Abstract
The discharge volume and composition of textile effluents gain scientific concern due to hazardous and biotoxic nature of azo dyes. Azo dyes are non-biodegradable due to its complex molecular structure and recalcitrant nature. Serious attempts have been made to synthesize and develop new materials to combat the environmental problems. The present study is designed for removal of azo dyes (Methyl orange, Congo red and Basic fuchsine) from synthetic aqueous solutions and real textile effluents. For this purpose, cobalt-doped alumina hybrids are synthesized and applied as adsorbents in batch experiment. Two different aluminium precursor (aluminium nitrate and spent aluminium foil) and glucose are mixed following sol–gel method to get hybrids. The synthesized hybrids are characterized for surface and bulk properties using Fourier transform infrared, scanning electron microscopy and energy dispersive X-ray techniques. The characterization of materials under Fourier transform infrared revealed that –OH (3487–3504 cm−1), C–H (2935–2985 cm−1), Al–O–C (∼1380 cm−1), Al–O (∼ 800 cm−1), Al–O–Al (659–669 cm−1) and Co–O (583–589 cm−1) groups participate in the binding of dyes onto the surface of hybrids. Amorphous shaped particles and elemental composition (carbon, aluminium and oxygen) are demonstrated in scanning electron microscopy–energy dispersive X-ray micrograph. Time-dependent batch experiments under identical experimental parameters showed 74% of basic fuchsine, 68% of methyl orange and 85% maximum removal of Congo red onto the surface of cobalt-doped alumina hybrids probably through ion exchange mechanism. The experimental data when treated with adsorption models and isotherms, pseudo-second-order kinetic and Freundlich isotherm are found to be best fitted. The present study accomplishes the successful synthesis of novel and efficient cobalt-doped alumina hybrids provides environmental friendly and economical alternative option to the commercial adsorbents for the treatment of textile effluents.
Introduction
Textile industry is a flourishing industry in Pakistan due to high demand of cotton fabric. It serves as backbone to the economy of Pakistan. Textile industry alone contributes 46% of the total exports. It equals to US$9.6 billion and 8.5% of total GDP of the country (Khan and Khan, 2010). It provides employment to 38% of the workforce in the country, which sums up to 15 million people (Iqbal et al., 2010). Pakistan is ranked the eighth largest exporter of textile products worldwide (Dey and Islam, 2015). Textile industry consumes two-thirds of the dyes manufactured in the world and discharge effluent directly in the water bodies. It is highly polluted with azo dyes. It is applied to fibres to give permanent colour, i.e. resistant to water, light and soap (Sayal et al., 2012).
Sayal et al. (2012) reported that every dyestuff consists of one or more compound obtained from distillation of coal tar such as anthracene (C14H10), naphthalene (C10H8), benzene (C6H6), acridine (C13H9N), phenol (C6H5OH) and quinolone (C9H7N) (Das, 2011). Dyes are not only aesthetically undesirable but it also reduces sunlight transmission into water bodies affecting aquatic plants and have toxicological effects such as allergic conjunctivitis, dermatitis, occupational asthma, rhinitis and allergic reactions (Lorenc-Grabowska and Gryglewicz, 2007). Research work has been carried out for the treatment of dye from wastewater using different technologies, e.g. chemical coagulation–flocculation (Riera-Torres et al., 2010), oxidation (Yonar, 2011), membrane-based separation processes (Amini et al., 2011), etc. These technologies have efficiency for remediation of dyes but initial and operational costs are very high. Adsorption technique (Sulak and Yatmaz, 2012) is quite popular due to its simplicity because it does not require additional chemicals during pre- and post-treatment, efficient and cost effective for the treatment of non-biodegradable pollutants (including dyes) from wastewater. Several adsorbents have been studied like rice husk (Han et al., 2008), wood bottom ash (Leechart et al., 2009), chitosan-based composites (Perju and Dragan, 2010), activated carbon (Lu, 2011), amine functionalized silica SBA-3 (Anbia and Salehi, 2012) for the removal of Congo red, basic dye, methylene blue, azo reactive, red reactive141, Congo red and direct blue, acid dyes. The quest for more efficient and improved material introduced hybrid materials with its unique properties. Hybrid materials are composites of inorganic and organic moiety at molecular or atomic level with novel features that are different from individual one (Kickelbick, 2007). Several hybrid materials have been reported such as EDTA-modified silica–chitosan (Repo et al., 2011), amine-modified polyglycidyl methacrylate grafted cellulose (Anirudhan and Senan, 2011), poly(acrylic acid)-modified core-shell-type resin (Yavuz et al., 2011), chitosan-modified palygorskite composite (Peng et al., 2013), superparamagnetic graphene oxide–Fe3O4 hybrid composite (Xie et al., 2012), chitin hydrogel/SiO2 and chitosan hydrogel/SiO2 hybrid materials (Copello et al., 2011), polymer-grafted alumina (Wåhlander et al., 2014). The present study is an endeavour to synthesize efficient and economical adsorbent, i.e. cobalt-doped alumina hybrids for the treatment of textile effluent.
