Abstract
Adeno-associated virus (AAV)-based vectors are the most frequently used platform for retinal gene therapy. Initially explored for the treatment of loss-of-function mutations underpinning many inherited retinal diseases, AAV-based ocular gene therapies are increasingly used to transduce endogenous cells to produce therapeutic proteins, thus producing site-specific biofactories. Relatively invasive ocular routes of administration (ROA) mean prominent procedure-related in-life, and histopathological findings may be observed with some regularity. Test article-related findings may vary with the ROA and cell populations transduced, with retinal pigmented epithelium (RPE) changes prominent (ranging from pigment alteration through degeneration, with or without associated degeneration of the overlying retina) with subretinal ROA, and more anterior changes (iris, ciliary body) generally observed with the intravitreal ROA. Ocular inflammation is the most frequent finding that occurs nonclinically and in patients, and is particularly pronounced with intravitreal administration. Extraocular findings may be observed in extraocular muscles, regional ganglia, or central visual pathways with multiple ocular ROA. Work is still needed to understand the mechanisms underpinning many of these ocular and extraocular findings. Emerging patient data is helping to clarify both the potential for translating nonclinical findings to predict possible human responses and the applicability of nonclinical biomonitoring methods to the clinical setting.
Introduction
Retinal gene therapy is an emerging field with great promise for treating ocular diseases. Direct injection of a gene therapy vector into the ocular compartment offers the potential to both ameliorate previously untreatable inherited retinal diseases (IRDs) and dramatically improve quality of life for patients with acquired disease.
Adeno-associated virus (AAV)-based vectors are currently the most commonly used platform for retinal gene therapy. 20 The first AAV therapeutic to treat ocular disease was voretigene neparvovec-rzyl (LuxturnaTM), an AAV2 gene therapy delivering a working copy of the retinoid isomerohydrolase RPE65 gene to treat Leber’s congenital amaurosis that was approved by the United States Food and Drug Administration (FDA) in 2017. 42 Since then, the number of AAV-based retinal gene therapies in development has increased dramatically, with more than 45 products currently in clinical trials (www.clinicaltrials.gov).
The eye is an attractive candidate for viral gene therapy-based approaches, for several reasons. The eye is relatively compartmentalized, both physically and immunologically. This feature provides two key advantages in treating ocular diseases. First, therapeutic agents may be delivered locally, to specific target sites, thereby enabling the use of relatively low numbers of vector genomes. Second, the low dose levels decrease the propensity to develop some of the characteristic “class effects” in distant organs (eg, dorsal root ganglia, heart, liver) that are associated with systemic administration of AAV-based gene therapies.24,43 However, although an exciting prospect, ocular gene therapies are not without problems. In humans, ocular inflammation is a well-recognized and common complication of viral vector-mediated ocular gene therapy.11,14,20 The severity of inflammation has been shown to vary dramatically but is often related to vector dose and is impacted by the chosen intraocular route of administration (ROA). 14 Clinical trial data have shown that retinal gene therapies in patients may be associated with severe ocular inflammation leading to vision loss.20,37 The high clinical incidence of inflammation associated with retinal gene therapies has resulted in the regular implementation of courses of prophylactic anti-inflammatory and immunosuppressive treatments in clinical trials.
This article is based on a talk entitled “Developing AAV-Based Ocular Gene Therapies,” which was delivered during a half-day scientific session on “AAV-Based Therapies” offered during the 2024 annual symposium of the Society of Toxicologic Pathology (STP) in Baltimore, Maryland in June. The talk and this companion article highlight the nonclinical histopathologic findings identified to date following administration of ocular gene therapies, both within the eye and outside of it, to aid pathologists in preparing reports grounded in consistent lesion descriptions and diagnostic terminology. Additional topics include key considerations in the development of ocular gene therapies, such as the common in-life clinical correlates of histologic lesions in the eye; utilization of immunosuppressive and anti-inflammatory treatments nonclinically; and the importance of communication between the veterinary ophthalmologist and toxicologic pathologist nonclinically as well as the medical ophthalmologist clinically to facilitate data interpretation, correlation of findings, and assessment of translatability and human risk.
