; Ravanan et al., 2014). The influence mass of AC on CV removal reveal that change in removal percentage from 40% to 98% by enhance in AC mass from 0.8 to 1.8 g (Figure 6(c)). These expected results are attributed to increases in the number of available adsorption sites and progress in the uptake of the CV. At lower amount due to probable saturation of reactive centers a significant decrease was seen in removal percentage.
Effect of initial dye concentration
The effect of the initial concentration of CV on the removal efficiency in the process of surface adsorption was studied and investigated by changing the initial concentration from 10–40 mg/L, in the condition of other parameters being constant. As it is clear in Figure 6(d), the efficiency of dye removal by the used adsorbent decreased with the increase of dye concentration, which is normal, because with the increase of the initial dye concentration, its residual amount also increases, and therefore its removal efficiency decreases, and the reason Another is the saturation of the absorbent surface at high concentrations of dye (Ghaedi et al., 2011, 2012). The maximum absorption capacity was 23.86 mg/g for the concentration of 10 mg/L CV dye and other optimal conditions.
Adsorption equilibrium study
The adsorption process is usually expressed through isotherms, which is equivalent to the amount of adsorbed substance on the absorbent surface and is a function of the pressure or concentration of the adsorbed substance under constant temperature conditions. In this case, the surface adsorption isotherm process describes the type of interaction of the CV dye with the prepared activated carbon. To calculate CV dye surface absorption isotherms, the data of optimal conditions, i.e., the effect of initial dye concentration, were used. The adsorption isotherms used in this research are Langmuir, Freundlich and Tempkin surface adsorption isotherms (Ghaedi et al., 2014; Ho et al., 2000; Freundlich, 1906; Theydan and Ahmed, 2012). Data related to Langmuir, Freundlich and Tempkin surface adsorption isotherms are presented in Table 2 and Figure 7(a) to 7(c). The results show that the maximum surface adsorption capacity in the Langmuir isotherm model for CV dye using activated carbon is equal to 23.86 mg/g. This absorption capacity indicates that the adsorbent is very effective for removal CV dye from aqueous solutions. According to the obtained results, the surface absorption process of CV dye by the adsorbent can be described well with the Langmuir isotherm model, the correlation coefficient of 0.996, which indicates the absorption of a single layer and the uniform nature of the adsorbent.

(a) The Langmuir; (b) Freundlich; (c) Tempkin plots for the adsorption of CV on AC (adsorbent dose: 1.4 g/25 mL, pH: 7.5, 38 min agitation time at speed of 400 rpm); (d) First-order; (e) Pseudo-second order; (f) Elovich kinetic plots for the adsorption of CV on AC (dye concentration: 10 mg/L, adsorbent dose: 1.4 g/25 mL, pH: 7.5, stirrer speed: 400 r/min).
Isotherm parameters for adsorption of CV on AC

Kinetics evaluation
The kinetics of surface absorption is one of the most important factors in the design of the surface absorption system, predicting the speed of the absorption process, the factors affecting it and how to do them (Lagergren, 1898; Azizian, 2004; Azizian and Naviri-fallah, 2010; Azizian and Bashiri, 2008; Brdar et al., 2012; Khansorthong and Hunsom, 2009). Contact time data were used to calculate surface adsorption kinetics. For this purpose, in this research, the description of the kinetic behavior of surface adsorption of CV dye on the surface of activated carbon was studied using pseudo-first-order and pseudo-second-order kinetics models, as well as elovich and intraparticle diffusion kinetics models. Surface adsorption kinetic data are presented in Table 3 and Figure 7(d) to f. The results show that the absorption of CV dye by AC with a correlation coefficient equal to 0.993 follows the pseudo-second order kinetic model. In addition, in the pseudo second-order kinetic model, the calculated surface absorption capacity (Qcal) is closer to the measured surface absorption capacity (Qe). These results show that the pseudo-second-order kinetic model includes all steps of surface adsorption and can describe the mechanism of surface adsorption well.
Kinetic parameters for the adsorption of CV on AC

