Abstract
Background:
Depression and cognitive impairment often coexist in older adults. The relation between depression and cognitive impairment is complex. The objective of this article is to review recent literature on cognitive impairment in older adults with depression and provide clinicians an update.
Methods:
We searched PubMed, Google Scholar, Science Direct, and Psych Info for the articles published in the English language related to late-life depression (LLD)/geriatric depression and cognitive impairment. We considered original research articles, relevant systematic reviews, chapters, and important conceptual articles published in the last 9 years (2011–2019). We selected relevant articles for this narrative review.
Conclusion:
The concept pseudodementia, indicating depression with cognitive impairment mimicking dementia, is now seen only as a historical concept. The current literature strongly agrees with fact that cognitive deficits often exist in LLD. The cognitive deficits in depression were initially seen as trait marker; however, some recent studies suggest that cognitive deficits persist even in the remission phase. There is heterogeneity among the studies in terms of the nature of the cognitive deficits, but higher number of studies reported impairment in attention and executive function. LLD with cognitive deficits is at a higher risk of progression to dementia. In older adults, depression with cognitive impairments requires a comprehensive evaluation. Electroencephalography, event-related potentials, fluorodeoxyglucose–positron emission tomography, amyloid positron emission tomography, and CSF amyloid will supplement clinical evaluation in differentiating functional depressive disorder with cognitive impairment from depression with an underlying degenerative condition.
Patients with depressive disorder often report forgetfulness along with typical symptoms of depression. This is particularly evident in late-life depression (LLD) when compared to depression in younger age group.
1
Cognitive deficits could be the primary reason for consultation in LLD. It is often difficult to tell whether depression is implicated in cognitive deficits or the initial cognitive decline in a neurodegenerative condition leads to depression as a reaction to it. Longitudinal studies have reported depression as one of the important risk factors for dementia.
2
The current classificatory systems, International Classification of Diseases and Related Health Problems (
Method
We searched PubMed, Google Scholar, Science Direct, and Psych Info for the articles published in the English language related to late-onset depression (LOD) or geriatric depression and cognitive impairment, using terms such as “pseudodementia,” “neuropsychology in LLD,” “dementia and depression,” “cognitive deficits in LOD,” “social cognition in LLD,” “neurobiology of cognitive impairments in depression,” and “evaluation of cognitive impairment in LOD.” For this review, we considered original research articles, relevant systematic reviews, chapters, and important conceptual articles published in the last 9 years (2011–2019). We obtained 2423 articles in PubMed, 59 articles in Google Scholar, 1141 articles in Science Direct, and 236 articles in Psych Info. We selected relevant articles for this narrative review. This review is an update of our previous review article published in 2010. 7
Pseudodementia
Pseudodementia, often a misunderstood term, is a much-debated concept with respect to its validity and use. The term was first coined in the 1880s by Wernicke, describing the chronic hysterical states mimicking mental weakness. This concept went into disuse for nearly half a century. Later in 1952, Madden reintroduced this term to describe patients with clinical features of dementia that disappeared with successful treatment. In 1961, Kiloh described ten cases presenting with dementia who recovered with treatment. He used this term to decrease the nihilism among clinicians regarding dementia.
5
In 1979, Wells, in an article describing ten patients, used pseudodementia as a diagnosis for the first time.
8
There is no unified definition or consensus on the description of what constitutes pseudodementia. The commonly accepted definition is that pseudodementia is a condition presenting with cognitive deficits due to an underlying primary psychiatric disorder mimicking dementia. These deficits are mostly reversible and without any organic etiology. Most commonly, the term pseudodementia is used for cognitive deficits in major depressive disorder. In the
Cognitive Deficits in LOD
The studies related to cognitive deficits in LLD can be broadly classified as those comparing LOD with healthy controls, LOD with Alzheimer’s dementia (AD) or mild cognitive impairment (MCI), and LOD with early-onset depression (EOD).
The cross-sectional studies comparing the LOD with healthy controls have uniformly reported poorer performance in neuropsychological tests in LOD. Impairment in processing speed, executive function, response inhibition, learning and memory, language, arithmetic, and clock drawing ability has been reported.13–20 Most of the studies agree about the presence of executive dysfunction in LOD.
