Abstract
Background:
The inflammatory state that characterizes COVID-19 may contribute to depression, anxiety, other morbidities, and mortality. Interleukin-6 blocker tocilizumab has been used as a treatment modality in COVID-19 as an anti-inflammatory agent. Tocilizumab has also been studied for its potential use in preventing depression in other patient groups, with mixed results. This study was designed to study subsequent depression, anxiety, and quality of life (QOL) in patients treated with tocilizumab plus standard care versus standard care alone, for moderate to severe COVID-19 pneumonia.
Methods:
Patients admitted with moderate to severe COVID pneumonia, treated with tocilizumab plus standard treatment, and discharged from the hospital were identified. Age- and gender-matched controls who had received standard treatment alone for COVID-19 pneumonia during the same time frame were identified. After obtaining consent, these patients were followed up and assessed at 3 and 6 months on measures of depression (PHQ-9), anxiety (GAD-7), and QOL (EQ-5D-5L).
Results:
39 patients in the tocilizumab group and 41 in the control group were followed up at 3 and 6 months. Patients in both groups were comparable in sociodemographic and clinical parameters. The prevalence of clinically significant depression in the tocilizumab group at 3 and 6 months was 33.33% and 5.12%, respectively, whereas in control group it was 31.7% and 4.87%, respectively. Analysis of EQ-5D health profiles revealed that the maximum problems were reported in the dimension of mobility: 43.7% at 3 months and 35% at 6 months.
Conclusions:
Depressive and anxiety symptoms and impairment in QOL were reported more frequently and with greater severity in patients in the tocilizumab group at three months but not at six months. Psychological morbidity and impairment in QOL were modest and improved from 3 to 6 months.
We investigated the impact of tocilizumab as a treatment modality for moderate to severe COVID-19 pneumonia compared to patients who had received standard care at three- and six-month follow-ups on self-ratings of depression, anxiety, and quality of life (QOL). Variables found to be statistically significantly different in patients with and without clinically significant depression were the use of tocilizumab, presence of clinically significant anxiety, days of hospitalization, and EQ-5D index-score.Key Messages:
COVID-19 is associated with neuropsychiatric manifestations, including depression and anxiety. 1 While the exact pathophysiology of these manifestations is a subject of ongoing research, it is postulated that the inflammatory state brought about by the cytokine storm associated with COVID-19 may be contributory. 2 Previous studies have shown a causative relationship between inflammation that is characteristic of infective disorders and subsequent psychopathology, especially depression and anxiety. 3 Studies have also investigated the possible role of inflammatory modulators in treating depressive disorders. 4 A study on naïve rats suggested that long-term inhibition of peripheral TNF-alpha IL-6 improves anxiety and cognitive function in rats, leading to increased Brain-Derived Neurotrophic Factor expression in the brain. 5 In this context, the interleukin-6 (IL-6) blocker tocilizumab has been investigated as a possible preventive agent or treatment strategy for depression, with mixed results. Two studies in patients with rheumatoid arthritis concluded a beneficial role for tocilizumab in this respect, but these had a small sample size and did not have a control group.6,7 Another methodologically sound study in patients receiving allogeneic hematopoietic stem cell transplantation reported more depressive symptoms in patients who received tocilizumab than the controls. 8 Population-based studies have also shown that higher levels of serum IL-6 and c-reactive protein are associated with fatigue and impaired sleep but not hopelessness. Cytokine-induced somatic symptoms may affect mood by reducing rewarding experiences and so could mediate the relationship between inflammation and depression. 9
Severe COVID-19 is characterized by a pro-inflammatory state leading to morbidity and mortality, and for this reason, tocilizumab has been used in India and elsewhere.10,11 Given the high incidence and prevalence of depression and anxiety in post-COVID patients, it has been postulated that blockade of inflammation associated with COVID-19 with an agent such as tocilizumab may be associated with fewer neuropsychiatric sequelae, including depression and anxiety. 12 Also, given the number of patients with severe COVID-19 treated with immunomodulatory agents, including tocilizumab, and the high prevalence of depression and anxiety in post-COVID in these patients, there is an opportunity to assess whether or not an immunomodulatory agent such as tocilizumab can influence the development of future psychopathology.
