Abstract
The increased global demand for lithium is rapidly depleting the lithium ore reserves. Therefore, attention has turned to the recovery of lithium from aqueous solutions, such as lithium-containing brine. Compared with other methods of lithium recovery, adsorption is energy efficient and simple to implement, increasing demand for selective lithium adsorbents. In this study, a selective lithium-ion adsorbent, H4Ti5–
Introduction
Lithium is among the lightest and rarest strategic metals.1,2 Specifically, owing to its applications in energy storage, lithium has become key to global development and economic growth.3–10 In addition, lithium and its compounds are used in bauxite ceramics, medicines, metals and alloys, military technology, and battery storage and power generation.11–13 However, as a result of the large-scale use of lithium as cathode and anode material in lithium-ion batteries for consumer electronics and electric vehicles, it is being rapidly exhausted,14–17 and the global demand for lithium sources, which are primarily lithium ore and salt-lake brine, has increased significantly.18,19 As a result of the depletion of high-grade lithium ore reserves, lithium extraction from water bodies, such as seawater and saltwater lakes, has drawn increasing attention.20,21
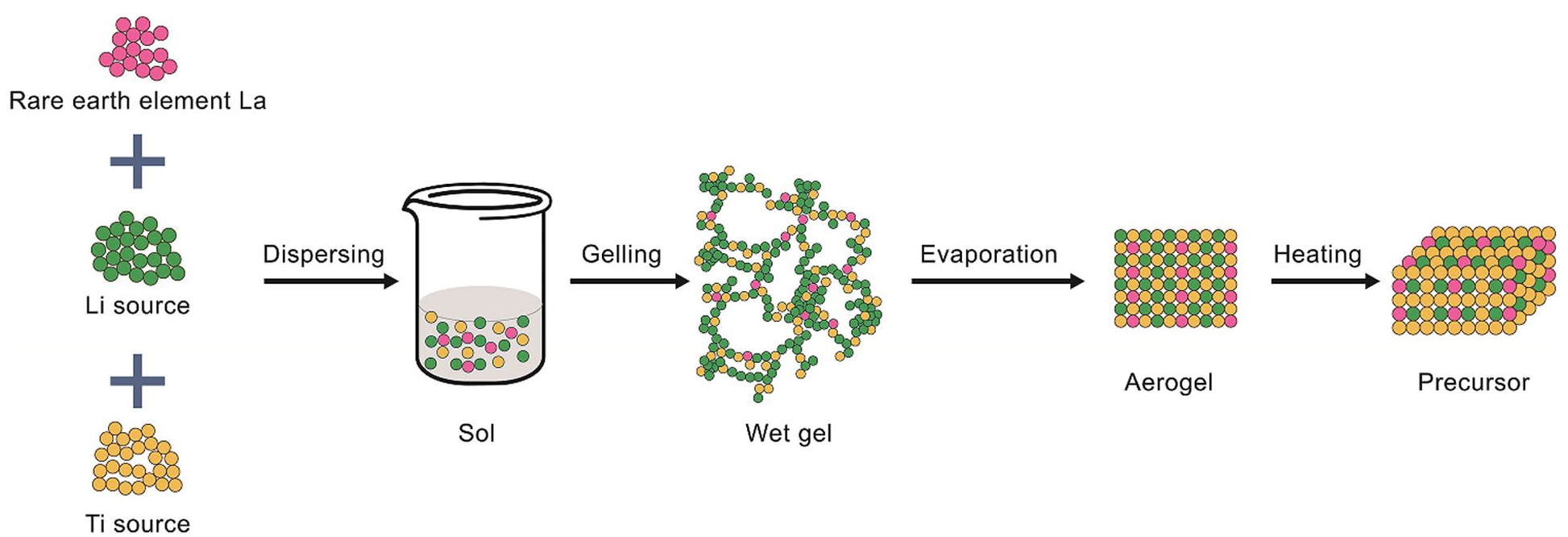
Several methods are utilized for recovering lithium from salt-lake brines, including precipitation, 22 solvent extraction, 23 membrane filtration, 24 electrochemical extraction,25,26 and ion-exchange adsorption. 27 Precipitation-based methods are typically used to recover lithium from brine having high lithium-ion concentrations, as well as a low magnesium-to-lithium ratio. In contrast, solvent extraction is used to extract lithium from brine having a high magnesium-to-lithium ratio. Furthermore, membrane-based and electrochemical methods can selectively separate divalent Mg2+ and monovalent Li+; however, they usually suffer from low recoveries and require the input of a large amount of energy. 28 Therefore, as a result of their high lithium-ion selectivity and simplicity, adsorption-based processes have been recognized as an ideal choice for the recovery of lithium from low-grade sources. Consequently, in this study, we prepared lanthanum-doped lithium-ion sieves using the sol–gel method to yield an adsorbent having high crystallinity, few impurities, and highly selective lithium-ion adsorption. A schematic of the synthesis of the ion-sieve precursors is shown in Figure 1.
