Abstract
As one of the promising adsorbents, activated carbon fibers were modified in order to enhance their sulfur dioxide removal efficiency by depositing strong oxidants. Potassium permanganate (KMnO4) was found to be an effective promoter for activated carbon fibers. The optimum content of KMnO4 on activated carbon fibers was 43.34 wt% prepared from 6 wt% KMnO4 solution. The composition of the challenge gas was found to affect the SO2 adsorption capacity of KMnO4 modified activated carbon fibers. The presence of water in the mainstream improved the desulfurization properties. Prehumidification of the challenge gas increased the breakthrough capacity. KMnO4 modified activated carbon fibers were durable in desulfurization properties during the wet-laying process and was made into activated carbon fiber papers with copolyester as the binder. KMnO4-ACFP was found to be able to maintain the SO2 capacity of KMnO4 modified activated carbon fibers with a breakthrough capacity of 76 mg SO2/g modified activated carbon fiber and 90% saturation capacity of 186.75 mg SO2/g modified activated carbon fiber. Sulfur content on KMnO4 modified activated carbon fibers was increased to 9.47 wt% from 0.045 wt% during the adsorption process which proves the adsorption of sulfur on fiber adsorbents.
Introduction
Activated carbon is a well-known adsorbent because of its capability to remove particulate materials, heavy metals, organic materials, and other air toxics in filtration. Among the different shapes of activated carbons, activated carbon fibers (ACFs) are one of the most promising adsorbents which have been studied for manufacturing, structure, and adsorption performances at conventional scale as well as nanoscale. Compared with other activated carbon materials, ACFs have the advantages of small diameter, less blockage, lower pressure drop, faster adsorption rate, and flexibility [1–10]. Removal of sulfur-containing contaminant in gas stream is one of the significant applications of activated carbons. In order to enhance their adsorption properties, they are modified with a variety of approaches and promoters, such as heat treatments and modification with metal [11–14].
Heat treatment and impregnation of metal require high temperature and inert gas, which are energy consuming. Impregnating ACFs with alkali and oxidants will address this problem without the need of nitrogen as the protecting gas and high temperature. However, alkali cannot stand wet-laying process in which activated carbons are subjected to water, which limits the application of ACFs [3]. In this paper, strong oxidants, potassium permanganate (KMnO4) and potassium chlorate (KClO3) are investigated as desulfurization promoters and applied onto ACFs. Modified ACFs are found to possess increased desulfurization capability. Preparation of KMnO4 and KClO3 modified ACFs do not need high-temperature heat treatment, which is regarded as an energy-saving approach in comparison with metal treated and heat modified ACFs. Strong activated carbon fiber papers (ACFPs) were manufactured from modified ACFs which can be used as filter media for filtration systems.
Experimental
Materials
Porous properties of the materials used.
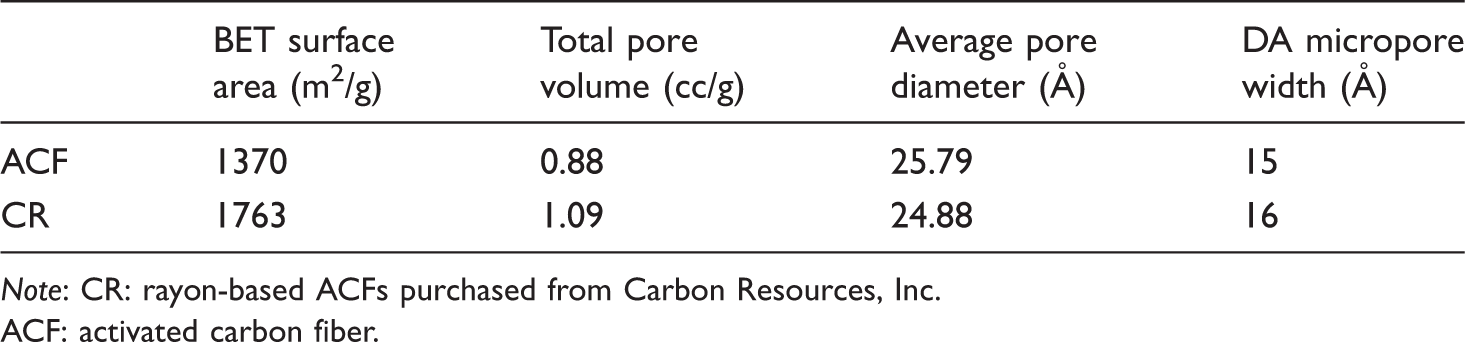
Note: CR: rayon-based ACFs purchased from Carbon Resources, Inc. ACF: activated carbon fiber.
