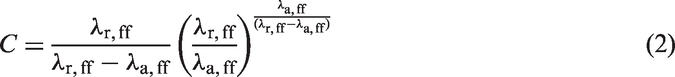Abstract
This work is framed within the Eighth Industrial Fluid Properties Simulation Challenge, with the aim of assessing the capability of molecular simulation methods and force fields to accurately predict adsorption in porous media for systems of relevant practical interest. The current challenge focuses on predicting adsorption isotherms of n-perfluorohexane in the certified reference material BAM-P109 standard activated carbon. A temperature of
Keywords
Introduction
The aim of the Eighth Industrial Fluid Properties Simulation Challenge (IFPSC) is to assess the capability of force fields and molecular simulation methods to accurately predict adsorption in porous media for systems of relevant practical interest. Specifically, the adsorption of n-perfluorohexane in the certified reference material BAM-P109 standard activated carbon is the purpose of the current IFPSC.
Perfluorinated molecules are known to be challenging to describe. Their structural and thermophysical properties are different to those of their analogous hydrocarbon compounds, in part due to the size, polarisation, and highly electronegative nature of the fluorinated group, which gives rise to the particularly steep nature of the repulsive interactions between perfluorinated groups (Maitland et al., 1981; Yee et al., 1992). Force fields based on a generalised Lennard-Jones (Jones, 1924a, 1924b; Lennard-Jones, 1931) or Mie potential (Grüneisen, 1912; Mie, 1903), where the repulsive and attractive exponent can be varied, allow a fine tuning of the steepness of the potential well and the range of the interaction, which have proven to be especially useful to describe the fluid phase behaviour (Potoff and Bernard-Brunel, 2009; Rubio et al., 1985) and second derivative thermodynamic properties of perfluoroalkanes (Lafitte et al., 2013).
As a further complication, the use of force fields developed for bulk fluids to describe fluid–solid interactions is not a straightforward task. In most cases empirical combining rules (e.g. the traditional Lorentz–Berthelot) are used to obtain the unlike-interaction parameters. The validity of such combining rules is however restricted to systems with similar type of molecular interactions (Desgranges and Delhommelle, 2014; Haslam et al., 2008). Although not the focus of this work, it is imperative to find robust and reliable approaches to obtain the fluid–fluid and fluid–solid interactions particularly in the case of systems with limited experimental data. It is also important to recognise that coarse-grained (CG) solid–fluid interactions will depend in a complex way on the morphology of the surface (Forte et al., 2014).
In our current work, we use CG intermolecular potential models to represent the fluid molecules and a collective of atomistically detailed graphite slit pores (kernel) to represent the microporosity of the reference solid. The CG force fields employed in our molecular simulations are developed using a top-down approach (Müller and Jackson, 2014), as this provides robustness in the predictions of the key properties for adsorption such as condensed liquid densities and vapour pressures. An essential element for top-down development of force fields is an accurate equation of state (EoS) characterised by a well-defined Hamiltonian enabling one to establish a formal link between target macroscopic thermodynamic properties and the CG force field. The latest version of the statistical associating fluid theory for potentials of variable range based on the Mie interaction, SAFT-VR Mie (Lafitte et al., 2013), and its group-contribution variant SAFT-γ Mie (Papaioannou et al., 2014) has been shown to be a reliable and robust method to develop CG force fields for direct use in molecular simulation (Avendaño et al., 2011, 2013; Herdes et al., 2015; Lafitte et al., 2012; Lobanova et al., 2015; Müller and Jackson, 2014; Theodorakis et al., 2015). The same approach is also used here to develop SAFT-γ Mie CG force-field parameters that are then assessed by molecular dynamic (MD) simulations and used within Grand Canonical Monte Carlo (GCMC) simulations to predict the adsorption of the system of interest.