Poorer performance in LOD compared to EOD in verbal learning and memory, fluency, language, visuospatial skills, and clock drawing ability has been observed. Overall, the LOD group has been noted to have greater deficits compared to EOD.1,16,17,21 However, a similar level of impairment in terms of executive function and response inhibition was observed irrespective of the age of onset of depression, too. 16
Studies have compared cognitive deficits in LLD, MCI, and AD, predominantly related to the profile and pattern of deficits. The difference was more in terms of the degree of cognitive impairment. AD patients have severe deficits, followed by severity by MCI and LOD patients. In terms of the pattern of cognitive deficits, AD and MCI patients performed poorly in learning and memory tasks and language tasks (fluency and semantic tasks).13,14,18 Dillon et al. reported that depressive spectrum disorder in older adults had a subcortical type (attention and executive functions) of cognitive deficits, whereas AD patients had cortical type (impairment in memory with good recognition) of cognitive deficits. 22
Only a few prospective studies related to LOD and cognitive deficits have been reported during the period of our review. In a prospective study from the USA, older adults with the LOD (
Three studies investigated the risk factors for cognitive deficits in LOD during our review period. A prospective study involving LOD patients (
To summarize, most studies report cognitive deficits in older adults with depression irrespective of age at onset and severity. The majority of the studies reported that cognitive deficits were more in LLD compared to those with EOD. A few studies that compared LLD with MCI and mild AD reported that the difference was more in terms of severity, with LLD patients having relatively milder cognitive deficits. The findings involved impairment in attention, working memory, and executive dysfunction and good recognition memory.
22
This could be explained by the involvement of subcortical structures in LLD, and it is known that subcortical circuits have a role in executive functions.
30
Most of the studies were cross-sectional, involving outpatient clinics/hospitals. In most of the studies, depression was diagnosed using
There was heterogeneity in the kind of instrument for the assessment of cognitive functions in the reviewed studies. The comparison between EOD and LOD in older adults that many studies had investigated did not consider the number of depression episodes. Factors such as medical illnesses, anticholinergic burden, and electroconvulsive therapy, which can potentially contribute to cognitive deficits and can act as confounders, were not taken into consideration. Also, factors such as poor motivation, anhedonia, catatonic features, and poor cooperation can affect the performance during the neuropsychological assessment.
Social Cognition Deficits in LOD
There is emerging research on social cognition and its relation with the nature of deficits, severity, and its role in depressive disorders. The domains of social cognition include theory of mind, attributional bias, emotion processing, and social perception. Persons with major depressive disorder have performed poorly on overall social cognition compared to healthy controls.31,32 There were two reports on social cognition in older adults with depression. Szanto et al. assessed the relationship between social emotion recognition and attempted suicide in LLD using Reading the Mind in the Eyes (RME) test. RME is a measure of mentalizing ability, which is tested using 36 grayscale photographs of eye regions. Each photo with an emotion is shown along with four words. The participants have to choose a word/phrase that best describes the emotion. 33 The study reported that those who had attempted suicide made significantly more errors in social emotion recognition compared to nonattempters with LLD and healthy controls. 34 Another study compared facial emotion recognition ability among older adults with and without depression using static and dynamic stimuli. The study found that older adults with depression were better in recognizing negative emotions, such as sadness and anger, but less accurate in recognizing happiness emotion. 35
Risk of Progression to Dementia
A retrospective study reported that the risk of dementia increased by 20% for mid-life depressive symptoms and 70% for late-life depressive symptoms.
36
Among the prospective studies, the Framingham study cohort is an important one, consisting of 949 participants assessed at baseline and after 17 years. At baseline, 13.2% of subjects had depressive symptoms. At follow-up, 21.6% of those with depression developed dementia compared to 16.6% of nondepressed individuals.
37
In the Women’s Health Initiative Memory Study, 6376 postmenopausal women were followed up annually for 5.4 years for depression and MCI/dementia. The study reported that depression at baseline was associated with a higher risk of MCI (hazard ratio—1.98, 95% CI = 1.33–2.94, P < 0.001) and probable dementia (hazard ratio—2.03, 95% CI = 1.15–3.60, P = 0.01).
38
A prospective study from Italy that compared the risk of progression to AD among patients with LOD or amnestic-MCI (aMCI) noted that the risk of progression to AD in 4 years was 63.33% for the aMCI group and 49.57% for the LLD group (P = 0.02).