Manufacturing of ACFP
Considering the advantage of flexibility of ACFs, they are made into paper shape, which can be used as filter media directly, through the wet-laying process [3]. Desulfurization promoters are able to stand the wet-laying process. ACFP was manufactured from the slurry of ACFs with binding agents using the wet-laying process with the help of surfactant which was used to increase dispersion of ACFs and binder fibers in the slurry. Cellulose fibers and bicomponent polyester (CoPET) fibers were used as binding agents to optimize the physical and mechanical properties of ACFPs, such as tensile strength and uniformity. The strength of ACFP was increased with the improvement in uniformity. Different compositions of ACFP with several curing conditions were studied. The optimum combination and curing conditions were found to be 70% ACF/30% CoPET and 20 min curing at 200 ℃, which was applied to potassium permanganate modified ACFs to manufacture paper (K-ACFP) for the purpose of desulfurization [3].
Surface morphology and element analysis
Surface characteristics of bare ACFs, modified ACFs, and ACFP prior to and after SO2 adsorption were investigated on scanning electron microscope (SEM, Zeiss® EVO 50). Sulfur element contents on fiber surfaces were analyzed on SEM/EDS system (JEOL 7000 FESEM).
Desulfurization analysis
Desulfurization analysis of modified ACFs prior to and after washing procedure and ACFP was conducted on SO2 adsorption test bench developed for this purpose [3]. Modified ACFs and KMnO4-ACFP (0.5 g) were exposed to 50 ppm SO2 carried by humidified air with a face