Abstract
Adeno-associated virus (AAV) gene therapy vectors are an accepted platform for treating severe neurological diseases. Test article (TA)-related and procedure-related neuropathological effects following administration of AAV-based vectors are observed in the central nervous system (CNS) and peripheral nervous system (PNS). Leukocyte accumulation (mononuclear cell infiltration > inflammation) may occur in brain, spinal cord, spinal nerve roots (SNRs), sensory and autonomic ganglia, and rarely nerves. Leukocyte accumulation may be associated with neuron necrosis (sensory ganglia > CNS) and/or glial changes (microgliosis and/or astrocytosis in the CNS, increased satellite glial cellularity in ganglia and/or Schwann cellularity in nerves). Axonal degeneration secondary to neuronal injury may occur in the SNR (dorsal > ventral), spinal cord (dorsal and occasionally lateral funiculi), and brainstem centrally and in nerves peripherally. Patterns of AAV-associated microscopic findings in the CNS and PNS differ for TAs administered into brain parenchyma (where tissue at the injection site is affected most) versus TAs delivered into cerebrospinal fluid (CSF) or systemically (which primarily impacts sensory ganglion neurons and their processes in SNR and spinal cord). Changes related to the TA and procedure may overlap. While often interpreted as adverse, AAV-associated neuronal necrosis and axonal degeneration of limited severity generally do not preclude clinical testing.
Neurological diseases afflict millions of people around the world, striking at any time from before birth to old age. The intricacies of neural circuitry and physiology make pharmaceutical development for such conditions quite complex, and until recently few companies possessed the patience and resources to work consistently in this space. The recent pivot toward personalized medicine has resulted in the advent of many new modalities for treating neural diseases, such as biologics (nucleic acids and proteins), cell and gene therapies (also including gene editing), and medical devices (e.g., electrostimulation). Innovative treatments offer substantial promise for specifically targeted intervention to delay, ameliorate, or even reverse neurological diseases.
Adeno-associated virus (AAV)-based gene therapy vectors have demonstrated considerable success as therapies for various neurological diseases. 39 In brief, AAV vectors are used to deliver therapeutic transgenes into cells, thus restoring gene activity sufficiently to improve the patient’s clinical signs and symptoms. For neurological diseases, AAV therapies are typically delivered centrally by either intraparenchymal (IPa) infusion into the brain at one or more often several sites or by delivery into the cerebrospinal fluid (CSF) by one of several intrathecal (IT) routes: intracerebroventricular (ICV), into one or both lateral ventricles; intracisternal into the cisterna magna (ICM); or intracisternal into the lumbar cistern (ICL). 62 Administration of AAV vectors is associated with a constellation of neuropathological findings in the central nervous system (CNS) and peripheral nervous system (PNS), although the pattern and severities of the changes differ depending on many factors. Transduction of CNS domains may be observed when AAV-based vectors are delivered by other routes if the injection site connects directly to the circulatory system (e.g., intravenous [IV] 62 ) or to the CNS (e.g., intracochlear [ear],1,56 intranasal [nose],4,23 subretinal [eye]10,25), so AAV-related findings may develop if vectors are delivered by these indirect routes as well.
This article is based on a talk entitled “Nervous System Pathology for Adeno-Associated Virus (AAV)-Based Gene Therapy Products,” which was delivered during a half-day scientific session on “AAV-Based Therapies” offered during the annual symposium of the Society of Toxicologic Pathology (STP) in Baltimore, MD on June 17, 2024. The goals of this article are to review our collective understanding of AAV-related neuropathological findings in the CNS and PNS, and to contemplate how to distinguish test article (TA)-related findings from overlapping trauma-related effects associated with the delivery procedure.
Neuropathological Findings Associated With AAV Vectors