Core Considerations in the Development of Ocular Gene Therapies
In the United States, gene therapies are regulated through FDA’s Center for Biologics Evaluation and Research (CBER). Two guidance documents produced by the FDA help to direct the development of retinal gene therapies: Human Gene Therapy for Retinal Disorders: Guidance for Industry (FDA, 2020) 18 and Preclinical Assessment of Investigational Cellular and Gene Therapy Products (FDA, 2013). 17
From a nonclinical development perspective, some specific components should be incorporated in the safety assessment package for retinal gene therapy test articles (TAs). Notably, however, no “one-size-fits-all” approach is possible in developing retinal gene therapies. Therefore, the paradigm implemented for an individual product development program should be data-driven according to a rational scientific justification. The main elements that are particularly relevant to the pathologist’s work flow are a suitable rationale and proof-of-concept (efficacy) data to support ocular administration to a specific patient population, acquisition of toxicity data utilizing the intended clinical route/procedure/device and clinical-grade TA, and conduct of toxicity studies in an appropriate test system—typically a large animal species with a bigger globe that more closely approximates the size of the human eye. If feasible, the test system (eg, a spontaneous or engineered animal disease model) may possess a target cell population that can assess the pharmacological activity of the human-derived transgene product.
Rationale and Proof-of-Concept for Ocular Administration to a Specific Patient Population
Currently, administration of one AAV-based gene therapy precludes the administration of a subsequent AAV-based therapy to the same patient. This “one and done” situation stems from priming of an anti-AAV host immune response by the first product, which may limit or entirely negate the therapeutic effects of any subsequent AAV agent. 47 Accordingly, it is vital that strong proof-of-concept data are available to support both TA efficacy for a particular patient’s disease and effective dose selection before an AAV-based gene therapy is administered. 24 Both in vitro and in vivo data can be utilized to provide this rationale.
For some conditions, particularly monogenic inherited retinal diseases, in vivo studies in genetically modified animal models bearing the genetic mutation of interest (usually rodents, although in some cases spontaneous canine models) provide excellent proof-of-concept data.30,32 For gene-agnostic approaches, such as those assessing optogenetic targets (where the aim is to confer photoreceptor-like function on nonphotoreceptor cells in the retina), in vivo studies in models of retinal degeneration (genetic or induced) may also provide good efficacy data. However, for certain indications (eg, geographic atrophy), reliable animal models that reproduce the findings and pathogenesis of the human disease are not available. In these cases, in vivo proof-of-concept studies in wildtype animals may be less helpful, and a combination of human data and in vitro work may afford increased confidence that efficacy may be attained in human patients.
Comprehensive Safety Assessment With the Intended Clinical Route/Procedure/Device and Clinical-Grade Test Article
For retinal gene therapy candidates, the nonclinical study design should closely mimic the planned clinical trial design. The principal considerations in these regards are that nonclinical studies should utilize the intended clinical ROA (eg, intravitreal, suprachoroidal, subretinal); dosing procedure (including pre-operative preparation and intra-operative approach); and dosing devices as far as possible. In general, nonclinical studies designed to enable an Investigational New Drug (IND) or comparable application should be Good Laboratory Practice (GLP)-compliant and should employ the optimized clinical-grade TA.
From a safety perspective, the treatment should be considered permanent—it cannot be stopped or reversed in the case of lack of efficacy or development of an adverse event. Toxicity studies must assess both local and systemic toxicities, and the nonclinical program needs to capture potential acute, chronic, and/or delayed onset toxicities as well as the potential for resolution of toxicity. 31 For gene therapies, study duration goals are currently considered on a case-by-case basis, although standard nonclinical bioassay designs (13-week subacute studies, 26-week chronic studies) are usually appropriate to enable dosing in patients. 31 For ocular toxicity studies, in-life evaluations may be used to help understand some of the more acute effects in the context of longer-duration studies. In selecting among many design options, it is important to consider the types of findings that are likely to be observed, what potential progression and/or resolution might develop over time, and what the impact of those findings might be in terms of assessing risk to human patients.