velocity of 33 cm/min at 20 ℃. SO2 adsorption capabilities and breakthrough curves are used to evaluate desulfurization properties of the samples. To study the effect of challenge gas conditions on desulfurization process, KMnO4 modified ACF was also exposed to dry nitrogen, dry air, and humidified nitrogen. The desulfurization test bench was designed based on the ASTM D6646 as described in another article [3,10].
Results and discussion
Sulfur dioxide adsorption properties of modified ACFs
Potassium chlorate modified activated carbon fibers (ACF/KClO3)
SO2 breakthrough capacities of modified ACFs with KClO3 through impregnation are shown in Figure 1. SO2 breakthrough capacity is promoted almost linearly by loading KClO3 on ACFs characterized with equation y = 1.5869x + 3.1853 with a correlation factor of 0.8154. However, KClO3 is not durable on ACFs and easily removed by exposure to water. ACF/KClO3 almost lost all SO2 adsorption capacity during washing and thus it is not suitable for wet-laying process.
SO2 breakthrough capacity of ACF/KClO3 before and after water exposure.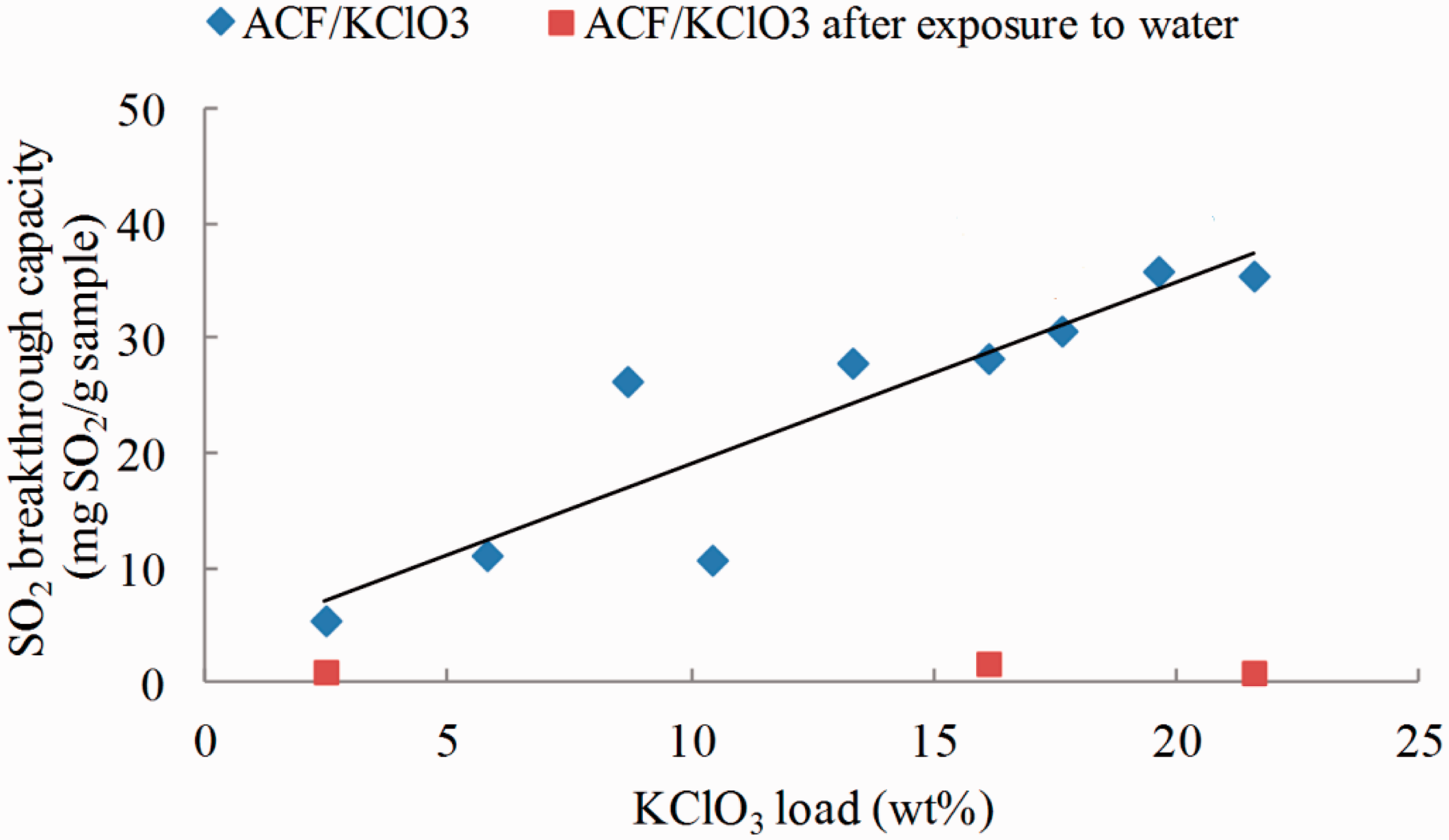
Potassium permanganate modified activated carbon fibers (ACF/KMnO4)
SO2 breakthrough capacities of KMnO4 modified ACFs are shown in Figure 2. The capability of ACF/KMnO4 in SO2 purification is slightly affected by washing indicated by similar SO2 breakthrough capacity prior to and after washing, which indicates the suitability and durability of ACF/KMnO4 as a candidate for wet-laying process to make ACFP. The maximum SO2 capacity of ACF/KMnO4 was obtained with 43.34 wt% KMnO4 load.
