Abstract
Background and Objective:
United States Food and Drug Administration (USFDA) recently approved a novel combination of olanzapine-samidorphan (OLZSAM) for managing olanzapine-associated adverse events (weight gain) in adult patients with schizophrenia and bipolar disorder. To opine about the safety and efficacy of OLZSAM, authors performed a systematic review and meta-analysis to convene justifiable evidence.
Methods:
A thorough literature search was performed through the databases Embase, Cochrane Library, PubMed, and clinicaltrials.gov, from inception to September 2022, with the keywords: ‘olanzapine and samidorphan’ and schizophrenia; and “ALKS3831” and “lybalvi.” Clinical trials published in English that analyzed the efficacy and safety of OLZSAM were included. The significant outcomes included in this study were change from baseline (CFB) in Positive and Negative Syndrome Scale (PANSS) at the end of the study, the proportion of patients with weight gain at the end of the study, the proportion of patients with at least one adverse event, and the incidence of drug discontinuation due to adverse events.
Results:
The change in PANSS score at the end of the study was comparable among groups receiving OLZSAM and olanzapine alone: standardized mean difference (SMD) = 0.04; 95% CI = -0.09 to 0.17; p = 0.57. The OLZSAM group reported less incidence of weight gain: risk ratio (RR) = 0.91; 95% CI = 0.62–1.34; p = 0.63, and any adverse event: RR = 0.99; 95% CI = 0.90–1.09; p = 0.81. Drug discontinuation incidence was higher in the OLZSAM group: RR = 1.22; 95% CI = 0.84–1.79; p = 0.30.
Conclusions:
The combination OLZSAM showed comparable efficacy to olanzapine alone in schizophrenia patients, with relatively less incidence of weight gain and adverse events; however, the drug discontinuation due to adverse events was more in the OLZSAM group.
Schizophrenia, which is considered one of the top 10 causes of disability globally, is characterized by disrupted mental processes.1,2 Intriguingly, this disorder, with a lifetime prevalence of 1%, is associated with a massive economic burden and comparatively shorter life expectancy.1-3 For the disorder having onset in late adolescence or early adulthood, several antipsychotic drugs are available. 4 These include D2 receptor antagonists (e.g., chlorpromazine, triflupromazine, prochlorperazine, and haloperidol), 5HT2A blockers with D2 antagonism (e.g., clozapine, olanzapine, quetiapine, and risperidone), and partial D2 agonists (e.g., aripiprazole and brexpiprazole). 5 Olanzapine (OLZ), a second-generation antipsychotic with preeminent action on the 5HT2A receptor, is one of the most commonly prescribed drugs for schizophrenia. It is efficacious in resolving both positive and negative symptoms.6,7 Its added advantage is that it normalizes the affected cerebral regions (related to cognitive functions and emotional processing) and provides a longer time-to-treatment discontinuation, thus contributing to better patient adherence, especially to chronically ill patients.7,8 However, there are significant drawbacks concerning OLZ’s long-term use, such as weight gain, dyslipidemia, cardio-metabolic syndrome, and diabetes mellitus.9-11 To reduce its adverse effects, a novel opioid receptor modulator, samidorphan (SAM), was introduced to be administered in combination with OLZ. Structurally related to naltrexone, SAM is also an antagonist at the mu-opioid receptor and a partial agonist at the kappa and delta opioid receptors.12-14 Attributable to SAM’s mechanism (mu opioid receptor antagonism, thus inhibition of mesolimbic reward system), it was observed that the use of olanzapine-samidorphan combination (OLZSAM) in the management of schizophrenia or bipolar disorder led to reduced weight gain and decreased possibility of worsening of cardiometabolic risk factors.10,12,15,16 The United States Food and Drug Administration (USFDA) approved the said combination in May 2021 for treating schizophrenia and bipolar disorder I in adults. 17 Of the few systematic reviews and meta-analyses, we could identify that compared OLZSAM with OLZ, all reported on the cardiometabolic profile and weight gain mitigation effects.9,18-20 An evidence-based review scrutinized eight pivotal clinical trials, including open-label trials, randomized controlled trials (RCTs), and ongoing phase III clinical trials, to determine the efficacy and safety (ECG parameters, movement disorders, suicides, and adverse events) of the new combination among psychiatric patients. 9 Srisurapanont et al., in their analysis, included four short-term clinical trials (≤24 weeks) with a primary focus on changes in weight as well as a comparison of changes in cardiometabolic profile and dropout rates. 18 Laguado et al. evaluated those clinical trials, wherein OLZ was administered with an opioid antagonist (for allaying weight gain by OLZ). Of the six clinical studies they reviewed, only five had a head-on comparison of OLZSAM with OLZ, and the results of their review were again centered around changes in the weight or body mass index of the participants. 19 Jawad et al., in their systematic review, included eight clinical studies, both RCTs and open-label trials, with the primary intent to investigate the effect of the combination on weight gain and metabolic parameters. 20 Against this background, we conducted a systematic review and meta-analysis to recognize how efficacious (with Positive and Negative Syndrome Scale [PANSS]) and safe (in terms of both common adverse events and dropouts due to adverse events) OLZSAM compared to OLZ by critically analyzing only the RCTs comparing these drugs head-on.
Methods
The authors performed the systematic review and meta-analysis as per the guidelines of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 21
Search Strategy
A thorough systematic search was conducted of databases, such as, Embase, PubMed, Clinical Trial Registry (
Study Selection Process.
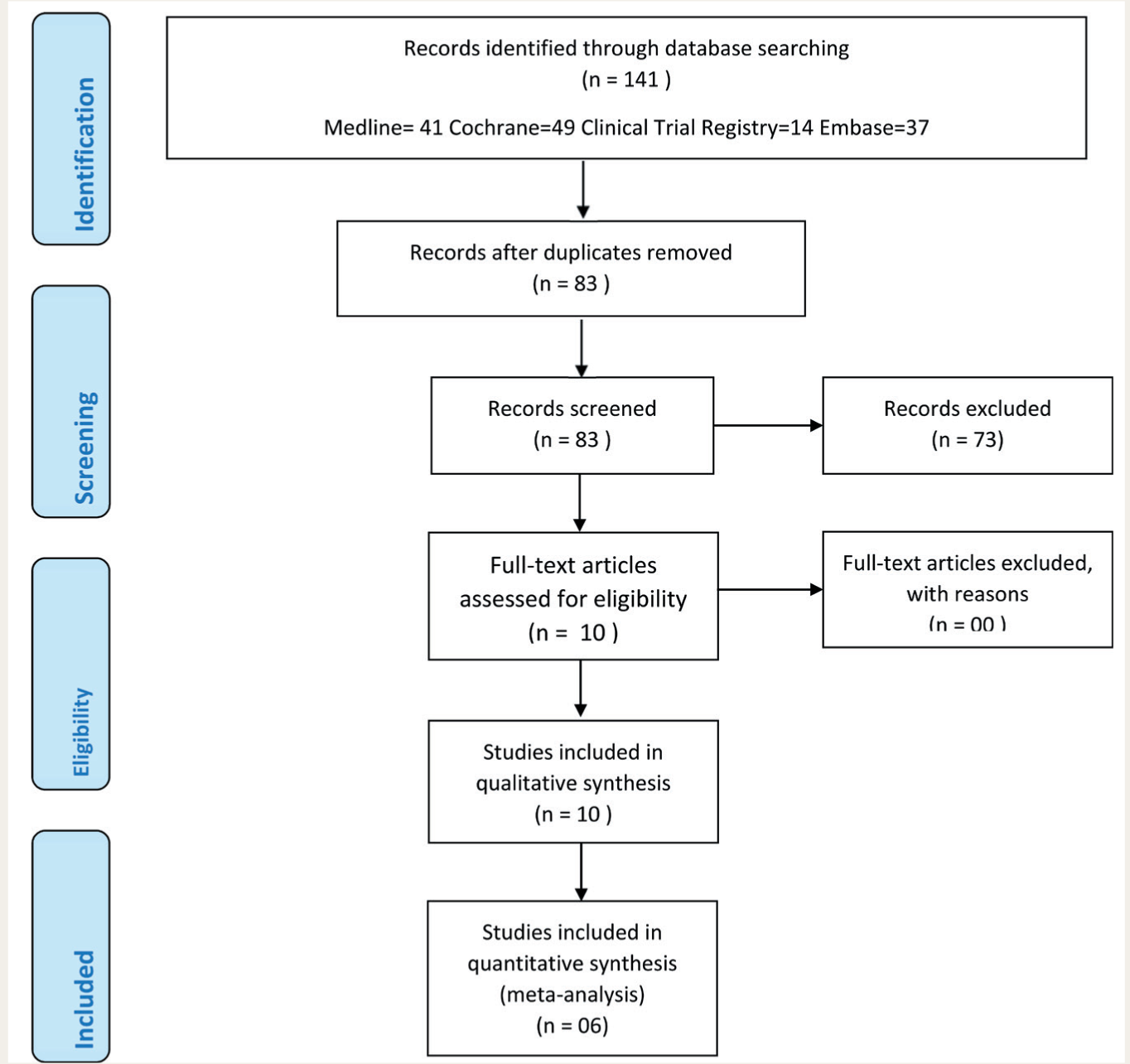
Study Selection
RCTs that recruited patients with a psychiatric disorder (such as schizophrenia) or healthy volunteers of age >18 years and compared OLZSAM with OLZ in either phase of clinical trials were included for analysis. If the studies were available as conference paper abstracts or posters, or had insufficient trial details, or were reviews (narrative and systematic) on OLZSAM, then they were not included.