Our collective experiences as well as a plethora of recent publications have defined a general pattern of AAV-related pathological findings affecting the CNS and PNS. This pattern may be interpreted as a “class effect” related to shared attributes of AAV delivery systems, a concept analogous to the “class effects” defined for groups of therapeutic TAs that share such properties as molecular (chemical) similarities or related pharmacological actions. Importantly, this AAV class effect may not be induced by the AAV vector (e.g., capsid proteins) but instead may reflect the efficiency of AAV vectors in facilitating transgene transduction, where high levels of transgene expression have been implicated as a principal mechanism of toxicity to cells.14,30,45,55 However, the AAV pattern depends on many parameters. Among these, principal variables include the AAV dose, delivery approach, injection variables, route of administration (RoA), test species, and vector attributes. This section defines the typical pattern of AAV-related neuropathological findings and describes how the basic spectrum of changes is altered by these characteristics. In general, AAV-related findings relate to primary parenchymal injury (neuronal degeneration and/or necrosis) with secondary responses reflecting nerve fiber (axonal) degeneration, leukocyte accumulation, and/or glial reactions. The character of these changes differs depending on whether they develop in the CNS or PNS.
Microscopic Findings in the PNS
Ganglionic neurons are well-recognized as a target for the “neurotoxicity” induced by AAV-based vectors,5,6,22,29,34,45,46,55 and as such represent the main class effects for this category of TAs. In our experience, this class effect occurs to some degree (often but not always modest) during nonclinical studies at clinically relevant doses. For PNS-localized effects, active neuron injury manifests in rats and nonhuman primates (NHPs) as early as 8 days after IT (ICL) or IV delivery (based on rising blood levels of neural biomarkers, such as neurofilament light chain [NF-L] 22 ) and peaks at approximately 30 to 45 days after administration. The degree of tissue destruction (neuroaxonal degeneration/necrosis and active inflammation) falls with time after exposure (as early as 3 months but mainly at 6 or more months).16,17,29,45
For PNS-localized effects, the pattern reflects primary injury to neurons. Findings related to PNS tissue injury are commonly observed in dorsal root ganglia (DRG) and trigeminal ganglia (TG), which are readily collected at necropsy, as well as other somatic ganglia (e.g., spiral ganglia [ear]) and autonomic ganglia (e.g., ciliary ganglia [near the eye] 10 ) that are sampled only in special circumstances or when incidentally collected in sampling other protocol-specified tissues. Secondary effects occur in CNS and PNS sites where neurites (neuronal processes [axons and dendrites]) attached to ganglionic neurons are found, including nerve trunks peripherally and spinal nerve roots (SNRs) (dorsal > ventral) and the spinal cord centrally.
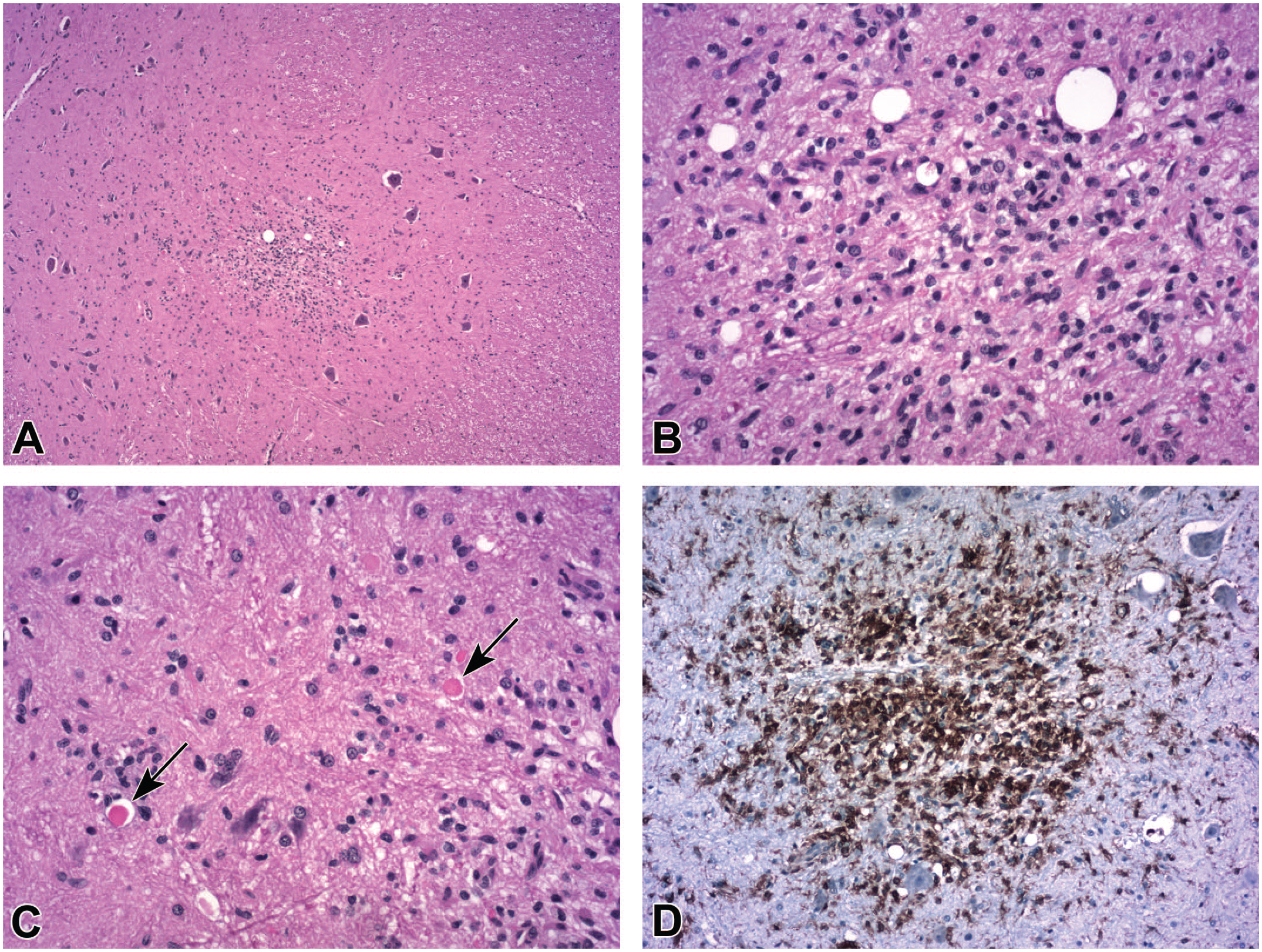
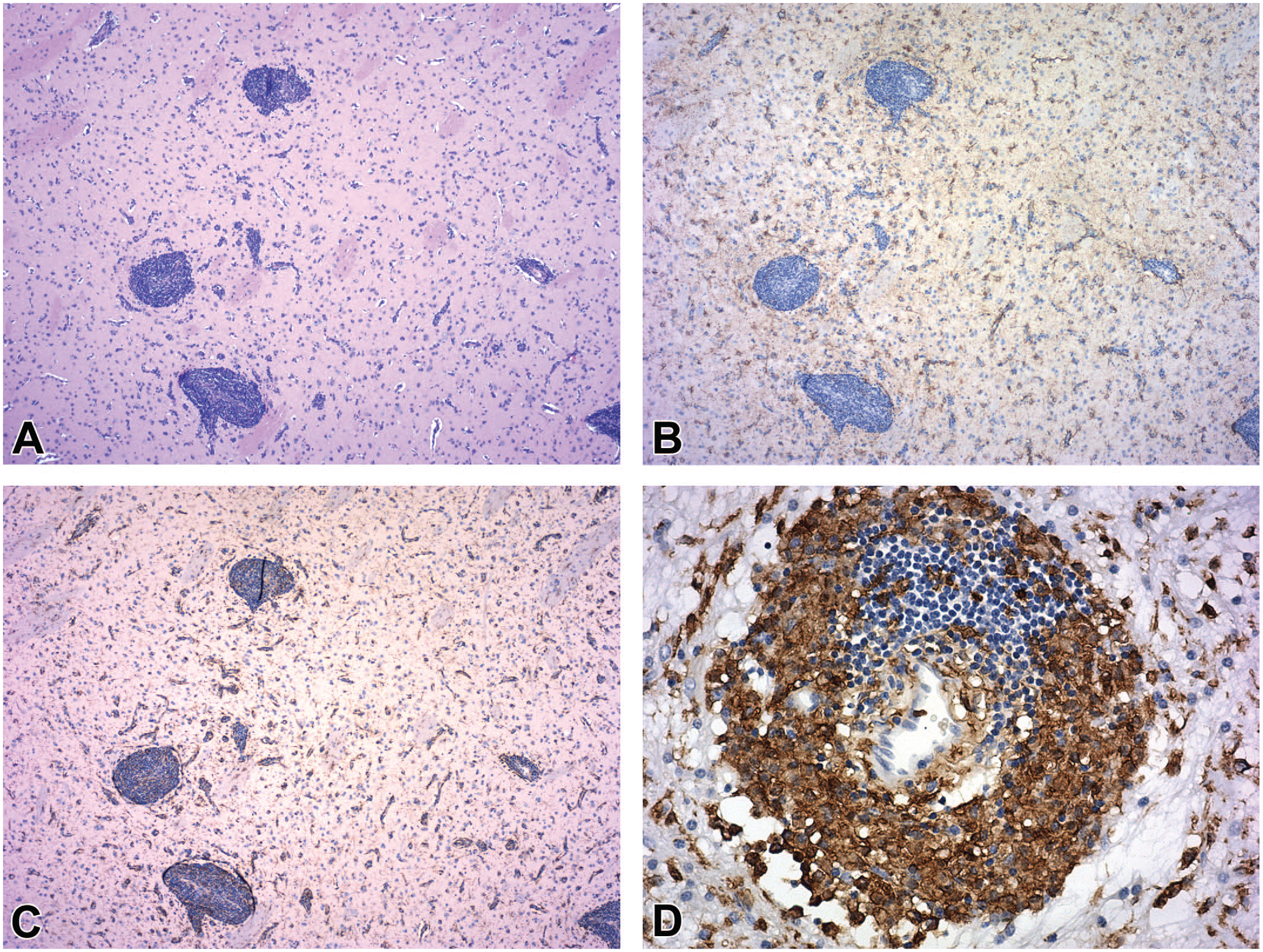
In somatic sensory and autonomic ganglia, the principal findings are neuronal degeneration and necrosis, nerve fiber degeneration (reflecting axonal changes secondary to neuron injury), increased satellite glial cellularity (a secondary glial response in ganglia in response to primary neuron damage), and leukocyte (typically mononuclear cells [lymphocytes and macrophages in variable proportions] but rarely mixed cells [i.e., mononuclear cells with neutrophils and less often eosinophils]) infiltration and/or inflammation ( Figure 1 ). 6 The effects on ganglionic neurons are generally more severe in lumbar and sacral DRG, followed by cervical DRG and TG.29,41,54,61 The AAV-related neuronal necrosis in ganglia, and rarely in the CNS, may lead to depletion of neuronal numbers (International Harmonization of Nomenclature and Diagnostic Criteria for Lesions [INHAND] diagnosis: “reduced cellularity, neuronal”; Figure 1B ).
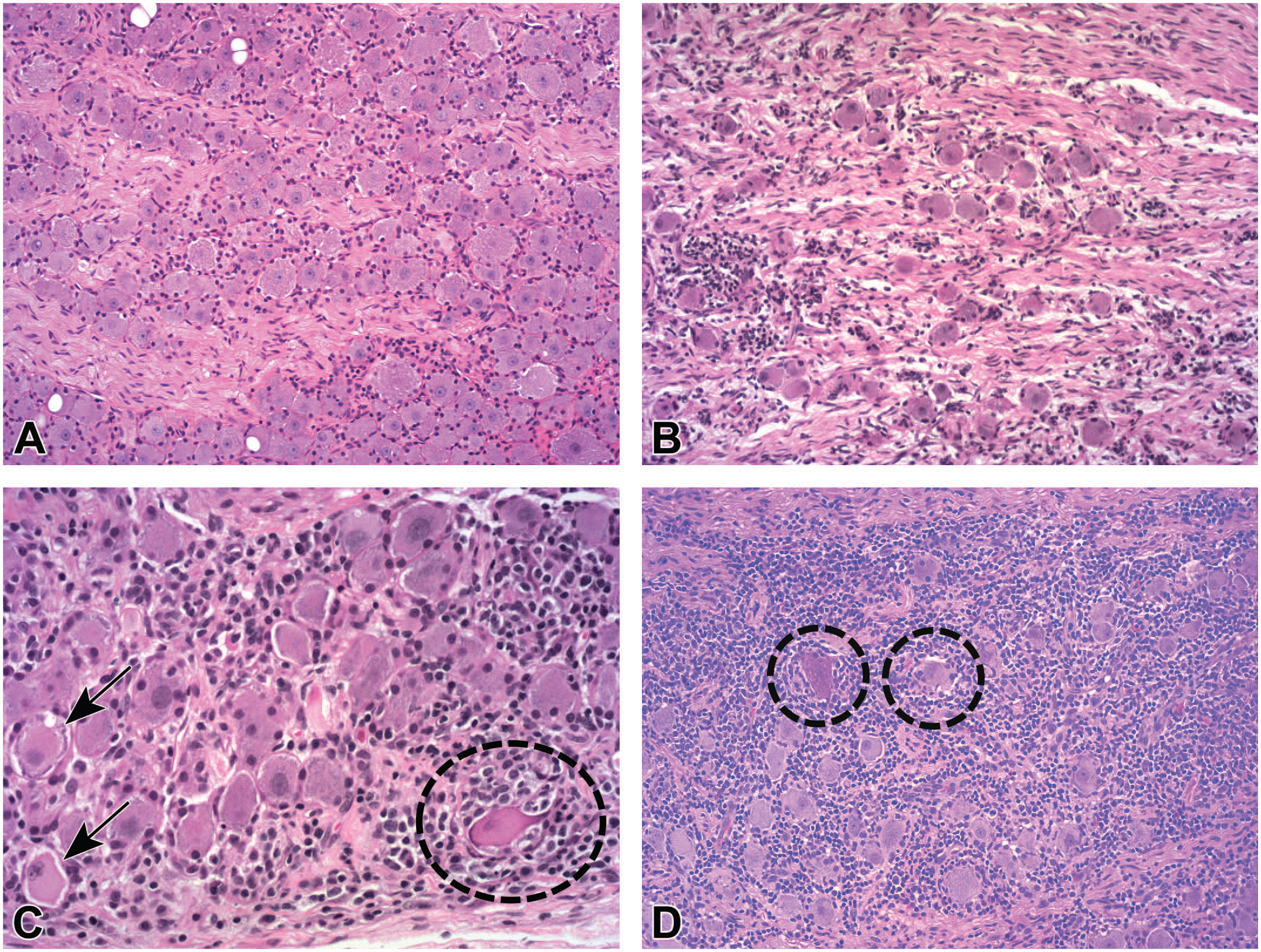
Representative characteristics of dorsal root ganglion (DRG) findings after administration of an adeno-associated virus (AAV) vector.
