Abstract
Restrictive ventilatory patterns (RVPs) in older adults may contribute to morbidity and decreased quality of life. The purpose of this study was to begin to understand (a) the number of older adults residing in Continuing Care Retirement Communities with RVPs, (b) factors associated with RVP, and (c) whether RVP is associated with ambulation level. This descriptive study was conducted at three sites. RVP was evident in 34 of the 65 participants (52%), and 33 (97%) of these were either moderate or severely restricted. Dementia, dyspnea, kyphosis, and lower muscle strength were significant predictors of RVP. Eighty percent of the nonambulatory participants had RVP and 38% of the ambulatory participants had RVP. Designing interventions to address modifiable factors associated with RVP may prevent respiratory infections, help people to achieve better lung health, and increase physical activity tolerance.
People with a restrictive ventilatory pattern (RVP) have lung expansion impeded by one or more factors, resulting in a decreased lung volume, an increased work of breathing, and possibly inadequate ventilation and oxygenation. RVPs may contribute to two prevalent problems in older adults, the development of pneumonia and declining physical activity (Falcone, Blasi, Menichetti, Pea, & Violi, 2012; Fragoso, Enright, McAvay, Van Ness, & Gill, 2012). Residents in long-term care have high rates of pneumonia and decreased physical activity, but RVPs in this population have received little attention (Galik, Resnick, Hammersla, & Brightwater, 2013; Polverino et al., 2010).
Restrictive ventilation may be due to a decrease in the elasticity of the lungs or a problem related to the expansion of the chest wall during inhalation. Several lung diseases are classified as restrictive, such as sarcoidosis, pulmonary fibrosis, and chronic thromboembolic pulmonary hypertension (Criado et al., 2010; Seeger et al., 2013). While these medical conditions and their relationship to RVP are well defined, pain, obesity, kyphosis, and weak respiratory musculature are possible extrapulmonary causes of RVPs (Fimognari et al., 2007; Jones & Nzekwu, 2006).
Obesity is associated with decreases in lung volumes and a more RVP (Zammit, Liddicoat, Moonsie, & Makker, 2010). Weight on the anterior chest wall decreases chest wall compliance and respiratory muscle endurance that results in increased work in breathing and airway resistance (McClean, Kee, Young, & Elborn, 2008). Abdominal adipose tissue hampers the movement of the diaphragm, decreases expansion at the base of the lungs during inspiration and can cause hypoxemia (Malhotra & Hillman, 2008).
Respiratory muscles also need to function normally for ventilation to be effective. The primary inspiratory muscles of respiration are the diaphragm and external intercostals that elevate the ribs and sternum. The primary expiratory muscles are the internal intercostals, intercostalis intimi, and subcostals that depress the ribs and sternum (West & Luks, 2015). Sarcopenia, or the decline of skeletal muscle mass and strength with age, decreases the strength of the diaphragm and intercostal muscles and decreases the body’s ability to overcome upper chest wall restrictive forces (Arnall, Camacho, & Tomás, 2014; de Souza et al., 2016; Simões, Castello, Auad, Dionísio, & Mazzonetto, 2009).
During aging, changes in the thoracic cage and decreased mobility of costovertebral joints can restrict ventilation. Kyphosis is an exaggerated curvature of the upper thoracic spine and estimates are that 20% to 40% of older adults have a severe hunchback known as hyperkyphosis (Kado, Prenovost, & Crandall, 2007). Kyphosis can result from a variety of sources including developmental problems, degenerative joint diseases, compression fractures associated with osteoporosis, or trauma to the spine (Katzman, Wanek, Shepherd, & Sellmeyer, 2010). Depressed ribs commonly accompany thoracic kyphosis and the resulting decrease in the size of the thoracic cavity impedes the expansion of the rib cage.
Dyspnea leads to increased ventilatory drive, resulting in tachypnea with greater dead space ventilation or ventilation without perfusion. Decreased expiration accompanied by hyperinflation forces the inspiratory muscles to operate at a shorter, disadvantageous sarcomere length. The inspiratory muscles then function at a mechanical disadvantage that can cause an RVP that increases the amount of muscle effort required for a given ventilation. Some evidence supports that pulmonary stretch receptors and chest wall proprioceptors sense respiratory muscle underperfusion and respiratory muscle fatigue, and transmit the sensation of distress to the cerebral cortex (Parshall et al., 2012).
