Abstract
The increasing specificity of novel druggable targets coupled with the complexity of emerging therapeutic modalities for treating human diseases has created a growing need for nonhuman primates (NHPs) as models for translational drug discovery and nonclinical safety assessment. In particular, NHPs are critical for investigating potential unexpected/undesired on-target and off-target liabilities associated with administration of candidate biotherapeutics (nucleic acids, proteins, viral gene therapy vectors, etc.) to treat nervous system disorders. Nervous system findings unique to or overrepresented in NHPs administered biomolecule-based (“biologic”) test articles include mononuclear cell infiltration in most neural tissues for all biomolecule classes as well as neuronal necrosis with glial cell proliferation in sensory ganglia for certain viral vectors. Such test article-related findings in NHPs often must be differentiated from procedural effects (e.g., local parenchymal or meningeal reactions associated with an injection site or implanted catheter to administer a test article directly into the central nervous system) or spontaneous background findings (e.g., neuronal autophagy in sensory ganglia).
Keywords
Introduction
Neurological diseases afflict millions of people worldwide. 1 The most common in terms of global disease burden are severe cephalgia (“migraines”), cerebral infarction (“strokes”), and various dementias (Alzheimer’s disease [AD], frontotemporal dementia, Huntington’s disease, Lewy body dementia, etc.). Numerous other conditions also exert their primary effects on the central nervous system (CNS) or peripheral nervous system (PNS). The most serious disorders are progressive neurodegenerative diseases (amyotrophic lateral sclerosis [ALS], dementias, Parkinson’s disease, spinal muscular atrophy [SMA], etc.); lysosomal storage diseases (mucopolysaccharidoses, neuronal ceroid lipofuscinosis, oligosaccharidoses, sphingolipidoses, etc.); autoimmune diseases (e.g., multiple sclerosis [MS], Guillain-Barré syndrome [GBS]); and nervous system tumors. These conditions collectively are major unmet medical needs. In particular, the cumulative health care liability posed by progressive inherited and adult-onset neurodegenerative diseases is predicted to become a major socioeconomic burden in future years.1,2 Accordingly, neurodegenerative diseases are frequent indications for drug discovery and development investment.
To meet this need, biopharmaceutical research companies are currently developing over 260 novel medicines for treating 29 major neurodegenerative diseases. 3 Many additional therapeutic candidates are being investigated in earlier stages of the drug discovery pipeline. These novel test articles are capable of targeting either specific molecular components that mechanistically drive the disease pathophysiology (e.g., altered neurotransmitter levels, beta-amyloid plaques, tau protein tangles) or discrete processes that inflict damage directed against one or more neural cell populations (e.g., neuronal necrosis, immune and inflammatory responses, metabolic changes). Besides small molecules (“conventional” drugs) and large biomolecules (e.g., monoclonal antibodies [mAbs], recombinant proteins) that already feature prominently in the armamentarium for treating neural diseases, many new modalities now are being developed for this purpose. Among these innovative approaches, biomolecules designed to replace, remove, or silence a particular gene are gaining favor as targeted agents for correcting cell-type-specific molecular defects in the nervous system. In this regard, major modalities that have catapulted to the fore in the last decades include various nucleic acids (e.g., antisense oligonucleotides [ASOs], messenger RNAs [mRNAs], microRNAs [miRNAs], small interfering RNAs [siRNAs]), numerous proteins (e.g., antibody-based fusion proteins), and increasingly gene editing (e.g., clustered regularly interspaced short palindromic repeats [CRISPR]) and especially gene therapy (e.g., nonviral and viral vectors). Neural stem cell implantation also shows great promise as a means of replenishing depleted cell populations or cell secretions (e.g., growth factors, neurotransmitters) in neural tissues. Key advantages and disease targets of these novel therapeutics and delivery platforms are briefly summarized in Table 1.
Novel biotherapeutic modalities and delivery platforms that target the nervous system.
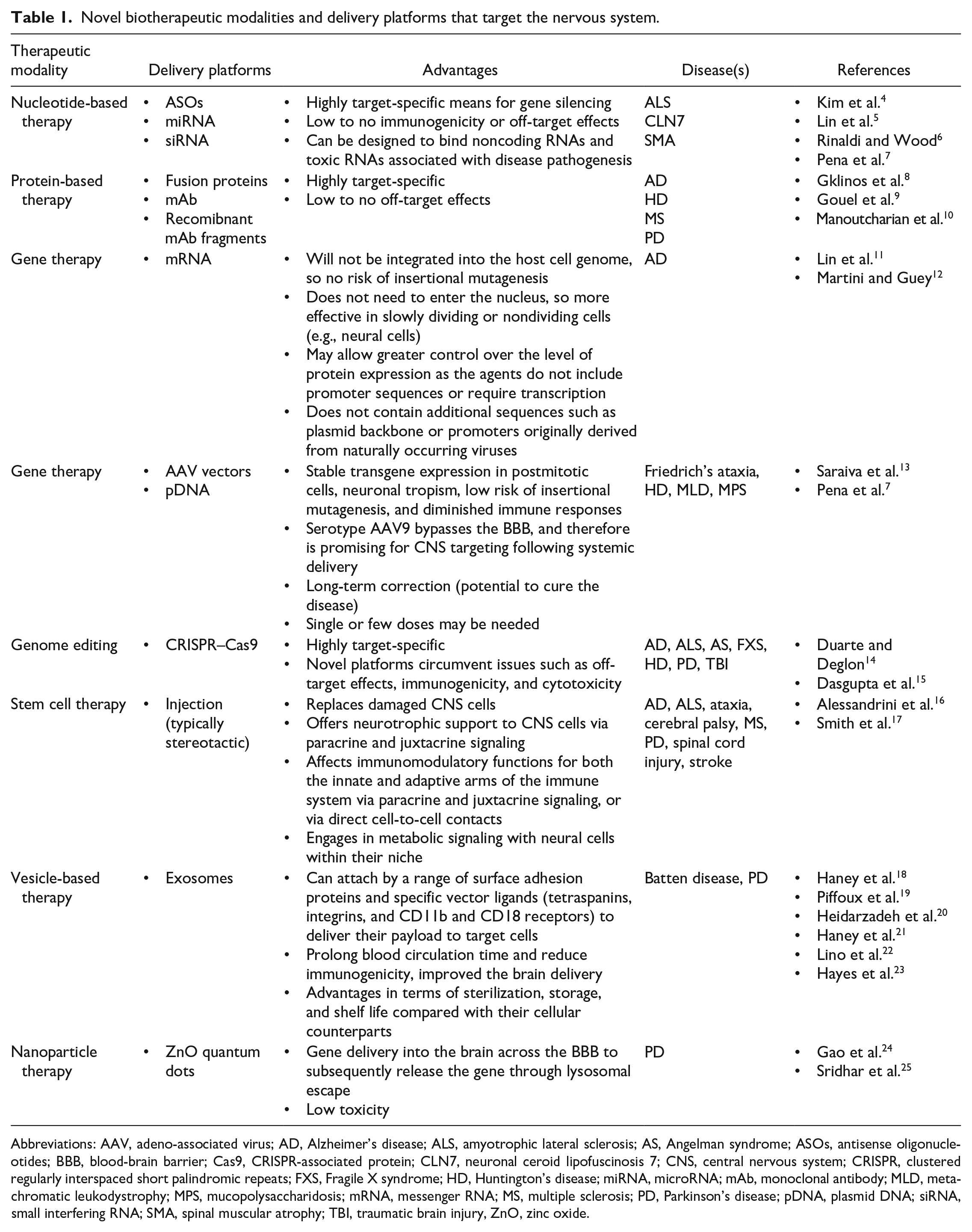
Abbreviations: AAV, adeno-associated virus; AD, Alzheimer’s disease; ALS, amyotrophic lateral sclerosis; AS, Angelman syndrome; ASOs, antisense oligonucleotides; BBB, blood-brain barrier; Cas9, CRISPR-associated protein; CLN7, neuronal ceroid lipofuscinosis 7; CNS, central nervous system; CRISPR, clustered regularly interspaced short palindromic repeats; FXS, Fragile X syndrome; HD, Huntington’s disease; miRNA, microRNA; mAb, monoclonal antibody; MLD, metachromatic leukodystrophy; MPS, mucopolysaccharidosis; mRNA, messenger RNA; MS, multiple sclerosis; PD, Parkinson’s disease; pDNA, plasmid DNA; siRNA, small interfering RNA; SMA, spinal muscular atrophy; TBI, traumatic brain injury, ZnO, zinc oxide.
Development of these novel biomolecule-based test articles for treating neurodegenerative diseases is fraught with unknowns in comparison with conventional protein-based (mAbs, recombinant proteins) and small molecule drugs. The new biotherapeutic modalities possess many unique biological properties, including unusual molecular structures, the need to deliver extremely high doses (e.g., 10e14 or more particles per patient) directly into the CNS, complex biodistribution (i.e., equivalent to absorption, distribution, metabolism, excretion [ADME] and pharmacokinetic [PK] properties of small molecules), and potential cell tropism that permits transduction of a broad range of peripheral organs. Furthermore, pharmacodynamic (PD) on-target (desired and unexpected) and off-target effects may be incompletely characterized. Finally, the chemistry, manufacturing, and control (CMC) processes to produce consistent large-scale amounts of high-quality biologic test article suitable for nonclinical and clinical testing are technically sophisticated. The complicated nature of the development pathway for these novel biotherapeutics often requires a case-by-case approach to designing individual safety studies and entire product development programs. Regulatory guidance for developing biologics depends on the nature of the product. For example, oligonucleotides, while based on naturally occurring nucleotide sequences, are synthesized by conventional chemical processes and thus are regulated according to ICH (International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use) M3(R2) similar to conventional pharmaceutical molecules. 26 In contrast, protein-based biologics are regulated by ICH S6(R1) 27 while cell and gene therapies currently are developed using various national or regional guidelines.28-30
Nonhuman primates (NHPs) are integral to the development of biomolecule-based test articles for treating neurodegenerative diseases, for many reasons. 31 This article briefly reviews these reasons, concentrating in particular on the biological basis for NHPs as a preferred species for nonclinical neurotoxicity testing, major aspects of nonclinical study design and conduct for neurotoxicity evaluations in NHPs (emphasizing but not limited to primary neuropathological endpoints), and common biomolecule-related neuropathology findings and their implications for human risk assessment.
Core Biological Considerations for Neurotoxicity Testing in NHPs
The primacy of NHPs as a key animal model for drug discovery and development in many respects reflects the biological attributes common to all primates, including humans. First, genomes for NHPs including principal proteins in the nervous system are highly conserved compared with humans (97% or higher depending on the NHP species).32-34 This molecular equivalence often permits the administration of human-based proteins to NHPs with a reduced NHP immunologic response relative to other animal species.35,36 Moreover, the molecular structures of many human-derived biomolecules render them active only in humans or closely related NHPs, and not in other test animals. 37 This ability for human-derived molecules to function in a test species is essential in assessing off-target effects related to the pharmacological activity of the test article, and for many biologics cannot be evaluated in animal species other than primates. 38 Second, NHPs possess neurobiological complexity equivalent to that of humans. The brain mass of NHPs (approximately 100 g) is about 10% of the human brain mass (range, 1000-1500 g depending on body size) and is appreciably larger than the brain mass of other common test species (about 0.4 g for mouse, 2 g for rat, and 72 g for dogs). 39 The large neocortex and intricate pattern of gyri and sulci distinctive of the gyrencephalic (convoluted surface) NHP brain are more human-like than those of other gyrencephalic test animals (e.g., dogs, minipigs) and especially lissencephalic (smooth surface) rodents and rabbits.40-46 These neuroanatomic traits reflect the increased regional cross-connectivity and synaptic density needed to support the functional distinctions characteristic of NHPs and humans, including enhanced associative thought via a large prefrontal cortex with elaborate inter- and intra-hemispheric circuitry as well as exquisite fine motor control (especially for opposable “thumbs” and other digits) centered in the expanded cerebellar hemispheres. Finally, modern genome editing procedures offer the opportunity to modify NHPs to better model human neurological diseases.47,48 This opportunity is important because in many respects the complex behaviors common to primates (NHPs and humans) are examined most readily in a primate test system.
In addition to the structural, physiological, and molecular similarities between humans and NHPs described above, another major advantage of NHPs as animal models for neurotoxicity assessment of candidate biotherapeutics is the relative ease of administering test articles directly to the CNS. The ability is essential because biomolecules typically cannot cross the healthy blood-brain barrier (BBB); for this reason, direct delivery is frequently used to achieve optimal biodistribution to the target cell population and minimize off-target systemic liabilities. That said, some neural elements (notably dorsal root ganglia [DRGs]) lack a barrier and therefore will be exposed to materials delivered centrally or peripherally. To bypass the BBB, biomolecule-based test articles often are delivered to the CNS by direct intraparenchymal injection (e.g., into a specific brain region) or injection or infusion into the cerebrospinal fluid (CSF) via intracerebroventricular (ICV) injection into a lateral ventricle, intracisternal (ICM) injection into the cisterna magna, or intrathecal (IT) injection into the lumbar cistern over the caudal spinal cord. The total CSF volume of NHPs is approximately 15 ml (vs 100-150 ml in humans) and the lateral ventricle volume of NHPs is around 0.25 ml (vs 25 ml in humans), but the turnover of CSF is comparable in both species (4-5 complete replacements per day). 39 Dogs have a similar CSF volume (approximately 12 ml) and CSF turnover rate (about 5.75 replacements daily) but have a quadrupedal posture rather than the bipedal (human) or semi-bipedal (NHPs) posture of primates. 39 By comparison, rodents have much higher CSF turnover (12.5 replacements per day in mice, nearly 29 per day in rats) and very small CSF volumes (approximately 0.04 ml in mice, 0.15 ml in rats). 39 The larger size of the NHP nervous system typically supports administration using the same catheter or needle intended for clinical use, while rodents often cannot accommodate such devices. The greater physiological similarity of NHPs and humans coupled with the higher likelihood that human-based biomolecules might exhibit on-target PD activity largely explains the preference for NHPs as test systems for evaluating potential neural effects of biomolecules being developed to treat neural diseases.
Design and Conduct of NHP Neuropathology Evaluations for Biomolecules
Potential neurotoxic liabilities of biomolecule-based test articles are typically investigated by integrating data acquired using multiple analytical techniques. Evaluation of neural function in primates may include conventional in-life observations, detailed neurological examinations, neurobehavioral testing (e.g., functional observational battery [FOB], electrophysiology),49,50 and CNS safety pharmacology.51,52 Biodistribution studies assess the presence of the test article or (for entities designed to modulate gene expression like ASOs, siRNAs, and viral gene therapy vectors) altered levels of gene transcripts or proteins in expected cell types. 53 Neuropathology evaluation is essential for examining effects in neural tissues. Good Laboratory Practice (GLP)-compliant nonclinical toxicity studies always include anatomic pathology endpoints but also often incorporate clinical pathology analyses as well. In most cases, conventional anatomic pathology endpoints (e.g., macroscopic and microscopic evaluations, brain weight) are more informative than clinical pathology endpoints (e.g., CSF chemistry and cytology analyses). A detailed consideration regarding all aspects of designing NHP studies (e.g., selection of doses, dosing interval, necropsy times) to assess neurotoxicity is beyond the scope of this article, so the remainder of this section is focused on key points related to neuropathology evaluation in NHPs that have been given biomolecule-based test articles.