Data Extraction
Both authors performed the data extraction using Microsoft Excel 2016. Extracted data included demographic information, inclusion and exclusion criteria, treatment schedule, study design, and all outcomes. Any missing information was obtained from the clinical trial registration site’s protocol and statistical analysis plan. Subsequently, all the relevant data were analyzed using Review Manager 5.4 (RevMan v5.4) for Windows. The risk of bias for the individual study was assessed using both Risk of Bias (RoB) and Risk of Bias-2 (RoB2) assessment tools. The older RoB tool was utilized for preparing a summary of findings (SoF). However, the risk of bias is presented in results using the RoB2 tool.22,23 The biases assessed for each study included selection bias, performance bias, detection bias, attrition bias, and reporting bias, as per the older RoB tool. The RoB2 tool was used to evaluate numerous other biases, such as bias arising from the randomization process, bias due to deviations from intended interventions, bias due to missing outcome data, bias in the measurement of the outcome, and bias in the selection of the reported result. Publication bias was inspected using a funnel plot for each pre-determined outcome. The strength of evidence was judged with the GRADE approach considering the risk of bias, inconsistency, indirectness, imprecision, and publication bias. 24
Outcomes
The efficacy and safety endpoints included were as under:
Change from baseline (CFB) in PANSS at the end of study (EOS). The proportion of patients with weight gain at EOS. Incidence of somnolence at EOS. Incidence of dry mouth at EOS. Incidence of headache at EOS. Incidence of Any Adverse Event at EOS. Incidence of Serious Adverse Events (SAEs) at EOS. Incidence of Drug Discontinuation due to Adverse Events at EOS.
Subgroup Analysis
We further analyzed these endpoints across two participant settings, that is, among patients and healthy volunteers.
Statistical Analysis
The standardized mean differences (SMDs) and relative risks (RRs), with their 95% confidence intervals (CIs), were used for continuous and dichotomous data, respectively. The true heterogeneity among the included studies was assessed with I2 statistics. The data were considered heterogenic if the I2 was >50%. 25 The authors used a random effects model to conduct this meta-analysis.
Results
Baseline Characteristics
In the present review, 1,655 patients were included from six RCTs.26-31 The leading reason for excluding the studies was that they were either available as conference paper abstracts or posters, with insufficient trial details, or were reviews (narrative and systematic) on OLZSAM.
The study selection process has been illustrated in Figure 1. The baseline data from the included studies have been enumerated in Table 1. Overall, 72.9% of patients were male, and 53.5% belonged to the black race. All except for one trial had established clinical diagnosis of schizophrenia as per the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5) criteria. 32 In the exception one, the diagnosis was based on DSM-IV criteria. 30 Among the six selected trials, four had recruited patients of schizophrenia, while the remaining two had enrolled healthy volunteers. The duration of these trials ranged from 3 to 60 weeks. The risk of bias in different domains for the individual trials has been presented in Table 2. Two of these trials were found to be at high risk as per the RoB2 tool.28,31 Toledo et al. had a high risk due to bias arising from improper randomization process and inadequate addressing of missing data; on the other hand, in Silverman et al., the randomization process could have been more precise.
Baseline Characteristics of Included Studies.
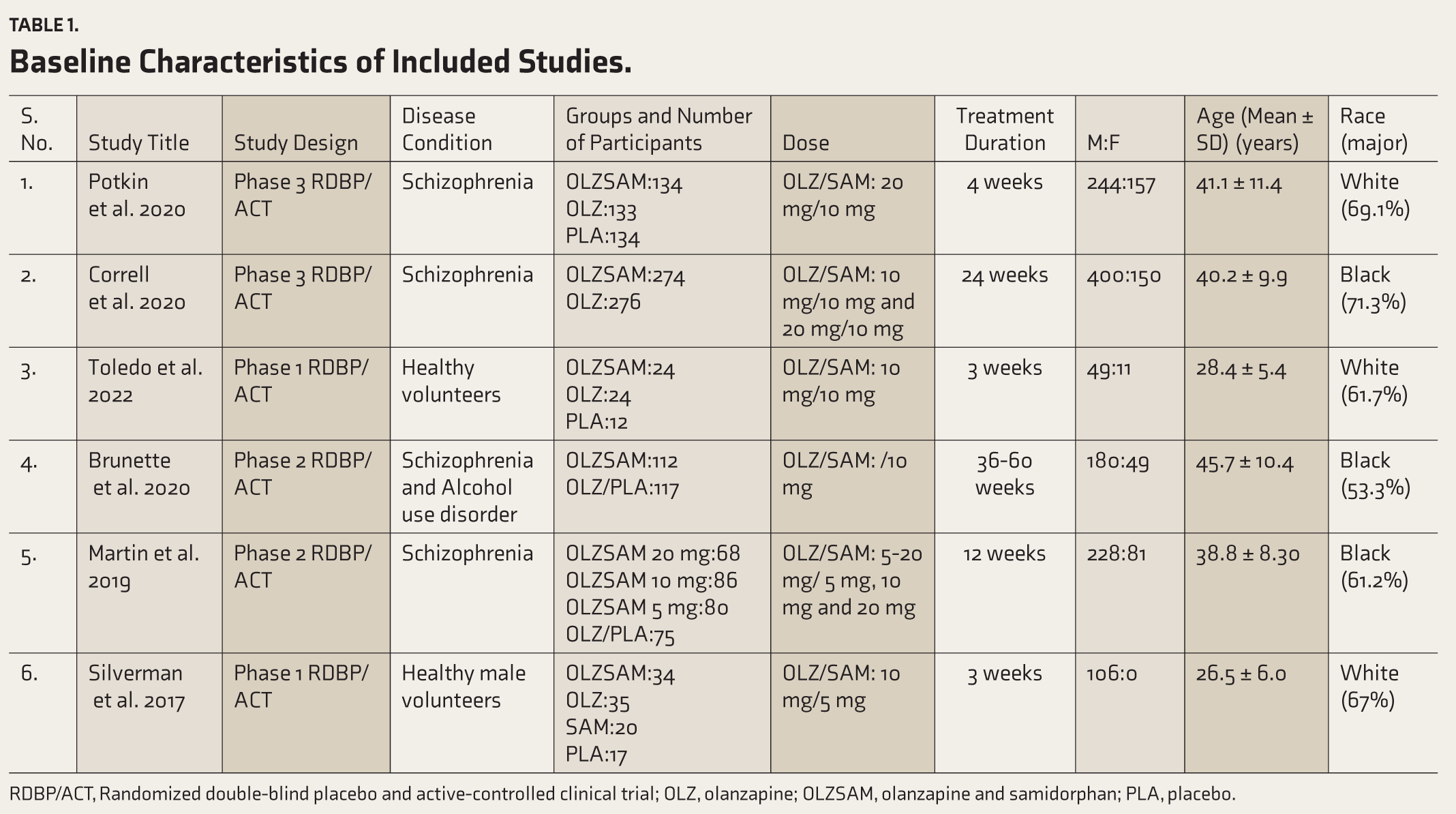
RDBP/ACT, Randomized double-blind placebo and active-controlled clinical trial; OLZ, olanzapine; OLZSAM, olanzapine and samidorphan; PLA, placebo.
