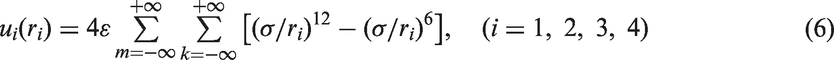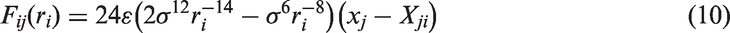Abstract
Adsorption and diffusion of lithium-ion on and between graphene layers are investigated by an analytical model, employing a pairwise potential, which can be approximated by the Lennard–Jones potential to express the interaction between lithium-ion and each carbon atom of graphene. The equilibrium position and binding energy of lithium-ion at three particular adsorption sites (hollow, bridge, and top) are calculated, and the adsorption stability is discussed. The results show that hollow site is the most stable adsorption site, and top site is the most unstable. The adsorption and diffusion of lithium-ion on different sizes of monolayer graphene are investigated and proved to be size and edge dependent. Moreover, lithium-ion would rather diffuse on the surface of graphene than through a hexagonal carbon ring to the other side no matter what the graphene sheet size is. In addition, two kinds of vacancy defects in graphene are considered to study the diffusion of lithium-ion. The vacancy defect can improve energy barrier, and if a vacancy defect is big enough, lithium-ion migrating through the vacancy area from one layer to another is feasible. The possible applications of present study include rechargeable lithium-ion graphene battery and Li storage in carbon material.
Introduction
Graphene, the thinnest known two-dimensional (2D) nanomaterial, has many unique characteristics, such as high electron mobility, the lowest resistivity, and extreme mechanical strength, etc. (Babichev et al., 2013; Koshino, 2009; Lau et al., 2012; Voloshina et al., 2011; Zhang and Zhao, 2013). As such, it has become a promising candidate for applications in nanodevices and been widely investigated in the field of biology, chemistry, and physics (Chi and Zhao, 2009; Chi and Zhao, 2012; Huang et al., 2014; Li et al., 2014; Liu et al., 2013; Zhang et al., 2013). Especially, the interaction between Li-ion and graphene has been a subject of intensive study motivated by their potential for the development of rechargeable Li-ion graphene battery and Li storage in carbon material (Buldum and Tetiker, 2013; Hassoun et al., 2014; Kucinskis et al., 2013; Lee et al., 2013; Liu et al., 2014; Sharifi et al., 2012; Yoo et al., 2008). However, there is still a common unsolved major scientific question for these applications. It is to develop an analytical model to quantitatively describe the adsorption and diffusion of atom/ion on and between graphene layers with a finite dimension, which can be further applied to the interaction between nanoparticles or atomic force microscopy (AFM) and graphene surface.
For this problem, a multitude of theoretical and experimental researches have been made, and quite a few interesting research results are achieved. Density functional theory (DFT) method was performed to investigate the surface diffusion and coverage effect (calculated by dividing the number of Li atoms by the number of carbon atoms in graphene) of Li on graphene (Ji et al., 2013). The adsorption phenomena was considered using a wide range of coverage of Li on graphene and different surface models as supercells, and the stability order of adsorbed sites were found to be hollow > bridge > top. Moreover, the equilibrium distance of Li above graphene surface followed the general trend (hollow < bridge < top), in which the distance increased with increasing coverage. For the Li diffusion, the path which Li migrated from a hollow site (H-site) to a neighboring H-site through the bridge site (B-site) between them should be the minimum energy path. Using DFT method, the feasibility of modifying graphene to store Li more efficiently was discussed (Liu et al., 2013). It was found that certain structural defects in graphene could stably bind Li, yet a more efficacious approach was through substitution doping with boron (B). In particular, the layered C3B compound stood out as a promising Li storage medium, and its capacity was about twice larger than graphite, with comparable power density and small volume variation during discharge/charge cycles. These results are helpful to clarify the fundamentals of Li storage in low-dimensional materials, and to guide the reasonable design of nanodevices for energy storage. By the same method, Li adsorption and diffusion on graphene with point defects was studied (Zhou et al., 2012). The results showed that Li preferred the H-site above the center of an octagonal (heptagonal) ring to the one of pentagonal (hexagonal) ring if they existed in defective graphene. The interaction between a Li and a divacancy (DV) defect was attractive, and it was stronger than that between a Li and a Stone–Wales (SW) defect. It suggested that the intentional creation of point defects before adsorbing Li would significantly enhance the Li adsorption. Successively, Li adsorption and diffusion on graphene with grain boundaries were studied by the same researchers (Zhou et al., 2014). They concluded that not only the edge, point defect, and dopant, but also the grain boundary could enhance the Li adsorption of graphene, which would be very helpful for the design of graphene-based anode materials of Li-ion secondary batteries (LIBs).
