Abstract
Background:
Persons with previous history of a suicide attempt are at increased future risk of death by suicide. These vulnerable individuals, however, do not seek receive or seek help from mental health services. Telephone-based psychosocial interventions are potential strategies in augmenting mental health care in such persons.
Methods:
We aim to compare the efficacy of telephone-based psychosocial interventions (TBPI) with routine telephone reminders in persons with recent suicide attempts using a multi-site, parallel group, rater-blind, two-arm randomized controlled trial design in 362 participants. In the first group, participants will receive three sessions of TBPI comprising of brief supportive interventions, problem-solving strategies, and reminders for adherence to prescribed mental health treatment at weekly intervals. In the second group, participants will receive three telephone reminders for adherence to prescribed mental health treatment at weekly intervals. We will follow up participants for 6 months. Primary outcomes are suicidal ideation scores on Beck’s Scale for Suicide Ideation and number of repeat suicide attempts. Secondary outcomes are scores on Beck’s Hopelessness Scale, Beck’s Depression Inventory, Connor–Davidson Resilience Scale and Visual Analogue Rating Scales for acceptability of interventions. Outcomes will be assessed at 1, 3, and 6 months after receiving telephone interventions or reminders.
Results:
The trial is currently underway after prospective registration under Clinical Trials Registry of India and has recruited 260 participants till August 15, 2020.
Conclusion:
This study has potential to generate evidence on additional strategies for use along with standard mental health treatments in management of high-risk suicide behaviors.
Keywords
Brief contact interventions including telephone counseling are a potential useful strategy in augmenting standard mental health treatments in individuals with high suicide risk. There is lack of evidence in type of telephone interventions that could be useful in individuals with previous suicide attempt. This trial describes the protocol of a multi-centric randomized control to compare the efficacy of a telephone based psychosocial intervention with routine telephone reminders on suicidal ideation in such individuals.Key Messages:
Suicide is a significant cause of mortality in India. 1 Suicidal behaviors, such as suicide attempt and deliberate self-harm, have shown significant associations with death due to suicide. 2 The National Mental Health Survey showed that prevalence of high risk of suicidal behaviors in India was 0.9% with the national suicide rate for reporting year 2014 being 10.6 per lakh population. 3 Risk factors for suicidal behaviors in India were of biopsychosocial origin. 4 Prevalence of depression and other mental disorders in persons who attempted suicide ranged from 22%–59% in Indian studies. 5 Long-standing psychosocial vulnerabilities were definite risk factors for repetition of a suicide attempt. 6 Gender-related factors were implicated from findings that showed suicide death rates were higher than expected in women taking account the country’s sociodemographic index level. 7 Impulsivity and hopelessness were significant variables that appear to have significant interactions with stressful life events in persons with suicide attempts in Indian settings. 8
India has a wide mental health gap and this makes it necessary to focus on suicide prevention as a top priority. 9 A large proportion of mentally ill individuals do not seek help from mental health services prior to dying due to suicide. 10 It is thus necessary to consider alternate strategies that could deliver or augment mental health care in this vulnerable population. 11
Low-cost psychosocial interventions of brief duration are useful in suicide prevention. 12 Brief contact interventions that involve regular, structured contact by non-specialist health workers with the affected individuals through telephones, mobile messages, and post cards are being studied in suicide prevention. 13 In a study conducted in Chennai general hospital settings, the group that received a detailed individual information session on risk factors associated with suicide and help seeking avenues followed by repeated follow-up contacts through telephone for a period of 1 year showed significant reductions in deaths due to suicide and repeat suicide attempts as compared to treatment as usual group. 14 This finding of significantly lesser deaths due to suicide in individuals who received brief interventions along with periodic follow-up contact was noted across eight countries and led to the conclusion that periodic follow up of individuals at high risk of suicidal behaviors could be a potential low-cost public health strategy to reduce suicide related mortality. 15
Telephones in India are potential delivery channels of psychosocial interventions that could help individuals with suicide attempt access mental health services in India. 16 A systematic review of 16 studies across the world that used mobile phones, text messaging, and other technological modes of brief interventions in individuals with suicidal behaviors demonstrated an encouraging trend toward reduction in suicidal ideation, hopelessness, and depression in such individuals. 17
Thus, while follow-up phone contacts in persons with suicidal behaviors are useful as additional treatments in persons with suicidal behaviors, there is a need to further evaluate the type of telephone interventions that could be useful in Indian settings. This study, using a randomized controlled design, aims to compare the efficacy of telephone-based psychosocial interventions (TBPI) with routine telephone contacts (TC) in individuals with recent suicide attempt on future risk factors, such as persistent suicidal ideation, repeat suicide attempts, persisting depression, persisting hopelessness, and protective factors, such as resilience. This study also aims to study and compare the acceptability of these two types of telephone communications.
