Abstract
Many potential drugs for treatment of neurodegenerative diseases, particularly antisense oligonucleotides (ASOs), are administered via lumbar intrathecal injection, because these drugs do not cross the blood–brain barrier. Intrathecal injection is a well-established method in cynomolgus monkeys, a species that is used in preclinical safety assessment when other nonrodent species cannot be used. The authors completed intrathecal ASO administration in over 30 preclinical safety studies (>1000 animals and >4500 dose administrations) during which we observed 3 cases of procedure-related spinal cord necrosis (incidence <0.1%). We describe clinical symptoms, diagnostic approaches, morphological features, and prognosis of this rare injury, and compare these findings with typical drug-related findings of ASOs dosed by intrathecal injection. The low incidence of procedure-related and dose-limiting lesions confines this analysis to a small sample set. The pattern of effects is similar across all monkeys despite differences in age, body weight, and intrathecal injection site. All 3 cases presented a combination of the following findings: blood in cerebrospinal fluid at time of injection, clinical signs that increase in severity within a day of dosing, lameness of both hind limbs, reduced muscle tone, and loss of patellar, foot grip, and/or anal reflexes. In all cases, magnetic resonance imaging (MRI) showed a linear hyperintense lesion in the lumbar spinal cord. In 2 cases, this hyperintensity was associated with evidence of spinal cord edema. We conclude that a pattern of in-life and pathology findings, including noninvasive MRI assessment, is indicative of procedure-related effects.
Introduction
Numerous drugs are currently in development for the treatment of neuronal diseases. 1 -3 Many of these drugs, including antisense oligonucleotides (ASOs), require direct administration into the cerebrospinal fluid (CSF) as they do not cross the blood–brain barrier. 2,3 In this regard, intrathecal (IT) bolus delivery is a well-established route of administration in cynomolgus monkeys (Macaca fascicularis), a species that is used for preclinical safety assessment when other nonrodent species are not sufficiently predictive, for example, due to lack of pharmacological activity. Herein, we describe the clinical symptoms, diagnostic approaches, morphological features, and prognosis of a procedure-related spinal cord lesion that very rarely occurs after lumbar IT administration in cynomolgus monkeys. Differentiating procedure-related findings and drug-related findings is essential in correctly defining a maximum tolerated dose and no observed adverse effect level (NOAEL) in toxicology studies.
Materials and Methods
Animal Experiments
All experiments were conducted in the state of Northrhein-Westphalia, Germany. Protocols were reviewed and approved by the local authority (Landesamt für Natur, Umwelt und Verbraucherschutz) and an Institutional Animal Care and Use Committee. All in vivo studies were conducted in accordance with EU Directive 2001/20/EC and German Drug Law to support clinical trials. Details of the 3 animals reported herein are given in the Results section.
Intrathecal Bolus Administration
Following sedation with ketamine (intramuscular [im] 25 mg/mL for 0.1 mL/kg) and medetomidine (im 1 mg/mL for 0.06 mL/kg), animals were placed in abdominal recumbence on a curved metal grid (Figure 1); the arms were fixed in order to prevent any movement under anesthesia. Lumbar puncture was conducted between L2 and L6. Determination of the lumbar vertebra: The L6 spinous process is identified below or incorporated within an imaginary line drawn from 1 iliac crest to the other over the dorsum. From this defined line, the next cranial intervertebral space is considered to be L5/L6. From there, the other intervertebral spaces are counted cranially. For animals aged older than 12 months, standard intervertebral injection is at L5/L6 or L4/L5.
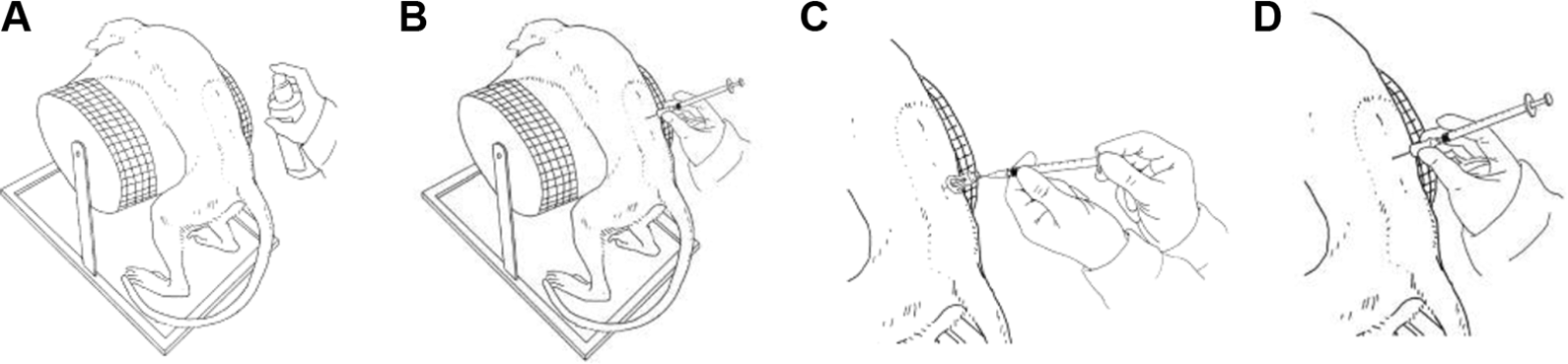
Schematic drawing of the lumbar intrathecal dosing procedure. A, Skin disinfection; B, Insertion of blunt spinal needle through previously created skin incision; C, CSF withdrawal; D, dose administration (owned by author). CSF indicates cerebrospinal fluid.