SO2 breakthrough capacity of ACFs modified with different ratio of KMnO4.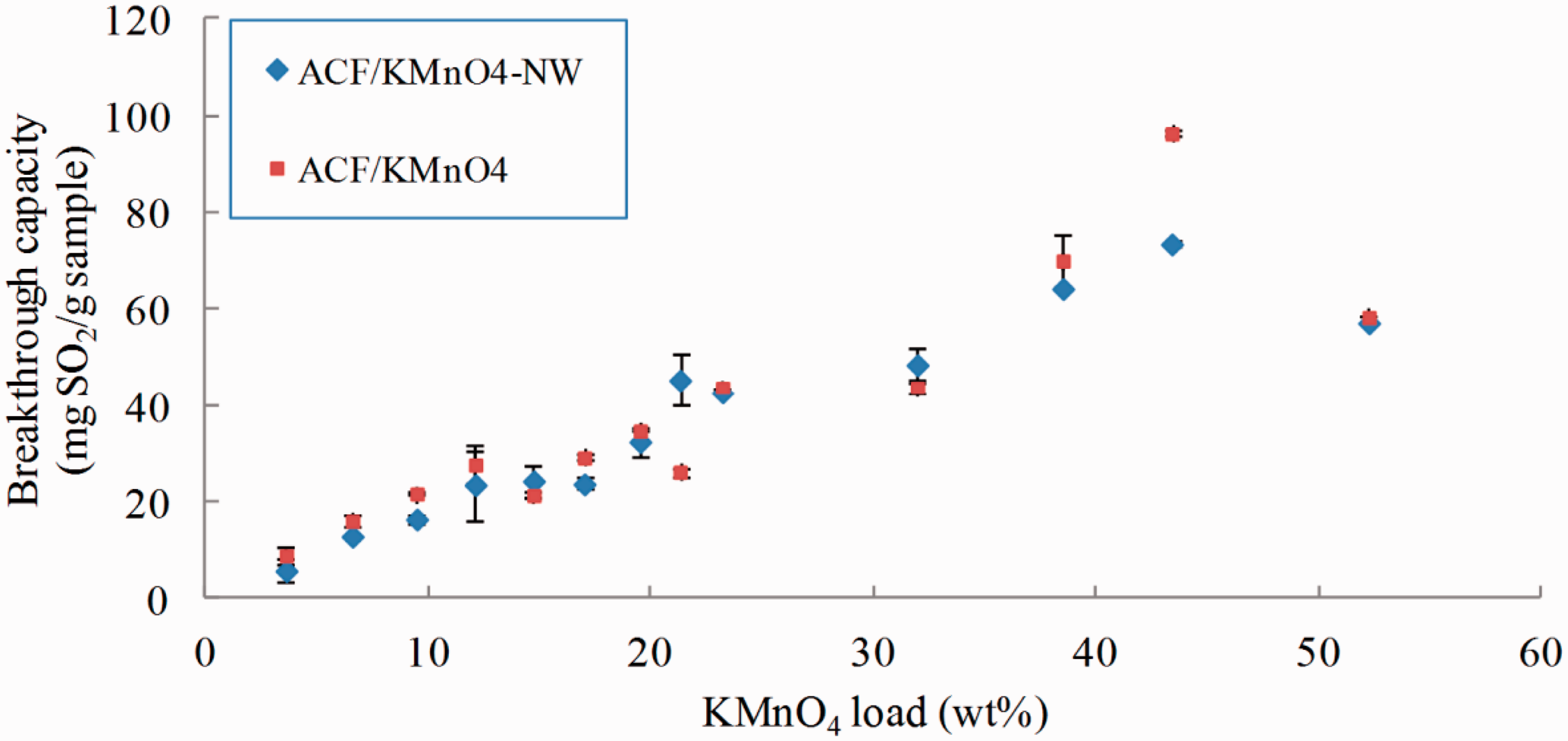
When the same amount of KClO3 and KMnO4 (e.g. 15 wt%) was applied to ACFs, ACF/KMnO4 shows a higher breakthrough capacity, about 40 mg SO2/g sample, compared to about 27 mg SO2/g sample of ACF/KClO3. The breakthrough curves of ACF/KMnO4 prior to and after exposure to water are shown in Figure 3. They are similar to each other except the slightly increased slope of the washed sample, which is probably due to the increased contact efficiency caused by redistribution of KMnO4 on ACF during washing. Although the breakthrough capacity of the washed sample is higher, its saturation capacity of 210 mg SO2/g sample is lower than that of 317 mg SO2/g sample before washing.
Breakthrough profiles of ACF/KMnO4 before and after exposure to water.
It has been reported that experimental parameters affect desulfurization properties including gas component [15]. ACF/KMnO4 (43.34 wt%) was exposed to different challenge gases, namely, SO2, SO2 + air, SO2 + H2O, and SO2 + H2O + air; their breakthrough profile characteristics are compared in Figure 4, which exhibits drastically different SO2 capacities and breakthrough curve patterns. Breakthrough profiles of SO2 + air and SO2 + N2 appear to be close with similar adsorption rates. Without the presence of water, presence of oxygen in air alone does not dramatically affect the adsorption process. When water vapor is present, SO2 breakthrough capacity was greatly increased indicating that water vapor accelerated the SO2 removal process. Additional oxygen contributes to the reaction more with the presence of water vapor. In other words, both the presence of oxygen and water vapor promote the desulfurization ability of ACF/KMnO4 while co-presence of them enhances the ability further. The improvement of SO2 capacity caused by water vapor appears to be more than that caused by oxygen.