A literature review revealed a scarcity of data, with conflicting findings. One study concluded that at three months, immunomodulatory therapy, including that by tocilizumab, was beneficial in protection from depression. 12 This study assessed the effects of tocilizumab individually in addition to other immunomodulatory agents. Another study found no benefit of immunomodulatory therapy, at three months, in depression or anxiety. 13 This study, however, did not evaluate the effects of tocilizumab individually.
Moreover, COVID-19 can potentially worsen patients’ overall health by limiting their capacity to live well and diminishing productivity, thus negatively impacting their health-related QOL. Impaired QOL may contribute to subsequent psychopathology. The measurement of QOL becomes important to capture the broadened definition of health that goes beyond accounting for just the traditional measures of mortality and morbidity.
Against this background, this study was conducted with the aim of evaluating the effect of tocilizumab on the development of subsequent depression and anxiety in patients with COVID-19 compared to age- and gender-matched controls with COVID-19 who were not treated with tocilizumab. This study aimed to assess if administration of tocilizumab in patients with moderate to severe COVID-19 pneumonia leads to reduced occurrence of anxiety and depression and improved QOL, compared to patients who receive standard care.
Materials and Methods
Study Design and Population
The study protocol was approved by the Institute Ethics Committee. This non-funded study was conducted in a cohort of patients selected from patients discharged from the COVID-19 hospital within a tertiary hospital in North India. Records of patients aged ≥18 years who had received tocilizumab while admitted in the COVID hospital for COVID-19 pneumonia, from February 2021 to June 2021, were traced. The study period corresponded to the second wave of the COVID-19 pandemic in India, which was characterized by widespread morbidity and mortality. 14 Controls were selected from age- and gender-matched patients discharged during the same period from the COVID hospital who had received treatment for COVID-19 pneumonia but had not received tocilizumab. The protocols followed for use of tocilizumab are described elsewhere. 11 Briefly, patients in the two groups were admitted to the hospital with PaO2/FiO2 < 200 and received at least standard care as per the hospital treatment protocol. Standard care included oxygen supplementation; intravenous (i.v.) remdesivir loading dose of 200 mg on day 1, followed by 100 mg for the next four days; i.v. dexamethasone 6 mg for 10 days; therapeutic low-molecular-weight heparin 1.5 mg/kg/day; and proning. Within 48 hours of the initiation of standard care, if a patient showed clinical worsening, they received a single i.v. infusion of tocilizumab 6 mg/kg plus standard care of 6 mg dexamethasone for 10 days. The low dosing of tocilizumab was based on a previous study and due to paucity of the availability of the drug during this time. 15 Thus, patients in both the groups had been treated for moderate to severe COVID pneumonia during the hospitalization.
Patients thus identified were approached for participation in the study at follow-up or telephonically. Written informed consent was obtained from all who agreed to participate. Baseline sociodemographic and clinical details were obtained from the case records. These patients were then evaluated at 3- and 6-months post-discharge. All the interviews were administered in Hindi language, which is a primary language of interaction in the region of the country where the study hospital is located.
Instruments
The following instruments were used at the 3-month and 6-month follow-up evaluation.
Patient Health Questionnaire-9 (PHQ-9): This is a commonly used self-rated questionnaire for diagnosing and quantifying depression. Validated translations in Hindi and Punjabi are available. 16
General Anxiety Disorder Scale-7 (GAD-7): This is a commonly used self-rated questionnaire for diagnosing and quantifying anxiety. 17
EQ-5D-5L: This is a commonly used instrument for the measurement of health-related QOL. 18 This self-rated instrument measures QOL in the domains of mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. These domains can be scored at five levels of severity. These values are then used to generate a single EQ-5D utility score ranging from 0 to 1. A score closer to 1 indicates better QOL. In addition, a visual analog (EQ-VAS) assesses self-rated overall QOL and is marked from 0 to 100. Higher scores are indicative of better QOL. A recently published Indian value set for this instrument was utilized in this study. 19 Existing evidence also suggests that the EQ-5D has benefits for capturing anxiety severity and is responsive when severity changes over time. 20
Patients having current or past mental illness or receiving any psychotropic medication were excluded from the study.