Fixation depends on the study objectives. In general, NHP nonclinical toxicity studies assess the CNS and PNS as two of many systems to be evaluated, even if the test article is introduced directly into the CNS. In this setting, neural tissues may be harvested at necropsy and then fixed by immersion in neutral-buffered 10% formalin (NBF, which is available commercially as an aqueous solution containing 3.7% formaldehyde with ~1% methanol as a stabilizing agent to prevent formaldehyde cross-linking). 54 If the main reason for the study is to better characterize effects in neural tissues, then intravascular perfusion fixation using NBF or methanol-free 4% formaldehyde (MFF, pH 7.4, known colloquially as 4% paraformaldehyde) is preferred because fixative is delivered directly into deep tissues; this rapid introduction of fixative is necessary because lipid-rich neural organs slow the penetration of aqueous fixatives. 55 The rationale for selecting MFF as a fixative for neural tissues is that this solution omits the methanol, which may minimize lipid extraction. Perfusion with chilled saline may be used when downstream analysis precludes perfusion with fixative. Alternatively, perfusion with mixtures of formaldehyde (which penetrates quickly) and glutaraldehyde (which cross-links molecules more tightly) followed by postfixation by immersion in 1% osmium tetroxide (a lipid-stabilizing agent) may be necessary if fine architectural detail of myelin sheaths is the primary study objective. 56 In the authors’ collective experience, careful tissue removal and handling with immersion fixation at room temperature in NBF is acceptable for NHP nonclinical toxicity studies to assess neural effects of candidate biotherapeutics.
The neural tissues to be collected and evaluated for NHP nonclinical studies of neuroactive biomolecules are dictated by the study objectives. For general toxicity studies where CNS and PNS are two of many systems to be assessed and neurotoxicity is unknown or not expected, brain (7 coronal hemi-sections), spinal cord (transverse and longitudinal or oblique sections of cervical, thoracic, and lumbar divisions), and one sciatic or tibial nerve represent the minimum neural tissue list for analysis.54,56,57 Where the test article is suspected or known to produce neurotoxicity, more brain regions as well as select ganglia (e.g., DRGs, trigeminal ganglion [cranial nerve V]) and nerves should be evaluated.56,58,59 Examination of more tissues is consistent with the authors’ experiences where guidance by regulatory agencies in letters to sponsors suggests sampling and evaluation of more CNS and PNS tissues (i.e., “expanded neurohistopathology”) to improve the likelihood of identifying biomolecule-related changes that might predict an impact on neural structures and functions. In general, CNS and PNS tissues should be collected generously at necropsy, even if they are only archived and not slated for immediate processing and evaluation. The reason for extensive collection is that unsampled tissues will be discarded with the carcass and thus permanently unavailable for assessment.
Alternative protocols may be employed in collecting and processing neural tissues for analysis. For example, instead of working with brain hemi-sections (which fit readily on standard 75 mm × 25 mm glass slides), full coronal brain sections may be prepared and placed on large (75 mm × 50 mm) glass slides to permit bilateral evaluation of brain regions. Another option is to collect serial coronal slices and then fix alternating slices by immersion in either NBF (for routine processing into paraffin) or MFF (for routine processing into paraffin or cryosectioning to demonstrate fixation-sensitive molecules). For cryotomy, fixation is performed for 12 to 24 hr at 4°C, after which slices may be transferred to a cryoprotectant solution (e.g., 30% sucrose containing 0.01% sodium azide) until saturated and then stored at –80°C until sectioning (typically 10-20 µm) or using a freezing sliding microtome (typically 20-40 µm). Vibratome sectioning of tissue fixed in MFF and embedded in an agarose block might be useful for obtaining better histomorphology on thick brain sections (typically 30-50 µm) immuno-stained by free-floating immunohistochemistry (IHC). Tissue samples may be flash-frozen without cryoprotection to permit molecular (DNA, RNA, and/or protein) analyses from tissue homogenates; care must be taken to avoid cross-contamination of samples, such as using fresh instruments for each animal or each organ and frequent changes of gloves. In the authors’ experience, these ancillary sampling procedures and anatomic pathology methods typically are not used for general toxicity screens but may be incorporated into biodistribution studies, discovery studies (e.g., proof-of-concept), or combined efficacy/toxicity studies.
Histological preparation of NHP neural tissues follows routine procedures for animal tissues. Processing conditions may need to be adjusted for tissues of larger NHP species to optimize tissue preservation. 60 In general, all neural tissues are processed into paraffin and sectioned at 4 to 5 µm. In some instances, a nerve block (usually a mixed trunk such as the sciatic or tibial nerve) in transverse orientation is embedded in plastic to better preserve myelin architecture; hard plastic (e.g., epoxy resin) rather than soft plastic (e.g., glycol methacrylate) provides superior resolution in such situations. 56 Plastic blocks are sectioned at 0.5 or 1 µm, and sections are stained with toluidine blue.
The stain battery for NHP neuropathology evaluation also depends on the study objectives. For general toxicity studies where the test article is not known to cause neurotoxicity, routine hematoxylin and eosin (H&E) is acceptable for screening. Additional special neurohistological stains (including immunostains and silver impregnations) may be performed as warranted to characterize any neural effects; these other methods are the second arm of an “expanded neurohistopathology” (i.e., “expanded neuropathology”) analysis sometimes requested by regulatory agencies. Common stains for the CNS include Fluoro Jade (FJ) B or C, which detects necrotic neurons in the CNS; biomarkers for reactive glial cells such as anti-glial fibrillary acidic protein (GFAP, for astrocytes) and anti-ionized calcium-binding adaptor molecule 1 (Iba1, for resident microglia and infiltrating macrophages), Bielschowsky’s silver or anti-neurofilament protein (NFP) for axons, and/or Luxol fast blue (LFB) or anti-myelin basic protein (MBP) for myelin integrity in white matter tracts. 54 In the PNS, ganglia may be stained to show satellite glial cells (SGCs; using GFAP or anti-glutamine synthetase), infiltrating mononuclear cells (e.g., CD3 for T-lymphocytes, CD68 or Iba1 for macrophages); and if needed the axonal and myelin stains noted above. 56 The use of special stains often increases sensitivity of the histopathologic evaluation by demonstrating subtle parenchymal responses (e.g., glial activation) in regions where substantial tissue damage is not visible in H&E-stained sections.
Successful neuropathology evaluation of NHPs requires special technical skills from involved staff. Prosectors responsible for tissue collection at necropsy should be well trained in terms of basic neuroanatomy knowledge and procedures for correctly finding, sampling, and processing neural tissues. In general, this information is maintained in detailed standard operating procedures (SOPs) and reinforced by experience. Pathologists performing NHP neuropathology evaluations should possess a solid understanding of neuroanatomy (e.g., via a medical or veterinary medical education) as well as familiarity with fundamental principles of neuropathology associated with biomolecules. This neuropathology background may be built by perusing reference texts61-63 but is sustained by regular reading of scientific literature (e.g., Bradley and Chilton, 64 Brennan et al., 65 Leach et al., 35 and Pardo et al. 66 ) and continuing education.
Common Neuropathological Findings in NHPs Administered Biotherapeutics
Pathology findings in NHPs that have received biomolecule-based test articles usually present as histopathologic findings rather than macroscopic lesions or brain weight changes. In this section, common neurohistopathologic changes observed in NHPs are presented and, where possible, distinguished as test article-induced, procedure-related, or incidental changes. The key points of this section are summarized in Table 2 for ready reference. To provide a scaffold for understanding, the findings are organized based on the neural cell population exhibiting the change. Globally harmonized diagnostic terminology for neural findings should be used to provide consistency among study reports and facilitate understanding among various constituencies.67-69
Summary of common biomolecule-related neuropathology findings in NHPs.
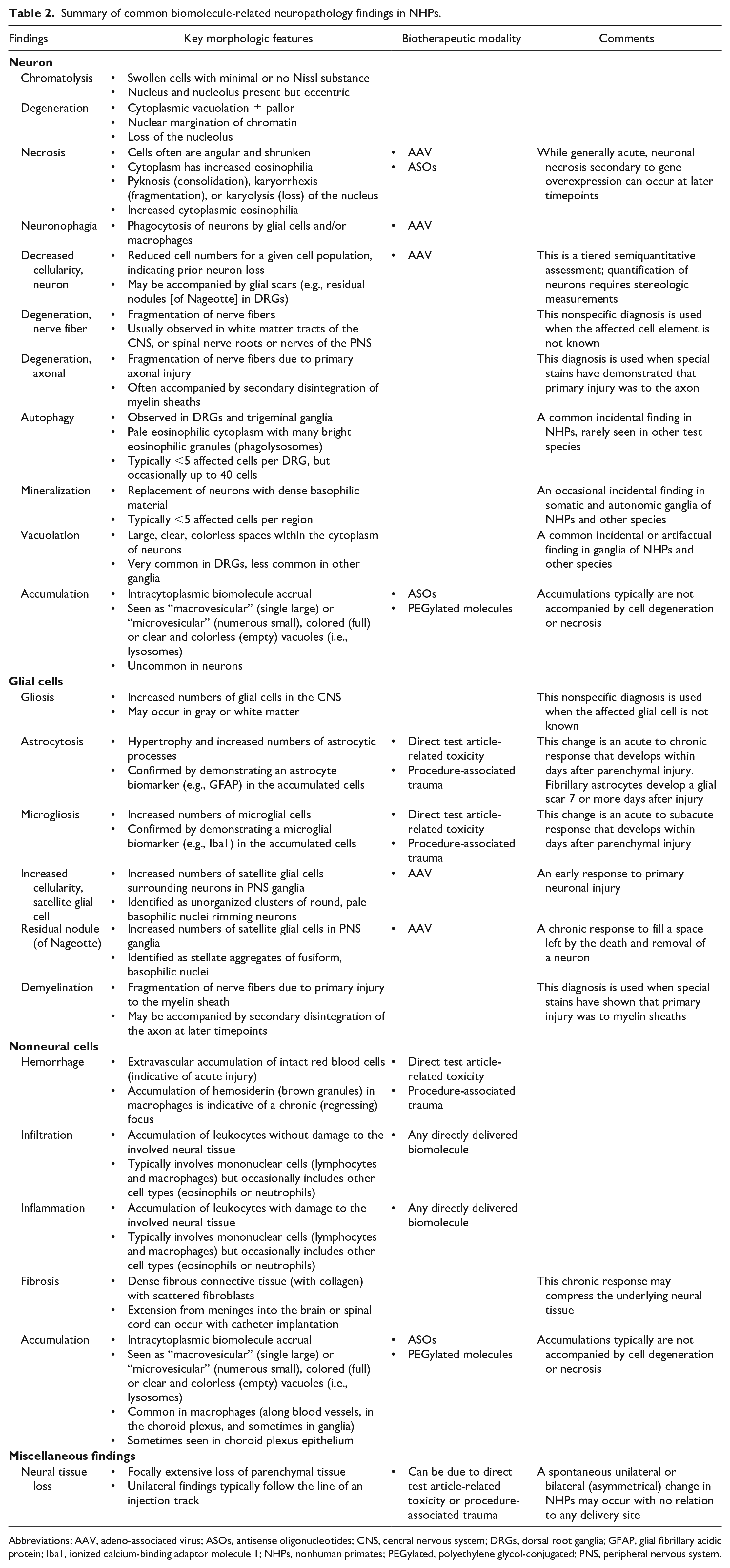
Abbreviations: AAV, adeno-associated virus; ASOs, antisense oligonucleotides; CNS, central nervous system; DRGs, dorsal root ganglia; GFAP, glial fibrillary acidic protein; Iba1, ionized calcium-binding adaptor molecule 1; NHPs, nonhuman primates; PEGylated, polyethylene glycol-conjugated; PNS, peripheral nervous system.
Neurons
Neurons in the CNS (gray matter of the brain and spinal cord) and PNS (somatic, autonomic, and enteric nervous systems) are a frequent site for intended (on-target) biotherapeutic action. Neurons also are a common locus of off-target biomolecule-associated toxicity. The affected neuronal population is influenced by both the test article class and the route of administration. For example, in the authors’ collective experience, adeno-associated virus (AAV) gene therapy vectors often impact DRG neurons whether administered directly into the CNS or peripherally (e.g., by intramuscular [IM] or intravenous [IV] injection), 70 while ASOs seldom affect DRG neurons after IT injection (unpublished observations). Other sites of neuronal degeneration and necrosis in biomolecule-exposed NHPs include cerebral cortex, hippocampus, and cerebellum in the brain and the large pyramidal neurons in the spinal cord ventral horns.
The typical neuron-focused effects imputed to biomolecule test articles are “degeneration” and “necrosis” (often combined in data tables as “degeneration/necrosis”). This finding may be seen in all common test species but in the authors’ experience is more commonly observed in NHPs. By definition, degeneration is a potentially reversible disturbance in neuronal function, while necrosis is an irreversible event that culminates in cell death. The pathogenetic mechanisms likely resulting in both of these changes may involve direct toxicity of the biomolecule (e.g., an overexpressed human protein) toward the neuron70,71 or an immune response mounted against a molecular target that is present within the nervous system. 72 Features of neuronal degeneration may include cytoplasmic pallor due to variable dispersal of Nissl substance (RNA-rich rough endoplasmic reticulum), vacuolation at the cell periphery, and often nuclear chromatin margination and/or loss of the nucleolus. In contrast, neuronal necrosis generally is seen as hypereosinophilic cytoplasm with complete loss of Nissl substance, fragmentation or loss of the cell nucleus (Figure 1), and often cell shrinkage or neuronophagia (i.e., superimposition of infiltrating mononuclear leukocytes over disintegrating neuronal cell bodies; Figure 2). Sections stained with FJ reveal neuronal necrosis as green-highlighted cells and neurites in brain and spinal cord sections. 54 Interestingly, FJ does not consistently label necrotic DRG neurons. 56 The time course of neuron necrosis induced by biomolecules differs from that associated with small molecule administration. For example, following AAV administration, death of DRG neurons can be detected at acute timepoints (less than 14 days) 73 and typically peaks between 2 to 4 weeks after injection, 70 while death of neurons induced by small molecules usually reaches a maximum at 2 to 4 days after exposure. 74 Neuronal degeneration and necrosis must be distinguished from “autophagy,” an incidental background finding seen often in NHPs but rarely in other animal species.68,75,76 Histologically, autophagic neurons are of normal to slightly enlarged size and are distinguished by pale eosinophilic cytoplasm with multiple eosinophilic granules and free-floating amphophilic chromatin globules or spicules (Figure 3). The number of autophagic neurons in a single section of ganglion from control cynomolgus monkeys typically is fewer than 5 but can range as high as 40 (unpublished observations); in most cases, NHPs either have no DRGs with autophagy or many ganglia with autophagy. 76 In the authors’ experience, exacerbation of this spontaneous background change by biologic test articles has not been seen (unpublished observations).
