Abstract
In this paper, scanning electron microscopy, low-temperature N2 adsorption and CH4 isothermal adsorption experiments were performed on 11 coal samples with Ro,max between 0.98 and 3.07%. The pore structure characteristics of coals (specific surface area, total volume distribution) were studied to assess the gas adsorption capacity. The results indicate that there is significant heterogeneity on coal surface, containing numerous channel-like pores, bottle-shaped pores and wedge-shaped pores. Both Langmuir volume (VL) and Langmuir pressure (PL) show a stage change trend with the increase of coalification degree. For different coalification stages, there exist different factors influencing the VL and PL values. For low-rank coals (Ro,max < 1.1%), the increase of VL values and decrease of PL values are mainly due to the abundant primary pore and fracture within coal. For middle-rank coals (1.1% < Ro,max < 2.1%), the moisture content, vitrinite content and total pore volume are all the factors influencing VL, and the reduction of PL is mainly attributed to the decrease of moisture content and inertinite content. Meanwhile, this result is also closely related to the pore shape. For high-rank coals (Ro,max > 2.1%), VL values gradually increase and reach the maximum. When the coal has evolved into anthracite, liquid hydrocarbon within pore begins pyrolysis and gradually disappears, and a large number of macropores are converted into micropores, leading to the increase of specific surface area and total pore volume, corresponding to the increase of VL. In addition, the increase of vitrinite content within coal also contributes to the increase of VL. PL, reaches the minimum, indicating that the adsorption rate reaches the largest at the low pressure stage. The result is mainly controlled by the specific surface area and total pore volume of coal samples. This research results will provide a clearer insight into the relationship between adsorption parameters and coal rank, moisture content, maceral composition and pore structure, and it is of great significance for better assessing the gas adsorption capacity.
Introduction
Coal is a natural organic rock with complex surface properties and porous structures. Because of the existence of unsaturated energy on the surface of pore structure within coal, these kinds of energy can be combined with nonpolar gas molecules to produce force, called Van der Waals force. The effect of Van der Waals force makes the gas adsorption on coal surface, producing the gas adsorption effect. There are many factors influencing the gas adsorption, including the coal rank, temperature, pressure, moisture content, maceral composition and pore structure, etc. (Crosdale et al., 2008).
It is widely accepted that coal rank is one of the main factors influencing gas adsorption, and there is a U-shaped relationship between coal rank and adsorption capacity. Temperature and pressure are also indicators influencing adsorption capacity. There is a positive correlation between adsorption capacity and pressure, a negative correlation between adsorption capacity and temperature (Meng et al., 2016; Pan et al., 2015a). In addition, many scholars have studied the influence of other factors on the adsorption ability. Hou et al. (2016) investigated the influence of maceral composition on adsorption capacity in low-rank coals, proving that the adsorption capacity showed a positive correlation with vitrinite, a negative correlation with inertinite. Jian et al. (2015) also analyzed the relationship between adsorption capacity and maceral composition in low-rank coals, but they got the inconsistent conclusion. They indicated that the adsorption capacity showed a “three-step-style” change with the increase of vitrinite and inertinite. Liu et al.(2016) found that adsorption capacity was mainly controlled by micropores in middle-rank coals. With the increase of coal rank, the adsorption capacity and micropores showed positively proportional (Wang et al., 2014). It is generally accepted that there exists adsorption competitiveness between moisture molecules and gas molecules (Busch and Gensterblum, 2011; Romanov and Soong, 2008), but this competitiveness gradually weakens with the increase of coalification degree (Liu et al., 2016), due to affinity to water in high-rank coals becoming smaller compared with low-rank coals (Gensterblum et al., 2014). In conclusion, although the influencing factors (including the coal rank, temperature, pressure, moisture content and pore structure) of Langmuir volume (VL) have been intensively studied, the research results exist contradictorily, while the effect of these factors on Langmuir pressure (PL) has not been comprehensively evaluated. It is generally known that PL is also an important index for gas adsorption. Thus, research on the relationship between PL and comprehensive factors is of great importance for the prediction of the adsorption rate of gas. In addition, previous studies have focused on a single coal rank, and few studies refer to the wider coal ranks.