The main change affecting neurites is nerve fiber degeneration. This finding may be observed in several sites. First, nerve fiber degeneration may be seen in somatic nerve trunks ( Figure 2 ), where the affected processes are chiefly extensions of AAV-transduced DRG sensory neurons (perhaps with a smattering of axons originating from transduced ventral horn motor neurons of the spinal cord). Similar findings may be evident in autonomic nerves (if such samples are available for evaluation) due to AAV transduction of autonomic neurons, but in our experience effects at these autonomic sites are uncommon compared with changes in somatic PNS elements; this divergence might reflect the fact that autonomic ganglia are infrequently included in the list of protocol-specified tissues compared with DRG and TG. The glial response in nerves (seen with relative ease, albeit occasionally, in thickly myelinated, relatively acellular somatic nerves) is characterized by enhanced numbers and/or sizes of Schwann cells near affected nerve fibers (INHAND diagnosis: “increased cellularity, Schwann cell”). Other findings 46 are rare in nerves and may be diagnosed when present using standard INHAND terminology.11,19,35
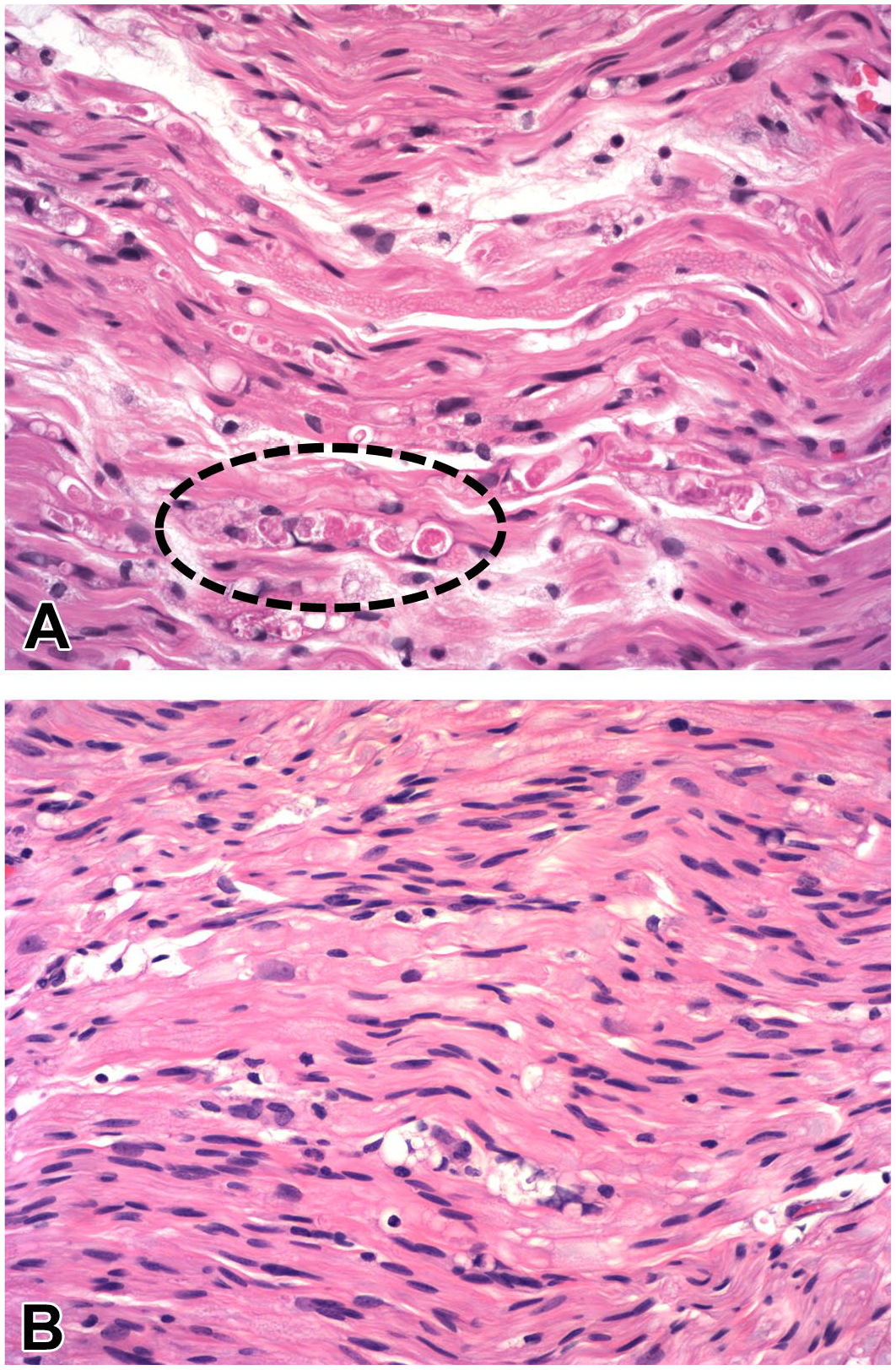
Representative attributes of nerve fiber degeneration in the sciatic nerve after intrathecal administration of an adeno-associated virus (AAV) vector.
Nerve fiber degeneration (affecting axons from ganglionic neurons) is also evident in SNRs (part of the PNS) and in certain white matter tracts of the spinal cord (i.e., CNS domains that support PNS sensory function [
Figures 3A
and
Representative features of axonal degeneration in the spinal cord white matter, a common finding after intrathecal injection of an adeno-associated virus (AAV) vector.
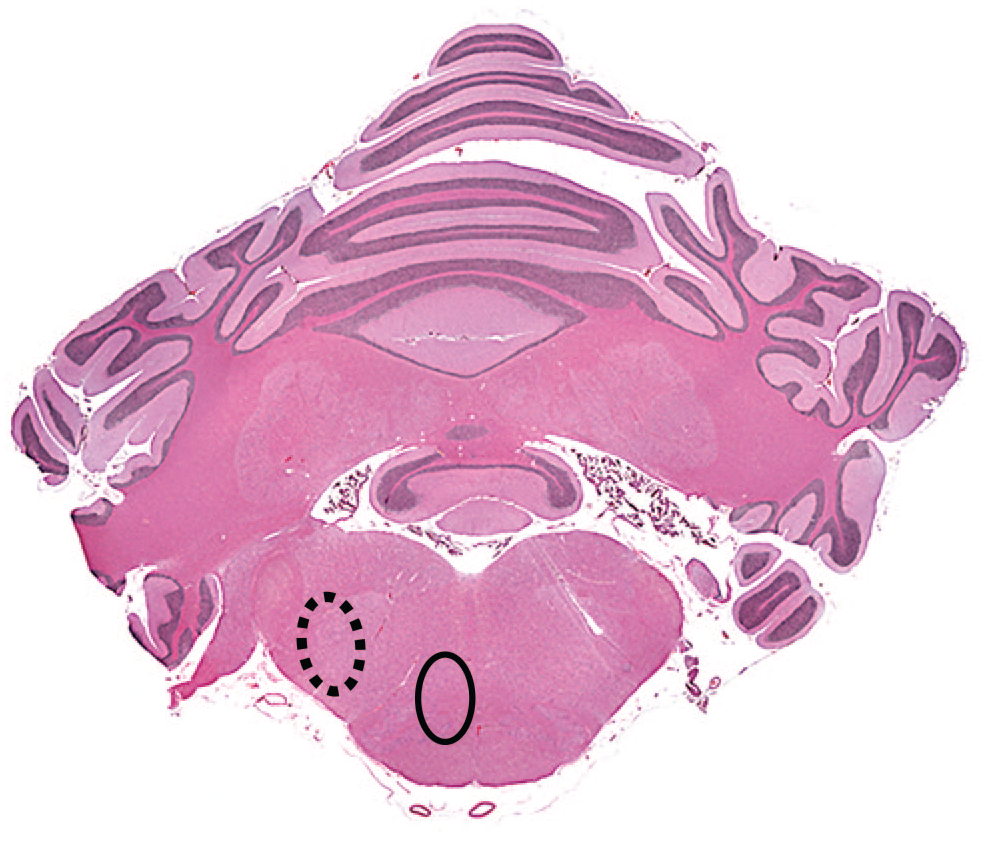
Cross-section of the hindbrain showing the approximate locations of fiber tracts that carry ascending axons derived from somatic sensory neurons in ganglia. The medial lemniscus (solid oval), the rostral continuation of the dorsal funiculus in the spinal cord, conveys second-order axons from the contralateral cuneate and gracile nuclei; these nuclei in the caudal medulla oblongata are the sites where ascending first-order axons from forelimb (cuneate) and hind limb (gracile) dorsal root ganglia synapse. The trigeminal sensory tract (dashed oval) carries axons that originate in the ipsilateral trigeminal (cranial nerve V) ganglia. Cynomolgus macaque. Original objective magnification = whole organ screen capture from a digitized whole slide scan. (Image reproduced with added tract annotation from Bolon B, et al (2013), 9 by permission of Sage Publications).
Microscopic Findings in the CNS
For CNS-localized effects, the distribution depends on many factors, particularly the RoA and vector dose. The influence of these factors on lesion distribution in the CNS is discussed below. This section is designed to introduce the primary neuropathological changes that constitute the recognized spectrum of AAV-related class effects.