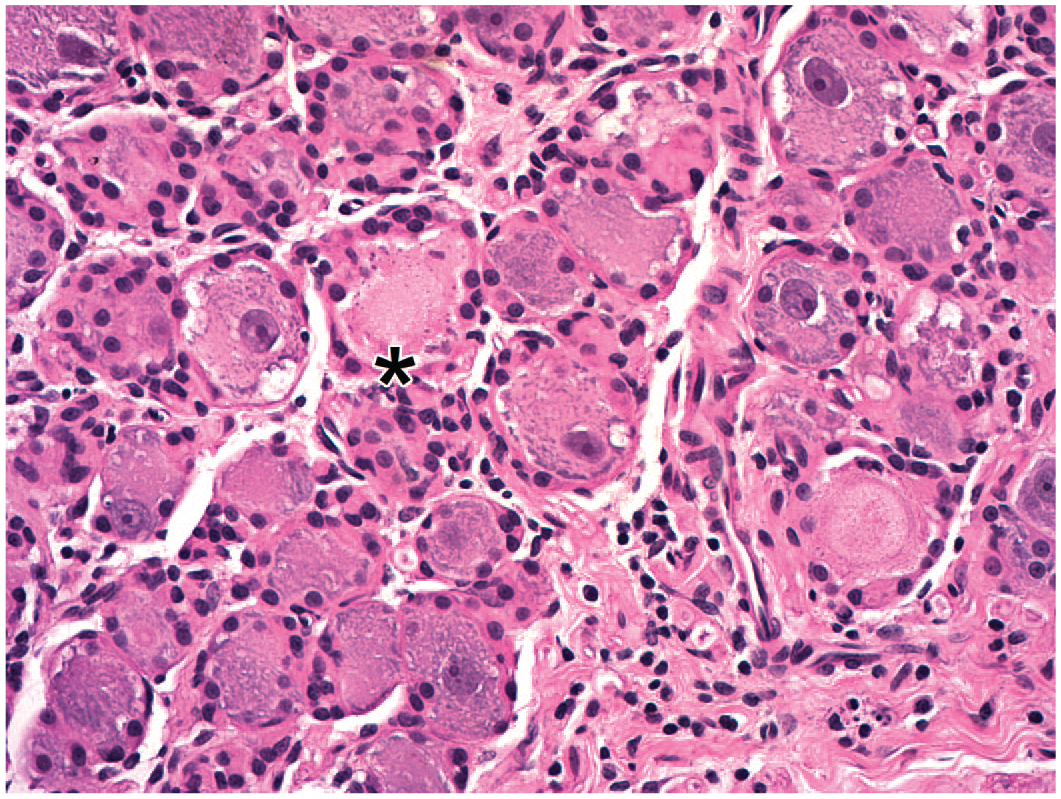
Neuronal necrosis in a lumbar dorsal root ganglion following intrathecal administration of an adeno-associated virus (AAV)–based gene therapy vector in the lumbar cistern. Necrotic neurons (above the asterisk) have eosinophilic cytoplasm and lack nuclei and Nissl substance. Juvenile cynomolgus monkey. Hematoxylin and eosin.
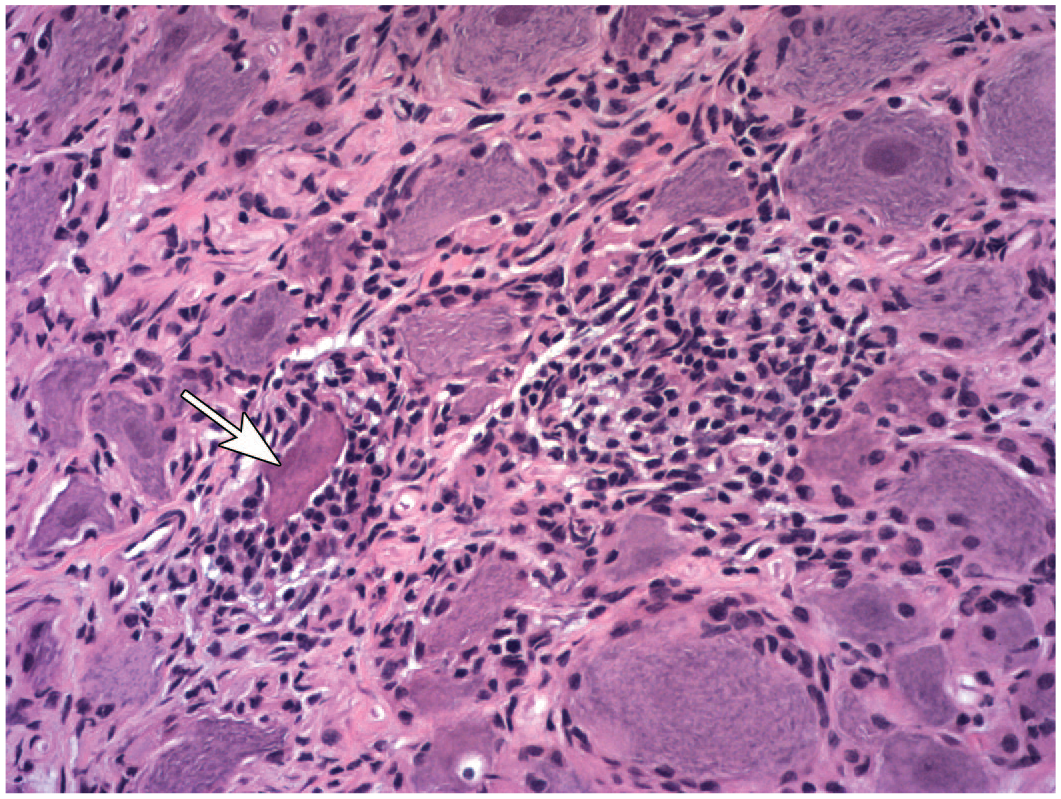
Neuronophagia of a necrotic neuron (arrow) associated with multifocal mononuclear cell inflammation in a sacral dorsal root ganglion following intrathecal administration of an adeno-associated virus (AAV)–based gene therapy vector in the lumbar cistern. The necrotic neuron possesses dark eosinophilic cytoplasm and lacks a nucleus and Nissl substance; neuronophagia is indicated by the scalloped neuronal borders. Juvenile cynomolgus monkey. Hematoxylin and eosin.
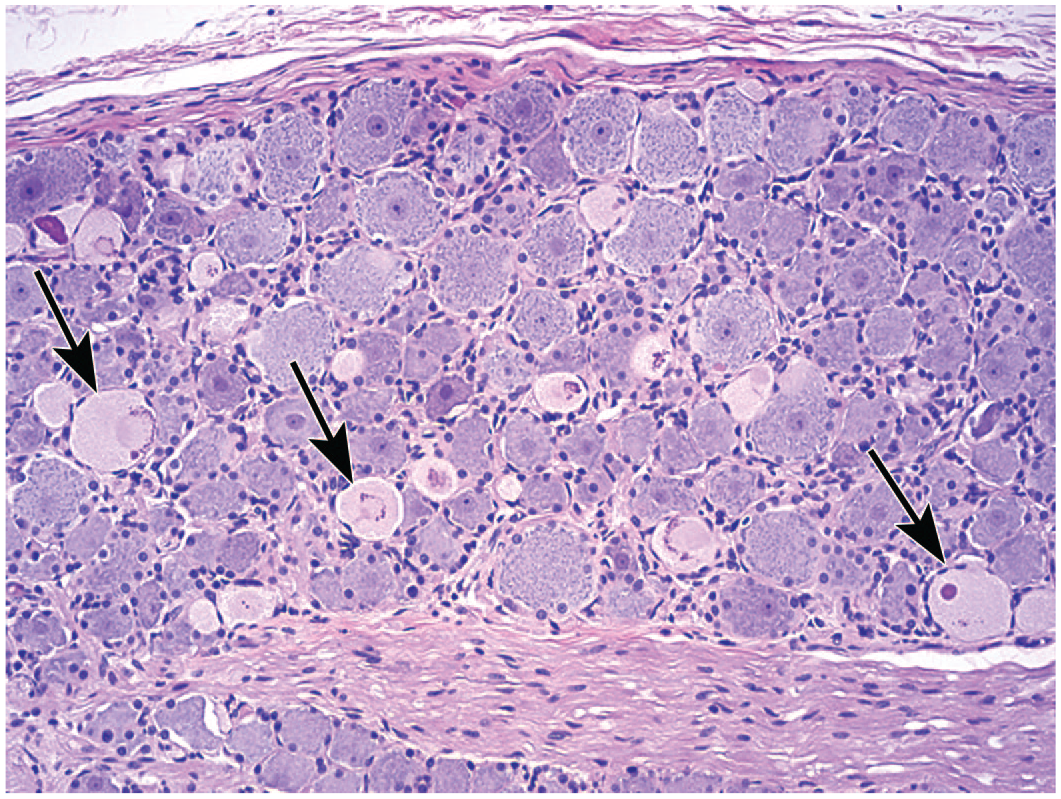
Autophagy, a background (“incidental”) finding, in numerous neurons of a lumbar dorsal root ganglion of a control juvenile cynomolgus monkey. Affected cells (arrows) are swollen and have pale eosinophilic cytoplasm with many punctate granules and scattered amphophilic chromatin globules or spicules. Note the dense bed of neurons surrounding the central core of parallel nerve fibers. Hematoxylin and eosin.
Over time, loss of many neurons may reduce regional cell numbers—a finding diagnosed as “decreased cellularity, neuronal” (Figure 4). Decreased neuron cellularity is relatively frequent in lumbosacral DRGs of NHPs administered an AAV-based test article. However, decreased cellularity should be assigned as a diagnosis only after careful comparison with concurrent control tissue sections in the equivalent orientation because minimal anatomic differences over short distances in DRGs and also various brain regions substantially alter neuronal numbers available for evaluation.
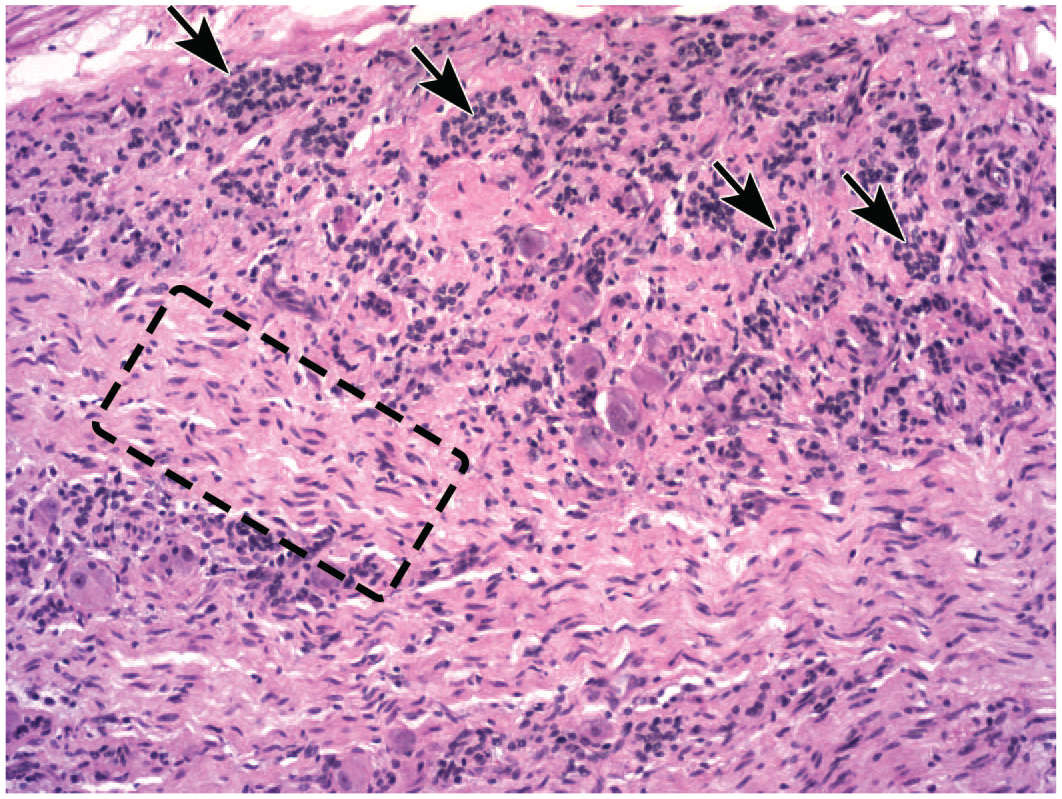
Decreased neuronal cellularity associated with increased satellite glial cellularity in a lumbar dorsal root ganglion (DRG) following intrathecal administration of an adeno-associated virus (AAV)–based gene therapy vector in the lumbar cistern. Compared with a similarly oriented lumbar DRG of a control animal taken at the same magnification (see Figure 3), neuron numbers are markedly reduced and unorganized, coalescing clusters of satellite glial cells (arrows) are widespread. The number of spindle-shaped Schwann cell nuclei in the central core of nerve fibers is modestly higher (see region enclosed by dashed box, in comparison with the nerve fiber-rich region at the bottom edge of Figure 3). Juvenile cynomolgus monkey. Hematoxylin and eosin.
Degeneration of neuronal processes is a secondary change often associated with primary injury (especially necrosis) of neurons. For example, damage to DRG sensory neurons often leads to degeneration affecting spinal cord white matter bilaterally in the dorsal funiculus (Figure 5) and, less often, the lateral funiculus because these tracts carry ascending axons from DRG neurons. Similar changes are seen frequently in brain white matter (e.g., the pyramids of the brainstem), spinal nerve roots, and peripheral nerves. The nonspecific term “degeneration, nerve fiber” is most appropriate if the finding is diagnosed in H&E-stained sections because the site of primary damage (i.e., axon vs myelin sheath) typically cannot be determined in such preparations. 69 If the initial sites of injury can be identified definitively using special procedures (e.g., biomarkers or electron microscopy), the specific terms “degeneration, axonal” (for primary damage to axons) and “demyelination” (for primary damage on myelin) are used instead. 67 In the authors’ experience, approximately 90% of nerve fiber degeneration results from initial axonal injury.