Safety studies may use healthy animals and/or an animal model of disease (where the latter provides combined safety and efficacy data). However, although efficacy studies are often conducted in rodent disease models, IND-enabling toxicity studies typically should be conducted in large animal species, such as the rabbit, dog, pig, or nonhuman primate (NHP). The preference for nonrodent test species is that their bigger globes better simulate the anatomy of human eyes, thereby facilitating assessment of intraocular biodistribution (BD) and pharmacokinetics (PK); evaluation of the dosing procedure and delivery devices; and the suitability of translatable techniques for monitoring TA effects on ocular tissues.
In-Life Evaluations in Ocular Toxicity Studies
In-life ophthalmology evaluations are critical to the successful identification and interpretation of structural findings in ocular toxicity studies. In-life techniques provide longitudinal, noninvasive, and directly translatable monitoring tools for assessing ocular health. Moreover, such methods frequently have morphological endpoints, thereby serving as gross correlates for findings subsequently identified and characterized during the histopathological evaluation. The ability to identify during life when lesions first occur and to follow their evolution over time can be extremely helpful in understanding the pathogenesis.
Slit lamp biomicroscopy and indirect/direct ophthalmoscopy, combined with scoring of ocular inflammation, are the mainstays of in-life structural ocular evaluations. These methods should be repeated at suitable intervals throughout the duration of the study to capture the evolution or resolution of findings. Slit lamp biomicroscopy provides a high-resolution, three-dimensional (3D) assessment of the anterior globe, while ophthalmoscopy enables morphological assessment of the posterior globe and retina. In addition, tonometry (which measures intraocular pressure, providing functional assessment of aqueous humor production and outflow) and electroretinography (ERG, which assesses retinal phototransduction) are frequently included as measures of global and retina-specific ocular function.
Advanced in-life techniques should be considered on a study-by-study basis. Principal options include wide-field color fundus imaging; optical coherence tomography (OCT, which offers high-resolution, cross-sectional images of the retina); and scanning laser ophthalmoscopy (scLO, which produces high-contrast, 2D surface images of the retina). These in-life methods provide detailed longitudinal, morphologic assessment of the retina and are easily implementable in patients. A more comprehensive description of these in-life ocular evaluation techniques in nonclinical toxicity studies may be found in the “Eye” chapter of Haschek and Rousseaux’s Handbook of Toxicologic Pathology, 4th ed., vol. 4. 9
Prior to histopathologic evaluation of an ocular toxicity study, the pathologist should have an awareness of the in-life observations related to the visual system (eye and associated brain centers).8,40 Moreover, the pathologist should consider (after discussion with the study ophthalmologist) correlating the morphologic findings with relevant in-life ophthalmology findings in the pathology report. In the case of ocular toxicity studies, as the eye is not generally opened at necropsy, data from a pre-terminal in-life ophthalmic examination is usually the most relevant (and impactful) macroscopic endpoint. Correlations of in-life and histopathology findings should be integrated into the final study report by the study director, ideally in consultation with the study pathologist and ophthalmologist.
Pathology Considerations in Ocular Toxicity Studies
Ocular gene therapy studies frequently utilize complex dosing procedures, with the most common intraocular ROA being intravitreal, subretinal, and suprachoroidal (Figure 1). 20 It is important to understand that a given ROA may employ substantially different procedures depending on such factors as the nonclinical species, investigator preference, or the development of devices to aid administration. For example, subretinal doses can be administered via either a suprachoroidal or a transvitreal approach; total or partial vitrectomies may be performed to aid dosing placement; a pre-bleb of sterile saline may be injected into the subretinal space prior to TA administration to facilitate dose distribution; the dose may be administered at various subretinal locations (eg, central, peripheral, submacular), and may even be split among multiple smaller blebs. These factors can all impact the location(s), character, and severity of pathological findings in and near the eye.
