Abstract
Background:
Substance use among adolescents is increasing rapidly and becoming a global health concern worldwide. Because of the changing trends and rising magnitude in India, there is an urgent need to adapt and validate instruments to assess adolescents’ substance use. The study translated the Teen-Addiction Severity Index (T-ASI) into Hindi and assessed the psychometric properties.
Methods:
The instrument (T-ASI) was translated from English to Hindi using: (a) forward translation: translation from the source language into the target one, (b) comparison and synthesis of the two translated versions, (c) blind backward translation, (d) comparison of the two back-translated versions and development of prefinal version, (e) conceptual evaluation of items and pilot study, and (f) assessing the psychometric properties.
Results:
The content validity of Hindi T-ASI was quite high (0.97). A significant positive correlation
Conclusion:
The translation and validation of Hindi–T-ASI show adequate psychometric properties and can be recommended for use in treatment settings across the country.
Adolescent substance abuse in India has reached an alarming state. Considering the present situation, an adequate assessment of the impact and addiction severity is highly needed. The study provides a valid and reliable version of the Teen-Addiction Severity Index which is a widely used instrument in adolescent substance use.Key Message:
Considerable recent epidemiological evidence from India suggests high rates of substance use among adults but, more importantly, among adolescents.1, 2 The adolescent population reportedly uses both legal and illegal substances. The national survey 1 reported the prevalence of alcohol (1.3%), cannabis (0.9%), opioids (1.8%), and inhalants (1.17%) among children and adolescents (10–17 years) in the community. The proportion of young substance users in treatment settings has increased over time in India. 3 There is thus an urgent need to adapt and validate instruments to assess adolescents who use substances. Research over the past two decades has provided health professionals with various English-language instruments for assessing drug-related problems among adolescents 4 that are developmentally appropriate. 5 Among these, the adolescent version of the Addiction Severity Index (ASI), 6 the Teen Addiction Severity Index (T-ASI), is relatively brief and easy to administer in a face-to-face interview. It is a structured interview developed to assess seven domains: chemical use, school status, employment-support status, family relationships, peer-social relationships, legal status, and psychiatric status. This instrument helps identify the severity of addiction among the youth and its potential consequences in different domains. Accurate screening and identification of substance use behaviors is a prerequisite for planning treatment, assessing service needs, and evaluating outcomes of interventions.
T-ASI has good reliability (inter-rater
This study aimed to translate T-ASI into the Hindi language, validate the Hindi version, and examine its psychometric properties among adolescents with substance use disorders. It was assumed that this validated and cross-culturally adapted version would assist the assessment of substance-using Indian adolescents (SUA) and make between-country comparisons feasible.
Material and Methods
The sample comprises 82 adolescent males aged 12–19 years: 32 SUA consecutively seen for treatment between August 2016 and December 2018 at the adolescent’s specialty clinic run by a tertiary care center and 50 normal healthy adolescent siblings recruited from the SUA’s families. Those having any history of severe psychopathologies like psychosis, organic mental disorders, mental retardation, severe depression, or seizures and those abusing tobacco only (in case of SUA) were excluded. The Institute Ethics Committee approved the protocol. No compensation for participation was provided.
The study had two phases. In the first phase, the instrument was translated from English to Hindi using the World Health Organization (WHO) guidelines 12 and the methodology recommended by the available literature. In the second phase, the instrument was validated for the Indian population using a representative sample, and the psychometric properties of the Hindi version of the test were evaluated.
Instruments
Teen-Addiction Severity Index (T-ASI) 8
It is a semi-structured interview developed to fill the need for a reliable, valid, and standardized instrument for a periodic evaluation of adolescent substance abuse. It has 154 questions containing seven domains and takes 20 min to 45 min to administer and 10 min to score.
Youth Self-Report Inventory (YSR) 13
It was designed for use with adolescents between 11 and 18 years. Using the child-report measure, it assesses problem behaviors using two “broadband scales”–internalizing and externalizing. It scores eight empirically based syndromes and DSM-oriented scales. There are 112 items, which take about 15 min to complete and 10 min to score with clinical cutoffs.