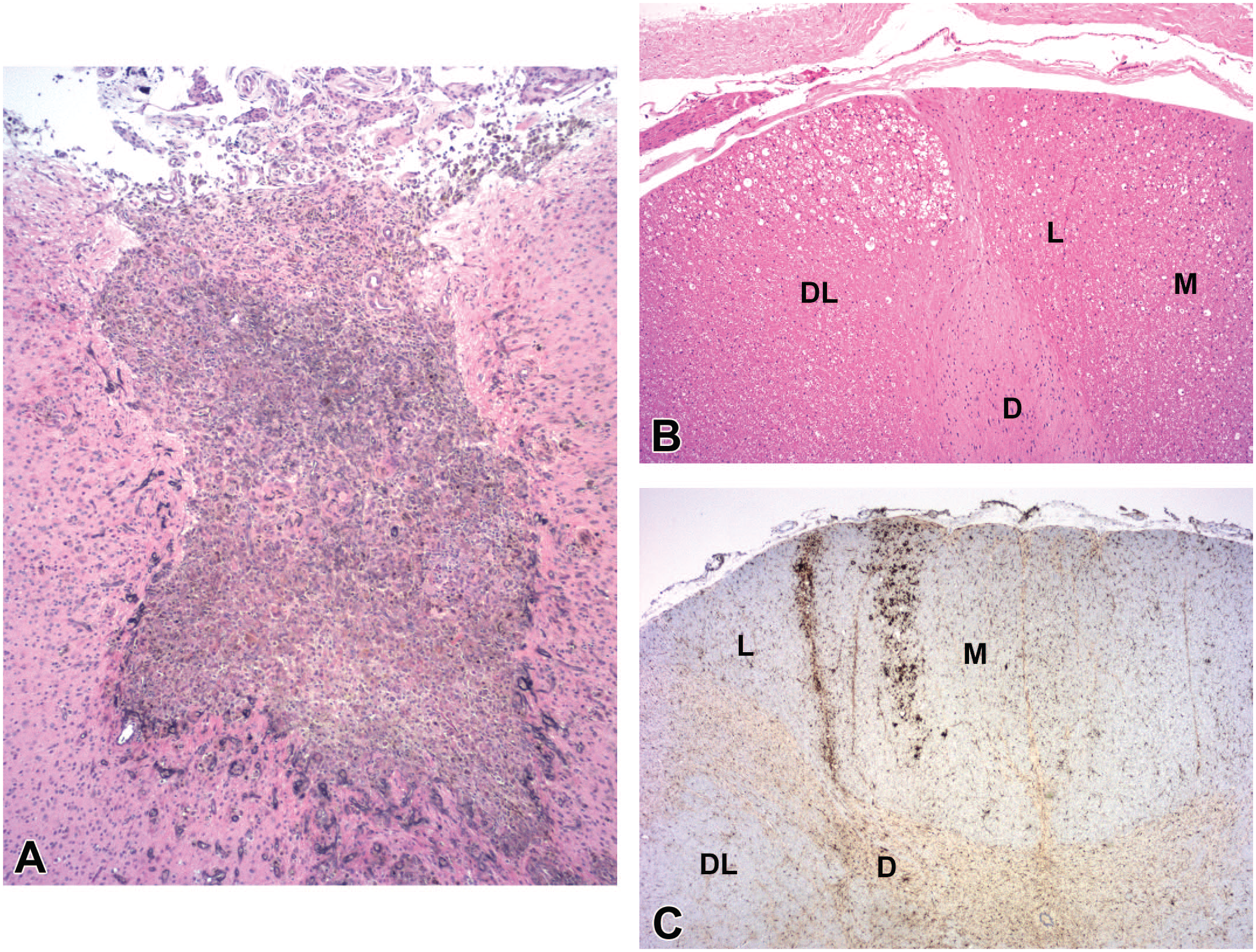
Findings related to direct CNS tissue injury are observed principally along injection tracks ( Figures 5 and 6 ), as procedural effects perhaps exacerbated on occasion by reflux (“backflow”) of the TA, as well as in the neuronal populations of the brain and less often the spinal cord (mainly the ventral horn). This distribution reflects the major sites of AAV vector distribution and transgene expression following IPa or IT administration. As described above, findings reflecting secondary CNS effects of primary damage to the PNS are typically localized to the ascending white matter tracts of the spinal cord (mainly the dorsal and lateral funiculi) and brainstem (in locations that represent the rostral extensions of these spinal cord tracts as well as the tracts that carry fibers originating in the TG).
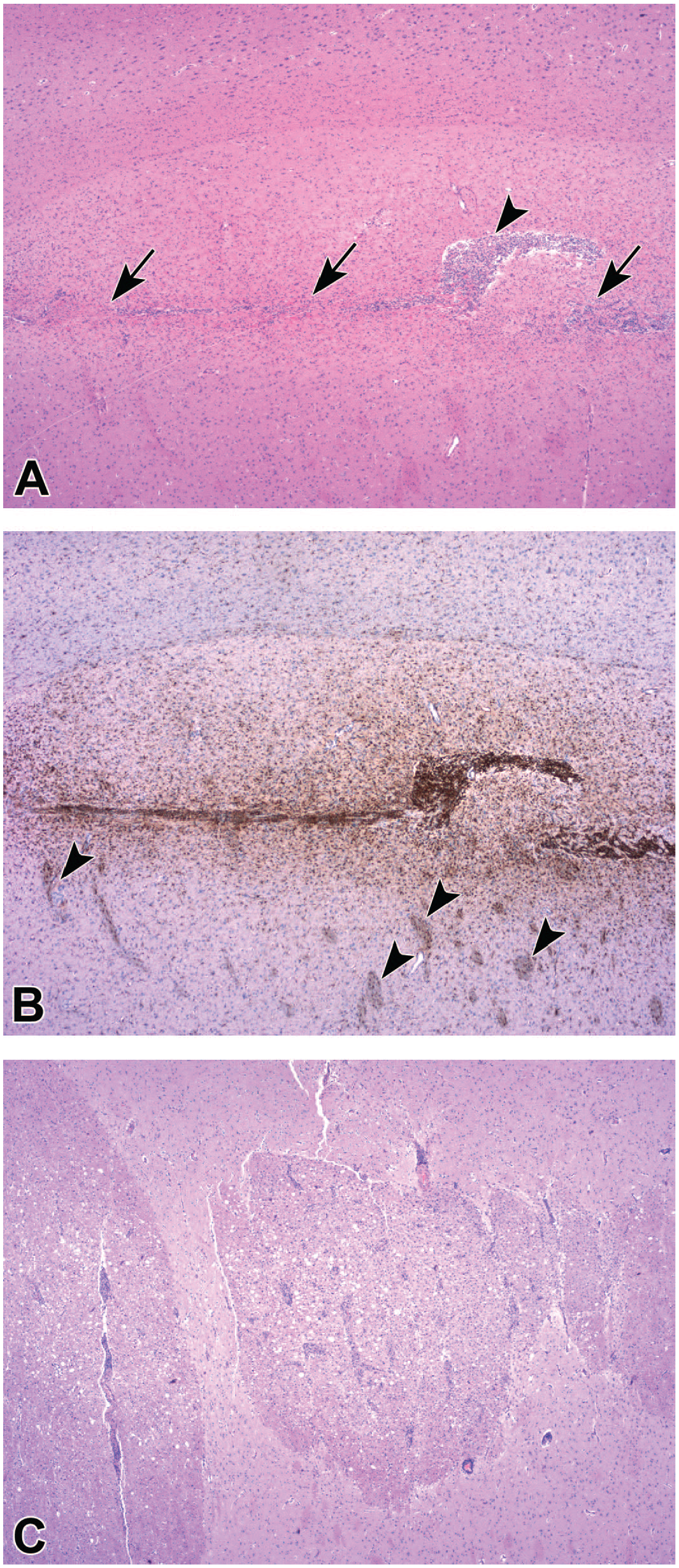
Representative attributes of intraparenchymal injection sites in deep brain nuclei after delivery of an adeno-associated virus (AAV) gene therapy vector. The most frequent changes include narrow linear cannula tracks (long arrows [
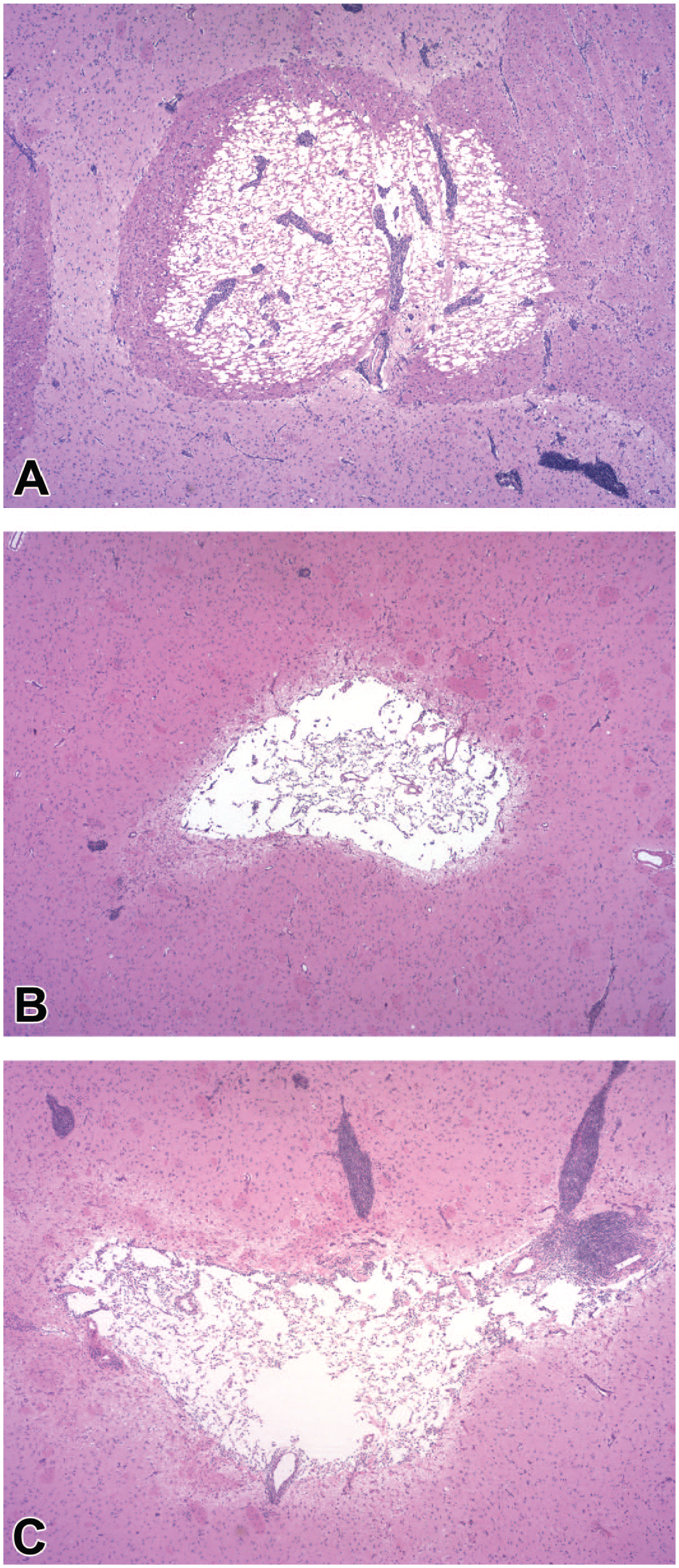
Occasional attributes of intraparenchymal (IPa) injection sites in deep brain nuclei after delivery of an adeno-associated virus (AAV) gene therapy vector. Common changes include small foci of mononuclear cell infiltration (all panels) with localized parenchymal vacuolation (
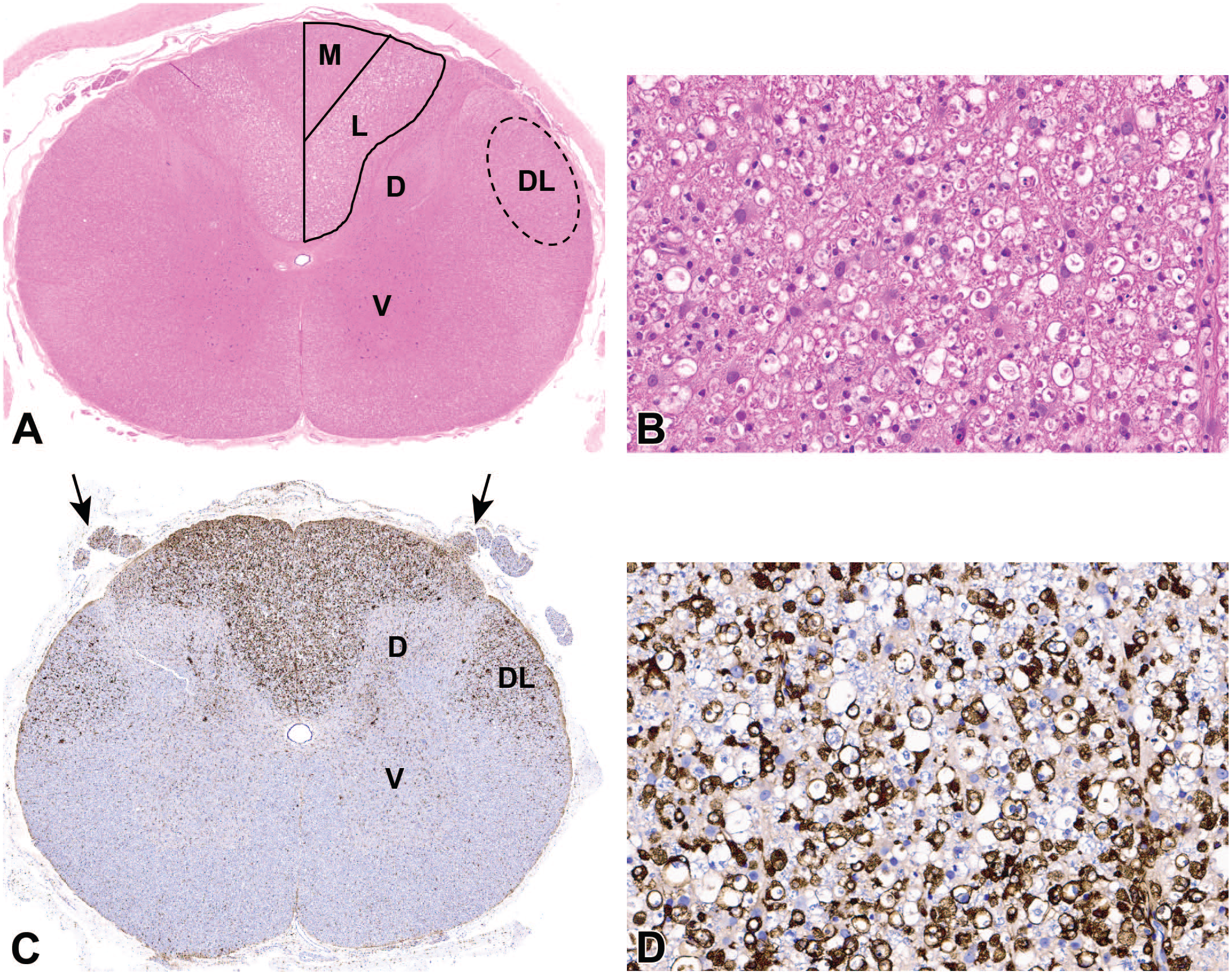
Common findings related to transduction and expression of transgenes delivered using AAV-based vectors involve CNS neurons and their axons, and sometimes glial cells (oligodendrocytes and/or astrocytes). Neuronal degeneration in the brain and spinal cord may present as scattered cells or focally extensive cell fields with reduced amounts or complete absence of cytoplasmic Nissl substance (INHAND diagnosis: “degeneration, neuronal”). Neuronal necrosis appears as shrunken profiles with hypereosinophilic cytoplasm and condensed and/or fragmenting nuclei (classic “red dead” neurons, INHAND diagnosis: “necrosis, neuronal”). Because these two changes often occur together, the diagnosis “degeneration/necrosis, neuronal” may be assigned as a composite INHAND term. Necrotic neurons may exhibit surface divots containing closely apposed glial cells and macrophages (INHAND diagnosis: “neuronophagia”). The neuropil around affected neuroaxonal elements may contain clear, colorless spaces (“vacuoles”) that range in size from small (INHAND diagnosis: “vacuolation” [
Figure 5C
]), which reflects parenchymal porosity due to loss of neuronal processes) to large (INHAND diagnosis: “necrosis,” which indicates tissue death with no or minimal tissue removal [
Figure 6A
]). Vacuolation may not be diagnosed by the study pathologist as neuronal degeneration/necrosis alone is sufficient evidence that neural injury has occurred while obvious necrosis is typically included in the diagnosis list. In time, necrotic tissue may liquefy to yield a cavity (INHAND diagnosis: “neural tissue loss,”
Figures 6B
and
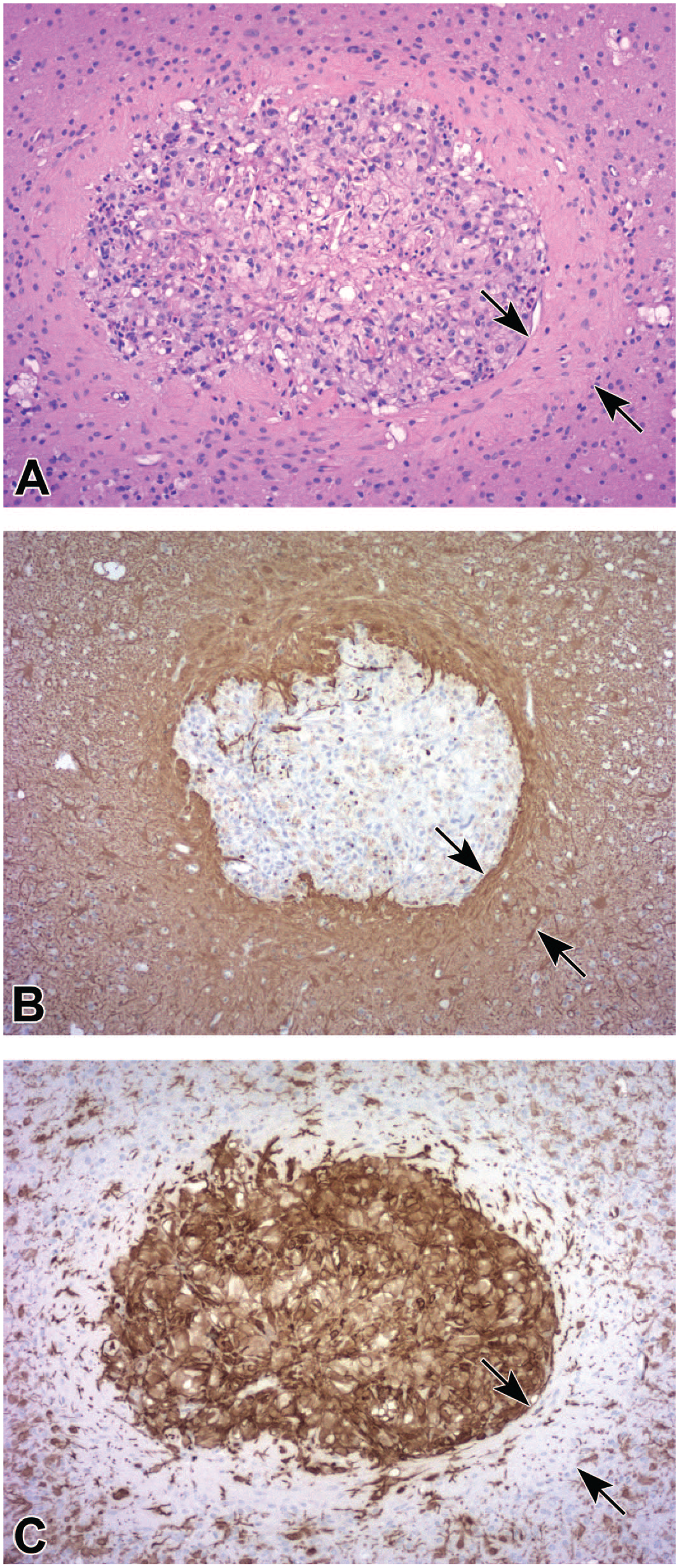
Gliosis (INHAND diagnosis: “gliosis, not otherwise specified”) with or without neuronal necrosis is an occasional consequence in spinal cord gray matter after intrathecal injection of an adeno-associated virus (AAV) vector. By H&E (
Accumulations of glial cells are a common secondary response to AAV-related and procedure-related neural tissue injury. The appearance and thus the diagnosis accorded these findings vary with the histological method used to screen the tissue. In conventional hematoxylin and eosin (H&E)-stained sections, one or a few randomly distributed clusters of small, fusiform to oval cells (INHAND diagnosis: “gliosis”) may be seen in the gray matter (often clustered more densely near neurons,
Figure 7
) or white matter (typically in the deep cerebral white matter of the brain or dorsal and lateral funiculi of the spinal cord). Special neurohistological methods to highlight glial cell type-specific immunohistochemical (IHC) markers often show greater numbers of glial foci in which the reactive cells are increased in size and/or number; the preferred INHAND diagnosis for the glial aggregates depends on the expressed marker, with the term “microgliosis” given to clusters labeled by the microglia/macrophage marker IBA1 (ionized calcium-binding adaptor molecule 1,
Representative features of an intraparenchymal injection site in the deep cerebral white matter following insertion of a rigid cannula to deliver an adeno-associated virus (AAV) vector. By H&E (
Representative appearances of increased glial cellularity and perivascular mononuclear cell infiltrates at intraparenchymal injection sites in deep brain nuclei following injection of adeno-associated virus (AAV) vectors. Increased glial cellularity is indicated by higher cell density in the parenchyma (
Procedure-related effects associated with insertion of a cannula (