Comparator
Studies on telephone interventions in individuals with suicidal behaviors in India have typically been in the form of regular, repeated, brief TC that focused on encouraging them to seek help from mental health service and adhere to prescribed mental health treatment. 14 Thus, there is a need to include this component in all telephone communications. Any study on telephone interventions in this high-risk population should also include specific inquiry into persistent suicidal ideation and behaviors so that urgent psychiatric care can be recommended as needed. Therefore, to control for these effects, participants in both groups in this study would receive an empathetic inquiry into the presence of suicidal ideation, reminders for treatment adherence, and follow up with their respective mental health care services as part of all telephone communications. We hypothesize that while participants in control group would receive some benefits, this would be lesser as compared to those in the intervention group.
Methods and Methodology
Study Overview
Objective
Our research hypothesis is that TBPI are more efficacious than TC in reducing future risk factors in individuals with recent suicide attempt.
Therefore, the primary objectives are as follows:
to study whether TBPI are more efficacious than TC in reducing suicidal ideation in individuals with recent suicide attempt on Beck’s Scale for Suicidal Ideation (BSS); to study whether TBPI are more efficacious than TC in reducing number of attempts to self-harm in individuals with recent suicide attempts. to study whether TBPI are more efficacious than TC in reducing hopelessness on Beck’s Hopelessness Scale (BHS); to study whether TBPI are more efficacious than TC in improving resilience on Connor–Davidson Resilience Scale (CD-RISC); to study whether TBPI are more acceptable than TC on Visual Analogue Likert Scale (VAS).
Design
This is a multi-site, parallel group, observer-blinded, two-arm randomized controlled trial, with participants receiving either TBPI or TC as the control condition (Figure 1). The protocol follows Standard Protocol Items: Recommendations for Interventional Trials guidelines (see Table 2).
