Abstract
In order to reduce the toxic effect on solid oxide fuel cells performance caused by biogas contaminated with hydrogen chloride and hydrogen sulphide, the purification of biogas is essential. Adsorptive gas purification is a highly auspicious technology to provide pollution-free biogas for solid oxide fuel cell-based power units. In this work the authors examined the influence of different parameters onto the adsorption capacity of three commercially available sorbents. Experimental runs in a laboratory glass downflow fixed-bed reactor were carried out to analyse the adsorption capacity of a potassium carbonate impregnated activated carbon and two sorbents based on a mixture of aluminium oxide and silicon dioxide. Hydrogen chloride removal was accomplished with the impregnated activated carbon and metal oxide-based sorbents. Hydrogen chloride adsorption capacity was analysed under space velocities 8000 and 16,000 h−1. In addition, the effect of a hydrogen chloride inlet concentration of 100 and 1000 ppmv was investigated. Furthermore, pellets in the size of 3–4 mm in diameter were crushed into a fraction between 500 and 1000 µm to investigate the influence of particle size on hydrogen chloride adsorption capacity. Additionally, the combined adsorption of hydrogen chloride and hydrogen sulphide was realized using the impregnated activated carbon. The experimental runs and the results obtained in this work provide useful data for designing an adsorption reactor to clean up biogas and optimizing the process.
Introduction
The high interest in alternative fuels and power sources is the result of the contemporary demand for environmentally friendlier and more efficient power production (Dincer and Zamfirescu, 2014). Despite being one of the oldest energy conversion technologies, the study of fuel cells for commercial use only began within the last decade. As can be seen all around the world, the combustion of fossil fuels has resulted in severe air pollution and extensive exploitation of the world’s oil resources. Additionally, to being hazardous for many species, pollution is indirectly causing the change of our world’s atmosphere (global warming) (Turco et al., 2016). The immense rise of the world’s population and its evergrowing hunger for electricity will only accelerate the combustion of fossil fuels and global warming. Apart from health and environmental issues, the world’s reserves of fossil fuels are plummeting (Shafiee and Topal, 2009).
In the past, a large number of electrochemical energy conversion technologies have been developed. These systems are being continuously optimized in terms of cost, lifetime and performance, leading to their further expansion into existing and emerging market sectors (Badwal et al., 2014). Fuel cells have been identified as one of the most promising technologies to accomplish these goals. A fuel cell is a galvanic cell which converts the chemical reaction energy of a continuously supplied fuel and an oxidizing agent into electrical energy. The major application for fuel cells are stationary electric power plants (e.g. solid oxide fuel cells (SOFCs) and molten carbonate fuel cells). This includes cogeneration units used as motive power for vehicles, as on-board electric power for space vehicles (proton exchange membrane fuel cell) or in other closed environments (Evers, 2003). SOFCs have higher fuel flexibility than other types of fuel cells. They can be operated with different types of fuels like biogas, natural gas, gasoline or diesel (Levin and Chahine, 2010).
Biomass is a versatile, renewable, widely available and potentially sustainable energy source. Besides that, it is almost carbon neutral (Aravind and De Jong, 2012). Anaerobic conversion of organic compounds (biomass) by microorganisms produces biogas. Optimizing biogas becomes increasingly crucial since it is a renewable and clean form of energy which is able to replace the conventional sources of energy, e.g. fossil fuels (Andriani et al., 2014).
Biogas composition is strongly dependent on the source it is obtained from and can consist of 40–70 vol% CH4 and up to 50 vol% CO2. An overview of various biogas compositions from different sources compared to natural gas is given in Table 1. The most commonly present components in biogas are water vapour, hydrogen sulphide (H2S), hydrogen chloride (HCl), ammonia (NH3), O2, CO and N2. The conversion of biogas into energy can be negatively affected by some of those trace components. For example, NH3 can also be considered as fuel for SOFCs. Due to the high operating temperature of the cell, the NH3 is reformed directly in the cell. Ni et al. (2009) performed experiments with NH3-powered SOFCs and came to the following conclusion: NH3 is a technically feasible fuel for direct use in SOFCs and the performance of NH3-fed SOFC is comparable to that of the H2-fed SOFC. The different amounts of H2S that can be present in biogas result in the corrosion of processing equipment and damage of the fuel cells. The content of H2S in biogas depends on its production source and the level of the upgrading process. It is commonly recognized that sulphur compounds are the main source of damage to fuel cell systems (Riegraf et al., 2014). Błesznowski et al. (2013) reported that even 2 ppmv H2S in fuel gas has a visible influence on SOFC performance. Sasaki et al. (2011) carried out experimental runs with 5 ppmv H2S in fuel gas and observed a slightly higher cell degradation rate compared to fuel gas without impurities. Current research results show that state-of-the-art Ni/YSZ cermet anodes can tolerate up to 1 ppm H2S and 10 ppm HCl without significant performance degradation (Błesznowski et al., 2013).
Different biogas compositions compared to natural gas (Wolak, 2012).

