Abstract
In 2016, the publication of the Society of Toxicologic Pathology’s (STP) “best practice” recommendations on determining and communicating adversity and the European Society of Toxicologic Pathology’s (ESTP) expert working group report on adversity were key milestones in addressing adversity determinations for nonclinical studies as translational tools for assessing human risk. Since then, many publications attest to the ongoing difficulty in adversity decision-making posed by unique context-specific challenges. The STP gathered input on current adversity practices from Society members via an open discussion at the 2024 STP Town Hall session (held at the STP 43rd Annual Symposium) and by a subsequent online survey. Most STP pathologists make adversity determinations by applying the STP and/or ESTP recommendations at their discretion. Adversity decisions are generally made for pivotal toxicity studies but occasionally may be assigned for other study types. Adversity determinations are difficult for certain organ systems (immune, reproductive, and endocrine) and product classes (eg, cell and gene therapies, proteins, and small molecules). Most pathologists assign adversity based on direct effects of the test article, but other factors (eg, secondary pharmacology, species relevance, adaptive responses) are also considered. Procedural adversity (eg, effects of administration/implantation) is a key factor in some circumstances.
This article is an opinion piece submitted to the Toxicologic Pathology Forum (TPF). This perspective is the view of the authors and does not represent an official position of the Society of Toxicologic Pathology (STP), British Society of Toxicological Pathology (BSTP), or European Society of Toxicologic Pathology (ESTP), nor should it be considered to reflect the opinions, policies, or positions of regulatory agencies (including the US FDA) or the authors’ employers/organizations. The Toxicologic Pathology Forum is designed to stimulate discussion of relevant issues in regulatory toxicologic pathology. Readers of Toxicologic Pathology are encouraged to send their thoughts on TPF opinion articles or ideas for new discussion topics to toxicologicpathologyforum@toxpath.org.
The 2024 Society of Toxicology (STP) Town Hall session of the STP 43rd Annual Symposium considered the topic “Adversity 2.0.” The Town Hall session focused on recent questions and trends with respect to determining and communicating adversity decisions for nonclinical toxicity studies. The session consisted of short presentations to frame the adversity discussion followed by panel members sharing their perspectives. Audience members responded to several poll questions to provide a collective summation of their own experiences, after which additional input from Society members was gained by polling the audience for additional perspectives and comments. This article briefly summarizes the main discussion points for this critical topic as deliberated during that Town Hall session and further explored by a post-meeting survey on the same issue.
Organization of the 2024 STP Town Hall Meeting
Dr Kathleen Funk served as moderator for the session. Drs Sabine Francke and Brad Bolon were chosen as panelists based on their prior experiences communicating the global toxicologic pathology community’s views on adversity decisions. Drs Francke and Bolon were coauthors of the 2016 STP Scientific and Regulatory Policy Committee’s (SRPC) “best practices” paper for determining, communicating, and using adverse effect data from nonclinical studies 10 (hereafter cited as Kerlin 2016); this policy paper incorporated feedback from the 2014 STP Town Hall session of the STP 33rd Annual Symposium, which considered adversity determinations as the Town Hall topic. Dr Francke was also a coauthor for the 2016 European Society of Toxicologic Pathology (ESTP) expert workshop paper on characterizing adversity for nonclinical studies 11 (henceforth cited as Palazzi 2016). These seminal 2016 papers on adversity were followed by numerous subsequent publications1-3,6-9,12-15 attempting to further hone various context-specific aspects of adversity decisions. This body of recent literature and ongoing international working groups of expert toxicologic pathologists tasked with considering organ-specific toxicity (including ocular and male and female reproductive systems) were kept in mind during the 2024 STP Town Hall audience discussion of context-specific factors that impact adversity decisions.
The initial presentation at the 2024 STP Town Hall session (Dr Funk) provided an overview of the Kerlin et al 10 and Palazzi et al 11 papers. Thereafter, panelists subsequently touched on regulatory considerations (lead by Dr Francke); organ-specific issues (Dr Bolon addressing unique considerations for neuropathology and Dr Renee Hukkanen speaking on reproductive endpoints); and product-specific parameters (Dr Hukkanen discussing anti-cancer chemotherapeutic agents and Dr Lyn Wancket describing medical devices). Dr Lila Ramaiah concluded by highlighting clinical pathology elements and their contribution to adversity decisions in a weight of evidence (WoE) approach.
Pathologist Perspectives Raised During the 2024 STP Town Hall Meeting
Several themes were explored during the 2024 STP Town Hall. These topics were addressed by the panelists or raised and discussed by audience members during the subsequent question-and-answer discussion.
Defining “Harm”
A main discussion point addressed the concept of “harm” as a primary factor in making adversity decisions. The topic was introduced because “harm” is the first parameter in the STP “best practices” paper used in assigning adversity. 10 Panelists and audience members acknowledged a lack of consistency regarding whether adversity is described for a specific test article (TA) dose versus identifying specific adverse changes and then noting which dose(s) induced unacceptable levels of those changes. Several audience members argued strongly against reporting the status of each finding as either adverse or nonadverse in a contributing scientist report. Others expressed concern that Study Directors may over-interpret or under-interpret “harm” (eg, changes were judged to be nonadverse if an animal was still able to walk around its enclosure) and advocated for developing a clearer definition of “harm” beyond the definitions provided in Kerlin et al 10 and Palazzi et al. 11 This clarification is needed because there are differences in the definitions of “harm” between the two 2016 papers, meaning that adversity decisions may differ depending on which definition is used as a rationale to support the adversity determination. Opinions were shared that “harm” must be based on more than a tissue change accorded a higher semi-quantitative severity grade, whereas others stated that taking the Kerlin 2016 information out of context might be counterproductive as it was the first paper to provide an approach on organizing adversity that considers “harm” occurring at one or many levels in an organism (ie, cell, tissue, organ, whole animal). Therefore, in considering the 2016 adversity “best practice” recommendations, the actual work of defining harm and its clear communication in the study report remains with the pathologist, as it is often study-specific and/or TA-specific.