Respiratory networks become dysfunctional in neurodegenerative illnesses, including those associated with dementia. Multiple mechanisms of action have been studied including alterations to the serotonergic system and degeneration of brainstem respiratory neurons that are associated with impairments in respiratory drive (Hilaire et al., 2010; Zhou, Jung, & Richards, 2012). In addition, older adults and those with dementia may decrease movement of thoracic and abdominal muscles in an attempt to reduce their pain and discomfort. This may reduce lung volume, increase airway resistance, and increase retention of sputum and secretions because of coughing hesitance, which subsequently leads to atelectasis and pneumonia (Middleton, 2003).
The extent of RVPs and extrapulmonary factors associated with RVPs in older adults in Continuing Care Retirement communities (CCRCs) has not been studied. Identifying those who are at risk for RVP and understanding the modifiable factors associated with RVP can inform the development of interventions to prevent and reduce RVP.
Purpose
This study was undertaken to begin to understand (a) the number of older adults in CCRCs with spirometry-confirmed RVPs, (b) extrapulmonary factors associated with RVP, and (c) whether RVP is associated with ambulation level. The primary hypothesis was as follows: Controlling for body mass index (BMI), muscle strength, kyphosis, dyspnea, musculoskeletal pain, and dementia will predict an RVP.
Method
Design, Setting, and Sample
This descriptive cross-sectional study was conducted by convenience at three CCRCs that provide access to older adults who live independently as well as those who need almost complete assistance with activities of daily living. The study was approved by an institutional review board and written consent to participate was obtained from the legally authorized signatory. Participants included those living in independent apartments, assisted living, and skilled care. Exclusion criteria were as follows: (a) admitted for short-term rehabilitation, (b) lung disease that is classified as restrictive, (c) acutely ill, and (d) no myocardial infarction, eye or lung surgery within the previous 8 weeks (pulmonary function testing contraindications). Participants who were unable to perform spirometry testing were dropped. Sample size was based on an odds ratio of 2.5 (small to medium), power level of 0.8, and probability (alpha) level of .05. Using G Power, a minimum sample size of 69 is required per power analysis (Faul, Erdfelder, Buchner, & Lang, 2009). Two hundred fourteen people were solicited for participation and consent was received from 90.
Measurement
Restrictive breathing patterns
Spirometry testing was performed using the MicroLoop battery-operated portable spirometer (MicroDirect Inc., Lewiston, ME). One trained research nurse conducted all tests using the standards and guidelines of the American Thoracic Society (Brusasco, Crapo, & Viegi, 2005; Levy et al., 2009). Disposable valved mouthpieces were used and calibration with a certificated calibration syringe was done per manufacturer’s guidelines. Our consulting pulmonologist also examined values, flow-time curves, and flow-volume loops for each pulmonary function test to judge the validity of the test.
The participants were initially divided in three groups: normal spirometry (NS; forced expiratory volume in 1 s/forced vital capacity > 0.70), obstructive (forced expiratory volume in 1 s/forced vital capacity < 0.70), and restrictive pattern (forced vital capacity < 80% predicted, forced expiratory volume in 1 s/forced vital capacity ⩾ 0.70; Fimognari et al., 2007). Because interpreting lung function tests with fixed cut points to determine whether a test is abnormal has been prone to some error (Miller, Quanjer, Swanney, Ruppel, & Enright, 2011), a consulting pulmonologist verified each finding. Also, because normative lung volumes for those beyond age 85 are not available, we further restricted the designation of RVP for our inferential testing to those deemed to have moderate or severe RVP by the pulmonologist.
