Abstract
Four activated carbons (ACs) activated by different activation methods from different precursors were heat-treated at 600℃ to decrease surface oxygen functional groups and to separate the effect of pore texture from surface chemistry on their sulfur adsorption performance. The ACs and the heat-treated ones were used as adsorbents for thiophene (T), dibenzothiophene (DBT), and 4,6-dimethyl dibenzothiophene (4,6-DMDBT) for model fuels with or without a competing benzene. The correlations of surface functional groups and pore textures to their adsorption capacities for T, DBT and 4,6-DMDBT were investigated. Results indicated that the heat treatment not only decreased surface oxygen content, but also caused a pore texture modification. The adsorption isotherms can be well fitted by the Langmuir–Freundlich single solute isotherm. The sulfur adsorption capacity of ACs after the treatment for T, DBT, and 4,6-DMDBT has a good linear relationship with the pore volume of pores within 0.536–1.090, 0.536–1.179, and 0.679–1.268 nm, respectively. The decreased percentage of surface oxygen of ACs by the heat treatment is correlated linearly with the decreased percentage of the sulfur adsorption capacity for all the thiophenic compounds. Micropore filling via the π–π interaction is the dominant adsorption mechanism of thiophenic compounds for both the untreated and heat-treated ACs. Surface oxides contribute to the thiophenic adsorption by a cooperative interaction of the π–π interaction and the acid–base interaction between the basic sulfur atom and the acidic carbon atoms linked with oxygen in graphene edges. A flat adsorption is the most possible geometry in the adsorption of thiophenic compounds.
Introduction
Sulfur compounds in fuel oils can cause environmental problems because they are converted to SOx during combustion, which not only contributes to acid rain but also poisons the catalytic converters for exhaust emission treatment. More and more environmental protection acts worldwide require a deep sulfur removal in fuel oils below 10 ppm (Khan and Reynolds, 1996; Lee et al. 1993; Unzelman 1993; US EPA, 1999). Moreover, near zero-sulfur fuel is needed as hydrogen source for proton-exchange membrane fuel cells (Song, 2001). The challenge for the deep desulfurization with conventional hydrodesulfurization is the difficulty to remove the refractory sulfur compounds such as 4,6-dimethy ldibenzothiophene (4,6-DMDBT) without sacrificing octane number and with an acceptable cost (Song and Ma, 2003). Among the desulfurization methods currently investigated, such as reactive adsorption, oxidative desulfurization, biodesulfurization, extractive desulfurization and physical adsorption with activated carbons (ACs) or nanoporous carbons, the last one is attractive in that it can be operated at ambient condition, and ACs used are abundant, have a high adsorption capacity for refractory sulfur compounds and can be regenerated (Kim et al., 2006; Song, 2003; Zhou et al., 2006). An integrated process is possible for deep desulfurization by combining AC adsorption with hydrodesulfurization based on the advantages of these two processes. There are many investigations on adsorptive desulfurization with porous carbons to correlate their surface chemical and porous properties to adsorption capacity and selectivity. However, these correlations from different researchers are quite different, even discrepant with each other, and mechanisms proposed are correspondingly quite different. Seredych and Bandosz (2010) found that adsorption capacities of DBT and 4,6-DMDBT on nanoporous carbons from polymer source and air-oxidized ones have a good linear relationship with the volume of pores smaller than 1.0 nm and surface functional groups play an important, but not dominant, role in the adsorption. Ania and Bandosz (2005) made a trial to correlate the DBT adsorption capacity to pore texture and surface functional groups for ACs from different sources before and after (NH4)2S2O8-oxidation. They found that the adsorption capacity has a linear relationship with the volume of narrow pores below 0.7 nm for the unoxidized ACs and with the density of acidic sites from the Boehm method for both the oxidized and unoxidized ACs. Zhang et al. (2014) prepared phenolic resin-based spherical ACs and revealed that the DBT adsorption capacity can be best fitted linearly to the volume of micropores between 0.6 and 1.2 nm. Zhou et al. (2009) made an effort to correlate adsorption capacity per unit area of the oxygen or HNO3-oxidized ACs with kinds and amounts of surface functional groups evaluated by X-ray Photoelectron Spectrocopy (XPS). They found that sulfur adsorption capacity per unit area can be related linearly to COO, O–C, and CO + COO functional groups and concluded that the adsorption may occur through the interaction of the acidic oxygen-containing groups with thiophenic ring or acid–base interaction mechanism. They also found that the selectivity for thiophenic compounds in presence of arenes increases slightly for the microporous carbons and decreased severely for the mesoporous carbons. However, Bu et al. (2011) found that DBT adsorption capacity can be well correlated linearly to the mesopore volume of seven ACs from different manufacturers and the π–π dispersive interaction between benzene rings is dominant in adsorption affinity. Yu et al. (2009) and Zhang et al. (2012) found that thermal and oxygen plasma oxidation can increase surface acidic functional groups and therefore DBT adsorption was enhanced. Yang et al. (2007) modified AC by steam activation followed by concentrated H2SO4 oxidation, which significantly improved selectivity of DBT in the presence of arenes. They ascribed the improvement in selectivity to surface acidic oxygenated groups.