Flow chart of trial
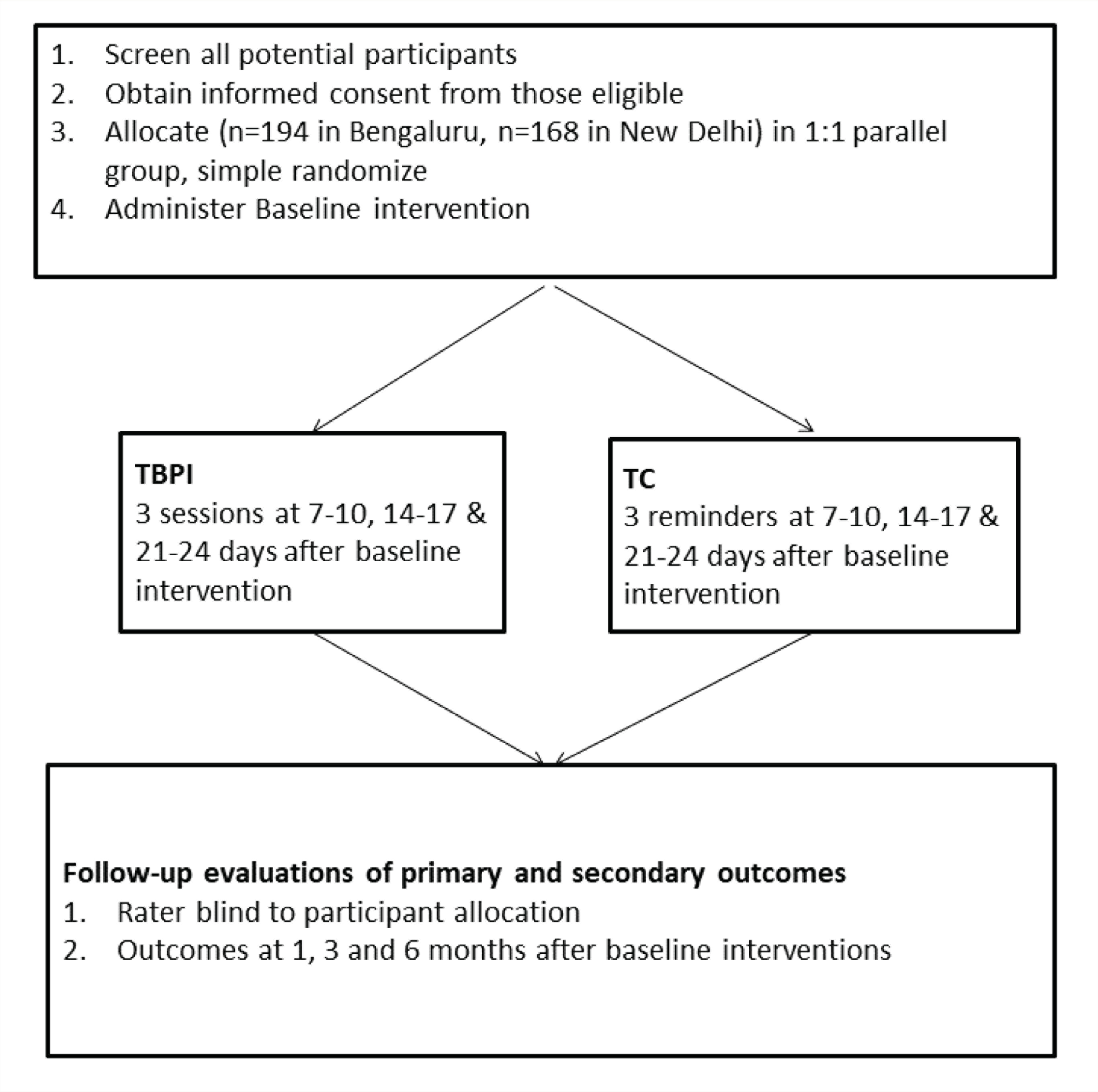
Participant Recruitment
This will be a multi-centric study with two sites in general hospital psychiatry settings in metropolises in India. The sites are Departments of Psychiatry at St John’s Medical College and Hospital (SJMCH) in Bangalore and Atal Bihari Vajpayee Institute of Medical Science and Dr Ram Manohar Lohia Hospital (ABVIMS-RMLH) in New Delhi. Both sites are referral centers that treat large number of individuals with suicide attempts for their medical and psychiatric conditions. We will recruit participants from out-patient, in-patient psychiatry settings, emergency, and other medical specialties.
Inclusion and Exclusion Criteria
These are outlined in Table 1. For purposes of this study, we will define a suicide attempt as “any non-fatal suicidal behavior and refers to intentional self-inflicted poisoning, injury, or self-harm, which may or may not have a fatal intent or outcome” as defined according to the World Health Organization. 9 We will exclude individuals with substance dependence (except nicotine dependence) as they will require specific motivation enhancement and relapse prevention strategies using M.I.N.I. version 6.0. 18 We will exclude individuals with current psychotic symptoms as they would require specific pharmacological treatments and psychoeducation.
Inclusion Criteria and Exclusion Criteria

Participant Consent Process and Screening
Research associates (RAs) will identify and screen potential participants and explain study objectives and procedures in detail. Once the participant assents, RAs will assess to check eligibility for recruitment according to inclusion and exclusion criteria. Subsequently, RAs will obtain written informed consent. A copy of the subject information sheet that provides all necessary details about study aims, objectives, and contact details of principal investigators will also be made available to the participants.
