Abstract
Breakthrough experiments are essential for the characterization of the adsorption capacity and micropore volume of activated carbon respiratory cartridges and for the validation and determination of cartridge service life models. In an effort to gain better control over environmental conditions in breakthrough tests and to obtain reliable data, a novel experimental approach using a miniaturized (Mini) cartridge was designed to replicate a small section of a respiratory cartridge. The Mini device and the organic vapor respiratory cartridge were tested in single and parallel experiments where in the former, one filter was tested one at a time and in the latter both devices were exposed simultaneously to the same conditions. The Mini device gave comparable results to the 10% breakthrough times and adsorption capacities of the organic vapor cartridges. The reproducibility of the packed carbon bed of the Mini provided strong support for using the Mini in breakthrough experiments for the characterization of the activated carbon adsorption capacity and estimation of cartridge service life.
Keywords
Introduction
Respirators are commonly used to protect workers against the toxic inhalation of dust, fumes, and organic vapors (OVs) (National Center for Health Statistics, 2011). Among all the respirator masks, air-purifying respirators are the most widely used. The National Institute for Occupational Safety and Health and the Bureau of Labor Statistics reported that air-purifying respirators were used in 95% of all the establishments requiring respirators used in the United States following the 2001 survey (Doney et al., 2005). Typically, the preferred sorbent material for OV respirator cartridge is activated carbon due to its high porosity, low cost, and good adsorption capacity for many vapors (Bradley, 2011; Rodriguez-Reinoso, 2001). Determining the cartridge service life is a complex task because a limited number of End Service Life Indicators (ESLI) for OVs are available (Favas, 2005).
Therefore, mathematical models are recommended to estimate the time at which the cartridge reaches the saturation point or is no longer able to filter out the contaminant (Occupational Safety and Health Administration, OSHA, 1998). To use such prediction models, knowledge of both the sorbent capacity and breakthrough behavior are essential for the prediction of their performance (Wood, 1993a, 1994). This information can be obtained by characterizing the vapor concentration (or relative concentration) versus time profile of the effluent, termed “breakthrough curve” (Wood, 2002a; Wood and Stampfer, 1993). Even so, breakthrough curve experiments are laborious, time consuming, and the control of the environmental conditions can be challenging. Furthermore, these breakthrough curves depend simultaneously on the physical and chemical characteristics of the sorbent material (activated carbon) (Wood, 1992; Wood and Stampfer, 1993) and the adsorbate vapor (Freedman et al., 1973; Moyer, 1983; Nelson and Correia, 1976), presence of other contaminants (Lara et al., 1995), and the environmental conditions such as temperature, air flow, relative humidity (RH), and vapor contaminant concentration (Nelson and Correia, 1976; Nelson and Harder, 1972; Nelson et al., 1976; Ruch et al., 1972). Hence, controlling the environmental conditions to obtain reliable breakthrough curve data is essential to assess the cartridge service life model validity and to estimate its parameters within a range of the expected working conditions (DeCamp et al., 2004).
Most cartridge service life predictive models assume that each contaminant breakthrough curve is a symmetric sigmoidal curve. Yet, some studies have shown that for some contaminants, at high breakthrough fraction above 50%, the experimental data deviate from symmetry particular at low concentrations and at high humidity (Wood, 1993a, 2002b). Thus, obtaining reliable and accurate data from breakthrough experiments in controlled environmental conditions that closely match the working settings is desired.
Early works from Cohen et al. have described in a series of papers the use of a filter device, which they termed “small respirator carbon tubes (RCT)” with the aim to estimate cartridge service life in workplace conditions (Cohen et al., 1991a, 1991b, 1990; Cohen and Garrison, 1989). The RCT was filled with the same sorbent material contained in the respirator cartridge and was connected to a pump for laboratory testing and to a small personal pump for field testing. The breakthrough time and the bed residence model method (Ackley, 1985) were applied and compared favorably with the cartridge data for CCl4 at dry and humid conditions and for multiple exposures with n-hexane and pyrimidine. However, only a fraction of the breakthrough profile was measured (up to the 10% breakthrough fraction) rather than the entire breakthrough curve. This limits the applicability of the RCT method by missing important information, which is needed to assess the validity of their device throughout the entire filtration process.