Family Environment Scale (FES) 14
It has three forms: R (real–people’s perceptions of family functioning), I (ideal–people’s perceptions of the family they would ideally like), and E (expectations–what people expect a family climate to be like). The FES comprises 10 subscales measuring three underlying dimensions of the family environment. The FES subscales have been found to have good internal consistency and test–retest reliability.
Procedure: Study Phases
After obtaining written informed consent and assent from the adolescents and their parents or guardians, the adolescents were assessed on the instruments. The translated Hindi T-ASI was administered to 32 SUA along with Achenbach’s YSR and FES. Besides, the original and translated versions of T-ASI were administered to 50 bilingual (fluent in the original language and the target language) normal healthy male adolescents.
Phase 1: Cross-cultural adaptation—this involved five steps:
Step1: Forward Translation
Two independent translators forward translated the T-ASI English version into Hindi. Thus, two translated versions that covered both the medical jargon and the usual spoken language with its cultural nuances were generated.
Step 2: Comparison of the Two Translated Versions
The instruments were compared for their semantic, idiomatic, conceptual, linguistic, and contextual differences, and the two forward-translated versions were reconciled into one.
Step 3: Blind Back Translation of the Preliminary Initial Translated Version
The reconciled initial Hindi translated version was translated back into English by two independent translators, with the purpose of identifying words that were not clear in the target language, resulting in two back-translated English versions.
Step 4: Comparison of the Two Back-Translated Versions
This process resulted in a refined prefinal second draft of the Hindi translation.
Step 5: Pilot Testing with a Monolingual Sample
Cognitive debriefing: the prefinal Hindi version was pilot-tested on 10 participants (apart from the 32 SUA) whose primary language was the target language (Hindi). Instructions, response format, and items of the instrument that were found to be unclear by at least 20% of the sample were reevaluated. 15 Therefore, the minimum inter-rater agreement among the sample was 80%. Following this process, a final version of the translated and culturally adapted Hindi T-ASI entered the cross-cultural validation phase.
Content Validity Index at the Item Level (I-CVI)
An expert panel of six members evaluated each item for content equivalence (content-related validity). For each item, the I-CVI was computed as the number of experts giving a rating of either 3 or 4 divided by the total number of experts. Items that were classified as 1 or 2 and did not achieve the minimum acceptable indices were revised and reevaluated as per the Lynn criteria. 16
Content Validity at Scale Level (S-CVI)
The S-CVI was computed using the S-CVI/Ave method. Researchers often use the guidelines offered by Waltz et al. 17 of using the S-CVI/Ave approach with 0.90 as the standard for the index of average congruity. All items with unacceptable I-CVIs were revised and reevaluated to meet the standard criteria.
Phase II: Cross-Cultural Validation and Assessment of Psychometric Properties
The Hindi T-ASI was administered on 32 SUA. These participants also completed YSR and FES for comparison of T-ASI composite scores with corresponding domain scores of these instruments. Psychometric properties were assessed on the 32 SUA, viz. concurrent validity by comparing the T-ASI family functioning domain with FES, and convergent validity by comparing (a) chemical use domain with YSR behavior problems scale and (b) psychiatric status domain with YSR clinical domains and internalizing and externalizing scores.
The discriminant validity was assessed by comparing the 32 SUA with 50 NSA. Similarly, for cross-cultural validation, the new instrument (T-ASI Hindi) was pretested on the NSA (12–19 years) along with the original version (English). Ideally, the bilingual sample should be from the target population in which the instrument will be used, but we were unable to get bilingual SUA because most of them were coming either from lower socioeconomic background or were school dropouts. So, according to the provision given in the literature,18, 19 the other alternative was chosen.
Cronbach’s α was calculated for finding the reliability of the newly validated instrument.
Data Analysis
Data analysis was done using IBM SPSS Statistics for Windows, version 23.0 (IBM Corp., Armonk, NY, USA). Pearson’s correlation coefficient was used to assess the correlation of FES and YSR internalizing, externalizing, and behavior problem scales with Hindi T-ASI. The Independent sample
Results
A detailed account of the findings are given below:
The demographic characteristics of the sample(SUA and NSA) show that majority of them are Hindu and having a mean age of 15.91 & 14.36 for SUA and NSA respectively (see Table 1).