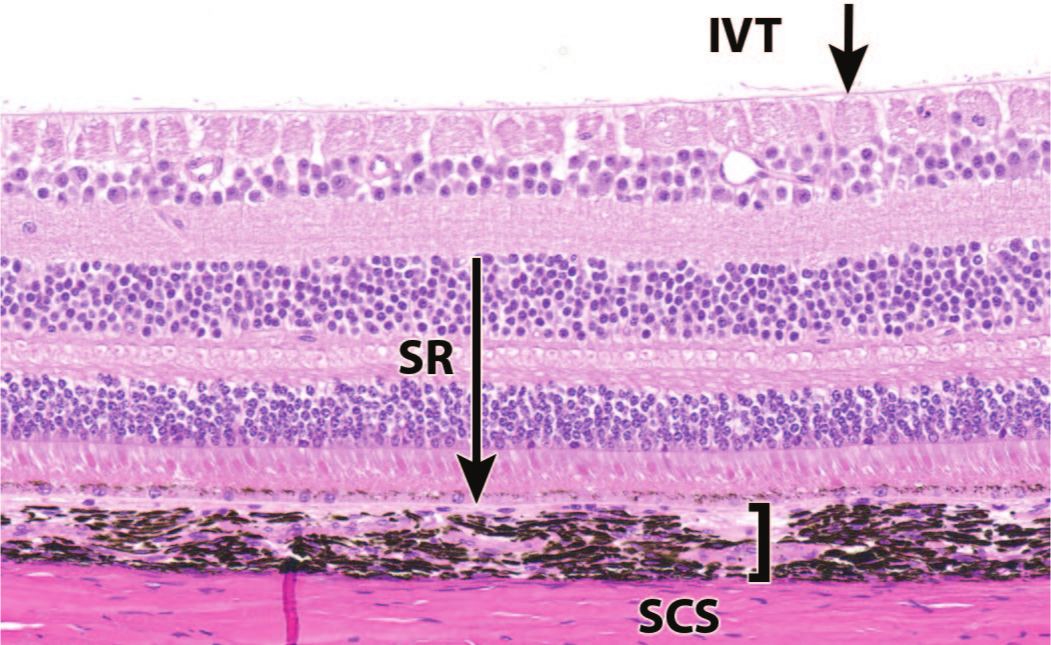
Commonly utilized ocular routes of administration for AAV-based retinal gene therapy vectors. IVT indicates intravitreal; SR, subretinal; SCS, suprachoroidal. Nonhuman primate.
In line with a recent “points to consider” publication for fixation, trimming, and sectioning eyes during nonclinical studies, 8 expanded ocular sampling will be required routinely for toxicity studies of retinal gene therapy products. Knowledge of the dosing procedure is impactful for designing the most suitable trimming and sampling procedures for the eye (and extraocular tissues). For subretinal and, to a lesser degree, suprachoroidal ROA, the administration site will usually be the region of highest transduction and therefore most susceptible to TA-related effects and procedure-related changes. To enable interpretation and contextualization of ocular findings occurring during a study, histopathologic evaluation of the eye must be conducted with knowledge of the in-life findings and the precise location and procedure for dose administration. When describing lesions associated with the introduction of a retinal gene therapy product, the location and extent (proportion of retina examined, location within or peripheral to the “bleb” [administration site], etc) should be detailed where possible.
Collection of in-life and postmortem tissue samples for assessing TA BD in the eye is a common request for ocular AAV studies. Separate PK studies are not usually conducted. Because of such requests, bilateral dosing is usually recommended for ocular AAV studies, enabling one eye to be used for histopathologic evaluation and molecular localization (spatial biology) (Figure 2), while the second is dedicated to in-life and postmortem sampling to assess BD. Aqueocentesis (fluid collection from the anterior chamber) and vitreocentesis (fluid collection from the posterior compartment) are invasive procedures that break down blood-ocular barriers and increase the potential for ocular inflammation. In addition, in the case of vitreous aspirates during life, it must be remembered that the vitreous is not homogenous, and the distribution of TA, transgene product, and downstream biomarkers may not be uniform between the gel and liquid phases. Accordingly, the vitreous sample collected will only sample the liquid phase, which may impact the BD data collected.