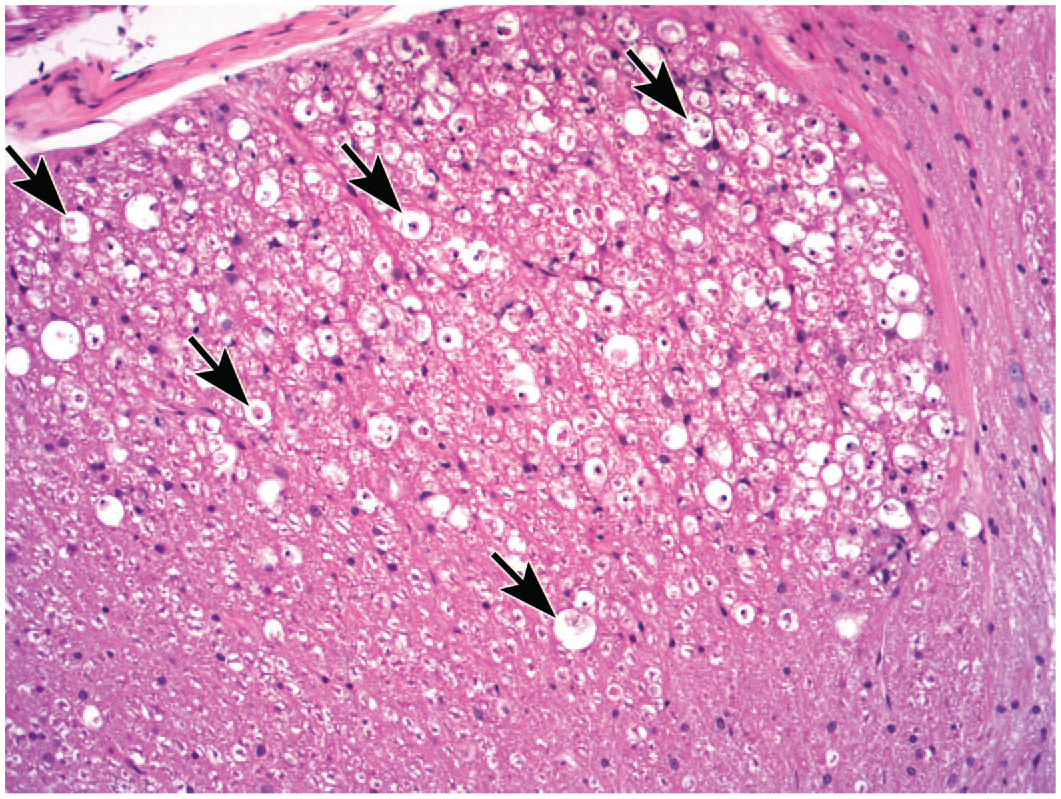
Nerve fiber degeneration (presumably axonal due to the underlying cause) indicated by increased space surrounding axons and the presence of macrophages and cellular debris within digestion chambers (arrows) of the medial fasciculus of the dorsal funiculus in the cervical spinal cord after intrathecal delivery of an adeno-associated virus (AAV)–based gene therapy vector in the lumbar cistern. Juvenile cynomolgus monkey. Hematoxylin and eosin.
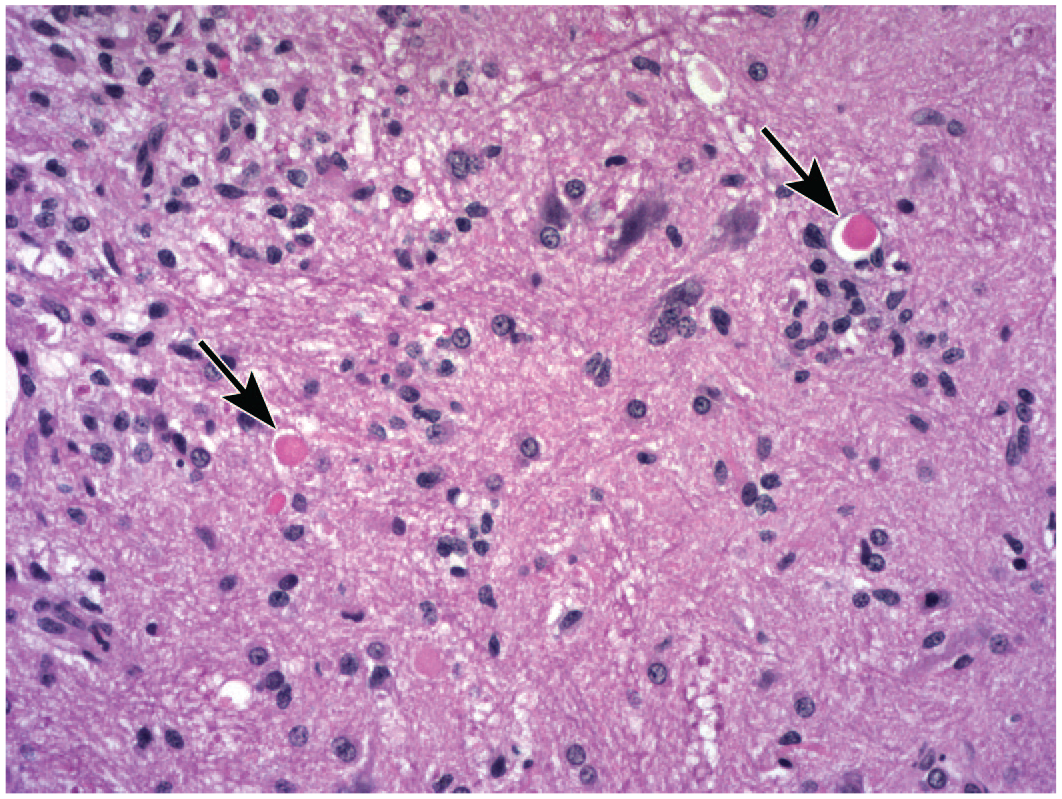
Several miscellaneous neuronal findings are observed frequently in neural tissues of NHPs. “Mineralization, neuronal” (Figure 6) and “vacuolation, neuronal” (Figure 7) are incidental background findings that may be observed to affect one or more neurons in PNS ganglia (in DRGs mainly but also autonomic and trigeminal ganglia). 66 These findings occur in control animals and in the authors’ experience are not exacerbated by administration of biomolecule-based test articles (unpublished observations). Neuronal vacuolation occurs as a single very large or several medium-sized to large, clear, colorless vacuoles in the cytoplasm of sensory neurons, sometimes containing small tangles of fibrillar material. These vacuoles may be either spontaneous background findings or possibly artifacts 77 ; importantly, these vacuoles are not consistent with the many small vacuoles (reported to be enlarged mitochondria) that form in DRG neurons as an artifact of delayed fixation. 78 Pathologists may or may not include these two findings in their histopathology data tables, depending on both personal and institutional preference. “Dark neurons” (Figure 8) are an artifact affecting some neurons—commonly those of the superficial cerebral cortex, hippocampus, Purkinje cells of the cerebellum, and pyramidal neurons of the spinal cord gray matter. 79 As an artifact produced by postmortem manipulation imposed on inadequately fixed tissue and/or by delayed fixation, 80 this finding is not diagnosed. The key point with respect to dark neurons is that they should not be interpreted as evidence of test article-induced injury indicative of degeneration or incipient necrosis.
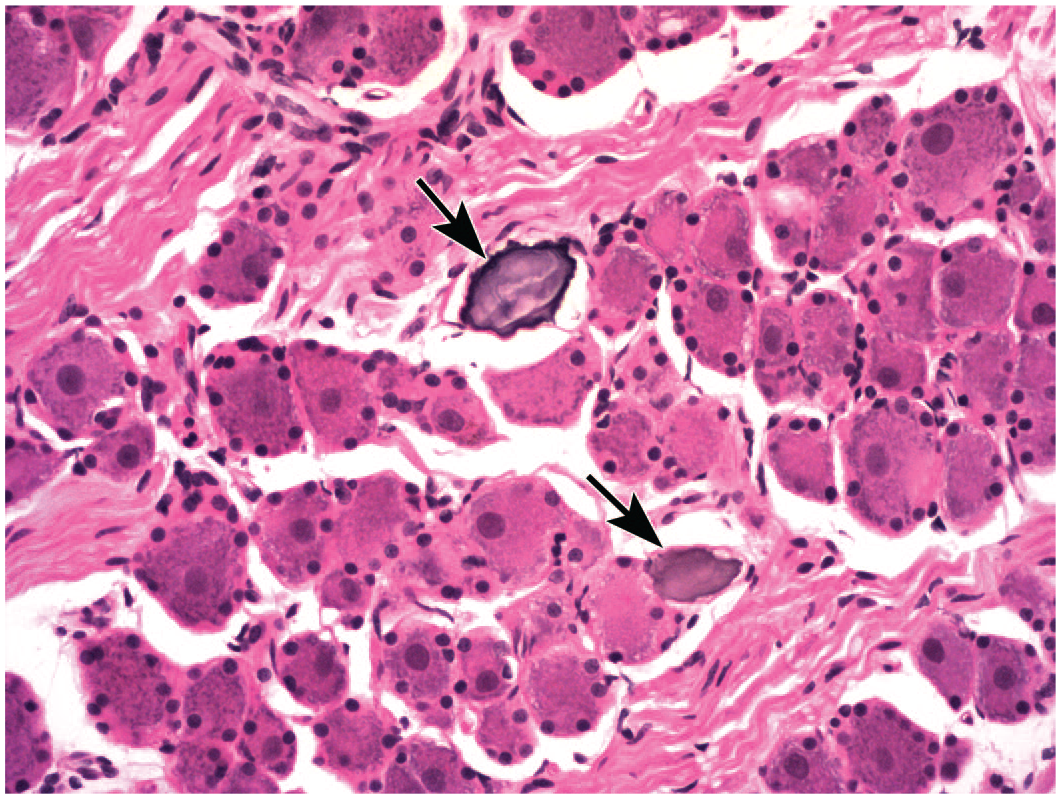
Mineralization of sensory neurons (arrows) in a cervical dorsal root ganglion after administration of an antisense oligonucleotide (ASO) intrathecally in the lumbar cistern. This finding is an incidental background change. Juvenile cynomolgus monkey. Hematoxylin and eosin.
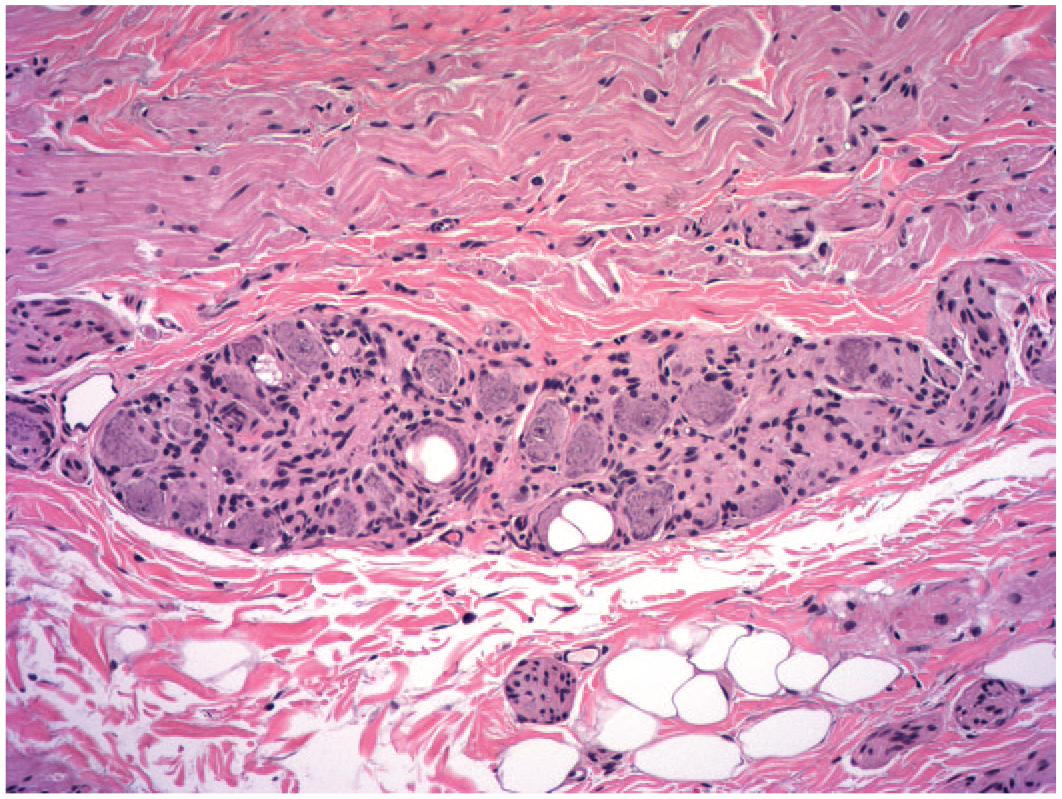
Vacuolation of sensory neurons in the atrioventricular ganglion of the heart showing large, clear, colorless cytoplasmic vacuoles. Affected neurons exhibit no evidence of degeneration. This finding may develop as a processing artifact or as an incidental background change. Juvenile cynomolgus monkey. Hematoxylin and eosin.
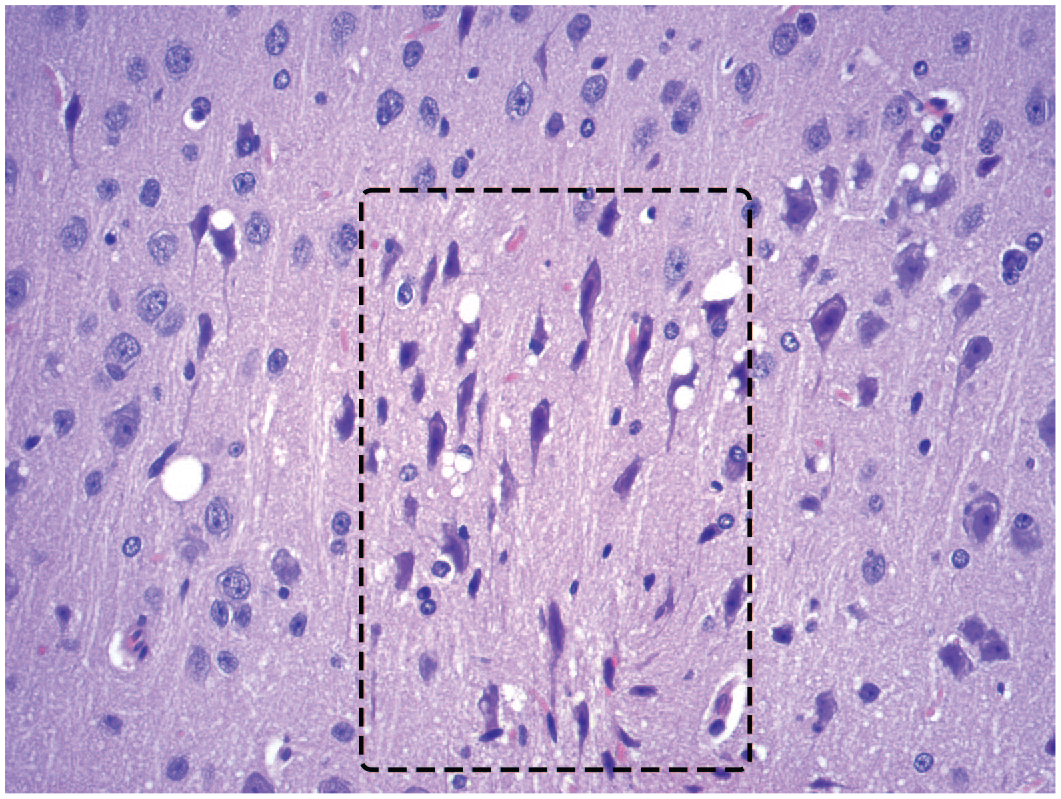
Dark neurons (mainly inside dashed box) in the cerebral cortex of a control animal are a processing artifact characterized by elongated contours and dark basophilic cytoplasm and nuclei. Dark neurons lack visible nucleoli and sometimes possess prominent, twisted axons. Morphologically unaffected neurons outside the box retain their round profiles and possess distinct nuclei, nucleoli, and cell margins. Juvenile cynomolgus monkey. Hematoxylin and eosin.