Materials and methods
Synthesis of alumina hybrid
Alumina hybrids were synthesized using two inorganic precursors (i.e. spent aluminium foil (green method), aluminium nitrate nanohydrate (reference method)) and one organic (glucose) precursor following sol–gel protocol (Rogojan et al., 2011; Zhou et al., 2012) to hybridize inorganic and organic moiety. These products are coded as G-AH and R-AH.
Synthesis of cobalt-doped alumina hybrids
An equimolar volume of dopant (cobalt nitrate hexa-hydrate aqueous solution) is added into mixture of aluminium and glucose (1:1) solution following sol–gel method to enhance the adsorption efficiency. It is expected that modifications improve the sorption capacity of the synthesized materials. These products are coded as G-AH-Co and R-AH-Co.
Characterization
The synthesized hybrids were characterized using different spectroscopic techniques, i.e. Fourier transform infrared (FTIR) spectroscopy to study the structural, energy dispersive X-ray (EDX) for compositional and scanning electron microscopy (SEM) for morphological study of alumina and cobalt-doped alumina hybrids.
Batch adsorption experiment
Batch mode experiments were conducted to evaluate the adsorption efficiency of synthesized alumina hybrids against selected azo dyes at their respective pH, i.e. Congo red = 5, methyl orange = 7 and basic fuchsine = 8. After giving appropriate contact time (2–5 min) to adsorbate and adsorbent, filtrate was run on pre-calibrated UV–visible spectrophotometer (UV-1601, Shimadzu Japan) at respective λmax. The removal efficiency and adsorption capacity (qmax) of dye were calculated using these equations
Adsorption kinetics and isotherms
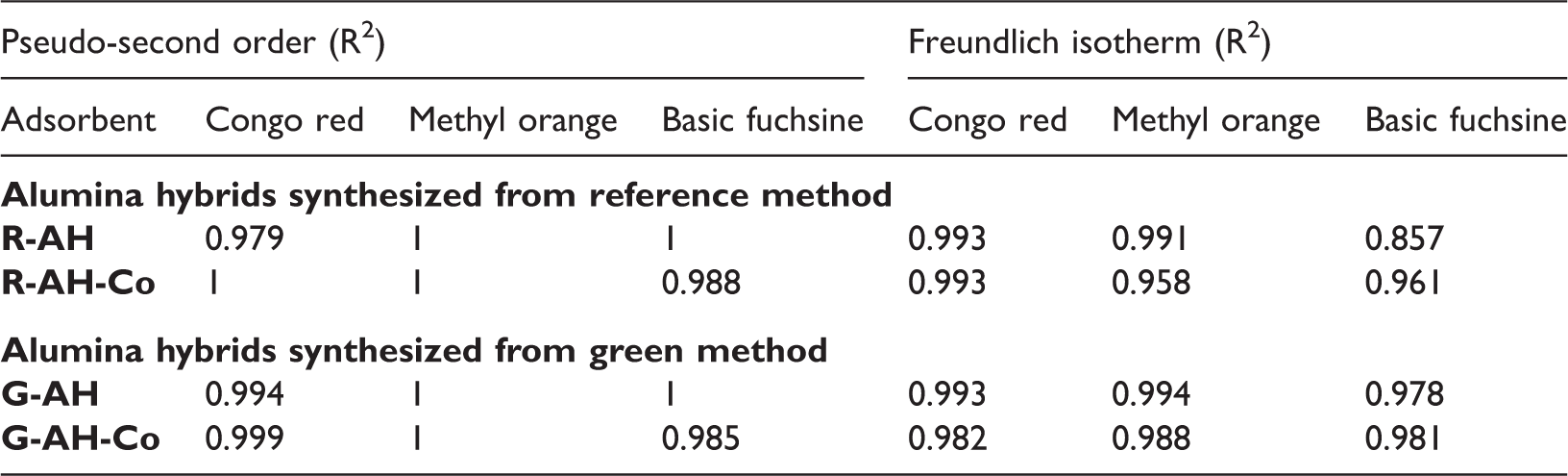
The well-established adsorption kinetics (first, second, pseudo-second and intraparticle diffusion) and adsorption isotherms (Langmuir and Freundlich) were applied on experimental data to evaluate sorption capacity of synthesized alumina hybrids. The best fitting of each kinetics and isotherms were evaluated by linear regression (R2) value.
Results and discussion
Fourier Transform Infra-Red (FTIR) Spectroscopy
FTIR frequencies of alumina hybrid and cobalt-doped alumina hybrid from reference (R-AH, R-AH-Co) and green method (G-AH, G-AH-Co).
FTIR: Fourier transform infrared.
Scanning Electron Microscopy (SEM)
