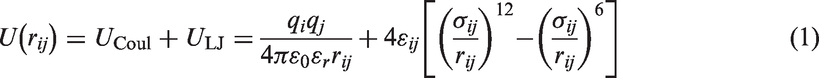Abstract
The adsorption isotherm of n-perfluorohexane on BAM-P109 activated carbon was predicted using an ab initio force field and a slit-like pore model representing the activated carbon. The force field parameters were derived from data calculated using quantum mechanics density functional theory with dispersion energy correction, and the total adsorption amount was calculated as the sum of the adsorption amounts of microscopic and mesoscopic pores. It was found that the Grand Canonical Monte Carlo method can efficiently calculate adsorption on microscopic pores but not on mesoscopic pores. Thus, we divided the mesoscopic pores into surface and free-volume regions and calculated the density and thickness of the adsorbed layers and the density of the vapor phase using canonical ensemble simulations. Using this protocol, we can calculate the adsorption isotherms in good agreement with the experimental data.
Introduction
Owing to its unique physical and chemical properties, perfluorocarbon (PFC) is widely used in semiconductor manufacturing processes and as a fire suppression agent, precision cleaning solvent, and atmospheric tracer. However, PFC is a major greenhouse gas, and the global warming potential of PFC is estimated to be 7000–9000 times greater than that of CO2 (Van Hoeymissen et al., 1996). To control PFC emissions, PFC adsorption using porous materials such as activated carbon has been studied (Ahn et al., 2006; Tom et al., 1994; Wang et al., 2014). The 8th Industrial Fluid Properties Simulation Challenge focused on predicting the adsorption isotherm of n-perfluorohexane (n-C6F14) on BAM-P109 activated carbon using molecular simulation method (Industrial Fluid Properties Simulation Collective (IFPSC), 2014).
Among the various computational methods Grand Canonical Monte Carlo (GCMC) method (Allen and Tildesley, 1987; Landau and Binder, 2000) is the most popular for predicting the equilibrium adsorption isotherms of gas molecules on porous materials. Two important factors are generally acknowledged to contribute to successful GCMC simulations: (1) an accurate force field to describe the adsorbate–adsorbent and adsorbate–adsorbate interactions; and (2) a valid simulation model (Calero et al., 2004; Fu and Sun, 2009; García-Pérez et al., 2006) to represent the experimental samples. In this work, we selected and validated several force fields to describe adsorbate–adsorbate interactions, and then developed force field parameters for adsorbate–adsorbent interactions using quantum mechanics density functional theory (QMDFT) with dispersion energy corrected functional. We used a popular slit-like model (McEnaney, 1988) for activated carbon (Jorge and Seaton, 2003; Jorge et al., 2002). Despite careful consideration of these factors, we found that adsorption is generally overestimated in comparison with the experimental data. Detailed analysis indicated that the overestimation is due to the fact that the grand canonical ensemble (NVT) representing an infinitely large reservoir of adsorbate with a constant chemical potential is not equivalent to NVT at the small length-scale (Neimark and Vishnyakov, 2006). This problem is not obvious for adsorption in microscopic pores but is rather pronounced for adsorption in mesoscopic pores. Using NVT simulation to determine the actual adsorption amount in mesoscopic pores, we obtained predictions in close agreement with the experimental data for Ar, CO2, and perfluorohexane on BAM-P109 (IFPSC, 2014).
Models and methods
The density functional theory (DFT) calculations were performed using the rVV10 van der Waals correlation functional (Sabatini et al., 2013) as implemented in Quantum ESPRESSO 5.1 (Giannozzi et al., 2009). Cutoff energies for the wave functions and charge density were set to 80 and 640 Ry, respectively. A 4 × 4 × 1 k point mesh was employed. The adsorbent was modeled by a 3 × 3 graphene sheet with a vacuum thickness of 3 nm in a 3D periodic box. Spurious electrostatic interactions between periodic images of the graphene layer were removed by using the effective screening medium method (Hamada et al., 2009). The adsorbate molecules included Ar, CO2, and C6F14. The geometries of graphene and the adsorbate molecules were optimized using the Broyden–Fletcher–Goldfarb–Shanno (BFGS) algorithm, and single-point energy calculations were conducted to scan the interactions between graphene and adsorbate molecules at various distances.