Some biomolecule-based test articles may lead to accumulation of test article in neurons. For example, ASOs may produce large (“macrovesicular”) or small (“microvesicular”) vacuoles in neurons of NHPs following IT injection. Macrovesicular vacuolation is characterized by a single big (up to 40 µm in diameter), clear vacuole in the perikaryon of affected neurons (Figure 9). 81 Microvesicular vacuolation appears as multiple small vacuoles in the perikaryon of neurons spread throughout the CNS, including cerebral cortex, cerebellum (Purkinje cells), brainstem, and spinal cord (Figure 10). Similar microvesicular vacuoles occur occasionally in various neurons as well as choroid plexus epithelium and/or macrophages after administration of polyethylene glycol (PEG)–conjugated biomolecules. 82 Macrovesicular vacuolation has been shown to represent lysosomal accumulation of the test article using immunohistochemistry and transmission electron microscopy, 81 and occurs without inducing neuronal degeneration or necrosis. Importantly for ASOs, macrovesicular vacuolation is observed in fixed but not flash-frozen tissue, indicating that it represents extraction of ASOs as a processing artifact and is of no relevance to human risk assessment. 81 Microvesicular neuronal vacuolation could be due to lysosomal accumulation of ASO material. Investigative work is ongoing to confirm the mechanism of this change.
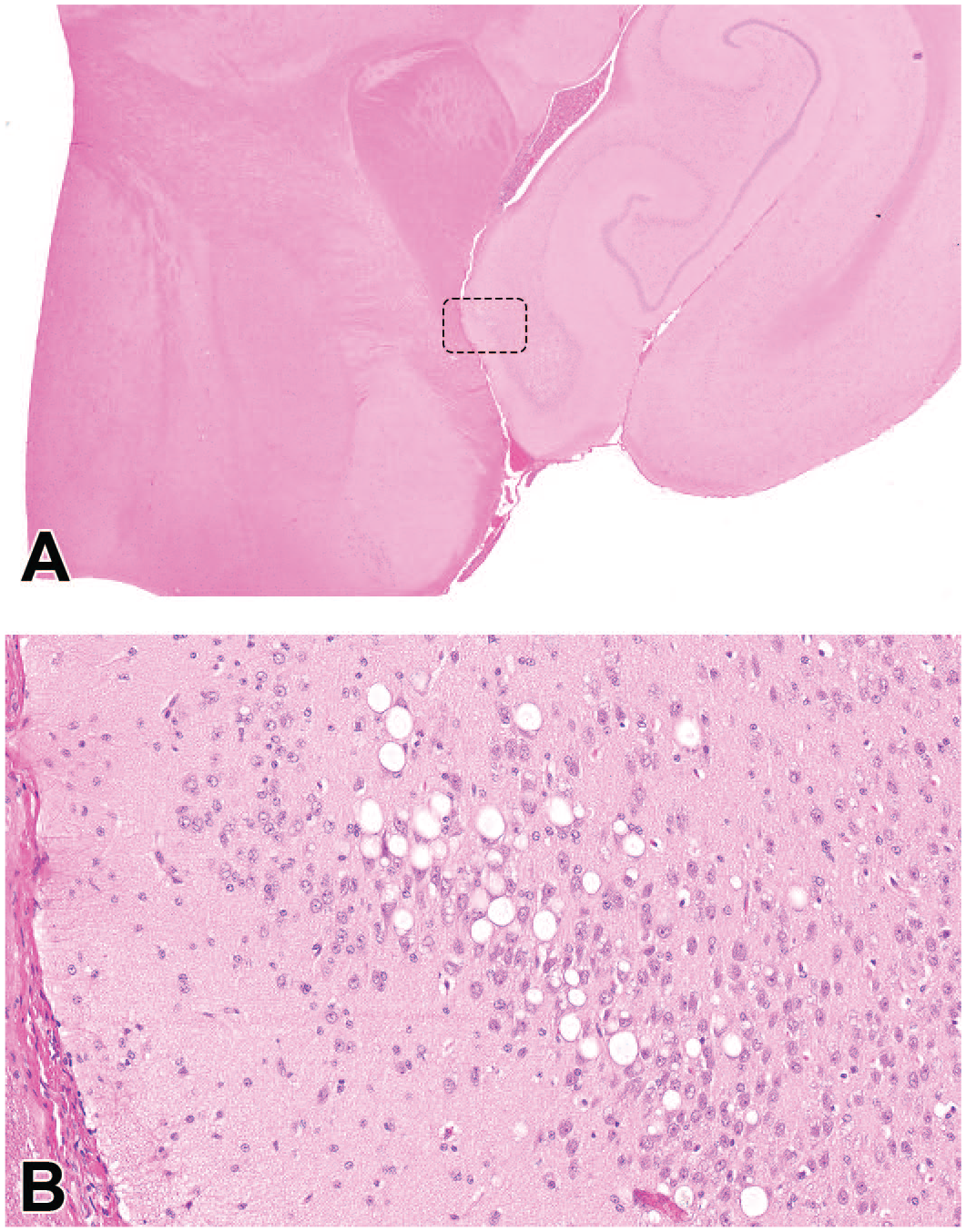
Macrovesicular vacuolation of hippocampal neurons following intermittent intrathecal administration of an antisense oligonucleotide (ASO), typically noted in the hippocampal CA1 region. Neuronal cytoplasm of many neurons in a small domain of the hippocampus (dashed box in Panel A) are greatly expanded by round, often large, clear, colorless vacuoles (Panel B) representing lysosomes that have accumulated ASO (which was lost in processing). The neuronal cytoarchitecture is within normal limits, indicating that the cells are viable and not degenerating. Juvenile cynomolgus monkey. Hematoxylin and eosin.
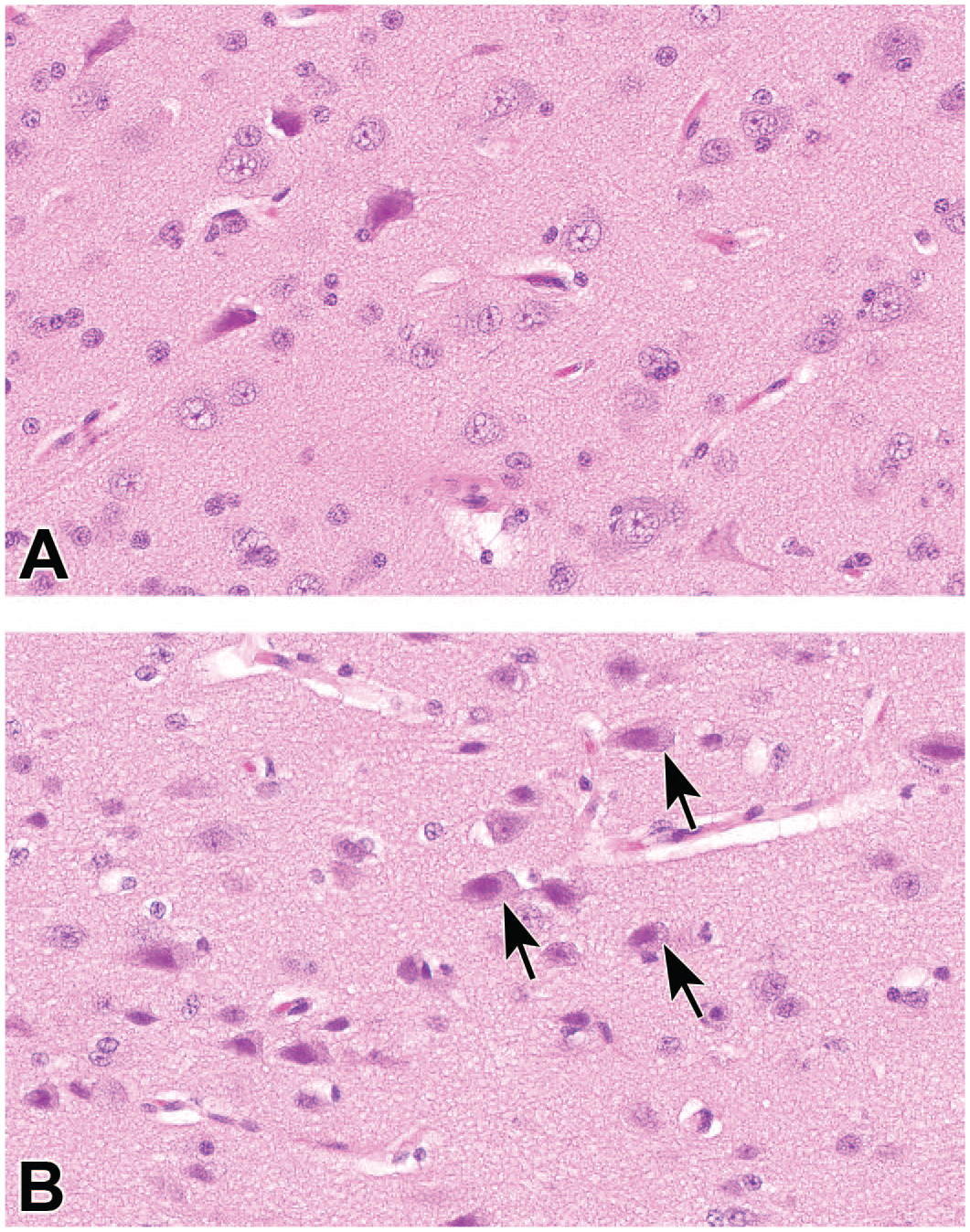
Microvesicular vacuolation in neurons of the cerebral cortex following intermittent intrathecal administration of an antisense oligonucleotide (ASO). Relative to neurons in a control animal (Panel A), the cytoplasm of many pyramidal neurons (arrows) in a treated animal (Panel B) is slightly expanded by very small vacuoles. The pericellular clear spaces, dilated perivascular (Virchow-Robin) spaces, and slightly dark nuclei represent artifacts of the immersion fixation process. Juvenile cynomolgus monkey. Hematoxylin and eosin.
Glial Cells
Multiple glia exist in various neural tissues. 83 Astrocytes are the major glial cells in the CNS, fulfilling such functions as metabolic support for neurons (including production and clearance of neurotransmitters), microenvironmental control, and protection (by maintaining the BBB). Microglial cells are responsible for CNS immune surveillance and synapse pruning during postnatal development. Oligodendrocytes produce the myelin sheaths that insulate CNS nerve fibers, thus speeding the rate of impulse transmission. In the PNS, SGCs in ganglia fulfill many astrocyte functions while Schwann cells are the principal myelinating cells. Key cell type–specific glial biomarkers used in nonclinical toxicity studies include GFAP for astrocytes, Iba1 for microglia, oligodendrocyte transcription factor 2 (Olig2) or Sox-10 for oligodendroglia, glutamine synthetase for SGCs, and Sox-10 or S100 for Schwann cells. 56 Where warranted, other biomarkers also are available for demonstrating various glial cells. 84
The chief glia-focused effects induced by biomolecule test articles in NHPs are related to increased cell numbers (sometimes in association with enhanced cell size). “Gliosis” is a nonspecific term indicating a nonneoplastic rise in CNS glial numbers (Figure 11). This term generally is assigned for H&E-stained sections where the glial cell type has not been confirmed using a cell type–specific biomarker. Gliosis reflects activation of astrocytes (“astrocytosis”; Figure 12) and/or microglial cells (“microgliosis”; Figure 13), or less frequently oligodendrocytes. 67 The cell type(s) in foci of gliosis may be predicted in H&E sections based on cytoarchitectural features (e.g., cell distribution, shape, and size), but the specific diagnoses are assigned when the cell identities have been confirmed using an IHC method. The use of IHC methods to detect biomarkers that are upregulated in activated glia or where glial cell numbers are increased substantially enhances the sensitivity of neuropathological evaluation as a means of assessing subtle injury in CNS tissues, particularly where overt neuronal effects (e.g., necrosis or decreased cellularity) are lacking.
Gliosis of the gray matter in the ventral horn of the cervical spinal cord following intrathecal administration of a recombinant protein. Clusters of plump oval to fusiform cells likely represent reactive microglial cells. Expected glial cell density is visible in the lower right quadrant of the image. Axonal dystrophy (brightly eosinophilic “spheroids”; arrows) is indicative of potentially reversible degeneration associated with altered axonal transport. Juvenile cynomolgus monkey. Hematoxylin and eosin.

Astrocytosis in the cerebellum following intracisternal administration of an adeno-associated virus (AAV)–based gene therapy vector is shown by enhanced labeling for the biomarker glial fibrillary acidic protein (GFAP) in astrocyte processes (dark brown focus at the image center) in the molecular layer (M). The expected degree of GFAP labeling is seen in the upper right quadrant of the image. Juvenile cynomolgus monkey. GFAP immunohistochemistry with hematoxylin counterstain. G indicates granule cell layer; W, white matter.
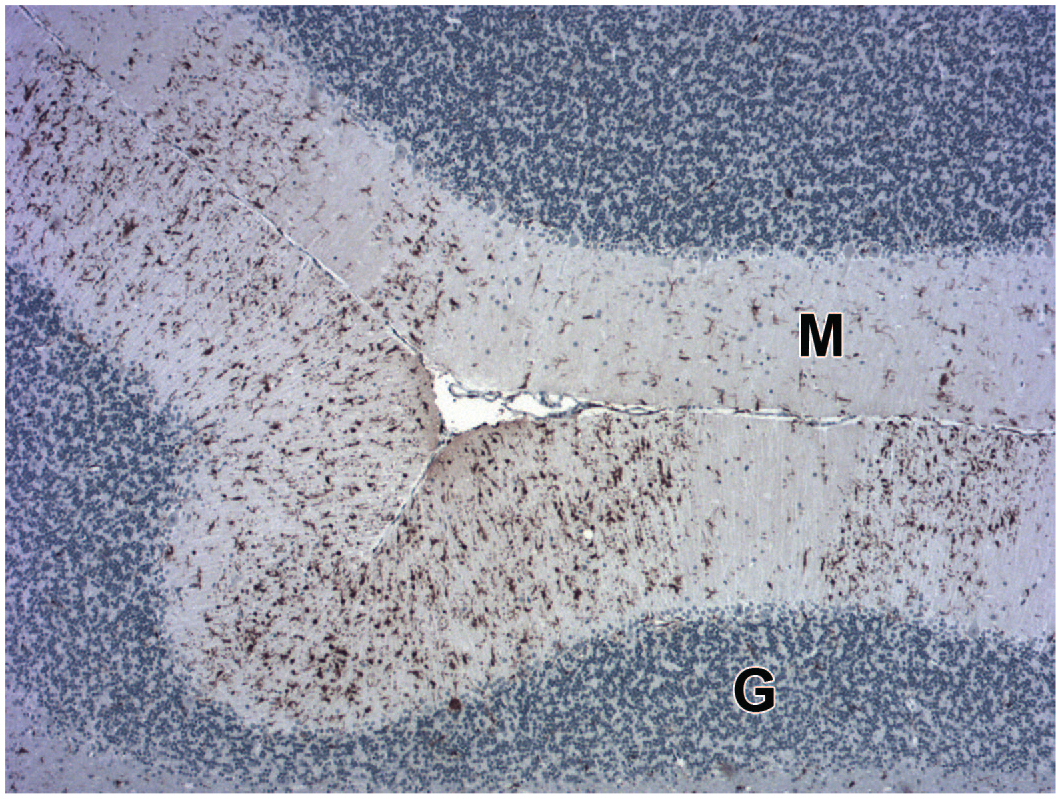
Microgliosis in the cerebellum following intracisternal administration of an adeno-associated virus (AAV)–based gene therapy vector is shown by enhanced labeling for the biomarker ionized calcium-binding adaptor molecule-1 (Iba1) in microglia (dark brown cells) in the molecular layer (M). The expected degree of Iba1 labeling is seen in the molecular layer to the right and below the “M.” Juvenile cynomolgus monkey. Iba1 immunohistochemistry with hematoxylin counterstain. G indicates granule cell layer.