In our previous paper (Janvier et al., 2015), the micropore structure of activated carbons of 10 commercially available respirator cartridges was characterized. It was demonstrated that breakthrough experiments, which were used to obtain adsorption isotherms from five OV substances of different vapor pressures at a concentration of 500 ppm and 40% of RH could be used to obtain the micropore volume of activated carbons. Using the OV approach, the adsorption capacity was successfully predicted.
To improve control over environmental conditions in breakthrough tests and to obtain reliable data, a novel experimental approach utilizing a miniaturized (Mini) cartridge was designed to replicate a small section of a respiratory cartridge. To evaluate whether the breakthrough curves obtained with the Mini were a suitable representation of the full size respirator cartridge, repeatability and reproducibility studies were conducted in single experiments (Mini and OV cartridge separately) and in parallel experiments (Mini and OV cartridge at the same time).
Materials and methods
Miniaturized (Mini) and OV cartridges
Figure 1 illustrates the Mini device and the vertical fall tube specially designed in house for this study. The Mini is made of stainless steel with a cylindrical collection container with an inside radius of 1.14 cm and a length of 5.00 cm. The packed carbon was kept in place by two wired meshed screens and filters (Texel, TRIBO 60 HJ, St-Elzéar-de-Beauce, QC) tightly screwed in at each end. These dimensions for a packed bed activated carbon filter ensured a linear flow throughout the Mini device.
Photograph of the miniaturized (Mini) cartridge used in this study and the vertical fall tube.
Structural properties of the activated carbon characteristics of the media and the experimental conditions.

OV: organic vapor.
OV micropore volume and B are derived from the Dubinin–Radushkevich equation using breakthrough experiments as described in Janvier et al. (2015).
Assumptions of the Mini
The approach of the Mini relies on two fundamental assumptions: (1) identical carbon bed density and (2) equivalent linear air through velocity to the OV Cartridge. To fulfill the first assumption, an equivalent amount of carbon obtained from the respiratory cartridge must be added to the Mini device. By considering that for a given area, the Mini should have the same carbon thickness and bed density as the OV cartridge, an equivalent amount (W) in grams of activated carbon was calculated using equation (1) and was transferred to the Mini device using the following equation

The second requirement is fulfilled by the estimation of an equivalent flow rate (Q) to the 24 l/min, which passes through the OV cartridge bed area. By assuming that for a given area of the Mini that the throughout linear air velocity for a plug flow through the bed is identical to that of the OV cartridge, the corresponding flow rate (
Design of breakthrough experiments
Complete breakthrough experiments of the Mini and the OV cartridge were conducted in single (Mini or OV Cartridge) and parallel experiments (Mini and OV Cartridge) to compare breakthrough data useful for the prediction of cartridge service life. A single experiment is defined as a breakthrough measurement performed with only one filter device, whereas in the parallel experiment, both devices (Mini and OV cartridges) are exposed simultaneously to the same contaminated airstream. The advantages of using the latter method are sustainability, time saving, and that it removes the experimental variables because both carbon beds are exposed at once to the same conditions. Thus, for this study, the parallel experiments are the preferred testing method. However, single initial experiments were performed to verify the repeatability and reproducibility of the test system.