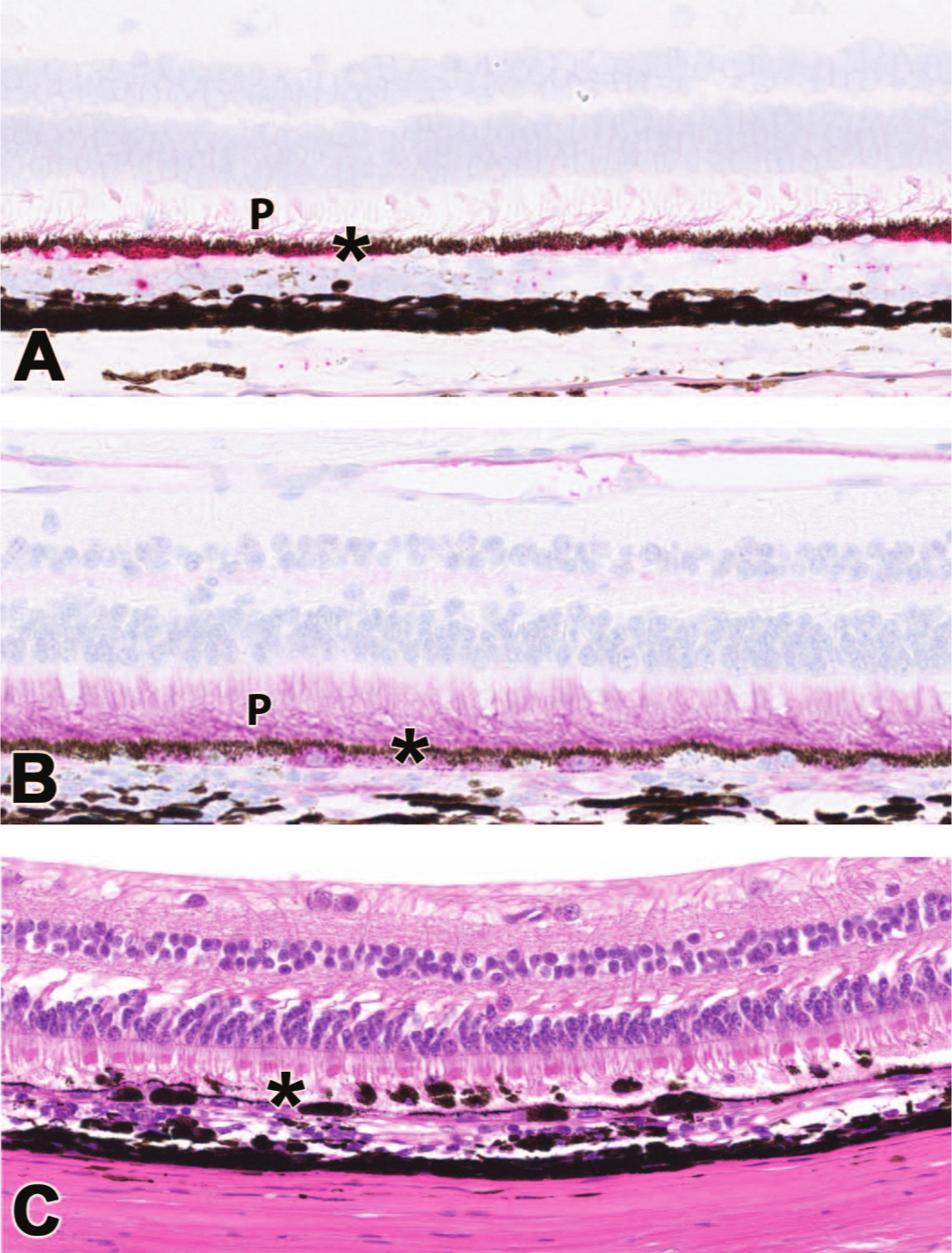
Molecular localization can be used to aid the assessment of biodistribution (BD) in the context of pathology findings. (A) In situ hybridization for transgene mRNA (red dots in the retinal pigment epithelium [RPE] and sclera) demonstrating transcription; (B) immunohistochemistry (IHC) for transgene product (pink hue in the photoreceptor layer and RPE cells), showing translation to produce the protein of interest; and (C) correlation of molecular distribution, and high levels of expression with regions of histopathologic findings in another eye (RPE degeneration). Asterisks = RPE; P = photoreceptor layer. Nonhuman primate.