Enhanced glial numbers in PNS ganglia are diagnosed as “increased cellularity, satellite glial cells,” especially where the SGCs form unorganized aggregates (Figure 4). At the discretion of the pathologist, SGC aggregates that exhibit a stellate arrangement (indicative of a persistent glial scar to fill space left by a lost neuron) 66 may be diagnosed separately as “residual nodules” or “nodules of Nageotte” (Figure 14).
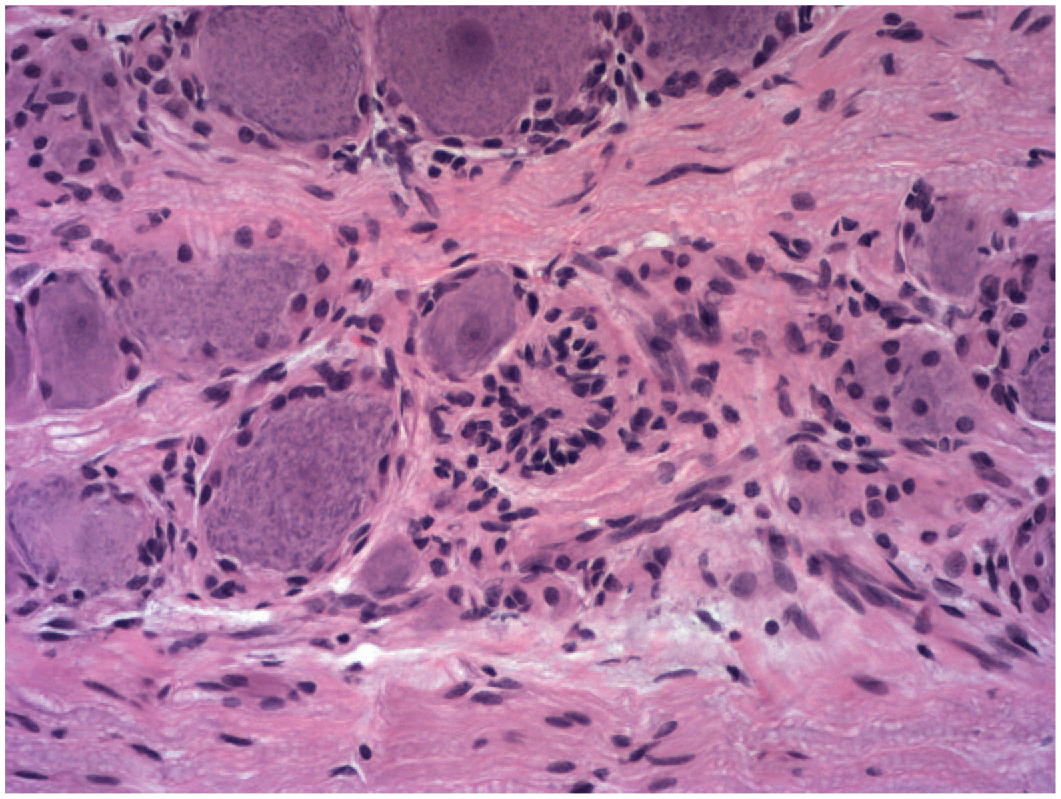
A residual nodule (of Nageotte) in a sacral dorsal root ganglion following intrathecal administration of an adeno-associated virus (AAV)–based gene therapy vector in the lumbar cistern. Such nodules represent foci of satellite glial cell proliferation (i.e., increased cellularity) exhibiting stellate (or starburst) organization which form at sites where necrotic neurons have been lost. Juvenile cynomolgus monkey. Hematoxylin and eosin.
Demyelination (i.e., primary damage to myelinating cells and/or myelin sheaths) is not a common feature in the CNS or PNS following administration of biomolecule-based test articles.
Nonneural Cells
Hemorrhage (i.e., extravascular blood) is a frequent finding following CNS administration of biomolecules. This finding reflects the modest but unavoidable trauma to neural tissues caused by inserting a rigid delivery apparatus (e.g., catheter or needle) into the neuraxis. It rarely presents in perivascular foci of infiltrating leukocytes that have disrupted the mural integrity of the affected vessel. Acute foci are characterized by accumulation of intact red blood cells, while chronic (regressing) foci exhibit clusters of hemosiderin-laden macrophages often along regional blood vessels or within any disturbed local tissues. 85
Leukocyte accumulation is a frequent finding in neural tissues following administration of biomolecule-based test articles directly into the CSF. The change may occur as “infiltration,” which is an influx without damage to the neural parenchyma, or less often as “inflammation” in which the incoming leukocytes are associated with damage in affected neural tissue. The accumulating cells generally are lymphocytes and macrophages, thereby warranting a diagnosis of “infiltration, mononuclear cell.” In some instances, other leukocyte types (usually acute effector cells of the innate immune system such as eosinophils or neutrophils) may occur as well, leading to a diagnosis of “inflammation, mixed cell.” For intraparenchymal and ICV injections, leukocyte clusters may be concentrated near the injection tracks and/or in the peri-ventricular tissues. 85 For ICM and IT injections, the leukocyte clusters typically occur as scattered small foci in the leptomeninges (Figure 15), choroid plexus (Figure 16), and/or in the perivascular (Virchow-Robin) spaces adjacent to penetrating blood vessels (Figure 15) in the brain and spinal cord. Similar clusters may be evident in PNS ganglia (Figure 2) and nerves (Figure 17). Such accumulations may represent a secondary, nonspecific immunostimulatory reaction rather than a direct effect of administering a nucleic acid,86,87 protein, 88 or viral gene vector,89,90 and they also may be observed as incidental background findings.67,91
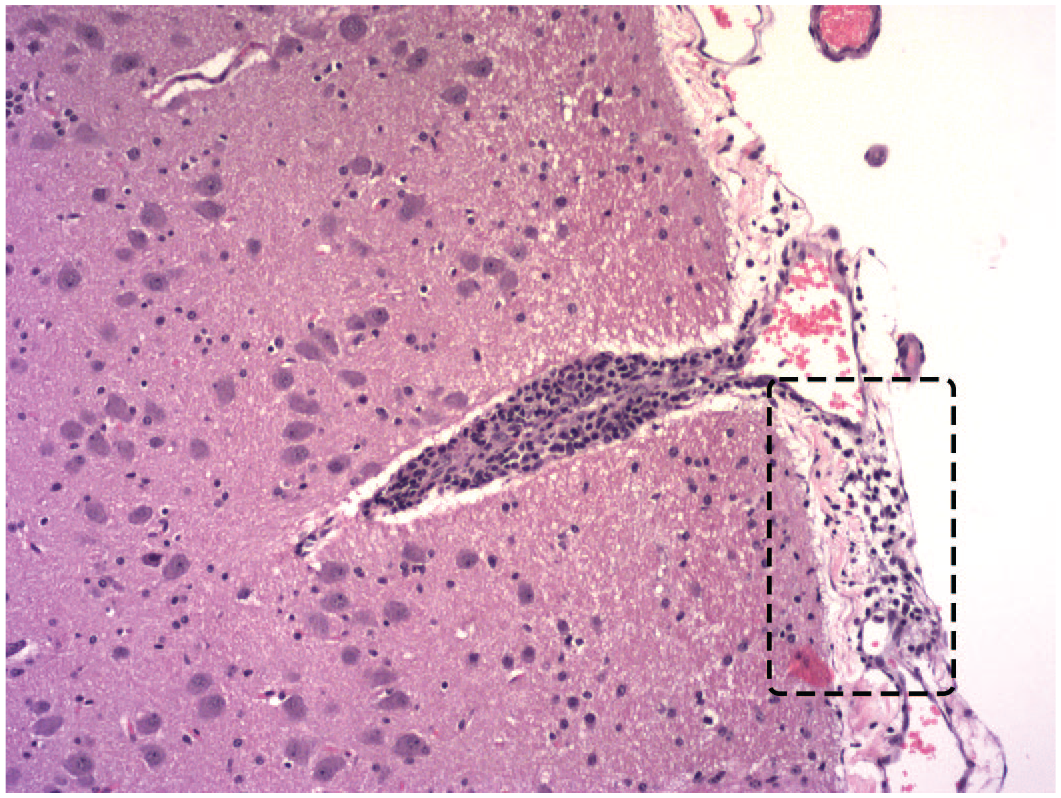
Mononuclear cell infiltration in the leptomeninges (inside dashed box) and filling a nearby perivascular (Virchow-Robin) space of the temporal cerebral cortex following intrathecal administration of an antisense oligonucleotide. The accumulating cells have not damaged the involved neural tissues. Juvenile cynomolgus monkey. Hematoxylin and eosin.
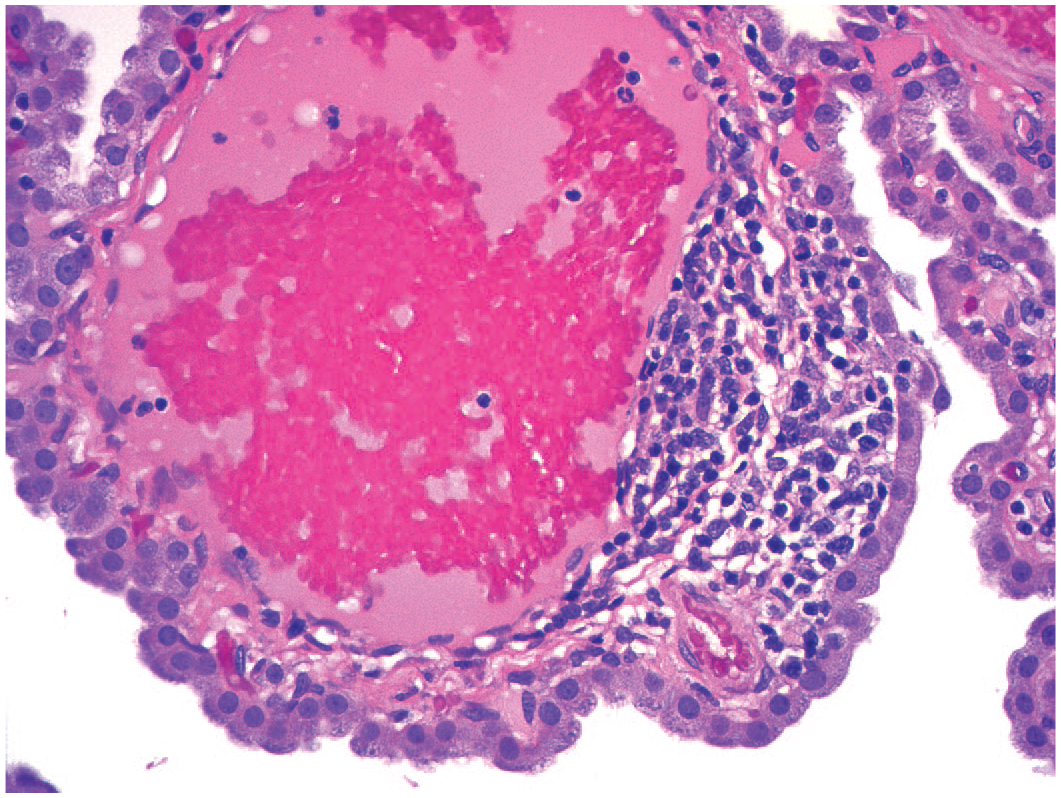
Mononuclear cell infiltration in the interstitium of the choroid plexus in a lateral ventricle of a control animal. The focus appears to contain lymphocytes (small dark cells) and macrophages (larger cells with visible cytoplasm). Juvenile cynomolgus monkey. Hematoxylin and eosin.
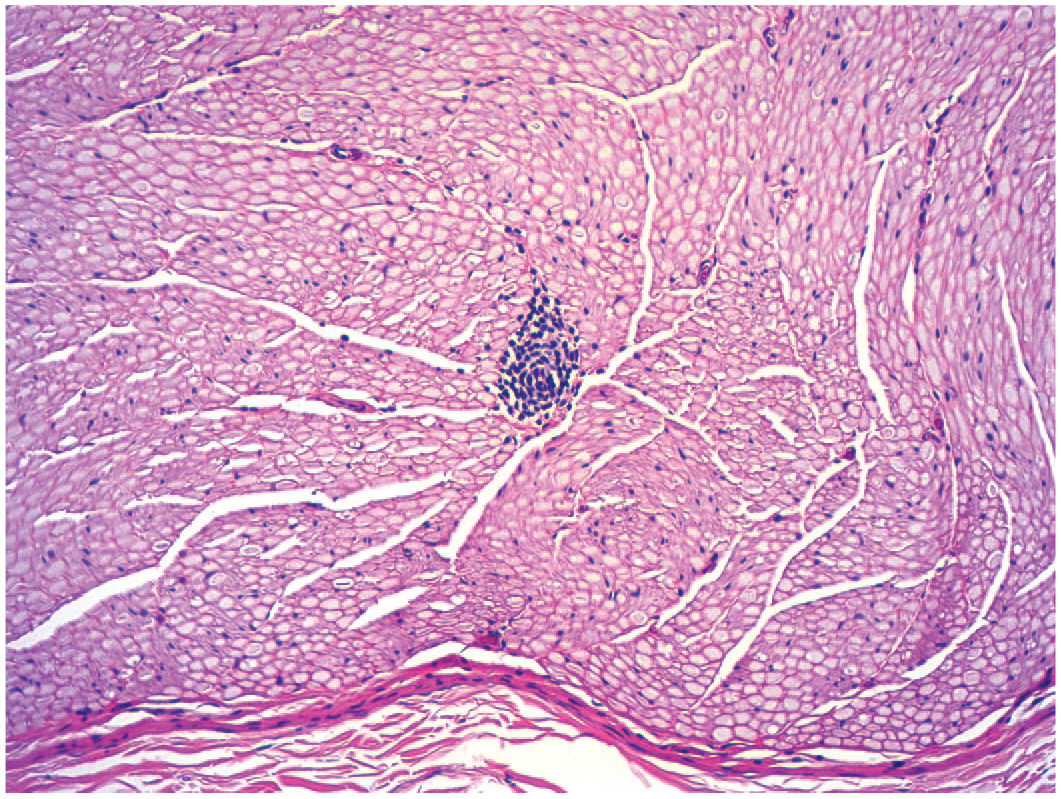
Mononuclear cell infiltration in the sciatic nerve of a control animal. The focus appears to contain mainly lymphocytes (small dark cells). Juvenile cynomolgus monkey. Hematoxylin and eosin.