Effect of temperature
Temperature has important effect on adsorption process. Adsorption of CV at different temperatures in the range of 10–50 °C using 1.4 g of AC and pH 7.5 was studied. The experimental results show that the surface absorption process of CV by activated carbon is controlled through endothermic absorption. In addition, the increase in absorption with temperature may be due to the high diffusion rate of dye molecules and the low viscosity of the dye solution compared to the absorbent particles. Also, at the evaluated temperatures, the absorption capacity increases with increasing temperature. Increasing the movement of dye molecules can gain enough energy to interact with the active sites on the surface of the adsorbent. An increase in temperature can also create a swelling effect within the internal structure of the absorbent, which causes the dye to penetrate further into the internal cavities. This confirms the endothermic nature of dye adsorption (Azizian, 2004; Azizian and Naviri-fallah, 2010; Azizian and Bashiri, 2008; Brdar et al., 2012; Khansorthong and Hunsom, 2009; Gupta et al., 2011).
Thermodynamic parameters including change in Gibb's free energy (ΔG0), entropy (ΔS0) and enthalpy (ΔH0) for the adsorption of CV over prepared adsorbent have been determined by using the Equations (3) and (4).



Thermodynamic parameters for the adsorption of CV dye on AC.

Various adsorbent for CV removal
Many CV removal processes using various adsorbents were reported in the literature (Mittal et al., 2010; Malarvizhi and Ho, 2010; Bertolini et al., 2013; Gandhimathi et al., 2012; Eren and Afsin, 2007; Abd-Elhamid et al., 2019; Wu et al., 2023; Putri et al., 2020) and their performance for CV removal was compared in term of adsorption capacity, amount of adsorbent and contact time. It can be seen that the removal of CV by AC (Table 5) has a noticeable superiority in terms of absorption capacity considering other conditions (23.86 mg g−1).
Comparison of adsorption results of previously reported CV removal with the proposed adsorbent.

Ability to reuse the absorbent
In order to check the stability and recovery ability of the adsorbent, the desired adsorbent was used during four periods of absorption and desorption according to the proposed method. For this purpose, every time at the end of the experiment, the absorbent was collected and then washed well with distilled water and dried in an oven. The results are shown in Figure 8. Considering that some of the absorbent may be lost during the washing of the absorbent, and there is also a possibility that a small amount of dye will remain on the surface of the absorbent during the washing process, so a small amount of reduction in absorption can be seen in Figure 8. As it is known, the prepared adsorbent can be used for at least four times without significant reduction in absorption capacity.

Recoverability and reusability of AC.
Conclusion
The results of this research show that activated carbon prepared from oak tree fruit waste can be used as an effective and efficient adsorbent to remove CV dye from aqueous solutions. The studies to determine the properties of the adsorbent according to various analyzes showed that the adsorbent has functional groups, size, particle distribution, specific surface area and appropriate morphology, which is a special point for removing dyes from aqueous solutions by surface absorption. The extent of adsorption has been investigated with various operating parameters, like; adsorbent dose, contact time, pH, initial dye concentration, and temperature. The use of 1.4 g of adsorbent, 38 min of contact time and 10 mg L−1 of CV at pH 7.5 were determined as optimum conditions to achieve maximum removal of 98% for adsorption process. In the following, isotherm and kinetic studies of the adsorption process were investigated, and the results show that the adsorption process fits well with the Langmuir isotherm model and pseudo-second-order kinetics. In addition, the thermodynamic parameters showed that the surface adsorption process for CV dye by the prepared adsorbent is endothermic and is associated with an increase in disorder. The reuse of the prepared adsorbent showed that the adsorbent can be recycled in 4 steps with an efficiency of more than 93%. Finally, according to the characteristics of the prepared activated carbon, such as naturalness, availability in large quantities, non-toxicity, low cost of the preparation method and the possibility of recycling, it can be used to remove toxic dyes from aqueous solution with high efficiency.
Footnotes
Acknowledgement
The author acknowledges the Farhangian University for supporting this work.
Data availability statement
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.