For younger animals (from 9 months up to 15 months), the intervertebral injection is carried out standardized in L4/L5 or L3/L4. The first injection should be attempted at the lowest possible vertebral space, for example, L5/L6 for older animals and at L4/L5 for animals to 15 months.
For anatomical reason, it might be necessary to choose different intervertebral injection sites in some individuals. This is the veterinarians’ decision. The actual injection site dosed at is documented. The area was shaved and a microincision of the skin was conducted using a 20G needle. Thereafter, 0.5 mL of CSF was withdrawn. A Pencan Paed pencil-point needle (25 G, B. Braun, Germany) was used to administer the test article over approximately 1 minute. Before injecting the test item, it was confirmed that CSF flow was present, and the position of the needle opening was facing cranially. Dose volumes were between 0.75 and 2.0 mL, always followed by 0.25 mL of a CSF to flush the needle. Subsequently, the animal was transferred into a box where it was placed in lateral recumbence for approximately 15 minutes. Thereafter, medetomidine was antagonized with atipamezole (im antisedan, at 0.3 mg/kg), and the animal was returned to its home cage.
Magnetic Resonance Imaging
Magnetic resonance imaging (MRI) of the lumbar spine (lower spinal cord, medullary cone, cauda equina, lumbar vertebral bodies, lumbar intervertebral discs, intervertebral foramina, and surrounding soft tissues) was performed using a Signa HDxt 8CH 1.5T scanner (GE Healthcare). The examinations were performed under general anesthesia (same as for IT dosing) on fasting animals with or without administration of a gadolinium contrast medium (Gadovist, Bayer). Animals were scanned in a prone position with ear protection with the use of 4 channel knee quad coil, in humans used for knee joint MRI scans or 4 channel head coil or 8 channel neurovascular coil. At the level of standard IT administration of drugs (approximately at L3/L4), the marker was positioned (vitamin A + D capsule) fixed with an adhesive plaster to the skin. Lumbar spine MRI scan parameters include field of view (FOV) 18 or 22 depending on the size of the animal; number of excitations 1 or 2; slice thickness 2.0 mm; and spacing 0.2 mm. Magnetic resonance imaging protocol included sagittal, coronal and axial T2-weighted images, sagittal T1-weighted and short τ inversion recovery (STIR) images before contrast administration and in the case with intravenous contrast also sagittal and axial T1-weighted images. The MRI scan covered lumbar spine, with the main focus on the spinal cord (lower spinal cord, medullary cone, cauda equina).
Results
In our laboratories, we identified a combination of findings indicative of rare adverse procedure-related effects that could be confused with drug-related effects. Differentiating between procedure-related and drug-related effects is essential in appropriate characterization of the NOAEL and maximum tolerated dose. To differentiate between procedure-related and drug-related findings, we compared rare instances of procedure-related effects with typical findings in our laboratory. Based on our existing experience with 30 IT toxicology studies using ASOs including >1000 total cynomolgus monkeys on study, with >4500 separate dosing occasions, we noted a >99.9% success rate for IT dosing in cynomolgus monkeys with ASOs. Adverse procedure-related findings were observed in 3 animals, <0.1%.
To differentiate between adverse procedure-related findings and drug-related effects, we identified a pattern of findings for procedure-related effects that are inconsistent with typical drug-related findings for ASOs. The pattern of findings was generally consistent across 3 cases of procedure-related adverse effects, yet different from drug-related or incidental findings. Based on our case studies, we prepared a rubric to differentiate procedure-related injury from drug-related effects (Table 1). Details of the 3 case studies are included below. In summary of these case studies, we found that procedure-related effects can be differentiated from drug-related effects based on findings during the dosing procedure, the dose–response relationship, type of clinical signs, frequency and timing of findings in a study, duration of findings, neurological assessments, MRI scans, anatomic findings and histological evaluations, as well as increased systemic exposure with concurrent increased histological findings in systemic tissues of the affected animal (egg, kidney vacuolation consistent with increased systemic exposure to ASOs).
Comparison of Typical ASO Postdose and Procedure-Related Effects in Cynomolgus Monkeys Dosed by Intrathecal Injection With Oligonucleotides.

Abbreviations: ASOs, antisense oligonucleotides; CSF, cerebrospinal fluid; MRI, magnetic resonance imaging.