These discrepancies might be caused by a wide variety of pore texture and surface chemistry of ACs in combination with the complex interactions between porous carbon and thiophenic compounds. Another factor is that a correlation of pore size with adsorption capacity has not been excluded from the effect of surface functional groups in previous investigations. As most carbons contain more oxygen than can be accounted for by the observed functional groups using the Boehm method (Boehm, 2002) due to ether-type oxygen or to carbonyl groups that do not react with NaOC2H5, these kinds of functional groups have not been considered in correlating sulfur adsorption capacity with surface functional groups and pore texture for some researchers.
In this paper, instead of oxidation to increase surface oxygen functional groups widely investigated by many researchers, four ACs from different precursors and different activation methods, which have widely varied pore texture and surface properties, were heat-treated at 600℃ to decrease the contents of surface functional groups. The purpose of the heat treatment is to separate the effect of pore texture from the surface chemistry to give different perspectives in terms of the role of pore and surface properties in thiophenic adsorption in fuel oils.
Experimental
Materials
Four ACs were used in the study. AC is a commercial product from Fujian Ningde Activated Carbon Plant (Fujian, China), 2100 and 2800 are provided by Institute of Coal Chemistry, Chinese Academy of Sciences (Shanxi, China), which were activated by KOH with a KOH/petroleum coke ratio of 3 and 4, respectively. 3 k is supplied by University of Missouri-Columbia (MO), which was obtained by phosphoric acid activation of carbonized corn cob followed by KOH activation. The four ACs were heat-treated at 600℃ for 3 h under nitrogen flow to reduce surface oxygen-containing functional groups. The heat-treated ACs were marked as H-AC, H-2100, H-2800, and H-3 k.
Characterization
The surface areas and pore texture of all samples were determined by N2 adsorption using a Micromeritics ASAP 2020 instrument (Micromeritics Inc., TX). The samples were degassed under vacuum at 150℃ for 15 h prior to the measurements. The surface area (SBET) was calculated from the N2 adsorption data using the standard Brunauer, Emmett and Teller (BET) equation in the relative pressure (p/p0) between 0.05 and 0.2. The pore size distributions were obtained using density functional theory, assuming a slit-shaped pore geometry as this model gives a best fit to the adsorption isotherms. The volumes of micropores between different size intervals were calculated from the cumulative pore volume versus pore size. For example, the volume of pores between 0.536 and 1.090 nm, V0.536–1.090 nm, was calculated by the cumulative volume of pores less than 0.536 nm, V0.536 nm, deducted from that of pores less than 1.090 nm, V1.090 nm.
The elemental compositions and surface functional groups in the samples were analyzed by a British Kratos Axis Ultra DLD X-ray photoelectron spectrometer (XPS; Kratos Inc., Manchester, UK) using Al Kα radiation (1486.6 eV). All binding energies were calibrated based on the C1s line (284.6 eV) of adventitious carbon. The XPS analysis was done at ambient temperature and the base pressure in the measurement chamber was less than 10−8 Pa. Prior to analysis, the samples were degassed in a vacuum oven overnight.
Adsorption
Model fuels were prepared by dissolving thiophene (T), DBT, or 4,6-DMDBT in n-octane. Other thiophenic model fuels were made to include 10 wt% of benzene as a competing agent. In each run, 10 g model fuel was mixed with 0.1 g sample in a flask and put into a shaking bath at 30℃ for 48 h to ensure an equilibrium adsorption. The sulfur adsorption capacity was calculated with the following equation:
The sulfur concentration in fuel oils was analyzed using a GC-2014C gas chromatograph (Shimadzu Corp., Tokyo, Japan) equipped with a capillary column (MXT-5, 60 m in length, 0.25 mm in internal diameter and 0.25 µm film thickness, Restek, America) and a flame ionization detection (FID) for identification and quantification of the thiophenic compounds in the model fuels before and after adsorption. Diphenyl sulfide was used as an internal standard. The column temperature was programmed to hold at 220℃ for 3 min, to rise from 220℃ to 280℃ at 4℃/min and to hold at 280℃ for 10 min for DBT and 4,6-DMDBT. For T, the column temperature was programmed to hold at 40℃ for 7 min, to rise from 40℃ to 260℃ at 5℃/min and to hold at 260℃ for 10 min. The temperatures for injector and detector were 250℃ and 310℃, respectively. The injection volume of each sample was 1 µL and the splitting ratio was 100:1.