This work consisted of four investigations: The first two involved corroborating the assumptions in the design of the Mini in terms of the equivalent weight and flow rate. The third investigation was reproducibility and repeatability study with selected solvents in single experiments, and the last one consolidated all the results in parallel experiments. The four experiments are listed below:
Reproducibility of the carbon bed thickness of the Mini. Flow rate experiments from 1.30 to 1.55 l/min for the Mini in dichloromethane (DCM). Single experiments of the Mini and OV cartridge tested in triplicate in n-hexane and DCM. Parallel experiments (Mini and OV cartridge simultaneously) conducted in DCM, n-hexane, methyl isobutyl ketone (MIBK), toluene, and m-xylene.
Experimental setup
A schematic diagram of the atmosphere generation system used for exposing the Mini and the OV cartridge is illustrated in Figure 2. A MNR (Miller-Nelson, HCS-501, Miller-Nelson Research, Inc., Dublin, Calif.) flow controller was used in this setup. The vapor gas introduced in the mixing chamber was generated with a syringe pump (KDS Legato 210, Holliston, MA). All solvents were HPLC grade from Sigma-Aldrich: DCM, MIBK, n-hexane, m-xylene, and toluene. The mixing chamber contained a fan, which allowed a uniform distribution of the airstream into the exposing chamber. The feeding rate of the exposure chamber was set to 30 l/min to generate a targeted concentration of ∼ 500 ppm with an overall ± 3% coefficient variation for the OV at 21 ± 1℃ and 40 ± 1% RH. The calibration of the generated vapor was performed by using calibration curves obtained with five concentrations points for each tested vapor. The temperature and humidity were controlled and monitored by means of a direct reading of a hygrometer. The OV cartridge and the Mini were either run in single or parallel experiments and were connected to separate vacuum pump lines, operating at 24 l/min for the cartridge and at the equivalent flow rate of 1.55 l/min for the Mini and were measured with TSI Mass Flow meter (4040, Shoreview, MN). The airstream was monitored by flow meters throughout the experiments. The inlet and the outlet vapor concentrations of the Mini and the OV cartridge were measured at 30 s intervals with a gas chromatograph (HP, 5890 Series II, Alto Palo, CA, USA) equipped with a flame ionization detector (FID) and a selection valve (Model A60, VICI, Houston, TX, USA). A deactivated silica column (40 cm × 0.32 mm I.D.) was attached to the injection port with a loop of 250 µl. The carrier (helium) flow rate was 4.6 ml/min, the detector makeup gas (nitrogen) flow rate was 30 ml/min, the hydrogen and airflow rates were 30 and 400 ml/min, respectively. The oven temperature was set at 200℃ and the FID temperature was at 250℃.
Schematic diagram of atmosphere generation system for the single and parallel experimental set-up of Mini and OV cartridge. OV: organic vapor.
The experimental activated carbon adsorption capacities (We exp) were measured gravimetrically after the downstream concentration (Cx) had reached ∼100% of the upstream concentration (Co). Prior to experimentation, empty OV cartridges and Mini were weighed and removed from cartridge total weight to determine the initial activated carbon weight. A blank experiment (no vapor) of the OV cartridge was run for 8 h at 40% RH, at 24 l/min, and showed that only 0.7% of water was adsorbed, which was judged negligible.
The calculated adsorption capacity (We calc) was approximated by using the midpoint of the breakthrough time at 50% saturation (tb50%) with (Wood, 1994)

Commonly, the time at 10% saturation (tb10%) is used to define the cartridge service life and it was also obtained from the breakthrough curve measurements of the Mini and OV cartridge. The carbon bed length, which corresponds to the thickness of the respiratory cartridge, was measured by using a caliper. Both values (tb10% and We) were used to compare Mini versus OV cartridge filtering capacity.