Macrophages that have infiltrated neural tissues may accumulate test article. For instance, clusters of macrophages within the interstitium of the choroid plexus (Figure 18) or PNS ganglia often develop minute, clear, colorless vacuoles as they remove PEG-conjugated biomolecules. 82 Vacuolated macrophages, sometimes containing basophilic granules, are commonly found in the meninges or in the perivascular space of the brain or spinal cord following IT administration of ASOs (Figure 19).
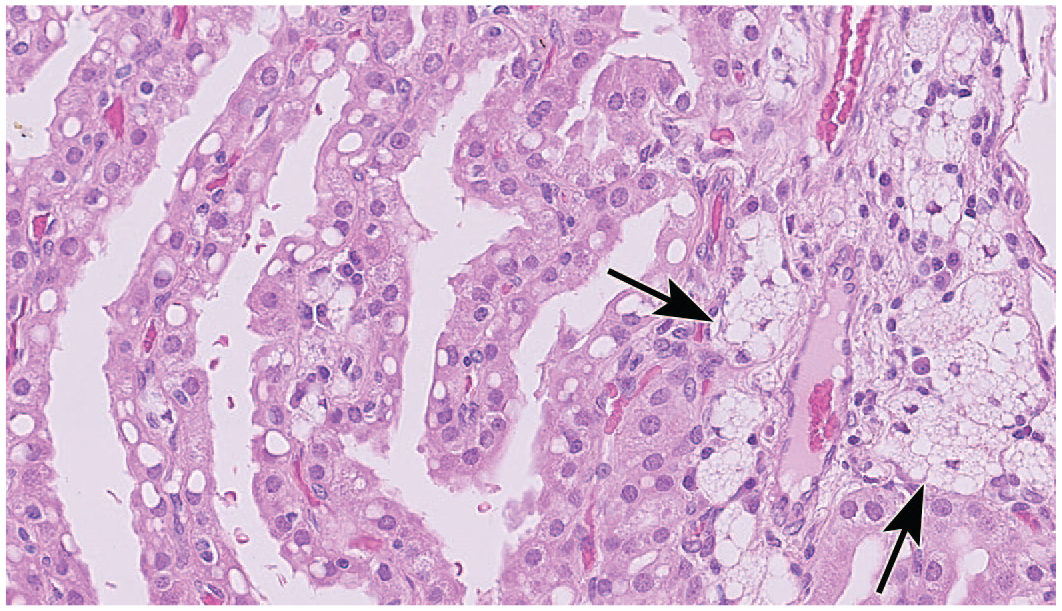
Vacuolation of interstitial macrophages (clustered cells, arrows) and choroid plexus epithelium. Clear, colorless, cytoplasmic vacuoles are filled with polyethylene glycol (PEG) following administration of an unidentified PEGylated biopharmaceutical by an unspecified systemic route. Cynomolgus monkey. Hematoxylin and eosin. (Image republished from Irizarry Rovira et al., 2018, 82 by permission of SAGE.)
Basophilic granules in cytoplasmic vacuoles of macrophages due to accumulation of an antisense oligonucleotide following intermittent intrathecal administration. The affected cells (asterisks) are viable as indicated by their normal cytoarchitectural features. Juvenile cynomolgus monkey. Hematoxylin and eosin.
Meningeal fibrosis is a frequent procedural effect related to chronic implantation of an indwelling catheter to permit intermittent infusion of biotherapeutic agents. 85 This change typically appears as dense connective tissue with relatively few fibroblasts, appearing as an empty ring (Figure 20) or flat plaque (if viewed in longitudinal orientation). In a few cases, the fibrotic reaction also may contain small spicules of foreign material (eosinophilic or sometimes translucent) surrounded by mononuclear and/or multinucleated giant cells (Figure 21). On occasion, fibrosis extends into the brain along the path followed by ICV or intraparenchymal catheters. Fibrous tissue may be differentiated from glial processes by Masson’s trichrome or picrosirius red stains or by IHC methods demonstrating the presence of various collagen types.
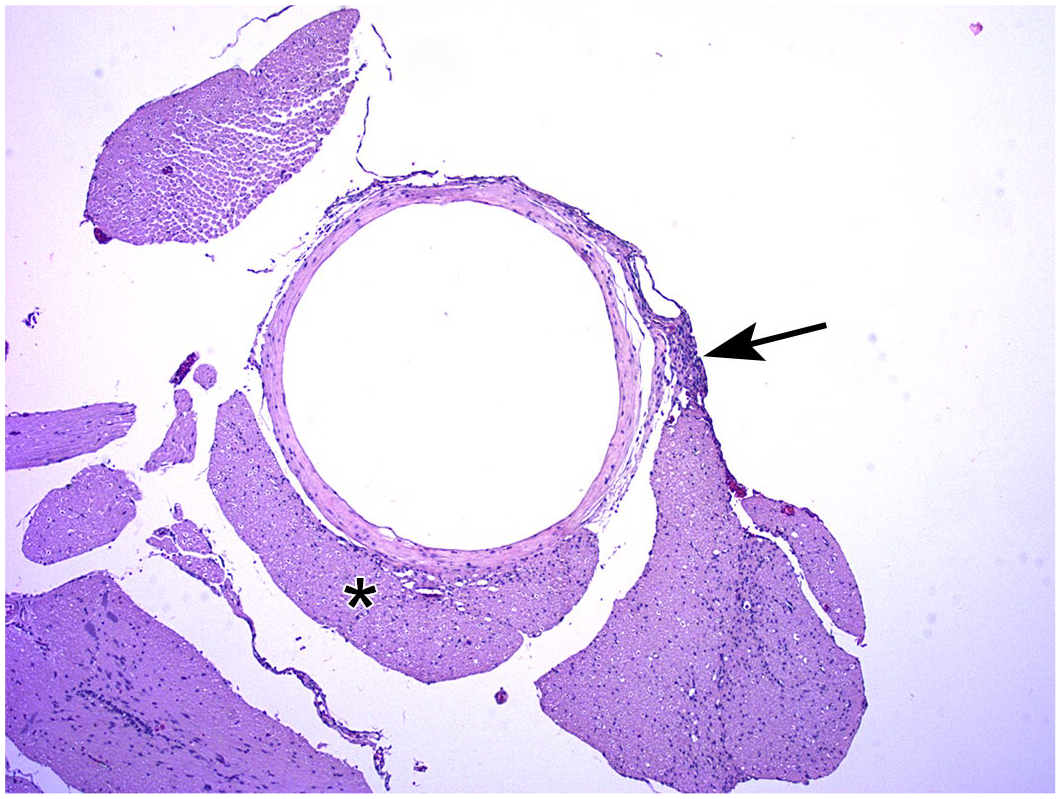
Fibrosis of the meninges surrounding an empty central channel that once held an indwelling catheter for intermittent infusion of a candidate biotherapeutic. A small focus of mononuclear cell infiltration (arrow) is located adjacent to the outer surface of the fibrotic ring. A nearby spinal nerve root (asterisk) is slightly compressed where it follows the surface of the fibrotic tissue. Juvenile cynomolgus monkey. Hematoxylin and eosin.
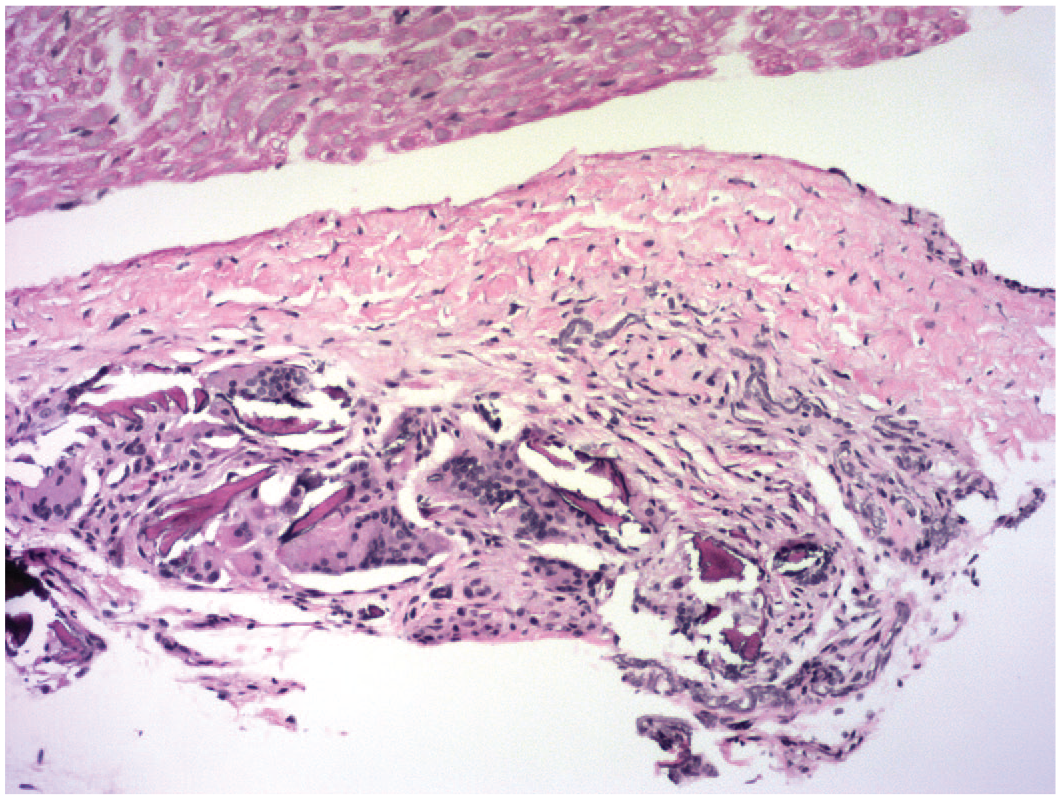
Localized fibrosis (adjacent to a spinal nerve) containing multiple elongated spicules of dark eosinophilic material associated with several multinucleated giant cells. Juvenile cynomolgus monkey. Hematoxylin and eosin.
Melanocytes are occasionally seen as an incidental finding near small blood vessels in the brains of NHPs. Isolated or small clusters of brown-black spindle cells may be observed in the superficial brain (Figure 22) or in the choroid plexus (Figure 23). Melanocyte numbers have not been seen to be altered by administration of biomolecules.
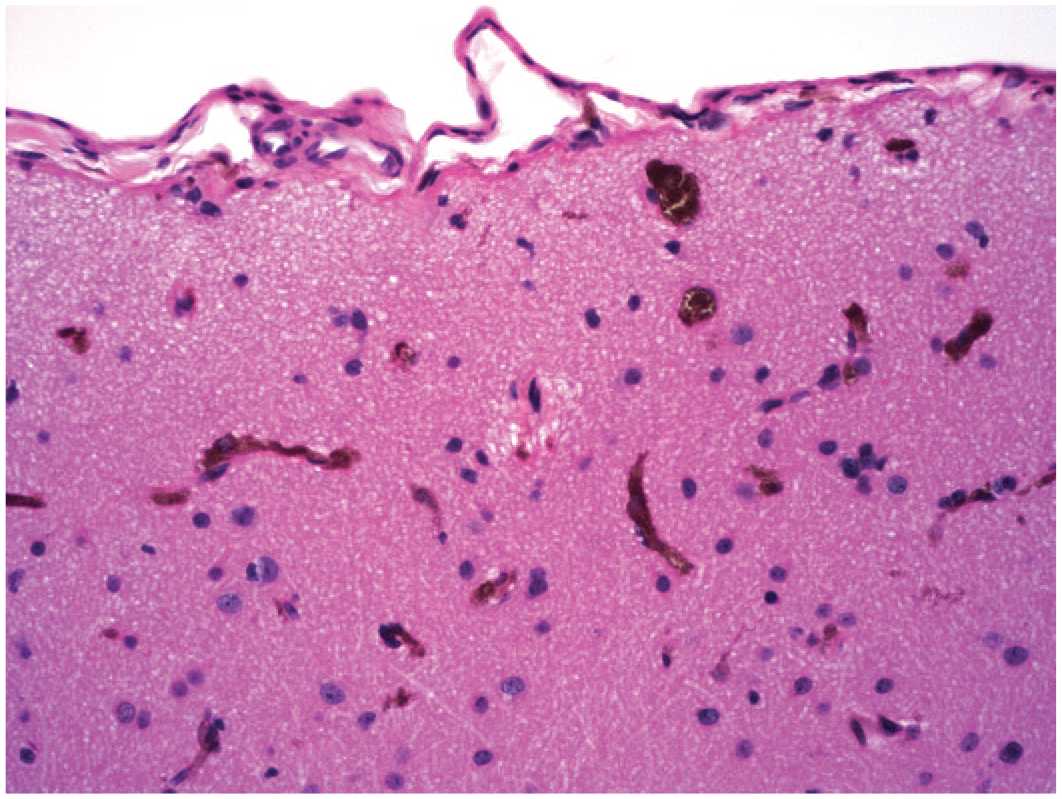
Multiple clusters of melanocytes (dark brown cells) in the superficial cerebral cortex of a control animal. Several elongated curved columns represent capillaries. This change is an incidental finding. More importantly, these cells should not be interpreted to be hemosiderin associated with chronic hemorrhage. Juvenile cynomolgus monkey. Hematoxylin and eosin.
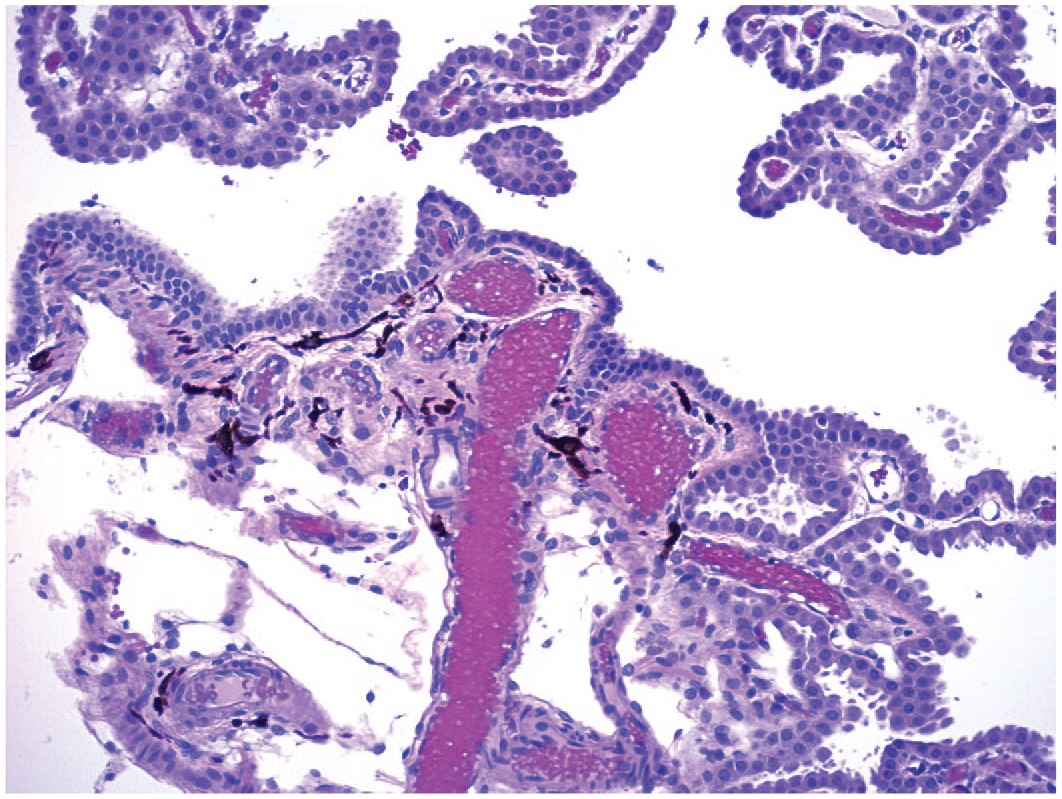
Multiple individual melanocytes (dark brown, stellate cells) in the interstitium of the choroid plexus of a control animal. This change is an incidental background finding. Juvenile cynomolgus monkey. Hematoxylin and eosin.