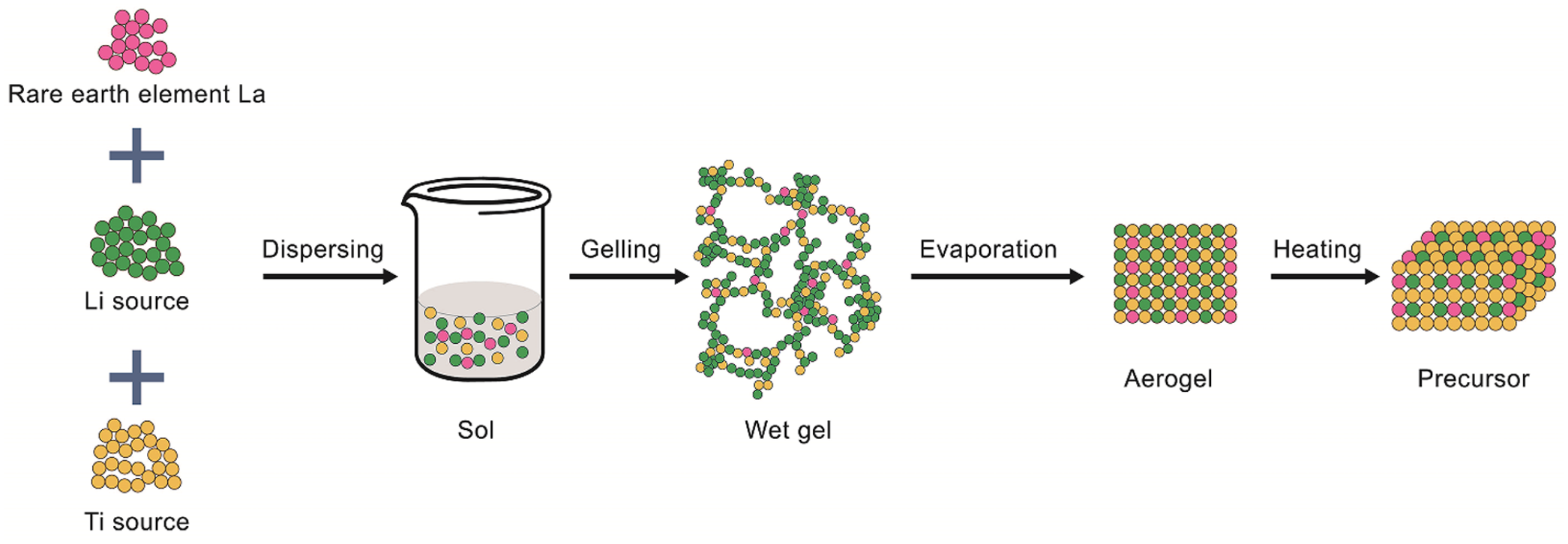
Preparation of the doped ion-sieve precursor by the sol–gel method.
Lithium-ion sieves are a safe, reliable, and efficient lithium extraction technology. Particularly, as a result of their efficient ion-sieving ability and high lithium-adsorption capacity, ion sieves can separate lithium from complex aqueous systems;29,30 the lithium extraction process using a lithium–titanium ion sieve is shown in Figure 2. 31 The selectivity for lithium ions is achieved via size and sieving effects, and, in such sieves, the partition coefficient for lithium ions is much larger than that of other metal ions. Specifically, the separation coefficients of tested metal ions are greater than 1, indicating that the adsorption capacity for lithium ions in a mixture is higher than for other metal ions. 32 The crystal structure of Li4Ti5O12 is shown in Figure 3; it has a unique spinel structure that provides a large lithium-adsorption capacity and good sorption cycling performance. In addition, Li4Ti5O12 has a stable structure, strong Ti–O bonds, and superior acid resistance; however, it generally contains rutile TiO2 and Li2TiO3 phases, which can reduce the structural stability and adsorption capacity. 33

Schematic of lithium extraction using an ion sieve (denoted LIS).

Crystal structure of Li4Ti5O12 showing the 16d and 8a sites.
However, doping Li4Ti5O12 with a small amount of rare earth elements can enhance its structural stability and adsorption capacity, thus making it suitable for use as an ion sieve.34,35 In this study, lanthanum ions were selected as the dopant ions, and the lithium-ion adsorption capacity enhancement was assessed. Consequently, we determined that the improvement in the performance of the ion sieve has several causes. The doping of La3+ cations into the Li4Ti5O12 lattice stabilizes the crystal structure; thus, structural changes during Li+ adsorption are less likely to occur. In addition, particle agglomeration is reduced, resulting in a lower ion-migration resistance. Furthermore, La3+ has a larger ionic radius than Ti4+ (
Results and discussion
As shown in Figures 1 and 2, the precursor ion sieves were prepared via the sol–gel method, followed by roasting. Subsequently, the ion sieves were obtained by acid washing to exchange Li+ for H+. To identify the optimal synthetic conditions, X-ray diffraction (XRD) and scanning electron microscopy (SEM) measurements of the precursors treated at different temperatures and in different acid concentrations were carried out. In addition, the loss of lithium and titanium from the ion sieves during acid washing was assessed. Thus, we obtained the optimal synthetic conditions, characterized the lithium-ion adsorption capacity and selectivity, and determined the mode of adsorption using kinetics analysis and isotherm fitting.
XRD and SEM characterization
XRD patterns of the undoped and doped powders roasted at different temperatures were obtained for phase analysis and are shown in Figure 4. To determine the effect of the dopant content (

(a–c) XRD patterns of lanthanum-doped lithium–titanium ion-sieve precursors roasted at 700, 800, and 900 °C. (d) Magnification of the (111) reflection of the precursors roasted at 800 °C.