Halogenated compounds lead to corrosion in delicate power plant components and measures have to be taken in order to keep the concentrations low (Ryckebosch et al., 2011). The effect of chlorine gas in conjunction with SOFC was analysed in several articles. Sasaki et al. (2007) published experiments which show that fuel gas containing 5 ppmv of Cl does not cause a cell degradation or voltage drop in the SOFC. Błesznowski et al. (2013) investigated the effect of HCl-contaminated fuel gas and concluded that at 10 ppmv a recoverable voltage drop is identified. When increasing concentration levels to 1000 ppmv the cell voltage starts to decrease continuously at a rate of 9.6% over 100 h. Trembly et al. (2007) investigated the effect of HCl on the SOFC at 800 and 900°C. The study indicates that introduction of 20–160 ppmv HCl leads to a performance loss of about 13–52%. He suggested the reaction between Ni and HCl which leads to formation of NiCl2. Besides that, the formation of NiCl2 should be paid attention to in regards of the durability of SOFC anodes because the sublimation temperature of NiCl2 is 985°C and that is near the SOFC operating temperature. Tjaden et al. (2014) suggested subliming NiCl2 at Cl concentrations >100 ppmv. As a conclusion, the HCl has to be removed before the gas is fed to SOFCs for two reasons: (1) HCl can cause corrosion of system components and (2) it can react with components of the SOFC anodes and cause cell degradation. Krishnan et al. (1996) calculated the equilibrium partial pressures of both HCl and metal chloride vapour as a function of temperatures for various alkali and alkaline earth compounds under coal gasification product gas conditions. These calculations predict the theoretical limit of the concentration of HCl that can be reduced to by various oxide and carbonate sorbents. Among the alkali and alkaline earth carbonates only sodium and potassium compounds have equilibrium HCl pressures less than 1 ppm at 773 K. In general, the HCl separation can be carried out by adsorption or absorption. HCl absorbers (scrubbers) are carried out industrially with or without additional neutralization (e.g. by NaOH). The addition of NaOH to the absorbent neutralizes the absorbed HCl, allowing for a more effective process. Since the absorption of HCl in H2O is highly exothermic (ca. 2100 kJ/kg), appropriate heat removal must be ensured at high concentrations. In addition to the adsorption on impregnated activated carbons also sorbents from limestone are used for HCl separation. However, this method does not allow to achieve the high levels of purification required for SOFC applications because of the low conversion efficiencies (Partanen et al., 2005; Rong et al., 2003).
Since little or no data for low-temperature HCl removal for SOFC application is available, this paper provides useful data for process optimization and adsorption tower design. The focus of this work lies in the investigation of the separation of HCl from the gas phase. Besides various sulphur impurities, HCl is one of the primary fuel impurities in biogas feedstocks which should be removed to a certain degree for the successful operation of SOFCs. Test gases with different HCl content were passed through a custom-built lab-scale unit. A custom-made gas wash bottle filled with water and mounted with a pH electrode was installed following the reactor. The amount of HCl that passed the reactor, or in other words which was not adsorbed by the sorbent, dissolved in the water in the gas wash bottle and, therefore, a drop in the pH value could be measured.
Experimental section
Sorbents
The physical characteristics of the potassium carbonate (K2CO3) impregnated activated carbon and the two sorbents based on a mixture of aluminium oxide (Al2O3) and silicon dioxide (SiO2) are listed in Table 2. Highly volatile and harmful substances such as H2S and HCl are only adsorbed in a very small extent on activated carbon. Therefore, a pure physical adsorption process is not practicable. Such pollutants can be removed by chemisorption, an interaction much stronger than physical adsorption. The interaction is a real chemical bond where electrons are shared between the gas and the solid surface. While physical adsorption takes place on all surfaces if temperature and pressure conditions are favourable, chemisorption only occurs on certain surfaces and only if these surfaces are clean. Chemisorption ceases when the adsorbate can no longer make direct contact with the surface. Therefore, it is a single-layer process. In general, physisorption and chemisorption are equally fast. The rate-determining step in the adsorption is not the adsorption itself. The mass transport through the boundary layer surrounding the particles as well as the mass transport in the pores of the adsorbent forms the rate-determining step. With heats of adsorption >70 kJ/mol, as they can occur in chemisorption, according to Bathen and Ciprian (2006) an influence on the total kinetics is to be expected. Aluminium oxide-based sorbents offer a high surface area which can be processed by surface modifiers. In addition to their chemical properties, adsorbents must have a large surface area in relation to their mass, or in other words a high internal or specific surface area. Adsorbents have a more or less regular, interspersed with pores structure in which the pores are interconnected. These pores are divided into micropores (<2 nm), mesopores (2–50 nm) and macropores (>50 nm). The actual adsorptive effect and thus the active surface are largely based on the micropores. The adsorptive is transported through the macro and mesopores. They can be considered as accesses to the actual active surface (Bathen and Ciprian, 2006). A series of experiments on the elimination of H2S with adsorbents led to the initiation of this work. Metal oxide-based sorbents were investigated under various operating conditions to ensure their ability of biogas desulfurization for feeding a SOFC (Weinlaender et al., 2016). Apart from the negative effect on SOFC performance caused by biogas contaminated with HCl, a negative impact on H2S removal capacity of zinc oxide-based sorbents by HCl was reported (Jun et al., 2004).
Physical properties of the investigated sorbents.

