Abstract
The safety of a 2’-O-methoxyethyl antisense oligonucleotide (ASO) was investigated in Mauritius cynomolgus monkeys in a 41-week Good Laboratory Practice (GLP) toxicity study after multiple intrathecal (IT) administrations. Histopathological examination revealed ectopic formation of lymphoid follicles in the spinal cord (SC) at the injection site at all doses and the presence of granular material in neurons of the SC in high-dose animals. The granular material was seen in all the segments of the SC, but mainly in the lumbar segment and persisted at the end of the 26-week recovery period, while the lymphoid follicles showed a reversibility trend. Findings associated with repeated IT administration of ASOs have been described in nonhuman primate (NHP) toxicity studies, specifically in the brain, but findings in the SC are rarely reported. In the present study, we report a high incidence of findings in the SC compared to brain, especially in the lumbar segment in proximity to IT injection sites. An extensive panel of immunohistochemistry markers showed that the ectopic lymphoid follicle formation (LFF) had a cellular composition and organization consistent with tertiary lymphoid structure (TLS) without associated axonal damage in the adjacent nervous tissue. In situ hybridization with an miRNA probe complementary to the ASO revealed that the granular material represented a dose-dependent ASO accumulation in the cytoplasm of neurons without inducing cell death or apoptosis. Glial and ependymal cells in the SC also showed dose-dependent accumulation of the ASO preceding detection of granular material by hematoxylin and eosin (H&E). Based on these molecular localization data, the presence of LFF in SC suggests a chronic local immune activation. Considering the absence of neuronal dysfunction or injury and transient clinical signs previously reported with other 2’-MOE ASOs, the presence of TLS and ASO was considered non-adverse.
Introduction
Antisense oligonucleotides (ASOs) represent an emerging class of therapeutic agents that have achieved significant progress in recent years, with approximately 20 ASOs approved globally for clinical use; most of which have been authorized in the past four years.6,10 The burgeoning success of ASOs is largely due to substantial advancements in chemistry, target specificity, and drug delivery systems, coupled with the efficient synthesis of sequence libraries tailored against validated disease targets. 13 Although the nonclinical safety assessment and toxicological effects of ASOs are well-documented following systemic administration to laboratory animals, especially rodents,1,9 there is scant literature on the toxicity following intrathecal (IT) administration in nonhuman primate (NHPs). Published ASO-related effects post-IT administration in NHPs include neuronal vacuolation (related to fixation artifact primed by intracellular ASO) and gliosis in the brain, as well as mononuclear cell (MNC) infiltration and macrophage accumulation in the meninges of the brain, spinal cord (SC), and associated nerve tissues.3,12 The study involved multiple IT administrations of 2’-O-methoxyethyl antisense oligonucleotide (2’-MOE ASO) for 41 weeks and revealed novel SC findings in addition to findings in the brain as described in the literature, persisting after a recovery period. Notably, lymphoid follicle formation (LFF) at the injection site and gray to brownish granular material in SC motor neurons were observed in animals at all doses. This case report details our investigative efforts to characterize the SC findings and to elucidate their nature and potential pathogenesis using molecular techniques to support assessment of any potential adversity.
Materials and Methods
Animal Study Design and Material
The NHP animal study was performed at Labcorp Early Development Services GmbH, Münster, Germany, and was performed in compliance with the GLP Regulations, as outlined in German Chemical Law, annex 1 to §19a Chemikalien Gesetz; the Organization for Economic Co-operation and Development (OECD): Principles of GLP, ENV/MC/CHEM (98) 17; the Consensus Document, The Application of the OECD Principles of GLP to the Organization and Management of Multi-Site Studies, ENV/JM/MONO(2002); and according to the study plan and subsequent amendments. Studies conducted in compliance with the preceding standards are in accordance with the GLP standards required by US Food and Drug Administration (FDA) GLP regulations.
The cynomolgus monkeys used in this study were from Mauritian origin; at initiation of dosing, animals were approximately 2 to 3 years old and weighed 2.7 to 4.1 kg.
A 2’-MOE ASO compound was administrated at 3 different dose levels (low, intermediate, and high) by multiple IT administrations (lumbar bolus of 1 mL volume) for a duration of 41 weeks followed by a 26-week recovery period. Three dose sites were utilized for IT administration of the test item, L3-L4, L4-L5, and L5-L6. However, microscopic findings reported were observed at L4-L5 and L5-L6. Groups of n = 6 animals (n = 3 animals per sex) were used for each test article dose in the main study phase. Two groups of n = 4 animals (n = 2 animals per sex) treated either with vehicle or high-dose 2’-MOE ASO was maintained on the study for a 26-week recovery phase. The IT route of administration was chosen because it represents the human therapeutic route. The vehicle used in this study was an artificial cerebrospinal fluid (CSF).
Clinical observations, body weights, estimated food consumption, physical and neurological examination, ophthalmology, electrocardiography, and clinical pathology evaluation were performed during the study.
Blood and CSF were collected for toxicokinetic (TK) evaluation and/or anti-drug antibody (ADA) determination. Blood was collected for neurofilament light chain (NfL) measurement. At necropsy, tissues from each animal were collected in neutral buffered formalin with an appropriate fixation time allowing immunohistochemistry (IHC) investigation. The blocks were subsequently trimmed and embedded in paraffin wax, sectioned at a nominal 5 µm, and stained with hematoxylin and eosin (H&E). The central nervous system (CNS) and peripheral nervous system (PNS) sampling were done according to the Society of Toxicologic Pathology (STP) position paper 5 and included brain, dorsal root ganglion at cervical, thoracic, and lumbar levels (taken at mid-cervical C6 to C8, mid-thoracic T3 to T5, and mid-lumbar L1 to L2 and L4 to L5), trigeminal ganglion, injection site, sciatic nerve, SC (cervical, lumbar, thoracic), and spinal nerve root (cervical, lumbar, thoracic). The left hemisphere was preserved for microscopic observation.
All tissues were examined microscopically by the study pathologist, and molecular localization work was performed on the same tissue blocks prepared for the histological examination consecutively to the H&E slide.
Determination of 2’-O-Methoxyethyl Antisense Oligonucleotide Levels
The blood samples (0.2 ml for at least 0.1 ml plasma) were collected from the vena cephalica antebrachii or vena saphena into K2EDTA tubes from all animals at different time points, including samples prior to dosing, multiple time points after the first dose and at the end of main and recovery periods. The blood samples were centrifuged to prepare plasma and were stored at 70°C or below until analysis. Quantification was performed by using biotin capture reagents and Alexa Fluor detection reagents. The solutions were passed over streptavidin beads within the Gyrolab Bioaffy CD microstructure (Gyros Protein Technologies AB, Uppsala, Sweden). The Alexa Fluor reagent bound to the 2’-MOE ASO allows the assay to be visualized by laser-induced fluorescence detection. The concentration of the 2’-MOE ASO in sample was back-calculated from a calibration curve.