Risk of Bias Assessment as per RoB2 Tool.
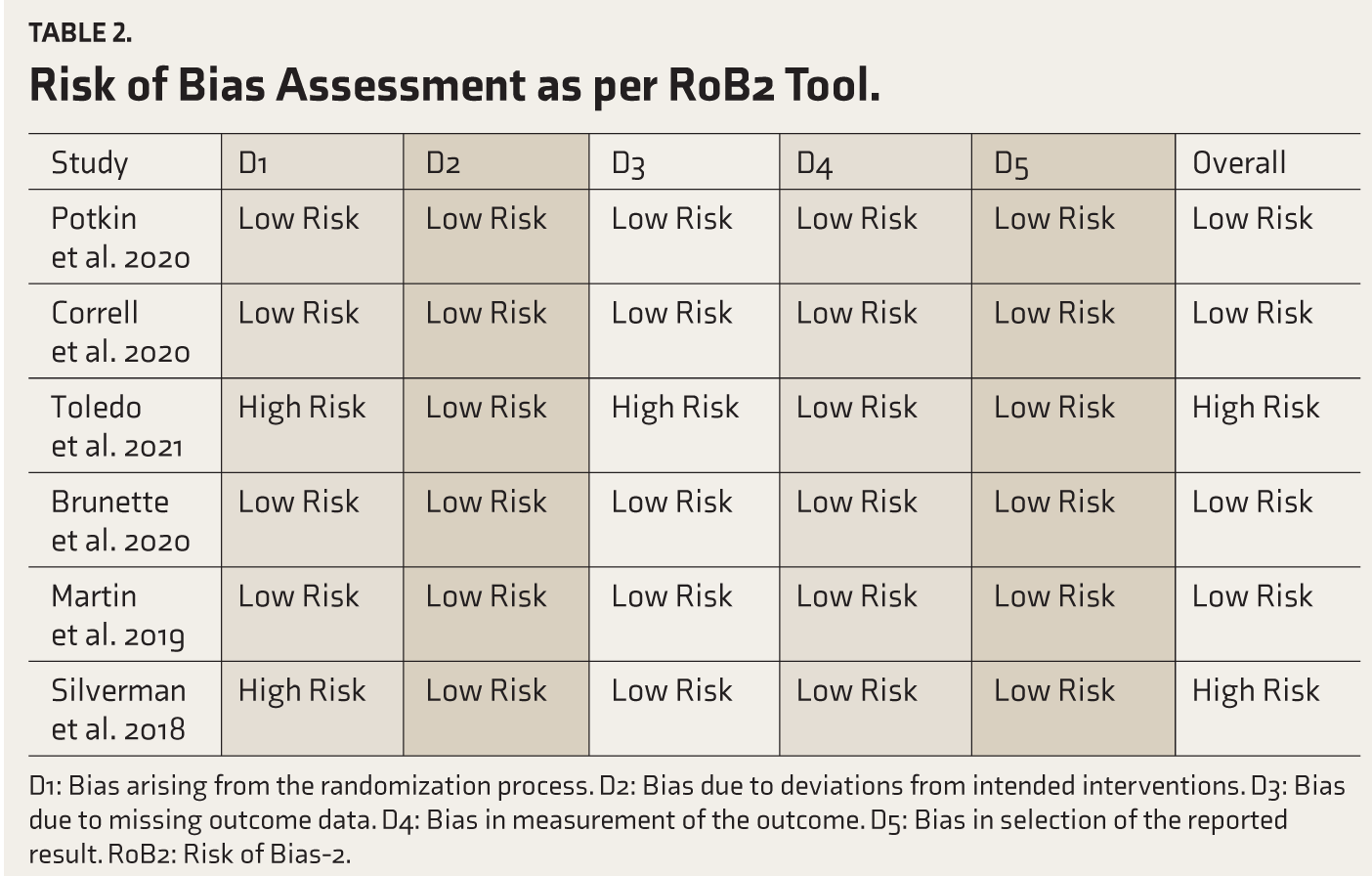
D1: Bias arising from the randomization process. D2: Bias due to deviations from intended interventions. D3: Bias due to missing outcome data. D4: Bias in measurement of the outcome. D5: Bias in selection of the reported result. RoB2: Risk of Bias-2.
Efficacy Endpoint
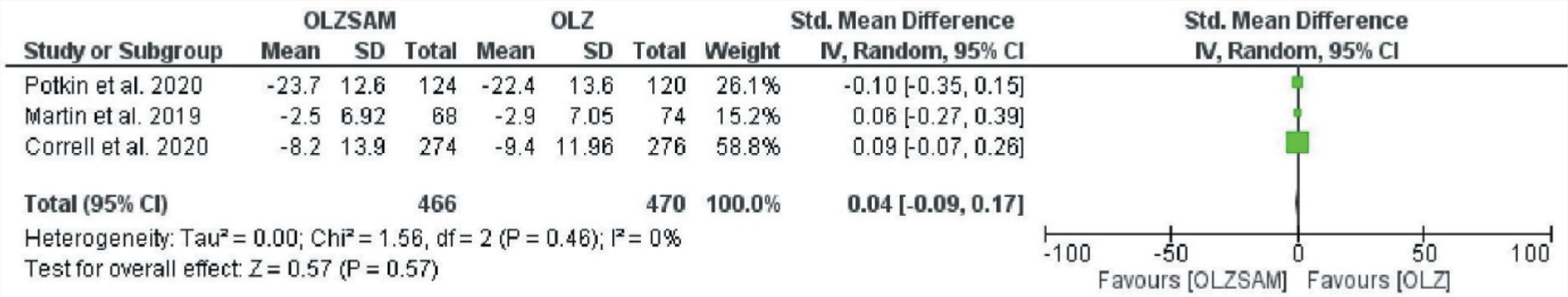
The funnel plot obtained for the efficacy outcome, that is, CFB in PANSS at the EOS, was symmetrical; hence, no publication bias was observed. Further, there was no heterogeneity observed for this outcome (I2 = 0%, p = 0.46) (Figure 2). The SMD for change in PANSS was 0.04 (CI -0.09 to 0.17; p = 0.57) (Figure 2). Therefore, both the groups, that is, OLZSAM and OLZ, were comparable regarding efficacy.
Forest Plot for Change in PANSS.
Safety Endpoints
Funnel plots for individual outcomes have been presented as supplementary files. Among the safety endpoints, the incidence of weight gain was higher in the OLZ than OLZSAM group (23.6% vs. 19.6%) but not statistically significant; RR = 0.91; 95% CI = 0.62–1.34; p = 0.63 (Figure 3). The funnel plot was symmetrical, and heterogeneity was observed, again not statistically significant (I2 = 49%, p = 0.12). The incidence of somnolence was more in OLZSAM group (18.2% vs. 15.4%); RR = 1.18; 95% CI = 0.88–1.57; p = 0.27 (Figure 4). Though the funnel plot was symmetrical, no heterogeneity was observed (I2 = 0%, p = 0.46). The incidence of dry mouth was also more in the OLZSAM group, and the difference was statistically significant (12% vs. 6.3%) RR = 1.73; 95% CI = 1.14–2.64; p = 0.01 (Figure 5). The asymmetrical funnel plot suggested publication bias without heterogeneity (I2 = 2%, p = 0.40).