Statistical Analysis
The independent sample t-test was used to assess the presence of statistically significant differences between the tocilizumab and control groups in terms of mean age and mean duration of hospitalization, whereas the chi-square test was used to assess the difference in terms of sex, presence of comorbidities, and history of substance abuse. Further, independent sample t-test was used to compare the groups in terms of mean PHQ-9 score, mean GAD-7 score, mean EQ-VAS score, and EQ-5D utility score at both 3- and 6- months post-discharge evaluations. The proportion of patients reporting problems in different attributes of EQ-5D-5L was assessed in both study groups. A bivariate analysis was performed to compare the differences in clinically significant depression across sex, age, days of hospitalization, substance abuse, clinically significant anxiety, history of having received tocilizumab, EQ-5D index scores, presence of diabetes, and presence of hypertension. For this analysis, all the patients with PHQ-9 scores ≥5 at three months follow-ups were considered to have clinically significant depression, and patients with GAD-7 scores ≥5 were considered to have clinically significant anxiety.
Results
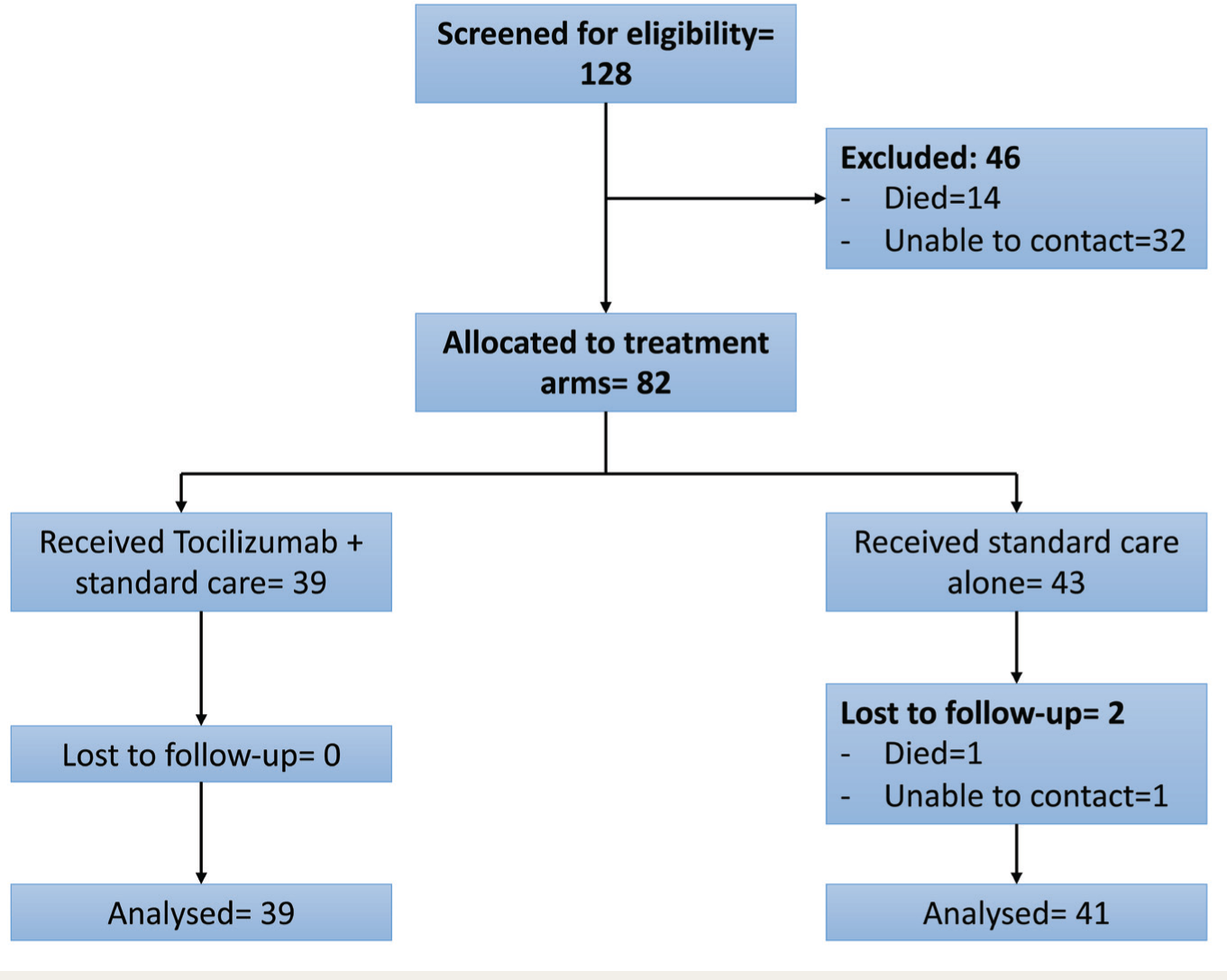
We identified 60 patients who had received tocilizumab plus standard care and 68 age- and gender-matched controls who had received standard care alone. 11 Seven of the patients in the tocilizumab group died before they could be contacted, and 14 were not traceable (tocilizumab group n = 39). Among the controls, seven died before they could be contacted, and 18 could not be traced (control group n = 43). All the patients in the tocilizumab group who entered the study at three months post-discharge could be followed up at six months. Among the control group, one patient died, and one could not be traced at the second follow-up (control group n = 41) (Figure 1). None of the patients had any self-declared mental illness. At three months, face-to-face follow-up was done in 87.8% of the patients, and telephonic follow-up was done in 12.2% of the patients. At six months, face-to-face follow-up in was done in 86.25% of the patients, and telephonic follow-up was done in 13.75% of the patients.