Modelling approaches and force fields
SAFT-VR Mie EoS
Here we use an extension of the SAFT EoS (Chapman et al., 1989, 1990) referred to as SAFT-VR Mie (Lafitte et al., 2013). The generic SAFT approach stems from the thermodynamic perturbation theory of Wertheim (1984a, 1984b, 1986a, 1986b, 1986c, 1987) to explicitly take into account the non-sphericity and directional interactions of molecules. The SAFT-VR Mie (Lafitte et al., 2013) methodology is a reformulation of the SAFT-VR EoS (Galindo et al., 1998; Gil-Villegas et al., 1997) for Mie potentials with enhanced accuracy in the description of the fluid phase behaviour (particularly in the near-critical region) and second derivative properties of fluids. Full details of the development of the theory are given in Lafitte et al. (2013).
Within the SAFT-VR Mie formalism molecules are described as chains of m tangent segments that interact through a Mie potential (Grüneisen, 1912; Mie, 1903)
The equation of state for a non-associating Mie fluid is written in a closed form in terms of the Helmholtz free energy A as a sum of the following contributions
Molecular models
The SAFT-VR Mie (Lafitte et al., 2013) (and SAFT-γ Mie (Papaioannou et al., 2014)) EoS is based on the assumption that the basic building blocks of a molecule are spherical segments. This is particularly useful to describe molecules in a CG manner: we dispense with the atomistic detail and represent molecules in terms of a tangent chain of beads. In our current study, the carbon dioxide molecule is modelled as a tangent dimer, n-perfluorohexane as a rigid trimer, and argon as a sphere. Note that potentials developed with this methodology are referred to as SAFT-γ Mie force fields in recognition of the more generic group-contribution nature of the approach (Müller and Jackson, 2014).
There is a corresponding-states relationship between the exponents in a Mie potential in such a way that an infinite number of pairs (
The estimation of the three remaining parameters ( Coexistence of vapour–liquid densities, vapour pressures, and surface tensions for argon. The dashed red curve corresponds to the SAFT-VR Mie EoS calculations, the continuous blue curve corresponds to correlated experimental data (Lemmon et al., 2014), and the orange circles correspond to molecular dynamics simulations, using the same parameters as the theory (color online). Coexistence of vapour–liquid densities, vapour pressures, and surface tensions for carbon dioxide. Symbols as in Figure 1. Coexistence of vapour–liquid densities, vapour pressures, and surface tensions for n-perfluorohexane. Symbols as in Figure 1. The SAFT-γ Mie CG force fields for the fluids of interest (the attractive exponent is fixed at 



Molecular simulation of fluid phase equilibria
Direct MD simulations of the vapour–liquid equilibria (VLE) of carbon dioxide, argon, and n-perfluorohexane are carried out using the GROMACS open source suite (Van Der Spoel et al., 2005). In the direct technique, a film of liquid surrounded by vapour (with two stable vapour–liquid interfaces) is simulated to obtain the VLE and interfacial properties of the system.
The simulations are performed in the canonical (NVT) ensemble, where the total density and temperature are kept constant. The parallelepiped cells have aspect ratios of
The MD simulations are thermostated every 2 ps using a Nose–Hoover algorithm (Frenkel and Smit, 2002; Hoover, 1985; Nosé, 1984), and all non-bonded interactions are truncated at
Molecular simulation of the adsorbed phase
The adsorption isotherms are determined using GCMC simulations in the
The unlike-interaction parameters between segments of different type (in this case fluid and solid segments) are obtained using the Lorentz–Berthelot combining rules, i.e.


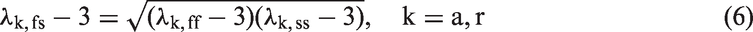
The GCMC simulations are carried out using a standard procedure starting with an empty pore which is filled until equilibrium is attained (Allen and Tildesley, 1989; Frenkel and Smit, 2002; Nicholson and Parsonage, 1982). For each Monte Carlo cycle, both the displacement/reorientation of a randomly chosen fluid molecule and a random creation/destruction of a fluid molecule are attempted. The systems are equilibrated at each pressure level and then averages are determined from a further two million cycles. Block averages are also obtained every 10,000 cycles and are used to compute standard deviations to calculate uncertainties.