Results and discussion
Porous texture of ACs
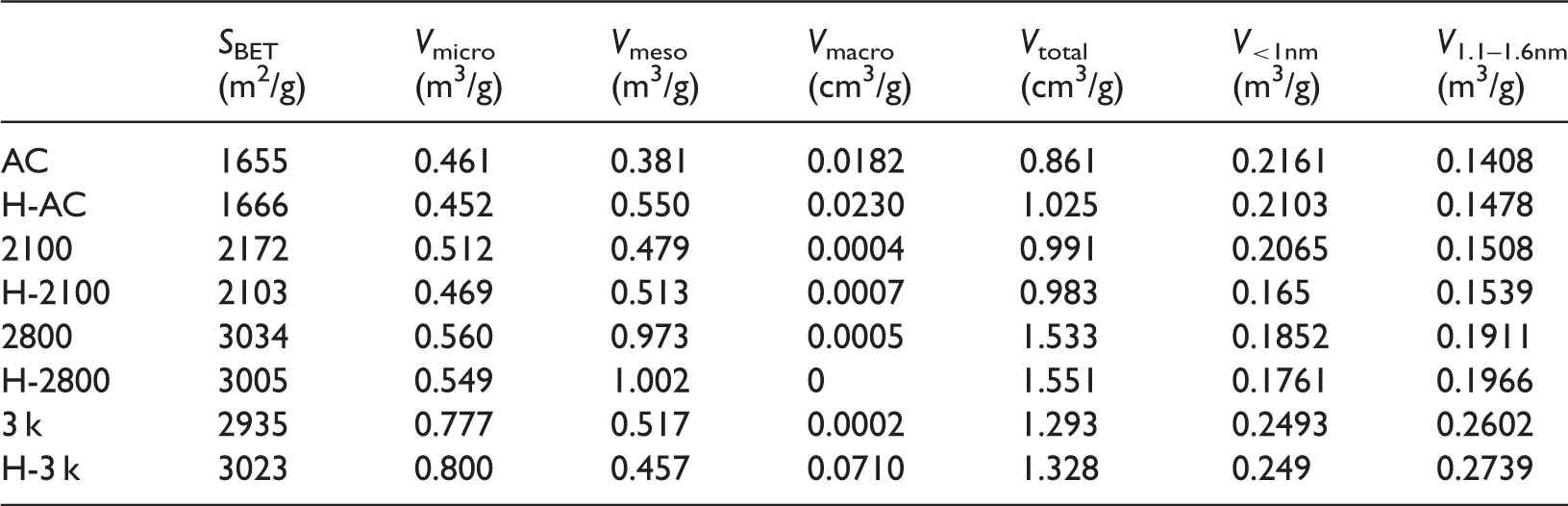
Figure 1 shows porous structure of the ACs before and after the heat treatment. Samples 2100, 2800, and 3 k with and without the heat treatment exhibit type adsorption isotherms according to International Union of Pure and Applied Chemistry (IUPAC) classification (Singh et al., 1985), indicating that they are mainly microporous. Samples AC and H-AC reveal a sharp increase of adsorption at the low limit of p/p0 similar to the other samples, and in addition have a hysteresis loop above p/p0 of 0.4, indicating that they are rich both in micropores and mesopores. We have tried several DFT-based models to fit the adsorption isotherms and found that the model based on non-local density functional theory (NLDFT) with a slit-shaped pore geometry gives a best fit. This is the basic requirement to calculate reliable porous texture. The conclusions reached by the shape of adsorption–desorption isotherms are in good agreement with the pore size distributions fitted by the NLDFT model. The cumulative pore volumes of all samples with pore size are obtained by the NLDFT model, from which volumes of pores at different size intervals are calculated. The correlation between sulfur adsorption capacity and volumes of pores at different size intervals in what follows is based on this calculation. The porous properties of all samples are listed in Table 1. From Table 1, it is found that the micropore volumes decrease and mesopore volumes increase after the heat treatment for samples AC, 2100, and 2800, but inverse trend is found for sample 3 k. The total pore volumes of all samples increase after the heat treatment. In micropore regime, the volumes of pores less than 1 nm decrease while those of pores between 1.1 and 1.6 nm increase, indicating the heat treatment causes a collapse of pores less than 1 nm, which contributes to a pore volume increase of pores between 1.1 and 1.6 nm. The BET surface areas of the heat-treated ACs change little as compared with the untreated ones.
(a) Adsorption–desorption isotherms, (b) pore size distributions, and (c) cumulative pore volumes of the activated carbons before and after the heat treatment. Porous parameters of the ACs.