Synthetic biogases
The composition of biogas has already been discussed in the ‘Introduction’ section and an overview of the different biogas compositions depending on the source of the gas is given in Table 1. In essence, the different biogases vary in the concentrations of the trace gases H2S, NH3 and halogenated compounds (Schneider, 2007).
In order to analyse the influence of HCl on the adsorption capacity, test gases with 100 and 1000 ppmv HCl content balanced with nitrogen (N2) were used. The gases were mixed and bottled by a commercial supplier. The relative measurement uncertainty of the certified test gases was 3%. This means that the accuracy of the 100 ppmv HCl gas mixture was 3 and 30 ppmv for the 1000 ppmv HCl gas mixture. To investigate the simultaneous adsorption of HCl and H2S, a certified gas mixture with 100 ppmv HCl and 200 ppmv H2S was used.
The influence of the main components of biogas, namely CH4 and CO2, on H2S adsorption performance was investigated by the authors in an earlier research (Weinlaender et al., 2017). Examination of a sorbent based on a mixture of CuO–MnO showed a significant influence of the gas composition on the breakthrough time. Expressed in numbers, the time to breakthrough of 28.1 h for an inlet gas consisting of 200 ppmv H2S in N2 decreased to 22.2 h for an inlet gas consisting of 200 ppmv H2S, 40 vol% CO2 and 59.98 vol% CH4. The authors are aware of this negative impact, but focused primarily on the investigation of the HCl adsorption capacity of the sorbents in the presence of N2. This approach allows a comparison with other results because adsorption capacity is best observed in the presence of an inert gas (e.g. N2 or He). An investigation focusing on the influence of other biogas components on the HCl adsorption capacity may possibly follow in the future. An overview of the test gases used in this research is given in Table 3.
Gas mixture compositions.

Test rig
The experimental set-up is schematically described in Figure 1. The custom-built lab-scale test unit consists of the following parts: certified test gases mixed by a commercial supplier, a mass flow controller, an adsorber, a custom-built gas wash bottle which can mount a pH electrode, a pH meter with electrode and a gas analyser for monitoring H2S content (only used for gas mixture III).