In the GCMC simulations the temperature and the activity (Müller, 2010) (which is directly related to the chemical potential) are specified. Simulations are also performed for bulk fluids that would hypothetically be in equilibrium with the adsorbed fluid (i.e. these are performed at the same temperature and activity) so that the density of the bulk fluid can be calculated as a simple average. This density is then used to determine the pressure at a given temperature using the SAFT-VR Mie EoS. Since the EoS reproduces faithfully the volumetric properties of the fluids (see Figures 1 to 3), this procedure is in essence equivalent to generating these pressures from pure component simulations, albeit more efficiently.
Development of a kernel of adsorption isotherms
In order to characterise the experimental data provided in the challenge documentation, we built a GCMC kernel of isotherms which are used to represent the known experimental adsorption isotherms of argon and carbon dioxide in BAM-P109. This procedure guarantees that we obtain a pore size distribution (PSD) compatible with the molecular modelling strategy avoiding any uncertainties in the underlying theoretical treatment as would be the case if the PSD provided in the challenge were used directly.
The pore widths that constitute our kernel are chosen to be consistent (or commensurate) with the expected PSD of BAM-P109 as suggested in the challenge documentation. We employ 11 values: H = 0.320, 0.488, 0.656, 0.824, 0.992, 1.160, 1.328, 1.496, 1.664, 1.832, and 2.0 nm. The distances between the solid surfaces, measured from the centre of the carbon atoms, correspond to
Unique PSD curve and estimation of the pore volume for BAM-P109
An important step towards the prediction of the adsorption isotherm is the determination of a reliable PSD of the BAM-P109 activated carbon. The porosity analysis, provided as part of the challenge documentation, comprises three differential pore volume distributions, two from argon (using quenched solid density functional theory, QSDFT, and non-local density functional theory, NLDFT, at 87 K) and another from carbon dioxide (using NLDFT at 273 K), which we reproduce in Figure 4. Several problems are evident from an inspection of Figure 4. One of the most obvious is the non-uniqueness of the PSD, as the analysis for each fluid (argon and carbon dioxide) and each temperature differs considerably. Another evident problem is the suggestion of unrealistically small pore sizes by means of the more refined theory (QSDFT).
Differential pore volume distribution of BAM-P109. The blue curve (triangles) corresponds to the quenched solid density functional theory analysis performed with argon at T = 87 K, the green curve (squares) to the non-local density functional theory (NLDFT) analysis performed with argon at T = 87 K, the red curve (diamonds) to the NLDFT analysis performed with carbon dioxide at T = 273 K. Information taken from the benchmark data of the Challenge. The black curve (filled circles) is the PSD determined for use in our current work (color online).