CONSORT Figure Demonstrating the Recruitment of Study Participants.
The tocilizumab and control groups were comparable on most sociodemographic and clinical parameters (Table 1). This included the mean number of days spent in the hospital. As has already been mentioned, patients in both groups were treated for moderate to severe COVID pneumonia. The severity and classification of depression and anxiety symptoms and QOL at 3- and 6-months post-discharge are presented in Table 2. The tocilizumab group had significantly more depressive and anxiety symptoms and worse QOL than the controls at 3-months follow-up. These differences were not statistically significant at six months. The change in scores of depressive and anxiety symptoms and QOL within the two groups at 3- and 6-months follow-up is presented in Table 3. The tocilizumab group had more and statistically significant reduction with regards to depressive and anxiety symptoms and QOL than the control group.
Sociodemographic and Clinical Profile
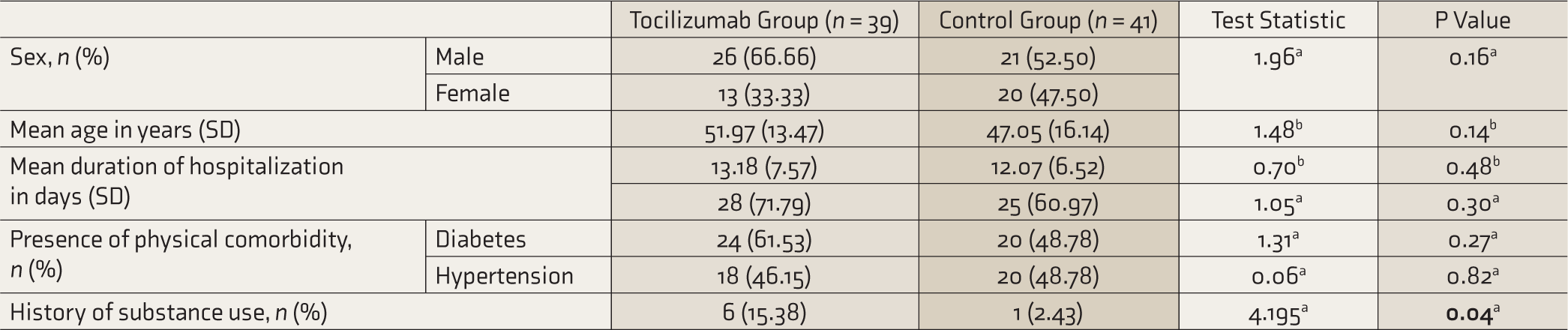
aChi-squared test. bt-test. P significant when ≤ 0.05 (significant values in bold).
Depression and Anxiety Symptoms and QOL at 3- and 6-Month Post-discharge.