Demographic Characteristics of the Sample (SUA and NSA)
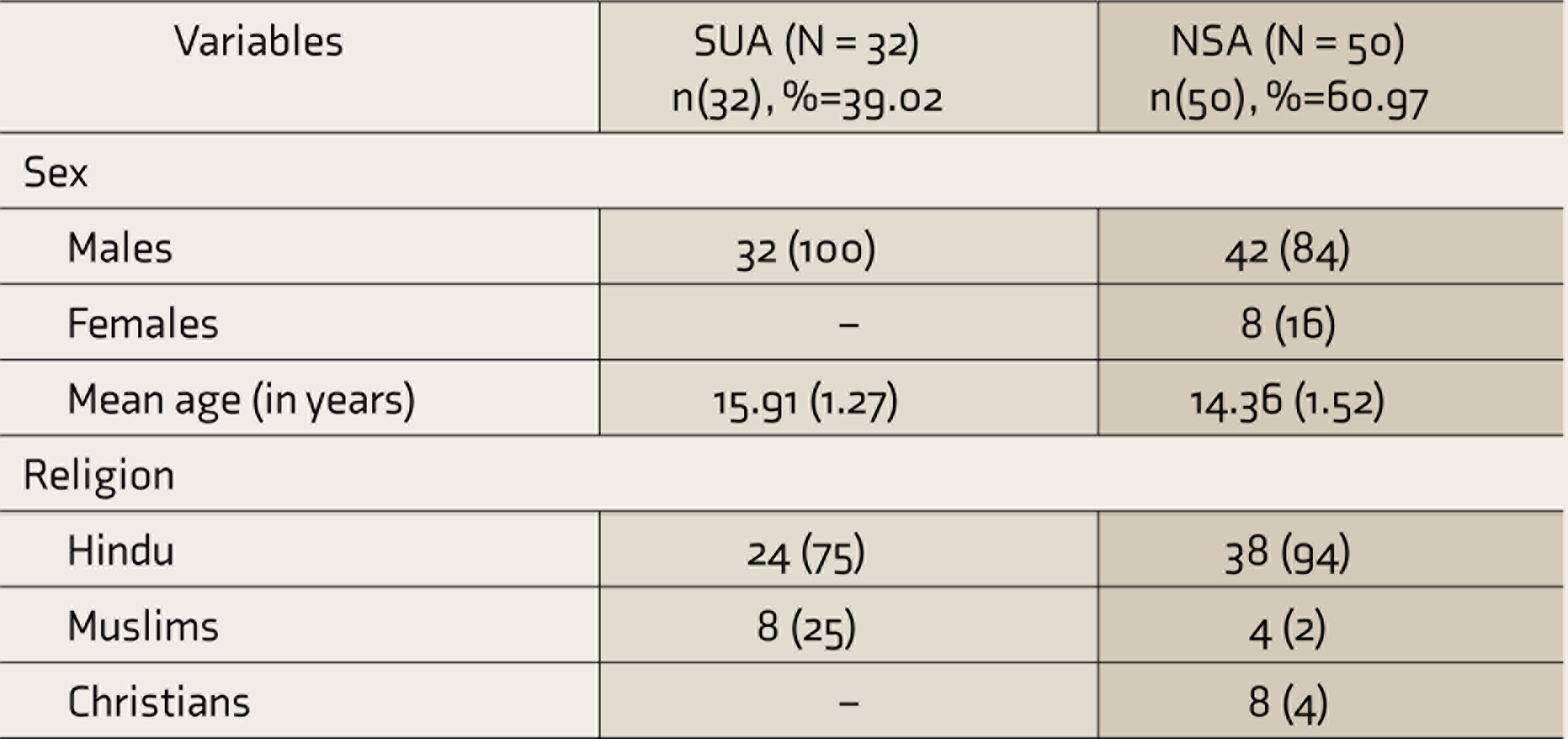
SUA, substance-using adolescents; NSA, nonsubstance-using adolescents.
Clinical Characteristics of the SUA and NSA
The mean age of the onset of substance use was 11 years (SD = 2.32, range 7–16). The mean year of substance use was 5.2 years (SD = 2.8). There was a wide variation in the amount spent monthly on substance use (₹100–3,00,000 per month). Tobacco was used by all the participants, 32 (100%). There were 21 (65.6%) adolescents using street heroin/smack, 19 (59.4%) were using alcohol, and 10 (31.3%) used IV and inhalants both. A small group of adolescents used cannabis 1 (3.1%) and cocaine 2 (6.3%). Majority 27 (84.4%) reported school-related negative consequences followed by familial 26 (81.3%), social 24 (75%); and psychological 17 (53.1%). Only 12 (37.5%) reported legal consequences.