Results
Case 1
A 3-year-old male cynomolgus monkey (3 kg) of Asian origin was dosed with an ASO (day 1). At 30 minutes postdosing, this animal showed clinical signs of lameness and reduced muscle tone of both hind limbs as well as loss of patellar and foot grip reflexes, leading to paresis. On day 6, MRI analysis showed a linear hyperintense lesion within the spinal cord in T2-weighted images and STIR images visible from the level of mid L1 (first lumbar vertebral body) to lower end plate of L3. The dimensions of the linear hyperintense lesion were 5.9 (anteroposterior), 7.0 (laterolateral), and 16.4 (craniocaudal). Additionally, the animal presented normal lumbar lordosis, normal height, morphology, and signal of vertebral bodies and intervertebral discs.
The clinical signs resolved without veterinary intervention within 3 weeks (day 22). A second scan 9 weeks after day 1 presented a linear hyperintense lesion in T2 weighted images and STIR images visible from the level of mid L1 (first lumbar vertebral body) to lower end plate of L3 of the sizes: anteroposterior: 2.8 mm, laterolateral: 3.5 mm, and craniocaudal: 40 mm (Figure 2A). The lesion presented higher signal intensity in T2-weighted images and STIR images than in initial scan. Although there were no macroscopic observations at necropsy (day 89), histological assessment revealed moderate myelomalacia of the dorsal portion of the lumbar spinal cord, characterized by loss of grey and white matter associated with phagocytosis and a cavity containing cellular debris (Figure 3A-D).
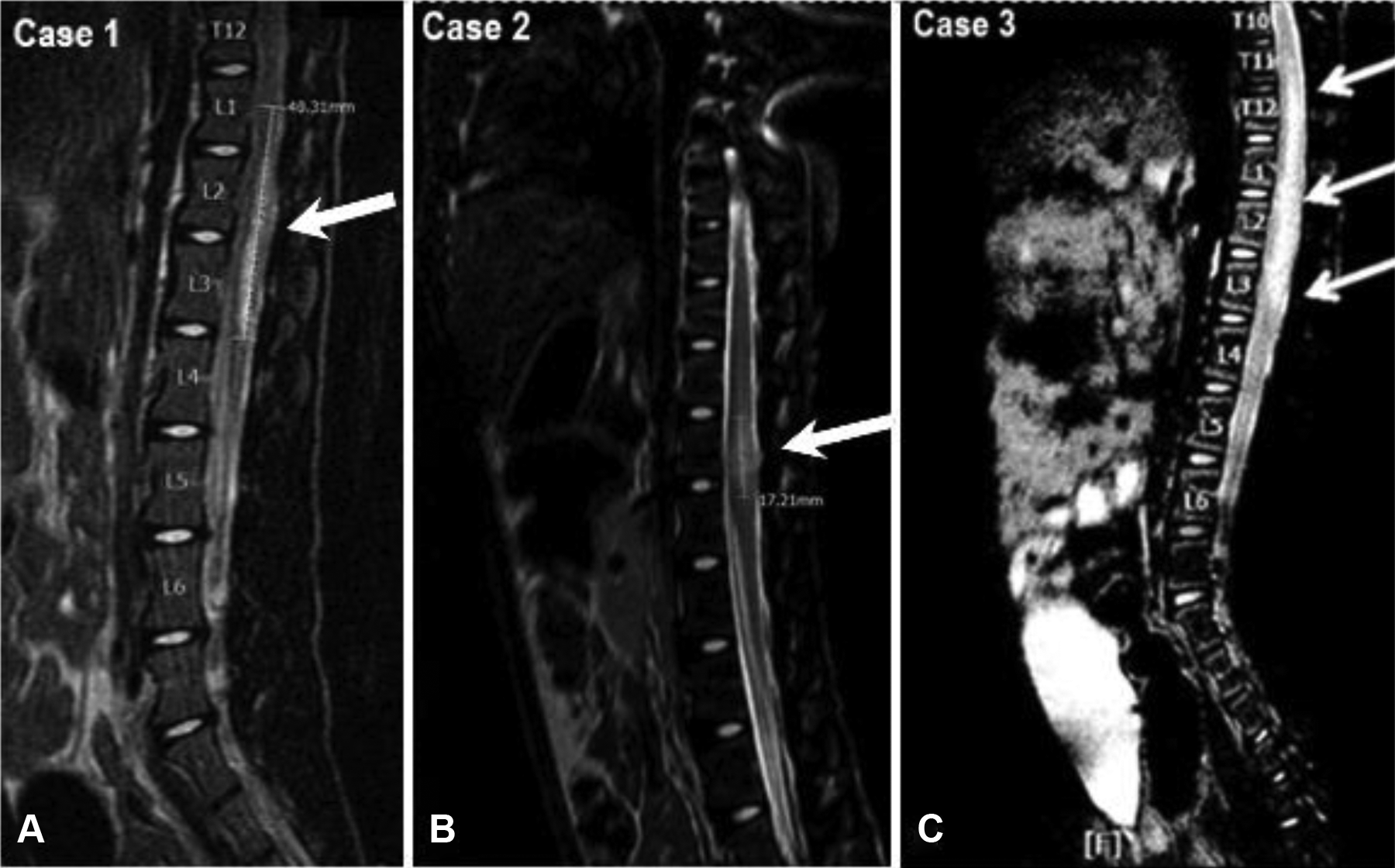
Comparison of MRI of the lumbar spine in case 1 to 3 after intrathecal bolus administration. A, Animal (case 1) presenting a linear hyperintense lesion in T2-weighted images and STIR images visible from the level of mid L1 (lumbar 1 vertebral body) to lower end plate of L3 of the sizes 2.8 mm (anteroposterior), 3.5 mm (laterolateral), and 40 mm (craniocaudal). B, Animal (case 2) presented spinal cord edema at the level of L2/L3 (lumbar 2 and 3 vertebral bodies) with markedly reduced fluid buffer space surrounding the spinal cord and the presence of a pathological linear hyperintense lesion in T2-weighted and STIR images within the spinal cord. C, Animal (case 3) presented at the level of L2 with signs of spinal cord edema (spinal canal stenosis). MRI indicates magnetic resonance imaging; STIR, short τ inversion recovery.