Next, we assessed the effect of the roasting temperature. In the XRD pattern of the sample roasted at 700 °C (Figure 4(a)), the baseline is noisy, and impurity peaks are observed. However, the XRD pattern remains consistent with the spinel structure. In the XRD pattern of the sample roasted at 800 °C (Figure 4(b)), the baseline is smoother, with fewer impurity peaks and sharper diffraction peaks. In the XRD pattern of the sample roasted at 900 °C (Figure 4(c)), the number of impurity peaks is increased, suggesting that this temperature resulted in the degradation of the Li4Ti5O12 phase. Therefore, the optimum roasting temperature was selected as 800 °C.
We further analyzed the effect of doping on the crystal structure of the sample treated at 800 °C by examining the (111) reflection, as shown in Figure 4(d). As shown, with the increase in La3+ content, the (111) reflection shifts to a lower angle, indicating an increase in the corresponding crystal plane spacing. This is a result of the inclusion of the larger La3+ ions, which cause unit cell expansion. For the application as an ion sieve, La3+ doping enlarges the channels available for lithium-ion adsorption, which lowers the resistance to the migration of lithium ions and, thus, improves the ion-sieve performance. Considering the impurity phases, crystallinity, and crystal structure, the sample having
SEM images of the Li4Ti4.99La0.01O12 sample after roasting at 700, 800, and 900 °C are shown in Figure 5(a–c), respectively. After treatment at 700 °C, the particles are clustered owing to the physical mixing of Li2TiO3 and TiO2, which is consistent with the XRD results. After treatment at 800 °C, a spinel-type oxide Li4Ti5O12 can be observed, and the particles are uniform in size with perfect grain sizes. After treatment at 900 °C, the product is still spinel Li4Ti5O12. Figure 5(d) shows an SEM image of the undoped sample after roasting at 800 °C, and Figure 5(e) shows an SEM image of the H4Ti5O12 phase obtained after acid washing the undoped precursor treated at 800 °C. As shown by comparison with Figure 5(b), the particle morphology was maintained after acid washing. Figure 5(f) shows an SEM image of the acid-washed sample after the adsorption of lithium ions; the lithium ions appear to have adhered to the surface of the lanthanide-doped ion sieve after adsorption.

SEM images of (a–c) Li4Ti4.99La0.01O12 roasted at 700, 800, and 900 °C, respectively. (d) SEM images of undoped Li4Ti5O12 roasted at 800 °C (e) after acid washing and (f) after lithium-ion adsorption.
Next, we investigated the effect of the concentration of the acid used to prepare the ion sieves on the degree of lithium and titanium leaching. To ensure a high performance and an ability to recover the adsorbed lithium, the degree of lithium leaching should be maximized. In contrast, to ensure stability, the degree of titanium leaching should be minimized. Figure 6 shows the degrees of Li+ and Ti4+ leaching with respect to the HCl concentration. The highest degree of Li+ leaching was obtained using 0.1 mol L−1 HCl; at the same concentration, the degree of Ti4+ leaching did not exceed 1%. Between 0 and 0.1 mol L−1 HCl, the Li+ leaching increased with the increase in acid concentration. However, over 0.1 mol L−1 HCl, the Li+ leaching plateaued, and the Ti4+ loss increased markedly, revealing that high acid concentrations damage the crystal structure. Therefore, a hydrochloric acid concentration of 0.1 mol L−1 was selected as the optimal concentration for acid washing.
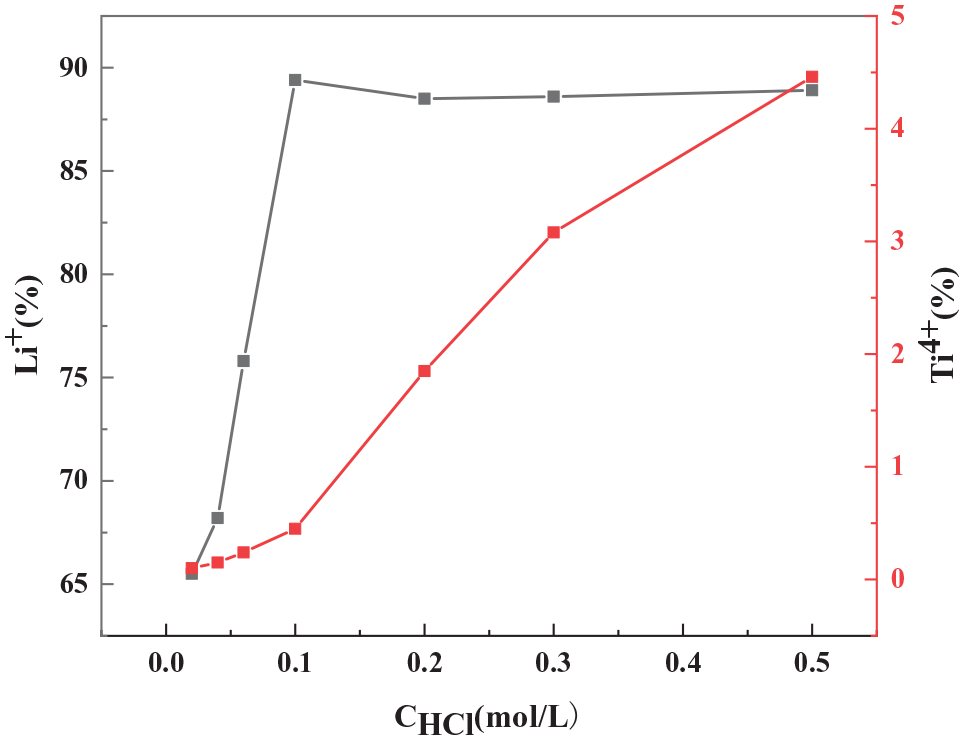
Degree of Li+ and Ti4+ leaching with respect to acid concentration.