SEM of alumina and cobalt-doped alumina hybrids synthesized from green and reference method were subjected for morphological analysis (see Figure 1).
SEM micrograph of alumina and cobalt-doped alumina hybrids synthesized from green (a = G-AH b = G-AH-Co) and reference (c = R-AH and d = R-AH-Co) methods. SEM: scanning electron microscopy.
The hybrid synthesized from aluminium foil (green method) showed irregular shaped blocks and crevices and pores with coarse particles. The incorporation of cobalt into the synthesized hybrids revealed interesting morphology as shown in Figure 1(b) and (d). The successful incorporation of cobalt into the matrix of hybrid is clear with deeper holes, interspaces and cluster formation. It led to a grain growth due to agglomeration. It is regarded as mesoporous material. This phenomenon is supported by Visinescu et al. (2010) narrating cobalt oxide doping enhances densification and promotes the grain growth of nano-powder. Alumina hybrids synthesized from spent aluminium foil (green method) have morphology comparable with standard aluminium salt (reference method) with no remarkable differences.
Energy Dispersive X-ray (EDX) Spectroscopy
EDX scans of synthesized hybrids from green and reference method shown in Figure 2 and data are tabulated (see Table 2). Oxygen, aluminium and carbon are the main identified elements. The successful doping of cobalt into the matrix of hybrids is confirmed by emergences of sharp peak in EDX. It is also noted that cobalt is doped to its higher content when reference method is adopted in comparison to green method.
EDX pattern of alumina and cobalt-doped alumina hybrids synthesized from green (a = G-AH, b = G-AH-Co) and reference (c = R-AH, d = R-AH-Co) methods. EDX: energy dispersive X-ray. Elemental composition (%) of alumina hybrids using EDX. EDX: energy dispersive X-ray.

Batch experiment
Each batch experiment was directed at 0.1 mg/l concentration of adsorbate and 5 mg dose of adsorbent till the attainment of equilibrium. Definite contact time (2–5 min) was provided for adsorbate and adsorbent. Then solution was filtered and run on UV–visible spectrophotometer (UV-1601, Shimadzu Japan) at their respective λmax (Congo red at 495 nm, methyl orange at 465 nm and basic fuchsine at 544 nm) and pH (Congo red = 5, methyl orange = 7 and basic fuchsine = 8) for the determination of residual dyes concentration.
The absorbed concentration was determined from standard calibration curves.
