Abstract
Background:
Cognitive impairment is the core outcome defining feature in schizophrenia. Schizophrenia, in the context of a broader neurodegenerative conceptualization, may have shared etiology with major neurocognitive disorders (MNCD). To elucidate this association there is definite need to explore the familial loading of dementia, in families of patients with schizophrenia.
Methods:
The authors compared relatives including parental generation and siblings of 100 cases (schizophrenia probands) and 100 controls (anxiety disorder) in order to assess the familial co-aggregation of MNCD. All cases and control were screened with Mini International Neuropsychiatric Interview screen for psychiatric morbidity. The pedigree analysis was conducted by family history method and Family Interview for Genetic Studies. Cognitive impairment in pedigree was screened by community screening instrument for dementia.
Results:
There was nonreporting of MNCD in the total 2538 relatives (proband siblings +parental generation) of both cases and controls. Diabetes mellitus was the most common somatic morbidity, found significantly more among the parental generation of cases than healthy controls (χ2 (1, 1713) = 6.452, P < 0.05). The odds of having various psychiatric and medical morbidities in the schizophrenia families compared to control are less than 1.
Conclusion:
There is no familial co-aggregation of MNCD in schizophrenia probands and common etiology between the two is less likely. Either schizophrenia could be counter-intuitively protective for MNCD or a reversible risk factor that can be prevented by effective treatments
100 families of schizophrenia probands (cases) were compared to 100 families of anxiety disorder (controls) for familial loading of dementia via pedigree analysis. There was no familial coaggregation of MNCD in schizophrenia probands and common aetiology between the two is less likely.Key Messages:
Dementia or major neurocognitive disorder (MNCD) is characterized by cognitive decline and impairment in activities of daily living. With a prevalence of around 5%–6% in persons older than 60 years, MNCD causes significant impairment and places a substantial global burden worldwide.1, 2 This prevalence is expected to double every 20 years, due to improving life expectancy and population demographics.2, 3
Schizophrenia is characterized by abnormalities of thought (content or from), perception (hallucinations), and communication. 2 It affects 1% of the population worldwide. Historically, schizophrenia was named “dementia praecox” by Kraepelin, as deficits in attention, memory, and visuospatial orientation were considered hallmarks of the disorder. Deficits in these domains often lead to substantial functional impairment and social disability and determine recovery. 4
Growing evidence predominantly supports a neurodevelopmental origin for schizophrenia (i.e., genetic vulnerabilities interact with prenatal or perinatal exposures) than neurodegenerative (i.e., progressive deterioration of the central nervous system).5, 6
Critically speaking, although schizophrenia may not fulfill a narrow definition of classical neurodegenerative disease (chronicity and neuroprogression, specific biochemical changes leading to distinct histopathological and clinical syndromes, Mendelian-like inheritance), its progression cannot be solely seen from the context of neurodevelopmental mechanisms. 6 Interestingly, in the context of a broader neurodegenerative conceptualization, schizophrenia probands have also been theorized to have accelerated aging. This is not only due to the association to age-related disorders, such as cardiovascular disease, diabetes mellitus, and cancer, but also because of shared risk factors between schizophrenia and age-related disorders (i.e., advanced paternal age, low birth weight, and specific genes).2, 7 Therefore, in order to elucidate the shared etiology between schizophrenia (as a risk factor) and neurodegenerative disorders like MNCD, several studies have attempted to study the association in case-control and prospective cohort designs. The available evidence is largely inconsistent.2, 8–10
The common basis of these two disorders could be better elucidated by assessing the clustering of the conditions in the families of each one. The association can be said to be true when the two conditions occur in a family with greater frequency and not due to chance factors alone. Several studies have addressed the familial coaggregation of the two disorders and have yielded incongruent results.11–15 Narayanaswamy et al. 16 reported significantly higher morbid risk for psychosis in early-onset dementia (EOAD) proband families and proposed a possibility of genetic overlap between dementia and psychosis. Thereby, in continuation of the proposal, there is a definite need to study the familial loading of dementia in families of patients with schizophrenia, to establish familial coaggregation.
The primary objective of our study was therefore to assess the familial loading of dementia in families of patients with schizophrenia. We studied patients with anxiety disorders as the control sample. An ancillary objective of the study was also to compare the familial loading of other general medical and psychiatric disorders between the two study groups.
Methods
This was a cross-sectional study conducted at a tertiary care mental health institute.