Contact angle tests
The wettability of the ion sieve has a considerable effect on its adsorption performance because Li+ is generally present in aqueous solutions as hydrated ions. In particular, the use of hydrophilic materials can enhance ion desolvation, thereby facilitating the interaction of Li+ with the adsorption sites. Therefore, increasing the hydrophilicity of the ion sieve can enhance metal-ion desolvation, facilitate ion entry and exchange with H+, and improve the adsorption performance. 37 Therefore, we carried out contact angle analysis using deionized water droplets at different positions on the surface of the ion sieves, as shown in Figure 7. The contact angles of the lanthanide-doped lithium–titanium ion sieves were 41.65° and 41.55°, indicating that the samples have good hydrophilicity; this is consistent with the presence of multiple hydrophilic functional groups on the material surface, which, as explained above, should enhance the desolvation of Li+ and accelerate its adsorption.

Contact angle analysis of deionized water droplets on the ion sieves.
Effect of temperature and pH on adsorption capacity
Figure 8 shows the Li+ adsorption capacities of H4Ti5O12 and H4Ti4.99La0.01O12 at 5, 15, 25, 35, and 45 °C. Between 5 and 15 °C, 15 and 25 °C, and above 25 °C, the Li+ adsorption capacity, respectively, increased rapidly, slowed, and plateaued. Spinel-type ion sieves contain many pores. In particular, the vacant site at the tetrahedral 8a position allows only small ions, such as Li+ and H+ to enter and exit. Li+ adsorption or insertion is a diffusion process that requires energy input. Consequently, increasing the temperature increases diffusion into the pores and, thereby, adsorption. 38 In addition, the Li+–H+ ion-exchange process requires the breakage of the O–H bonds, which also requires energy input, thus explaining the increase in Li+ adsorption with the increase in temperature. 39 Based on the obtained results, 25 °C was selected as the optimal reaction temperature.

Effect of temperature on the Li+ adsorption capacity.
The pH of the solution also has a significant influence on the Li+ adsorption capacity. Therefore, the effect of pH on Li+ adsorption by the H4Ti5O12 and H4Ti4.99La0.01O12 samples was assessed, as shown in Figure 9. Between pH = 2 and 7, the adsorption capacity increased relatively slowly; however, it increased rapidly after pH = 7, reaching a maximum between pH = 10 and 12. This indicates that alkaline solutions are more favorable for Li+ adsorption, as reported previously. 40 Therefore, pH = 12 was chosen for further experiments.

Effect of pH on Li+ adsorption capacity.
Adsorption kinetics
Next, we assessed the mode of adsorption of lithium ions by the ion sieves by analyzing their adsorption kinetics. Figure 10 shows the kinetics of Li+ adsorption for the H4Ti5O12 and H4Ti4.99La0.01O12 samples. Between 0 and 25 h, the adsorption capacity increased rapidly, subsequently plateauing because the sites for Li+ adsorption had been occupied. Therefore, the adsorption capacity of the ion sieve reached equilibrium after approximately 24 h. At this time, the saturated adsorption capacity of the H4Ti5O12 ion sieve was 21.98 mg g−1, whereas that of the H4Ti4.99La0.01O12 ion sieve was 23.96 mg g−1.
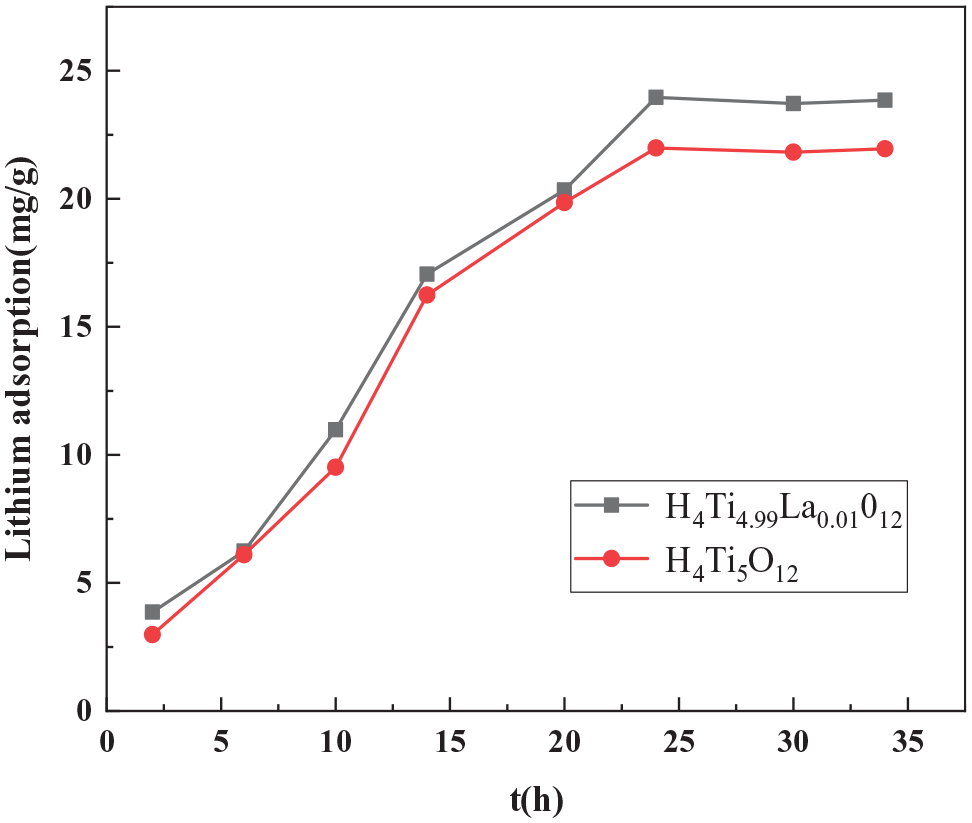
Effect of adsorption time on the adsorption exchange capacity of the doped and undoped samples.