Predictive model
Analysis of breakthrough curves of the OV cartridge and the Mini was performed using a simple semi-empirical model, the Wheeler–Jonas equation (Jonas et al., 1975), which relates the breakthrough time tbin minutes with


A plot oftbversus 
Equations (5) and (6) were used to simulate breakthrough curve. It is noted that A is equal to
Results and discussion
Reproducibility of the carbon bed thickness of the mini
The carbon loading of Mini is shown in Figure 1 and mimics the snow filling method (Jones and Smith, 1992), a typical protocol used by manufacturers. This method allows reproducibility of packing density, absence of voids, and even distribution of carbon (Jones and Smith, 1992). The equivalent amount of activated carbon calculated with equation (1) was added to the Mini to match the packed bed density of the OV cartridge and resulted in a carbon-bed length thickness of 1.66 ± 0.05 cm for N = 23. This value is in good agreement with the reported thickness of the OV cartridge of 1.65 cm listed in Table 1. This result indicates that the carbon bed of the Mini is packed uniformly to a maximum density similar to the corresponding cartridge, thus validating the packing method and the carbon bed density assumption. Though, it is well known that a small carbon bed may result in uneven packing due to channel formation (Schwartz and Smith, 1953), this was not observed in the Mini.
Validation of the flow rate for the mini
The equivalent flow rate calculated with equation (2) was verified by varying the flow rate of the Mini from 1.30, 1.35, 1.45, and 1.55 l/min in DCM at 500 ppm while the OV cartridge flow rate remained at 24 l/min. As described in the literature (Wood and Moyer, 1991), the Wheeler–Jonas equation presented in equation (4) is essentially flow rate dependent; therefore, a change in the flow rate is expected to affect the breakthrough profile.
Overall, the equivalent flow rate (1.55 l/min) of the Mini provided a good estimation of the breakthrough curve of the respirator cartridge as shown in Figure 3. In comparison with the smallest flow rates (1.30, 1.35, and 1.45 l/min), the breakthrough profile of the Mini using the equivalent flow rate of 1.55 l/min coincides well with the 10 and 50% breakthrough times of the full-size respirator cartridge. This result indicates that the estimated equivalent flow rate of the Mini is valid within a certain range. The difference in the shape and steepness of the breakthrough curves between the two filtration media is probably due to the difference in the cartridge configuration, which produces distinct adsorption kinetics inside the carbon bed (Wood, 2002a).
Effect of the flow rate on the breakthrough curves of the Mini for DCM. As a reference, the breakthrough curve for the OV cartridge at 24 l/min was included in this figure. DCM: dichloromethane; OV: organic vapor.
Reproducibility and repeatability study
Results of the breakthrough times of the repeatability and reproducibility study of the Mini and OV cartridge.

OV: organic vapor; RSD: relative standard deviation of the concentration.
tb10% is the 10% breakthrough time.
tb50% is the 50% breakthrough time.
tb100% is the 100% breakthrough time.
conc. is the concentration.
The resulting breakthrough curves are presented in Figure 4 for the Mini (opened blue symbols) and OV cartridge (closed red symbols) for DCM and in n-hexane. The superimposition of the triplicate curves of both Mini and OV cartridge using DCM and n-hexane corroborates the stability and reliability of the experimental setup. The sigmoidal shape of the breakthrough curves in DCM and h-hexane for the Mini confirms the even packing of the carbon bed, which implies that the diffusion of the vapor inside the filter device is uniform. On the other hand, the breakthrough curves of the OV cartridge in Figure 4(d) exhibits some spreading in n-hexane and deviates from the sigmoidal shape. The spreading in n-hexane is more pronounced for the respirator cartridge than the Mini, which is probably due to the difference in the cartridge configuration. Moreover, the adsorption rate being more rapid for low boiling points, and volatile compounds than higher boiling points contaminants may enhance this effect (Stampfer, 1982).