The potential to consider some changes as “inherently adverse” (eg, necrosis of some non-regenerating tissues), as stated in Palazzi et al, 11 was discussed. Audience members noted that some inherently adverse findings may be interpreted as nonadverse (ie, biologically inconsequential) depending on such factors as their distribution (localized vs widespread), reproducibility among all animals within a dose group, and severity (minimal or mild vs moderate or greater). In general, findings attributed to a TA are more likely to be deemed adverse if they are widespread, consistently present, and of moderate or greater severity.
Adversity and the No Observed Adverse Effect Level
The translatability of adverse findings from nonclinical animal studies to predict human risk was another major theme of discussion. Adversity determinations are specific to the animal tested under the study conditions tested, and adversity in an animal study does not necessarily correlate well (or at all) to adversity in humans. It was noted that human susceptibility to administered TAs may change over time (eg, long-term cancer patients and their evolving response to anti-cancer chemotherapeutics). It was emphasized that a no observed adverse effect level (NOAEL) (or other threshold value) is set for the specific conditions tested in animals during a nonclinical toxicity study and that regulatory documents used for assessing human risk apply a WoE approach to nonclinical animal data (including NOAELs) developed across several studies at different TA doses administered to various test species. The recommendation from the 2016 STP “best practices” paper (Kerlin et al 10 ) that adversity of pathology findings should be communicated in the pathology report but that the NOAEL should be reported only in the main study report was reiterated. In addition, while most participants in the Town Hall focused on adversity decisions in pivotal Good Laboratory Practice (GLP)-compliant toxicity studies, it was noted that Kerlin 2016 does not preclude adversity determinations in other study types (eg, dose range-finding [DRF] studies).
Communication of Adversity
Integrated communication was identified as a critical factor when reporting adversity decisions for nonclinical studies. Ideally, this integration would include participation from all applicable experts, including pathologists (clinical and anatomic) and toxicologists, and consideration of all study endpoints when establishing a NOAEL. A concern was raised by audience members that pathologists may be more isolated from other study team members due to their remote work locations as well as the often-limited information sharing between organizations (eg, sponsor and contract research organization [CRO] employees). Several audience members emphasized that the inclusion of all relevant scientists was essential to successfully apply the STP “best practice” recommendations in Kerlin 2016 and shared concerns that toxicologists had had limited involvement in drafting these 2016 recommendations. Other audience members responded that toxicologists were part of both the STP and ESTP and that both the Kerlin 2016 and Palazzi 2016 manuscripts were provided to members of the authoring societies for review and comment prior to their respective publications; moreover, the American College of Toxicology (ACT) formally endorsed the adversity “best practices” in Kerlin 2016. 10 Still other audience members reiterated the perspective that training in whole animal physiology and disease patterns makes pathologists ideally suited for integrating and interpreting animal datasets when providing adversity decisions with respect to pathology findings.
Regulatory Review and Risk Assessment
The inclusion of a written rationale in the pathology/study report, outlining how adversity was assessed and determined, was emphasized as being of key importance to regulatory reviewers. It was noted that the data available to individual study pathologists are typically less than what are provided to reviewers. The access to more data sets (eg, transcriptomic data) for a given study may aid regulatory reviewers in determining potential human risk. In addition, even if such ancillary non-pathology data sets are made available during analysis, many pathologists have minimal or no training/experience with interpreting this information. While monitorability and reversibility of findings does not affect the adversity determination itself, 10 the ability to monitor findings in the clinic as well as their potential for eventual reversal can have a major impact on the risk assessment decision for humans.
Adversity Considerations for Specific Organ/Systems and Product Classes
Comments from several clinical pathologists suggested that relatively few clinical pathology findings may be considered adverse on their own. 14 Exceptions include large decreases in the numbers of erythrocytes, neutrophils, and platelets. 14 Even in cases where a clinical pathology alteration may be life-threatening (eg, electrolyte abnormalities), it is often difficult to differentiate a primary TA-related clinical pathology effect from a change secondary to moribundity. Moreover, some dramatic changes (eg, electrolyte levels) may have no obvious microscopic correlates to further inform the interpretation. Several clinical pathologists reiterated that adversity decisions based wholly or partly on clinical pathology data may accurately be summarized as “it depends.”
The idea that “adversity” may not be the most relevant question for some studies was discussed. One situation in which this question might arise involves exposure to specific therapeutic products. For example, most tissue responses for some TA classes are localized (eg, implanted medical devices, long-acting injectables) and to some degree are determined by the method/route of administration (eg, intrathecal) more than by attributes of the TA. Such responses may be more appropriately characterized by terms such as “local tolerability” or “procedural adversity.”4,5,15 Similarly, the potential impact of stress on lymphoid alterations and the relevance of immune responses in animal species that mount immunogenic attacks on TAs composed of foreign molecules (eg, nucleic acids and proteins) were mentioned as specific challenges in making adversity decisions for many immunomodulatory therapeutics.