Subjects
Cases: Subjects (n = 100) aged <45 years meeting the diagnostic criteria of schizophrenia as per DSM-IV TR diagnosis (probands) were approached. The Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I) was used for diagnosis. The subjects were recruited from patients attending the inpatient and the outpatient services, in a consecutive manner, irrespective of the schizophrenia type, from January 2009 till May 2010. The investigator was blind to the positive or negative status of the family history of the subjects when each subject was recruited. Only those probands who were accompanied by reliable informants were considered eligible. Patients with mental retardation were excluded; no other comorbid psychiatric disorder was excluded.
Controls: Subjects (n = 100) with diagnostic criteria of anxiety disorder as per DSM-IV TR diagnosis were included as controls. SCID-I was used for diagnosis. Age- and gender-matched subjects were recruited from the inpatient and the outpatient services. Only those subjects who were accompanied by reliable informants were considered eligible. Patients with mental retardation were excluded; no other comorbid psychiatric disorder was excluded. Subjects with a history of psychosis in first-degree relatives were also excluded.
The sample size was determined based on the researcher’s convenience and available time.
Assessments
Schizophrenia probands and the control group were screened for comorbid psychiatric disorders with the Mini International Neuropsychiatric Interview [M.I.N.I.] screen). 17 Assessment of dementia was according to the SCID-I criteria for dementia. The patients’ sociodemographic and vascular risk factor profiles were obtained through sociodemographic and risk factor questionnaires. The reliable informants of probands with schizophrenia and the control group were then interviewed to obtain a pedigree that was mainly focused on the parental generation and the proband’s siblings. If any members of the pedigree screened positive for dementia, the Community Screening Instrument for Dementia (CSI-D)—the informant interview—was planned for confirming the loading of dementia in the family. 18 The Family Interview for Genetic Studies (FIGS) was applied on the members of the pedigree for the presence of major psychiatric illnesses in the family. 19
Pedigree Drawing
A pedigree was drawn as part of the initial ascertainment to determine whether a particular family is suitable for study. If the proband could not provide the information to draw the pedigree, another family member provided the data. If it was not drawn before the interview, it was done as part of the interview before beginning the FIGS. At a minimum, the pedigree included the proband and their parents, grandparents, siblings, aunts, uncles, offspring, and spouse. The pedigree indicates the presence of bilineality. When a proband’s relatives include affected individuals on both the mother’s and the father’s side, the family was said to be bilineal. Regarding screening for bilineality, a general rule was that in any generation, when there are ≥1 affected offspring, the parent relative’s spouse and the spouse’s first degree relatives should be screened by the FIGS. The pedigree may also indicate extenders. Extenders are any of the proband’s second-degree relatives (grandparent, uncle or aunt, half-sib, niece or nephew, or grandchild) who are affected. An extender expands the pedigree so far as the first-degree relatives of an extender are included in the pedigree for study. The interviewer enquired information regarding pathology in any of them.
The pedigree also indicated pointers. Pointers are relatives who are not affected but who have offspring who are affected. For example, if the proband’s aunt is not affected but has a daughter who is affected, the daughter’s sibship is studied and the aunt’s spouse and his family is checked for bilineality. The interviewer also enquired information regarding pathology in any of them.
The reliability of the pedigree charting was assessed by corroborating it between two available informants. When corroboration was not found, it was confirmed from a third informant.
Ethical Considerations
The study had the approval of the Institute Ethics Committee. Written informed consent was obtained from all the subjects after explaining fully about the procedures and only then they were enrolled into the study. Confidentiality of all the information obtained was maintained, and subjects had the right to withdraw the consent at any stage.
Statistical Analysis
The data were suitably coded and extracted on an SPSS 15.0 spreadsheet. The comparisons between cases and controls were done using the prevalence of psychiatric morbidity. χ2/Fisher’s exact test was used for all discontinuous variables and Student’s t-test for continuous variables. The morbid risk was estimated by computing the odds ratio (OR).
Results
Sociodemographic Characteristics
There were 100 probands of schizophrenia (cases) and 100 controls. The sex distribution among the cases and controls was 60 males and 40 females. The mean ages were 30.07 ± 6.81 years and 29.77 ± 6.38 years, respectively, for cases and controls. The urban/rural status revealed that 57% of cases came from rural areas, and in controls 64% belonged to rural areas. The information regarding the type of informants available and education level in the two groups is presented in Table 1. Fifty percent of the cases had two informants, and the remaining had one informant. For controls, 42% of the controls had two informants, and the remaining had one informant. Two informants were required when the information was primarily gathered from the participant’s spouse or if the information was considered less consistent.