Kyphosis
Thoracic kyphosis was isolated for measurement because it is the spinal abnormality common in older adults, especially those with osteoporotic vertebral fractures, and is associated with the stooped posture that we hypothesize restricts ventilation. The index of kyphosis was calculated by applying the formula, (h/l) × 100 to a paper tracing of a flexicurve measurement of the spine (Yanagawa, Maitland, Burgess, Young, & Hanley, 2000). Length (l) was determined by measuring in centimeters a straight line from the ruler position of C7 to T12 and height (h) was determined by drawing a line from the highest point in the thoracic curve to the point at which it intersected the straight line drawn from C7 to T12. All measures were taken by one research assistant trained by a physical therapist and checked for interrater reliabilities consistently above .85.
Muscle strength
Muscle strength was measured in pounds using a Jamar® Smart Hand digital dynamometer (Patterson Medical Inc., Warrenville, IL). Dynamometrically measured grip strength is a valid measure of muscle strength, correlating –.611 to –.843 with the Timed Up and Go (TUG) test in older adults (Schaubert & Bohannon, 2005) Test–retest reliability over 12 weeks revealed ICC coefficients of .954 and .912 for the left and right hands, respectively (Bohannon & Schaubert, 2005). In another study of women aged older than 75 years, it was found that handgrip strength measured by hand dynamometer correlated more highly with measures of upper body strength (r = .417 to .566) than lower body strength (r = .225 to .369; Tietjen-Smith et al., 2006). A percentage of predicted grip strength score was calculated using normative grip strength data for male and female adults based on 5-year age groupings from 55 to 59 to 90 to 99 (Bohannon, Bear-Lehman, Desrosiers, Massy-Westropp, & Mathiowetz, 2007; Bohannon, Peolsson, Massy-Westropp, Desrosiers, & Bear-Lehman, 2006).
Pain and dyspnea
Observational tools specifically developed for adults who cannot communicate symptoms were used. The five-item Pain Assessment in Advanced Dementia (PAIN-AD) Scale captures musculoskeletal pain with movement (Warden, Hurley, & Volicer, 2003). Possible scores range from 0 to 10 and cutoff scores reported for the tool are as follows: 1 to 3 = mild pain, 4 to 6 = moderate pain, 7 to 10 = severe pain. Internal consistency was .722 in this study. Construct validity is supported by correlations between the PAIN-AD and PAIN-Visual Analog Scale (VAS) during pleasant activity of .89 to .95 and of .82 to .91 during an unpleasant activity (Warden et al., 2003).
Dyspnea, which is defined as difficult breathing or shortness of breath, was measured with the eight-item Respiratory Distress Observation Scale (RDOS; Campbell, 2008; Campbell, Templin, & Walch, 2010). The possible range of scores are 0 to 16 with a score ⩾3 signifying dyspnea and respiratory comfort is signified by a score <3 (Campbell & Templin, 2015). Cronbach’s alpha internal consistency for the RDOS was .79 in this study. Interrater reliabilities were 1.0 and validity was supported by a correlation of .71 between the RDOS and verbal report by 210 cognitively intact individuals as well as a correlation of –.369 between oxygen saturation and RDOS scores (Campbell et al., 2010).
Other measures
BMI was calculated by dividing the persons’ weight in pounds by their height in inches squared, and then multiplying the results by a conversion factor of 703. A diagnosis of dementia was obtained from record review. Ambulation was measured from record review or staff report. Smoking history was collected by self-report and reported in pack years. Pack years was calculated by multiplying the number of packs of cigarettes smoked per day by the number of years the person has smoked.
Procedures
Following consent, the Mini–Mental Status Exam (MMSE) was administered and the resident record was reviewed for exclusion criteria, demographics, ambulation status, height, and weight. The PAIN-AD and RDOS scales were completed by observing the person midmorning on the testing day. Observations were done at least 20 min past any potentially stressful or discomforting activity in 5 min epochs for each tool. To obtain the angle of kyphosis, a flexicurve ruler was aligned to the spine from C7 to T12 with the person standing up as straight as possible. Nonambulatory residents were helped to a standing position by one or two nursing assistants. The curve of the ruler was traced onto paper.