Figure 11 and Table 1 show the results of fitting the Li+ adsorption kinetics data for the H4Ti4.99La0.01O12 sample to the pseudo-first-order (PFO) and pseudo-second-order (PSO) kinetic models (see section “Experimental” for further details of the fitting, as well as definitions of the parameters). The coefficient of determination obtained for fitting to the PSO model is higher than that obtained for fitting to the PFO model (
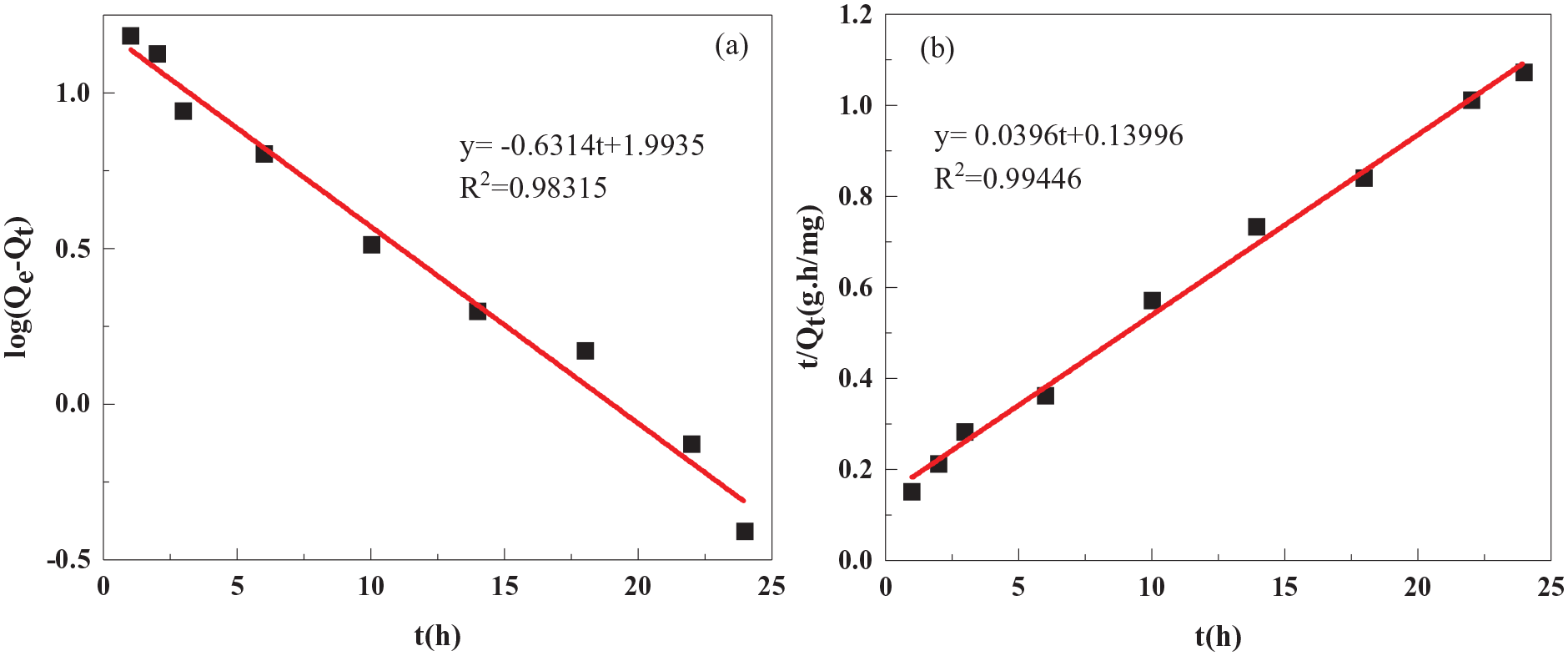
Linear fits to (a) PFO and (b) PSO models.
Fitting to adsorption kinetics.

PFO: pseudo-first-order; PSO: pseudo-second-order.
Adsorption isotherms
Figure 12 shows the effect of the initial Li+ concentration on the adsorption capacity of the doped and undoped samples. For [Li+] < 100 mg L−1, the adsorption capacity of the ion sieve increased with the increase in the initial Li+ concentration. However, the adsorption capacity began to plateau for [Li+] > 100 mg L−1. These results suggest that the optimal initial Li+ concentration is 100 mg L−1.