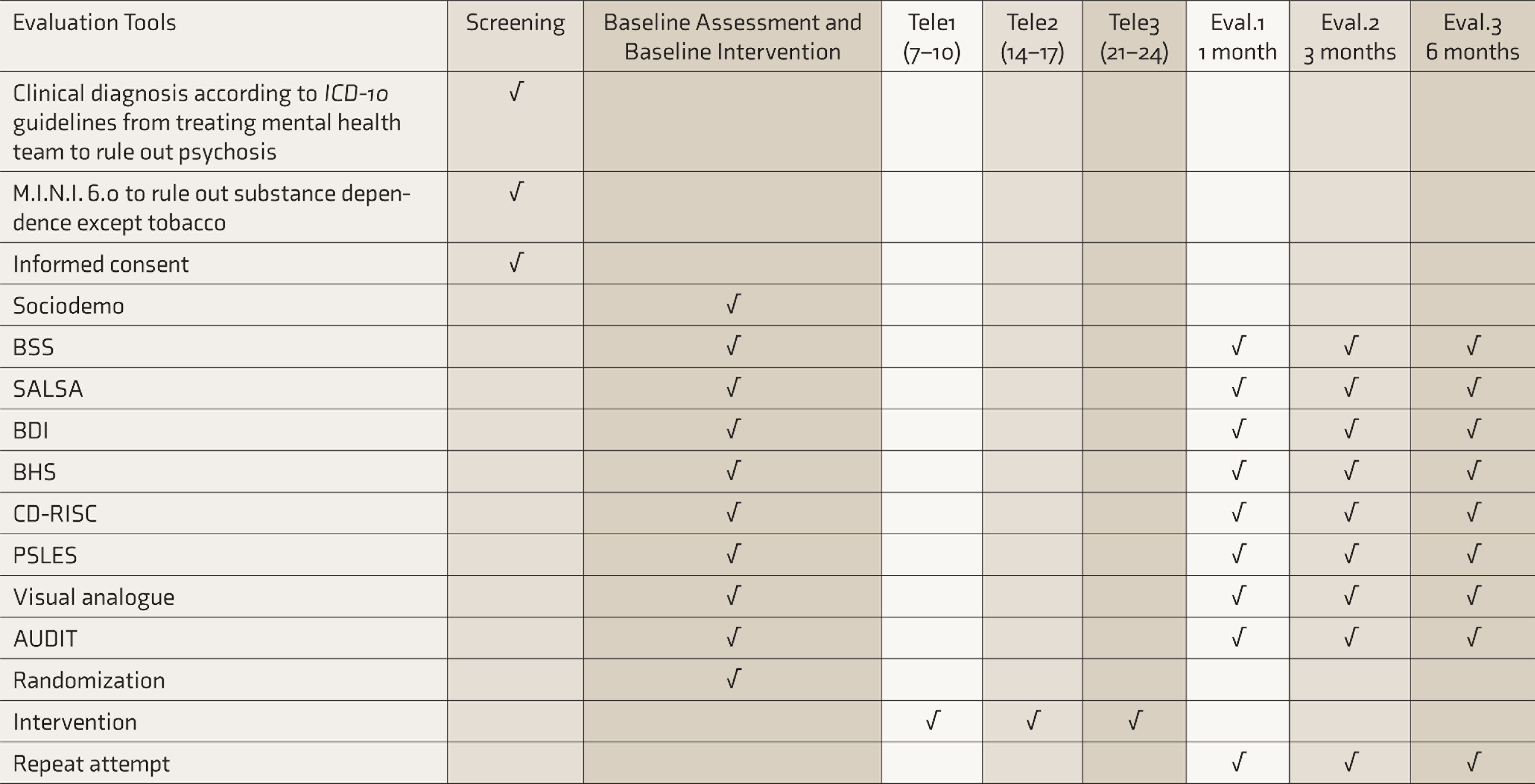
Baseline Assessment
After written informed consent, research associate 1 (RA1) will perform a baseline assessment using instruments. Instruments used as part of baseline assessment are as follows.
Outcome Measure
Primary outcome measures will be group comparisons on persistent suicidal ideation using BSS and number of repeat suicide attempts. Secondary outcome measures will be group comparisons on depression (BDI), hopelessness (BHS), and acceptability of interventions using VAS.
Randomization
We will allocate participants to either TBPI or TC in 1:1 ratio using simple computerized randomization in blocks of ten performed separately at each site. The randomization sequence will be performed by study personnel who will not be involved either in delivery or evaluation of TBPI and TC. RAs will remind participants during the evaluation process not to divulge contents of their phone conversations to prevent unblinding.
Procedure of Study
After RA1 completes baseline assessment, research associate 2 (RA2) will deliver a baseline psychosocial intervention to all participants. This intervention will consist of information on the following:
risk of repeat suicide attempt and death; need to adhere to prescribed treatment and follow up with mental health services; avenues of seeking help during times of crisis; suggestion of strategies that will help in coping and problem solving.
We will derive these strategies from a planned menu of options using principles from supportive, cognitive, and dialectical behavioral schools of psychotherapy. 29 Subsequently, RA2 will deliver the allocated TBPI or TC on three occasions: 7–10 days, 14–17 days, and 21–24 days after baseline interventions. The details of the interventions are mentioned as follows.
Telephone-Based Psychosocial Intervention (TBPI)
In initial two sessions, RA2 will inquire about stressors and whether the participant attempted to use strategies suggested during baseline intervention. Subsequently, RA2 will discuss additional strategies that participant could try out.