Another situation in which “adversity” may not be the most relevant question depends on the affected organ system(s). For instance, changes in systems required for endurance of the species but not for survival of the individual animal (eg, findings in male and female reproductive organs) will require a nuanced rationale for assigning adversity given that “harm” in such cases does not apply to the affected animal but rather to the potential for perpetuation of the species. Similarly, the apparent lack of adversity in the nonclinical setting must be balanced with the absence of certain data points in conventional study designs. In this regard, it was noted that minimal to mild neuropathologic findings may not have detectable or biologically consequential electrophysiological effects or neurological deficits in nonclinical models (assuming that electrophysiological testing and neurological evaluations are performed), whereas in humans, the first evidence that structural findings might exist may be signaled by verbal reports of sensory symptoms from patients.
STP Assessments of Adversity Practices
Based on these discussion points from the 2024 STP Town Hall, two STP member surveys were organized to capture perspectives of practicing pathologists with respect to making adversity decisions. The first was conducted by a brief real-time poll for the approximately 200 attendees at the 2024 STP Town Hall session (of which 129 participated in the poll) that covered three benchmarking questions. Several months after the 2024 STP Town Hall session, a brief online survey encompassing the three benchmarking questions asked during the 2024 STP Town Hall session (adapted as questions 1 to 3 of the online survey) as well as five additional benchmarking queries were sent to the entire STP membership (approximately 1380 members of which 190 or 13.8% responded). (The final online survey is reproduced in Supplemental Table 1.) The 2025 STP online survey included an optional 13-question demographic section.
Three-Question 2024 STP Town Hall survey
Three benchmarking questions were asked during the 2024 STP Town Hall session to obtain a snapshot of current adversity decision practices by STP members. The first asked whether audience members relied on the STP “best practices” (Kerlin 2016), ESTP expert workshop (Palazzi 2016), both papers, or other resources in making adversity decisions. Many 2024 STP Town Hall participants (44.3%) used both Kerlin 2016 and Palazzi 2016 at their discretion in guiding their adversity decisions, while a lower percentage (17.4%) used neither publication.