Repeatability and reproducibility of the single experiments for Mini and OV cartridges in 500 ppm DCM and n-hexane at 40% RH and at flow rates of 24 l/min for the cartridge and 1.55 l/min for the Mini. (a) DCM, (b) n-hexane, (c) DCM, and (d) n-hexane. DCM: dichloromethane; OV: organic vapor.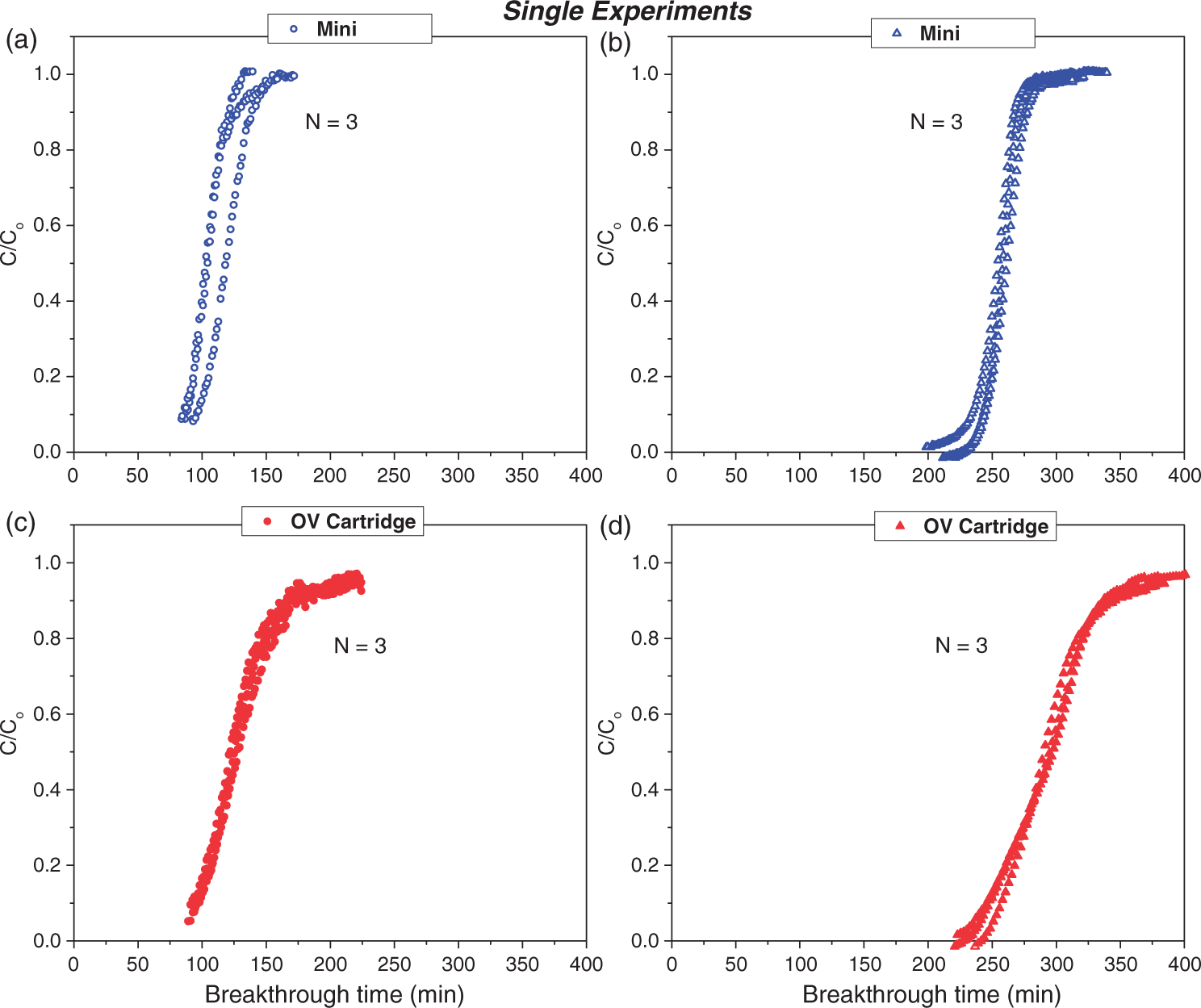
Breakthrough curves in parallel experiments
Once the reliability of the test system was confirmed, parallel experiments with both the Mini and the OV cartridge were tested. The breakthrough curves of the Mini and OV cartridge obtained with single and parallel experiments using DCM and n-hexane are compared in Figure 5. The superposition between the breakthrough curves of the single and parallel experiments of the Mini and the OV cartridge shows that there are no differences between the two experimental approaches. This is an important factor because it ensures that the Mini is exposed to identical conditions as the OV cartridge, therefore allowing an accurate comparison between the Mini and the OV cartridge in real time.