Surface chemistry of ACs
Figure 2 shows high resolution C1s spectra of the ACs before and after the heat treatment. The C1s peaks for samples were curve-fitted with five peaks (Yue et al., 1999) representing graphitic carbon (C=C at 284.6 eV), carbon present in phenolic, alcohol, ether (C–O at 286.1–286.3 eV), carbonyl or quinone groups (C=O at 287.3–287.6 eV), carboxyl or ester groups (COO at 288.4–288.9 eV) and carbon present in carbonate groups and/or adsorbed CO and CO2 (at 290.4–290.8 eV). A Shirley background subtraction was made in the fitting. The binding energies, full widths at half maximum (FWHM) and relative contents of these functional groups together with the oxygen contents by XPS are listed in Table 2. It is found that the surface functional groups of the ACs are quite different from each other. The absolute content of C=C group increases after the heat treatment. The oxygen content and the relative content of C–O group decreases, and COO group disappears after the heat treatment. Although there is no C=O group found in samples 2100, 2800, and 3 k, it appears after the heat treatment for samples 2800 and 3 k, indicating that decomposition of thermal unstable COO group leads to the formation of thermal stable C=O group. Although the relative content of C=O group increases after the heat treatment for sample AC, their absolute content decreases slightly. The decrease of the absolute content of C–O group is larger than that of C=O group, indicating that C=O group is more stable than C–O group. The thermal stability of the oxygen functional groups follows the order: C=O > C–O > COO, which is in good agreement with the results in the literature (Figueiredo et al., 1999).
High resolution C1s spectra of activated carbons before and after the heat treatment and their fitted components by deconvolution (Figure available in colour online). Carbon functional groups from deconvolution of C1s spectra.

DBT adsorption isotherms on ACs
Adsorption isotherms of DBT on all samples were obtained by changing the DBT concentration in the model fuels without benzene under the same dose of ACs. Figure 3 illustrates the adsorption isotherms for DBT adsorption on all samples investigated. The DBT adsorption isotherms have a L-type shape according to the Giles classification (Giles et al., 1960). The adsorption isotherms were fitted to the Langmuir–Freundlich single solute isotherm (Derylo-Marczewska et al., 1984), which has the equation:
DBT adsorption isotherms on the activated carbons at 30℃. The fitting parameters of DBT adsorption isotherms to the Langmuir–Freundlich equation.
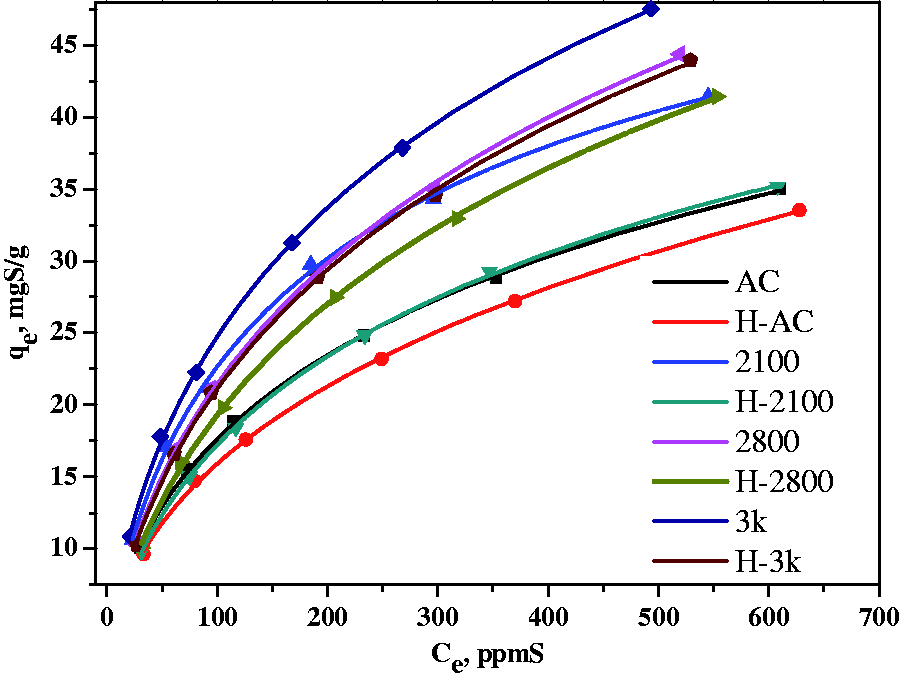

The value of exponent n is related to the degree of surface heterogeneity. The smaller the value, the more energetically homogeneous the surface for adsorption. From Table 3 it can be seen that the maximum adsorption increases and the value of exponent n decreases after the heat treatment for the samples AC and 2100 while the sample 2800 shows an inverse trend, indicating that a more homogeneous surface is favorable for DBT adsorption. The values of exponent n of the samples 2800 and 3 k increase after the heat treatment while those of the samples AC and 2100 exhibit an inverse trend. This different changing trends of n with the heat treatment for different samples could be accounted for by the following reasons. The DBT molecule has a sulfur atom that can donate its lone electron pair and show Lewis base characteristic in adsorption via an acid–base interaction. Also DBT has two benzene rings that exhibit an affinity with graphene layers as pore wall of ACs via a π–π interaction. A more favorable adsorption should occur when the active sites are energetically homogeneous, which means a balanced interaction of the two. The heat treatment causes mainly a decrease of acid characteristic of ACs by a reduction of oxygen functional groups, but changes little for the graphene layers. A decrease of the values of exponent n implies a changing trend towards a balanced interaction. As the samples 2800 and 3 k have BET surface areas around 3000 m2/g, which are much higher than the samples AC and 2100, they should have more oxygen functional groups for the balanced interaction. Therefore, the heat treatment causes an increase of surface heterogeneity for DBT adsorption for these two samples. On the contrary, the samples AC and 2100 have low BET surface areas and should have a low content of oxygen functional groups for the balanced interaction, the heat treatment leads to a deceased surface heterogeneity.