Breakthrough profiles of ACF/KMnO4 in different challenge gases.
The potassium permanganate solutions used for ACF/KMnO4 preparation were all 6 wt% so far. It is possible that the desulfurization capability is influenced by the precursor concentration. A lower KMnO4 concentration (2 wt%) was used to treat ACFs and different weight ratios of KMnO4 were obtained. The adsorption properties of ACF with 20, 33, and 43 wt% KMnO4 prepared from 2 wt% solutions are illustrated in Figure 5. Their breakthrough capacities were calculated to be 34, 59.7, and 60.8 mg SO2/g sample, respectively. With the increase of KMnO4 content, SO2 saturation capacity increased but the breakthrough capacity increased first and then leveled off. For the same KMnO4 load (43 wt%) on ACF, samples prepared from 2 and 6 wt% solutions showed similar breakthrough curves and saturation process; however, ACF impregnated with 6 wt% concentration of KMnO4 solution exhibited a longer breakthrough time as shown in Figure 5.
Breakthrough profiles of ACF/KMnO4 prepared from 2 wt% KMnO4 solutions.
It was reported that prehumidification before activated carbons were exposed to SO2 improved the SO2 removal efficiency by forming a water film over carbon surface where the conversion of sulfur trioxide to sulfuric acid and elution of aqueous sulfuric acid from carbon surfaces were promoted. KMnO4 treated ACFs were exposed to humidified air (91% relative humidity) for 1 h before starting the SO2 adsorption test. The breakthrough time was shortened in comparison with dry ACF/KMnO4 (Figure 6). The adsorption rate was initially similar which is indicated by a similar slope of the breakthrough curve. After 80% of SO2 was released from the reactor, the adsorption rate of the prehumidified sample slowed down and became lower than that of the dry samples. On the other hand, the saturation capacity (85%) of prehumidified ACF/KMnO4 of 215 mg SO2/g sample is higher than that of dry samples of 205 mg SO2/g. Furthermore, as the adsorption process continues, a higher saturation capacity would be obtained if samples were prehumidified.
Sulfur dioxide adsorption properties of humidified ACF/KMnO4 in comparison with dry sample.
ACFP and sulfur dioxide adsorption properties
The SO2 adsorption profile is shown in Figure 7 in comparison with that of ACF/KMnO4 at fiber stage. When the same amount of sample size was applied, KMnO4-ACFP showed shorter breakthrough time than ACF/KMnO4 because of presence of 30 wt% bicomponent polyester. If copolyester binder is excluded during calculation, the breakthrough capacity of KMnO4-ACFP with respect to ACF/KMnO4 is 76 mg SO2/g modified ACF which is similar with breakthrough capacity of 74.78 mg SO2/g modified ACF before they are made into paper. Ninety percent saturation capacity of KMnO4-ACFP with respect to fibers is 186.75 mg SO2/g modified ACF which is less than that of fibers of 210.83 mg SO2/g modified ACF. KMnO4-ACFP exhibited quicker saturation illustrated by the steeper breakthrough curve, which is probably due to more efficient contact when ACFs are diluted by binders.
Breakthrough profiles of KMnO4-ACFP in comparison with ACF/KMnO4 with 50 ppm SO2 challenging gas carried by humidified air.
Fused Co-PET, which is used as binding agent, formed strong interaction in the KMnO4-ACFP. However, it is possible that the desulfurization capability of ACF/KMnO4 is reduced due to the existence of joint points where fused binder fibers reduced effective reaction area on fiber surface for SO2 removal. To investigate the effects of joints, the formed wet mat was dried naturally in air rather than at high temperature. As a result, binder fibers remained as they were and there were not joints in the fiber network. Fibers in unfused KMnO4-ACFP were individual fibers which were subjected to SO2 under the same conditions with those in fused KMnO4-ACFP. The resulting 90% saturation capacity was 217 mg SO2/g modified ACF, which is 14.3% higher than fused KMnO4-ACFP and 4.1% higher than ACF/KMnO4 at the fiber stage.