Experimental set-up for HCl adsorption measurements (A: test gas; B: mass flow controller; C: adsorber; D: gas wash bottle; E: pH electrode and pH meter; F: gas analyser; G: vent).
The test gas bottle (A) is equipped with a pressure regulator ensuring a stable pressure of 2.4 bar at the outlet. A flexible metallic gas hose is used to connect the test gas bottle (A) with the mass flow controller (B). The mass flow controller guarantees that the feed gas is dozed within an accuracy of 0.5% of reading 0.1% of full scale. All elements following the mass flow controller are made of polytetrafluoroethylene (PTFE) or glass to avoid undesired reactions. The adsorber is a glass-made cylinder with 150 mm length and an inner diameter of 17 mm. The adsorber is sealed with lab stoppers made of PTFE on both sides. This allows mounting different sorbents without major reconstruction. By installing the sorbent sample within two layers of glass packings with 2 mm diameter, a homogeneous distribution of the feed gas is ensured. The custom-made gas wash bottle is suitable for taking up a pH electrode. To prevent a possible falsification of measurements, the gas wash bottle was filled with deionized H2O. In order to maximize the solubility of the gaseous HCl in the deionized H2O, the gas wash bottle is equipped with a frit. The pH electrode which is mounted in the gas wash bottle is connected with a pH meter. The pH value in the aqueous phase was measured every 30 s with a Mettler Toledo Seven2GoTM pH meter (accuracy 0.002%). While performing test runs with gas mixture III, the H2S concentration in the gas stream at the gas wash bottle outlet was continuously measured with an ABB AO, 2000 gas analyser (accuracy 0.05 ppmv). The control of the mass flow controller and the recording of process data were achieved with the aid of LabVIEW software and hardware. During the test phase of this carefully developed system, comparative measurements using gas chromatography were performed. The experiments showed a very good agreement up to a pH of 3. Here, the deviations from the gas chromatography measurements were below 5%. At a pH value around 2, however, the deviations became much larger, which is also the reason for the chosen termination criterion. The fact that the measurements by gas chromatography are complex and no in situ measurements are possible led to the development of the measuring system described here. This test rig forms the basis for the design of a gas purification unit based on adsorption to operate a 10-cell stack SOFC. Compared with other gas purification concepts, e.g. spray towers or bubble column reactors, the adsorption offers a less consuming solution regarding equipment cost. Furthermore, this solution offers the possibility to operate SOFC with different gases. Switching from biogas to landfill gas, for example, may require siloxane removal. The use of a modular adsorber system would allow a quick installation of an additional adsorber into the gas purification unit.
Test procedure
Variant types of the test runs were carried out using the K2CO3 impregnated activated carbon and the two sorbents based on a mixture of Al2O3 and SiO2 listed in Table 2. The sorbents based on a mixture of Al2O3 and SiO2 were regenerated for 1 h at 250°C before usage. The test gas mixtures listed in Table 3 were passed through the glass-made adsorber and the gas wash bottle while monitoring the pH value of the deionized H2O in the gas wash bottle in time steps of 30 s.
These test runs were carried out to investigate the parameters space velocity, particle size and HCl inlet concentration on the HCl adsorption capacity of the sorbents. A typical test run was carried out according to the following procedure: the pH meter including the pH electrode was calibrated, a measured quantity of sorbents was loaded into the adsorber to achieve a desired space velocity (e.g. 8000 h−1) and 150 ml of deionized H2O was filled into the gas wash bottle with the installed pH electrode. After connecting all pipes, all seals were checked by passing N2 through the apparatus. Following the activation of the data logging, the test gas mixture was fed into the adsorber. The pH value was measured continuously and the value was recorded every 30 s. The test run was stopped after the pH value dropped below 3. After that the amount of HCl that passed the adsorber over the time of test run was calculated based on pH value shift. The test runs involving gas mixture III, or in other words the test performed with HCl and H2S in the fed gas, followed the same procedure with the following exception. After leaving the gas wash bottle, the gas was passed through the H2S gas analyser. After completion of the test run, all parts were disassembled, cleaned and dried. To minimize differences and eliminate measurement errors, all measurements were done by double identification. The calculation of the sorbent loading was carried out by mass balance. By forming the difference of the mass of HCl which was metered into the reactor by means of MFC and the solution of HCl in the bubbler, the sorbent loading resulted. The sulphur capture capacity of the sorbent samples was calculated using the following formula, which has been used by Kim et al. (2007)


Results and discussion
Effect of space velocity on adsorption performance
In order to reveal the influence of space velocity, it is necessary to define the parameter gas hourly space velocity (GHSV). GHSV refers to the quotient of the entering volumetric gas flow rate to the reactor volume.
Test runs were conducted at space velocities of 8000 and 16,000 h−1, respectively. These two specific GHSV values were used to allow comparison to previous research. The other parameters for those test runs were kept stable. The experimental test runs were carried out with biogas mixture II at a reaction temperature of 20°C and with pellets of size 3–4 mm. A pH value of 3 marks the interruption criterion for all the experiments performed in this research.
Figure 2 shows the results of the test runs performed at space velocity of 8000 h−1. It can be seen that after 22 min the pH value in the gas wash bottle dropped below 3 while using no sorbent sample in the reactor. Loading the reactor with the activated alumina samples resulted in breakthrough time of 66 and 71 min, respectively. The best performance, namely the HCl adsorption capacity of 0.0288 g/g, was achieved with the impregnated activated carbon. The results achieved by increasing the GHSV up to 16,000 h−1 are demonstrated in Figure 3. The first test run to be performed was without sorbent loading in the reactor and the pH value of the H2O in the gas wash bottle dropped below 3 after 12 min. For the impregnated activated carbon, the HCl adsorption capacity decreased from 0.0288 to 0.0172 g/g. On the other hand, using the sorbents based on activated alumina led to HCl uptakes of 0.0187, respectively, 0.0104 g/g. Comparing Figures 2 and 3 leads to the following conclusion: Doubling the space velocity from 8000 to 16,000 h−1 had a significant impact on the capture capacity. This capture capacity decrease is a result of the shortened contact time between gas and sorbent and is in line with Kim et al. (2007) and Weinlaender et al. (2016). The capture capacity of sorbents depends on the gas solid contact time in the reactor which decreases at higher space velocities. Furthermore, it can be concluded that the activated alumina sample I took up most HCl until breakthrough at space velocity of 16,000 h−1 while the impregnated activated carbon sample had the best capture capacity at 8000 h−1. The numbers in Table 4 represent the amount of HCl in g/g that had been adsorbed by the sorbent until the pH value dropped below 3. Additionally, a significant decrease of HCl uptake capacity on activated carbon sample and aluminium oxide II sample by doubling the space velocity can be seen. On the other hand, there is just a minor difference of HCl uptake capacity between space velocity 8000 and 16,000 h−1 for the aluminium oxide I sample.