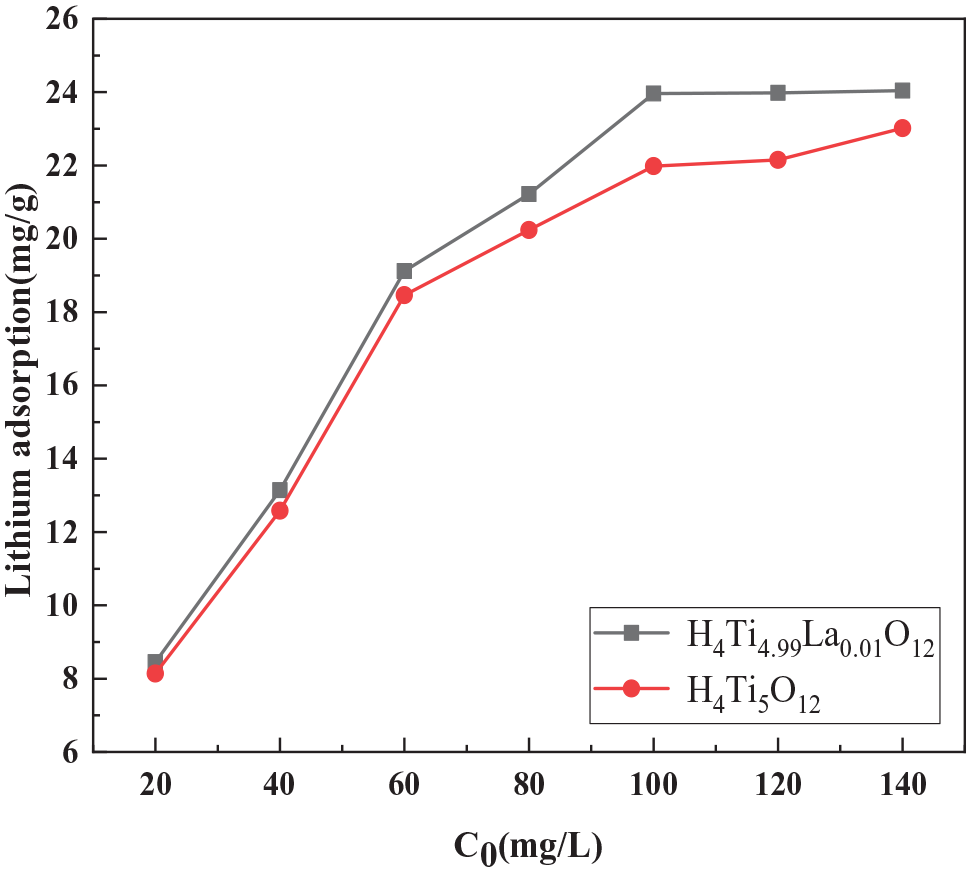
Effect of initial Li+ concentration on the adsorption capacity of doped and undoped ion sieves.
Next, these data were fitted to the Langmuir and Freundlich isotherms, and the results are shown in Figure 13 and listed in Table 2 (see section “Experimental” for further details of the fitting, as well as definitions of the parameters). As shown,
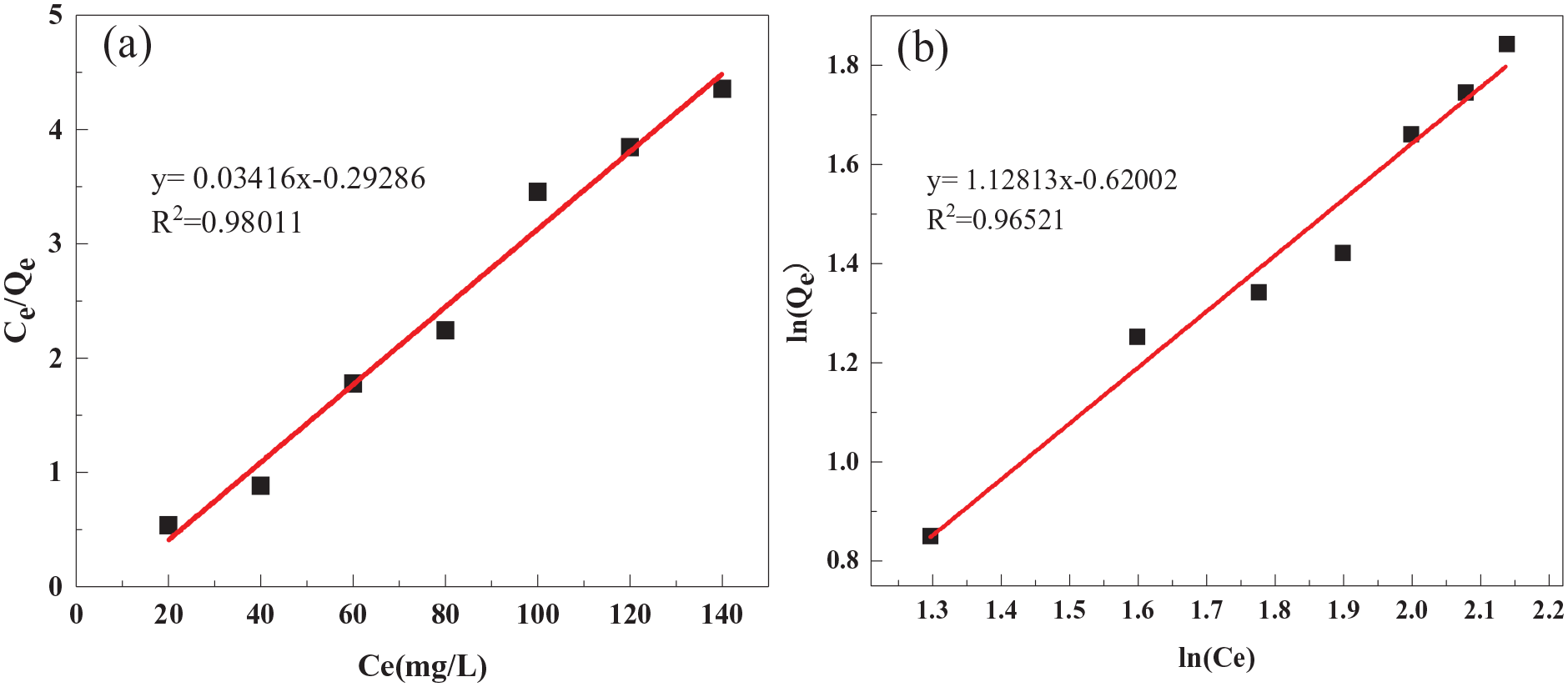
Fitting of the adsorption data to the (a) Langmuir and (b) Freundlich isotherms.