Nonclinical Findings Related to the Ocular Administration of Adeno-Associated Virus Agents
Various histopathologic findings have been identified following administration of AAV-based retinal gene therapies. Many ocular findings have correlates in-life that can be identified by slit lamp biomicroscopy, ophthalmoscopy, and/or advanced imaging techniques such as OCT. These findings may be observed within the eye and, to a lesser extent, outside it. The underlying pathogeneses of many such lesions are currently uncertain.
Ocular Findings
Procedure-related findings
During an ocular toxicity study, procedure-related findings are a common confounder in histopathology evaluation. These findings may be related to either the dosing procedure itself or to in-life or postmortem sampling of the eye conducted to enable BD or PK assessment in the aqueous and/or vitreous humors.
Currently, the most frequently utilized ROA for retinal gene therapies are intravitreal, subretinal, and suprachoroidal. All these intraocular routes are relatively invasive, so findings associated with the administration procedure are common in ocular toxicity studies. Procedure-related damage is a particular concern for subretinal ROA in which injection of the TA into the potential space between the neuroretina and retinal pigmented epithelium (RPE) forcibly detaches the retina. Procedure-related findings are sometimes ignored in making adversity decisions since the focus is on effects attributed to the TA. 26 However, such procedural effects should be described in detail and discussed with respect to their discrimination from TA-related findings.
When considering histopathologic findings related to the procedure, it is important to understand species differences in anatomy and physiology as well as any differences between the actual procedures applied to the eye. For example, different species have divergent propensities toward developing procedure-related findings. The comparatively large lens sizes in rodents or rabbits may predispose to iatrogenic lens trauma during intravitreal dosing in these species 16 while (in the authors’ experience) subretinal injection may result in extensive degeneration and atrophy of their relatively delicate retinas, in both cases to degrees not observed in the NHP. Therefore, from a risk assessment perspective, procedure-related findings in rodents and rabbits are often dismissed as not relevant to humans, although adverse to the test species. In contrast, in larger species (particularly NHP), procedure-related findings warrant greater consideration when utilizing clinically relevant dosing procedures given the significant similarities in anatomy and physiology of the NHP eye to the human eye.
Procedure-related findings are particularly common with the subretinal ROA and are most evident in the RPE. 34 The most frequently observed procedure-related findings are pigment alteration in the RPE cells, identifiable as increases or decreases in pigment quantity and distribution within the cytoplasm, as well as hypertrophy, detachment, and loss of polarity of the RPE cells (Figure 3). Focal subretinal fibrosis (see below for a more detailed description) may also develop at the administration site.