The other frequent AAV-related secondary response to primary parenchymal (neuronal and/or axonal) injury is leukocyte accumulation. In the CNS, this change often presents as multiple, usually small foci of mononuclear cell infiltration in the leptomeninges and/or perivascular (Virchow-Robin) spaces (
Biomarkers for Detecting Neuronal and Glial Effects
In principle, biomarkers in fluid samples (blood or CSF) may be used as comparatively non-invasive indicators of neural tissue damage. For example, neurofilament heavy chain (NF-H, 200 kD isotype) is a protein in large sensory neurons of common nonclinical species, including NHPs (macaques and marmosets), rodents (mice and rats), rabbits, and dogs.6,37 A counterpart protein, NF-L, may be elevated in conjunction with nerve fiber degeneration in sensory nerves of dogs with no apparent functional abnormalities (clinical or electrophysiological), 52 and increased plasma NF-L levels can be measured in rats and NHPs after ICL or IV delivery in conjunction with degeneration of ganglionic neurons and their processes. 22 Alternatively, increased circulating levels of GFAP may occur as an indicator of a CNS glial reaction. These fluid biomarkers may be elevated in many neurological conditions and thus are not specific for a given disorder. Nonetheless, for general toxicity studies undertaken to evaluate the safety of AAV-related vectors, especially for neural disease indications, fluid samples (CSF and/or plasma) suitable for assessing these biomarkers should be retained for potential future analysis. To our knowledge, no literature reports have systematically assessed whether or not routine evaluation of such biomarkers is a useful element of neuropathology evaluation for nonclinical safety testing. That said, such markers are sometimes included in nonclinical studies (with collection as one or two baseline readings, one or more in-life time points, and at necropsy) because any trend identified in animals offers an option for non-invasive monitoring of neuropathological sequelae in patients enrolled in clinical trials.
Variables Affecting the Spectrum of AAV-Related Neuropathological Findings
Many factors can influence the basic pattern and severity of AAV-related findings in the CNS and PNS. This section discusses the principal factors and their sway on the basic pattern. These insights are generic guidance only based on our collective experience. The actual impact of these factors on expression of AAV-related findings should be assessed on a case-by-case basis.
Dose
Neuropathological effects associated with AAV vectors may develop at any dose. One to a few animals may exhibit microscopic findings at lower doses while most or all animals typically have some findings at higher doses. Importantly, the pattern of findings needs to be judged on its own merits. For example, the most severe finding for a given study may occur in one or more lower-dose animals (often at only one or two sites per animal) while findings in the high-dose animals that received the same AAV-based TA may exhibit minimal or mild changes (though frequently at a greater number of sites per animal relative to the lower-dose animals). In terms of reporting such findings, care should be taken to record both the consistent dose-related pattern of findings (e.g., usually more widespread AAV vector distribution and/or transgene expression with more severe changes at higher doses) plus any key outliers that might provide additional context for risk assessment.
Depending on the experimental design, the severity of findings appears to vary with exposure as determined by both the total dose and the local dose concentration. In terms of total dose, findings are usually minimal to occasionally mild at doses up to 1 × 1012 and often 1 × 1013 vector genomes (vg) per animal or vg/kg and may range from minimal to marked at higher doses. This pattern primarily reflects the exposure of ganglionic neurons to AAV-based vectors administered by an IT route (ICL, ICM, ICV) or IV, which yield exposure via the CSF, and therefore is evident as effects localized in the PNS more than the CNS. In contrast, AAV-based vectors delivered into the tissue (i.e., IPa infusion into the brain) will result in locally high exposures at the site of administration and findings that are concentrated in the CNS more than the PNS.
In general, AAV-related neuropathological findings in the CNS and PNS of nonclinical species are not associated with measurable in-life functional aberrations. Of course, an important caveat to this observation is that standard designs for nonclinical screening studies typically are limited to a functional observational battery (FOB) and do not include neurological examinations and functional (e.g., electrophysiological) testing. Rarely, electrophysiological (nerve conduction velocity [NCV]) alternations with proprioceptive deficits have been described at very high doses of AAV-based TAs (commonly 1 × 1014 vg/animal or higher), and have been interpreted to reflect primary damage to ganglionic neurons.17,27 In general, in-life neurological deficits have not been reported in association with neuropathological findings of minimal to moderate severity.