In this paper, we used 11 coal samples with different coalification ranging from fat coal to anthracite to carry out our studies. The coal samples were investigated by proximate analyses, maceral analyses, scanning electron microscopy (SEM), low temperature N2 adsorption and CH4 isothermal adsorption. Combined with these experiment results, the relationship between adsorption parameters (VL and PL) and coal rank, moisture content, maceral composition and pore structure were analyzed comprehensively in different metamorphic stages. Meanwhile, the evolution characteristics of pore structure in coal were characterized from the angles of macroscopic and microscopic by using image analysis and gas adsorption analysis. This analysis allows us to more clearly understand the evolution characteristics of the pore structure, and gets the influencing factors for gas adsorption with different rank coals.
Experimental section
Experimental samples
Eleven coal samples with different ranks, from Jiulishan and Guhanshan in Jiaozuo mining (four samples), 8th and 10th mines in Pingdingshan mining (four samples), Hebi mining (one sample), Chaohua in Zhengzhou mining (two samples) were collected. The coals from Pingdingshan and Hebi mine area are of low-middle rank, those from Zhengzhou and Jiaozuo mine areas are of middle-high rank. All selected coal samples were immediately sent to the testing center after classification and wrapping.
Experimental methods
Proximate analyses of coal samples were performed using the automatic proximate analyzer TGA 701 according to Chinese National standards GB/T 30732-2014. The Chinese National standards GB/T 6948–1998 and GB/T 8899–1998 were respectively applied in maximum vitrinite reflectance (Ro,max) measurement and maceral analyses. Scanning electron microscopy (SEM) measurements were accomplished by S-3400 N SEM, following the GB/T 20307-2006.
Low-temperature N2 adsorption experiments were done by using the ASAP-2020 surface area analyzer equipment on 60–80 mesh samples (ASTM standard). The coal samples were outgassed at 100℃ for 12 h under a vacuum. The N2 adsorption experiment at 77 K was completed at relative pressure (P/Po) ranging from 0.01 to 0.994.
The CH4 isothermal adsorption experiment of coal samples was measured by WY-98B equipment following the GB/T 19560-2004 standard. The following is the specific experiment process: Approximately 30 g of coal samples were weighed, and then dried at 105℃ for 2 h in a vacuum drying oven to remove water. After the coal samples were cooled to room temperature, the samples were placed in a coal sample tank for evacuation at 70℃ for 8 h under vacuum (<4 Pa) prior to test. The experimental temperature and maximum equilibrium pressure were set to 30℃ and 6 MPa.
Experimental results
Fundamental characteristics of coal
Results from proximate analyses and maceral composition identified of coal samples.

Note. -: no data
M: moisture; A: ash yield; V: volatile matter; d: dry basis; ad: air-dry basis; daf: dry-ash-free basis; FCad: fixed carbon; V: vitrinite; I: inertinite; E: exinite.
SEM results
SEM observation is one of the useful tools for describing the pore surface morphology and analyzing the discrepancy of pore structure of coal samples. SEM can magnify the coal samples 100,000 times. In this paper, all the tested samples are enlarged 1000 times. The SEM results are shown in Figure 1. Pore surface morphology of coals with different coalification degree show a significant difference, indicating that coal samples surface exist obviously heterogeneity. Samples P10-Y, JG-Y and JJ-Y are relatively smoothness in coal surface (Figure 1(a), (h) and (i)), and samples P8-G, P8-Y2, HB-Y, ZCH-G1, ZCH-G2 and JG-G are relatively roughness, manifesting that the latter possess more complex pore structure characteristics.
SEM images of the coal samples. SEM: Scanning electron microscopy.
Results of low temperature liquid nitrogen and CH4 adsorption method.

VL: Langmuir volume; PL: Langmuir pressure.
Pore structure characteristic and CH4 isothermal adsorption results
In this paper, we classified pores as micropores (<10 nm), transition pore (10–100 nm), and mesopores (>100 nm) (Hodot, 1961). Among them, micropores and transition pore are called adsorption pore, providing the main space for gas adsorption, while mesopores are called seepage pore, offering the main paths for gas diffusion and flow (Li et al., 2015; Squires and Quake, 2005; Yao et al., 2009). Low-temperature N2 adsorption isotherms for the coal samples are shown in Figure 2, and the results of pore structure parameters can be seen in Table 2. BET and BJH are separately used to calculate specific surface area (SSA), total pore volume (TPV) and average pore size of coal samples. The average pore size of coal samples ranges from 8.758 to 36.582 nm. SSA of the coal samples is between 0.0101 and 1.0494 m2/g with an average of 0.4475 m2/g. SSA of samples P8-G, ZCH-G1, ZCH-G2 and JG-G are higher than that of samples P10-Y, JG-Y and JJ-Y. From the results of SEM, the surface of samples P8-G, ZCH-G1, ZCH-G2 and JG-G are rougher than that of samples P10-Y, JG-Y and JJ-Y. The rougher the coal surface is, the greater the SSA, which is in agreement with Nie et al. (2015). TPV of coal samples is from 0.00231 to 0.0086 cm3/g with an average of 0.00709 cm3/g with almost all of that pore volume attributable to transition pores. With the increases of coal rank, the percentage of micropores volume gradually increased, and those for transition pores and mesopores volume doesn't change appreciably at first and then gradually decrease (Table 2). The TVP shifts from transition pores and mesopores to micropore and transition pores with the increase of coalification.