Forest Plot for Weight Gain.
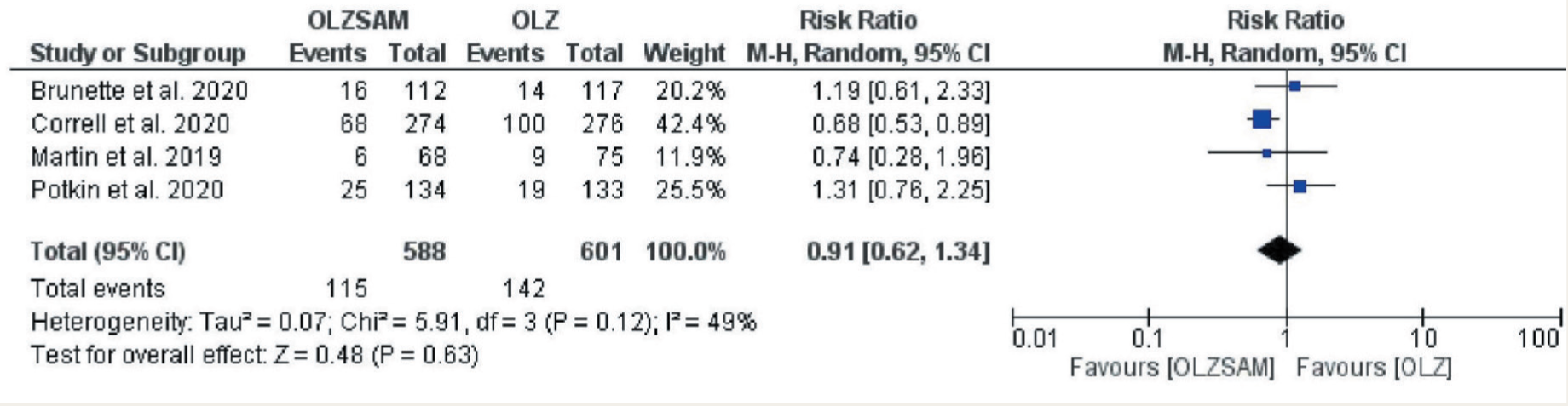
Forest Plot for Somnolence.
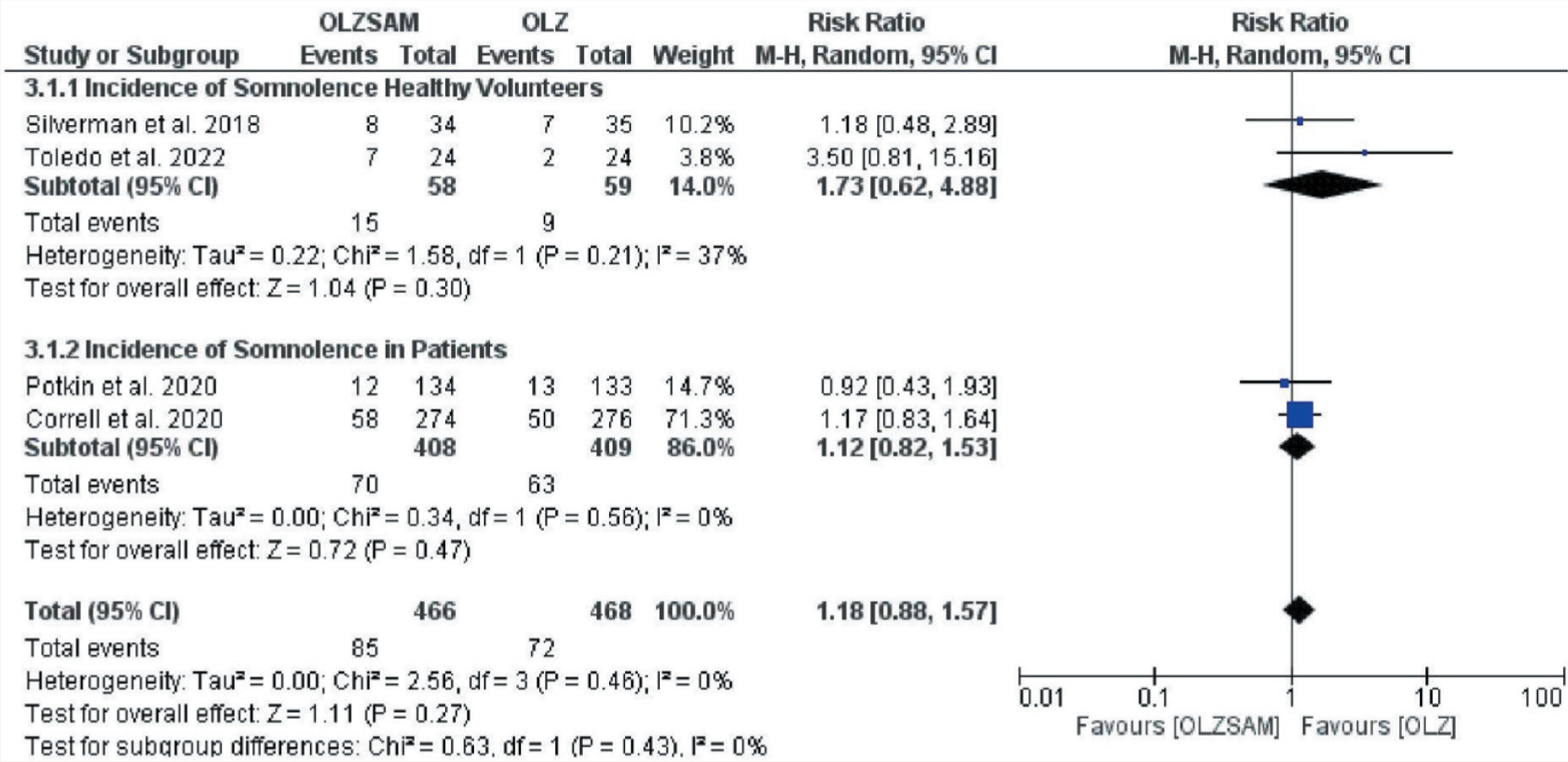
Forest Plot for Dry Mouth.