Among the NSA, the academic performance of these adolescents was either average 26 (52%) or poor 16 (32%) along with 3 (6%) school dropouts. Only a few of them 5 (10%) had good academic performance. Family conflict was also reported by 12 (24%) of the adolescents and 1 (2%) reported having drug-using friends and legal conflict.
Content Validity
The I-CVI of the retained items ranged from 0.83 to 1.0. S-CVI was 0.82. As an S-CVI of 0.82 is not acceptable according to the Waltz criteria, 17 items that did not achieve the minimum acceptable indices and had low agreement were rephrased and given an acceptance rating again by the reviewers. After revising the items, the S-CVI/Ave came out to be 0.97 (Table 2).
S-CVI of the Hindi T-ASI
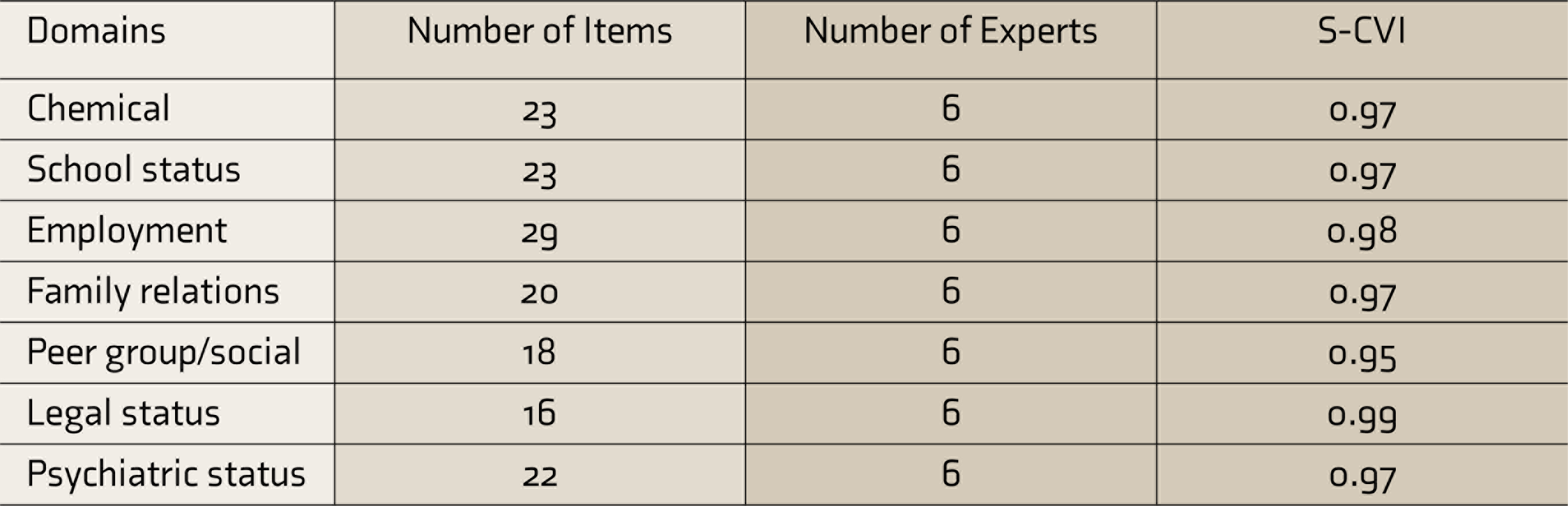
S-CVI, scale-level content validity index; T-ASI, Teen-Addiction Severity Index.