HCl adsorption performance of different sorbents. Reaction temperature 20°C; HCl inlet concentration 1000 ppmv; GHSV 8000 h−1; pellet size 3–4 mm.

HCl adsorption performance of different sorbents. Reaction temperature 20°C; HCl inlet concentration 1000 ppmv; GHSV 16,000 h−1; pellet size 3–4 mm.
Results for HCl adsorption capacity of different sorbents at GHSV 8000 h−1 and GHSV 16,000 h−1.

GHSV: gas hourly space velocity.
Results for HCl adsorption capacity of different sorbents with particle size 500–1000 µm.

Effect of sorbent particle size on adsorption performance
Besides space velocity and inlet concentration, the sorbent pellet size is one of the parameters with an influence on adsorption performance. In the adsorption tower design it is a limiting factor since apart from the tower height, the pellet size is decisive for the pressure drop (Donau Carbon GmbH, 2016). On the other hand, finely ground pellets provide shorter diffusion paths and this results in a larger mass transfer (Bathen and Ciprian, 2006).
The sample preparation was carried out the following way. Mortar and pestle were used to crush the pellets. After that a column of sieves was used to get a fraction of sorbent particles into the size between 500 and 1000 µm. This particle fraction was loaded into the reactor. In order to compare the results among each other, the tests were performed at space velocities of 8000 and 16,000 h−1. Figures 4 and 5 present the results for the particles at different space velocities and have to be compared with Figures 2 and 3.

HCl adsorption performance of different sorbents. Reaction temperature 20°C; HCl inlet concentration 1000 ppmv; GHSV 8000 h−1; particle size 500–1000 µm.

HCl adsorption performance of different sorbents. Reaction temperature 20°C; HCl inlet concentration 1000 ppmv; GHSV 16,000 h−1; particle size 500–1000 µm.
Comparing Figures 2 and 4 resulted in the following conclusions: Crushing the 3 mm impregnated activated carbon pellets into particles in the size of 500–1000 µm had no major influence on the adsorption performance for the experiments performed at a space velocity of 8000 h−1. The pH value dropped below 3 for both experiments after 95 min. In numbers (see Table 4) the HCl uptake increased from 0.0288 to 0.00292 g/g. Comparing the results of the activated alumina II sample led to a similar conclusion. There was almost no influence on HCl uptake or breakthrough time. However, the activated alumina I sample achieved a significant performance boost by crushing the pellets into particles. In numbers the HCl uptake increased from 0.0184 to 0.0406 g/g for the experiments performed at space velocity 8000 h−1 and from 0.0187 up to 0.0373 g/g at space velocity 16,000 h−1. Therefore, it can be concluded that the sorbents are not completely loaded at the selected termination criterion (pH value 3). All samples almost doubled their HCl uptake at GHSV 16,000 h−1.
Technically, crushing the pellets increases the surface which should have a positive impact as with smaller particles, the diffusion paths are shorter. Due to the longer contact time between the gas phase and the sorbent, this effect should also be monitored at GHSV of 8000 h−1 in order to compare pellets and particles. One reasonable explanation why only the aluminium oxide I achieved a better adsorption performance may be explained by an incorrect sample preparation for the activated carbon and the aluminium oxide II (e.g. crushing the pellets resulted in destroying the macroporosity).
Effect of HCl concentration on adsorption performance
Figure 6 shows the performance of the three different sorbents while using gas mixture I. Said mixture consists of 100 ppmv HCl balanced with N2 at 20°C and a space velocity of 16,000 h−1. To investigate the influence of the HCl inlet concentration on adsorption performance, Figure 6 needs to be compared with Figure 3 which was monitored under the same operational parameters except the HCl inlet concentration, which was as high as 1000 ppmv.