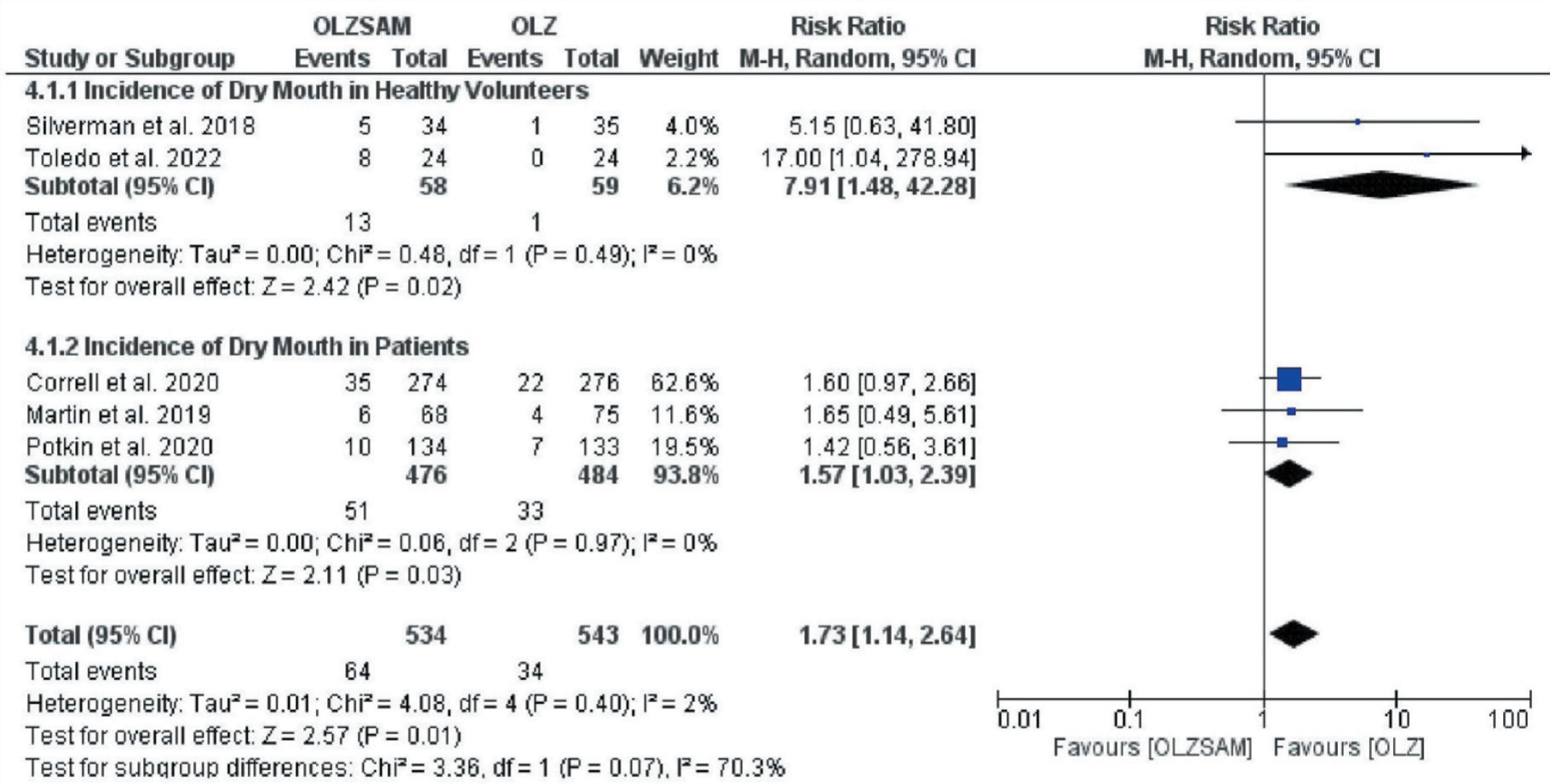
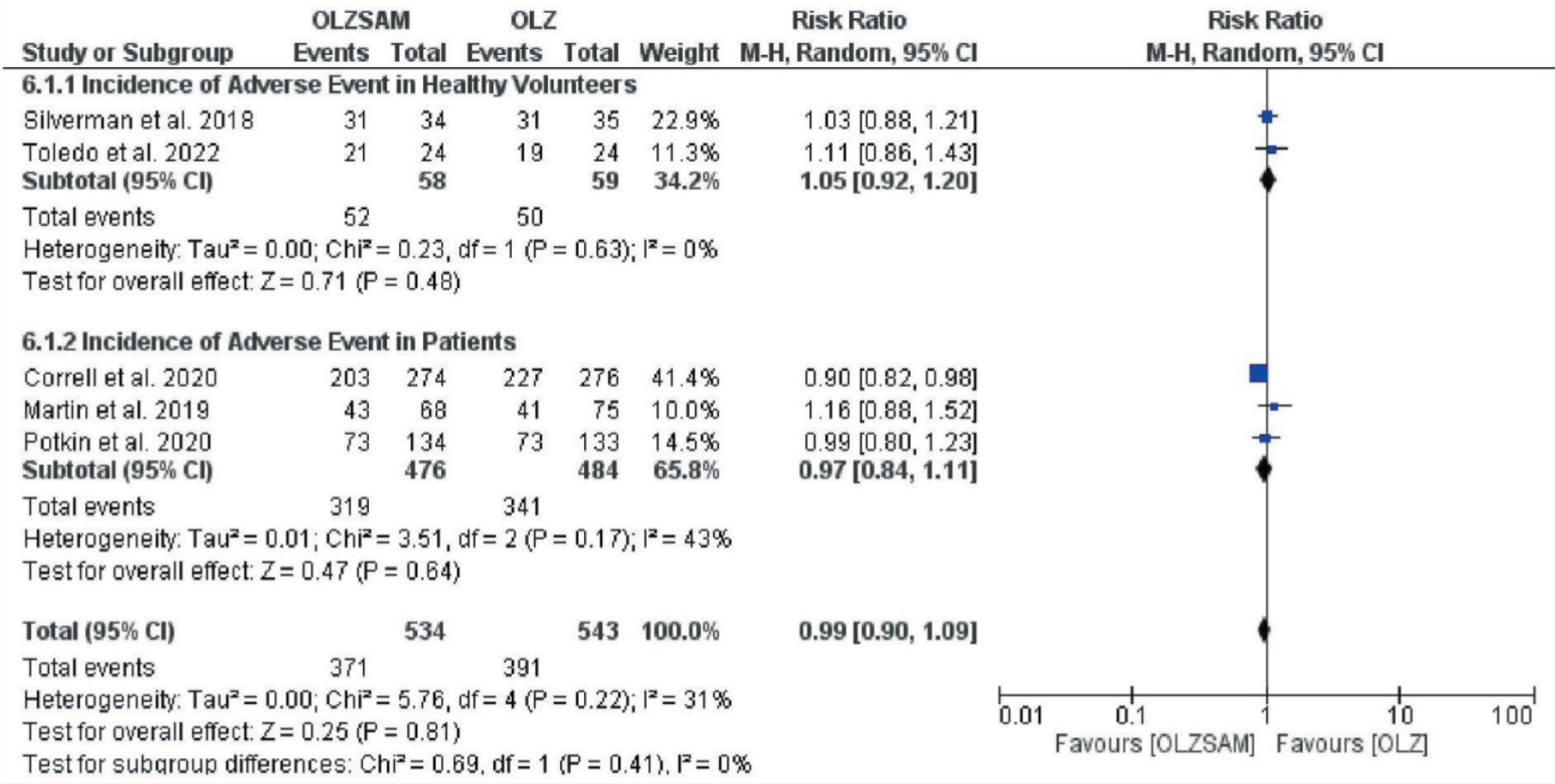
The incidence of headache was higher in the OLZSAM group (6.5% vs. 4.9%) but the difference was statistically insignificant; RR = 1.39; 95% CI = 0.50–3.88; p = 0.53 (Figure 6). The funnel plot attained was asymmetrical, indicative of publication bias, and statistically insignificant heterogeneity (I2 = 29%, p = 0.24). The incidence of experiencing any adverse event exceeded in the OLZ group (72% vs. 69.5%) but was not statistically significant; RR = 0.99; 95% CI = 0.90–1.09; p = 0.81 (Figure 7). The funnel plot was symmetrical, and we could observe heterogeneity, which was not statistically significant (I2 = 31%, p = 0.22).
Forest Plot for Headache.