Anti-Drug Antibody Detection
Blood samples (0.2 ml) for the detection of anti-2’-MOE ASO antibodies were collected, prepared for plasma, and stored the same way as for the determination of 2’-MOE ASO levels2’-MOEbio. The determination of ADA in plasma of treated animals was performed by an enzyme-linked immunosorbent assay (ELISA) using 96-well plates coated with 5’-end and 3’-end biotinylated 2’-MOE ASO oligos and a peroxidase-conjugated recombinant protein as a detection reagent. An anti-2’-MOE ASO monoclonal antibody was used as a positive control.
Neurofilament Light Chain Quantification
The serum samples analyzed were collected the same way as described above. The serum samples were thawed and analyzed for the detection of NfL at a 5-fold dilution using a qualified, bead-based immunoassay according to the manufacturer’s instructions (Quanterix Corporation, Billerica, Massachusetts, Simoa NF-light Advantage Kit for SR-X, reference #104073) and read on an SR-X Ultra-sensitive Biomarker Detection System (Quanterix Corporation). Results were fit to a Log-Log line and extrapolated in SoftMax Pro software v5.6 (Molecular Devices, San Jose, California).
Molecular Localization
The molecular localization work consisted of a panel of several localization methods, eg, special stains, IHC, in situ hybridization (ISH), autofluorescence performed on formalin-fixed paraffin-embedded (FFPE) tissue of the IT injection site and SC (lumbar section) sectioned at 3 µm thickness.
Immunohistochemistry in the LFF was performed using the following antibodies: CD21 (follicular dendritic cells), CD20 (B-cells), CD3 (pan T-cells), CD138 (plasma cells), immunoglobulin (Ig) G, and IgM (immunoglobulin deposition). The analysis was complemented with ionized calcium-binding adaptor molecule 1 (IBA1) for microglia activation, as well as NfL as a marker of axonal injury in the nerve.
The nature of the granular material on the SC was investigated with an anti-cytochrome C (mitochondria function); Nissl stain (nuclei and rough endoplasmic reticulum); anti-cleaved caspase 3 (apoptosis marker); ISH with an miRNA probe complementary to the ASO sequence (test article detection); anti-Lysosome-associated membrane protein 2 (LAMP2) (lysosomal membranes); and auto-fluorescent lipofuscin detection (neurodegeneration/lysosome trafficking). The details of the antibodies or ISH probes and protocol are available in Supplemental Table 7.1.
All the IHC staining was performed using the automated Ventana instruments Ventana Discovery XT or Discovery ULTRA (Roche Diagnostics Schweiz AG, Rotkreuz, Switzerland). All buffers and chemicals were purchased from Roche Diagnostics. The FFPE tissue sections were deparaffinized and rehydrated under solvent-free conditions, followed by an antigen retrieval step. The primary antibodies were incubated at room temperature for 1 to 6 hours, followed by a multimer secondary antibody and a 3,3’-Diaminobenzidine (DAB) chromogen detection (ChromoMap kit used following the manufacturer recommendations). Slides were counterstained with hematoxylin and bluing reagent and mounted using Eukitt (Medite, Dietikon, Switzerland) mounting medium.
The ISH method for the visualization of the test article 2’-MOE ASO was performed manually using the miRNAscope HD Detection—Red kit (ACD, reference 324530) provided by ACD (Bio-Techne AG, Zug, Switzerland). An miRNA Positive Control Probe-SR-RNU6-S1 (ACD, reference 727871-S1), targeting a common housekeeping gene, and an miRNA negative control Probe-SR-Scramble-S1 (ACD, reference 727881-S1) were used in addition to the miRNA probe of interest, named ASO, designed by ACD to hybridize to the test article 2’-MOE ASO. The tissue sections of SC were deparaffinized and post-fixed overnight in 10% NBF (J.T. Baker, reference 3933.9010) at room temperature, boiled in 1X Target Retrieval reagent for 15 minutes at 99°C, followed by a Protease III incubation for 20 minutes at 40°C in a HybEZ II Oven (ACD, 321720). The miRNA 2’-MOE ASO probe was hybridized for 2 hours at 40°C in the HybEZ II Oven. The signal was amplified using a multiple-step process following the supplier recommendations. Fast Red was used as a detection reagent and Hematoxylin II (Roche Diagnostics, 05277965001) and Bluing reagent (Roche Diagnostics, 05266769001) as counterstain. After drying, the slides were mounted with EcoMount (Biocare Medical, reference EM897L).
For the lipofuscin detection, the FFPE SC tissue sections were only stained with DAPI (NucBlue Fixed Cell ReadyProbes Reagent, Invitrogen R37606) for 10 minutes at room temperature after dewaxing and mounted with ProLong Gold Antifade Mountant (Invitrogen P36934). The slides were scanned on the Nanozoomer S60 slides scanner (scanning software NDP-Scan Version 2.5, Hamamatsu Photonics France, Swiss Office, Solothurn, Switzerland) in fluorescent mode using a TRITC red filter (excitation wavelength at 514 nm and emission wavelength at 600 nm) for the acquisition of the results using a 448-ms exposure time.
The Nissl body stains were performed on the FFPE SC tissue sections stained with FD Cresyl Violet solution (reference PS102, FD Neurotechnologies) for 6 minutes after dewaxing and briefly rinsed in 100% ethanol. Subsequently, slides were gradually dehydrated in ethanol and mounted using Eukitt mounting medium.
All the stained slides were scanned with the Nanozoomer S60 digital scanner (scanning software NDP-Scan Version 2.5, Hamamatsu Photonics France, Swiss Office), and the staining results were analyzed by visual examination. No quantitative image analysis was performed.
Results
All animals survived to their scheduled necropsy with no test article-related effect on the food consumption, ophthalmic, cardiovascular and physical examination, hematology, coagulation, serum clinical chemistry, and urine analysis during dosing and recovery phase. Transient test item-related neurological findings such as absent spinal reflexes were observed at all dose levels, with a dose-dependent increase in the number of affected animals, severity, and frequency of observations. These changes were recorded 4 hours postdose and fully reversed within 24b to 48 hours following dosing in all animals. These transient findings are well-described non-adverse class effects for ASOs administrated via IT injection. 4
Determination of 2’-O-Methoxyethyl Antisense Oligonucleotide Levels
In the plasma, the highest concentration of 2’-MOE ASO was observed between 1.00 and 4.00 hours post-dose both on the first day as well as on the last day of administration. The plasma exposure of the 2’-MOE ASO increased with the increasing dose across all dose levels after single and multiple administrations. No difference in the plasma 2’-MOE ASO exposure could be measured between the first and last day of dosing, and no sex difference was noted. Exposure to ASO in different brain regions increased with increasing dose. Spinal cord (lumbar > thoracic > cervical), frontal cortex, hippocampus and substantia nigra were generally most exposed, whereas the lowest exposure was measured in the putamen.