Adsorption of Congo red
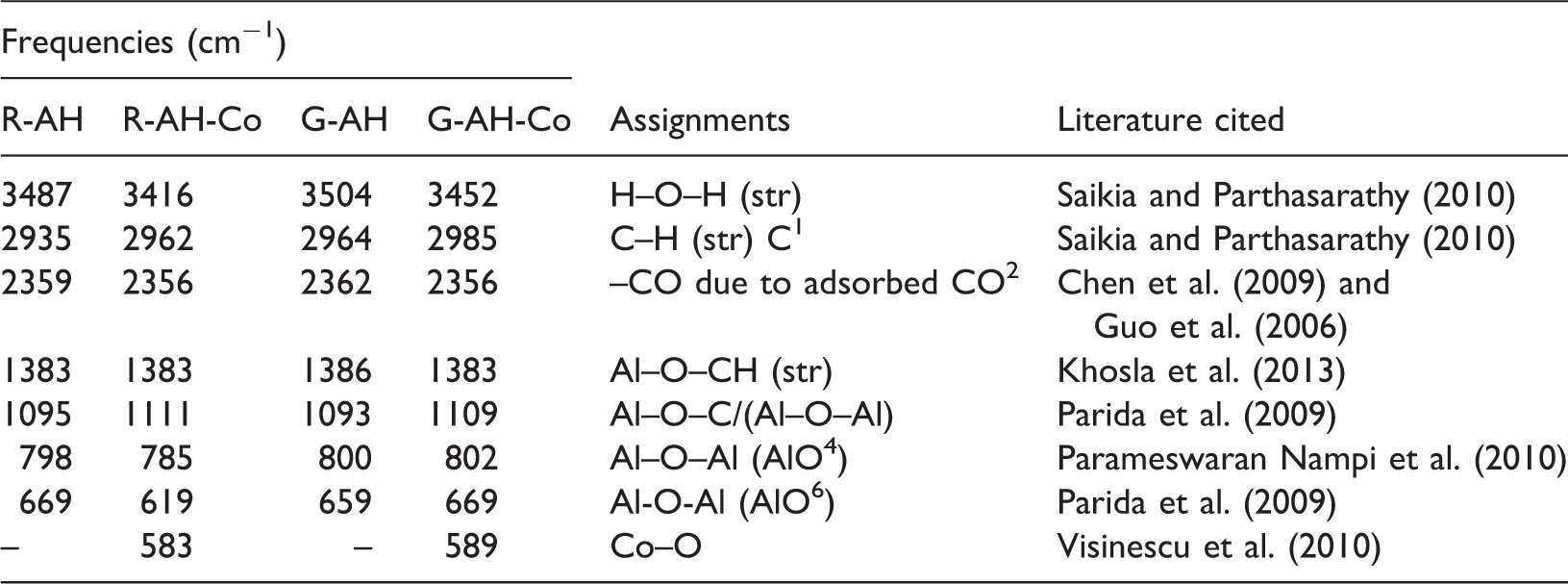
It is toxic dye having acidic nature and water solubility. It is sodium salt of 3,3′-([1,1′-biphenyl]-4,4′-diyl)bis(4-aminonaphthalene-1-sulfonic acid). It has affinity to cellulose fibres and that is why it is used in textile industry. Its metabolites are toxic in nature like naphthalene, aromatic amines, benzidine and sulfonic acid (Ben Mansour et al., 2007) and reported as mutagen, carcinogen, reproductive effectors, eye, skin and gastrointestinal irritant. The adsorption of Congo red onto alumina hybrids synthesized from reference method is shown in Figure 3(a). It started to adsorbed Congo red slowly and showed maximum 45% removal at 25 min. The adsorption pattern is slow and smooth up to equilibrium attainment. While cobalt-doped alumina hybrid (R-AH-Co) showed 83% maximum removal due to availability of more active sites on adsorbent surface and have ability to replace with protonated amine and sodium ion due to its cation exchange capacity. Alumina hybrid synthesized from green method showed comparable removal trend to reference method as shown in Figure 3(b). Alumina hybrid (G-AH) showed 41% removal of dye. Similarly cobalt doped showed 74% removal at 40 min. It takes more time to diffuse into the interior pores. The standard deviation (SD) of each adsorbent towards Congo red is calculated and reported in Figure 1. The percentage removal is highly significant and lies in the range of 0.000–0.002 at the level of 0.01 using statistical tool SPSS 16.