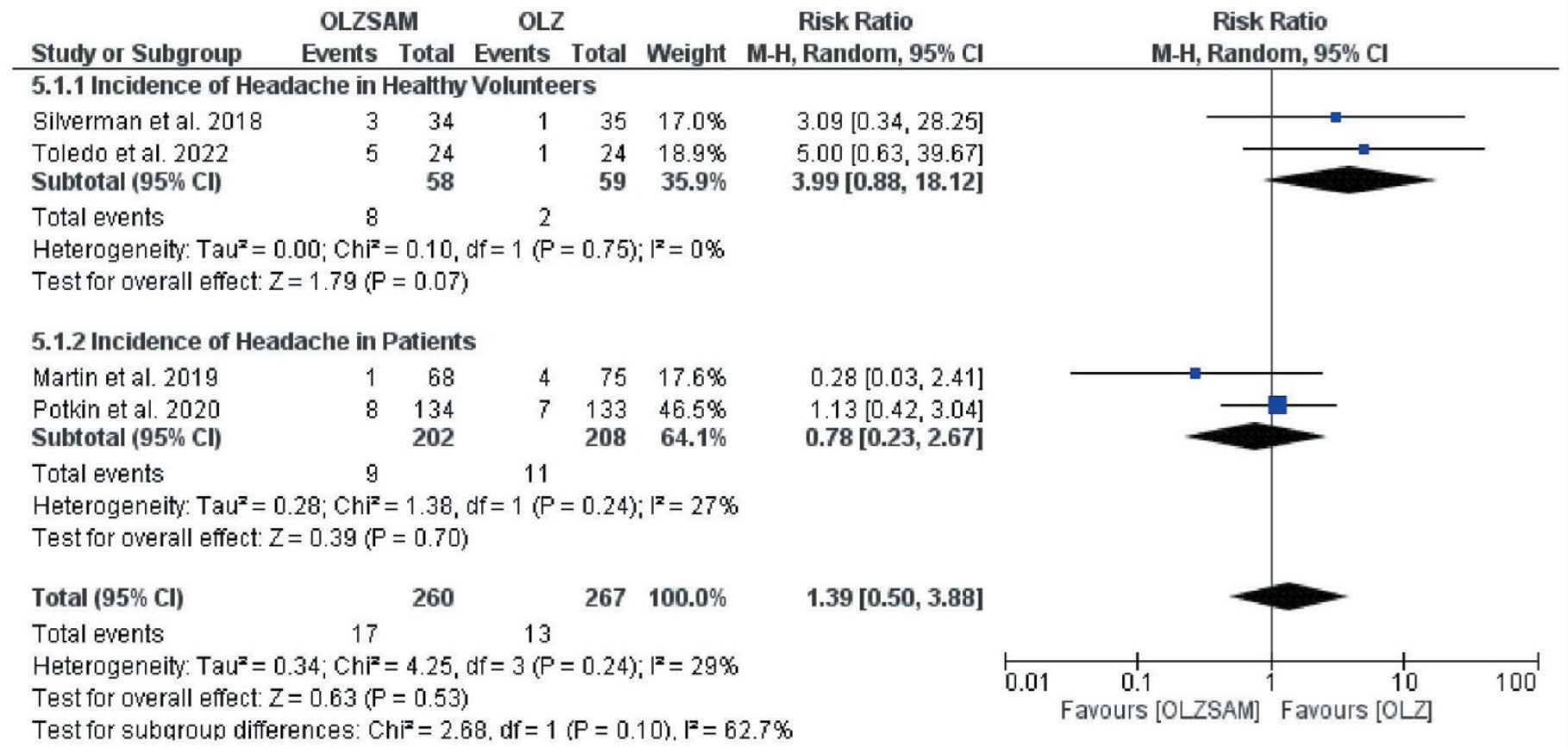
Forest Plot for any Adverse Event.
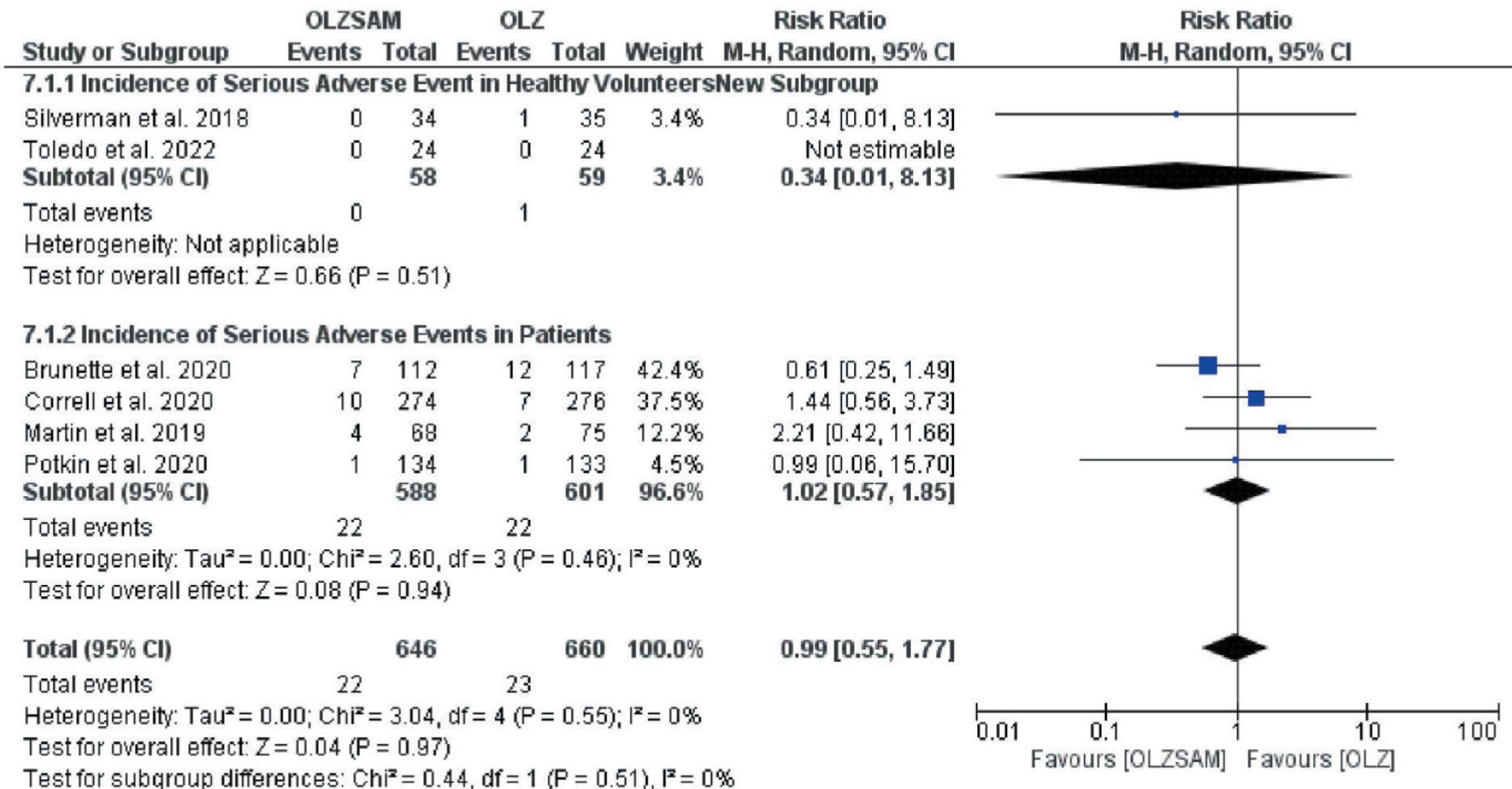
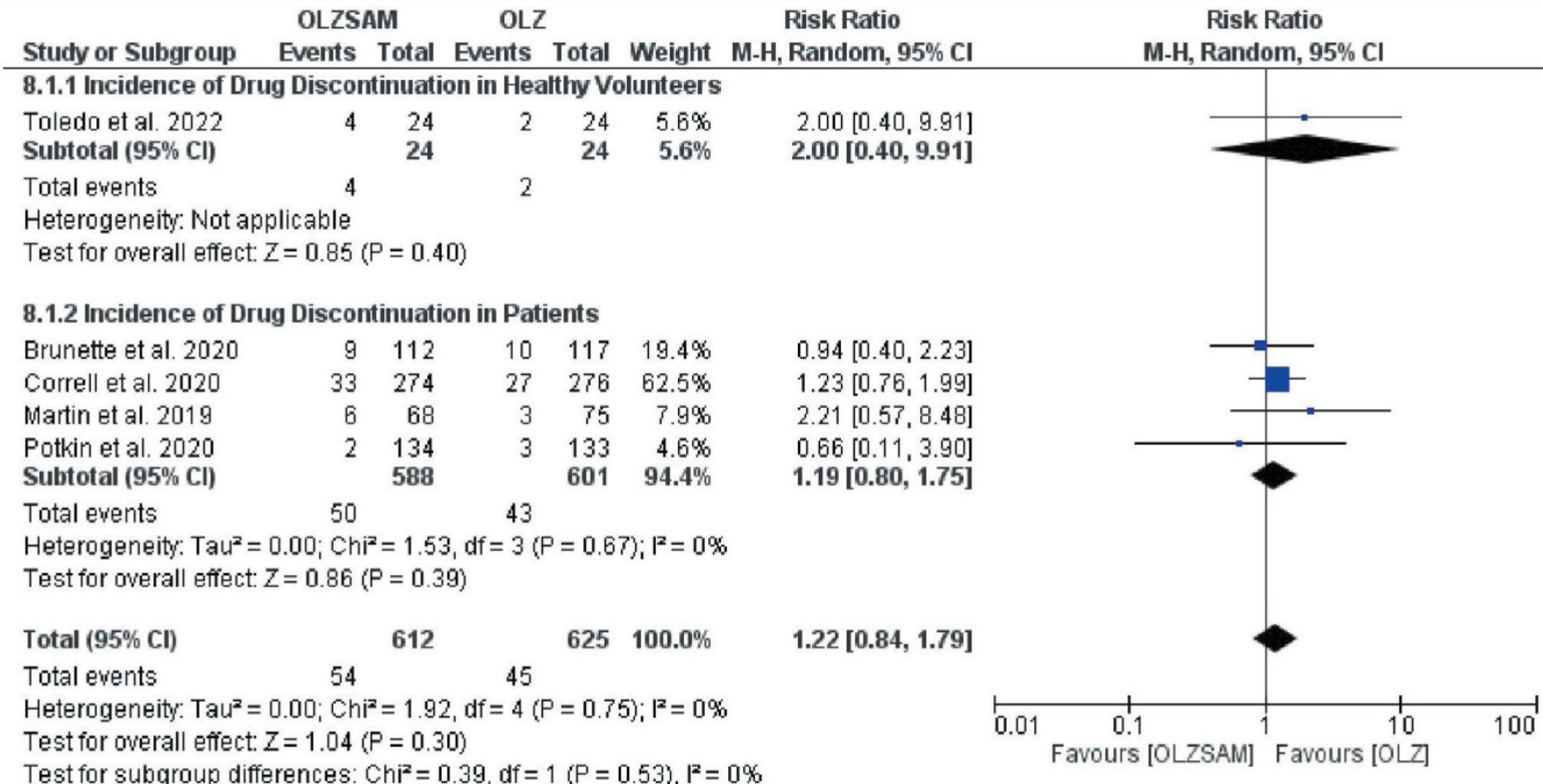
The incidence of encountering SAEs was again higher in the OLZ group (3.5% vs. 3.4%) even though it was not statistically significant; RR = 0.99; 95% CI = 0.55–1.77; p = 0.97 (Figure 8). The funnel plot captured was symmetrical without significant heterogeneity (I2 = 0%, p = 0.55). Contrarily, the incidence of drug discontinuation due to adverse effects was higher in the OLZSAM group (8.8% vs. 7.2%), but it was not statistically significant; RR = 1.22; 95% CI = 0.84–1.79; p = 0.30 (Figure 9). The funnel plot was symmetrical and exhibited no heterogeneity (I2 = 0%, p = 0.75).
Forest Plot for Serious Adverse Event.
Forest Plot for Drug Discontinuation.
The overall risk of bias was significant for two outcomes (incidence of headache and adverse events). No indirectness was observed in any of the outcomes. Inconsistency was observed with the incidence