Pulmonary function and grip strength testing were conducted with the resident seated in a comfortable chair and limbs not crossed. The procedures were explained and demonstrated by the research nurse prior to testing. Measures of grip strength were taken with the elbow flexed at 90°. The maximum reading after two grip strength attempts for the dominant hand was recorded. Spirometry testing of relaxed and forced lung function was conducted following the grip strength test. A nose clip was used and participants were taught to make a good mouth seal. The research nurse explained and demonstrated procedures prior to testing and continuously repeated instructions during testing. An initial relaxed vital capacity maneuver was done. Following a short rest period, the forced expiratory maneuver was done. The forced maneuver was repeated minimally two times, with rest periods between each test. Consistency of results were monitored by the research nurse using repeatability criteria recommended by the American Thoracic Society (Levy et al., 2009).
Analyses
Data were described with means, standard deviations, percentages, and ranges. Predictor variables that were significantly different between those with and without RVP using chi-square or t-test values at a .05 alpha level were entered into the logistic regression model to analyze the primary hypothesis. The RDOS scores were grouped into those with and without respiratory distress due to skew. The relationship between RVP and ambulatory status was examined with a chi-square and between RVP and pack years smoking with a Mann–Whitney U test. In addition, we describe the characteristics of participants with RVP stratified by diagnosis of dementia using frequencies and percentages.
Results
Of the initial consenting sample of 90, 23 people residing in skilled care with severe dementia were unable to follow directions to complete the pulmonary function testing, and an additional two were dropped from the study due to very poor spirometry output that, based on review by a pulmonologist, were not valid for reading. The resulting sample size was 65. The sample was primarily female (n = 47, 72%), White (n = 64, 98%), and had an average age of 87 (SD = 9.28, range = 57-100). The sample was highly educated with 63 (97%) having graduated high school, 49 (75%) attending college and 18 (28%) holding a graduate degree. While 28 participants had a diagnosis of dementia (37 had no dementia diagnosis), only 20 scored outside of the normal range on the MMSE (10 = mild, 6 = moderate, 4 = severe) and 25 scored in the normal range on the MMSE. The average length of stay was 40 months (SD = 35.17, range = 2-136). The three sites in which we collected data were all smoke-free buildings. Only two participants were current smokers (3%), 21 (32%) never smoked, and 42 (65%) were past smokers.
Description of Predictors
Twenty-eight participants (43%) had a diagnosis of dementia and the average BMI was 28.28 (SD = 8.61, range = 14.3-66.2). Muscle strength, as measured by grip strength, averaged 35.98 (SD = 16.59, range = 12.6-94.2). Twenty-two participants (36%) scored below 75% of predicted grip strength for age and gender, 23 scored between 75% to 100% of predicted, and 16 scored between 100% to 150% of normative grip strength predicted values.
More than half of the sample had dyspnea (n = 33, 51%) indicated by a score of 3 or higher on the RDOS (median = 3, range = 0-14). Pain was common, with 26 (40%) scoring in the mild pain range, 21 (32%) with moderate pain, and two (3%) having severe pain. Sixteen participants (25%) had no pain at the testing time.
The average index of kyphosis was 15.17 (SD = 4.52, range = 7.21-31.69). Using available normative values based on age and gender (Milne & Lauder, 1974), 11 participants (17%) had an angle greater than normal, 49 (75%) were considered normal, and five (8%) had an index of kyphosis that was less curved than normal.
RVPs and Factors Predicting RVP
RVPs were evident in 34 of the 65 participants (52%) and, according to our consulting pulmonologist, 33 (97%) of these were either moderate or severely restricted. Twenty-one participants (32%) had an obstructive ventilatory pattern and only 10 (15%) had normal spirometry readings.
Results of the logistic regression are presented in Table 1. Four people had missing grip strength tests, resulting in a sample size of 61 for the regression model. Two variables were not related to RVP (BMI, p = .452; musculoskeletal pain p = .857) and were not included in the model. The overall test for the model was statistically significant (χ2 = 22.023, p < .001). Seventy percent of cases were coded correctly and 69% of those with a moderate or severe RVP were predicted correctly. All betas were statistically significant. Those with RVP were more likely to have dementia, dyspnea, kyphosis, and lower muscle strength.
Logistic Regression Predicting Moderate or Severe Restrictive Ventilatory Pattern (n = 61).