Anti-Drug Antibody Formation and Correlation With Histopathology
The incidence of ADA induction to 2’-MOE ASO in plasma was increased in all groups during the dosing phase. Compared to vehicle control, ADA titers were considered positive in 37% of animals at low dose beginning on week 16, 31% of animals at intermediate dose beginning on week 8, and 44% of animals at high dose beginning on week 12.
The animals with ADA detected in plasma generally exhibited a higher exposure in plasma compared to the animals that were ADA-negative. The animals with ADA detected in plasma have been found to present a nondose-dependent positive correlation with the LFF at the injection site. The details of the animals with ADA formation and LFF are shown in Figure 1.
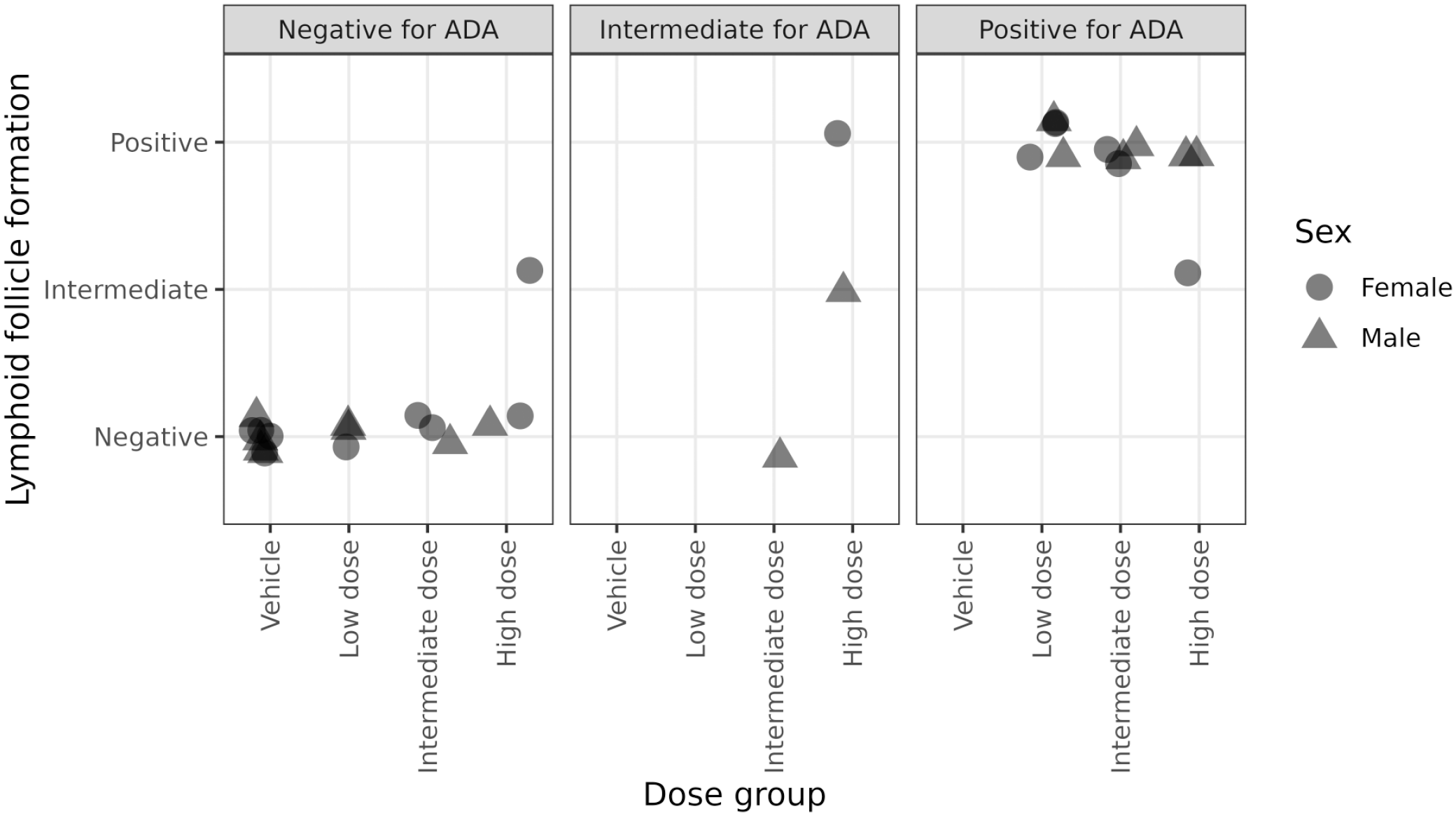
Individual correlation between ectopic lymphoid follicle formation (LFF) (no follicle, intermediate size, or well-formed) at injection site and the presence of anti-drug antibodies (ADA) in the plasma of 2’MOE-ASO-treated animals.
Neurofilament Light Chain Quantitation in Serum
No 2’-MOE ASO-related increase in serum NfL was observed relative to all pretest and vehicle control results. Levels of serum NfL above individual pretest and vehicle controls were observed at the end of dosing in one animal administrated at low dose and in one animal during the recovery phase previously administered at high dose. Levels of similar magnitude were observed prior to dosing in one female and in one female dosed with vehicle during the recovery phase. Such cases indicate that any apparent increase likely reflects biological variability or procedure-related changes as previously reported, 4 such as from sampling or IT administration.
All other NfL levels in animals administrated with 2’-MOE ASO test article were within the range of vehicle control or were of negligible magnitude and considered as likely biological variation (Figure 2).