39
In a community-based prospective study from Spain, LOD (
In a pooled analysis of 18 studies (13 case-control and 5 cohort studies), the risk of conversion to dementia was compared among MCI subjects with or without depressive symptoms. It found that the relative risk of progressing to dementia was 1.28 (CI = 1.09–1.52, P < 0.01) in patients with depressive symptoms. 41 A study that followed up 321 older adults with MCI for two years found that 27.5% of older adults with depression had progressed to dementia, compared to 14.8% of those without depression (P = 0.03). 42 In a study where amyloid-positive MCI subjects were followed up, the conversion rate in people with depression was 40.8% compared to 19.7% in those without depression (P < 0.01). 43 The presence of depressive symptoms and their severity were among the main risk factors for the progression of MCI to dementia. 44 From the above discussion, it becomes clear that depression is a risk factor for dementia. The risk is present irrespective of the age of onset of depressive disorder. The risk appears higher in late-onset compared to EOD.
Neurobiology of the Cognitive Deficits in Depression
A few hypotheses have been proposed for cognitive deficits in depression. The commonly known hypothesis for episodic memory deficits in depression is the reduction of hippocampal size.45,46 Hippocampus, the brain region concerned with episodic memory, is known to have a higher density of glucocorticoid receptors. The stress during depression causes an increase in circulating cortisol. This, in turn, acts on the hippocampal glucocorticoid receptors, leading to neurotoxicity. 47 Studies have reported a smaller hippocampal volume in adults with recurrent depression, which leads to cognitive deficits. 48 The second hypothesis is the role of brain-derived neurotrophic factor (BDNF) in cognitive impairment in LLD. BDNF is involved in maintaining neuronal homeostasis, synapse development, and strengthening through glutamate pathways, memory consolidation, and resilience against insults. 49 Another hypothesis is the vascular disease of the brain. The cerebrovascular disease pathology may cause frontostriatal damage and hippocampal volume loss, causing depression and cognitive impairment simultaneously.50,51 Functional neuroimaging data have reported conflicting results, with studies reporting hypoactivation and hyperactivation of prefrontal regions in depression. 52 Studies also have reported that in depressed patients, there is a failure in the deactivation of the default mode network during a cognitive task. 53 The close relationship between LOD and dementia is also observed at the genetic level. There are reports of a higher frequency of ApoE4 in LOD patients compared to controls. 54 ApoE4 is also a known risk factor for sporadic Alzheimer’s dementia. 55 This might explain ApoE4 contributing to depression and dementia at various time points. Some of the neurobiological markers in LLD that are pointers for higher risk of AD include medial temporal atrophy, positive cerebrospinal fluid (CSF) or PET amyloid markers, higher vascular lesions, and increased proinflammatory cytokines in the brain. 56
Antidepressants, Electroconvulsive Therapy, and Cognitive Deficits
Antidepressants, such as tricyclic antidepressants (TCA), due to their action at muscarinic, nicotinic, adrenergic, and histaminergic receptors, could contribute to cognitive decline. Particularly, the central anticholinergic action of TCAs is known to increase cognitive impairment. 57 A 10-year longitudinal study on community-dwelling older adults on antidepressants reported that subjects on TCA had poorer performance on verbal fluency, visual memory, and processing speed, whereas SSRIs had poor performance on verbal fluency and processing speed. However, over time, accelerated decline was not observed in either group. 58 A systematic review reported that antidepressants are associated with a twofold risk of cognitive impairment. 59 Another recent meta-analysis of 33 studies investigated the effect of antidepressants on specific cognitive domains. It reported that SSRIs have the maximum positive effect on all cognitive domains. In contrast, TCAs have a negative effect on divided attention and no significant effect on other cognitive domains. The studies that included SNRIs, atypical antidepressants, and stimulant antidepressants found either a modest positive effect or no significant effect on cognition. 60 Currently, SSRIs are reportedly safe for cognition, except for paroxetine, which has higher anticholinergic action. The evidence regarding other classes of antidepressants is limited. Electroconvulsive therapy is one of the effective treatments for depression in older adults. Though efficacious, it is known to cause transient cognitive deficits in older adults.61,62 The cognitive deficits with ECT can range from transient anterograde and retrograde memory deficits to delirium. In a recent case-control study on ECT in acute mental illness, where the mean age of the sample was 50 years, improvement in cognitive functioning was observed. 63 Another study also reported improvement in cognitive aspects with ECT in treatment-resistant depression in older adults. 64 Overall, ECT in older adults for depression may have a more positive effect on cognition though it is known to cause transient acute cognitive deficits.