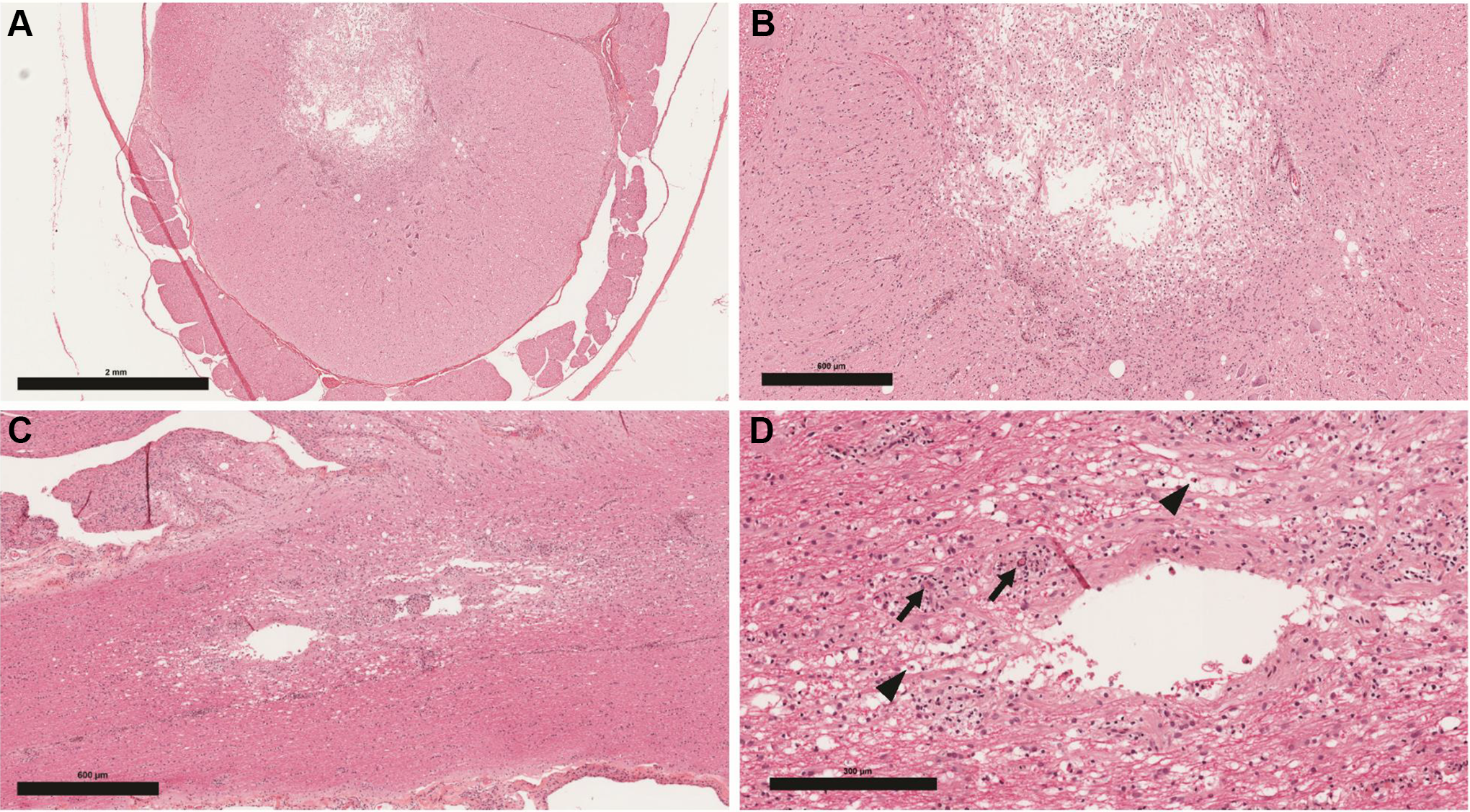
Injection site, intrathecal, transverse (A and B) and longitudinal (C and D) sections (H&E). A, Lumbar spinal cord with spinal nerve roots displaying focally extensive area of cavitation/myelomalacia in the grey matter; multifocal vacuoles in both grey and white matter. B, Cavitated area in lumbar spinal cord grey matter at higher magnification with gitter cells and few scattered neutrophils. C, Lumbar spinal cord with dorsal nerve roots; focally extensive area of cavitation/myelomalacia with cell debris and multifocal vacuolation. D, Lumbar spinal cord with dilated axonal myelin sheaths, gitter cell accumulation (digestion chambers, arrow heads); inflammatory cell cuffing (macrophages, lymphocytes, few neutrophils) around capillaries adjacent to cavitation (arrows). H&E indicates hematoxylin and eosin.