Correlation of adsorption capacity of thiophenic compounds to pore texture of ACs
The purpose of the heat treatment is to remove the majority of oxygen-containing functional groups to isolate the effect of pore size from that of surface chemistry on sulfur adsorption. So we use the data of the heat-treated ACs to correlate the sulfur adsorption capacity with pore texture.
Figure 4 shows the T adsorption capacity of the heat-treated ACs in relation to the pore volumes of pores at different size intervals (from 0.536 to 1.000, 0.536 to 1.090 and 0.536 to 1.179 nm) for the model fuels with and without 10 wt% benzene. It is found that the T adsorption capacity can be best correlated to the pore volume of pores at the size interval from 0.536 to 1.090 nm for both model fuels, indicating that the optimum pores for T adsorption are those between 0.536 and 1.090 nm. The pores less than 0.536 nm are inaccessible for T due to a size limiting effect. The pores larger than 1.090 nm contribute less effectively than those within 0.536 and 1.090 nm.
The relationship between pore volumes at different size intervals and the T adsorption capacity in the different model fuels with T in: (a) n-octane and (b) n-octane + 10 wt% benzene.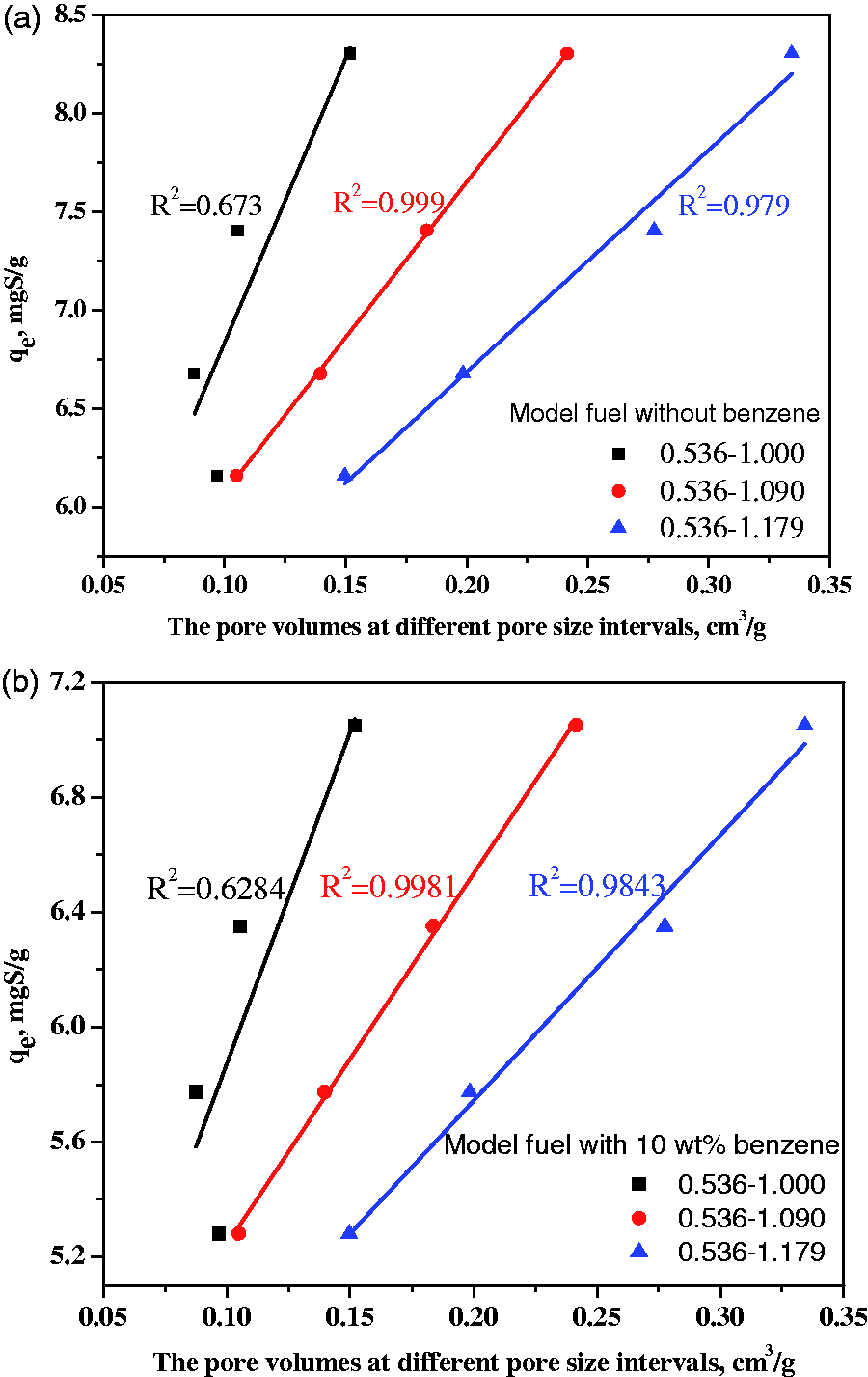
Figure 5 shows the DBT adsorption capacity of the heat-treated ACs in relation to the pore volumes of pores at different size intervals (from 0.536 to 1.090, 0.536 to 1.179 and 0.536 to 1.358 nm) for the model fuels with and without 10 wt% benzene. It is found that the DBT adsorption capacity can be best correlated to the pore volume of pores at the pore size interval from 0.536 to 1.179 nm for both model fuels, indicating that the optimum pores for DBT adsorption are those between 0.536 and 1.179 nm. The pores less than 0.536 nm are similarly inaccessible for DBT due to a size-limiting effect. The pores larger than 1.179 nm contribute less effectively than those within 0.536 and 1.179 nm.
The relationship between pore volumes at different size intervals and the DBT adsorption capacity in the different model fuels with DBT in: (a) n-octane and (b) n-octane + 10 wt% benzene.