Apart from density function theory method, molecular dynamics (MD) simulation methods have also been widely applied to study the interaction between Li-ion and graphene. The diffusion processes of Li-ion on a fluorinated graphene (F-graphene) surface were investigated by means of a direct molecular orbital–molecular dynamics (MO-MD) method (Tachikawa, 2008). It was found that the diffusion coefficient of Li-ion on F-graphene surface was close to that of hydrogen graphene (H-graphene), but the thermal behavior on the F-graphene surface was much different from that on the H-graphene surface. The direction of Li-ion diffusion could be controlled by fluorinated substitution (F-substitution) on the graphene sheet around room temperature. But at higher temperatures, the Li-ion diffused freely on surface and edge region of F-graphene. What's more, the effects of fluorine and hydrogen atom substitution of graphene edge site (F-edge region and H-edge region) on the diffusion of Li-ion were studied by direct MO-MD (Tachikawa et al., 2010). It was found that the Li-ion diffused freely on the surface, but the Li-ion could not approach the F-edge region of the surface because of repulsive interaction with positive charge of C–F carbon atom. On the other hand, the C–H carbon atom had a negative charge. This result strongly suggested that the Li-ion could preferentially go in and out of the H-edge region in a FH-graphene.
By comparison, there are relatively few experimental researches on adsorption and diffusion of Li or Li-ion on graphene surface. Li diffusion pathways with two types of graphene samples prepared by chemical vapor deposition (CVD) were explored (Yao et al., 2012). It was discovered that electrochemical reaction of the electrode (substrate/graphene) was not only related to the number of graphene layers but also relied on the defect sites on the basal plane of graphene. These results could help us in designing high capacity electrodes for Li-ion batteries. LiFePO4-graphene composites were successfully synthesized by the hydrothermal method (Nguyen and Gu, 2014). Different amounts of graphene were added to LiFePO4 to improve the Li-ion-diffusion ability of LiFePO4. It was confirmed that the extraction of Li-ions in LiFePO4-graphene was better than that in pure LiFePO4. LiFePO4-graphene delivered the fastest Li-ion diffusion and showed the highest specific discharge capacities.
To quantitatively understand Li-ion adsorption and diffusion on and between graphene layers from the prospective of atomic-scale, a discrete analytical model is put forward in this work based on the Lennard–Jones (L-J) potential. The equilibrium position, binding energy, energy barrier, and adsorption stability of Li-ion on monolayer graphene are calculated and analyzed. Moreover, the diffusions of Li-ion in bilayer graphene and on defective monolayer graphene are also considered.
The interaction between an adsorption atom/ion and graphene
First, a monolayer graphene is assumed to be perfect, flat, and infinite compared with atomic-scale. Because of the periodic structures of hexagonal ring, the positions of carbon atoms in monolayer graphene can be easily expressed in the Cartesian coordinate system as shown in Figure 1. The basic unit consists of numerous regular hexagons carbon rings, which are continuously arranged together along the y-axis, and four kinds of different carbon atoms along x-axis are defined in the unit as shown in Figure 1. An infinite graphene plane can be obtained by copying and moving the unit along the x-axis. Moreover, there are three particular adsorption sites of atom/ion on graphene surface, which are hollow (H-), bridge (B-), and top (T-) sites, respectively (as shown in Figure 1).
Schematics of graphene plane with a structural unit, which is defined by four kinds of carbon atoms along x-axis and three particular adsorption sites: hollow (H-), bridge (B-), and top (T-).