Adsorbate–adsorbate and adsorbate–adsorbent interactions were represented by pair-wise Coulomb and Lennard–Jones (LJ) 12-6 terms:
Ar was represented by a single particle model without charge. CO2 was represented by a three-particle model in which the C–O bond length was fixed at 1.16 Å and the C–O–C bond angle was fixed at 180°. The partial charges on the carbon and oxygen atoms were 0.70 and −0.35 e, respectively. C6F14 was represented by six united atoms (UA) with two UA types (i.e. CF3 and CF2), and the net charge of CF3 and CF2 sites was zero. The graphene layer was treated as a rigid body with interaction sites on carbon atoms without charge. Therefore, electrostatic interactions were employed only between CO2–CO2 pairwise.
Atom types and nonbond LJ-12-6 parameters used in this study.

The adsorbate–adsorbate parameters were validated by calculating vapor–liquid equilibrium (VLE) curves and saturated vapor pressures (SVPs) by using the Gibbs Ensemble Monte Carlo (GEMC) method (Panagiotopoulos, 1987). Two boxes representing the vapor and liquid phases were used in the simulations. The total number of molecule in the simulations were 350 for Ar, 250 for CO2, and 250 for C6F14. The GEMC moves included swapping molecules between the vapor and liquid boxes, volume exchanges, translations, and rotations of molecules in each of the two boxes. Each of the GEMC simulations ran 1 × 107 steps for equilibration and 1 × 107 steps for data collection.
The slit-like model (Figure 1), which consists of two carbon walls and a vacuum between the walls with pore width (w), was used in simulations to represent a pore in BAM-P109. The carbon wall was composed of three graphene layers with an interlayer space of 0.335 nm. The periodic boundary condition was applied so that the model represents an infinitely extended pore with equal width. The pore width was defined by the distance between two inner graphene layers in the slit pore (as shown in Figure 1), which was 3.4 Å greater than the effective pore size measured experimentally (Celzard and Fierro, 2005). Combining the simulation results performed on slit-like models and pore size distribution (PSD), which represents the specific volume of pore with pore width w, the adsorption isotherm curves of BAM-P109 were calculated by using the method described below.
Slit-like pore model used in this study.
The experimental PSD curve of BAM-P109 (Figure 2) has been determined by using the quenched surface model (QSDFT) to fit the experimental adsorption data. Different PSD curves may be obtained using different molecules. The PSD curve used in this work is based on Ar adsorption at 87 K. Based on PSD curve we divided the pores into two types: microscopic pores with widths less than 2 nm and mesoscopic pores with widths greater than 2 nm. The specific pore volumes and surface areas of the microscopic and mesoscopic pores can be calculated by integrating over PSD data. The calculated specific volumes of the microscopic (Vmicro) and mesoscopic (Vmeso) pores are 0.51 and 0.52 cm3/g, respectively, and the specific surface areas of microscopic (Smicro) and mesoscopic (Smeso) pores are 1253.12 and 74.8 m2/g, respectively. The total specific pore volume and surface area is 1.03 cm3/g and 1327.92 m2/g, respectively, which is in close agreements with the experimental values of 1.045 cm3/g and 1383 m2/g, respectively.
Experimental pore size distribution of BAM-P109.
The total adsorption number per gram BAM-P109 measured by the number of adsorbates, Ntotal (T, P), was then calculated as a sum of two parts:
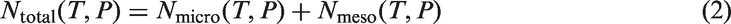
And the gravimetric adsorption amount (in cm3/g STP) was calculated as:

The adsorption amount per gram adsorbent in the microscopic pores, Nmicro (T,P), is related to the PSD via the adsorption integral equation (AIE) (Seaton et al., 1989)

In principle, the similar method can be used to calculate the adsorption amount per gram adsorbent in the mesoscopic pores, Nmeso (T, P). However, using GCMC simulation to calculate Density profiles calculated from (a) Grand Canonical Monte Carlo (GCMC) and (b) canonical ensemble (NVT) simulations of Ar in a pore with a width of 15 nm at pressures of 0.4 and 0.8 P0.