The second 2024 STP Town Hall question involved situations in which audience members assigned adversity during their professional activities. Study pathologists often assign adversity in the reports that they author (32.8%), often in conjunction with other scientists involved in the study (28.1%). Some members (21.1%) contributed to adversity decisions in multiple ways such as incorporating and/or reviewing adversity decisions made by others.
The final 2024 STP Town Hall question assessed situations in which the audience members made adversity determinations. Most members determine adversity in pivotal GLP-compliant pivotal toxicity studies (44.2%), whereas a substantial fraction also assign adversity in other study types including DRF and tolerability studies (28.7%).
Multi-question 2025 STP Online Survey
A total of 190 complete or partial responses were received to the 2025 STP online survey.
Of the 188 respondents who answered the question about their level of work experience, most respondents had been practicing for 20 to 25 years. All respondents identified themselves as toxicologic pathologists, whereas about 68% also identified as a diagnostic pathologist. More than 80% had formal pathology training obtained during a residency, roughly 65% had a PhD or equivalent degree, and 25% possessed a master’s degree or equivalent (Figure 1). Most respondents (85%) were certified in anatomic pathology. The most common credential was certification as a diplomate of the American College of Veterinary Pathologists (DACVP; Figure 2A), although some respondents also held other pathology and toxicology certifications (Figure 2A, 2B). Some individuals held multiple certifications (data not shown). Most respondents were either employed in North America (31%) or were members of multinational institutions (63%). Europe (4%) and Asia/India (1%) were also represented. Respondents were most often employed by biopharmaceutical companies and CROs (Figure 3). The most common employer sizes were 501 to 5000 employees followed by less than 50 employees (Figure 4).
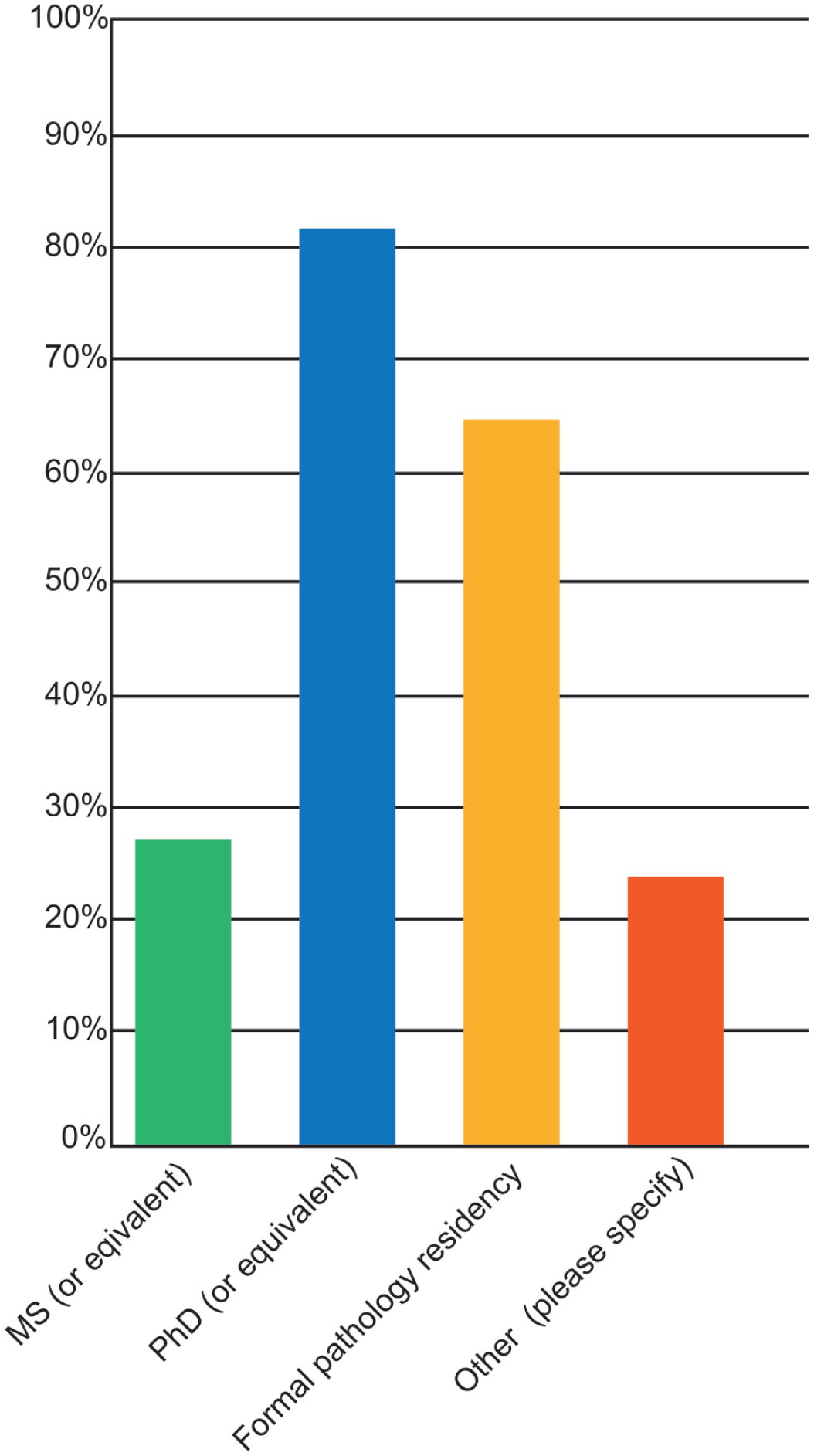
Pathology credentials held by respondents (N = 190) to the 2025 STP online survey on adversity practices.
Pathology certification types held by respondents to the 2025 STP online survey on adversity practices. A. Certification based on examination B. Certification based on credential review.
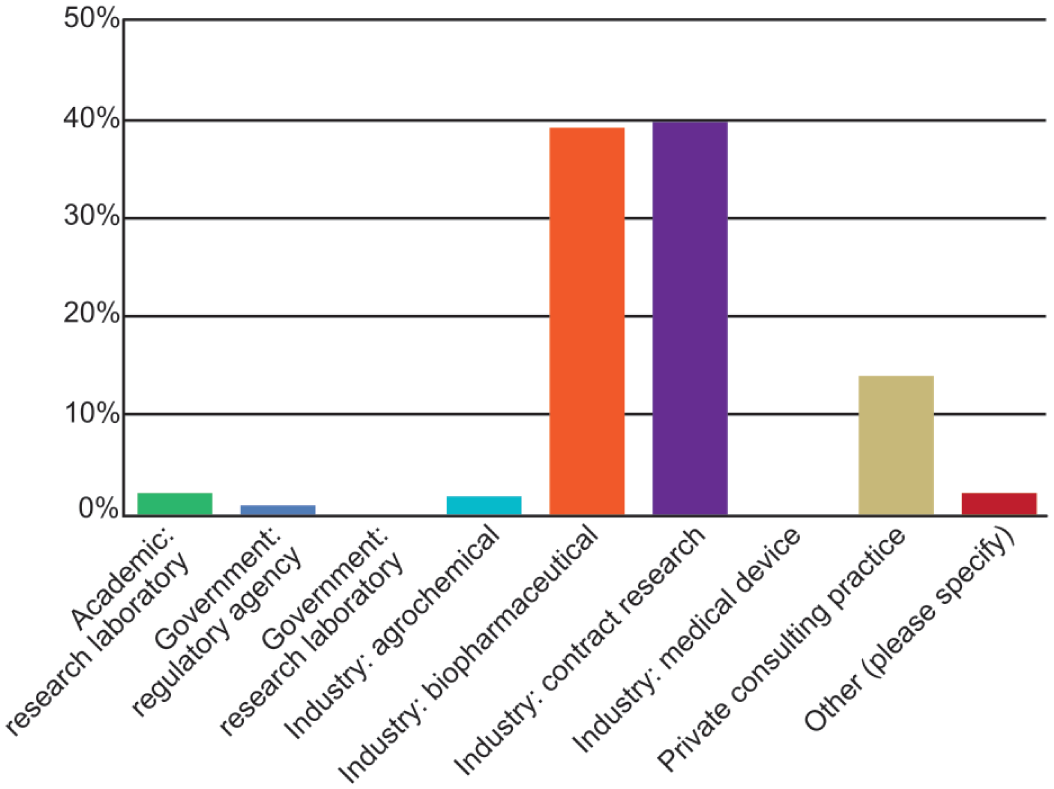
Types of institutions employing respondents to the 2025 STP online survey on adversity practices.