Comparison of the Socio-Demographic Profile and Clinical Variables of Cases and Control Population
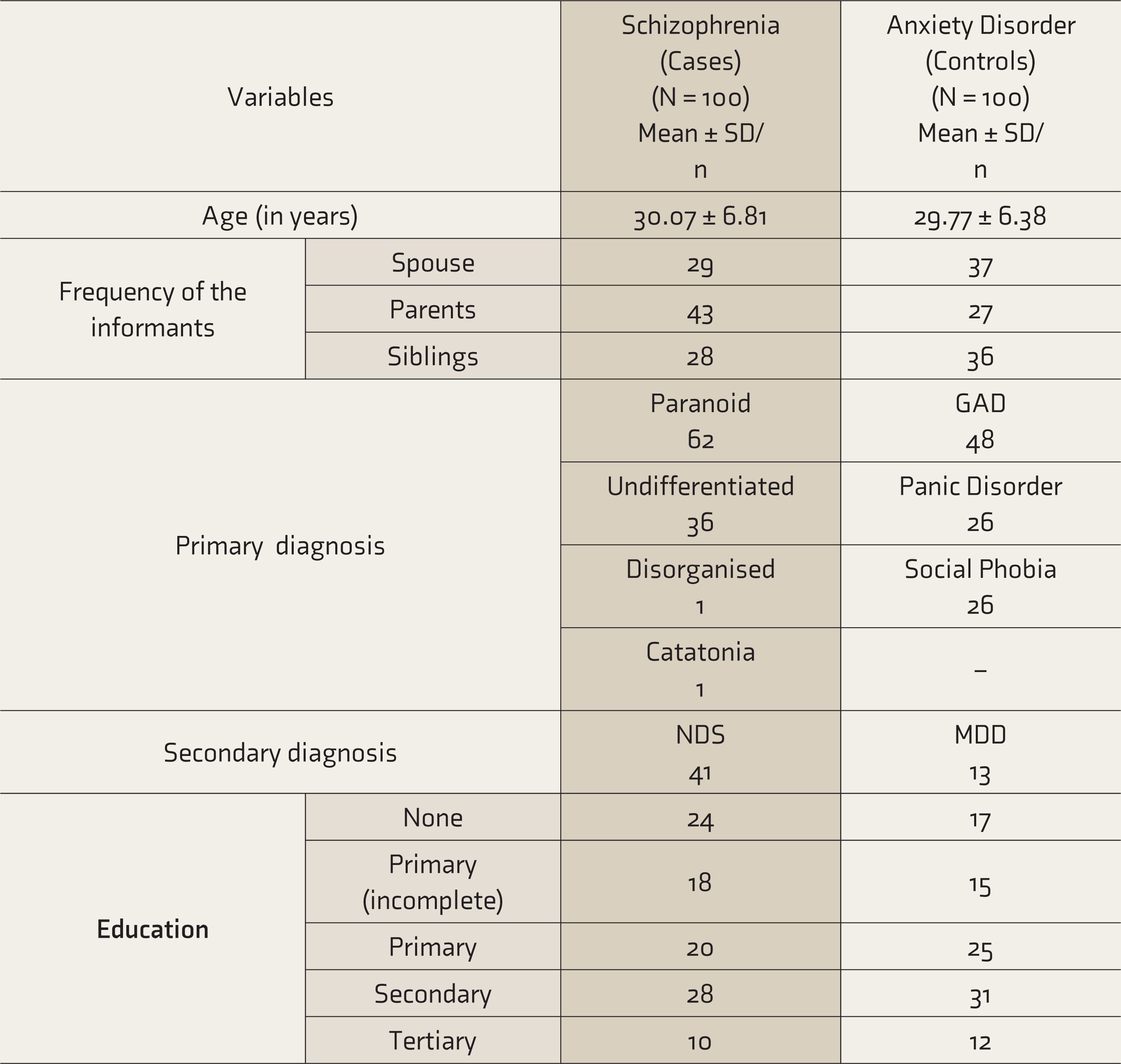
Schizophrenia Probands
The distribution of type of schizophrenia and the type of anxiety disorder in patients and lifetime diagnosis of a comorbid psychiatric illness in them is presented in Table 1.