To solve this problem, we divided the adsorption number in the mesoscopic pores into two parts: condensed adsorption near the surface, and gaseous adsorption in the free volume of the pore:
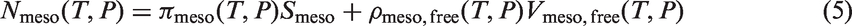

Chemical potentials (CPs) of Ar, CO2, and C6F14 calculated at different temperatures and relative pressures.
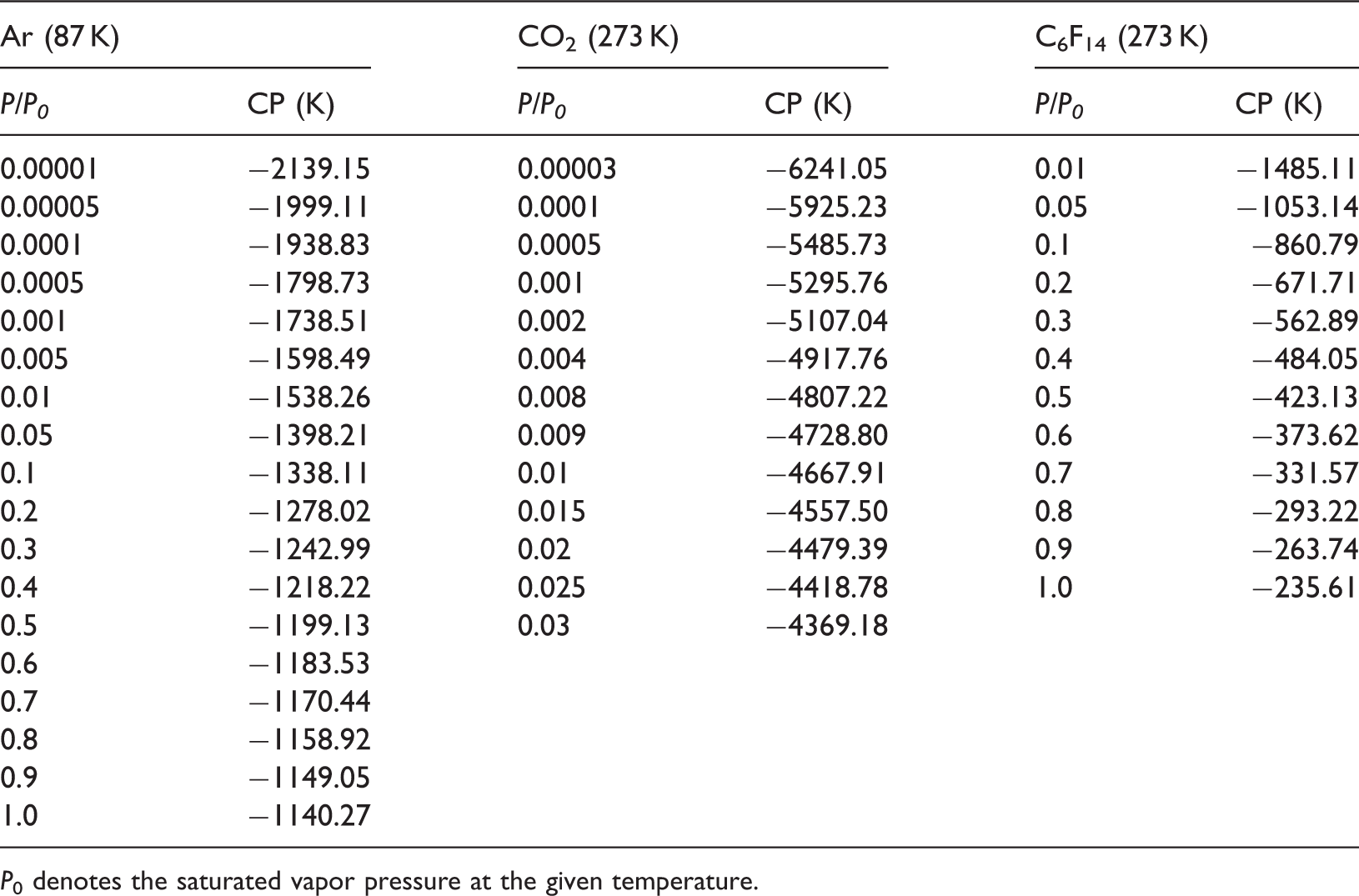
P0 denotes the saturated vapor pressure at the given temperature.