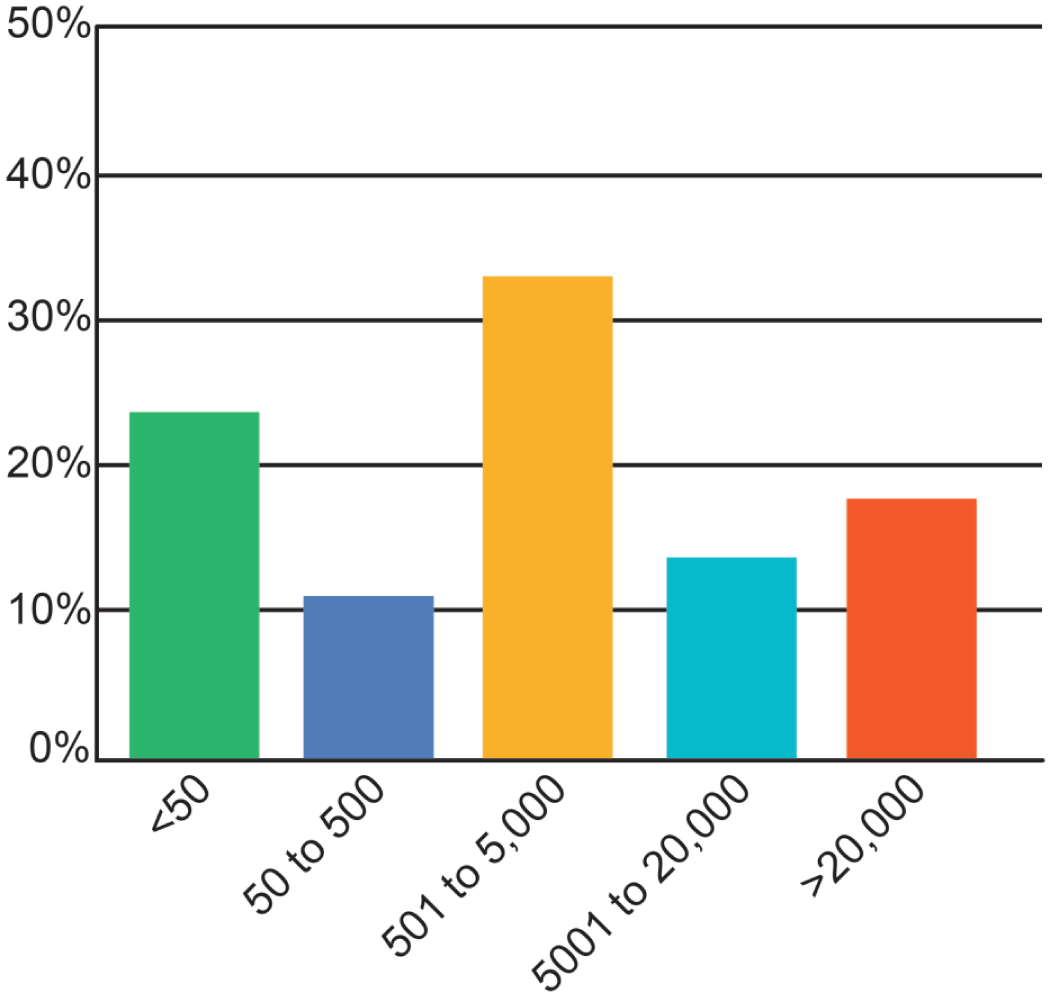
Sizes of institutions employing respondents to the 2025 STP online survey on adversity practices.
The vast majority (96%) of respondents were involved in adversity determinations as part of their jobs. Of those involved in adversity decisions, most (70%) use both Kerlin 2016 and Palazzi 2016, at their discretion, to help make their decisions. Fewer than 20% of respondents used Kerlin 2016 alone, ~5% relied on Palazzi 2016 alone, and the remainder used neither publication to guide their adversity decisions, instead relying on other resources.
The role of respondents in making adversity decisions varied considerably (Figure 5). Roughly one-third of respondents integrate adversity into pathology reports that they author, approximately 20% assign adversity in conjunction with the Study Director and clinical and/or anatomic pathologist, and 40% indicated they are involved in multiple steps; slightly less than 5% of respondents assign adversity as either sponsor or regulator. A small percentage (<3%) stated that their participation included involvement in authoring clinical (Investigator’s Brochure [IB]) and/or regulatory (Clinical Trial Application [CTA] and Investigational New Drug [IND]) documents as part of translational safety assessment. One response stated that it was not their standard practice to include adversity statements in the pathology report but that the Study Director was advised regarding their adversity concerns so that the information could be added to the main study report.