Case 2
A 5-year-old male cynomolgus monkey (4 kg) of Mauritian origin was dosed with an ASO (day 1). The animal showed body tension and muscle contraction immediately upon and during the dosing procedure while under sedation. Upon recovery of anesthesia and return to the cage, the animal showed signs of reduced muscle tonus, patellar, and foot grip reflexes in both hind limbs, with minor improvement over the next 48 hours. Thereafter, the animal showed improvement of clinical signs. On day 11, MRI analysis (Figure 2C) with and without contrast agent (Gadovist, Bayer) showed evidence of spinal cord edema on the level of L2/L3 with markedly reduced fluid buffer space surrounding the spinal cord. In addition, there was a linear hyperintense lesion within the spinal cord in T2-weighted images and STIR images at the level of L2 (lumbar 2 vertebral body) of the following dimension: anteroposterior: 2.4 mm, laterolateral: 3.4 mm, and craniocaudal: 17 mm. The lesion did not present enhancement following intravenous contrast agent administration.
Scheduled euthanasia of this animal was on day 28 after dosing. No macroscopic findings were noted, however, microscopic assessment showed slight focal axonal degeneration, foamy macrophages, and vacuolation of the lumbar spinal cord. In addition, slight multifocal vacuolation with axonal degeneration of spinal nerve roots at the lumbar IT injection site was observed (Figure 4A-C).
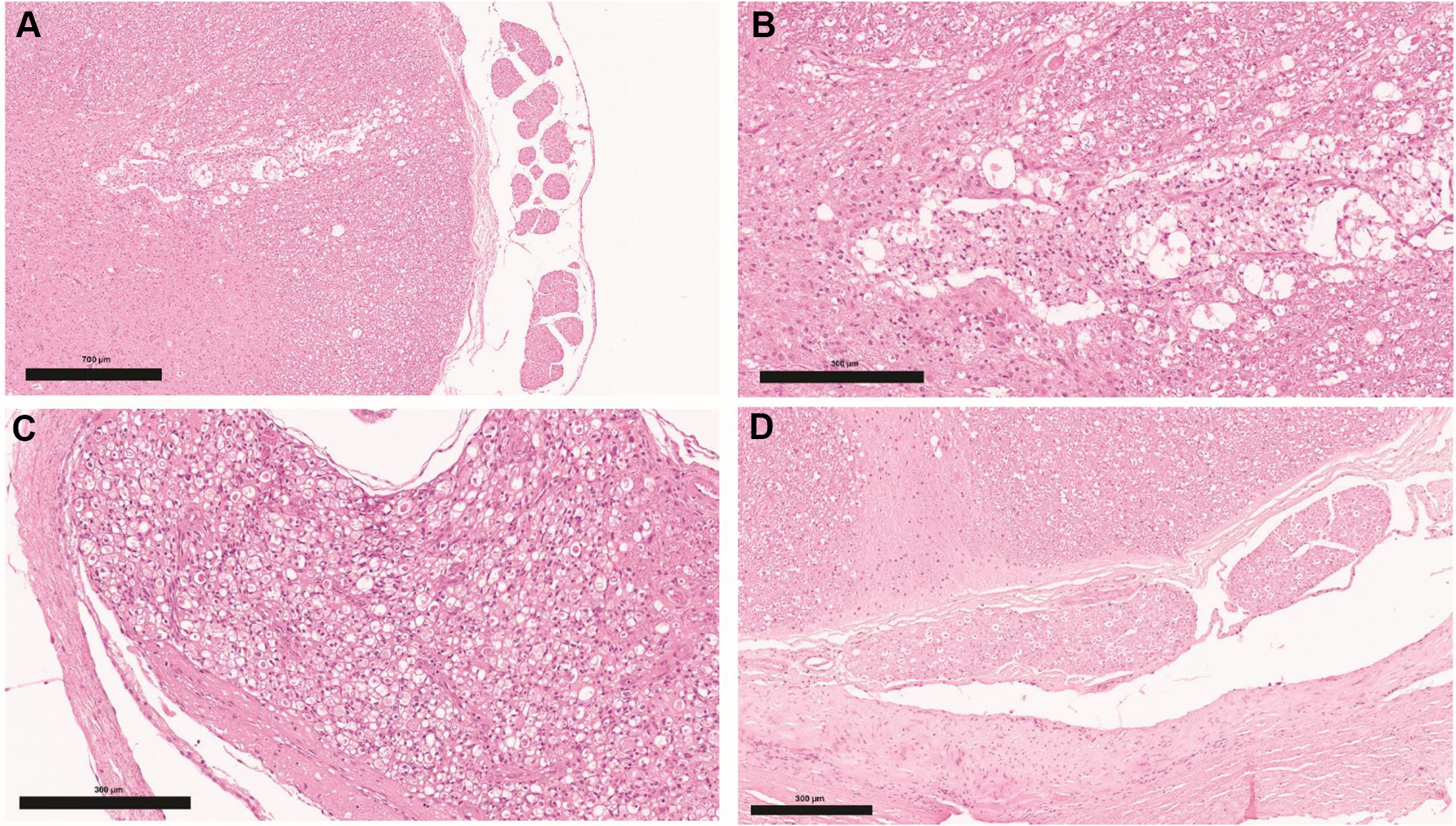
Injection site, intrathecal, transverse sections (H&E). A, Lumbar spinal cord and adjacent spinal nerve roots with area of cavitation and vacuolation in the ventral funicular white matter. B, Area with cavitation in the white matter of the lumbar spinal cord at higher magnification filled with numerous gitter cells; adjacent white matter displays dilated myelin sheaths and swollen axons (spheroids). C, Spinal nerve root adjacent to intrathecal administration site with numerous dilated myelin sheaths and spheroids. D, Spinal nerve root from a control animal. H&E indicates hematoxylin and eosin.