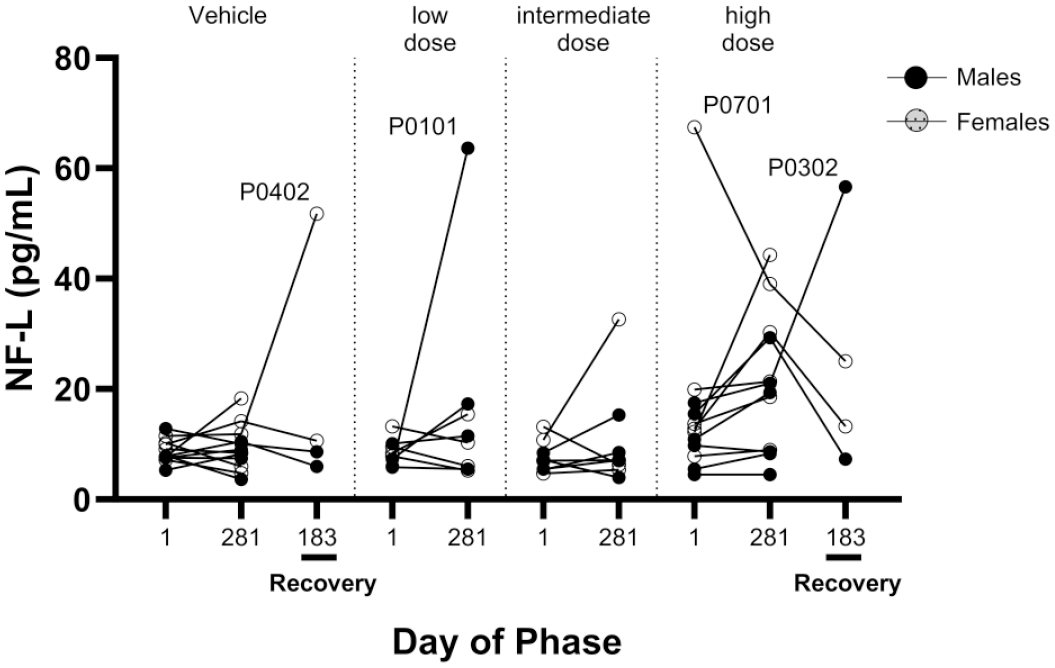
Determination of NfL level in plasma of 2’MOE-ASO animals at the end of treatment and after recovery.
Microscopic Findings in the Central and Peripheral Nervous Systems
End of treatment phase
The ASO-related findings were observed in the central and PNSs of animals at all doses.
Central nervous system
In the hippocampus, minimal cytoplasmic vacuolation of neurons was observed in two and six animals at intermediate-dose and high-dose groups, respectively. Vacuolation was characterized by the presence of a single, clear, well-defined large vacuoles (macrovesicular) or single or multiple smaller vacuole (microvesicular) in the cytoplasm of individual neurons. Both forms of vacuolation were generally noted in affected animals. Vacuolated neurons showed no degenerative changes and were not accompanied by glial cell reaction in the adjacent neuropil. In addition, minimal to moderate severity of macrophages containing cytoplasmic granular material (granular macrophages) were observed in the brain in several animals administered low dose, and all animals administered intermediate or high dose.
In the SC segments, minimal granular macrophages and minimal to slight MNC infiltrates were noted in the meninges and/or adjacent epidural fat tissue in several animals at all dose levels. In addition, minimal to slight MNC infiltrates were noted in the gray and white matter and/or within the ventral root spinal nerves of the lumbar SC. Neurons containing minimal to slight amounts of granular material were observed in the cervical, thoracic, and/or lumbar SC segments of several animals at high dose (Figure 3). The material was located in the perikaryon adjacent to the nucleus and was characterized by round, eosinophilic granules of approximately 1-3 µm in diameter, often surrounded by a pale halo giving the impression of microvesicular vacuolation. The observation was most prominent and with the highest incidence and severity in the lumbar SC in proximity to the IT injection site (L4-L5 and L5-L6).
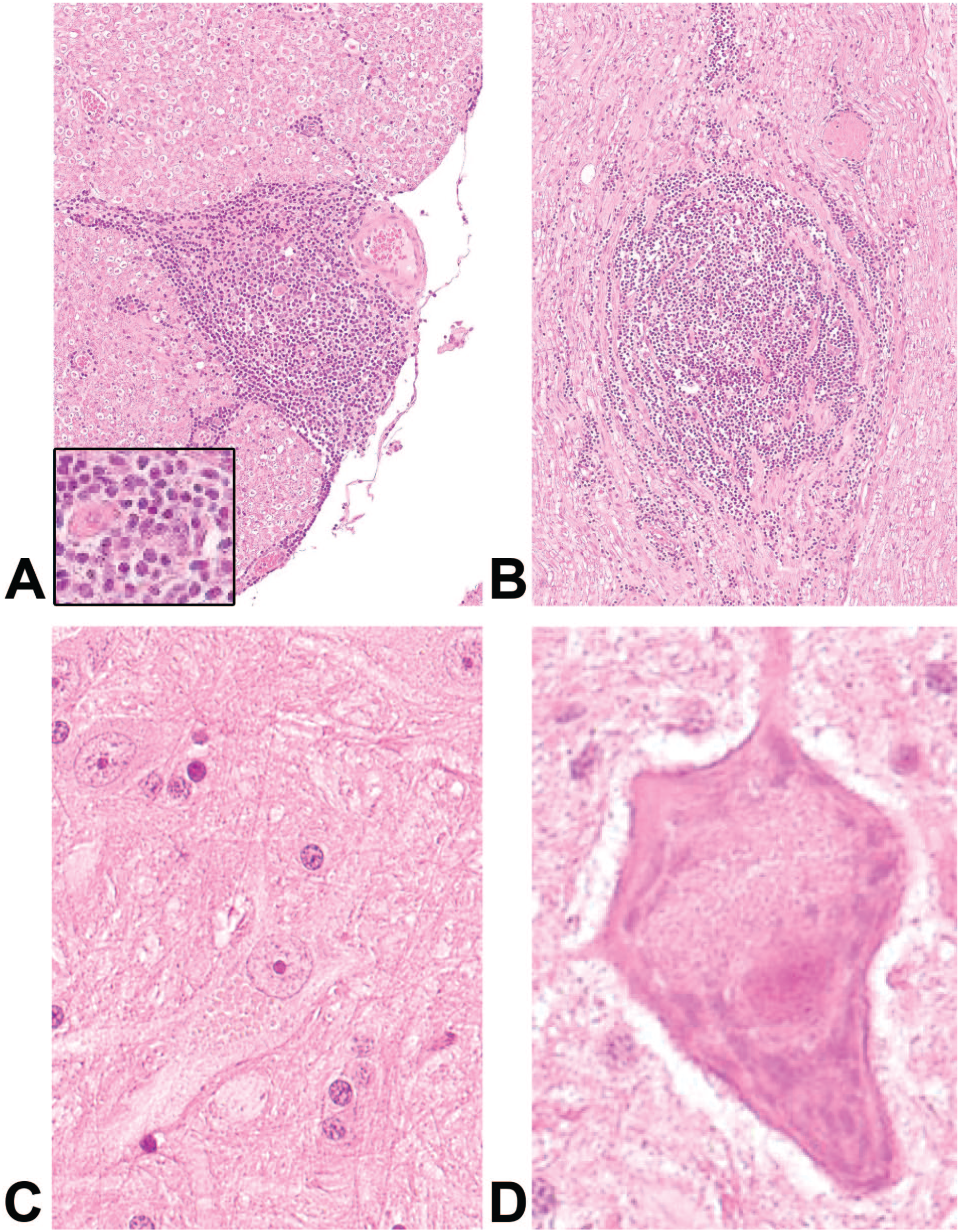
Histopathology of ectopic lymphoid follicle formation (LFF) at injection site in spinal cord at the end of treatment (A) and after recovery (B). Histopathology of granular material in motor neurons of lumbar spinal cord at the end of treatment (C) and after recovery (D). Magnification 40× (1A, B, C) and 80× (1D).