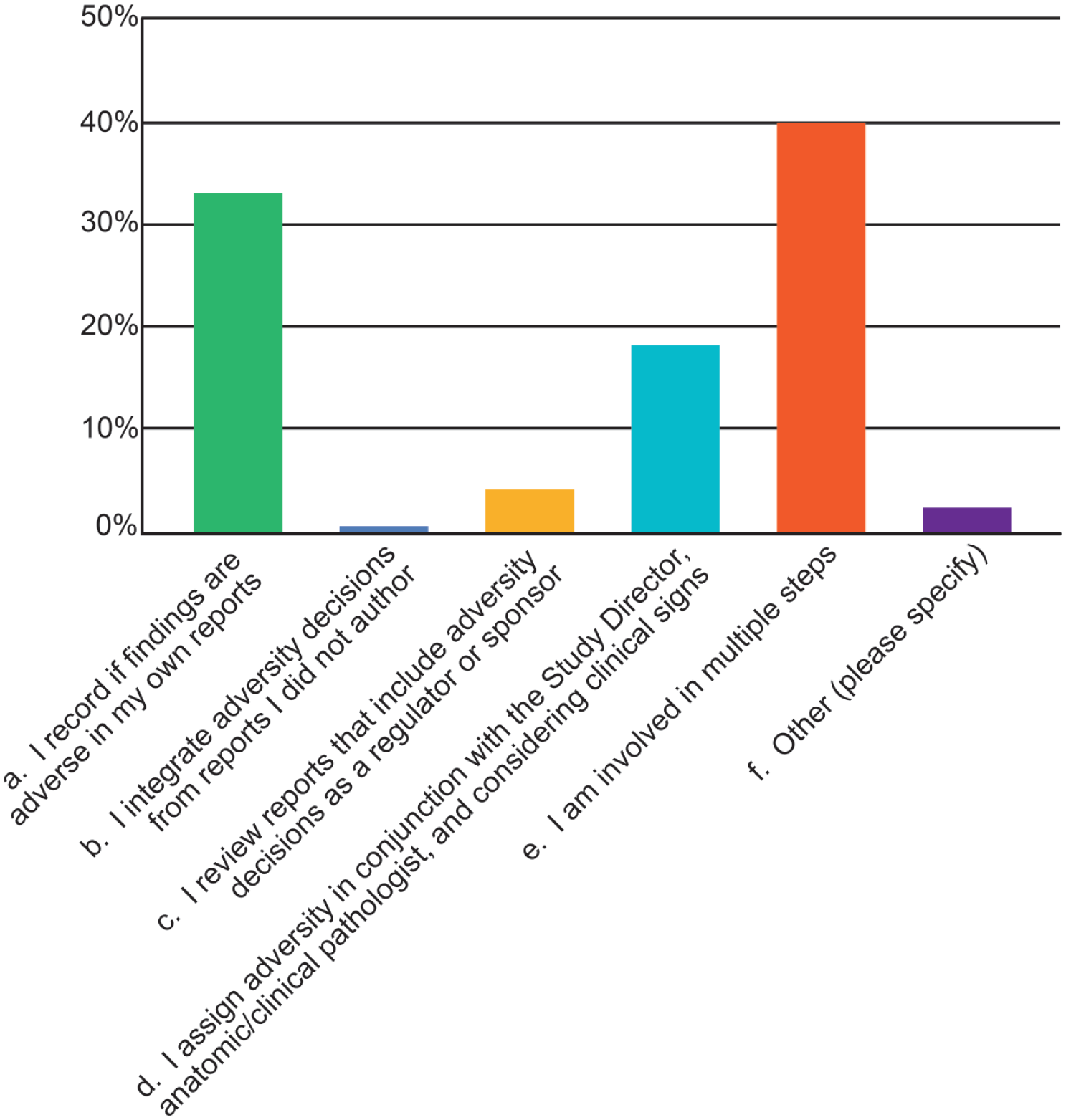
Steps in the adversity determination process fulfilled by respondents to the 2025 STP online survey on adversity practices.
Roughly 87% of respondents answered the question regarding the types of studies where they incorporated an adversity assessment. Most respondents (>86%) assign adversity in pivotal GLP toxicity studies, with such determinations being included in DRF studies much less often (second place, at 3.5%); adversity was assessed for tolerability and efficacy studies at 1% or less of the time. Approximately 8% of respondents added comments, including that the NOAEL was designated for pivotal GLP and non-GLP toxicity studies intended for submission; that maximum tolerated dose (MTD) and DRF studies ranging from 1 week to 9 months were primarily involved in adversity setting; and that most study types, except carcinogenicity studies, included adversity determinations.
Survey respondents were asked to identify up to three organ systems and three TA types where they encountered the most challenges when assessing adversity. The three most commonly identified systems were immune (55%), reproductive (48%), and endocrine (42%). Other systems included nervous (24%), hematopoietic (22%), hepatic (13%), ocular, (11%), cardiac (9%), and renal (8%), with a few respondents mentioning skin. Comments indicated that difficulties in adversity determinations depend on the nature of the findings and not on the types of affected organs or tissues. Moreover, a whole-body response sometimes needed to be considered and was occasionally critical in assessing adversity when selecting the starting dose for single dose clinical trials.
The TA types with which pathologists struggled most often were cell and gene therapies (including gene editing, 57%), proteins (51%), small molecules (42%), and nucleic acids (30%) (Figure 6). Medical devices and vaccines both ranked near 14%, though this apparent steep decline may reflect the relatively few pathologists who evaluate such TAs. Fewer respondents expressed concerns about making adversity decisions for “large molecules,” antibody-drug conjugates, inhaled products, chemicals, and agrochemicals. Many comments reiterated that adversity depends on the nature of the findings rather than the therapeutic modality. One respondent indicated that small molecules were the most idiosyncratic versus the platform-associated toxicities (eg, antisense oligonucleotides and adeno-associated viral [AAV] gene therapy vectors). Other concerns were the challenge of assessing adversity in underpowered studies (eg, non-rodent studies with small group sizes) and the lack of information about TA and biologics attributes from sponsor organizations.