Per cent removal of Congo red using synthesized hybrid from reference (a) and green (b) method as a function of time.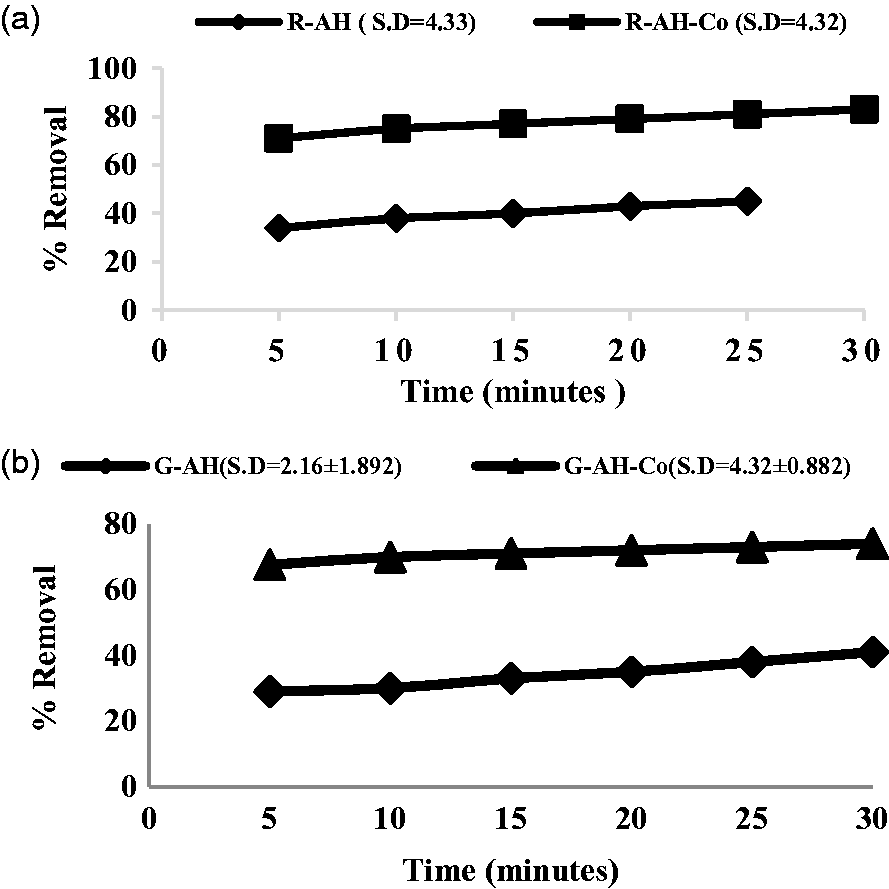
Adsorption of methyl orange
Methyl orange is of 2-(N,N-dimethyl-4-aminophenyl) azobenzene carboxylic acid. It can cause allergies and hypersensitivity. These dyes are very stable and show low biodegradability which makes their removal from wastewater difficult and costly. Alumina hybrid synthesized from reference and green method showed good uptake of methyl orange (see Figure 4). The results showed 29% removal of methyl orange by alumina hybrid (R-AH) synthesized from reference method as shown in Figure 4(a) due to the presence of repulsive force between negatively charged surface of adsorbent and anionic (methyl orange) dye. Removal of methyl orange is also studied by Haddadian et al. (2013). The cobalt-doped hybrid (R-AH-Co) showed increase in adsorption of methyl orange up to 68% than former one (R-AH) due to doping of cobalt metal on surface which provides more active sites towards anionic dye. Alumina hybrid synthesized from green route showed comparable removal trend as reference method shown in Figure 4(b). Alumina hybrid (G-AH) showed 43% removal of dye while cobalt-doped (G-AH-Co) alumina hybrid showed 65% removal at 30 min. The SD of each adsorbent towards methyl orange is calculated and reported in Figure 4. The percentage removal is highly significant and lies in the range of 0.001–0.01 at the level of 0.01 using statistical tool SPSS 16.