Comorbid Medical Illness
Medical illnesses such as hypothyroidism, vitamin B12 deficiency, and anemia, besides noncommunicable diseases, are common comorbidities in older adults. These medical illnesses present with various neuropsychiatric manifestations, including depression and cognitive impairment.65,66 This can be another explanation for the co-occurrence of cognitive deficits in older adults.
Clinical Evaluation and Management
Any older adult presenting with depressive symptoms and cognitive impairment requires a systematic evaluation. Three clinical scenarios might result out of the evaluation. The first is, the patient might have only depression, the other is depression with cognitive deficits, and the last scenario is where one might have coexisting dementia and depression. This differentiation will help choose the appropriate treatment. There can be medico-legal implications as well. To start with, a detailed history of the depressive symptoms, number of episodes, and severity of depression should be obtained. Patients with history of severe depressive symptoms, multiple episodes, and melancholic depression are more likely to have coexisting cognitive impairment. History should be obtained from many collateral sources, as a single informant might not be sufficient. At least a bedside cognitive assessment should be done in every case. Serial observations of the patient and careful recording of depressive symptoms and memory complaints are advisable. One should look for the impact of cognitive complaints on the daily activities and note for any discrepancies. Recording any variation in bedside cognitive assessment serially or even daily may aid in diagnosis. Thyroid function tests, serum vitamin B12 level, complete blood count, and serum electrolytes must be included in a comprehensive evaluation. Imaging (CT or MRI-brain), electroencephalography (EEG), and neuropsychological assessment are advised in older adults where there is a high suspicion of dementia.67–70
The management of these patients requires a multidisciplinary team to collaborate with patient and family members to take a collective decision and communicate a clear message about the treatment plan. In patients with depressive disorder and cognitive impairment, it is imperative to treat the depression first irrespective of its etiology, whether it is the result of underlying functional, vascular, or degenerative condition. Simultaneously, the medical comorbidities should be specifically addressed. Optimization of medications for chronic conditions is suggested in every case. Polypharmacy, commonly seen in the older adults, might be contributing to cognitive deficits. Specifically, medications with anticholinergic and/or antihistaminic effects and those causing sedation will need to be reviewed. Prospectively, it is important to evaluate these patients for cognitive decline even after remission of depressive symptoms. Serial cognitive assessments will generally help diagnose dementia in these patients. 71 Clinical variables helpful in differentiating depression with cognitive impairments and dementia with depression are shown in Table 1.
Differentiating Between Depression, LLD with Cognitive Impairment, and Coexisting Dementia and LLD
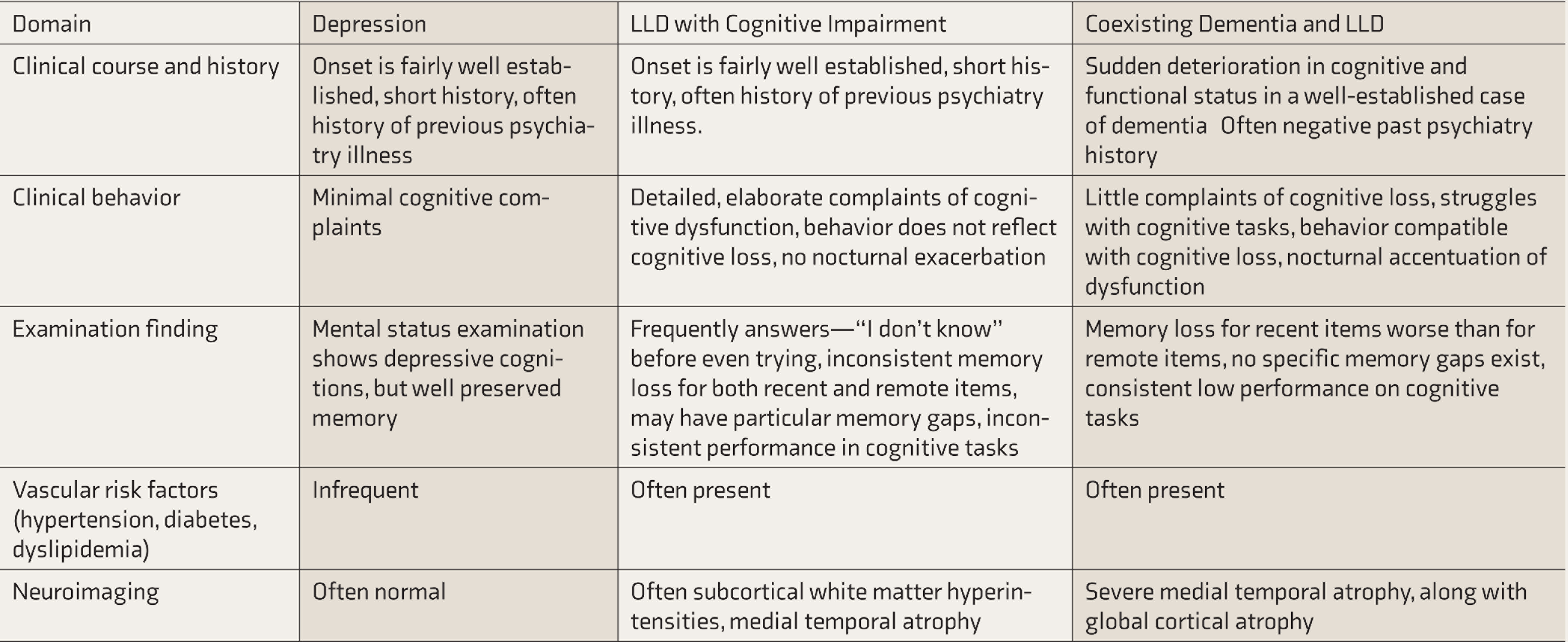
LLD: late-life depression.
Investigations to Differentiate Depression with Significant Cognitive Impairment and Alzheimer’s Dementia