In the third TBPI session, RA2 inquires and provides clarifications about previously suggested strategies.
As part of all three TBPI sessions, RA2 will inquire into persistent suicidal ideation and repeat suicide attempt in the participant. RA2 will advise the participant to continue their regular follow-ups with their mental health care providers, adhere to prescribed treatment, and avoid substance use. TBPI will last for a range of 5–15 min. TBPI thus primarily uses supportive and problem-solving strategies.
Telephone Contact (TC)
TC will be the comparator to TBPI. In all three sessions of TC, RA2 will inquire into persistent suicidal ideation and repeat suicide attempt in the participant. RA2 will advise the participant to continue their regular follow-ups with their mental health care providers, adhere to prescribed treatment, and avoid substance use. TC will last for a range of 3–5 min.
Ethics and Safety Measures
We will inform participants prior to obtaining written informed consent that while we would endeavor to maintain confidentiality as far as possible, as per best practice guidelines if we discover persistent suicidal ideation and behaviors during the course of our study, we will inform a designated care about this. This is to ensure that the participant receives urgent medical care. In order to ensure this, we will obtain the caregiver’s contact details. In this scenario, while we will go ahead with informing the caregiver about the suicide risk, we will aim to maintain confidentiality as far as possible with respect to other aspects. If the participant at this juncture expresses intent about not wanting to continue participation in the study, we will exclude the participant from the study. We will, however after this, follow up on phone at least once with this participant and caregiver to check whether they sought help and to encourage continued help-seeking.
Procedure
The participants will be assessed by RA1 who will be blinded to the allocation of participant. RA1 will evaluate participants at 30–33, 90–95, and 180–187 days after baseline interventions for primary and secondary outcome measures (Table 2).
SPIRIT: Showing Schedule of Visits
Quality Control
Training
Two RAs with a minimum of masters’ degree in psychology will be trained to assess participants and deliver TBPI and TC at each site. RAs will first observe mental health specialists evaluating and treating individuals with history of suicide attempts in both study sites. We will then train RAs using role plays. RAs will be permitted to evaluate participants and deliver TBPI and TC initially under supervision and subsequently on independent basis. They will continue to be under constant supervision during the course of this study.
Fidelity Assessments
We will aim to randomly record 10% of both the intervention as well as the control. These recordings will be assessed by an independent psychiatrist using a predetermined checklist of whether the RAs show fidelity in actual delivery of the interventions as compared to the manualized intervention.
Feasibility Study
We will perform a feasibility study at both sites to test feasibility and acceptability of the intervention and to identify and address any difficulties that arise prior to the conduct of main trial. Feasibility will be assessed using dropout rates while acceptability will be assessed using VAS scores. We will not study the primary outcomes or other secondary outcomes, such as hopelessness and resilience.
Sample Size Consideration
In a study by Guthrie et al., brief psychological interventions in individuals with deliberate self-poisoning reduced suicidal intent significantly on BSS with a mean difference of 4.9. 30 Using this as reference, we performed a power analysis using G-power software. Using an effect size of 0.5 with power of 80% and alpha error of 0.05 with an attrition rate of 25%, we obtained a sample size of 84 in each arm (N = 168) that would be adequate for both sites together. In accordance with recommendations from Indian Council Medical Research (ICMR), we intended to recruit this sample size for ABVIMS-RMLH site. For SJMCH, we calculated the sample using an effect size of 0.5 with a higher power of 85%, alpha error of 0.05, and attrition rate of 25% and obtained a sample size of 97 in each arm (N = 194). We estimated the larger sample size for SJMCH as that site already had an established assertive management of attempted suicide clinical service. 31
Data Entry and Data Safety Monitoring
ICMR has designed a custom password protected Microsoft Access based data repository software for entry and analysis. We will capture data in real time using tablets and enter the data into this ICMR repository. We will exercise utmost confidentiality while entering data along with the necessary security precautions using encryption. The data will be accessible to ICMR for further analysis in future.
Data Analysis
We will use a repeated-measures, mixed-effects model. Primary outcome measures will be scores on Beck’s suicidal intent scale and the presence of number of repeat attempts at two time points, first month and sixth month. Secondary outcomes will be scores on Beck’s hopelessness scale, BDI, Connors–Davidson Resilience Scale, and VAS. We will use an intent to treat form of analysis.