As mentioned above, the Cartesian coordinates of the four kind of different carbon atoms, (A
i
, B
i
, C
i
), (i = 1, 2, 3, 4), in the graphene can be expressed as following forms:
Commonly, the average interaction potential per atom/ion on graphene is defined by the total energy of an atom/ion, a sheet of graphene, and the graphene with atom/ion adsorption, which consider the effects of temperature, chemical bond, charge transfer, van der Waals interaction, etc. (Buldum and Tetiker, 2013; Chi and Zhao, 2009; Chi and Zhao, 2012; Ji et al., 2013; Sharifi et al., 2012; Zhou et al., 2014). In this article, the Cartesian coordinates of carbon atoms in graphene has been expressed above. For the convenience of discussion, the interaction potential is only defined by van der Waals interaction. Even so, the model in this article is also suitable for taking into account the effects of other factors mentioned above. Two empirical potential commonly used are the L-J potential and the Morse potential. By referring the reader for detail of the Morse potential and its applications (Qian et al., 2002; Wang et al., 1991), this article adopts the form of 6-12 L-J potential to determine the van der Waals interaction potential between an adsorption atom/ion and a carbon atom in the graphene,
Therefore, the total interaction potential between the adsorption atom/ion and the graphene can be defined as following form,
Furthermore, the normal and lateral interaction forces between the atom/ion and the graphene can be derived by
Results and discussion
Equation (8) defines the interaction potential between a single atom/ion and graphene via the L-J potential. Obviously, it's dependent on the position of the adsorption atom/ion on graphene and the parameters of L-J potential. In this work, Li-ion is selected as the adsorption ion to study the interaction characteristics. For a Li-ion, the L-J potential parameters can be specified as σ = 0.2473 nm, ε = 0.00433 eV (Chan and Hill, 2010; Hill et al., 2007a,b). As known from equation (5), with the increase of the distance between Li-ion and carbon atom, the interaction of them would be gradually weakened. Therefore, reasonable value ranges of m and k should be determined for approximate calculation.
There are three particular adsorption sites, hollow (H-), bridge (B-), and top (T-), as shown in Figure 1. When the interaction forces in the z-direction are equal to zero, the equilibrium heights (h0) at the three adsorption sites can be calculated, which are 0.23225, 0.24099, and 0.26324 nm, respectively, with the values of |m| ≥ 5 and |k| ≥ 5. It has a favorable agreement with the results in the literatures reported before (Chan and Hill, 2010; Ji et al., 2013). What is noteworthy is that the equilibrium height is quite important for the design of Li-ion battery based on graphene cathode, which can be used in predicting the minimum interlayer spacing 2h0 of two parallel graphene so that the embedded Li-ion undergoes no net force. According to our theoretical results, the minimum interlayer spacing for Li-ion should be approximate 0.46–0.52 nm.
Binding energy u0 of Li-ion on the three particular adsorption sites, with different values of m and k.
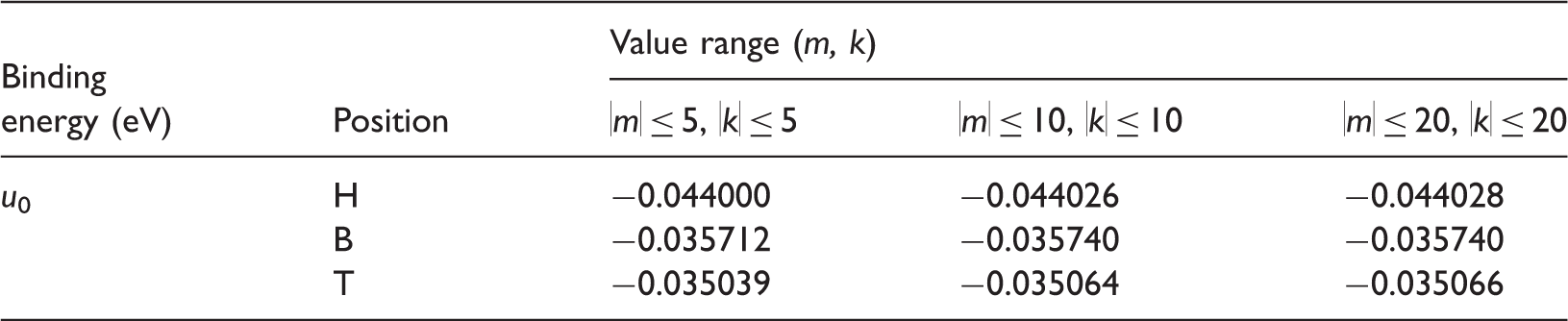
The equilibrium height and binding energy at the H-site are the lowest, followed by the B-site, and those at the top site (T-site) are the highest. Thus, H-site is the most stable adsorption site, and T-site is the most unstable. The results are in agreement with the counterparts in the other literatures (Chan and Hill, 2010; Ji et al., 2013; Zheng et al., 2011), which means that the theoretical model in the article is reliable to study the adsorption and diffusion of atom/ion on graphene surface at the atomic-scale.