Results of linear fits to the Langmuir and Freundlich isotherms.

Lithium-ion selectivity and adsorption–desorption cycling
The selectivity of the ion sieves for Li+ in a solution containing equal concentrations of Li+, Na+, K+, Mg2+, and Ca2+ was next assessed, and the results are shown in Figure 14. As shown, the adsorption capacities were 23.96, 4.15, 3.31, 2.05, and 1.28 mg g−1 for Li+, Na+, K+, Mg2+, and Ca2+, respectively, demonstrating the high selectivity of the prepared ion sieve for Li+.
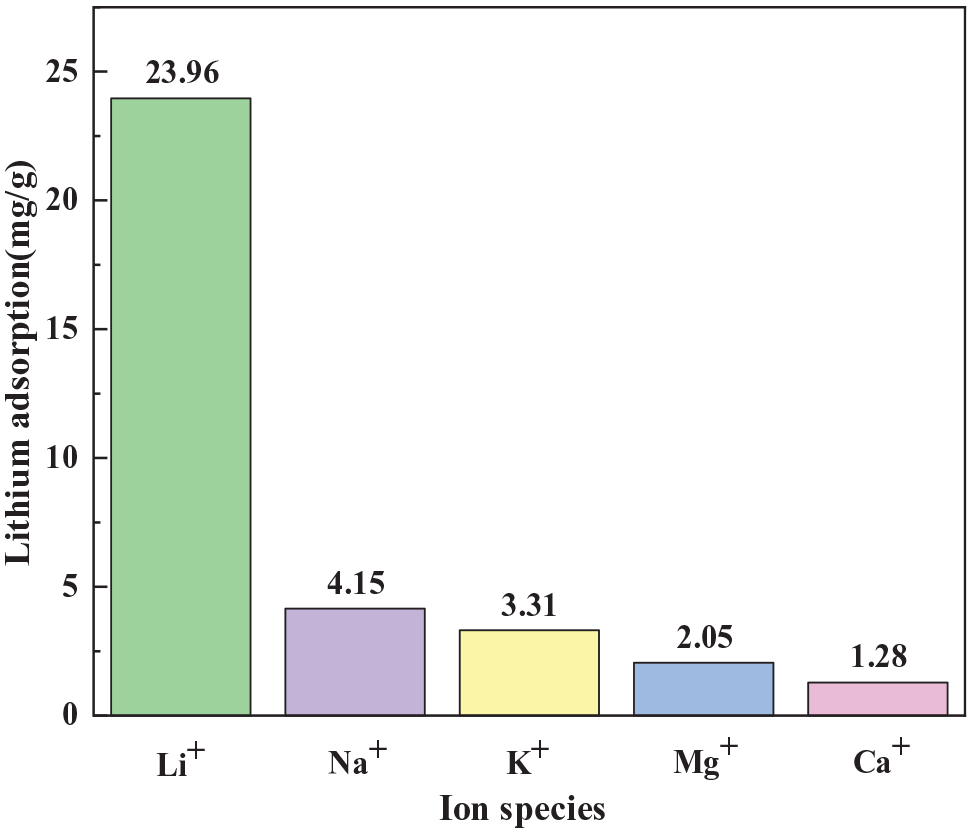
Selectivity of the doped ion sieve for Li+.
The ability to reuse the ion sieve is crucial for practical applications. Therefore, the Li+ adsorption capacity of the doped ion sieve over eight adsorption–desorption cycles was assessed. As shown in Figure 15, a decrease of only 4.5%, from 23.96 to 22.88 mg g−1, was observed from the first to eighth cycle, suggesting that the structure of the doped ion sieve is stable and demonstrating the potential of the sieve for real-world applications.

Cyclic stability of the doped ion sieve.
Conclusion
In this study, La3+-doped Li4Ti5–
La3+ can be doped into H4Ti5O12 without changing the crystal structure. The Li+ adsorption performance was found to improve after La3+ doping.
The doped sample with
La3+ doping improved the stability of the H4Ti5O12 crystal structure, prevented particle agglomeration, reduced the lithium-ion migration resistance, and improved the adsorption performance. Furthermore, the adsorption capacity was maintained after eight adsorption–desorption cycles, indicating that the La3+-doped ion sieve has good cycling stability.
Experimental
Materials and instruments
Materials
Lithium acetate, butyl titanate, lanthanum nitrate, anhydrous ethanol, glacial acetic acid, hydrochloric acid, lithium hydroxide, lithium chloride, sodium chloride, potassium chloride, magnesium chloride, and calcium chloride were obtained from (Tianjin Comio Chemical Reagent Co., China). All the reagents were analytically pure. Deionized water was prepared using a (Ultra Pure Water Machine YTUP-15S, Shanghai YETOP Technology Co., China).