At the IT injection site, minimal to moderate granular macrophages and slight MNC infiltrates were observed in the meninges and/or adjacent epidural fat tissue in several animals at all doses and were often accompanied by slight to moderate MNC infiltrates in the adjacent neuropil organizing into LFF (Figure 3).
Peripheral nervous system
In the cervical, thoracic, and/or lumbar dorsal root ganglia, minimal to slight granular macrophages were noted in a few animals at low or intermediate dose and in several animals at the high dose. Slight to moderate MNC infiltrates were noted in the thoracic and lumbar dorsal root ganglia, adjacent fat, and/or fibrous tissue, and in the dorsal root spinal nerves of the thoracic and lumbar SC segments of a few animals at low dose and several animals at intermediate and high doses. As in the SC, these infiltrates organized at higher severity into LFF. LFF was predominantly located around blood vessels. Likewise, granular macrophages in the brain, spinal cord, IT injection site, and DRG were predominantly located around blood vessels, but in the brain, they were also dispersed within the neuroparenchyma.
Overall, test article-related changes in the peripheral and/or CNSs included neuronal vacuolation, granular macrophages, and MNC infiltrates. In the SC, the findings were most prominent in the lumbar SC, associated compartments (lumbar dorsal root ganglion and lumbar spinal nerve), and the IT injection site. These findings clearly decreased in incidence and/or severity over the thoracic toward the cervical segment.
End of the 26-week recovery phase
The ASO-related findings at the end of the recovery phase were observed in both central and PNSs (evaluated in controls and high-dose animals only).
Following a 26-week treatment-free period, most test article-related findings had either reversed or had decreased severity and/or incidence. Mononuclear cell infiltration either decreased or was absent depending on location [still present in the IT injection site (Figure 3), thoracic and lumbar SC as well as dorsal root spinal nerves]. Similarly, granular macrophages were absent or decreased in severity and/or incidence (still present in the thoracic and lumbar SC, IT injection site, thoracic, and lumbar dorsal root ganglia). The presence of granular macrophages in several tissues including lymph nodes, central and peripheral nervous system is consistent with ASO uptake and accumulation. 8 The granulation of SC neurons was still present (Figure 3).
Molecular Localization Results
Analysis of ectopic lymphoid follicle formation and immune cells at injection site
The cell populations present in the ectopic LFF were identified by IHC. The staining for follicular dendritic cells (CD21) showed a strong positivity restricted to the center of the follicles. Hardly any CD21 positive cells could be detected in the 2’-MOE ASO-treated animals without LFF (Figure 4). Immunoreactivity for CD20 (B-cell marker) was also found in the follicles (Figure 4). The B-cells were strongly stained and located in the center of the follicles. The CD3+ cells (T-cell marker) instead aggregated at the periphery of these follicles (Figure 4). Frequent isolated CD3+ T-cells or organized as clusters were dispersed in the nerve sheath and in the meninges. The 2’-MOE ASO-treated animals without LFF also contained a high number of CD3+ T-cells, present as isolated single cells in the nerve and/or meninges. The control animals did not show any CD3+ cells.
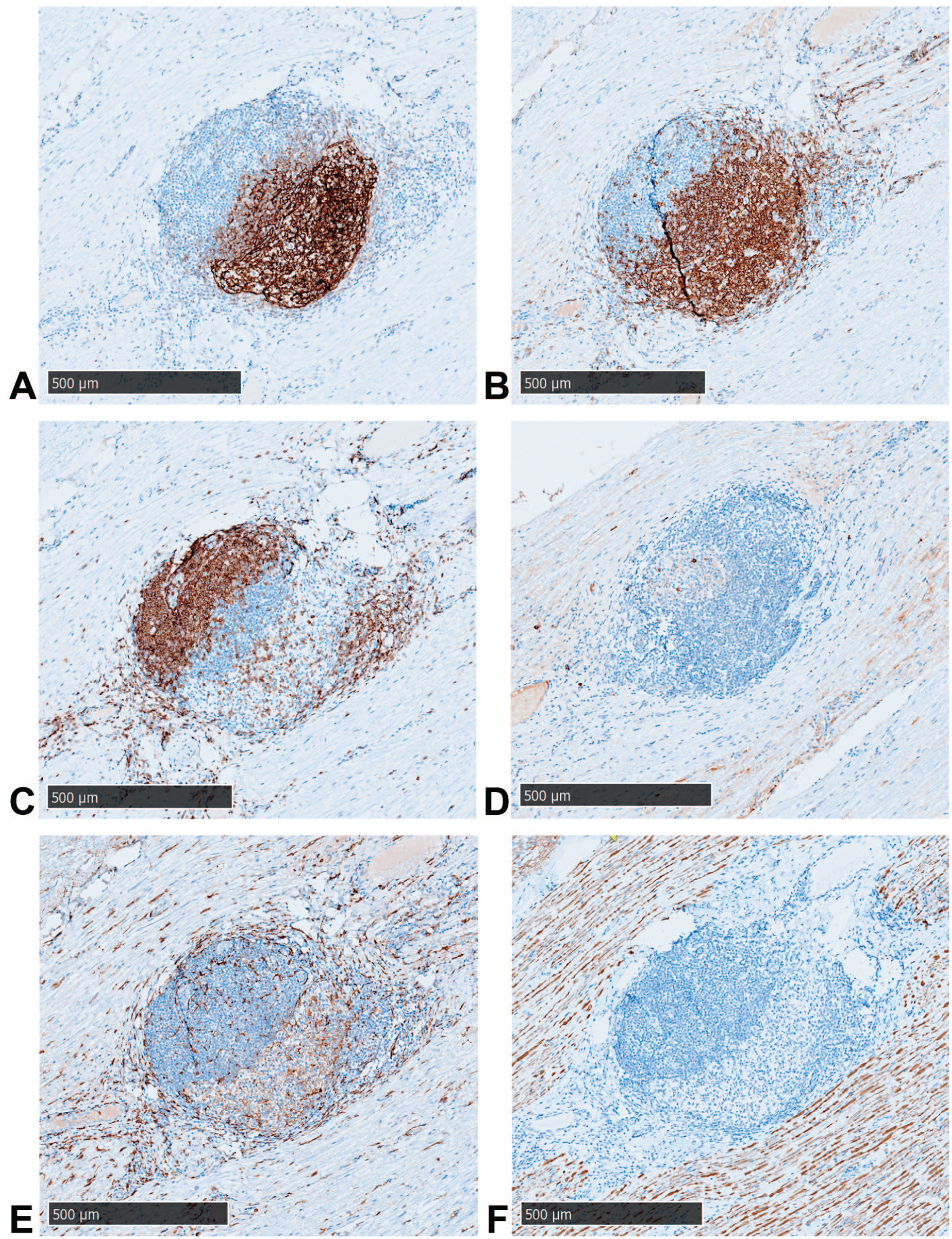
Characterization by IHC of cells present in the ectopic lymphoid follicles formation (LFF) at injection site after repeated treatment at high dose with 2’-MOE ASO (end of treatment period): anti-CD21 (A); anti-CD20 (B); anti-CD3 (C); anti-IgM (D); anti-IBA1 (E); and anti-NfL (F).