Route of administration
Neuropathological findings depend to a considerable degree on the RoA. The basic pattern of findings is approximately comparable across RoAs, but the severity of various changes within the pattern differs across the RoAs. The known class effects related to AAV-related ganglion toxicity occur following all direct central RoA (ICV, ICL, ICM > IPa) as well as by systemic RoA (e.g., intraperitoneal [IP], IV > intramuscular [IM], subcutaneous [SC]).
Delivery into the Brain Parenchyma
For administration directly into the brain (IPa), most findings are restricted to the brain parenchyma. The most substantial findings occur where the TA is introduced into deep tissue (
Several physical parameters related to the injection may impact distribution of an infused material at an IPa injection site. In general, fluid will move outward radially from the opening at the end of the delivery device. Inclusion of a gadolinium-based contrast agent in the dosing solution permits in-life monitoring of the injection site, yielding a locally hyperintense signal when viewed by magnetic resonance imaging (MRI)2,60; hyperintense foci, representing edema and inflammation, may persist for several weeks at injection sites in MRI brain images. Importantly, MRI may be employed in the clinic to monitor structural changes related to IPa infusion of AAV-based gene therapy vectors. The shape of the fluid dispersal field is influenced by the design attributes of the delivery device, including the diameter, position of the orifice (opening at the tip vs on the side near the tip), as well as the porosity and shape of the device walls.2,48 In addition, spreading away from the injection site depends on the character of the parenchyma at that administration site. For example, white matter tracts act as a barrier to flow for fluids delivered into gray matter, so that, the TA is constrained to diffuse mainly within the gray matter, while fluids injected into white matter readily disperse within the white matter. That said, injected fluid can move along the device walls away from the target site and reflux along the injection track, reducing the dose present at the intended site due to escape into the adjacent tissue.3,12,49 Judicious use of reflux and injection trajectories may be used to maximize fluid distribution at the intended injection site. 2 In general, the final distribution of a large volume of infused material cannot be predicted reliably merely by assessing hyperintense MRI signals since infused fluids containing AAV vectors disperse at a different rate compared with contrast agents. 13 Nonetheless, the general trends are that the extent of tissue damage at IPa sites tends to increase for larger infusion volumes, longer infusion times, higher infusion rates, and constant (instead of gradually ascending) infusion rates. 51 The final AAV distribution and size of the injection-related lesion depends on how these injection variables interact, the transgene properties, and the proximity of the tissue section to the actual administration site and injection track.
Delivery into Cerebrospinal Fluid
For central delivery directly into the CSF (ICV, ICL, ICM), the findings are more widespread and concentrated at the CNS surface (as multiple, usually mononuclear cell infiltrates in the meninges and topologically continuous perivascular spaces along penetrating blood vessels) and in the PNS (as primary injury to DRG and TG sensory neurons). The ganglionic damage leads to secondary changes in the spinal cord, SNRs, and nerves (as nerve fiber degeneration in some cases accompanied by a glial reaction [gliosis in the spinal cord and/or increased glial cellularity in ganglia and nerves]). In general, neuropathological findings within the brain parenchyma are constrained to focal or multifocal gliosis, which reflects the variable and typically lower level of neural cell transduction when AAV vectors percolate in from the organ surface.
For systemic delivery (IV, IM), findings are widespread and mimic the pattern described for direct delivery into the CSF. This pattern reflects the extensive capillary network and fenestrated endothelium (i.e., absence of a tight blood-ganglion barrier) in the DRG and TG, which means that these target organs are readily dosed by blood-borne TAs. With original-generation AAV-based vectors, comparatively little TA will cross the tight blood-brain barrier (BBB) found in the brain and spinal cord parenchyma. Next-generation AAV vectors can circumvent the BBB and provide better penetration and more uniform distribution into deep CNS tissues, but at present it is not clear if the enhanced AAV entry will yield a pattern of AAV-related effects consistent with deep tissue (IPa) injection compared with CSF (ICV, ICL, ICM) delivery.