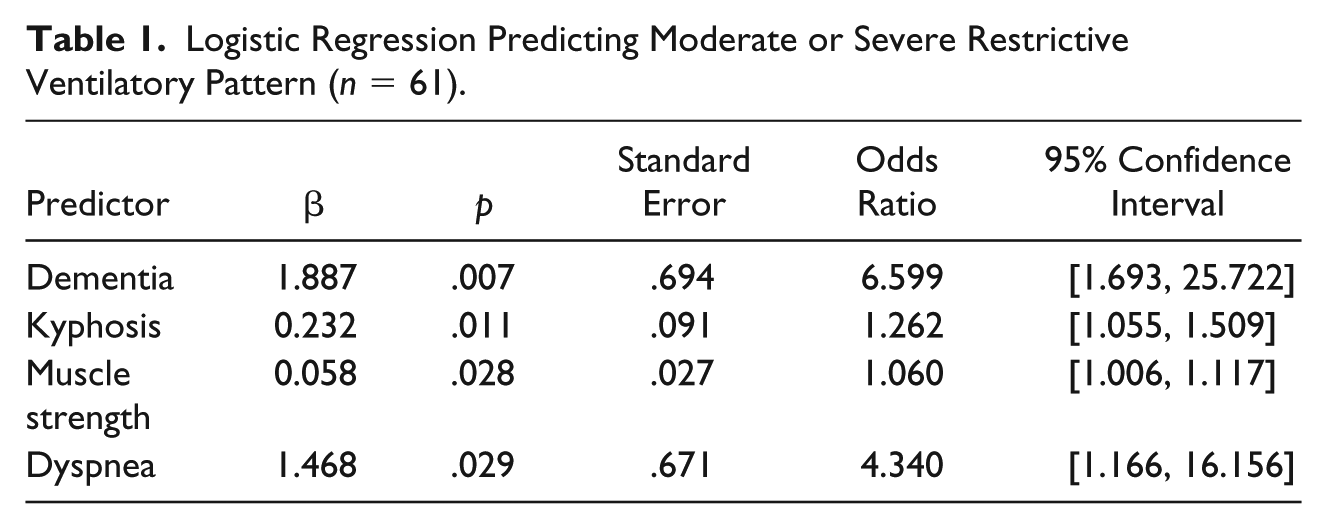
We explored the relationship between dyspnea and RVP further by examining correlations between continuous RDOS scores, forced vital capacity, and forced expiratory volume in the subsample of people with RVP. The significant negative correlations of forced vital capacity (r = –.518, p =.002) and forced expiratory volume (r = –.674, p < .001) with dyspnea indicate that participants who could exhale less had higher respiratory distress.
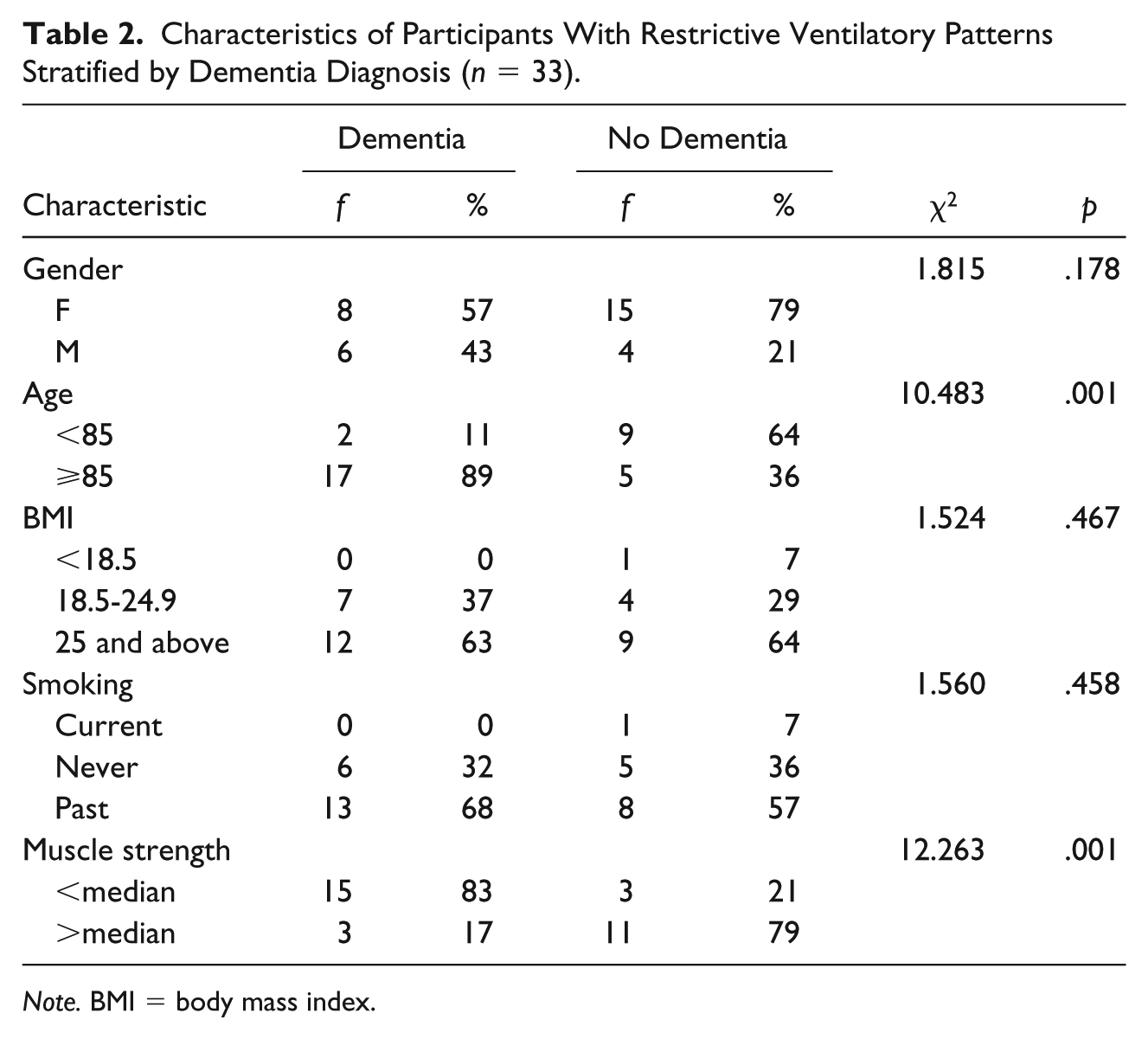
Table 2 presents characteristics of participants with RVP stratified by whether they had a diagnosis of dementia. Participants with RVP and dementia were more likely to be age 85 or older (χ2 = 10.483, p = .001) and to have less strength (χ2 = 12.263, p = .001) than those with RVP and no diagnosis of dementia.
Characteristics of Participants With Restrictive Ventilatory Patterns Stratified by Dementia Diagnosis (n = 33).
Note. BMI = body mass index.
RVP, Smoking, and Ambulatory Status
The median number of pack years smoked was 4.77, but the range was very large at 0 to 177. A Mann–Whitney U test revealed there was not a statistically significant difference (p = .971) in pack years smoked between those with and without RVP. As seen in Table 3, there was a statistically significant difference in ambulation between those with and without RVP. Eighty percent of the nonambulatory participants had RVP, and 38% of the ambulatory participants had RVP.
Prevalence of RVP Among Ambulatory and Nonambulatory Older Adults (N = 65).