Case 3
An 11-month-old male cynomolgus monkey (1.6 kg) of Asian origin was dosed with an ASO (day 1). At the second placement of the needle, the backflow of CSF was blood-filled instead of clear. Immediately after dosing, this animal had loss of patellar, anal, and foot grip reflexes. At 4 hours postdosing, it developed paraplegic lameness of both hind limbs. By day 2, the reflexes recovered and there was limited unilateral recovery of hind limb mobility. On day 2, MRI indicated clinical spinal cord infarction from the level of T11 to the lower end plate of L3 (anteroposterior: 6.2 mm, laterolateral: 6.2 mm, craniocaudal: 43.0-46.0 mm, Figure 2C) consistent with spinal cord edema (spinal canal stenosis). Furthermore, there was a markedly reduced fluid buffer space surrounding the spinal cord and the presence of a linear pathological lesion within the spinal cord, hyperintense in T2-weighted images and STIR images. Additionally, the animal presented normal lumbar lordosis, normal height, morphology, and signal of vertebral bodies and intervertebral discs. No signs of dehydration of the vertebral disc, abnormalities of vertebral end plates, or any disc protrusions/signs of narrowing of the lumen of intervertebral foramina were noted in the tested animal. No abnormalities were detected in the soft tissues surrounding the FOV. The animal was euthanized on day 3 due to persisting hind limb paralysis and diagnostic support of MRI assessment. The decision to euthanize this animal on day 3 was also informed by the laboratory’s prior experience with comparable procedure-related findings described in cases 1 and 2.
Macroscopic findings included red and swollen appearance of the lumbar spinal cord. Microscopic findings were comprised of necrosis (slight, multifocal), hemorrhage (moderate, multifocal), axonal degeneration (moderate, multifocal), and reactive astrogliosis (slight, multifocal) in the lumbar spinal cord. Axonal (Wallerian-type) degeneration was characterized by marked dilation of myelin sheaths, which enclosed multifocally swollen axons (spheroids) and/or vacuolated macrophages (digestion chambers), which were predominantly located in the dorsal funiculus gracilis, ventral funiculus, and to a lesser extent the lateral funiculus. These degenerative changes appeared more severe unilaterally when compared to the contralateral side (Figure 5A-C). In the kidney, minimal, multifocal basophilic granules were observed in the cytoplasm of proximal tubule epithelial cells consistent with higher systemic exposure in this 1 animal compared to other drug-treated animals in the same dose group (data not shown).