Given an experimental adsorption isotherm 
We aim to determine an averaged unique single-fluid independent PSD f(H) by minimising the quadratic error between the experimental adsorption isotherms
Results and discussion
In Figures 5 to 7 we show the results obtained by simultaneously fitting the experimental data to the kernels generated for each fluid: the averaged single fluid independent PSD f(H) (with the ancillary pore volume variable, which we estimated to be a value of 0.5802 cm3/g at Calculated probability density function for the pore size distribution of BAM-P109 (color online). Result of the fitting of the experimental data (continuous curve) and GCMC kernel (circles) for argon at T = 87 K. Result of the fitting of the experimental data (continuous curve) and GCMC kernel (circles) for carbon dioxide at T = 273 K. Predicted adsorption of n-perfluorohexane Γ(p) in BAM-P109 at 273 K and standard deviation, SD, for various pressures 

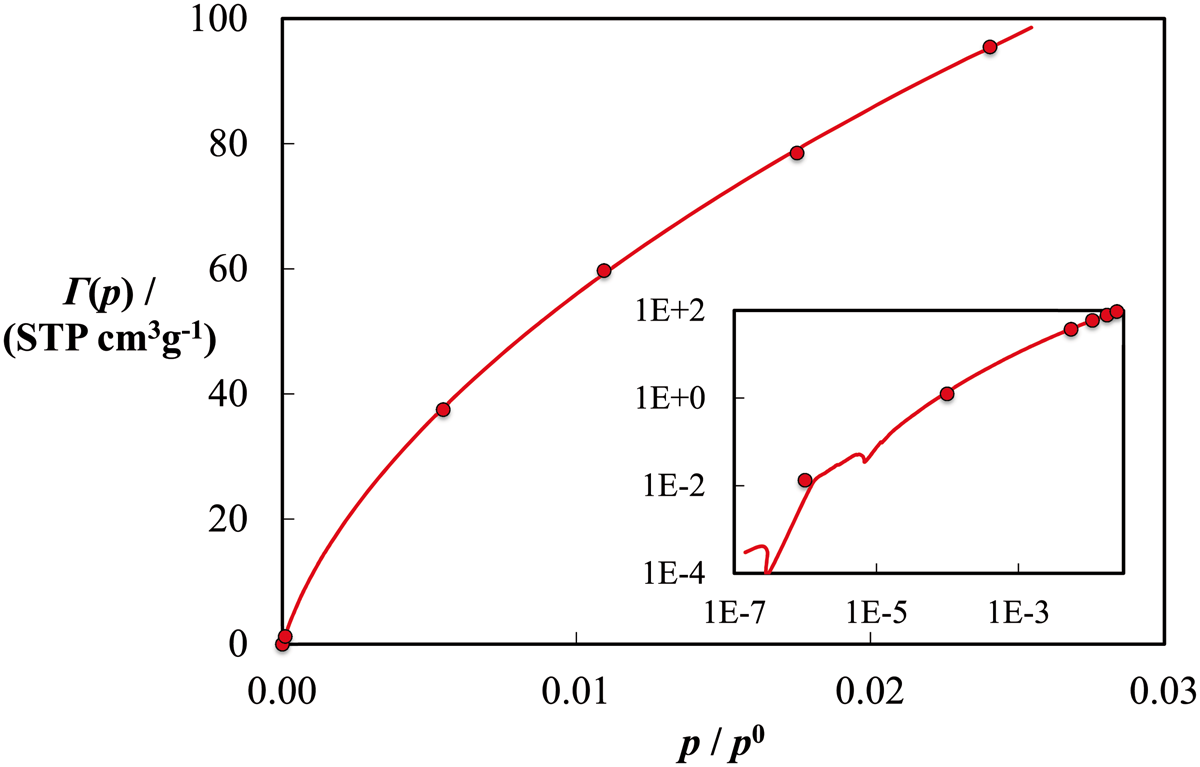

Conclusions
We have demonstrated a synergetic use of molecular simulation methodologies, accurate force fields, and experimental data for the prediction of adsorption isotherms of challenging fluid–solid systems. It is expected that the determination of a unique PSD based on simultaneous consideration of the isotherms of two distinct fluids will provide a sound qualitative prediction of the unknown adsorption of a fluid, as the main goal specified in the challenge. Had there been data for the adsorption isotherm of n-perfluorohexane, these could have been considered within our methodology to provide further confidence in the calculated values. The interaction between carbon and perfluorinated moieties is non-trivial and this is most likely to be the largest source of uncertainty in the predicted results.
After the disclosure of the Benchmark results (Ross et al., 2016), we were pleased to discover that our predictions had the correct behaviour. Our prediction and the experimental data (made available at a special session sponsored by CoMSEF at the AIChE annual meeting) are compared in Figure 8. As expected, we over-predicted the adsorption (by approximately 20%) as our model surface was a perfect graphite slit pore. In hindsight, the consideration of a structure with a tuneable degree of disorder such as random carbon platelets could (Kumar et al., 2011, 2012) have improved the accuracy of the predictions as was demonstrated by the entry of Sarkisov (2016) to this challenge.