In a few patients, even after thorough evaluation, it may not be possible to come to a conclusive diagnosis. In these patients and medico-legal cases, one might need to perform additional investigations. We have mentioned here some electrophysiological and other biomarkers that may help ascertain the diagnosis.
EEG may help differentiate depression with cognitive impairment from dementia. In AD, EEG shows abnormal slowing and higher global theta activity. 72 Quantity EEG is also found useful in this clinical scenario.73,74
Event-related potentials can also reliably diagnose patients with Alzheimer’s disease (AD) pathology. Patients with AD have increased P300 latency and decreased amplitude. In contrast, they are normal in healthy controls and depressed patients. 75
Studies have reported that fluorodeoxyglucose–positron emission tomography (FDG-PET) of the brain helps in differentiating both these conditions. FDG-PET shows hypometabolism in temporoparietal areas in patients with AD as compared to patients with LLD. 76
Biomarkers, such as CSF-Aβ42, may also help to differentiate AD from depression with cognitive impairment. In patients with AD, the levels of CSF-Aβ42 were abnormally low, whereas in patients with depression with cognitive deficits, the levels were normal.76,77 These biomarkers may not be readily available for clinical use besides in research settings. The recent developments in LLD with cognitive impairment are shown in Table 2.
The New Developments in the Past 9 Years on LLD with Cognitive Impairment

LLD: late-life depression, EEG: electroencephalography, FDG-PET: fluorodeoxyglucose–positron emission tomography.
Conclusions
Depression with coexisting cognitive impairment is a heterogeneous condition more often seen in older adults. Predominant cognitive complaints may be at the forefront rather than low mood. Simultaneously, there is also a risk of these being misinterpreted as transient subjective cognitive complaints during depression despite LOD being frequently associated with cognitive deficits. These patients have an increased risk of conversion to MCI and dementia. Cognitive deficits during depression increase functional impairment in older adults. Evaluation requires a systematic and comprehensive approach. It is suggested to look at multiple contributory factors causing cognitive deficits rather than a single etiology. Communicating the diagnosis to the patient and their families helps to allay apprehension and unnecessary investigations. In medico-legal cases, prospective examination and using appropriate objective tests may be helpful. There is variability in research findings regarding the nature of cognitive deficits in depression. This might be due to the heterogeneous samples, instruments, and settings. Social cognition deficits in LOD has been recently reported.
Future Directions
There is a need for prospective studies on depression in older adults, to improve the understanding on the association with cognitive deficits and develop objective markers to differentiate LLD from dementia early on in the presentation. Future studies can also incorporate biomarkers (amyloid PET or CSF Amyloid) to exclude AD pathology while studying cognitive impairment in LLD. There is a need to study the predictors of remission with respect to cognitive impairment in LLD.
Supplemental Material
Supplemental material for this article is available online.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