of weight gain, headache, and adverse events. Imprecision was noted in almost all endpoints. Overall certainty of the evidence for numerous outcomes ranged from very low to high. Detailed estimates with the certainty of the evidence for all the endpoints have been presented as a table of SoF (Table 3).
Summary of Findings.
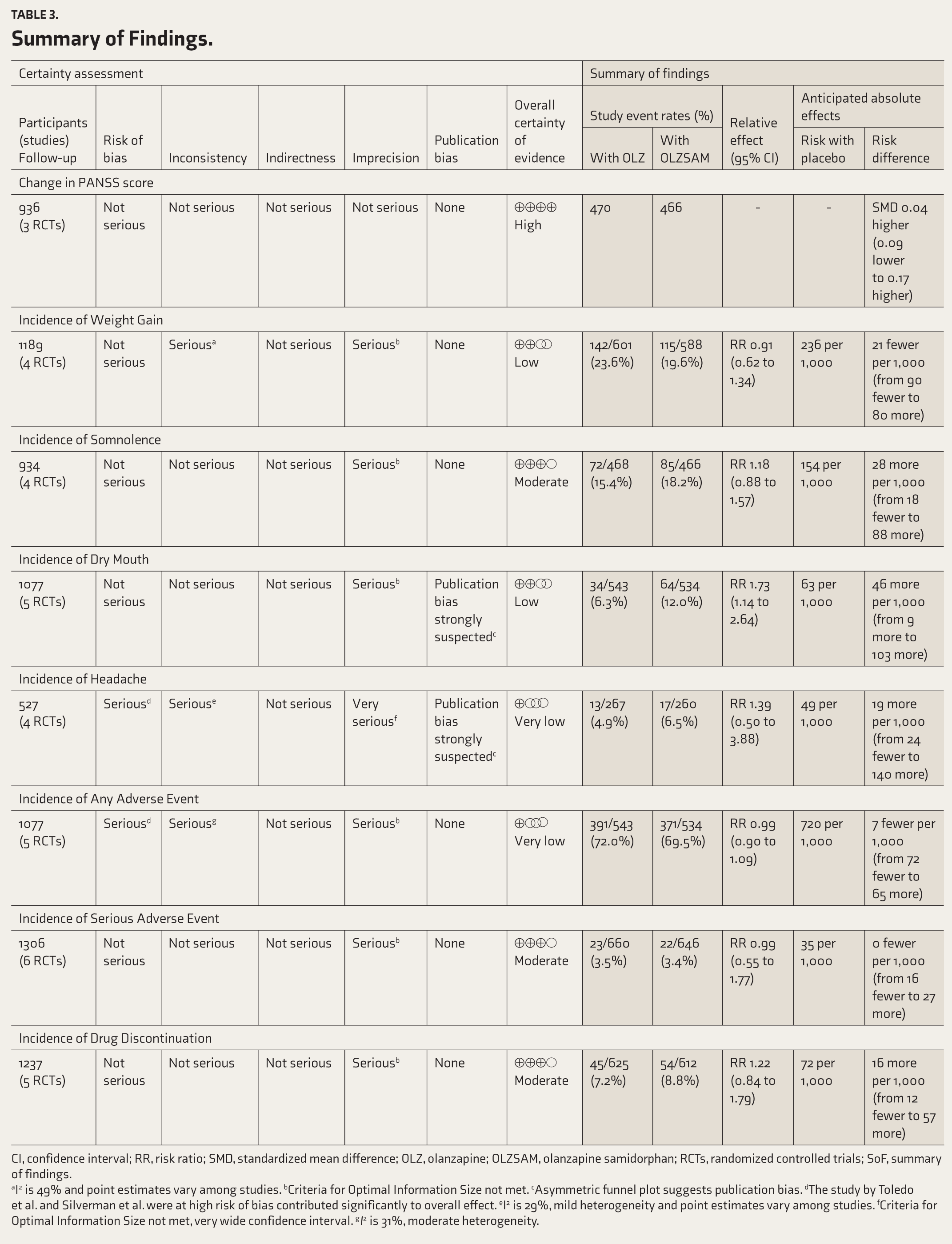
CI, confidence interval; RR, risk ratio; SMD, standardized mean difference; OLZ, olanzapine; OLZSAM, olanzapine samidorphan; RCTs, randomized controlled trials; SoF, summary of findings.
aI2 is 49% and point estimates vary among studies. bCriteria for Optimal Information Size not met. cAsymmetric funnel plot suggests publication bias. dThe study by Toledo et al. and Silverman et al. were at high risk of bias contributed significantly to overall effect. eI2 is 29%, mild heterogeneity and point estimates vary among studies. fCriteria for Optimal Information Size not met, very wide confidence interval. gI2 is 31%, moderate heterogeneity.