Figure 6 shows the 4,6-DMDBT adsorption capacity of the heat-treated ACs in relation to the pore volumes of pores at different size intervals (from 0.679 to 1.179, 0.679 to 1.268 and 0.679 to 1.358 nm) for the model fuels with and without 10 wt% benzene. It is found that the 4,6-DMDBT adsorption capacity can be best correlated to the pore volume of pores at the pore size interval from 0.679 to 1.268 nm for both model fuels, indicating that the optimum pores for the 4,6-DMDBT adsorption are those between 0.679 and 1.268 nm. The pores less than 0.679 nm are similarly inaccessible for 4,6-DMDBT due to a size limiting effect. The pores larger than 1.268 nm contribute less effectively than those between 0.679 and 1.268 nm.
The relationship between pore volumes at different size intervals and the 4,6-DMDBT adsorption capacity in the different model fuels with 4,6-DMDBT in: (a) n-octane and (b) n-octane + 10 wt% benzene.
The fact that large pores contribute less effectively than small pores should be attributed to the large adsorption potential for small pores (Bu et al., 2011). The presence of cut-off pore size is also related to the accessibility of pores to the adsorbates, so there is an optimum pore size interval for adsorption of T, DBT, and 4,6-DMDBT. With increasing molecular size of adsorbates, the optimum pore size intervals for their adsorption shifts to the high value direction, which is easy to understand. Similar pore size interval has been reported for DBT adsorption on spherical AC (Zhang et al., 2014). Therefore, the thiophenic adsorption on the heat-treated ACs is dominated by a micropore filling effect.
With increasing molecular size of the adsorbates, the sulfur adsorption capacity increases, indicating that dispersive interaction between the adsorbate and AC pore wall play an important role. The sulfur adsorption capacity is decreased when 10 wt% benzene is added in the model fuels, indicating a competing effect of benzene with the thiophenic compounds.
Correlation of adsorption capacity of thiophenic compounds to surface chemistry of the ACs
The heat treatment not only greatly decreases the content of surface oxygen functional groups, but also causes a slight change of pore texture, which leads to a decrease of the sulfur adsorption capacity. In order to separate the effect of micropore filling from the surface chemistry of ACs as the pore texture has been changed by the heat treatment, we calculate the contribution of pore texture to the sulfur adsorption capacity of the untreated ACs with the established correlation of the sulfur adsorption capacity with pore texture of the heat-treated ACs. The contribution of surface chemistry to the sulfur adsorption capacity was evaluated by deducting the contribution of pore texture from the total adsorption capacity of thiophenic compounds for the untreated ACs.