Given that the geometric sizes and edges could have influence on the adsorption and diffusion of an atom/ion on the graphene surface, five kinds of monolayer graphene sheets with different geometric sizes (n = 1, 7, 19, 37, and 61, where n is the number of hexagonal carbon rings as shown in Figure 2(a) to (e)) are constructed to analyze the interaction between the adsorbing Li-ion and graphene. The equilibrium height and binding energy of Li-ion on H-site of the graphene with different sizes are calculated and listed in the Table 2. The results coincide to the work previously reported (Chan and Hill, 2010), where predicted that equilibrium height and binding energy on H-site are 0.25 nm and 0.038 eV, respectively. It can be found that the equilibrium height and binding energy exhibit size effects. They both reduce with the increase of geometric sizes of the graphene until the geometric sizes are big enough. For five geometric sizes in the work, the variations of equilibrium height and binding energy can be neglected when the geometric size of graphene is n = 61. In other words, when the distance between a Li-ion and a carbon atom in graphene is about 10 times greater than the length of carbon–carbon bond, the interaction of them can be almost ignored.
Schematics of the graphene with different sizes (n represents the number of hexagonal carbon rings, and n = 1, 7, 19, 37, 61 in the (a) to (e), respectively), and (f) two kinds of vacancy defects in graphene (n = 61). (g) Bilayer graphene with size n = 61 and interlayer spacing d. Equilibrium height and binding energy on H-site of graphene sheet with different geometric size (n = 1, 7, 19, 37, and 61).

Furthermore, when a Li-ion diffuses along the x and y directions on the surface of the graphene with different geometric sizes, the variations of interaction potential can be illustrated in Figure 3(a) and (b) with respect to the diffusion paths in the x and y directions. The perpendicular distances between Li-ion and H-site of graphene are set to the equilibrium heights as listed in the Table 2. Apparently, with the increase of graphene sheet size, the interaction potential is tending towards periodic change. However, in the edge field of graphene, the interaction potential exhibits significant increase, i.e., the diffusion of Li-ion near the edge field has to overcome a higher energy barrier than in the central field of graphene sheet.
The variations of interaction potential between the Li-ion and monolayer graphene with different geometric sizes along the (a) x-direction, (b) y-direction, and (c) z-direction, respectively.
In addition, the migration of Li-ion in the z-direction of monolayer graphene is also discussed. Assuming that the Li-ion migrates through a hexagonal carbon ring from below up for different size graphene, the variations of interaction potential are shown in Figure 3(c). It can be found that the variation trends of the interaction potential are almost same for five kinds of graphene sheets. It means when the Li-ion migrates through monolayer graphene, the interaction potential and energy barrier principally depend on the hexagonal carbon ring, and the effect of other carbon atoms is insignificant. Moreover, compared with the results of Li-ion diffusion on graphene surface, the maximum energy barrier of migration in the z-direction is about 79.9 eV, which is three orders of magnitude greater than the energy barriers of in-plane diffusion, i.e., the diffusion of Li-ion through a perfect graphene is more difficult. Therefore, Li-ion would rather diffuse on the surface of graphene than through a hexagonal carbon ring to the other side, regardless of the size of graphene sheet.
As per discussions above, Li-ion prefers to diffuse on the surface of monolayer graphene than to penetrate it. In the same way, Li-ion would diffuse along the interlayer space rather than from one layer to another for a multilayer graphene. Therefore, the interlayer spacing d is an important parameter for Li-ion diffusion in the multilayer graphene, and the energy barrier of Li-ion diffusion should be dependent on its values. For a bilayer graphene with the size of n = 61, three kinds of interlayer spacing are selected, d = 0.36, d = 0.40, and d = 0.46 nm. The diffusion paths are from the center of the bilayer graphene to its edge in the x and y directions. As shown in Figure 4(a) and (b), the interaction potential change periodically in the two directions, and with the increase of layer spacing, the values of energy barrier gradually reduce. Theoretically, the greater interlayer spacing is, the easier Li-ion diffuses in bilayer graphene. As the interlayer spacing is finite, there should exist a minimum values for free diffusion of Li-ion in the bilayer or multilayer graphene. The migration of Li-ion through bilayer graphene (as shown in Figure 4(c)) is similar to that through monolayer graphene. No matter what the value of the interlayer spacing is, the energy barriers of Li-ion penetrating a perfect bilayer graphene are also about 79.9 eV.
The variations of interaction potential between the Li-ion and different interlayer spacing of bilayer graphene (n = 61) in the (a) x-direction, (b) y-direction, and (c) z-direction, respectively.