Per cent removal of methyl orange using synthesized hybrid from reference (a) and green (b) method as a function of time.
Adsorption of basic fuchsine
It is a tri-aminotriphenylmethane used for colouring the textile and leather materials. It may responsible for skin and eye irritation, spleen, liver and thyroid damages. In the present study, removal of basic fuchsine is also assessed using alumina and cobalt-doped alumina hybrids synthesized from reference and green methods (see Figure 5). The results depicted 47 and 74% removal of basic fuchsine using alumina hybrid (R-AH) and cobalt-doped alumina hybrid (R-AH-Co) synthesized from reference method. The regular increasing trend started from 5 min and end in 35 min as shown in Figure 5(a). Basic fuchsine binds with oxygen atom surrounding each octahedral vacancy forming bi-dentate or tri-dentate attachments. Khraisheh et al. (2005) reported the same adsorption behaviour for methylene blue (basic dye). Similarly alumina hybrid synthesized from green method showed 55% removal of basic fuchsine whereas addition of cobalt metal enhanced the sorption capacity up to 72%. It can be seen in Figure 5(b) that adsorption trend is smooth and regular. The SD of each adsorbent towards basic fuchsine is calculated and reported in Figure 5. The percentage removal is highly significant and lies in the range of 0.001–0.01 at the level of 0.01 using statistical tool SPSS 16.
Per cent removal of basic fuchsine using synthesized hybrid from reference (a) and green (b) method as a function of time.
Industrial application
For industrial application, the wastewater samples were collected from the outlet of textile mill. Alumina hybrid and cobalt-doped alumina hybrids were applied as adsorbent, for the adsorption of Congo red, methyl orange and basic fuchsine at their respective λmax (465, 495 and 544 nm). The concentration of selected dye in wastewater is pre-determined and illustrated in Figure 6. The following trend showed optimum removal of dyes using alumina hybrid and cobalt-doped alumina hybrid synthesized from reference and green method (see Figure 7(a) and (b)). It is observed that adsorbent removed less azo dye from industrial wastewater as compared to synthetic solution. This might be due to presence of degradable and non-degradable pollutants in textile effluent as compared to synthetic solution.
Concentration of Congo red (CR), methyl orange (MO) and basic fuchsine (BF) in textile effluent. Per cent removal of selected azo dyes using alumina hybrid and cobalt-doped alumina hybrid using reference (a) and green (b) method from textile effluent.
Each pollutant has a different affinity towards hybrid adsorbents. Further the competition for limited adsorption sites available on the adsorbent surface offers a win-win situation for pollutants having higher affinity. Less adsorption of dyes also might be the indication of instantaneous capability of adsorbent to adhere more than one pollutant at a time exhibiting multifunctional property.
Proposed sorption mechanism
Dyes containing amine, methyl and sulfonic groups attached to the naphthalene rings at the ends, respectively. These functional groups are responsible for the adsorption process. The amine, methyl and sulfonic groups protonated in water and become negatively and positively charged. Adsorption process is not always a single-stage process (Lin et al., 2000). Initially molecules escape from the aqueous phase near the solid surface and associated with it via varying types of forces, such as dipole–dipole, ion exchange, van der waals interactions and covalent as reported by Sakkayawong et al. (2005).