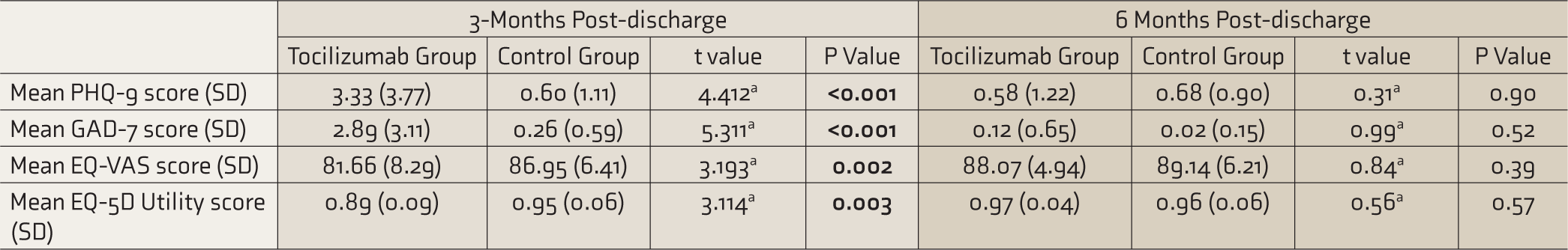
QOL: quality of life. at-test. P significant when ≤ 0.05 (significant values in bold font).
Change in Scores in the Tocilizumab and the Control Groups from 1st to 2nd Follow-up.
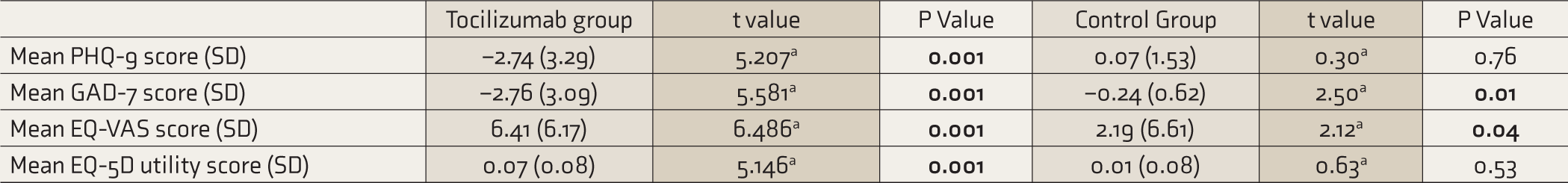
aPaired t-test. P significant when ≤ 0.05 (significant values in bold font).
Regarding the QOL, at the three months follow-up, maximum problems were reported in the dimension of mobility (43.7%). In the tocilizumab group, maximum problems were reported in performing usual activities (59%), closely followed by problems in mobility (56.4%). In contrast, in controls, maximum problems were reported in mobility (31.7%). Overall, the extent of reporting problems was higher in the tocilizumab group. Also, whereas 28.2% of the tocilizumab group reported having anxiety/depression of some degree, only 12.2% of the control group reported having anxiety/depression.
At the time of the second follow-up (six months post-discharge), similar to the findings of the previous follow-up, the maximum problems were reported in mobility (35% of the patients; 13 patients in tocilizumab group and 15 patients in control group). However, the percentage of patients reporting the problems decreased by 8.7%. At the time of the second follow-up, 33.3% and 36.6% of the tocilizumab and control groups reported mobility problems. Further, at six months post-discharge, 2.6% of the tocilizumab group reported the problem of anxiety/depression, whereas 28.2% had reported the same problems at the time of the first follow-up. None in the control group reported problems with anxiety/depression at six months post-discharge, whereas 12.2% had reported such problems at the first follow-up.
To identify the presence of clinically significant depression across different patient groups, we performed an exploratory subanalysis in which all the patients with PHQ-9 scores ≥5 at three months follow-up were considered to have clinically significant depression. Using the presence or absence of clinically significant depression as the dependent variables, we performed chi-square tests in which sex, age (≥ 45 years), days admitted in hospital (≥ 10 days), presence of substance use, presence of clinically significant anxiety at three months (≥5 on GAD-7), history of having received tocilizumab, EQ-5D index score at three months ≥0.90, presence of diabetes mellitus, and presence of hypertension were the independent variables. Variables found to be statistically significantly different in the two groups (clinically significantly depressed versus not) were tocilizumab (P = 0.001), clinically significant anxiety (P = 0.008), days of hospitalization (P = 0.002), and EQ-5D index-score (P = 0.001).