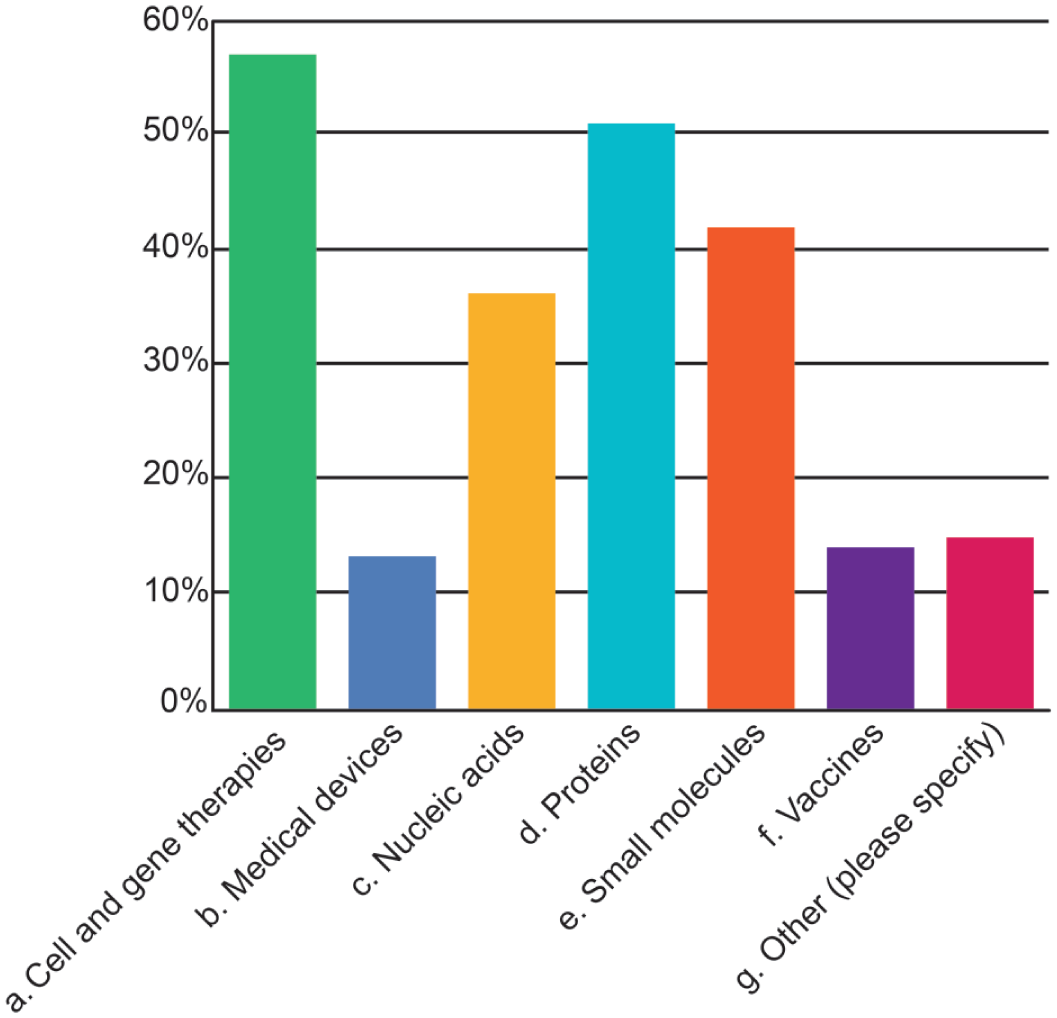
Types of test articles where adversity determinations are most difficult to categorize as identified in the 2025 STP online survey on adversity practices.
Participants were then asked which factors they considered when making an adversity assessment. Responders could choose up to five options including a write-in choice. The vast majority (96%) of respondents indicated that they considered direct effects of the TA in assigning adversity. Similar percentages of respondents selected secondary or indirect pharmacology, including species relevance (83%) and adaptive effects to TA pharmacology (77%) (Figure 7). Procedural effects were considered in assessing adversity 42% of the time. Numerous write-in comments provided additional perspectives. For example, some respondents evaluate the relationship between lesion severity/extent and imputed functional alterations as well as species-specific findings that could be adverse (even if not relevant for human risk assessment) but did not assign adversity to single histopathologic findings if there were no consistent consequences on organ systems or the animal’s well-being. Other participants added that additional data types such as qPCR (quantitative polymerase chain reaction) and TCR (tissue cross-reactivity assays) as well as any other in-life or postmortem findings should also be considered when making adversity decisions. Finally, some respondents stated that translational relevance to humans was not a consideration for determining adversity, although a separate comment regarding possible human relevance may be mentioned to communicate risk or add detail around potential impacts on reproductive capacity
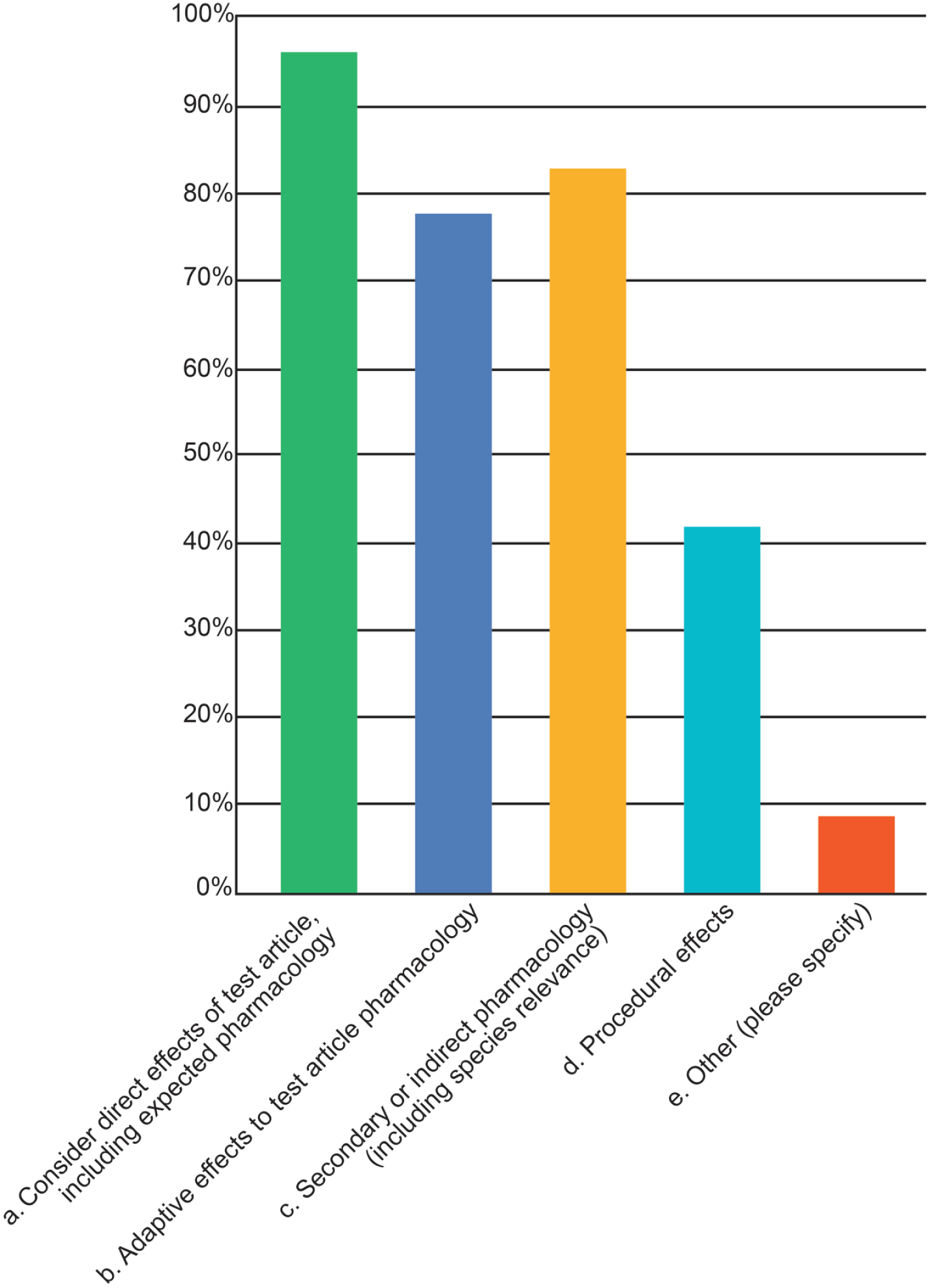
Common factors considered in determining adversity as identified in the 2025 STP online survey on adversity practices.
Most respondents (71%) indicated that adversity was still assigned in cases where there were no direct effects (on target toxicity, including expected pharmacology) but adaptive, secondary, and/or procedural effects were observed. The balance of respondents (30%) stated that adversity decisions relied on the direct TA-related effects.
Conclusions
The decade since the 2016 publication of the two initial pathology-oriented, globally recognized papers on adversity determinations has seen a wealth of debate regarding acceptable approaches to defining and communicating adversity in nonclinical animal toxicity studies. The perspectives gleaned on adversity determination practices from the 2024 STP Town Hall session of the STP 43rd Annual Symposium and the 2025 STP online survey, as summarized herein, indicate that the seminal Kerlin 2016 and Palazzi 2016 papers have provided substantial but