Low temperature N2 adsorption isotherms for the coal samples. (a) Low-rank coal samples (Ro,max < 1.1%); (b) Middle-rank coal samples (1.1% < Ro,max < 2.1%); (c) High-rank coal samples (Ro,max > 2.1%).
In this paper, we use the Langmuir adsorption equation to calculate the adsorption parameters (VL and PL) (Langmuir, 1918)
CH4 adsorption isotherms of coal samples.
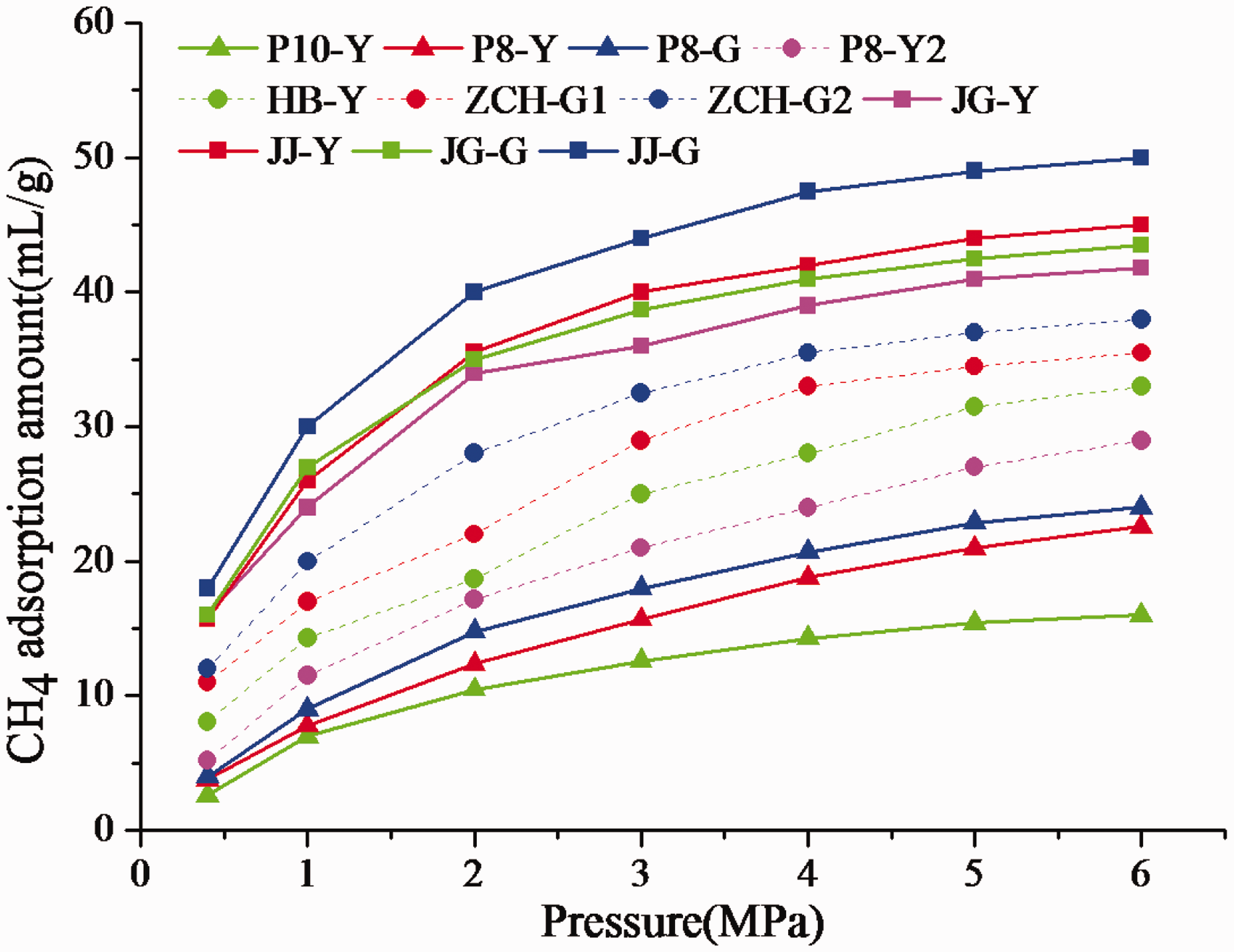
Discussion
Influence of coalification on Langmuir parameters
The relationships between Langmuir parameters (VL and PL) and coal rank are shown in Figure 4. These results indicate that both VL and PL have a stage change with coal rank. As seen from Figure 4(a), VL shows a linear increase before the Ro,max < 1.1%. For coal with lower coalification, the primary pores are relatively abundant, which provides the space for gas adsorption, corresponding to the increase of VL. But it can be seen that the VL is smaller compared to the other stages (Figure 4(a)). It is mainly ascribed to the existence of numerous oxygen-containing functional groups within coal during this stage (Billemont et al., 2014; Hao et al., 2013; Jin et al., 2014), which has an inhibitory effect on gas adsorption (Liu et al., 