As we know, the imperfection is inevitable in the graphene because of the restrictions in manufacturing. The defects in the graphene include vacancies of atoms, SW defects and line defects, etc. The influence of these defects on the diffusion of Li-ion on the graphene should be investigated. Two kinds of vacancy defects in a monolayer graphene sheet with the size of n = 61 are taken into account in the work. A single-atom vacancy defect (T1 as shown in Figure 2(f)) is induced by missing a carbon atom in the graphene, and another vacancy is a hexagonal ring defect (T1–T6 as shown in Figure 2(f)), which is induced by missing a hexagonal carbon ring in the graphene.
These two kinds of vacancy defects have been compared with perfectly 2D graphene by diffusion along the ρ-direction (60 ° angle from the x-axis and across defective areas, as shown in Figure 2(f)) and along the z-direction. As shown in Figure 5(a), Li-ion diffuses across the two kinds of defective areas from the center of a carbon ring H1 to another one H2 (as shown in Figure 2(e)) along the ρ-direction. The perpendicular distance of Li-ion and graphene is set to 0.23 nm. Because of the existence of defects, the interaction potential shows apparent variations compared with perfectly 2D graphene. In the center of vacancy defect area, the interaction energy has an obvious decline. However, the energy barriers increase at the boundary of vacancy defect area, i.e., the vacancy defects are against the diffusion of Li-ion when the Li-ion migrates across defective areas of graphene.
The variations of interaction potential between a Li-ion and monolayer graphene sheet (n = 61) with vacancy defects when the Li-ion migrates (a) across defective areas along the ρ-direction and (b) through vacancy defects from different sites along the z-direction.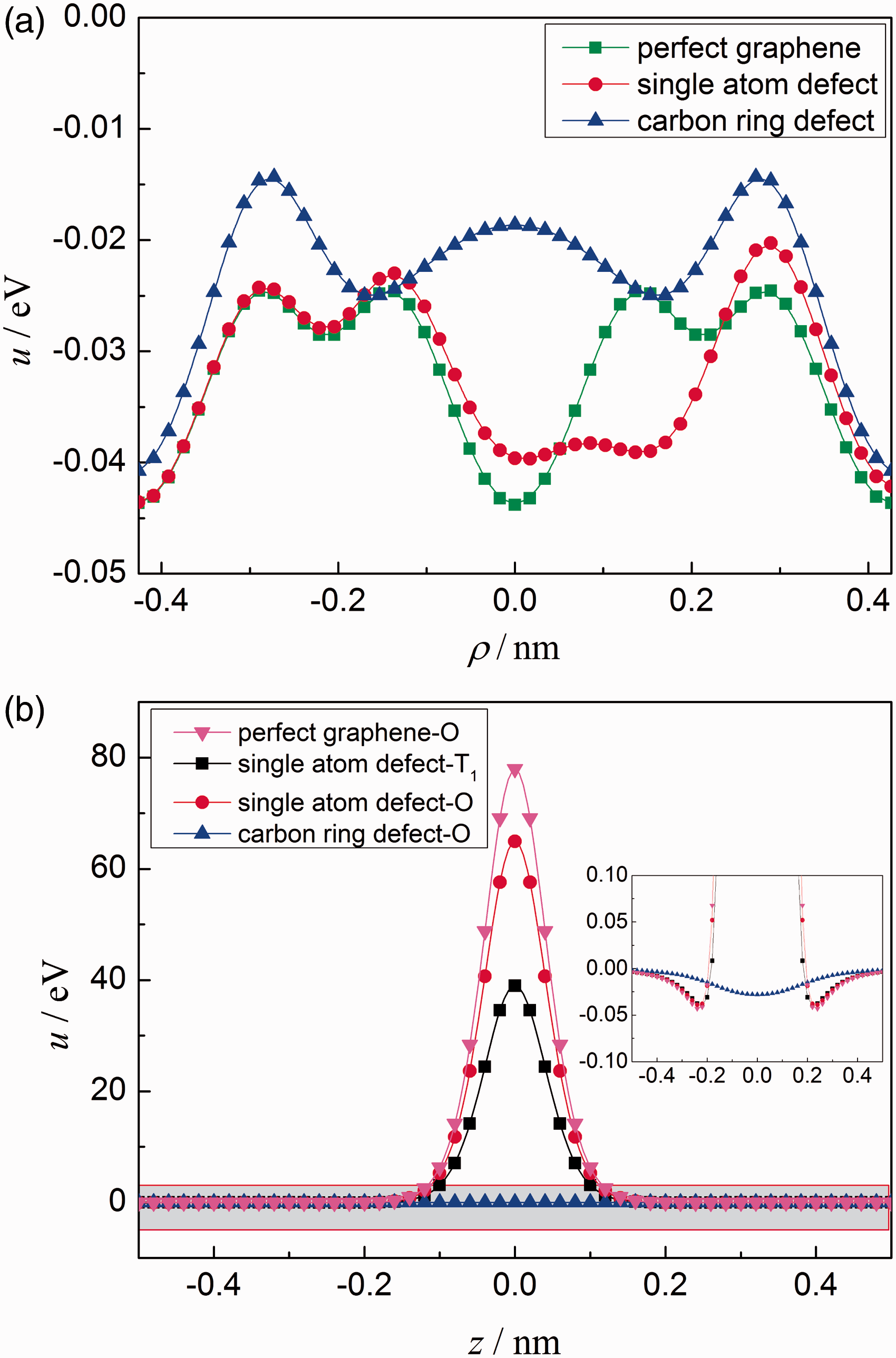
When Li-ion migrates through defective monolayer graphene from O-site and T1-site along the z-direction, respectively, the interaction potentials are plotted in Figure 5(b). Compared with the energy barrier about 79.9 eV for perfectly 2D graphene on the O-site, the single-atom vacancy defect and hexagonal ring defect have significant decline on energy barrier and are 65.0 eV and 0.05 eV, respectively. The energy barrier 0.05 eV is comparable with that of interlayer diffusion. Thus, for a hexagonal ring defect, it is possible that Li-ion migrates through vacancy area from one layer to another. The energy barrier is 39.0 eV when Li-ion migrates through the single-atom vacancy defect form T1-site. These results further confirm that when the Li-ion migrates through the layers of graphene, the interaction potential and energy barrier principally depend on the carbon ring, and the effect of other carbon atoms is insignificant. Moreover, for a vacancy defect big enough, Li-ion can migrate through vacancy area from one layer to another.