not all-inclusive guidelines for making adversity decisions. As the information summarized in this document clearly shows, the 2016 manuscripts by Kerlin and Palazzi have made a notable impact on the profession, providing helpful early concepts of categorizing and standardizing approaches in adversity determinations. However, ongoing challenges with pathology adversity assessments in specialized fields such as immunology, reproductive toxicology, and medical device pathology,1-3,6-9,10-15 among others, have shown that the 2016 STP “best practice” recommendations and the 2016 ESTP expert workshop proceedings are a “place in time” compilation of the then-current thinking of the pathology profession, and were not intended as a “one and done” permanent fix. The outcome of the STP “Adversity 2.0” Town Hall meeting clearly shows that periodic updates of “best practices” for determining adversity are warranted as the field of toxicologic pathology continues to evolve.1-15
Supplemental Material
sj-docx-1-tpx-10.1177_01926233251395000 – Supplemental material for Toxicologic Pathology Forum*: Summary of the 2024 Society of Toxicologic Pathology Town Hall and 2025 STP Member Survey on Determining and Communicating Adversity
Supplemental material, sj-docx-1-tpx-10.1177_01926233251395000 for Toxicologic Pathology Forum*: Summary of the 2024 Society of Toxicologic Pathology Town Hall and 2025 STP Member Survey on Determining and Communicating Adversity by Kathleen A. Funk, Lyn M. Wancket, Brad Bolon, Sabine Francke, Renee R. Hukkanen and Lila Ramaiah in Toxicologic Pathology
Supplemental Material
sj-docx-2-tpx-10.1177_01926233251395000 – Supplemental material for Toxicologic Pathology Forum*: Summary of the 2024 Society of Toxicologic Pathology Town Hall and 2025 STP Member Survey on Determining and Communicating Adversity
Supplemental material, sj-docx-2-tpx-10.1177_01926233251395000 for Toxicologic Pathology Forum*: Summary of the 2024 Society of Toxicologic Pathology Town Hall and 2025 STP Member Survey on Determining and Communicating Adversity by Kathleen A. Funk, Lyn M. Wancket, Brad Bolon, Sabine Francke, Renee R. Hukkanen and Lila Ramaiah in Toxicologic Pathology
Footnotes
Author Contributions
Authors contributed to Writing—original draft (KAF, LMW, BB, SF, RRH, LR); Writing—review & editing (KAF, LMW, BB, SF, RRH, LR); Conceptualization (KAF, LMW, BB, SF, RRH, LR); and Resources (KAF, LMW, BB, SF, RRH, LR).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Brad Bolon is a senior advisor to the Editor of Toxicologic Pathology and publishes regularly in the journal but did not take part in the peer review or decision-making process for this submission. The other authors express no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