Concurrent Validity
To check the concurrent validity of the Family functioning scale of the Hindi T-ASI, the scores obtained in it were correlated with FES scores. A significant positive correlation (
Pearson Correlations Between Hindi T-ASI Family Function Scale and FES
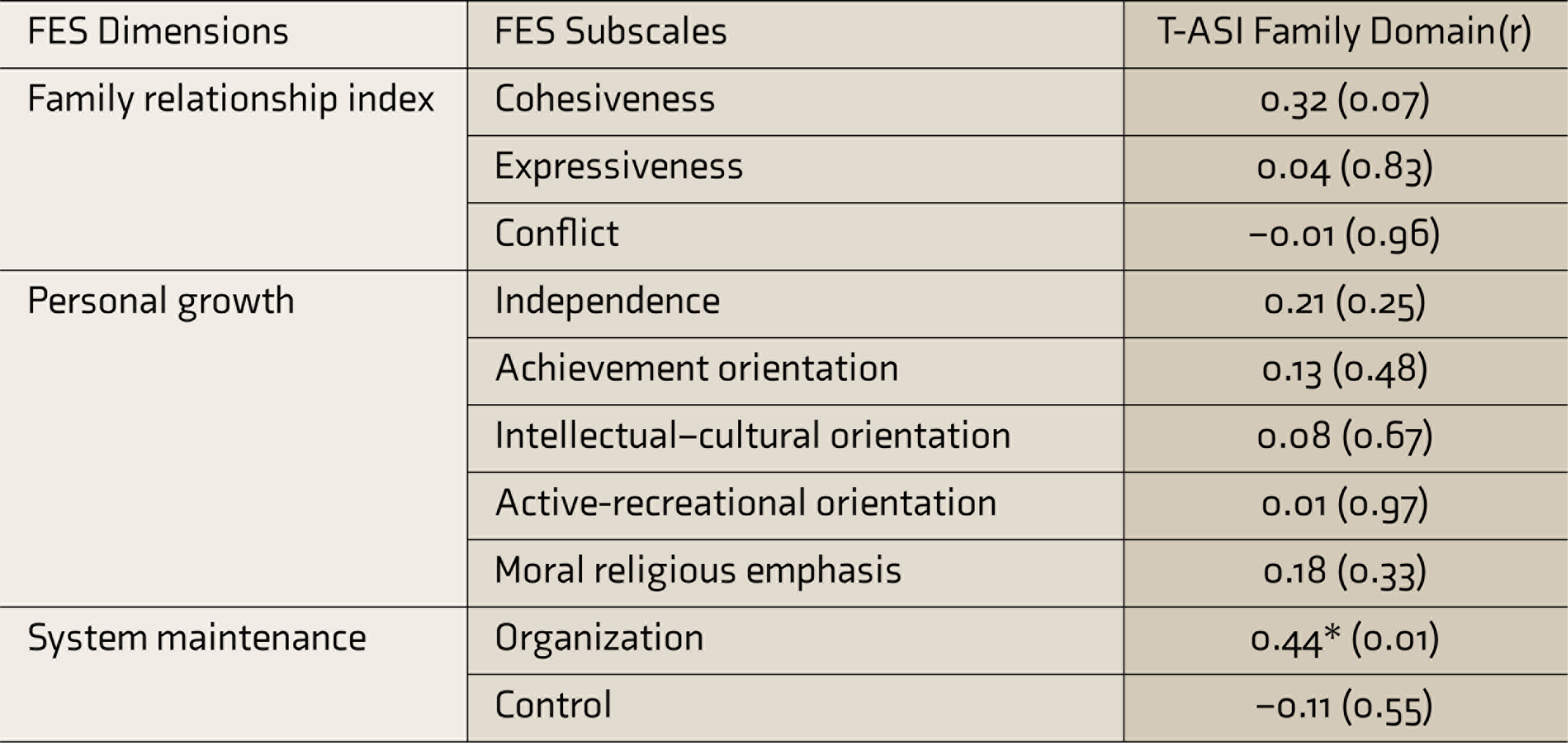
FES, Family Environment Scale; T-ASI, Teen-Addiction Severity Index; r, correlation coefficient.