Miscellaneous Findings
Several neural changes not related to a specific neural cell type may be observed in neural tissues of NHPs. These effects often reflect procedural changes or incidental findings but may be associated with test article toxicity.
In addition to the cell-focused changes described above, transient or permanent placement of a delivery device in neural parenchyma may lead to fluid accumulation or vacuolation that is not associated with specific cells. These findings often are localized closely around the injection track, and therefore are hard to see macroscopically. Frank liquefactive necrosis (diagnosed as “neural tissue loss, cerebrum”) of the cerebral hemisphere may occur as unilateral or bilateral (frequently asymmetrical) cavitation. 68 Such liquefied foci appear macroscopically as depressed, often brown-colored regions (resulting from accumulation of hemosiderin and/or lipofuscin); microscopically, the parenchyma is absent focally and/or is replaced by macrophages (gitter cells), astrocytic processes, or occasionally fibrous tissue as well as scattered newly formed capillaries. This cavitating change may occur infrequently in control animals as a spontaneous change or may develop at sites of intraparenchymal test article administration as a procedure-related effect.
Mineralization of the neural parenchyma and/or vascular wall may be seen as an incidental change in the forebrain (Figure 24). The change usually occurs in the thalamus or basal nuclei and appears as small, irregular collections of globular basophilic material.
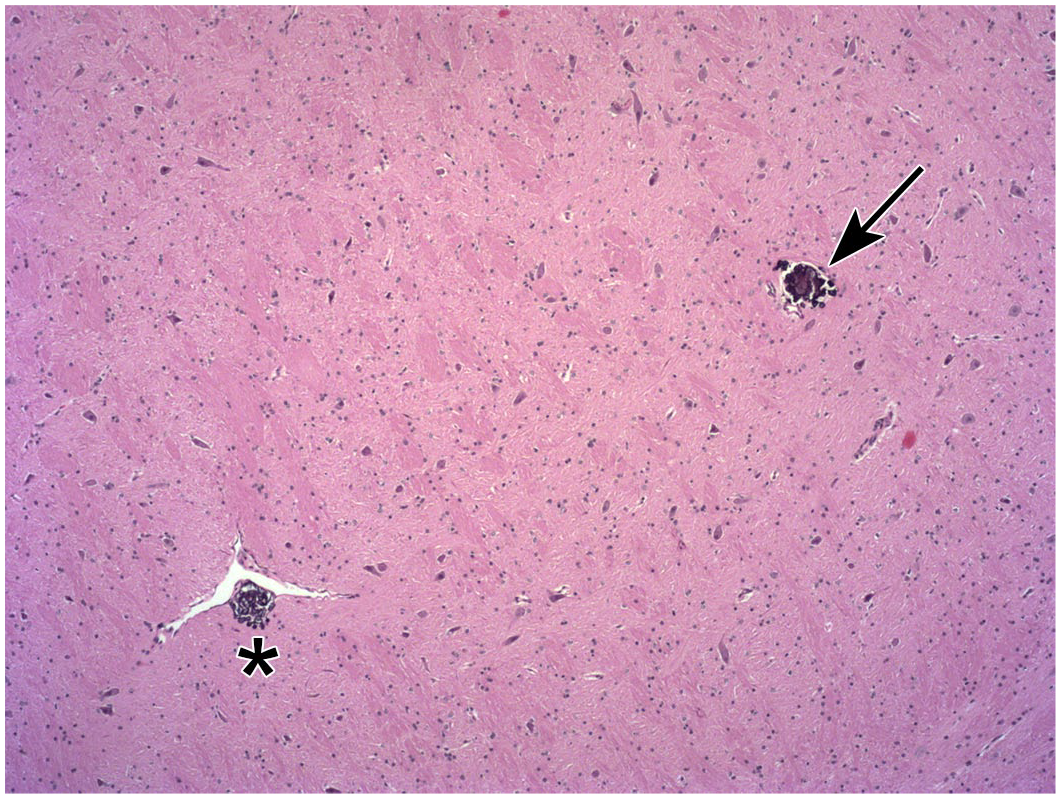
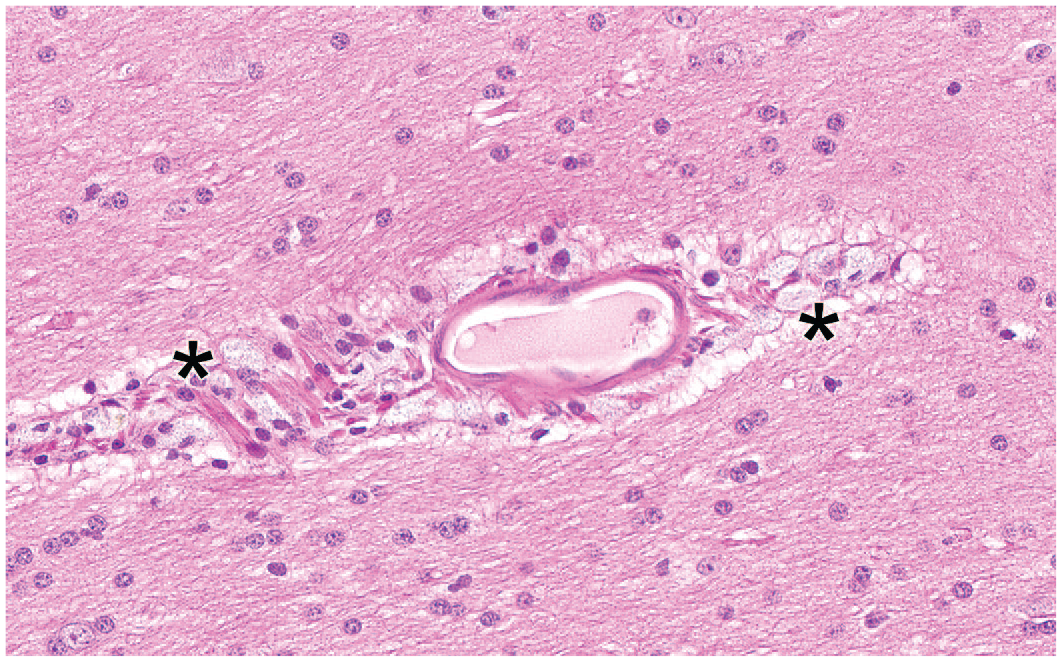
Mineralization of the neuropil (arrow) and an arteriolar wall (above asterisk) in the thalamus of a control animal. This change is an incidental finding. Juvenile cynomolgus monkey. Hematoxylin and eosin.
Mucocytes (metachromatic bodies or Buscaino bodies) are small irregular foci scattered in white matter tracts, most often in the cerebellum, brainstem, spinal cord, and optic nerve (cranial nerve II). The foci appear as empty vacuoles or contain pale blue, homogeneous (Figure 25), or wispy material. The exact nature of these bodies is uncertain, but they are thought to represent an artifact due to inadequate myelin fixation and lipid extraction during histological processing. 92 The important point is that this change should not be interpreted as a test article-related effect.
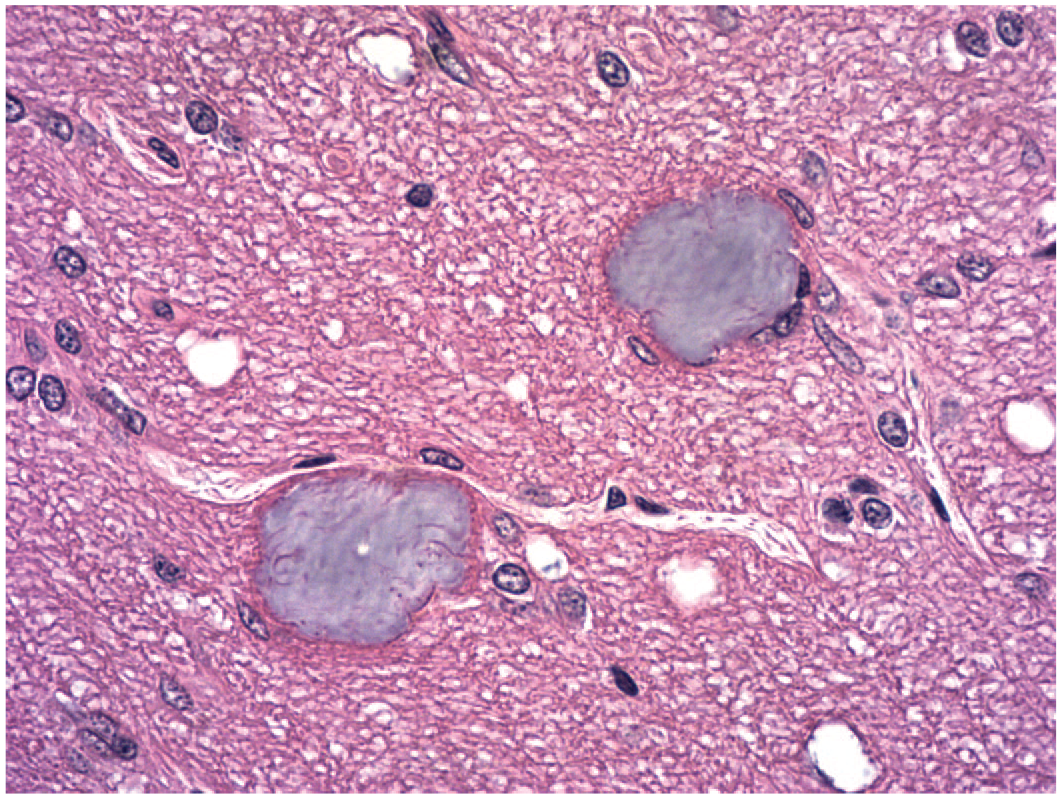
Two mucocytes (metachromatic bodies or Buscaino bodies) appear as irregular, basophilic foci in the optic nerve of a control animal. These foci are thought to be an artifact resulting from lipid extraction during neural tissue processing. Juvenile cynomolgus monkey. Hematoxylin and eosin.
Considerations for Interpreting Neuropathology Data From NHP Nonclinical Studies
Neuropathology evaluation is one aspect of the comprehensive pathology analysis that comprises a key component of nonclinical toxicity studies. As for other pathology endpoints, interpretation of neuropathology data is centered on such factors as their relationship to test article exposure (including the dose, duration, and frequency) and the administration method, character and severity of the diagnoses, and their potential for inducing harm.93,94 Direct delivery of biomolecules to the CNS leads to relatively high-dose exposures, which may induce direct toxic effects on neural cells and/or incite glial or inflammatory cell responses. 85 Direct test article-related changes often are superimposed over procedure-associated effects related to acute trauma (e.g., local hemorrhage or necrosis near injection tracks) or chronic responses (e.g., fibrosis or tissue compression around implanted catheters). 85
Pathology reports for biotherapeutics should clearly communicate several aspects of the neuropathology data set. Findings should be described in the narrative accurately but succinctly. Comparison of test article-treated animals with concurrent control animals is critical in discriminating biomolecules-induced effects from procedure-related changes. Such interpretations are especially challenging for NHP studies due to small group sizes (typically 3-5 animals/sex/group), variable tissue responses within and between studies, and the relative scarcity of relevant historical control data for most novel classes of biotherapeutics. In the end, a crucial element of nonclinical data interpretation is whether or not test article-related and procedure-induced findings are adverse (i.e., cause harm to the affected NHPs).93,94 For example, neuronal necrosis is considered to be inherently adverse due to the limited ability to replace neurons during adulthood. 94 However, modest degrees of neuronal necrosis in DRG that occur as a class effect following AAV administration typically do not produce in-life neurological signs or consistent electrophysiological deficits.70,71,73 Therefore, visible structural effects that do not elicit biologically significant functional disturbances support the continued development of biologic candidates that elicit some degree of unavoidable neuropathology for treating severe neurological diseases with few or no current effective therapeutic options.
Several major trends are helpful when interpreting neuropathology data from NHP studies. Neuronal and glial effects (e.g., degeneration, necrosis, and altered—decreased or increased—cellularity) affecting multiple sites often are direct effects of test article administration. In contrast, those effects found near the site of test article delivery may originate principally as procedure-related effects. 85 Microglia are activated (as indicated by locally increased numbers) within a few days after damage to neurons or neuropil, while proliferation of astrocytes typically develops only 10 or more days after injury occurs. These two glial findings typically indicate secondary activation (hypertrophy and/or hyperplasia) to primary neuronal injury, a reaction to procedure-related parenchymal trauma, or both. In addition, microglial cells as the CNS immune surveillance elements also may respond directly to the presence of biomolecule-based test articles, especially after intraparenchymal injections. Importantly, microglia can present biomolecules as foreign antigen to T-lymphocytes, thus resulting in the generation of an adaptive immune response. 95 Demyelination (i.e., primary damage to myelin sheaths around axons) is rarely induced in the CNS or PNS by biomolecule. Where seen, disintegration of myelin sheaths usually reflects a secondary response to primary neuroaxonal injury or nonspecific destruction of neural tissues at the site of administration.
Conclusion
Various NHP species, primarily cynomolgus (Macaca fascicularis) and rhesus (Macaca mulatta) macaques and sometimes common marmosets (Callithrix jacchus), are the preferred non-rodent animal models for human safety risk assessment of biotherapeutics.38,96-98 In terms of neurotoxicity testing, their relevance is found in both their remarkable neuroanatomic, neurochemical, and neurophysiological similarities to human brain biology and the fact that human-derived biomolecules may exhibit pharmacologic activity in NHPs but often not in other animal species.
Pathologists who analyze and interpret neuropathology findings related to biomolecule-based test articles must be comfortable with both fundamental neurobiological principles and proficient in recognizing common neural findings related to administration of biomolecules. In particular, care is necessary in distinguishing direct test article-induced effects from procedure-related changes and incidental background findings. This article affords a practical guide to diagnosing and interpreting common biomolecule-induced findings in neural tissues of NHPs and as such provides a valuable review of key information needed for the effective practice of NHP neuropathology for many innovative classes of neurotherapeutics.
Footnotes
Acknowledgements
The authors gratefully acknowledge Dr Martin Lamb for sharing his expertise and valuable insights in preparing this manuscript, and Mr Timothy Lanigan (Wadsworth, OH) for his assistance in preparing the figures for this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