The staining with an anti-CD138 (plasma cells) antibody could detect just few positive cells, randomly distributed in the follicles. The 2’-MOE ASO-treated animals without LFF showed a similar frequency of positive cells.
The detection of IgG+ IgM+ cells was challenging due to a strong background signal in sections, particularly in all blood vessels. Positive cells detected in the follicle were rare and often located in the external layers of the follicles (Figure 4).
The evaluation of the lymphoid cell populations in the follicles showed that the number and/or intensity of the positive cells was unrelated to the dose level of 2’-MOE ASO administered and with no sex difference. The results at the end of the dosing period and after the 26-week recovery period were similar and without any change in the proportion of the cells.
To evaluate the impact on tissue structures in proximity to the LFF, further staining for microglia (IBA1) and axonal damage (NfL) was performed on the tissue sections.
The IBA1 stained positively the microglia in the SC sections and in nerve sheaths of the vehicle-administered animals. In 2’-MOE ASO-treated animals, the microglia appeared sometimes to be focally activated (amoeboid shape and retracted processes), but this activation profile was limited to few animals in each group and was not dose related. In the nerve sheaths of 2’-MOE ASO-treated animals, the number of IBA1+ cells appeared clearly increased with cells distributing randomly within the nerve sheaths compared to control animals. The 2’-MOE ASO-treated animals with well-developed follicles showed additionally many IBA1+ cells infiltrating the LFF (Figure 4).
The NfL normally present in the neuronal cell body and axons can get depleted from neurons in case of injury and form compact aggregates in interstitium. In the control animals, a positive staining was found in all neurons of SC and homogenous stain in the axons of the nerve. It was noticed that few animals had minimal focal NfL positive aggregates likely related to the known procedural-related microdamage. In 2’-MOE ASO-treated animals, some axonal damages were detected focally in the nerve sheath and were independent of LFF (Figure 4).
Characterization of granular material in neurons (spinal cord)
To assess the different subcellular components of the cytoplasm, a set of stainings were performed—cytochrome C staining (mitochondria), Nissl stain (nuclei and endoplasmic reticulum), cleaved caspase-3 (apoptosis), lipofuscin (lysosomal trafficking), and LAMP2 (lysosome).
In control animals, staining for cytochrome C revealed a normally homogenous positive stain with a granular appearance in the gray matter of the SC and different levels of staining intensity in neurons; the large neurons tended to be weakly positive. The 2’-MOE ASO-treated animals at any dose level did not show any change in terms of intensity and appearance compared to the vehicle-administered animals (Figure 5). The large neurons, observed after a cresyl violet stain, did not reveal any evidence of dissolution of Nissl bodies and absence of chromatolysis. The only recurrent observation was a displacement of the Nissl bodies in the cytoplasm of the large neurons after 2’-MOE ASO administration at high dose (Figure 5). In line with the absence of chromatolysis, evaluation of apoptosis with an anti-cleaved caspase 3 did not detect any alteration of the neuronal cells after administration of 2’-MOE ASO (Figure 5).
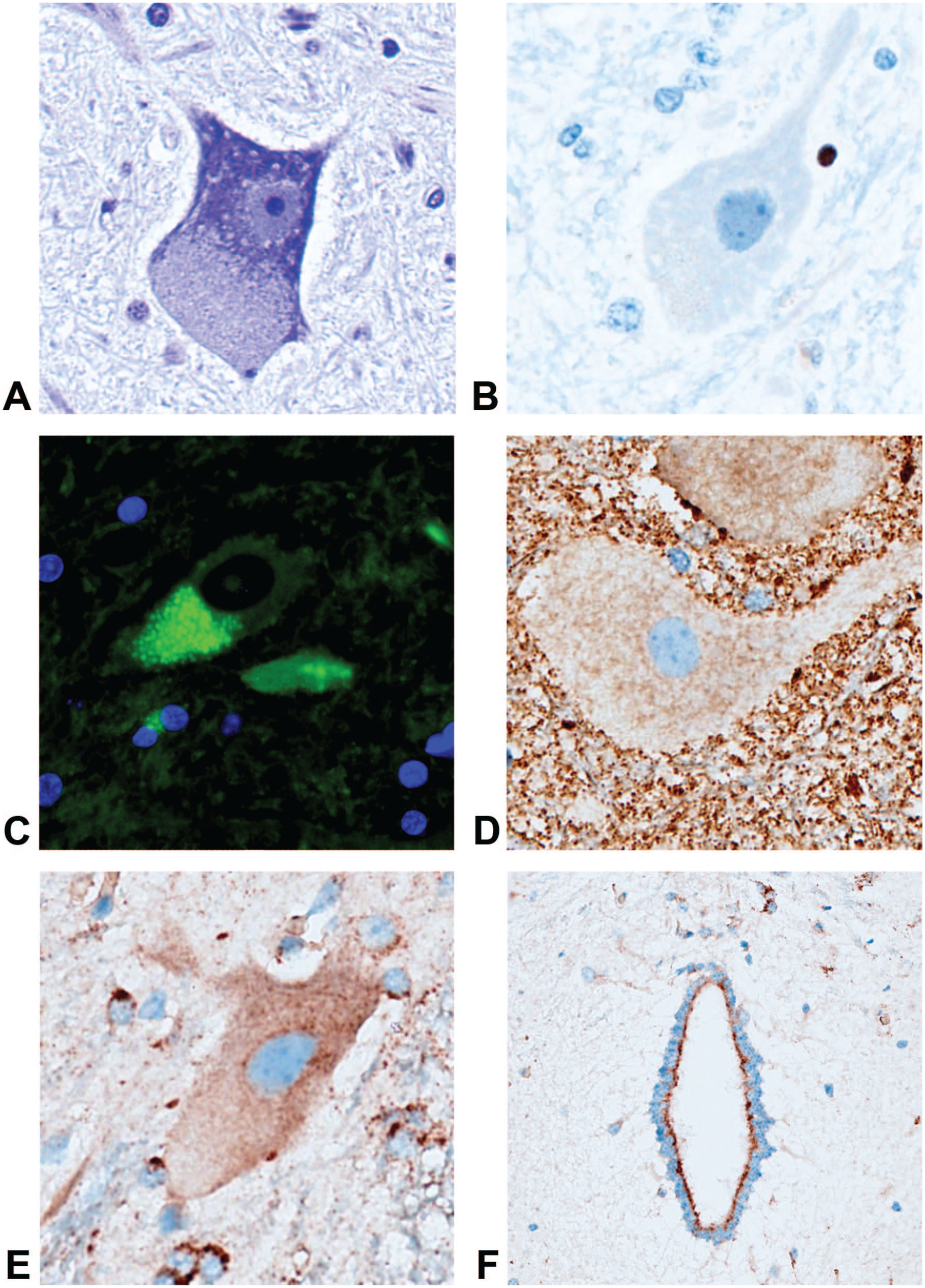
Characterization of the granular material in motor neurons: Nissl stain (A); anti-cleaved caspase 3 (B); auto-fluorescent lipofuscin (C); anti-cytochrome C (D); anti-LAMP2 in neuron (E); and in ependymal cells (F). Magnification 80×.