Evaluating Study Progress
This research proposal was planned in “ICMR–Dr RMLH–Pittsburgh University Capacity Building Workshop on Implementation Research under NMHP” held during November 14–19, 2016 in New Delhi. 32 The national coordinating committee set up in ABVIMS-RMLH by the ICMR will monitor the study progress in monthly meeting with principal investigator telephonically or on skype. This will be further assessed in every 6 months with mentors under capacity building workshop. Principal Investigators (PI) and RAs of both study site will confer every month to assess progress (Figure 1).
Ethical Standards
The authors assert that this study compels with all standards of Institutional Ethics Committee of SJMCH and ABVIMS-RMLH, Indian Council of Medical Research guidelines, and Helsinki declaration. We will recruit participants only after obtaining written informed consent. We will take utmost care in maintaining confidentiality during interventions, evaluations, and data entry. We will use alphanumeric codesign order to obscure identification. In the event of any suicide attempt or death during course of the study, we will inform Institutional Ethics committee and ICMR national coordinating committee.
Results
The study has been prospectively registered in Clinical Trials Registry of India on September 18, 2018. Participant enrolment began on November 15, 2018. Currently, both sites have randomized around 260 participants (till date August 15, 2020).
Conclusions
National Mental Health Program has the objective of making necessary mental health care accessible to all including most vulnerable and underprivileged sections of population. 33 This study outcomes have the potential to generate evidence relevant to this stated objective. This trial is designed as a pragmatic trial with the intention of intervening in vulnerable individuals through telephone calls as a large number of people in India have access to a telephone. 16 This study also aims to evaluate interventions that could supplement mental health care similar to others that have used telephone-based systems to augment psychiatric treatment and monitor suicidal ideation in high-risk individuals with severe mental illness. 34
An important aspect to be considered during the study conduct is that while all participants receive a detailed baseline intervention, the subsequent telephone communications occur through phone. Thus, it is essential to establish a good therapeutic alliance within a short period. In the absence of a therapeutic alliance, there might be high dropouts noted giving rise to an inherent limitation. A potential limitation of this study could be the relatively short follow-up period; however, this duration also makes it potentially more feasible. As this is a multi-center trial across general hospitals in two urban metropolitan cities situated in different regions in India, the study findings will have external validity. However, the authors are aware that these findings cannot be generalized to rural, community, and other settings. Also, the exclusion of individuals with substance-dependent disorders and current psychotic disorders limits the applicability of the findings in two subgroups of populations that are at high risk for suicidal behaviors. Other limitations include exclusion of vulnerable populations, such as adolescents and elderly.
Suicide prevention is a matter of immense importance for India. 35 However, the large mental health gap along with poor follow-up rates act as barriers for individuals with suicide attempts in India. The increase in number of telephone users in India along with reduced costs of phone calls and data could potentially circumvent these barriers. 16 Mobile phones could serve as conduits for additional mental health interventions that understand and prevent suicidal behavior. 36 By comparing the efficacy between two types of telephone outreach communications, this study would have implications for implementation research in suicide prevention in India. A pilot study from an academic hospital in India demonstrated that delivery of aftercare for patients with psychiatric disorders was perceived as acceptable and feasible in this current COVID-19 scenario. 37
Finally, National Mental Health Program in India has shifted focus to capacity building, task shifting, and collaborative care in order to improve availability of mental health services. 38 Telephone interventions proposed in this study could assist in this by providing potentially scalable interventions to other settings subject to nature of findings.
Footnotes
Acknowledgements
We thank Dr Soumya Swaminathan (then Secretary, Department of Health Research, DHR), Dr Balram Bhargav, current Secretary DHR, Professor VL Nimgaonkar, Dr Ravinder Singh, and Dr Harpreet Singh. We thank the faculty of “Cross-Fertilized Research Training for New Investigators in India and Egypt” (D43 TW009114, HMSC File No. Indo-Foreign/35/M/2012-NCD-1, funded by Fogarty International Center, NIH). We are also thankful to Dr Smita N Deshpande, Head of National Coordinating Unit of ICMR for NMHP Projects for their wholehearted support, constant encouragement, and guidance. We thank Data Management Unit of ICMR for designing the database for this project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This protocol paper describes the methodology of a research project funded under ‘Capacity Building Task Force for Mental Health Research in India’ funded by the Indian Council of Medical Research vide file number 5/4-4/151/M/2017/NCD-1. International mentors were funded by the training program ‘Cross Fertilized Research Training for New Investigators in India and Egypt’ funded by FIC, NIH (No. D43 TW009114).