HCl adsorption performance of different sorbents. Reaction temperature 20°C; HCl inlet concentration 100 ppmv; GHSV 16,000 h−1; pellet size 3–4 mm.
The contemplation of the abscissa of both figures demonstrates the major influence of the HCl inlet concentration. It can be easily identified that the test run duration increased 8–10 times before reaching the interruption criteria, a pH value of 3 in the gas wash bottle. The corresponding numbers are given in Table 6. Comparing the sorbent uptake capacity shows that using a higher HCl inlet concentration led to a 10% higher sorbent uptake. This can be explained by the concentration gradient. One of the driving forces of adsorptive separation is the concentration of the feed. In other words, the higher the dilution the lower the driving force and the higher the increase in technical effort. This means particularly that the extent of sorbent utilization was improved by the HCl content of the test gas.
Results for HCl adsorption capacity of different sorbents with HCl inlet concentration 100 and 1000 ppmv. Other parameters: reaction temperature 20°C; GHSV 16,000 h−1; pellet size 3–4 mm.

GHSV: gas hourly space velocity.
However, the increased test run duration until reaching the interruption criteria could be explained as follows: Less pollutant molecules (e.g. HCl) in the gas feed mean less saturation of free adsorbent sites on the solid. In other words, a minor feed concentration increases the duration until saturation of the sorbent is reached.
The sum of the conducted experiments in this work led to the conclusion that decreasing the space velocity at lower HCl inlet concentration will result in very long test runs with similar HCl uptake.
Simultaneous separation of HCl and hydrogen sulphur
Apart from static capacitance effect, steric effect and kinetic effect, the equilibrium effect is one of the four mechanisms the selectivity of adsorption is essentially based on. Since all thermodynamic systems strive to minimum of free energy, the component with higher energy of bonding is adsorbed preferably. Because of the high experimental effort, multi-component adsorption equilibria for the practical interpretation of adsorbers are only rarely fully measured. Instead, the adsorption is concentrated from multi-component mixtures to the measurement of the breakthrough curve. In particular, this provides information on kinetics and dilution effects (Bathen and Ciprian, 2006).
The activated carbon is impregnated with K2CO3, which allows a simultaneous adsorption of HCl and H2S. The reaction of K2CO3 with HCl leads to potassium chloride (KCl) which has a bond dissociation energy of −436 kJ/mol (chemical reaction equation (2)). On the other hand, the reaction between K2CO3 and H2S leads to potassium sulphide (K2S) which has a bond dissociation energy of −380.7 kJ/mol (chemical reaction equation (3))