Discussion
The current systematic review and meta-analysis were conducted with the intent to comprehend what substantial difference this new combination, OLZSAM, would bring about in resolving the symptoms of schizophrenia and whether it has an upper edge over OLZ in terms of producing fewer adverse events (especially weight gain). The outcomes are based on well-reported clinical trials published or available until September 2022.
The usual pattern of the demographic distribution of schizophrenia could be observed in the studies included in this analysis, with males being more commonly affected than females (even though two trials had included only healthy volunteers, especially one having exclusively male healthy volunteers) and black people being more afflicted.28,31,33-35 The mean age of recruited patients in the studies was ~40 years; however, as no information about the age at which schizophrenia was first diagnosed was available, it is dubious to comment whether the schizophrenia indeed developed in preadolescence or post-adolescence age. 36
Even though we wished to include trials of high quality, two of them (Toledo et al. and Silverman et al.) had a high risk of bias, thus affecting the results.28,31 On the whole, the certainty of evidence generated in this meta-analysis for the selected endpoints was of moderate to high quality, except for the incidence of weight gain, headache, dry mouth, and any adverse event, wherein the certainty ranged from low to very low (Tables 2 and 3), which again can be attributed to these two trials having a high risk of bias. Furthermore, the overall certainty was reduced as the sample size required to achieve optimal information was inadequate in these trials. Similarly, inconsistency and publication bias also contributed to the decreased certainty of evidence.
PANSS is a well-established gold-standard scale for assessing the symptoms of schizophrenia, as it takes into account both the positive and negative symptoms as well as general psychopathology. 37 Presumably, compared to the placebo, the combination OLZSAM does have the upper hand in reducing the PANSS score significantly. 38 Nevertheless, compared to OLZ, the change in PANSS score at the end of the study was comparable, with no statistically significant difference in their efficacy. This is similar to the finding proposed by Jawad et al., especially in acute psychosis. At the same time, according to Rehan et al., the combination can be helpful in the long-term treatment as it demonstrated a significant decline in PANSS score.9,20 The plausible reason behind their equivalent efficacy could be that olanzapine alone demonstrates an antipsychotic effect (via antagonism at D2 and 5HT2A receptors), while the effect of the addition of samidorphan (which acts on opioid receptors) is to blunt the weight gain associated with the use of olanzapine. This effect culminates from the antagonism of the µ-opioid receptor, inhibiting the mesolimbic reward system responsible for inducing food cravings.15,39
As quoted, the rationale for combining samidorphan with olanzapine was to counter the weight gain induced by the latter. Our analysis substantiated the same (incidence of weight gain was higher in the OLZ group than in OLZSAM, though statistically insignificant). Nevertheless, this fact should be looked at carefully owing to the low certainty of the evidence, that is, OLZSAM may not have a significant benefit of less weight gain. Srisurapanont et al. observed an insignificant change in weight between the group of patients who received OLZ and OLZSAM. 18 In contrast, Laguado et al. reported significantly lesser weight gain with OLZSAM as compared to OLZ (1.5-3.2 kg vs. 2.4-5.1 kg) and Jawad et al., in their systematic review, concluded that in contrast to OLZ, the combination aided in curtailing the rate of weight gain as well as facilitated in achieving a stable weight. 20 Since these previous studies reported weight gain as continuous data, we instead planned to fixate on the incidence of weight gain or the proportion of recruited individuals who experienced weight gain with these drugs.
While assessing the selected studies, we also identified a few commonly reported adverse events apart from the incidence of adverse events and SAEs. The trials have documented the good tolerability of OLZSAM and indicated that the incidence of adverse events with either OLZSAM or OLZ is comparable.26-28,30,31 Haddad et al. also had documented that OLZSAM exhibited fewer adverse effects than OLZ, and the same has been substantiated by Jawad et al., as they observed that in short-term studies, the addition of the µ-opioid receptor antagonist did not alter the safety profile of the OLZ.15,20 Interestingly, we observed that the incidences of various adverse events were either high in the OLZ group or the OLZSAM group. In the former group, the incidence of any adverse event and any SAEs was high, though statistically insignificant, while in the latter group, the incidence of somnolence, dry mouth, headache, and drug discontinuation due to adverse events was high, and a statistically significant difference was observed for the incidence of dry mouth (p = 0.01).
The possible justification for these findings is that two of the included studies28,31 had recruited a small number of healthy individuals. In contrast, other studies recruited patients with schizophrenia, contributing to heterogeneity. The incidence of somnolence, dry mouth, and drug discontinuation, which was more in the OLZSAM group patient sample, contributed to the majority of the overall effect, with a short confidence interval (Figures 4, 5, and 9). In contrast, two studies involving healthy volunteers28,31 contributed significantly less to the overall effect with a very wide confidence interval (Figures 4, 5, and 9). Hence, somnolence, dry mouth, and drug discontinuation are vital concerns.
The incidence of headache was also more in the OLZSAM group (Figure 6). The studies with healthy volunteers28,31 contributed to around one-third of the overall effect, contributing to heterogeneity. In the case of the incidence of headaches, these two studies brought about a one-sided shift in the entire result (as evidenced in their respective risk ratio (RR), which is in sharp contrast to studies with patients), thus indicating inordinate misrepresenting and over-reporting of these adverse events by healthy volunteers.
The incidence of any and SAEs was slightly higher in the OLZ group (Figure 7 and 8). The studies of healthy volunteers contributed to one-third of the overall effect for any adverse event incidence; despite heterogeneity, the RR in healthy and patient samples was similar, with a short confidence interval. Hence, each group contributed equally to this parameter, and the tolerability was comparable in healthy volunteers and patients. In contrast, the entire outcome is attributed to the patient sample due to sparse reporting of SAEs in healthy volunteers (Figure 8). Considering these details, the comparable tolerability of OLZSAM is debatable in healthy and patient populations.
Although we tried to include trials of high quality in our analysis, a few limitations need to be addressed. Firstly, we did not quantify publication bias. Nevertheless, we have incorporated funnel plots for all the endpoints. Also, in previous reviews, the comparison of weight gain has been quantified as continuous data. To avoid the reiteration, we have evaluated it as dichotomous data ( proportion of individuals who had weight gain).
Conclusions
For a chronic perplexing psychiatric illness having a smaller prevalence, schizophrenia is associated with substantial disability and a high risk of suicide.4,40 The available pharmacotherapeutic options are unquestionably clinically effective. Nonetheless, they engender troublesome adverse effects, predisposing the patient to various cardiometabolic risks (e.g., hyperglycemia, dyslipidemia, increase in waist circumference, weight gain, and metabolic syndrome) and compromising the already affected compliance.
In such a scenario, introducing a novel alternative that may yield equivalent clinical efficacy with a better tolerability profile would be a panacea. OLZSAM, in various clinical trials, has demonstrated that added edge. Even in our evidence review, with the variable certainty of the evidence, we could affirm that OLZSAM offers comparable clinical efficacy with a low risk of weight gain. However, all the results should be evaluated in light of the smaller number of participants (not able to achieve optimal information size). Appraising our analysis and the previous evidence reviews, OLZSAM might be superior to OLZ regarding safety profile; even so, it warrants having head-on trials in real-world settings to genuinely ascertain the tolerability of this newly approved combination to generate more credible evidence.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