The lipofuscin tends to accumulate in degenerating neurons or in case of lysosome trafficking perturbation. In fact, neurons in control animals showed just few granules of lipofuscin in the cytoplasm of certain neurons. Neurons in 2’-MOE ASO-treated animals at high dose demonstrated the presence of lipofuscin granules quite frequently, all size neurons being affected as well as a small number of glial cells (Figure 5). Lipofuscin granules were still present in the cytoplasm of the neurons from animals necropsied at the end of the recovery period. Complementing the lysosome assessment by an anti-LAMP2 staining, IHC from control animals revealed a faint cytoplasmic granular stain homogeneously distributed not only in all neurons but also in glial cells. The 2’-MOE ASO-treated animals at high dose showed an increased intensity of the LAMP2 stain mainly in glial cells, indicating lysosomal accumulation. Some animals showed additionally a heterogeneous distribution of the lysosomes in the cytoplasm of the large neurons (Figure 5). The LAMP2 increase was not reversible in animals necropsied at the end of the recovery period. It was interesting to note that the ependymal cells in the central canal of the SC were strongly stained in 2’-MOE ASO-treated animals at high dose (Figure 5).
Finally, we assessed the presence of the 2’-MOE ASO test article in the tissue using an miRNA probe. The SC tissue of control animals was completely negative as expected (Figure 6). Low-dose 2’-MOE ASO-treated animals showed some positivity in the vascular bed, and in glial cells, the cytoplasm of the neurons remained mostly negative (Figure 6). Intermediate dose of 2’-MOE ASO detected the test article in the cytoplasm of the neurons in addition to glial cells and ependymal cells. High dose of 2’-MOE ASO resulted in a positive stain covering most of the neurons and glial cells cytoplasm. Ependymal cells and vessels were also strongly positive (Figure 6). The exact same staining pattern was seen in animals at the end of the dosing period and at the end of the recovery period (data not shown).
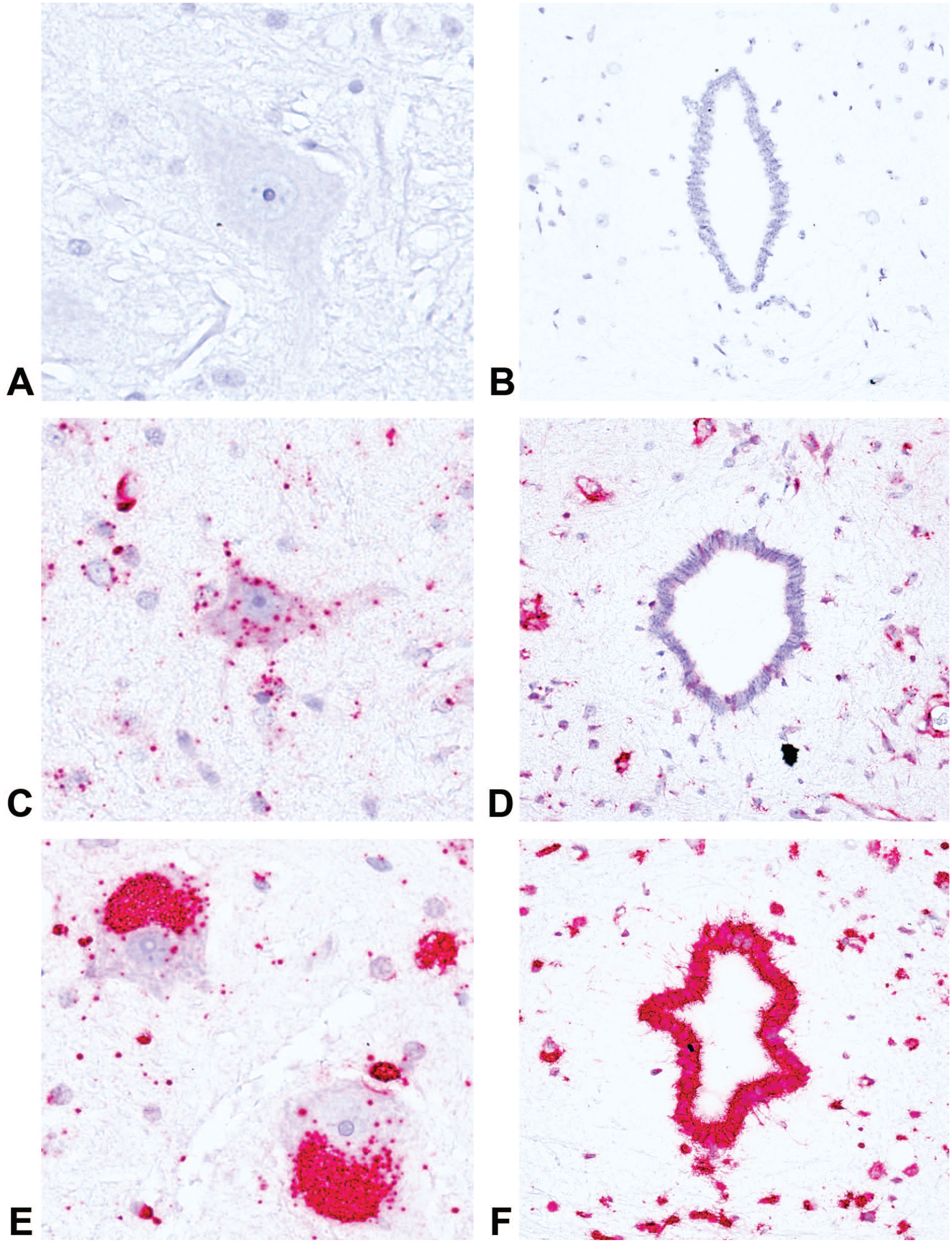
Detection of ASO with an miRNA ISH probe in lumbar spinal cord: vehicle animal (A) and (D); low-dose animal (B) and (E); and high-dose animal (C) and (F).