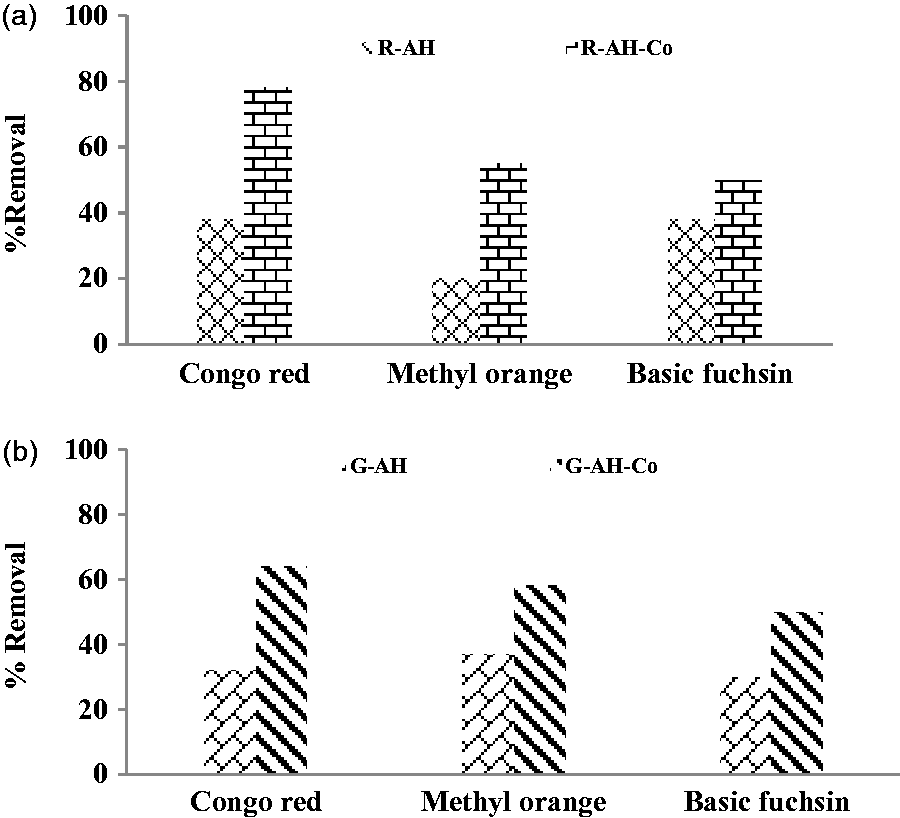
The surface of adsorbents (alumina hybrids) without dopant is negatively charged due to presence of oxygen. It showed interaction with basic fuchsine (cationic nature) through cation–anion exchange (Khan et al., 2015; Santos and Boaventura, 2016) with the high adsorption removal while it showed weak interaction towards Congo red (anionic nature) Basic Fuchsine > Methyl orange > Congo red
When dopant (cobalt) is doped onto alumina hybrids, nature of adsorbent surface is changed. It created more active site to cater more dye molecules on the surface. Doped alumina hybrid showed strong interaction towards Congo red (Khan et al., 2015) than methyl and basic fuchsine due to alteration of surface charge. It is observed that adsorption takes place through cation–cation exchange to replace with protonated amine, methyl and sodium ion Congo red >Basic Fuchsine > Methyl orange
Adsorption kinetic and isotherms
The value of best fitted regression value (R2) of pseudo-second-order kinetic and Freundlich isotherm.
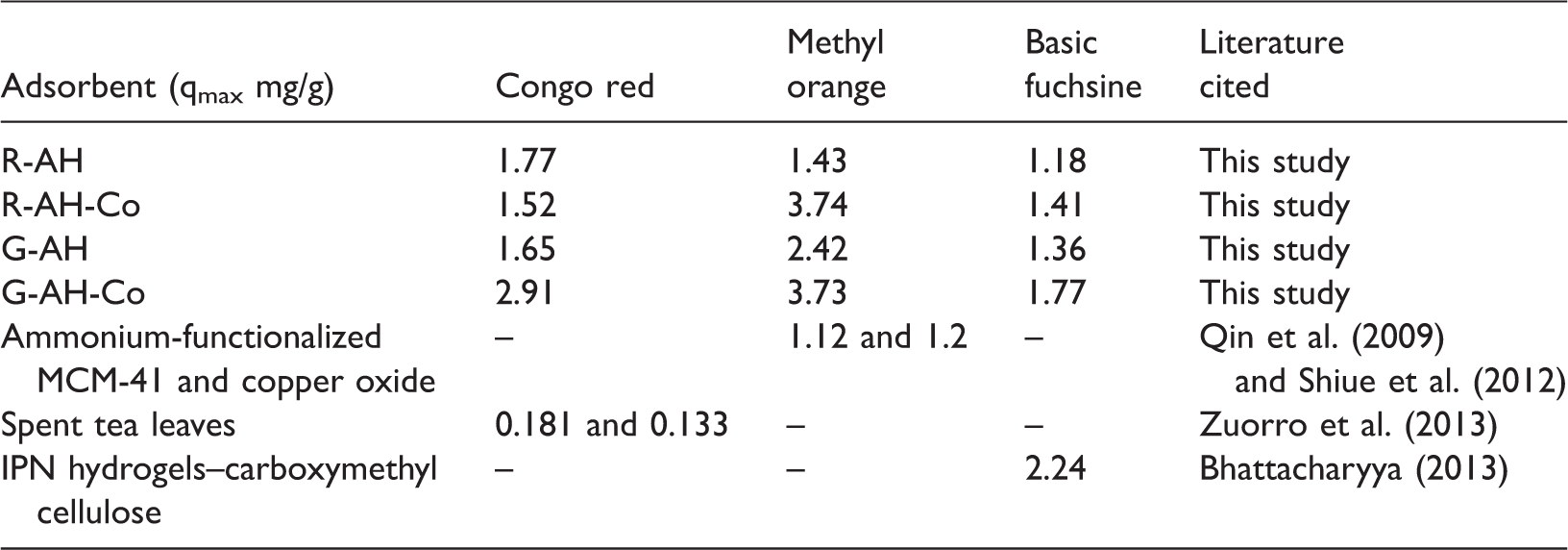
Comparison of adsorption capacity (qmax) of different adsorbents with present study.
Cost–benefit analysis
Selection of remediation technique at industrial scale depends on availability, ease of operation, regeneration and profitability. Adsorption technique fulfils all the characteristics.
It is a simple and efficient technique than other remediation approaches like precipitation, oxidation, gravity separation, coagulation, distillation, solvent extraction, micro and ultra-filtration, electrolysis. It does not require additional chemicals during pre- and post-treatment, easy to operate, availability of a wide variety of adsorbents, low operational cost and occupy less space for unit instalment. The cost of adsorption is also dependent upon the cost of adsorbent and regeneration ability (Al-Ghouti et al., 2009). The alumina and cobalt-doped alumina hybrid synthesized in this study presented comparison between reference (aluminium salt) and green (spent aluminium foil) route. The cost of aluminium nitrate nanohydrate is € 477/100 g while spent aluminium foil is waste material. Chemical residue generated from this technique can be recycled and reused.