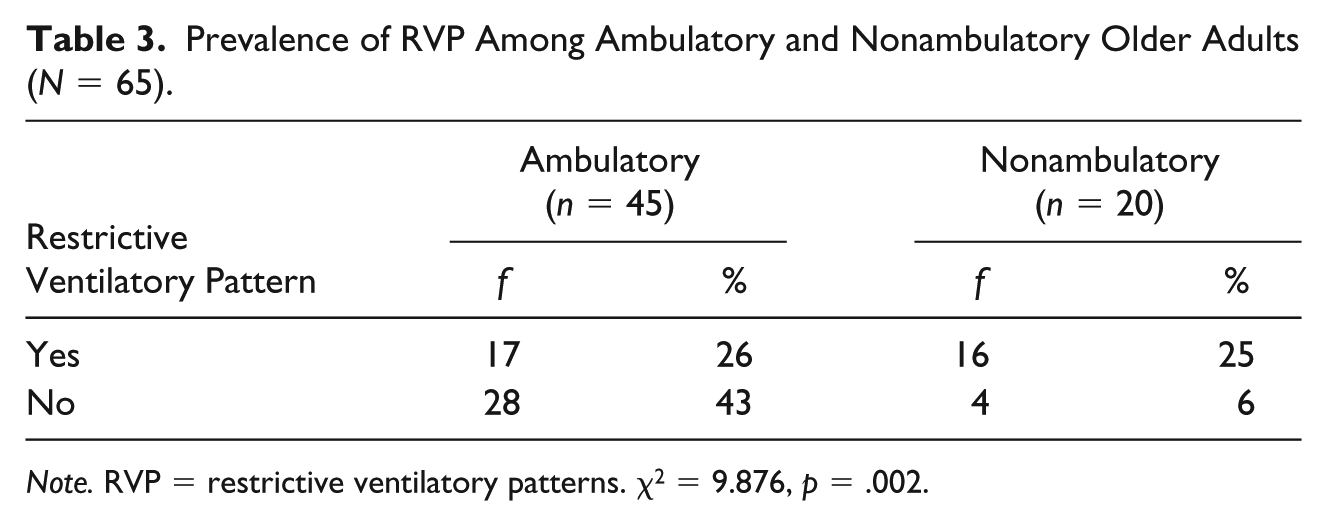
Note. RVP = restrictive ventilatory patterns. χ2 = 9.876, p = .002.
Discussion
This is the first study to describe RVPs in older adults residing in CCRCs. Our findings demonstrate that 52% had RVP, while one study found that 10.9% (n = 1,267) of community-dwelling older adults had RVP (Scarlata et al., 2008). All of the cases of RVP identified in our study did not have any known parenchymal lung disease associated with RVP. While the RVP we identified could be prodromal, early identification of RVP in relatively asymptomatic older adults may provide opportunities for preventive therapies or modification of identifiable risk factors.
The current study shows that CCRC residents with RVP were more likely to have dementia, lower muscle strength, kyphosis, dyspnea, and to be nonambulatory. Those with RVP and dementia were more likely to be older and to have less muscle strength than those with RVP and no diagnosis of dementia. To our knowledge, this has not previously been reported in older adults residing in CCRCs. A study of community-dwelling older adults showed that frail individuals are more likely to have airflow limitation and RVP compared with those who are not frail (Fragoso et al., 2012). Physical inactivity and functional impairment including slow gait speed and low grip strength were also associated with RVP among those who are frail (Fragoso et al., 2012; Mannino, Ford, & Redd, 2003). The finding that thoracic kyphosis was associated with RVP is consistent with other reports (Di Bari et al., 2004).
The relatively high correlations between dyspnea, forced vital capacity, and forced expiratory volume in those with RVP is consistent with research and clinical findings that RVP contributes directly to the development of dyspnea (Scarlata, Costanzo, Giua, Pedone, & Incalzi, 2012). People with RVP work harder to move air and therefore feel dyspneic. Finding the relationship in this sample adds credence to the significance of the problem of RVP in this population. Dyspnea is a highly uncomfortable symptom that affects quality of life and the ability to engage in activities of daily living (Arnold, Spertus, Jones, Xiao, & Cohen, 2009). If treating RVP improves symptoms of dyspnea, meaningful benefits for the individual in terms of quality of life and activities of daily living ability may be attained.
While our results are parallel to previous findings with community-dwelling older adult samples, we are not able to make temporal or causal assertions based on these correlational findings. The association between RVP and dementia, low muscle strength, kyphosis, and ambulation may be multidirectional and their combined effects may be particularly deleterious. Restrictive pulmonary dysfunction has been associated with mortality in community-dwelling older adults (Scarlata et al., 2008).