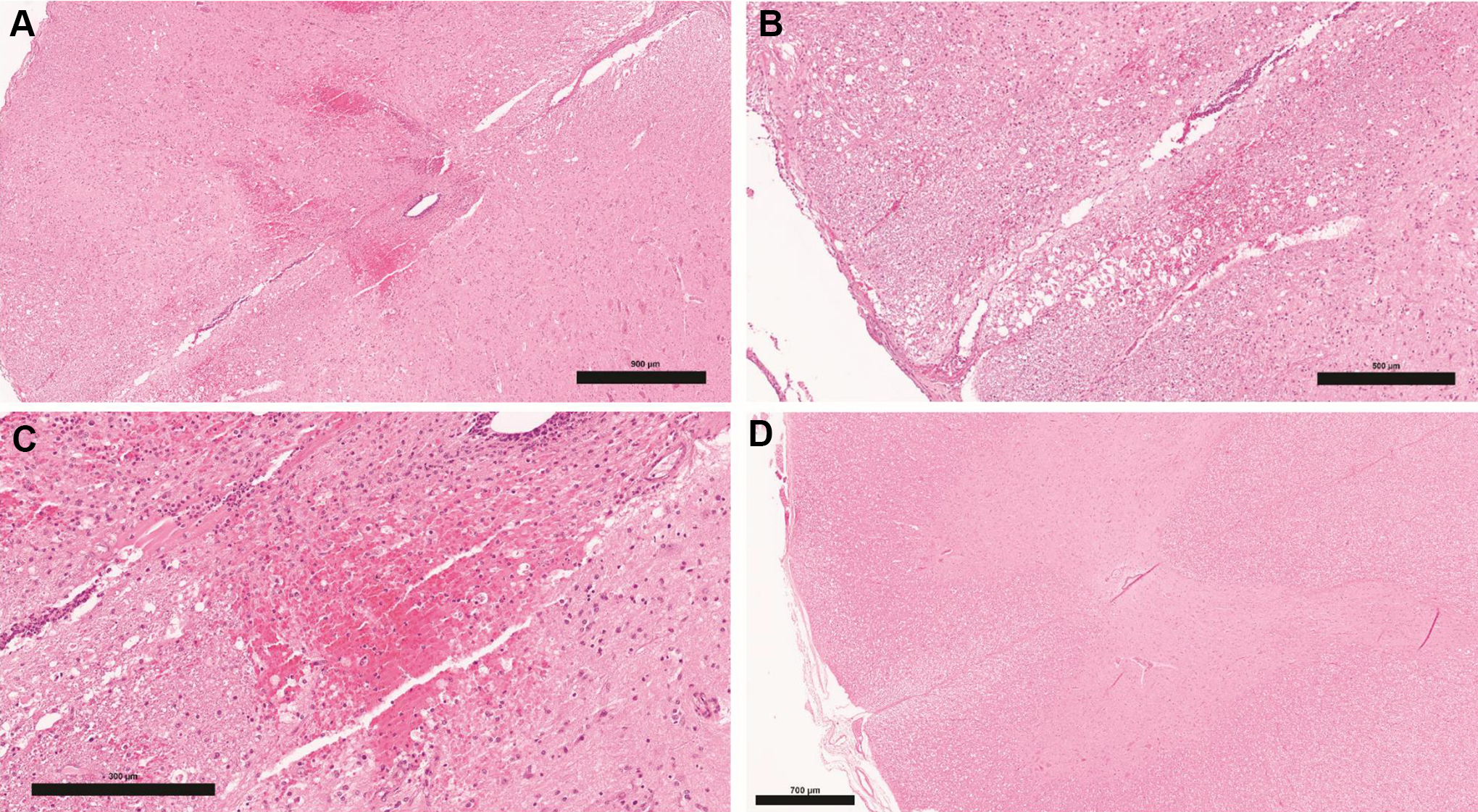
Injection site, intrathecal, and transverse sections (H&E). A, Lumbar spinal cord with multifocal hemorrhages in grey and white matter near the central canal. B, White matter adjacent to the ventral median fissure with focal hemorrhage, multiple dilated myelin sheaths, and swollen axons (spheroids). C, Focal myelomalacia and hemorrhage admixed with gitter cells in lumbar spinal cord grey matter. D, Lumbar spinal cord from a control animal. H&E indicates hematoxylin and eosin.
Discussion
With ongoing and increasing development of novel therapeutics for serious neurodegenerative diseases, more nonclinical toxicology studies will be conducted using IT injection as the dosing route in support of planned clinical trials. antisense oligonucleotides are one moiety of drug underdevelopment using this dose route. Based on our existing experience with 30 IT toxicology studies using ASOs including >1000 total cynomolgus monkeys on study, with >4500 separate dosing occasions, we noted a >99.9% success rate for IT dosing in cynomolgus monkeys with ASOs. With a very small set of animals, <0.1%, we identified a pattern of clinical signs, bioanalytical, MRI, and anatomic findings that characterize procedure-related effects that are inconsistent with typical drug-related findings for ASOs.
We present a pattern of findings that are secondary to procedure-related mechanical injury of the spinal cord. These findings likely apply to all drug classes, yet our data and discussion focus on ASOs because of our extensive preclinical data base for IT injection of ASOs in cynomolgus monkeys. We have conducted IT ASO administration in over 30 preclinical safety studies with >4500 individual dosing occasions in >1000 cynomolgus monkeys. Among those studies, we observed 3 cases of procedure-related spinal cord lesions, a rare traumatic complication encountered in IT administration.
These rare cases of procedure-related changes show a distinct pattern of elements that are different from effects commonly seen in cynomolgus monkeys dosed with ASOs after IT injection. A typical and normal dose of an ASO by IT injection in a monkey is characterized by a clear backflow of CSF after needle placement in IT space, transient clinical signs occurring after dosing with reversion to normal a few hours postdosing, a dose-related response pattern of clinical signs (eg, transient reduced hind limb mobility, transient uni- or bilateral reduction of reflexes (patellar, anal, and foot grip), lack of macroscopic findings, and limited dose-related microscopic changes in the spinal cord. These findings are not considered adverse as they are transient and/or of low magnitude.
In contrast, procedure-related findings after IT injection include blood present in CSF upon needle placement, lack of dose–response, clinical signs that increase in severity postdose, persisting clinical signs (eg, prolonged impairment of reflexes (patellar, anal, and foot grip), hind limb paralysis, nonreversible paresis, and macroscopic changes (eg, edema, necrosis, and hemorrhage). Spinal cord edema and necrosis can be determined in life by noninvasive MRI. Bioanalytical data may also indicate higher plasma and tissue exposure in animals with procedure-related findings, as well as drug accumulation, generally evidenced by typical microscopic granules. Such procedure-related findings are not relevant to inform and determine the NOAEL.