Conclusion
The present study concludes as follow:
It is found that alumina hybrids and cobalt-doped alumina hybrid are successfully synthesized from green (aluminium foil) and reference (aluminium nitrate) method through sol–gel process confirmed using different spectroscopic techniques (FTIR, SEM and EDX). Cobalt doping is the signatory mark of present study. FTIR analysis showed –OH, C–H, Al–O and Al–O–Al functional group on the surface of hybrid adsorbents. These groups are responsible for binding. The interaction of dyes with adsorbents is likely due to cation–anion exchange process. Hybrid formation is confirmed from absorption band appearing at 1383–1386 cm−1. Alumina hybrids are found to be amorphous that changes to semi-crystalline form upon cobalt doping. EDX patterns indicate 23–44% of carbon, 29–39% of aluminium and 11–20% of oxygen as elemental composition. The alumina hybrid and cobalt-doped alumina hybrid synthesized from green and reference method showed 74 and 83% removal of Congo red, 65 and 68% removal of methyl orange and 72 and 74% of basic fuchsine. It is also effective for the removal of dyes from real textile effluents exhibiting 60% removal of Congo red, 53% removal of methyl orange and 74% removal of basic fuchsine. Freundlich isotherm and pseudo-second-order kinetics are best explained by the experimental data.
Thus, the present work is a successful attempt in exploring eco-friendly and economical option rather than expensive synthetic adsorbent material (Adegoke and Bello, 2015; Kyzas et al., 2013) for the adsorption of textile effluents. It will also provide benefit in recycling of waste material into valuable product following the principle of green chemistry.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author acknowledges the support from Fatima Jinnah Women University for providing the lab facility for conducting research activities.