Discussion
Depression and impaired QOL have been reported frequently in patients with COVID-19. Given the pandemic nature of COVID-19 and the number of patients, psychological morbidity is a major public health crisis in this population. 21 The frequency of clinically significant depression at three months has been reported to be 11%–28% and is associated with female sex, previous history of depression, and initial psychopathology. The association of depression with inflammatory markers and disease severity is unclear. 22 However, as reviewed above, inflammation has been postulated to contribute to this association. Thus IL-6 blocker tocilizumab may have a potential role in preventing depression.23,24
Using validated self-rated instruments, we found that depressive and anxiety symptoms and impairment in QOL were more frequent and severe in the tocilizumab group at three months but not at six months. Contrary to expectation, the severity of depressive and anxiety symptoms was modest. This is in keeping with a follow-up study conducted in this center in the past on patients with asymptomatic to mild COVID-19, which revealed mild depressive symptoms at three months. 25 Similar results have been reported in other studies as well. 22 Lower-than-expected rates of depressive and anxiety symptoms may also be attributable to the self-rated nature of assessments. For instance, using a clinician-rated instrument, a study on the effects of immunosuppressive therapy in COVID-19 reported higher levels of depression at three months. 13
The mean EQ VAS score and EQ-5D-5L utility score of the Indian population has been reported to be 75.18 (95% CI: 74.51–75.85) and 0.848 (95% CI: 0.84–0.857), respectively. 19 Our patients reported higher EQ-VAS scores than this. One reason for this difference could be that our patients were predominantly from the North Indian states (Punjab, Haryana, Himachal, and Union Territory of Chandigarh), which have better socioeconomic indicators than the rest of India and hence a better baseline QOL.19,30 In contrast, the Indian valuation study was conducted on a nationally representative sample with respondents from states performing low on the indicators of income, health, and education, which are associated with lower QOL. For instance, in a study assessing QOL of patients with rheumatic fever (RF) and rheumatic heart diseases (RHD) in Punjab and the union territory of Chandigarh, the mean EQ VAS score for RF, RHD, and RHD with congestive heart failure were estimated as 94 (95% CI: 92.7–95.3), 84 (95% CI: 82.8–85.3), and 79.8 (95% CI: 78.5–081.1), respectively. 24 Similarly, a study conducted at this center has reported modest impairments in QOL in patients with COVID-19 who were mechanically ventilated. 27
Studies have shown that COVID-19 leads to impairments in QOL that gradually show an improving trend.28,29 Similar to results reported earlier in an Indian sample, in our study, QOL was most affected by the presence of pain/discomfort, followed by mobility problems, difficulties in self-care activities, difficulties in performing usual activities, and the presence of anxiety/depression. 30 This supports the modest levels of depression and anxiety symptoms reported on PHQ-9 and GAD-7. Further, in post-COVID patients, depression and anxiety may well be a part of the general impairment of QOL following an acute infection rather than an episode of endogenous major depression. Another potential confounding factor could be the patients whom we could not contact. Those may have been the patients with the worst outcomes and mortality following discharge.
Our study has many limitations. Firstly, only a single dose of tocilizumab could be administered to eligible patients due to the scarce availability during the pandemic, which may be inadequate to demonstrate the beneficial effects pertaining to neuropsychiatric symptoms. Secondly, although all the patients aged ≥18 years who had received tocilizumab during the study period were traced, the number of patients in both groups was relatively lesser. Thirdly, as the evaluations were done after 3 and 6 months post-discharge, the estimation of disease severity could not be done at the time of the acute episode of disease/admission. Though we had given the single i.v. infusion of tocilizumab to only those patients who showed clinical worsening within 48 hours of the initiation of standard care, as presented in Table 1, there was no difference between the two groups in the mean days of hospitalization, one of the indicators of disease severity. Fourthly, to minimize the loss to follow-up, some follow-up evaluations had to be done telephonically. However, it has been established that the internal consistency of the telephone-administered instruments assessing depression was high and close to the self-administered one, thus making it a reliable procedure. 31 Another limitation is that patients with PHQ-9 score ≥5 were considered to have clinically significant depression and patients with GAD-7 score ≥5 were considered to have clinically significant anxiety. These operational cut-offs were considered to differentiate between patients with no/minimal symptoms and those with mild to severe symptoms. Although similar cut-offs have been used in some earlier studies, 32 it contrasts with most of the other studies employing a cut-off score of 10, limiting the comparability of the results. 33
Our results suggest that depression and anxiety symptoms and impairment in QOL are modest and improve with time. The improvement in psychological morbidity and QOL during the 3–6 months of the follow-up can be a natural course of recovery from depression and hence reflected in improved QOL. Nevertheless, the measurement of QOL may be a useful component of follow-up care in patients who have had COVID-19. The unexpected nature of some of the results suggests the need for future studies in this area.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