For all simulations, LJ interactions were evaluated using a 12.8 Å cutoff with tail correction. Columbic interactions were calculated using the Ewald summation method. The block-average method was used to estimate prediction uncertainties at each pore size. Total deviations were calculated as:

Results
Validation of force fields for adsorbate molecules
The calculated and experimental VLE curves of Ar, CO2, and C6F14 are shown in Figure 4. The calculated curves are in excellent agreement with the experimental data for Ar and CO2. For C6F14, only liquid densities at temperatures of 293 and 313 K are available for comparison. The agreement obtained is reasonably good.
Comparison of calculated and experimental vapor–liquid equilibrium (VLE) coexistence curves.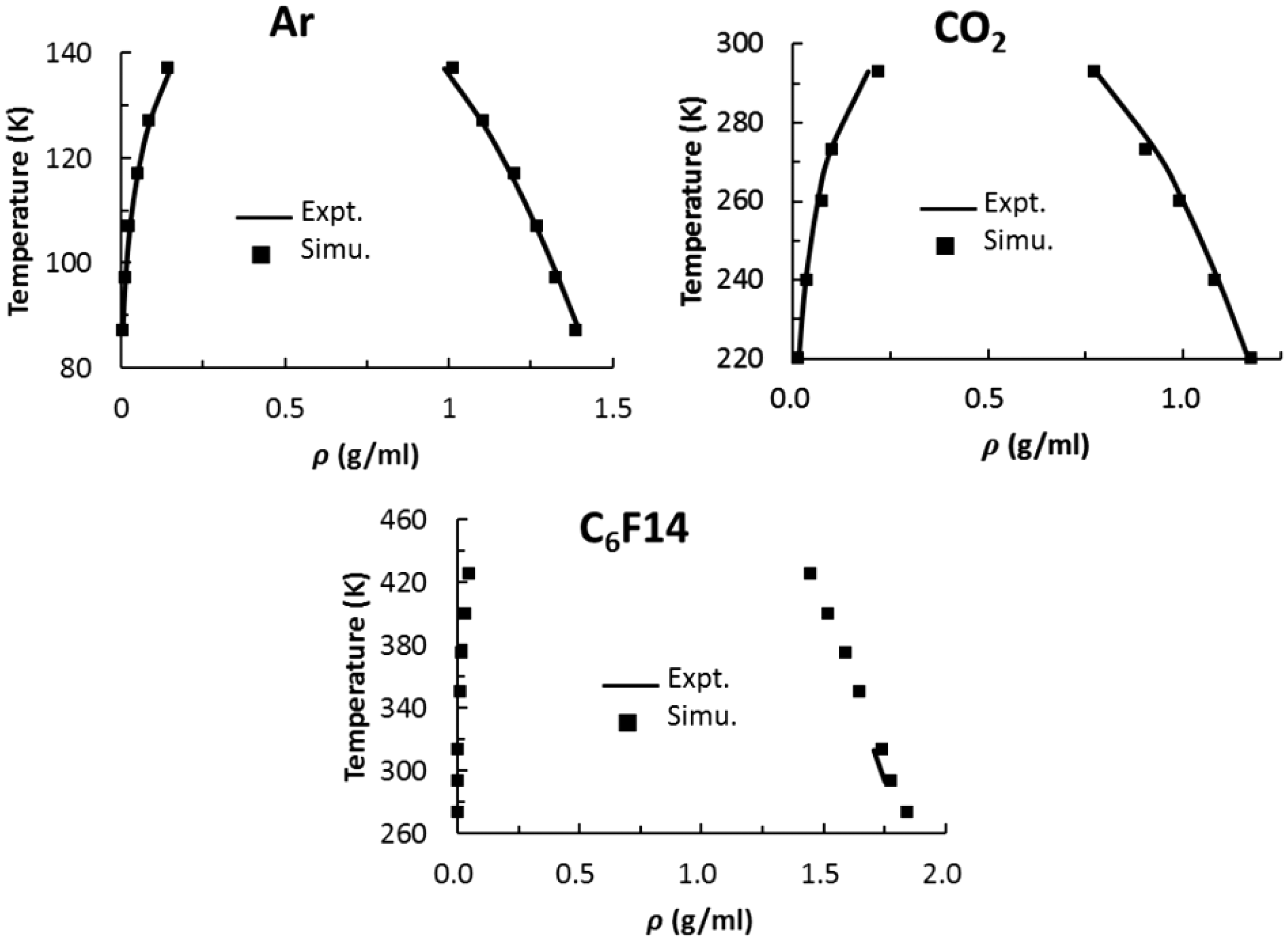
Comparison of calculated and experimental saturated vapor pressures (SVPs).
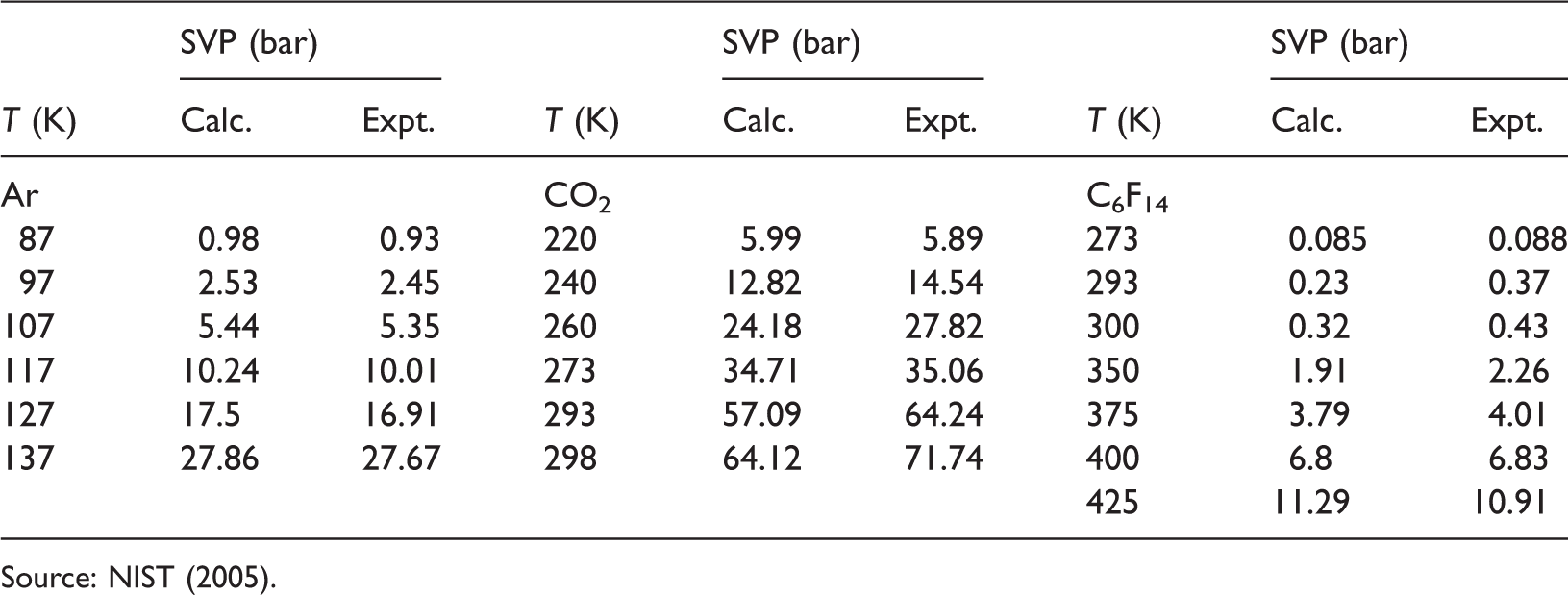
Source: NIST (2005).