Discussion
During microscopic evaluation, we observed that MNC infiltrations in the SC, IT injection sites, and dorsal root ganglia often coincided with the emergence of organized lymphoid follicle-like structures (aka tertiary lymphoid structure [TLS]) at all dose groups. Notably, the incidence of these structures was the highest near the IT injection sites and diminished progressively from the lumbar to the cervical SC segments. Although control animals also exhibited MNC infiltrations, the occurrence and intensity were lower, and no LFF was observed. The LFF was still observed after a 26-week recovery period in the high-dose group. The IHC analysis with a comprehensive panel of immune cell markers revealed that these LFF possessed a cellular makeup characteristic of TLS. 15 The INHAND nomenclature for NHPs prefers the term LFF over TLS but describes these as interchangeable. 7 Previous studies have reported MNC infiltrations in the central and PNSs of NHPs post-administration of biologic and nucleic acid-based therapies.2,13 However, to the best of our knowledge, this is the first report of TLS formation in association with such infiltrations. Furthermore, the presence of TLS strongly correlated with the presence of ADA in the affected animals, suggesting a potential role in ADA production. Importantly, we found no morphological signs of axonal swelling or degeneration near the TLS, as confirmed by both HE and NfL stainings and corroborated by the absence of plasma NfL concentrations in affected animals. The presence of LFF did not correlate with transient neurological findings of absence of spinal reflexes. Although certain animals with LFF exhibited these neurological findings, most animals displaying LFF lacked the findings.
The accumulation of ASOs within the cytoplasm of hepatocytes and renal epithelium, manifesting as basophilic granules, is a common finding in nonclinical toxicologic studies following systemic administration.6,8 It has been postulated that ASOs administrated via the IT route are widely distributed in the brain and SC and are taken up by all major cell types including neurons and glial cells, 2 resulting in microscopic findings of granulated or vacuolated macrophages in the meninges (brain and SC) and perivascular (Virchow-Robin) spaces, in addition to cytoplasmic vacuolation of neurons. 3 Despite these findings, the toxicological significance, precise neural distribution, and reversibility of ASO accumulation remain underexplored. Our histological assessment has detected granular material within SC neurons post high-dose exposure, with a notable concentration near the IT injection site, persisting into the recovery phase. The ISH has confirmed this granular material as therapeutic 2’-MOE ASO also present in glial and ependymal cells of the SC central canal. Further special histochemical stains and molecular localization work were performed to characterize the potential for cellular dysfunction caused by the 2’-MOE ASO accumulation. Despite the accumulation, our results, including cytochrome C and cleaved Caspase 3 IHC, along with Nissl staining, have not indicated any mitochondrial disruption, apoptotic induction, or morphological cellular damage in affected cells when compared to control specimens, suggesting a lack of overt cellular dysfunction from 2’-MOE ASO presence.
Lysosomal staining with LAMP2 IHC showed similar patterns in both 2’-MOE ASO-treated and control groups. However, a notable increase in lysosomal accumulation was observed in the glial and ependymal cells, which persisted at the end of recovery period. However, evidence of lysosomal accumulation was not associated with any morphological signs of cellular degeneration or necrosis. The underlying cause of this lysosomal accumulation in non-neuronal cells, especially glial cells, remains unclear. Following the initial internalization of oligonucleotides, they are trafficked through various intracellular vesicular compartments, such as endosomes, multivesicular bodies, lysosomes, and the Golgi apparatus. 11 It is hypothesized that the buildup of ASOs in neurons could lead to an overload of the lysosomal transport system, resulting in a compensatory upsurge in uptake by glial and ependymal cells. In addition, a heightened presence of lipofuscin, as evidenced by autofluorescence, was observed in the cytoplasm of cells in high-dose animals, predominantly within large neurons. This finding aligns with the ASO accumulation and points to a potential disruption in the lysosomal degradation of old residues within these cells, despite the absence of morphological or molecular indicators of cellular degeneration.
The TLS and accumulation of 2’-MOE ASO within the SC, particularly near the IT injection site, were not detected in short term GLP IT repeat-dose toxicity studies conducted on Mauritius cynomolgus monkeys. It is important to note that Mauritian monkeys, used for toxicologic studies involving this ASO, seem to exhibit a more pronounced immune response to ASO compared to Asian-sourced monkeys. This is evident through increased deposition of C3d/C4d on platelet surfaces and elevated serum levels of anti-GPIIb/IIIa IgG antibodies. 16 While thrombocytopenia was not observed in this study, it may suggest that Mauritian monkeys are potentially more susceptible to the immune activation effects of ASOs than their Asian counterparts.
In conclusion, these findings were deemed non-adverse. This conclusion is supported by the absence of cellular dysfunction or injury, as well as the lack of corresponding clinical manifestations. The findings suggest that the presence of TLS and 2’-MOE ASO in this context did not negatively impact cellular health or function or posed harm to the animals.
Supplemental Material
sj-docx-1-tpx-10.1177_01926233241311277 – Supplemental material for Characterizing Antisense Oligonucleotide-Induced Histopathology Findings in Spinal Cord of Mauritius Cynomolgus Monkeys by Molecular Localization Investigation
Supplemental material, sj-docx-1-tpx-10.1177_01926233241311277 for Characterizing Antisense Oligonucleotide-Induced Histopathology Findings in Spinal Cord of Mauritius Cynomolgus Monkeys by Molecular Localization Investigation by Valérie Dubost, Kuno Wuersch, Kelley Penraat, Eric Johnson, Anja Wekkeli, Ramprasad Ramakrishna, Aline Piequet, Géeraldine Greiner, Magali Jivkov, Esther Erard, Regine Hansen, Dominique Brees, Andreas Hartmann, Kamal Kumar Balavenkatraman and Jairo Nunes in Toxicologic Pathology
Footnotes
Author Contributions
Novartis contributed to the study design; participated in the collection, analysis, and interpretation of data; and participated in writing, reviewing, and approval of the final publication.
Declaration of Conflicting Interests
The authors are current or previous employees of Novartis and may hold shares and/or stock options in the company.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Novartis sponsored and funded the animal study described in this publication.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