2016), leading to the smaller VL. For PL, these values are relatively larger, suggesting that the desorption capacity is stronger (Ro,max < 1.1%) (Figure 4(b)). This is because coal contains abundant primary fracture during this stage, and it can provide a channel for the gas desorption, resulting in the higher desorption rate at low pressure stage.
Relationship between Langmuir parameters and coal rank: (a) VL; (b) PL.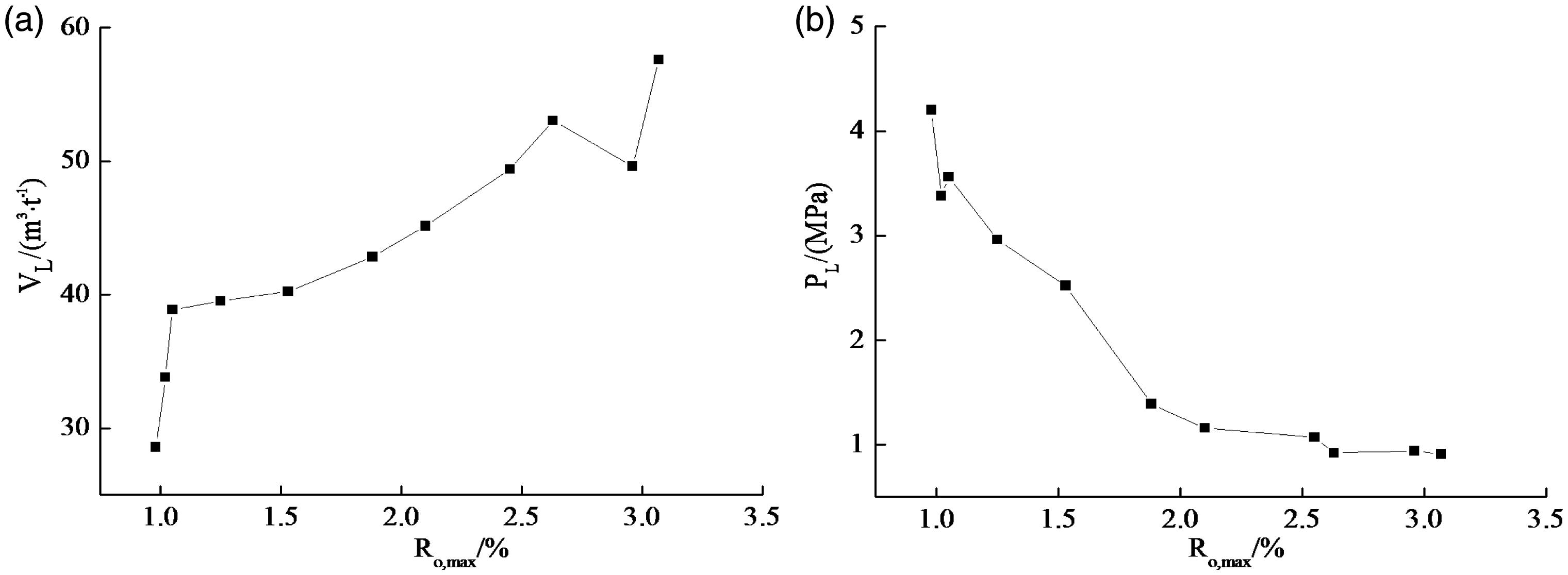
When Ro,max is from 1.1% to 2.5%, VL shows monotonous increase. There are many channel-like pores, bottle-shaped pores and wedge-shaped pores in coal surface (Figure 1(c) to (e) and (g)), which make it become the stronger heterogeneity, leading to the increase of VL. Conversely, PL shows a polynomial decay trend, indicating that the desorption capacity of coal gradually reduces. With the increasing coalification degree, the compaction effect of coal occurs and intensifies, then the micropores and microcracks are gradually formed (Liu et al., 2016), reducing the channel for gas flow, which leads to the decrease of desorption rate. For the anthracite coal stage (Ro,max > 2.5%), both VL and PL reach the maximum and the minimum, respectively.