Parameterization of interactions
LJ parameters between the adsorbent and adsorbate were optimized by fitting the ab initio binding energy data calculated along the probe paths from one carbon on the graphene surface to the center of mass of the adsorbate molecule. Two orientations of CO2 (i.e. parallel and perpendicular to the probe path) were calculated, and the CF3 UA type was represented by a smaller molecule, C2F6. Fixing the parameters for CF3, the CF2 UA type was parameterized using C3F8. For perfluoro molecule configurations optimized on the surface were fixed in the quantum mechanics energy scan calculations. A total of 155 energy data points were used to determine the LJ parameters. As shown in Figure 5, excellent fit quality was obtained for Ar and the perfluoro compounds, but poor fit quality was achieved for CO2. This poor fitting may have resulted from imbalances in the force field models used for CO2, which has electrostatic terms, and for graphene, which is neutral. We obtained the best fit for CO2 by compromising two probe paths. Finally, the LJ parameters of PFC molecules were validated by calculating the energy curve of C6F14. The results are also given in Figure 5, and excellent agreement is obtained.
Comparison of quantum mechanics density functional theory (QMDFT) (lines)- and force field (squares) calculated binding energy curves.
Adsorption isotherms
The predicted adsorption isotherms for Ar, CO2, and C6F14 are shown in Figure 6. Besides the simulation protocol combining GCMC for microscopic pores and NVT for mesoscopic pores (denoted as GCMC + NVT), the protocol with GCMC simulations for both microscopic and mesoscopic pores was also performed (GCMC + GCMC) for comparison. In the case of Ar, the GCMC + GCMC prediction (open squares) was overestimated at pressures larger than 0.8 P0. For CO2, the adsorption amounts were calculated in the low-pressure range, thus, the difference between the two protocols was not obvious, and the calculated adsorption isotherms were in good agreement with the experimental data. For C6F14, GCMC + GCMC protocol significantly overestimated the adsorption results. As shown in Figure 6(c), although the prediction at low pressure (0.1 P/P0) was very accurate, predictions were highly overestimated at high pressures. Using the GCMC + NVT protocol, the predicted values (solid squares) for C6F14 at relative pressure of 0.1, 0.3, and 0.6 were 53.16, 58.39, and 64.54 cm3/g, respectively, in excellent agreement with the experiment values.
Calculated and experimental adsorption isotherms on BAM-P109 of (a) Ar at 87 K, (b) CO2 at 273 K, and (c) C6F14 at 273 K as functions of relative vapor pressures P/P0.
Conclusions
In this work, we predicted the adsorption of Ar, CO2, and n-C6F14 on BAM-P109 activated carbon using an ab initio force field and slit-like pore model with an experimentally derived PSD. The intermolecular LJ parameters for interactions between the adsorbate and graphite carbon were obtained by fitting the potential energies calculated using QMDFT with a functional containing dispersion energy correction.
The pores were divided into microscopic and mesoscopic pores. For microscopic pores, direct GCMC simulation was applicable. For mesoscopic pores, however, GCMC simulation overestimated the adsorption at high pressures because the coexistence of a vapor–liquid phase cannot be accurately predicted by the simulation. Using NVT simulation to estimate the density and thickness of the adsorbed layers of molecules and the gas phase density for the free volume, we obtained predictions showing good agreement with the experimental data. The success of our work indicates that the ab initio force field developed using DFT with dispersion energy correction can accurately represent interactions between organic molecules and graphene-type carbon materials.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: partially funded by the National Science Foundation of China (No. 21073119, 21173146, 21473112 and 21403138), and National Basic Research Program (No. 2014CB239702), and the computational resources are supported by the High Performance Computing (HPC) Center of Shanghai Jiao Tong University.