Single (black) versus parallel (gray) experiments for the average breakthrough curves in DCM and n-hexane for the Mini (dotted lines) and the respirator cartridges (dashed lines) at 500 ppm and 40% RH.
Figure 6 shows all the breakthrough curves of the parallel experiments with the Mini (open blue symbols) and the OV cartridge (closed red symbols) for N = 3. Both Mini and OV cartridge start saturation at the same time and as the saturation fronts advance, the shape of the breakthrough curves of the Mini remains sigmoidal and symmetrical. Overlapping of the curves occurs up to the range of 0.3–0.9 saturation ratio of the upstream concentration. Moreover, as the volatility of the solvent vapor increased in the order of DCM >n-hexane > toluene > MIBK > m-xylene, the gap between the breakthrough profiles of the Mini and the OV cartridge increased. Similarly to single experiments, skewed breakthrough curves of the OV cartridge in parallel experiments are observed for all the contaminants especially for less volatile contaminants. Conversely, this trend is not observed in the breakthrough profiles of the Mini. The nonsymmetry of the breakthrough curve in respirator cartridges has been reported by others (Wood, 2002b) and is thought to originate from the heterogeneity of adsorption sites in the micropores of the activated carbon, which results in changes in the adsorption rates and/or mechanisms as the adsorption front of the vapor (Wood, 1993b, 2002b).
Experimental data of parallel experiments with the Mini (blue opened symbols) and the respirator cartridge (red closed symbols) and simulated breakthrough curves for DCM, n-hexane, MIBK, toluene, and m-xylene at ∼ 500 ppm and 40% RH. (a) DCM, (b) n-hexane, (c) MIBK, (d) toluene, and (e) m-xylene. DCM: dichloromethane; MIBK: methyl isobutyl ketone.
The difference in the shape of the breakthrough curve between the Mini and the OV cartridge further challenges the notion that skewed breakthrough curves of respirator cartridge are a result of the kinetic of the adsorber, which is influenced by the volatility of contaminants (Wood, 1993a, 2002b). Rather, the geometric configuration of the cartridge or the face velocity and the nonlinear air through profile throughout the cartridge seem to be the dominant factor affecting skewed breakthrough curves. A study evaluating the adsorption of OVs onto activated carbon has shown that surface diffusion or face velocity trumps over pore diffusion (Shiue et al., 2011). Therefore, as a consequence of the smaller surface of the Mini, the diffusion of the vapor molecule in the pores of the activated carbon is increased and reaches equilibrium faster as shown by the steeper and symmetrical breakthrough curves of the Mini compared to those of the OV cartridge in Figure 6.