A multidirectional relationship has important clinical and therapeutic implications. Restrictive breathing has been identified as a biologically plausible and modifiable risk factor for the increased risk of pneumonia, decreased functional activity, and immobility (Jary et al., 2017; Kochi et al., 2018; Salhi, Troosters, Behaegel, Joos, & Derom, 2010). Strength training for the upper torso, scapular wall slides, postural taping (Greig, Bennell, Briggs, & Hodges, 2008), and spinal extension exercises (Ball, Cagle, Johnson, Lucasey, & Lukert, 2009) can be tailored and tested to determine their effect on improving posture, lung capacity, and respiratory function, and ultimately reduce RVP among CCRC residents. Consequently, an improvement in respiratory function may affect physical activity levels and improve quality of life (Renno, Granito, Driusso, Costa, & Oishi, 2005). Strategies that target CCRC residents and common conditions, such as sarcopenia and osteoporosis, could potentially improve lung volume, ventilatory patterns, and pulmonary function.
Dementia was the strongest predictor of RVP in this study, which raises the question of how future interventions might be tailored to those affected by mild and more advanced forms of dementia. A myriad of techniques have been effectively used by clinicians and researchers to adapt interventions for people with mild and more advanced dementia. For example, Jablonski and colleagues (2011) discuss the use of priming, cueing, pantomime, bridging, hand over hand, and a technique they call mirror-mirror to accomplish oral hygiene in those with advanced dementia.
Limitations of this study include a relatively small sample size and possible errors in self-report of prior smoking status. The final sample of 65 was less than the 69 calculated in our power analysis. The smaller sample size may have increased the width of confidence intervals by increasing the standard error. To obtain a larger sample size that can perform pulmonary function testing, multiple sites may be needed and those with severe dementia should be excluded. Our estimates of RVP in this population could have been influenced by at least two factors. Our sample had an average age of 87, but the spirometric reference values available to identify pulmonary function predicted values only include older adults up to age 85. As a fairly substantial number of people with dementia were unable to perform spirometry, it is likely we have underestimated RVP in those with dementia. The correlation of hand dynamometer readings with upper body strength reported by Tietjen-Smith et al. (2006) increases our confidence that this was a reasonable proxy for intercostal muscle strength, but measures of maximal inspiratory pressure would have provided a more direct measure of respiratory muscle strength. Maximal inspiratory pressure can be measured with a respiratory pressure meter and is the classic test of inspiratory muscle strength. A simpler way to perform the test is the sniff nasal inspiratory pressure (SNIP) test of inspiratory muscle strength. This test has reference values for adults, may be better tolerated by frail older adults, and is a very good measure of inspiratory muscle strength (Terzi, Corne, Mouadil, Lofaso, & Normand, 2010).
Future work should evaluate interventions that target muscle strength, kyphosis, and dyspnea to prevent or reduce RVP and its possible sequelae. Respiratory muscle exercises combined with back extensor muscle strengthening, for example, may be beneficial if adapted to the unique needs of older adults residing in CCRCs. Furthermore, it is important for future research to determine if respiratory muscle function predicts changes in exercise tolerance, morbidity, or mortality in general populations of older adults. In a study of 145 males with chronic heart failure, exercise limitation was associated with reversible lung restriction and inefficient ventilation at rest and during exercise. Reductions in respiratory muscle strength, assessed by measuring the maximal inspiratory pressure, was associated with poor patient outcomes (Meyer et al., 2000). Thus, respiratory muscle training to strengthen breathing muscles may have the potential to improve the tolerance for physical activity and ambulation, and decrease the development of lower respiratory infections, particularly among nonambulatory, frail, community-dwelling older adults with dementia.
In conclusion, viewing aging as a dynamic process focuses attention more keenly on maintaining functional abilities, physical endurance, and physical health with concomitant attention to preventive therapies. This study found a high prevalence of RVP in people residing in CCRCs and identified four factors that were associated with RVP. Understanding the factors associated with RVP can inform the development of interventions as well as matching interventions to particular older adults. Designing interventions to address modifiable factors associated with restrictive breathing patterns may prevent respiratory infections, help people to achieve better lung health, and increase physical activity tolerance.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Bader Philanthropies and the Jewish Home Foundation.