Test species
Neuropathological findings in the PNS recognized as AAV-related class effects occur in many nonclinical species. These findings are common in sensory ganglia (DRG and TG) of NHPs, including juvenile and adult cynomolgus and rhesus macaques, as well as African green monkeys7,22,27-32,42,45,55; pigs27,58; rabbits18,53; and rodents (mice and rats).6,16,22,45,57 Similar findings have been reported in sympathetic ganglia of rats. 45 The pattern of changes is approximately similar across species but may be less severe in some (e.g., mice6,29 and rats 17 ) or develop at a different rate (e.g., inflammation may be more modest and slow to rise in rats compared with NHPs 45 ).
To our knowledge, AAV-related in-life neurological signs have been described in the nonclinical setting only in piglets. In this case, piglets developed ataxia (movement difficulties) and proprioceptive (sensory) deficits in association with substantial DRG neuron degeneration within 14 days after a single high-dose (2 × 1014 vg/kg) IV injection on postnatal day 7 or 30. 27 Abnormal NCV recordings have been reported from the sural nerve (a sensory-predominant trunk) in cynomolgus monkeys after ICL injection at 1.68 × 1014 vg/animal, but the decreased NCV was not associated with detectable in-life neurological signs. 16
Vector attributes
The AAV vector itself influences the pattern of neuropathological findings. Effects vary with the vector tropism (serotype [capsid complement]), which dictates which tissues are transduced and to what degree, and the payload (e.g., promoter, transgene), which affects the strength of transgene expression. Unfortunately, the study pathologist may not be apprised of such features prior to the microscopic evaluation, but where available such details may provide useful clues to explain the pattern and severity of AAV-related neuropathological findings.
The AAV serotype is a principal aspect of configuring AAV-based vectors for neurological indications. Serotype 9 (AAV9) capsid is the most common option for treating neurological diseases based on its proven neurotropism, but several other serotypes (AAVs 1, 2, 5, 8, rh10) are also employed for CNS applications.24,38,40 Certain AAV serotypes can spread more widely from the injection site 24 and preferentially target-specific neuronal and/or glial populations.43,59 Recently, capsid engineering innovations have been undertaken to optimize capsid-receptor interactions, thereby enhancing penetration across the BBB. 33
The transgene in an AAV construct influences both the extent of transgene expression and the immune system assault on transduced cells. 29 At present, transgene overexpression related to the design of the transgene genome is hypothesized to drive ganglionic neuron injury.29,30 The choice of promoter controls the strength of transgene expression and also directs transduction of various neural cell types (e.g., neurons and glia vs neurons alone). 43 Moreover, transgenes tested in the nonclinical setting generally encode a foreign protein (typically of human origin) while capsid proteins of AAV vectors are demonstrably foreign. Accordingly, their introduction into animals with an intact (i.e., wild-type), mature immune system drives some degree of anti-transgene immune response centered on the cells that express the foreign (“non-self”) transgene, suggesting that an immune response may also be a contributor.21,26,47 The immune battering may be magnified for transgenes that yield excessive accumulation of highly overexpressed DNA, RNA, or protein.45,57 Certain transgene products (often reporter proteins of non-mammalian origin) are highly immunogenic, such as the jellyfish-derived green fluorescent protein (GFP), 50 which boosts the attack on transduced cells.
Neuropathological Findings Associated With the Delivery Procedure
Procedure-related findings are influenced by several factors. The principal parameters are the cannula/needle (injection device), delivery approach (injection trajectory), and infusion rate. A variant of procedure-related finding is related to injury resulting from in-life sample acquisition (i.e., CSF collection). The common patterns of these procedure-related effects are outlined below, and may be explored further elsewhere.15,20
Medical Device for TA Administration
In the brain (ICV or IPa injections), insertion of a cannula or needle unavoidably causes physical disruption of the parenchyma through which the device is introduced (
An injection track associated with device placement in the brain exhibits reasonably regular features when examined microscopically. In longitudinal orientation, injection tracks are usually narrow and elongated ( Figure 5 ). In transverse orientation, injection tracks are typically small and round to oval ( Figure 8 ). In our experience with vehicle control animals, one or more modest expansions away from the injection trajectory may develop somewhere along injection tracks, where they represent a common feature of introducing fluid into a dense tissue with little interstitial space. However, such cavities are infrequent in control animals, especially at longer time points (3 months or more), and are small compared with cavities that may form at the site of TA deposition in the deep brain parenchyma ( Figure 6C ). Such expansions may be larger at higher TA doses (thus representing a dose-dependent TA-related effect) or for greater injection volumes (regardless of whether TA is present in the infusate). In the superficial cerebral cortex, initial penetration of the brain parenchyma by the delivery device produces a columnar void (apparent as a conical or rectangular profile in 2-dimensional tissue sections) of variable width that may contain foamy, sometimes brown-pigmented phagocytes ( Figure 10A ) or be empty. The parenchyma near the injection track may contain increased numbers of glial cells (“gliosis”), evident as increased nuclei in H&E-stained sections; scattered leukocytes (typically “infiltrate, mononuclear cell”); or sometimes a modestly greater number of capillary profiles (mainly at acute times, Figure 10A ). On occasion, superficial blood vessels at the IPa injection site may be damaged, leading to a focal hemorrhage or infarct characterized by parenchymal necrosis and variable gliosis.
When a delivery device is inserted into a CSF-filled cavity, procedural effects on the brain (ICM injection) or spinal cord (ICL injection) present a range of possibilities. No lesions may be observed for successful injections of spinal cord where the device does not enter the underlying parenchyma. An infrequent procedural effect of IT device insertion is a linear column of focal tissue disruption (typically evident as necrosis and/or gliosis) in the dorsal brainstem or dorsal lumbar/sacral spinal cord. As with brain injections, any exacerbation in effects observed in cannulated or injected animals that receive an AAV vector rather than vehicle alone are typically deemed to represent a TA-related effect.
Delivery Approach
For IPa injections in the brain, the delivery approach (i.e., injection trajectory) can influence the extent and severity of neuropathological findings. For brain diseases, efficacy of AAV-based vectors typically requires transduction of neuronal populations that occupy a fairly large volume in the outsized human brain. Effective targeting of widely dispersed neurons in the brain often necessitates that the TA be delivered in small aliquots at multiple sites (from 2 to 6 is common) bilaterally rather than as a single larger depot released at one site per hemisphere. An efficient strategy for accomplishing multi-site injections is to insert the cannula deep into the brain to deposit the first aliquot, followed by withdrawal of the cannula a small distance before infusing a second aliquot, followed by a third short withdrawal and injection, and so on. For this practice, a single injection track may be used for each targeted brain region rather than producing a separate track for each of several injection sites per region. Neuropathological findings may be further influenced by the choice of osteotomy sites when entering the cranial vault. For example, in targeting a bilaterally symmetrical brain nucleus, two osteotomy holes (one made in each half of the cranium) will yield a pair of non-intersecting IPa injection tracks while one hole will yield converging injection tracks that intersect at the brain surface. More osteotomy holes may be needed if multiple widely spaced neuronal populations have to be treated. The study pathologist should be informed of the number and location (relative to bregma) of injection sites, the delivery method (single site vs multiple sites along a given injection trajectory), and ideally ancillary data (e.g., intra-operative and post-operative MRI scans) to optimize the trimming scheme and microscopic evaluation.
Infusion Rate and Volume
The rate of fluid delivery directly impacts the pattern of AAV-related neuropathological findings, 3 especially when the TA is delivered into a dense tissue (e.g., IPa injection into the brain). As a procedure-related effect, larger delivered volumes (e.g., >100 µL total delivered at a single site in an NHP brain) may produce a bigger cavity at the injection site in comparison to a small injection volume. For the same volume, a variable infusion rate with gradual ramp-up (e.g., sequential 5-minute steps at 1, 2, 3, and 4 µL/min with a final rate of 5 µL/min for the remaining infusion) produces less damage extending outward for a reduced distance compared with the wider disruption produced by a higher constant infusion rate (e.g., 5 µL/min).
In-Life Sample Acquisition
When a cannula or needle is inserted into a CSF-filled space to harvest CSF samples for analysis, the brain (ICM sampling) or spinal cord (ICL sampling) may be injured by exuberant introduction of the device. For a single CSF collection, a linear column of minimal tissue disruption may be created in the dorsal surface of the brainstem (for ICM) or caudal spinal cord (for ICL); for the spinal cord, the damage may be produced in the caudal lumbar or sacral divisions. This procedure-related tissue disruption may appear as either focal degeneration of nerve fibers, gliosis, or frequently some degree of both. Rarely, for serial CSF collections, one or multiple, roughly parallel, vertically oriented linear columns of minimal to mild severity may develop in the brainstem or spinal cord ( Figure 10C ). Transient or chronic placement of a dosing cannula may yield superficial nerve fiber degeneration in the dorsal funiculus or dorsolateral portion of the lateral funiculus of the spinal cord ( Figure 10B ).
Collection of CSF from the lumbar cistern also may damage one or more SNRs (i.e., cauda equina) rather than the spinal cord. The key procedure-related findings in the SNRs are nerve fiber degeneration and sometimes increased Schwann cellularity. These findings may affect more nerve roots and be of greater severity following many episodes of collection. If the necropsy is performed soon after sampling, acute hemorrhage may be seen at the sampling site.