Figure 7 illustrates the flow pattern process in the two types of carbon beds: the Mini and OV cartridge. The geometry of the bed diameter of the Mini is cylindrical and exhibits a plug flow for which the face velocity is uniform and is represented by a large arrow. The OV cartridge, which has a trapezoid shape, has an average air-through flow velocity represented with a large arrow in Figure 7. Although the average flow throughout the cartridges is considered to be a constant flow across the section of the cartridge (large arrow) like in the Mini, in reality it is not, and the nonlinear flow profiles will affect the shape of breakthrough curve. Furthermore, the breakthrough curves of the OV cartridge in Figure 6 show that it becomes increasingly hard to achieve adsorption equilibrium at 100% saturation and consequently it will take a longer time to saturate as evidence by the tailing. Different zones in the carbon bed of the OV cartridge will have different velocities, especially around the wall of the bed. Lodewyck’s research team also reported a wall effect in activated carbon by visualization with X-ray microtomography for cylindrical configuration with CH3I, CCl4, and water mixtures (Lodewyckx et al., 2006; Léonard et al., 2006). A constant wave front was observed for the CCl4 study and a radial adsorbate concentration profiles were observed for different activated carbon bed thickness with the CH3I study. This phenomenon may be important to consider since most prediction models use linear velocity for vapor gas and neglect radial velocity profiles within the carbon beds. Nonetheless, in terms of characterization of the activated carbon from breakthrough curves, the Mini may prove to be a better choice because it may diminish the influence of the flow pattern.
Comparison between the flow profiles of the Mini and the OV cartridge. The large arrow is the average flow profile. OV: organic vapor.
Adsorption capacity
The experimental (We exp) and calculated (We calc) adsorption capacities of the Mini and OV cartridge for the five OVs tested are presented in Figure 8. Overall, the Weexp are smaller than the Wecalc values across the Mini and the OV cartridge. A possible explanation for this discrepancy may be due to the preadsorbed water on the activated carbon and in the air stream, which is desorbed by the more strongly adsorbed hydrophobic compound (Lodewyckx and Vansant, 1999; Nelson et al., 1976) resulting in the appearance of a low Weexp compared to the Wecalc. This is in agreement with another study, which described the breakthrough behavior difference of low boiling points solvents by stipulating they may have less adsorption affinity and that they are adsorbed less than contaminants at higher boiling point (Stampfer, 1982). Therefore, the calculated adsorption capacity obtained from the midpoint of the breakthrough curve may be more reliable than the gravimetric method because it is not influenced by the presence of water.
Comparison between the Mini and OV cartridge of the experimental and calculated adsorption capacity. OV: organic vapor.
When comparing the Mini versus OV cartridge, the capacity values of the Mini were slightly lower than that of the OV cartridge but in most cases, the differences were either small or showed no significance difference within experimental errors (see Table 4 in Appendix 1).
Breakthrough time
Even with the assumption that the Mini and the OV cartridge exhibit the same linear air through velocity, the difference in the flow rates between the carbons beds did not significantly affect the 10% breakthrough times. Figure 9 compares in (a) 10% breakthrough times and in (b) 50% breakthrough times between the Mini and OV cartridge. Though the 10% breakthrough times of the Mini were slightly smaller than the values of the OV cartridge, they were within experimental errors (less than 10 ppm difference). Whereas, the 50% breakthrough times of the Mini showed a greater variability from the OV cartridge, especially for toluene and m-xylene. This difference in these values is expected because the breakthrough curves of the Mini deviated from the OV cartridge at 0.5 of the saturation fraction (see Figure 6) and the OV breakthrough curves were asymmetric for the OV cartridge.
Comparison of breakthrough times between the Mini and the OV cartridge at 10% breakthrough time and 50% breakthrough time. OV: organic vapor.
The general trend observed both in the single and parallel experiments is that the Mini always reaches complete saturation before the OV cartridge. This implies that the data obtained with the Mini will have a safety factor when estimating the cartridge service life because the tb10% will always be slightly lower than the ones obtained with the OV cartridge data.
Simulation
Experimental parameters of the Mini used in the breakthrough curve simulation.