Figures 7 and 8 show the performance of the activated carbon while using gas mixture III, consisting of 100 ppmv HCl and 200 ppmv H2S balanced with N2, at 20°C and at space velocities 8000 and 16,000 h−1, respectively. A comparison of the two figures leads to the following conclusion: The measured H2S output concentration seen in Figure 7 seems to be stable at 160 ppmv H2S from 260 min until the test run stops. Doubling the space velocity up to 16,000 h−1 led to a H2S outlet concentration of 160 ppm after 90 min, increasing up to 180 ppm until the test run ended after 170 min. By doubling the GHSV the HCl breakthrough time had been halved from 300 to 150 min. The numbers for the HCl and H2S uptake are given in Table 7.
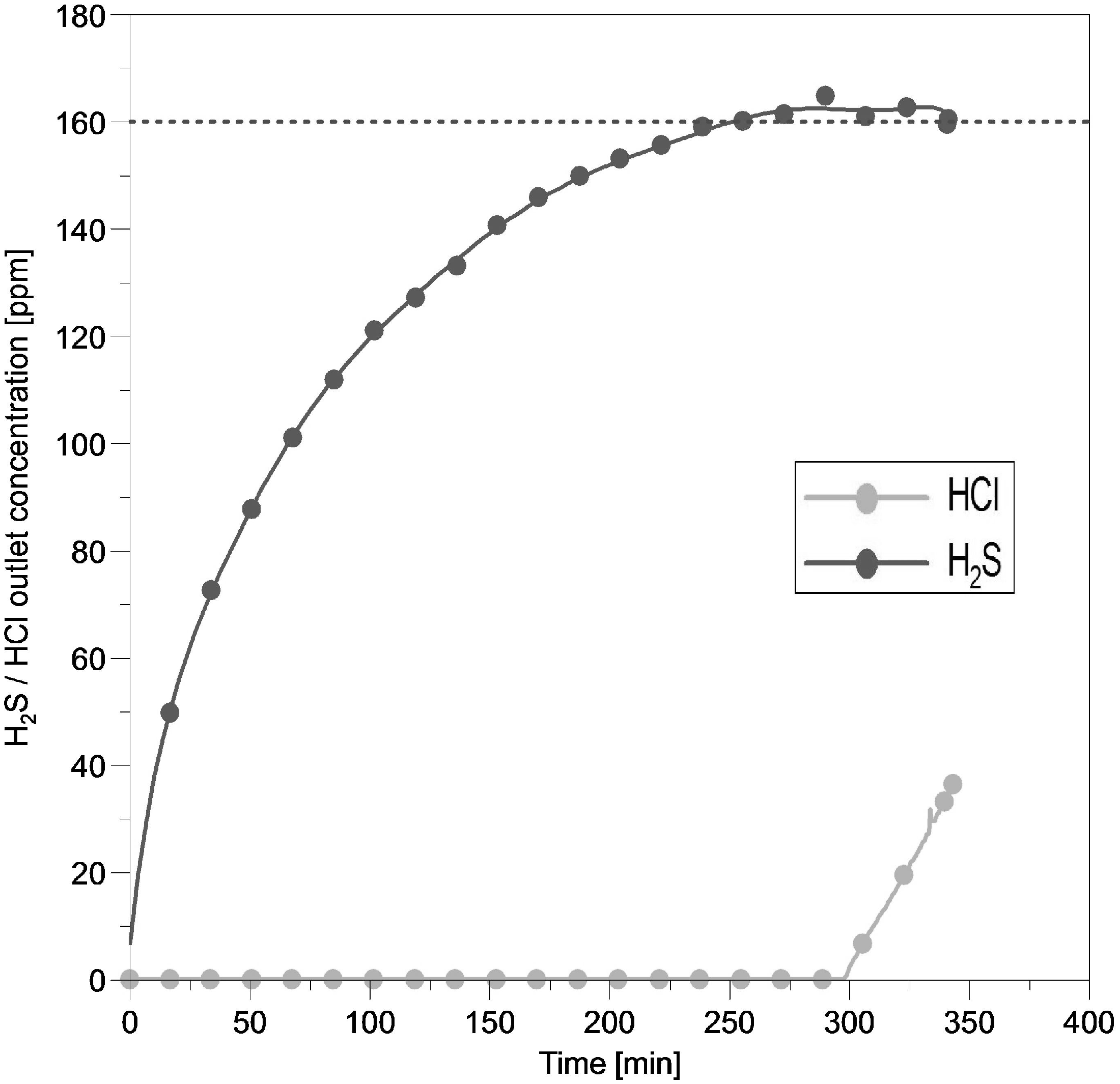
HCl–H2S adsorption performance of activated carbon. Reaction temperature 20°C; HCl inlet concentration 100 ppmv; H2S inlet concentration 200 ppmv; GHSV 8000 h−1; pellet size 3–4 mm.
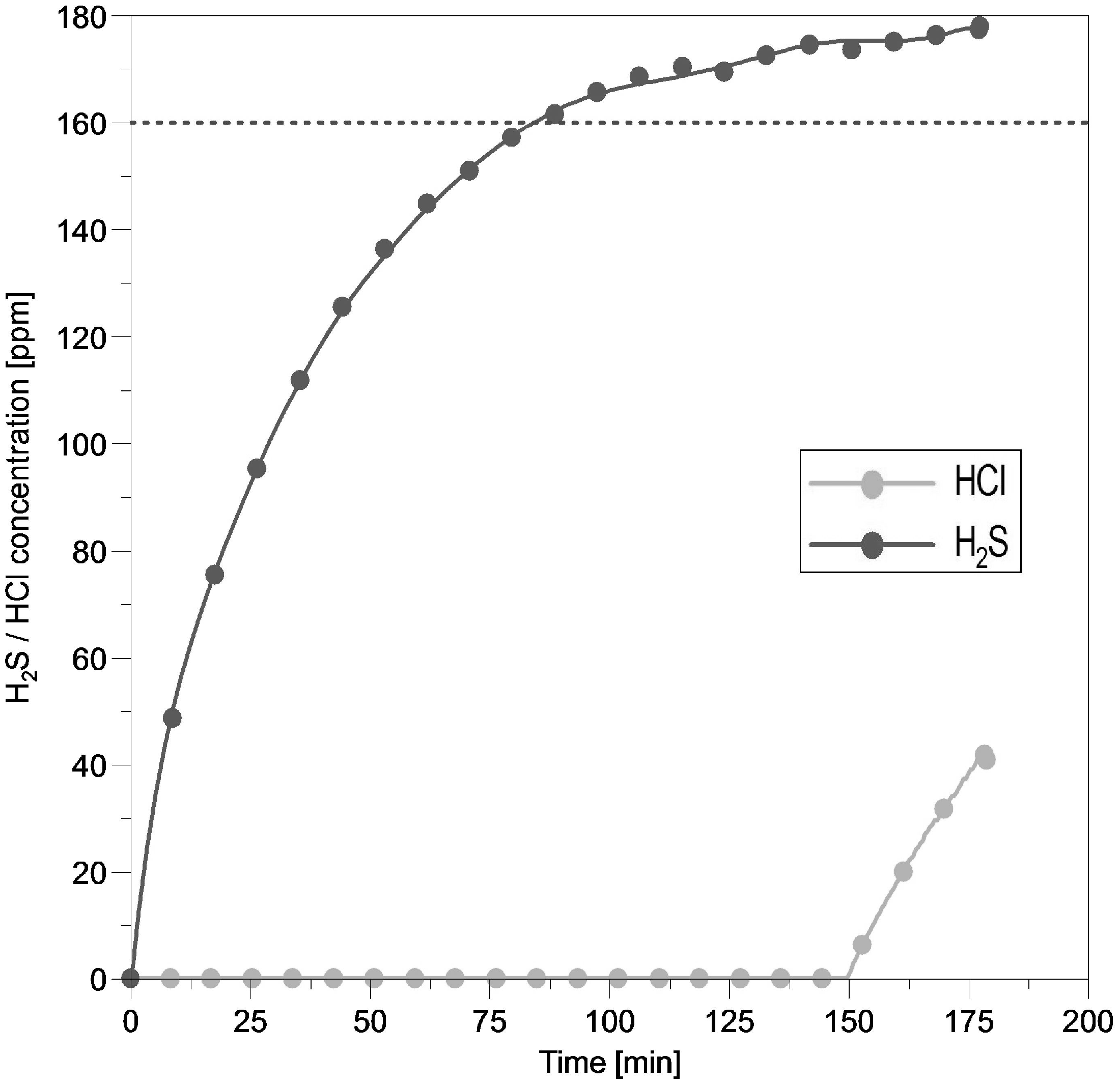
HCl–H2S adsorption performance of activated carbon. Reaction temperature 20°C; HCl inlet concentration 100 ppmv; H2S inlet concentration 200 ppmv; GHSV 16,000 h−1; pellet size 3–4 mm.
Results for H2S and HCl adsorption capacity of activated carbon at GHSV 8000 and 16,000 h−1.