Convergent Validity
This was obtained through the correlation between the Hindi T-ASI substance use domain and YSR behavior problems scale (
Pearson Correlations Between Hindi T-ASI Domains and External Validators: YSR and CBCL
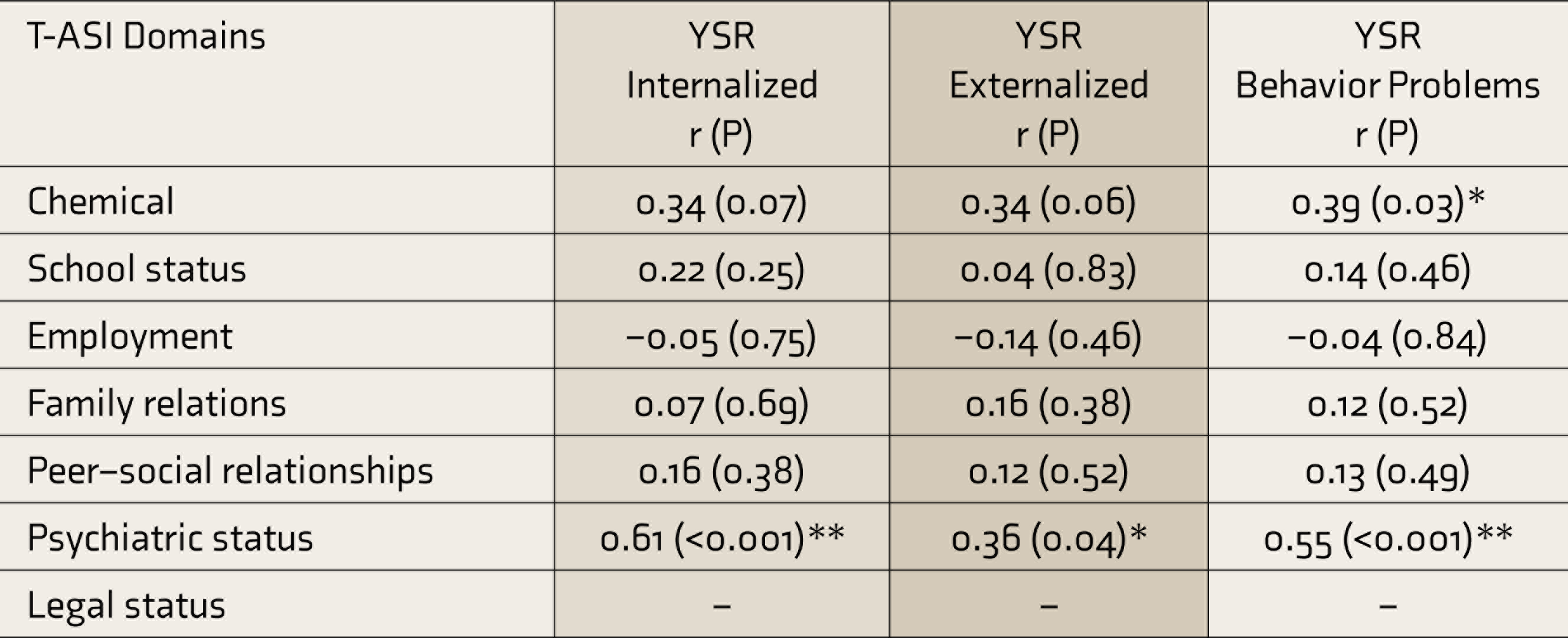
T-ASI, Teen-Addiction Severity Index; YSR, youth self-report; r, correlation coefficient.
Discriminant Validity
This was assessed by comparing scores of SUA with NSA. The independent sample
Cross-Cultural Validity
This was obtained through a paired
Reliability Coefficient
Cronbach’s α was calculated to see whether the test has satisfactory item-total correlations or not and was found to be α = 0.73.
Discussion
The T-ASI was translated to Hindi to make available a scale in Hindi to measure addiction severity among adolescent substance users in India. Using a step-by-step approach, it was translated from English into Hindi and back-translated into English and then validated. The process was a set of methods used for instrument translation15, 18 such as combined translation, pretest, and decentering.
Because the main emphasis was on translation and validation of the T-ASI for the Indian population, the instrument’s content validity was computed. The study provides a clear-cut distinction between content validity at the item and scale levels. 19 I-CVI of the retained items was quite high (5 out of 6 raters gave high acceptance for all the items) and finally ranged between 0.95 and 0.99, which is as high as what Davis 20 had recommended, thus establishing a satisfactory reason to accept the Hindi version of T-ASI. Detailed information about how the CVI values were calculated can help the potential users of the scale to draw informed conclusions about the scale’s content validity.