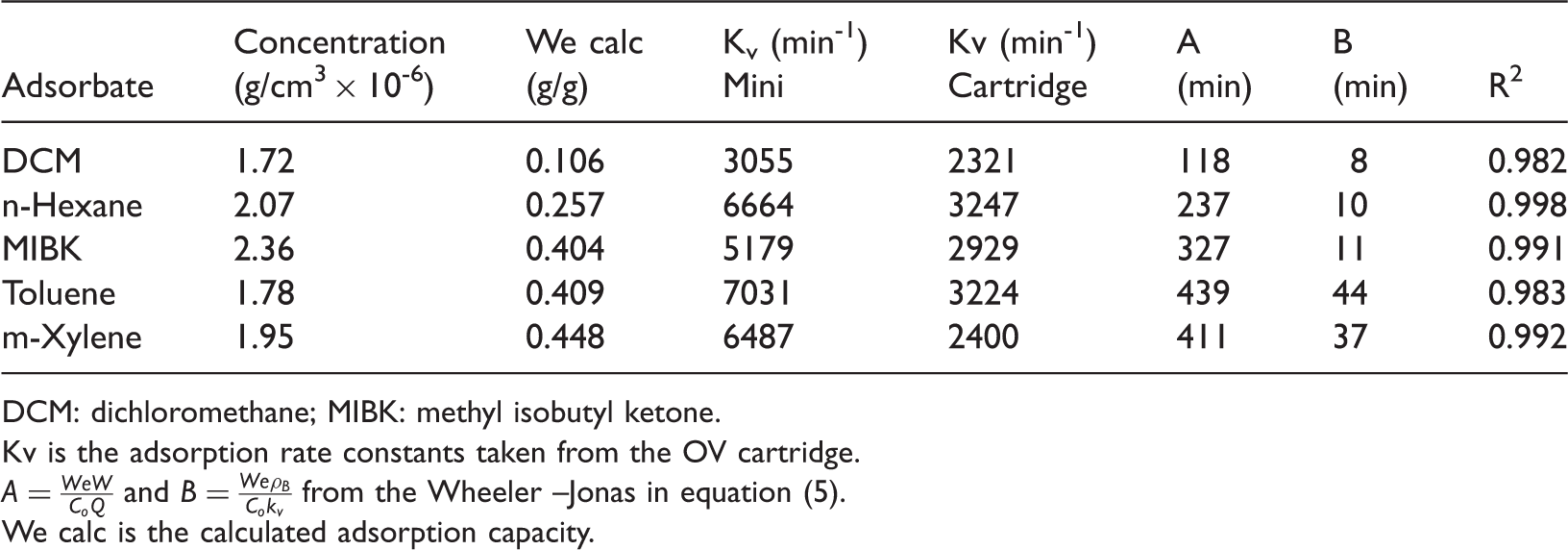
DCM: dichloromethane; MIBK: methyl isobutyl ketone.
Kv is the adsorption rate constants taken from the OV cartridge.
We calc is the calculated adsorption capacity.
Conclusions
The Mini approach discussed in this work is the first attempt to systemically evaluate the suitability of this novel experimental approach where the Mini represents a section of the full-size respirator cartridge and is tested in parallel breakthrough experiments with the corresponding respirator cartridge. This study demonstrated that assumption of equivalent amount of activated carbon of the Mini on the basis of having the same packing density and bed thickness of the respirator cartridge was justified. Whereas the equivalent flow rate showed some discrepancies in terms of matching breakthrough curves between the Mini and the respirator cartridge due to different flow profiles inside the carbon bed caused by geometric configuration of the cartridge. Nonetheless, the adsorption capacities and 10% breakthrough times of the Mini gave reasonably good estimation of the OV cartridge performance and its applicability may be useful in obtaining data for the model prediction for cartridges service life and characterization of the sorbent capacity.
Further work is in progress to evaluate the Mini with several other respirator cartridges and contaminants to gain a better understanding of the asymmetry of the breakthrough curve observed with OV cartridges and the underlying mechanism of the adsorption of OV onto activated carbon.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge project grant 0099-5080 from IRSST (Institut de recherche Robert-Sauvé en santé et en sécurité du travail) and F.J acknowledges financial support from IRSST, FRSQ (Fonds de recherche du Québec-Santé), and ÉSPUM (École de santé publique de l’Université de Montréal).