Comparison of the total adsorption predicted (diamonds) for n-perfluorohexane at T = 273 K and the experimental data (circles) released after the disclosure of the benchmark results. The curves are just guides to the eye.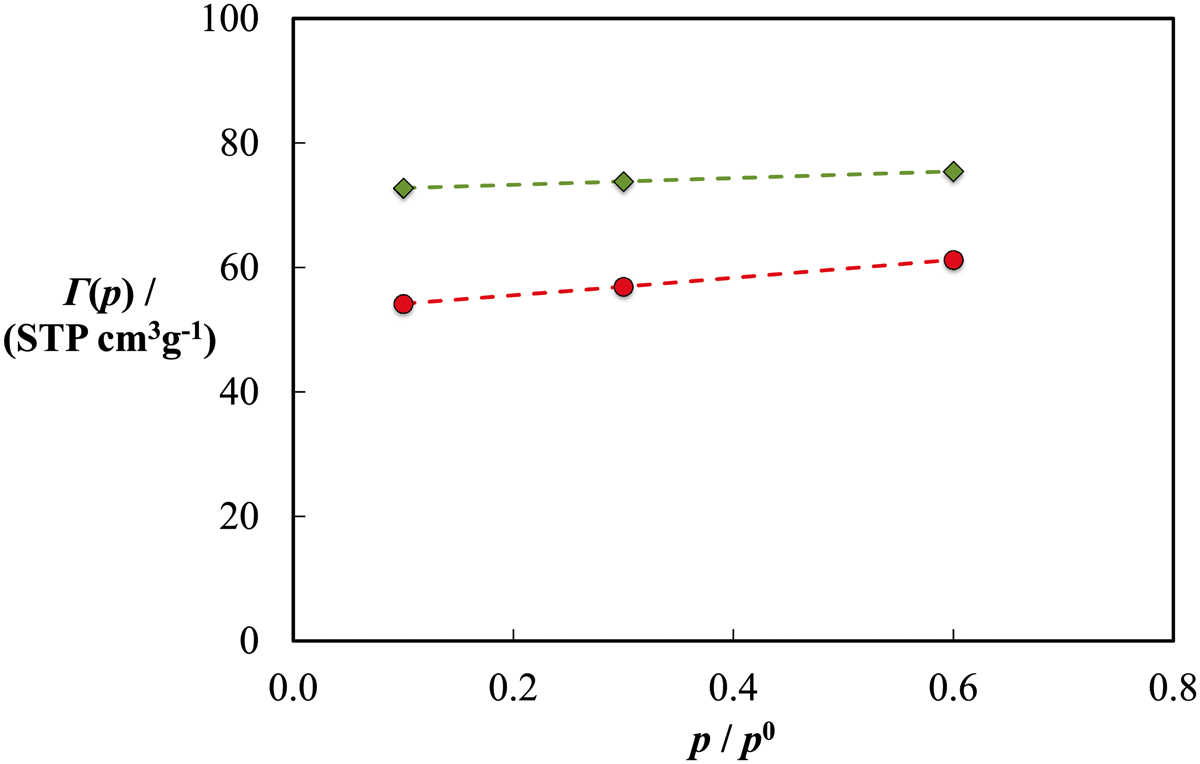
Footnotes
Acknowledgements
We are grateful to Olga Lobanova for her advice in the development of CG fluid models. Simulations described herein were performed using the facilities of the Imperial College High Performance Computing Service.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding of the Molecular Systems Engineering group by the Engineering and Physical Sciences Research Council (EPSRC) U.K. (Grant Nos. GR/T17595, GR/N35991, EP/E016340, and EP/J014958), the Joint Research Equipment Initiative (JREI) (GR/M94426), the Royal Society-Wolfson Foundation refurbishment scheme and support from the Thomas Young Centre through grant TYC-101 are also gratefully acknowledged.