GHSV: gas hourly space velocity.
Conclusion
It is essential to remove HCl from biogas to a certain level to ensure a fault-free operation of SOFCs. The adsorption performance of three different sorbents was investigated in this work. An activated carbon is impregnated with K2CO3 and two sorbents based on a mixture of Al2O3 and SiO2. In order to compare the sorbents under different conditions, the HCl uptake, a value for the extent of sorbent utilization, has been calculated. HCl adsorption capacity was investigated under different space velocities, inlet concentrations and particle sizes. Furthermore, the adsorption of HCl and H2S was investigated at the same time. The experiments performed in this work on low-temperature HCl adsorption using different commercially available sorbents have led to the following conclusions:
Investigating the influence of GHSV on HCl adsorption performance revealed a difference in the operating behaviour of the sorbents. The HCl capture capacity of sorbents is dependent on the contact time between the feed gas and the sorbent sample in the reactor. With higher GHSV this contact time decreases and the adsorption performance drops. The activated carbon sample achieved the best results at GHSV 8000 h−1. Increasing the GHSV up to 16,000 h−1 had a negative effect on all sorbents except the alumina oxide I sorbent. Crushing the pellets into a particle fraction between 500 and 1000 µm almost doubled the HCl uptake for all sorbents at the test runs performed at GHSV 16,000 h−1. Technically, crushing the pellets increases the surface which should have a positive impact as with smaller particles, the diffusion paths are shorter. However, the experiments conducted at a GHSV of 8000 h−1 do not confirm this theory. The activated aluminium oxide I sample achieved a better HCl uptake, while the activated carbon and the aluminium oxide II gained only a minimal benefit from crushing the pellets. The reason behind this may be explained in a defective sample preparation, e.g. crushing the pellets into particles destroyed the macroporosity. Increasing the HCl inlet concentration resulted in the following findings: A 15% higher sorbent uptake could be achieved due to the increased concentration gradient, which is a driving force of adsorptive separation. Additionally, the test run time decreased almost by a factor of 10 when the HCl inlet concentration was increased by a factor 10 from 100 to 1000 ppmv. In other words, more HCl molecules in the feed gas result in a faster saturation of the free adsorbent sites on the solid. K2CO3 is able to capture HCl and H2S. Due to the fact that thermodynamic systems strive to a minimum of free energy, the component with higher energy of bonding, namely HCl, is adsorbed preferably. Two test runs performed with space velocities of 8000 and 16,000 h−1 confirmed the possibility of simultaneous adsorption with the impregnated activated carbon.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank the Austrian Research Promotion Agency (FFG) for funding this project. The project title is SOFCool (grant number 843835, eCall 4157797).