Figure 7(a) shows the correlation of the sulfur adsorption capacities for T, DBT, and 4,6-DMDBT to the pore volumes of pores within the pore size intervals for the ACs before and after the heat treatment, and Figure 7(b) shows the reduction of sulfur adsorption capacities for T, DBT, and 4,6-DMDBT versus the reduction of oxygen content by the heat treatment for the model fuels without benzene. The former is related to the contribution of the micropore filling effect (pore texture) and the latter to the surface chemistry. It is found from these figures that the contribution of the micropore filling to the sulfur adsorption capacity is 6–8, 17–21, and 23–26 mgS/g for T, DBT, and 4,6-DMDBT, respectively. The contribution of surface chemistry to the adsorption capacity is 1–1.8, 1–2.3, and 2.3–4.1 mgS/g for T, DBT, and 4,6-DMDBT, respectively. The contribution of the former is much larger than the latter. This indicates that the micropore filling effect is also dominant even for the untreated ACs. The reduction of sulfur adsorption capacities for T, DBT, and 4,6-DMDBT by the heat treatment have a good linear correlation with the reduction of oxygen content (Figure 7b). This implies that the contribution of different oxygen-containing functional groups to the sulfur adsorption is similar, which means that ether-type oxygen or carbonyl groups that cannot be detected by the Boehm method contributes to the thiophenic adsorption. This result was also observed by Zhou,13 who showed that thiophenic adsorption capacity had a linear relationship with the amount of the CO + COO groups by XPS. This can be accounted for in the following. The C and O atoms have an electronegativity of 2.5 and 3.5, respectively, which means that the C atoms linked with O atoms have an electron deficiency and therefore are Lewis acid sites. From the fact that all oxygen-containing functional groups, C–O, C=O, and COO contribute to an increase of sulfur adsorption capacity and only phenolic COH and carboxylic COOH groups have a proton, it can be deduced that Lewis acidic sites of the C atoms linked to O atom be responsible for the acid–base interaction in the adsorption of thiophenic compounds. With an increasing of molecular size of the adsorbates, the contribution of oxygen functional groups to the sulfur adsorption capacity increases, which implies that there is a cooperative action of the dispersive π–π and acid–base interaction in the adsorption of thiophenic compounds. The sample 3 k has a COO content lower than the sample 2100, and COO group is removed completely by the heat treatment. It is found from Figure 7(b) that the reduction of sulfur adsorption capacities for DBT and 4,6-DMDBT is a little bit higher for the sample 3 k than the sample 2100, which suggests that the COO group contributes a little bit more to the sulfur adsorption capacity than the other oxygen functional groups. However, this phenomenon is not found for the T adsorption. The reason for this could be that T has a higher electron density on S atom than DBT and 4,6-DMDBT, which is not sensitive to the change of the strength of acidic sites on the surface of ACs. From all these observations, it could be deduced that a flat adsorption should be dominant, in which the thiophenic compounds lie in graphene layers which are the pore walls of ACs, causing the dispersive π–π between the hydrocarbon part of the thiophenic compounds and the graphene layers and also the acid–base interaction between the lone electron pair of S atom and the C atoms linked to O atoms. As a comparison, plug-in adsorption, the out-of-the-plane interaction of the S atom in the thiophenic compounds with COH and COOH via an acid–base interaction, is less likely to occur since the interaction in the case of the flat adsorption is energetically more favorable than that of the plug-in adsorption, an out of plane acid–base interaction. Figure 8(a) shows the correlation of the sulfur adsorption capacities for T, DBT, and 4,6-DMDBT to the pore volumes of pores within the pore size intervals for the ACs before and after the heat treatment, and Figure 8(b) shows the reduction of sulfur adsorption capacities for T, DBT, and 4,6-DMDBT versus the reduction of oxygen content by the heat treatment for the model fuels with 10 wt% benzene. The results from Figure 8 are quite similar to those from Figure 7 except that the sulfur adsorption capacity is decreased by adding 10 wt% benzene in the model fuels, which could be ascribed to a competing effect of benzene with the thiophenic compounds due to their structure similarity. The sulfur adsorption capacity for DBT adsorption decrease more obviously than those for T and 4,6-DMDBT because benzene has the most similar structure with the aromatic part in DBT. Ania and Bandosz (2005) and Seredych and Bandosz (2010) also reported that the acidic sites have a linear correlation with the thiophenic adsorption, but they estimated the acidic sites using the Boehm method that eliminates the ether-type oxygen or carbonyl groups. The importance of the dispersive π–π interaction in thiophenic adsorption is also found by Bu et al. (2011), but they stressed the importance of mesopores.