Family Relatives
The number of proband siblings was 421 for cases and 404 for controls. The mean age (in years) of proband siblings of cases and controls was 26.67 (SD 7.86) and 27.11(SD 8.24), respectively. The number of proband parental generation relatives was 872 for cases and 841 for controls. The mean age of proband parental generation relatives was 51.33 (SD 10.45) among cases and 53.97 (SD 11.55) among controls. There were only 277 relatives above 65 years in the parental generation among the schizophrenia probands.
Comorbidity and Morbid Risk Assessment
Table S1 compares various disorders in the parental generation between cases and controls. Dementia was not found in the parental generation of cases or controls. Table S1 shows the comparison of general medical and psychiatric comorbidities found in parents during the pedigree assessment. The proportion of DM (only somatic morbidity) was significantly more in individuals of schizophrenia probands parental generation compared to that of controls (χ2 (1, 1713) = 6.45, P < 0.05).
Discussion
In continuation with the prior study conducted at our institute where the significantly higher morbid risk for psychosis in EOAD proband families was reported using survival analysis, 16 we proposed to study the familial loading of dementia in families of patients with schizophrenia to establish the familial coaggregation.
We did not find any cases of dementia either in parental generation among 1,713 subjects comprising both cases and controls. This is in convergence to a recent population-based study which reported that schizophrenia proband families did not have an increased risk of Alzheimer’s [incidence rate ratio (IRR), 0.97; 95% CI, 0.88–1.07], vascular, or unspecified dementia. 15 Rohde et al., 15 in turn, postulated that there might be no common etiological basis between the two disorders and that the associations detected between the two in previous studies might11–15 be false because of higher rates of somatic comorbidity (like DM) among these individuals. But despite the increased proportion of somatic morbidity (like DM in schizophrenia probands families) in our study, there was nonreporting of MNCD. Therefore, implying that schizophrenia could be either counterintuitively protective for MNCD or a reversible risk factor that effective treatments can prevent. One reason for this lack of family loading could be that the mean age of the parental generation of cases and controls was in the early 50s, which is below the age of risk for dementia: only < 1/3rd of relatives in the parental generation were older than 65 years. Another explanation could be the sensitivity of the scale (CSI-D) used for assessing dementia, as it is just a screening scale. The family history method employed by us is prone to an information (awareness and recall) bias, since not all psychiatric disorders will be observed and reported by family members. This is a general limitation of this method of family genetic study, and hence it applies to this study as well. 20 Bradford et al. 21 reviewed various factors responsible for missed or delayed diagnosis of MNCD. Some of these factors responsible for not detecting the familial coaggregation in our study could be rural background, lower level of awareness, possibly lesser severity of dementia, and false assumption that cognitive changes are part of normal aging. Moreover, a caregiver who has come to address the problem of severe mental disorder may not consider the cognitive impairment a priority for discussion with the physician. 22 Underreporting of cognitive impairment is also a possibility because the proportion of somatic morbidity in our sample was less than the current prevalence. 23 An important limitation of our study is that prevalence of EOAD is very low, 24 requiring a larger sample size to detect it (type-2 error).
Moreover, the fact that this study was cross-sectional and did not include longitudinal, follow-up assessments is an inherent limitation. This study being part of a time-bound academic thesis, the sample size was determined based on the researcher’s convenience and available time. This arbitrary nature of the sampling limits the generalizability of our findings. Also, prudently, studies should focus on family members in the pedigree who have crossed the age of risk for dementia. This number being very low in our sample is another limitation. In fact, siblings and the parental generation less than 65 years of age were specifically included for assessment to look for cases of EOAD, if any.
Despite these limitations, the study has many strengths. The interviewer was blind to the family history status of probands and controls, even though blinding was not feasible for the diagnosis status of probands and controls. This study used a semi-structured instrument—FIGS—to ascertain family history.
To some extent, the lack of differentiation in the outcome between the groups might be because a patient control group was chosen rather than a healthy control group. However, the reason for choosing patient controls in our study was that the cases stemmed from the population catered to at a tertiary care mental health institute and not from the general population. Therefore, we deem that our results are better generalizable to hospital-based populations.
Conclusion
We conclude that there is no familial coaggregation of MNCD in schizophrenia probands attending tertiary care mental health services, and common etiology between the two is less likely.
Supplemental Material
Supplemental material for this article is available online.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