Instruments
The instruments used were collector magnetic stirrer (DF-101S, Shanghai Yixin Scientific Instruments Co., China); electric thermostatic blast dryer (101-1B, Shaoxing Luda Machinery Equipment Manufacturing Co., Zhejiang Province, China); muffle furnaces (SX2-4-10-A, Shanghai Heng Science Instruments Co., China); pH meter (pHS-3C, Shanghai Yidian Scientific Instruments Co., China); centrifuge (LSC-20, Yixing Huading Machinery Co., Jiangsu Province, China); thermogravimetric analyzer (FBS-TGA101, Nanjing Huicheng Instrument Co., Jiangsu Province, China); X-ray diffractometer (DX-2007X, Dandong Haoyuan Instrument Co., Liaoning Province, China; tube voltage = 40 kV, current = 100 mA, Cu-
Synthesis of doped ion-sieve precursors (Li4Ti5–xLaxO12)
First, butyl titanate (8.0002 g) was dropwise added to anhydrous ethanol (20 mL), yielding solution A. Subsequently, deionized water (20 mL) was mixed with glacial acetic acid (10 mL), and lithium acetate (2.2012 g) and lanthanum nitrate (0, 0.1, 0.2, or 0.3 g) were added, yielding solution B. Solution A was magnetically stirred for 20 min in a constant-temperature water bath at 25 °C; it was then added slowly and dropwise to solution B. The temperature was then increased to 65 °C with continuous stirring until a milky white solution was formed. The obtained solutions were aged at room temperature for 24 h, and then dried in an oven at 110 °C to yield dry white gels. The Li4Ti5–
Synthesis of doped ion sieves (H4Ti5–xLaxO12)
The Li4Ti5–
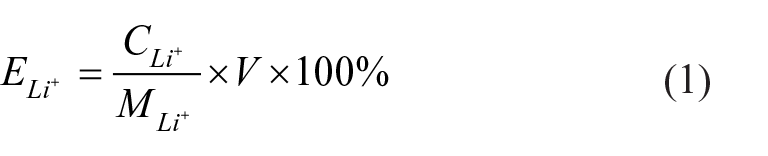
here,
Contact angle tests
For the preparation of the samples, an aqueous solution of HCl (0.1 mol L−1) was used, and the ion-sieve precursor having
Effect of temperature and solution pH on adsorption capacity
To determine the effect of temperature on the adsorption capacity, LiCl and LiOH were used to prepare aqueous Li+ solutions (100 mL, 100 mg L−1, pH = 12). Five doped ion sieve samples (0.1 g each) were added to the prepared Li+ solutions and stirred in a water bath at 5, 15, 25, 35, and 45 °C for 24 h. After standing, aliquots of the supernatant were removed, and the Li+ concentration was measured to calculate the adsorption capacity.
To determine the effect of pH on the adsorption capacity, five aqueous Li+ solutions (100 mg L−1, 100 mL), having pH values of 4, 6, 8, 10, and 12, were prepared. Five samples of the doped ion sieves (0.1 g each) were added to the prepared Li+ solutions and stirred in a water bath at 25 °C for 24 h. After standing, aliquots of the supernatant were removed, and its Li+ concentration was measured to calculate the adsorption exchange.
Adsorption kinetics
First, samples of the doped ion sieve (0.1 g) were added to aqueous Li+ solutions (100 mL, 100 mg L−1, pH = 12) and stirred in a water bath at 25 °C. Aliquots of the supernatant were then removed at different intervals (1, 2, 3, 6, 10, 14, 18, 22, and 24 h); after standing, the Li+ concentration was measured, and the saturated adsorption capacity was determined. The measured data were fit to the PFO and PSO equations to identify the mode of adsorption

here,
Adsorption isotherm analysis
First, samples of the doped ion sieve (0.1 g) were placed in aqueous Li+ solutions (100 mL, pH = 12) having different lithium salt concentrations (20, 40, 60, 80, 100, 120, and 140 mg L−1) and stirred in a water bath at 25 °C for 24 h. After standing, the supernatant was extracted at intervals, and the Li+ concentration in the supernatant was determined via ion chromatography. The data were then fitted to the Langmuir and Freundlich adsorption isotherms, as given by equations (3) and (4), respectively


here,
Selectivity and cyclic stability
Selectivity
Five samples of the doped ion sieve (0.1 g) were placed in an aqueous solution containing equal concentrations of Li+, Na+, K+, Mg+, and Ca2+ (100 mL, 100 mg L−1, pH = 12) and stirred in a constant-temperature water bath at 25 °C for 24 h. After standing, the supernatant was extracted, and the concentration of each metal ion was determined. Subsequently, the adsorption capacity for each ion was calculated to determine the selectivity of the doped ion sieve.
Cyclic stability
The doped ion sieve (0.1 g) was placed in an aqueous Li+ solution (100 mL, 100 mg L−1, pH = 12) and stirred in a water bath at 25 °C for 24 h. After standing, the supernatant was extracted, Li+ concentration was measured, and its saturated adsorption capacity was calculated. The doped ion sieve was then filtered, dried, and acid washed with 0.1 mol L−1 hydrochloric acid to desorb the lithium ions. Subsequently, the doped ion sieve was washed with deionized water to neutral pH, vacuum filtered, and dried. Thereafter, the adsorption–desorption process was repeated eight times, and the saturated adsorption capacity was determined for each cycle.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