Influence of moisture content on Langmuir parameters
Correlational analysis between Langmuir parameters (VL and PL) and moisture content are performed in Figure 5. For the low-middle coal rank stage (Ro,max < 2.1%), VL steadily increases with decreasing moisture content (Figure 5(a)). The decrease of moisture content can provide the sites for gas adsorption (Nie et al., 2016), leading to the increase of VL. PL shows a same change trend with moisture content which exhibits the monotone decreasing (Figure 5(b)). With the decrease of moisture content, the adsorption ability of coal is enhanced, which leads to the adsorption rate of coal at the low pressure gradually increased.
Relationship between Langmuir parameters and moisture content: (a) VL; (b) PL.
For the higher metamorphism degree (Ro,max > 2.1%), there shows roughly the same change trend between VL and moisture content which present sharply increase (Figure 5(a)). Due to the increase of fracture within coal particles during this stage, formation water flowing into fracture leads to the enhancement of moisture content (Zhang and Ma, 2008). According to the literature (Crosdale et al., 2008; Guo et al., 2015), the moisture content shows the negatively correlated with VL, but this paper presents the opposite conclusion. With the increase of coalification, the characteristic of the pore structure would be a dominant factor for the gas adsorption (Gareth et al., 2007; Levy et al., 1997; Liu et al., 2015), and the influence of moisture on adsorption capacity gradually weakens (Yao et al., 2009). Therefore, moisture content is not the main factor that influences gas adsorption during the stage. PL has a little change with the increase of moisture content and its value reaches the minimum, indicating the lower desorption rate during this stage. The reason for this phenomenon is that moisture content can be into the pore and fracture, producing the capillary force, thereby hindering the channel for gas flow (Nie et al., 2015), leading to the decrease of desorption rate.
Influence of maceral compositions on adsorption parameters
As shown in Figure 6, when Ro,max<1.1%, the vitrinte gradually decrease with the increase of VL and the reduction of PL. With the increasing coalification (Ro,max > 1.1%), the vitrinite content in coal is almost proportional to VL (Figure 6(a)), which is in agreement with Lamberson and Bustin (1993) and Hou et al. (2016). Vitrinite contains higher SSA and TPV of micropores (Gan et al., 1972; Unsworth et al., 1989), which provide the sites for gas adsorption (Harris and Yust, 1976; Walker et al., 2001), leading to the increase of VL. The PL shows the negative correlation for vitrinite with the increase of coalificaiton degree (Figure 6(b)). Due to the low porosity of vitrinite, the vitrinite increases with Ro,max increased, making that porosity of coal become more and more low, leading to the poor connectivity between pores. Namely, the higher the vitrinite content is, the more difficult the gas desorption in low pressure. Therefore, it is not conducive to gas desorption.
Relationship between Langmuir parameters and vitrinite content: (a) VL; (b) PL.
Relationships between VL, PL and inertinite content show the complex change trend during Ro,max < 1.1% (Figure 7). When Ro,max > 1.1%, a negative correlation between VL and inertinite content (Figure 5(a)) is found, and this result was supported by Clarkon and Bustin (1996), Crosdale and Basil (1998), Clarkson and Bustin (1999) and Mastalerz et al. (2004). Unsworth et al. (1989) indicated that inertinite has more macroporosity and less microporosity and there is positive correlation between adsorption capacity and micropores. So, the gas adsorption capacity gradually reduces with the increase of inertinite content. PL values and inertinite content keep almost the same change trend during this stage (Figure 7(b)). Namely, the smaller the inertinite content is, the larger the gas adsorption rate at the low pressure stage.