We propose the following approach to facilitate the differentiation of procedure-related findings (Table 1), bearing in mind that not every animal dosed by IT injection will necessarily display every finding. A clear conclusion that a set of findings is procedure-related instead of drug-related requires an assessment based on the totality of the study data, not individual findings. A determination that a set of findings are procedure-related is strongest when multiple lines of evidence indicate that the findings are secondary to a dosing procedure.
The inclusion of MRI assessments certainly adds value to the diagnosis of procedure-related effects, since MRI is a noninvasive tool that can guide early decision making prior to necropsy. Although our approach/list is based on case studies using ASOs dosed by IT injection, a common dose route for this drug class, it applies to all drugs classes dosed by IT injection in nonhuman primates and facilitates distinction of severe and nontransient procedure-related from more transient test item-related findings.
Similar to our findings in cynomolgus monkeys, neurological complications have been reported 4 -6 clinically during steroid injections of the lumbar spine to manage common back and leg pain. The complications are very rare as few cases of paraplegia of acute onset following image-guided selective steroid injection of the lumbar epidural space have been reported since 2002 in the literature. 6 Steroid injections of the lumbar spine carry a minimal, however inestimable, risk of sudden-onset paraplegia. The MRI findings in such cases were consistent with spinal cord injuries due to infarction of arterial origin.
The etiology of spinal cord infarction in patients is supposed to be local spinal cord ischemia following thrombosis of an extravertebral artery. In the clinic, infarction is tissue death (necrosis) due to inadequate blood supply to the affected area. It may be caused by artery blockages, rupture, mechanical compression, or vasoconstriction. Infarction occurs as a result of prolonged ischemia, which is the insufficient supply of oxygen and nutrition to an area of tissue due to a disruption in blood supply.
With ongoing and increasing development of novel therapeutics for serious neurodegenerative diseases, more nonclinical toxicology studies will be conducted using IT injection as the dosing route in support of clinical trials. Antisense oligonucleotides are one moiety of drug underdevelopment using this dose route. With a very small set of animals, <0.1%, we identified a pattern of clinical signs, MRI, and anatomic findings that characterize procedure-related effects that are inconsistent with typical drug-related findings for ASOs.
A comprehensive assessment of in-life and anatomical findings is essential to provide an appropriate diagnosis of procedure-related effects. The strongest comprehensive assessments that an apparent adverse effect is procedure-related is based on multiple lines of evidence, not a single observation. We believe that inclusion of MRI assessments adds value to the diagnosis of procedure-related effects because MRI is a noninvasive tool that can guide early decision-making prior to necropsy. Although our rubric is based on case studies using ASOs dosed by IT injection, a common dose route for this drug moiety, our recommended rubric applies to all drug classes for differentiating procedure-related findings dosed by IT injection in nonhuman primates.
Overall, differentiation of drug-related and procedure-related findings in nonclinical studies is essential in hazard identification and safety assessment, as procedure-related findings are not directly relevant for the safety assessment of drugs in nonclinical studies.
Conclusion
Differentiating between drug-related adverse effects and procedure-related effects is essential for appropriate selection of the NOAEL and maximum tolerated dose. With more than 4500 IT injections in more than 1000 cynomolgus monkeys, we have observed only 3 cases in which traumatic spinal cord necrosis was induced. Lumbar spinal cord necrosis is a very rare assumed mechanical, IT dosing-related complication in cynomolgus monkeys. We recommend diagnosing procedure-related adverse effects based on the totality of data including observations during dosing, the dose–response relationship, types of clinical signs, onset and duration of clinical signs, frequency of clinical signs, neurological assessments, MRI scans, macroscopic findings, and microscopic changes, as well as potential abnormalities systemic exposure with concurrent increase in findings in systemic tissues. Magnetic resonance imaging is a useful noninvasive tool that can be used to characterize this rare injury.
Footnotes
Authors’ Note
The authors of this manuscript certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or nonfinancial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript. Thus authors do work in the contract research industry and conduct preclinical regulatory safety assessment studies with nonhuman primates.
Author Contributions
Korte substantially contributed to conception or design and drafted the manuscript; Luft contributed to acquisition, analysis, or interpretation of data and drafted the manuscript; Keutz contributed to acquisition, analysis, or interpretation of data and drafted the manuscript; Runge contributed to acquisition, analysis, or interpretation of data and drafted the manuscript; Wozniak substantially contributed to conception or design and critically revised the manuscript for important intellectual content; Zander substantially contributed to conception or design and critically revised the manuscript for important intellectual content; Ludwig contributed to acquisition, analysis, or interpretation of data and drafted the manuscript; Pajaziti contributed to acquisition, analysis, or interpretation of data and critically revised the manuscript for important intellectual content; Romeike substantially contributed to conception or design and drafted the manuscript; Korytko contributed to acquisition, analysis, or interpretation of data and drafted the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