The family relations domain of the Hindi T-ASI showed a significant correlation with the organization dimension of FES, establishing its concurrent validity. The T-ASI psychiatric status domain correlated with YSR internalizing and externalizing scores and clinical domains, except for somatic and rule-breaking subscales. The chemical domain of the T-ASI correlated with the YSR behavior problems scale, thus establishing that the Hindi T-ASI is strongly correlated with other measures that are already existent and valid.
The construct validity (convergent and discriminant) was assessed through the known-groups technique.
21
The responses of the SUA on the Hindi version of T-ASI were compared with scores of the NSA. The independent sample
For the convergent validity, the chemical subscale and psychiatric status subscales were expected to correlate with YSR. The T-ASI Psychiatric Status scale correlated well with YSR clinical domains (except somatic and rule-breaking scales) and internalizing and externalizing scores. The chemical use subscale correlated with the YSR behavior problems scale. Thus, it verifies that the Hindi version of T-ASI is strongly correlated with other measures that are already existent and valid.
Cross-cultural validation is time-consuming and requires enough planning and expertise. It requires the adoption of a rigorous methodological approach to obtain good reliability and validity. This paper has reviewed and utilized the highly recommended methodological approaches and shows that the original instrument and another one that is culturally adapted are equivalent as no significant difference between the two forms was found. Further, the Hindi version of the tool was found to be reliable enough to use with SUA as the tool had good internal consistency.
Our findings are similar to the findings of the original study by Kaminer et al., 22 where the original T-ASI psychiatric status domain correlated significantly with both the internalizing and externalizing scores from the YSR. The findings are also consistent with the findings of Díaz et al. 10 who validated the Spanish version of the T-ASI and similarly found that the family relationship domain of the T-ASI correlated with the FES Family conflict scale and that the psychiatric status domain also had a good concurrent validity with YSR internalization, externalization, and behavior problem scales, thus showing no differences in terms of convergent validity between the Indian and the Spanish versions. Similarly, the findings are also consistent for discriminant validity as both versions showed good discriminant validity.
The Brazilian Portuguese version of T-ASI, 11 had good internal consistency, with Cronbach’s α coefficients as follows: chemical use- 0.89, psychiatric- 0.80, and legal- 0.81). The Hindi version of T-ASI has almost similar psychometric properties (Cronbach α = 0.73) and, therefore, shadows good cross-cultural validity.
The validity of the Hindi version assessed in different forms has got satisfactory values as generally, a correlation above 0.50 is assumed to be good. 23 Similar findings were reported by De Von and Ferrans 24 who examined the psychometric properties of four quality of life tools and found that a moderate correlation might be strong evidence of validity.
Various other studies have been done where different researchers developed/translated and validated the instruments to assess constructs like spirituality, pain, and QOL measures.25, 26 The methodology utilized in the translation, adaption, and validation of the instruments was almost similar to our steps in this study. Despite the disparity in the research areas and disagreement on some points, researchers were unanimous in considering reliability and validity as the instrument’s main measurement properties.
Thus, the literature provides good support for the Hindi version of the T-ASI as a reliable and valid assessment tool for adolescents with substance use and related behaviors. It also shows the clinical and research utility of the Hindi version of the T-ASI, extending the previous research on the English, Brazilian.
Limitations
The instrument was pretested on the general population rather than the target one, because of the unavailability of a bilingual sample. Second, the data collection was done from two clinical settings only and also from a tertiary care center, which causes a limitation in the generalizability of the test. Further, a big sample size could have provided better outcomes and wider generalizability.
Future Recommendations and Implications
Further analyses need to be conducted to examine the test–retest validity of the domains and validate comparisons with other published instruments. Further research on this Hindi T-ASI is needed to generate its use in other clinical and nonclinical settings. The steps of the validation process will also provide clear directions to other cross-cultural research in the area of validation of a research instrument or scale.
Conclusion
Hindi version of the T-ASI appears to be a valid instrument in terms of its relevance, clarity, simplicity, and purpose for measuring the severity of problems related to substance use among adolescents in Hindi-speaking population.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study has been supported by a grant from the All-India Institute of Medical Sciences (AIIMS), New Delhi as part of the intramural projects.