Conclusions
Theoretical analyses of interaction potential, equilibrium height, and energy barrier for a single Li-ion on and between graphene layers are presented based on an analytical discrete model, and the mechanism of adsorption and diffusion are revealed. The equilibrium height and binding energy on three particular adsorption sites are calculated, and the adsorption stability are discussed. The order from low to high for both equilibrium height and binding energy is H-site, B-site, and T-site, and H-site is the most stable adsorption site. The adsorption and diffusion of Li-ion on the graphene sheets with different sizes are investigated, and obvious size and edge effects are found. In the edge area of graphene, the energy barrier has a significant increase. Moreover, it is proved that Li-ion would rather diffuse on and between graphene than through a hexagonal carbon ring regardless of the size of graphene sheet, because the energy barriers of Li-ion penetrating a perfect graphene are far greater than that of Li-ion diffusing on and between graphene. In addition, the influence of vacancy defects in graphene is considered to study the diffusion of Li-ion. The interaction potential of Li-ion migrating across defective area has apparent variations, and a hexagonal ring defect has a greater effect than a single-atom vacancy defect. When the vacancy defect is big enough, Li-ion can migrate through the graphene form vacancy defective area to another side.
The results in this article are advantageous to improve the efficiency of drug delivery based on graphene, high-performance rechargeable lithium-ion batteries, and lithium storage in carbon materials, which can be further applied to manufacture more advanced technology equipment to enhance the energy transfer efficiency. However, there is a little inadequacy in the present study. The binding energy of Li-ion on the three particular adsorption sites are smaller than the other works (Zheng et al., 2011; Zhou et al., 2012, 2014), that is probably because the interaction potential is only defined by van der Waals interaction, ignoring the effects of chemical bond, charge transfer, etc. Nonetheless, the adsorption and equilibrium rule of Li-ion on graphene are the same. What's more, the graphene is assumed to be non-deformable and flat, and the influence of temperature and corrugation of graphene are not taken into account (Aitken and Huang, 2010; Chan and Hill, 2010; Gao and Huang, 2011). These factors will be considered in our future works.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work is supported by the National Natural Science Foundation of China (Grant no. 11372216, 11502167), the National Basic Research Program of China (Grant no. 2012CB937500) and the State Key Laboratory for Strength and Vibration of Mechanical Structures of Xi an Jiaotong University (Grant no. SV2015-KF-13).