Relationship between Langmuir parameters and inertinite content: (a) VL;(b) PL.
Influence of pore structure parameters on adsorption parameters
The impact of SSA and TVP on VL and PL are shown in Figure 8. It can be seen that when Ro,max < 1.1%, VL values increase with increasing SSA and decreasing TVP (Figure 8(a) and (c)). For the low coalification degree, the increase of SSA is due to abundant micopores and mespores, and the reduction of TVP is attributed to a large amount of bitumen, which can be block pores (Gürdal and Yalçın, 2001; Yu, 1992), leading to the reduction of TVP. So, the increase of VL during this stage is mainly contributed to abundant micopores and mespores. When Ro,max is between 1.1 and 2.1%, VL increase with the slight decrease of SSA and the sharply increase of TVP. During the process of coalification, with the increase of geothermal temperature and pressure, the polycondensation of coal molecules occurs and accompanies with complex chemical and physical changes generally (Radlinski et al., 2004), leading to the reduction of SSA. Meanwhile, the various oxygen-containing functional groups and aliphatic side chains fall out, forming small-molecule compounds, i.e. H2O, CO2, and CH4, which during this stage the TVP (Pan et al., 2015b). Thus, the increase of VL during this stage is due to the fall out of oxygen-containing functional groups and aliphatic side chains, leading to the increase of pore volume. When Ro,max > 2.1%, VL reaches the maximum with the obviously increase of SSA and no significant change of TVP values with it reached the maximum. When the coal has evolved into anthracite, liquid hydrocarbon within pore begins pyrolysis and gradually disappears, and a large number of macropores are converted into micropores (Gareth et al., 2007; Levy et al., 1997; Liu et al., 2015; Mathews and Sharma, 2012), leading to the increase of SSA. During this stage, the increase of VL is due to the comprehensive effect of SSA and TVP in coal.
Relationship between Langmuir parameters and pore structure parameters: (a) VL versus SSA; (b) PL versus SSA; (c) VL versus TPV; (d) PL versus TPV. SSA: specific surface area; TPV: total pore volume.
When Ro,max < 1.1%, PL decreases with the SSA increase and TPV reduction (Figure 8(b) and (d)). With the increase of coalification (Ro,max = 1.1–2.1%), PL is linear downward corresponding to the no obviously change of SSA and the increase of TPV. The decrease of PL represents the increasing CH4 adsorption rate at low pressure stage. For the middle-rank coal, there are many channel-like pores, bottle-shaped pores and wedge-shaped pores in coal surface (Figure 1(c) to (e) and (g)), and it can offer more adsorption sites for CH4 (Liu et al., 2016), which lead to the increase of adsorption rate. When Ro,max > 2.1%, the reduction trend of PL tends to flat with reached the minimum and the SSA and TPV reach the maximum. Both SSA and TPV provide more sites and larger space for gas adsorption, leading to the increase of adsorption rate.
Conclusion
In this paper, coal rank, moisture content, maceral composition, pore structure of 11 coal samples were analyzed, in response to the CH4 adsorption behavior. The results shown that:
Both VL and PL show a stage change trend with the increase of coalification degree. There are different influencing factors on VL and PL in different coalification stages. For the low-rank stage (Ro,max < 1.1%), the increase of VL and decrease of PL are mainly due to the abundant primary pore and fracture in coal. But the VL values are the lowest compared to middle-high rank stage. These results mainly attribute to the oxygen-containing functional groups in coal. For the middle-rank stage (1.1% < Ro,max < 2.1%), there exist three reasons that lead to an increase in VL values. First, the decrease of moisture content can provide the sites for gas adsorption. Second, the increase of vitrinite produces the higher SSA and TPV of micropores. Third, the various oxygen-containing functional groups and aliphatic side chains fall out, which enlarge the TPV. For PL, it shows linear decay. This result is mainly influenced by the reduction of moisture content and inertinite. In addition, it is closely related to the pore shape. For the high-rank stage (Ro,max > 2.1%), the VL values gradually increase and reaches the maximum. There are two reasons for this result. One is vitrinite content increased. The other is influenced by the combined effects of SSA and TPV. PL tends to flat and reaches the minimum, indicating that the adsorption rate reaches the largest at the low pressure stage. This result is mainly affected by the SSA and TPV.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China(41430640).