(a) Correlation of the sulfur adsorption capacities for T, DBT, and 4,6-DMDBT to the pore texture of activated carbons before and after the heat treatment and (b) the reduction of sulfur adsorption capacities for T, DBT, and 4,6-DMDBT versus the reduction of oxygen content by the heat treatment for the model fuels without benzene. (a) Correlation of the sulfur adsorption capacities for T, DBT, and 4,6-DMDBT to the pore texture of activated carbons before and after the heat treatment and (b) the reduction of sulfur adsorption capacities for T, DBT, and 4,6-DMDBT versus the reduction of oxygen content by the heat treatment for the model fuels with 10 wt% benzene.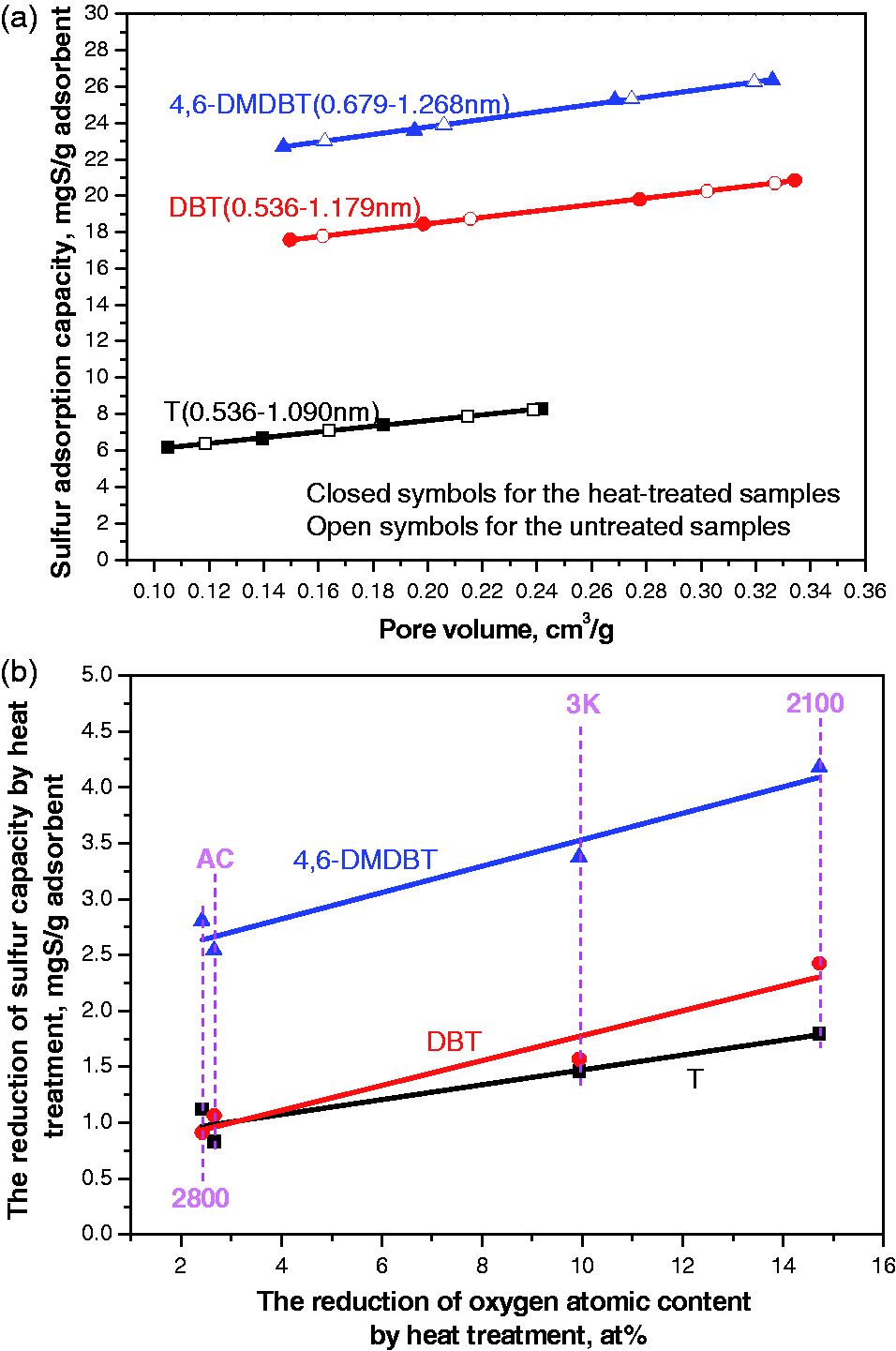

Conclusion
The micropore filling is the dominant adsorption mechanism for the thiophenic compounds in the activated carbons with or without the heat treatment. The optimum micropore size for the thiophenic adsorption increases with molecular size of a thiophenic compound. The flat adsorption is the most possible geometry in the adsorption. In the flat adsorption, the interaction of the basic S atom and the acidic C atoms linked with O atoms and that of the hydrocarbon part of the thiophenic compound and graphene layers on the pore wall of the activated carbons jointly determine the adsorption capacity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Capacity Building Program of Shanghai Local Universities (No. 12160503600), the First-class Discipline Construction Fund of Shanghai Municipal Education Commission (No. J201212), Nature Science Foundation of China (No. U1332107) and Key Discipline Construction Fund of Composite Materials of Shanghai Institute of Technology (No. 10210Q140001).