Post-Administration Tissue Reactions in the Brain
Certain pathological findings related to ICV or IPa injection are tissue reactions induced by the injection procedure and not the TA. These findings are evident in the brain and overlying meninges, occur on a sporadic basis, and persist for extended periods in various forms. Such findings are readily seen in standard H&E-stained sections. At the brain surface, tissue edges that border injection tracks may be raised modestly relative to the usual contours of the nearby brain surface. These bulges may be either smooth (which is consistent with protrusion of the brain parenchyma into the void left by an incompletely filled osteotomy site) or rarely tattered (which suggests focal disruption of tissue that had adhered to the overlying meninges,
Figure 11C
). The overlying meninges may exhibit foci of hemorrhage, where acute changes include intact erythrocytes while chronic and/or resolving lesions contain pigmented hemosiderophages and scattered erythrocytes (
Figures 11A
and
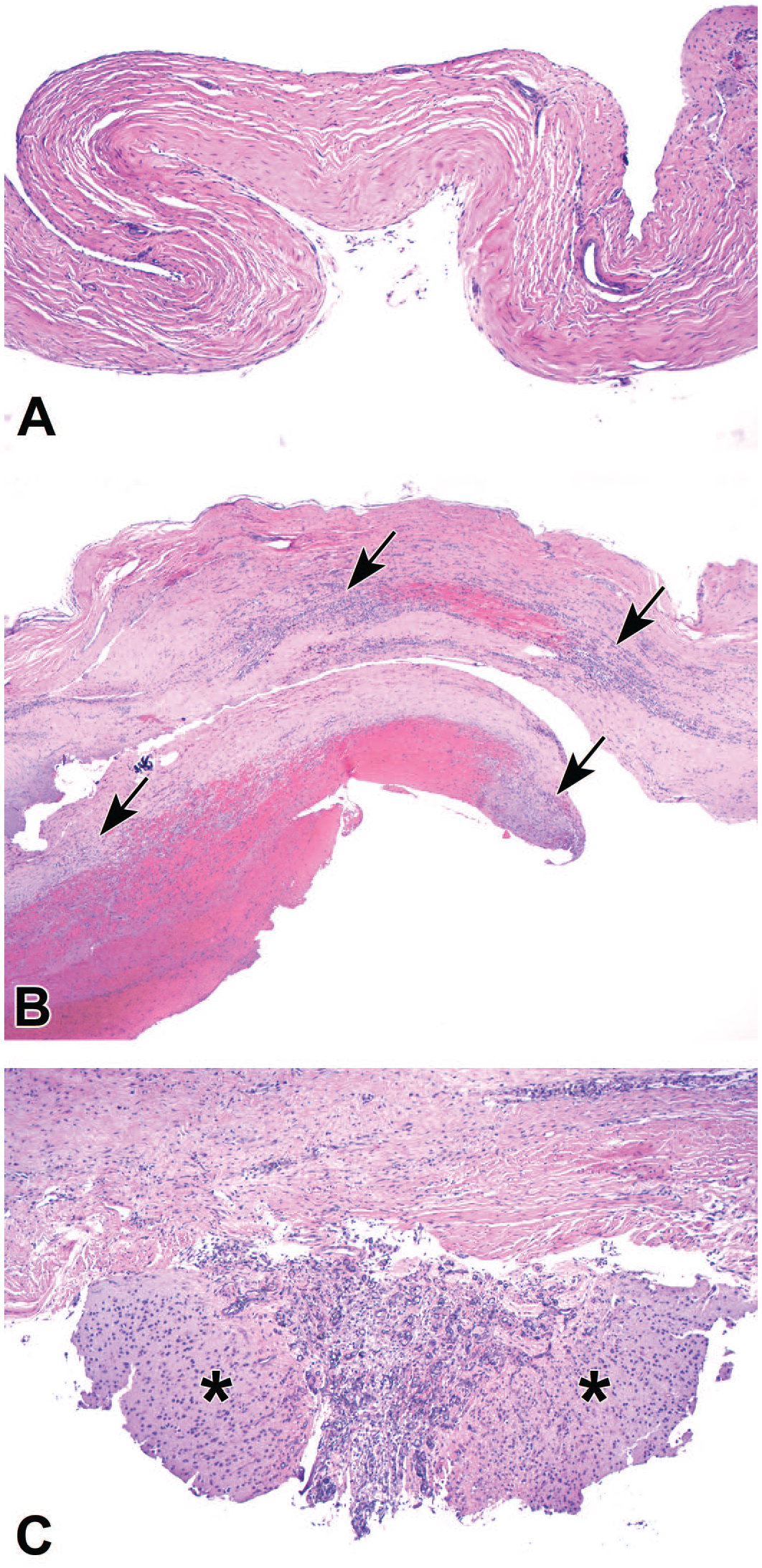
Representative post-procedural tissue responses of intraparenchymal injection sites at the brain surface following insertion of a rigid cannula to deliver an adeno-associated virus (AAV) vector into the brain. The overlying dura mater ranges from a uniform sheet of parallel dense fibrous connective tissue with rare small foci of acute hemorrhage (
Adversity of Neuropathological Findings Associated With AAV Vectors
Adversity as applied to pathology data generated for nonclinical studies has been defined in differing ways. A simple definition as stated in the STP’s “best practice” recommendations on determining, communicating, and using adversity delineates ten core principles, of which the first is that adversity reflects “harm” to the test animal. 36 A more elaborate definition proposed by a working group of the European Society of Toxicologic Pathology (ESTP) holds that adverse effects are TA-related changes in one of multiple biological traits (e.g., morphology, physiology, growth, development, reproduction, life span) leading to impaired functional and/or homeostatic capacities encountered in daily life and when undergoing a physiological challenge. 44 In general, adversity for AAV-based products is defined on a case-by-case basis. Several considerations pertain in making decisions when assessing the implications of AAV-related neuropathological findings.
Common AAV-related primary neuropathological findings in the CNS and PNS result from damage to neurons and their processes. In the CNS, neuronal degeneration and necrosis as well as nerve fiber degeneration are not repaired. In the PNS, injury to ganglionic neurons is not repaired while nerve fiber degeneration may be restored anatomically and functionally after some time. Neuronal and axonal damage of any degree, especially in the CNS, is often viewed as inherently adverse (as suggested by the ESTP 44 ). However, minimal to mild neuron degeneration/necrosis as well as minimal to mild nerve fiber degeneration typically occur in the absence of detectable functional effects, which is frequently construed as a lack of harm to the animal (as viewed by the STP36). The rationale for this latter adversity interpretation is the modest nature of the findings (i.e., effects are often limited to only one or a few neurons or nerve fibers at a limited number of sites) and the redundant functional support provided by intact neural fields that can compensate for this degree of injury; the caveat to this interpretation is that functional testing as practiced in routine nonclinical studies is a blunt tool for detecting neural deficits. In preparing pathology reports for nonclinical toxicity studies, the presence of such AAV-related class effects of limited degree may be interpreted as non-adverse for practical purposes. This interpretation requires detailed explication in the pathology report.
A complicating factor for adversity decisions produced by direct central administration of TAs is that they must be interpreted in the context of concurrent procedure-related effects. These procedural findings cannot be avoided for injections into the brain (e.g., ICV and IPa) and may be seen for injections into CSF-filled caverns (e.g., ICL and ICM). By convention, procedure-related findings are not considered when making adversity decisions with respect to the adversity of a TA, but such procedure-related effects should be addressed in crafting a pathology report. The relative contributions of any findings attributed to an AAV-based TA and the procedure-related tissue disruption (if any) must be assessed in the context of duration-matched vehicle controls.
Conclusions
Administration of AAV-based gene therapy vectors induce a spectrum of neuropathological findings in the CNS and PNS that represent documented class effects for this modality. These lesions may develop whether the AAV agent is delivered directly into the CNS or systemically. The most common changes are neuron degeneration and necrosis, especially affecting ganglionic sensory neurons; nerve fiber degeneration in the CNS (spinal cord white matter > brainstem) and PNS (ganglia as well as peripheral nerves and SNRs); and secondary responses like glial reactions and leukocyte accumulation (infiltration and/or inflammation). The changes are comparable across nonclinical species although for equivalent doses the severity is often more substantial in NHPs compared with rodents.
While the basic constellation of AAV-related neuropathological findings is similar across AAV constructs and species, subtle variations in the extent and severity may be affected by numerous factors. Findings are correlated with exposure, with neural tissues encountering higher AAV levels following direct central administration. Lesions are concentrated near the injection site and injection track for routes of administration that penetrate neural tissue (e.g., ICV and IPa) while changes are more widespread and follow the classic gangliocentric pattern5,6,29,34 more closely when the agent is delivered into CSF (ICV, ICL, and ICM) or systemically (e.g., IV > IM). The AAV-related findings overlap to some degree with procedure-related changes associated with direct CNS administration. The concordance of AAV-related and procedure-related effects requires careful consideration when making adversity decisions.
Footnotes
Acknowledgements
The authors gratefully thank Ms Beth Mahler for assistance in optimizing the figures.
Authors’ Note
Several authors have shifted to new institutions since acceptance of this article. Drs. Buza and Hordeaux have transitioned to GEMMA Biotherapeutics, Inc., Philadelphia, Pennsylvania, USA. Dr. Galbreath is now employed by Experimental Pathology Laboratories, Inc., Sterling, Virginia, USA.
Author Contributions
The authors are solely responsible for the contents and crafting of this paper. Brad Bolon delivered the specific STP talk on which this paper is based (with coauthors for this paper serving as co-presenters for the talk) and drafted the manuscript. All authors assisted in refining the concepts (including visual depictions) as well as editing the slides for the talk and the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The work reported in this article was conducted during the normal course of employment. Two authors (BB of GEMpath and EG of Advanced Pathology Solutions) are private practitioners who provide scientific consulting services to private and public institutions, while the other authors (EB and JH of the University of Pennsylvania and FC and JW of Spark Therapeutics) are scientists at institutions developing gene therapy agents. Different authors have presented these findings previously, in piecemeal fashion, at various public scientific meetings.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors were compensated by their employers for performing work in developing gene therapy agents, and some data were also supported by a grant from FA212 LLC. The funders had no role in the analysis and interpretation of the data included in this paper, the decision to publish, or the preparation of the manuscript.