Morphology of modified ACFs
The SEM images of ACF/KMnO4 with different chemical weight ratios are shown in Figure 8. At a low KMnO4 load (6.55 wt%), the distribution of KMnO4 on fiber surfaces is not uniform: some areas are smooth while KMnO4 is dense on some other areas causing coarse fiber surfaces. As the weight ratio increased to 23.16 wt%, the uniformity of KMnO4 distribution was enhanced. It seems that fiber surfaces were covered by KMnO4 at the weight percentage of 43.34 wt% and after exposure to water, KMnO4 seemed to be redistributed uniformly, which may contribute to efficient contact between the adsorbate and oxidant during adsorption tests, which is consistent with the sharper breakthrough curve as discussed in the previous section.
Surface morphology (×2000) of ACF/KMnO4. (a) 6.55 wt% KMnO4, (b) 23.16 wt% KMnO4, (c) 43.34 wt% KMnO4, (d) 43.34 wt% KMnO4 after exposure to water.
Surface morphology of ACF/KMnO4 (34.34 wt% KMnO4) after adsorption tests in different challenge gases (SO2 + N2, SO2 + air, SO2 + H2O + N2, and SO2 + H2O + air) was observed under SEM as shown in Figure 9. As ACF/KMnO4 was exposed to SO2 only, the fiber surfaces were similar to the fresh sample but the challenge gas mixture of SO2 and O2 without water resulted in rougher surfaces. The coarse surfaces are probably because of incomplete reaction between SO2 and KMnO4 with unreacted oxidant remaining on the fiber surfaces. However, the other two spent samples, which were exposed to the water-containing challenge gases, exhibited much smoother surface with few particles attached, which may be due to KMnO4 being consumed by SO2 during the reaction.
ACF/KMnO4 surface morphology (×2000) after exposure to (a) SO2 + N2, (b) SO2 + air, (c) SO2 + H2O + N2, and (d) SO2 + H2O + air.
Surface morphologies of fresh and spent KMnO4-ACFP were observed with SEM as shown in Figure 10. After SO2 adsorption test, ACF/KMnO4 fiber surfaces became smoother with some scale-like substances which are probably the adsorption products. CoPET binder fibers are fused and bond ACFs together to form a strong network in KMnO4-ACFP, which makes it a candidate for filter media.
SEM images of KMnO4-ACFP: before (left) and after (right) SO2 adsorption tests (×2000).
Energy dispersive X-ray (EDS) analysis
The EDS analysis was performed on a SEM/EDS system to analyze the element composition on ACF/KMnO4 fiber surfaces both before and after SO2 adsorption tests with the carrier gas of humidified air (Figure 11). Sulfur content on ACF/KMnO4 was initially very low (0.045 wt%), but increased to 9.47 wt% after adsorption tests. Sulfur species were caught by adsorbents effectively and the goal of desulfurization was achieved.
EDS element signal before (left) and after adsorption in the challenge gas of SO2 carried by humidified air.
Conclusions
Potassium permanganate was found to be an effective promoter for ACFs in sulfur dioxide removal. The optimum load of KMnO4 on ACFs was 43.34 wt% prepared from 6 wt% KMnO4 solution.
The composition of the challenge gas was found to be an influential factor in determining sulfur dioxide adsorption capacity of ACF/KMnO4. The presence of water improved the desulfurization properties dramatically. Prehumidification of challenge gas increased its breakthrough capacity.
KMnO4 modified activated fibers are durable during wet-laying process and can be made into ACFPs with copolyester as the binder which formed joints contributing to integrity and strength of ACFP. KMnO4-ACFP showed high performance on sulfur dioxide removal with a breakthrough capacity of 76 mg SO2/g modified ACF and 90% saturation capacity of 186.75 mg SO2/g modified ACF.
Sulfur content on ACF/KMnO4 increased to 9.47 wt% from 0.045 wt% during adsorption process which testified the adsorption of sulfur by fiber adsorbents.
Footnotes
Acknowledgements
The authors would like to thank the colleagues at the Center for Microfibrous Materials Manufacturing for their help.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