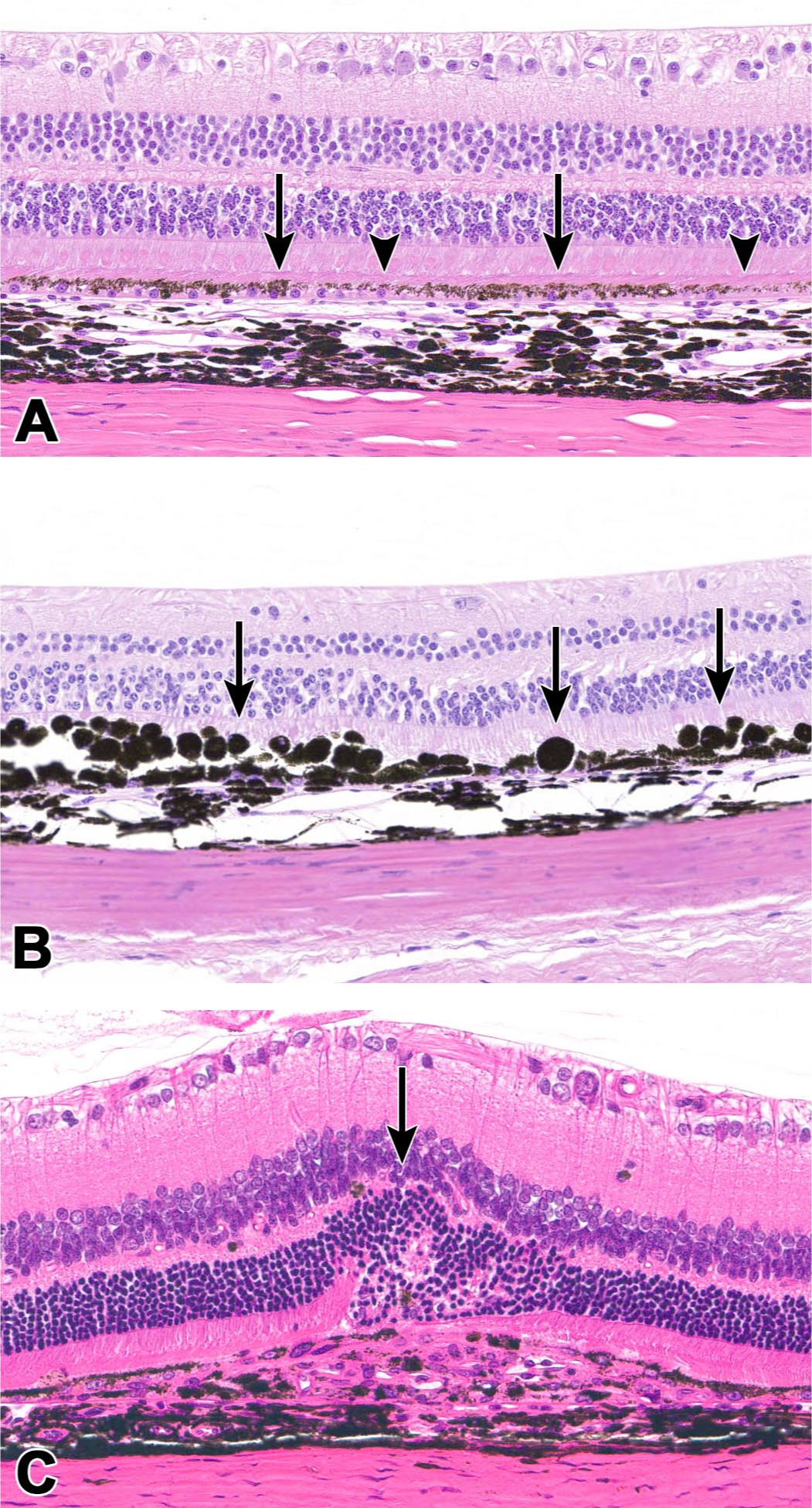
Procedure-related findings secondary to subretinal dose administration. (A) Retinal pigmented epithelium (RPE) demonstrating pigment alteration characterized by regional (extracellular and intracellular) increases/changes in pigment distribution (arrows) and decreases in pigment (arrowheads), often within the same field. Nonhuman primate (NHP). (B) RPE, hypertrophy and loss of polarity (arrows). NHP. (C) Focal subretinal fibrosis with RPE disruption and focal disorganization of the overlying retina, associated with the administration site (arrow). Rat.
In-life evaluations can aid in determining whether a finding is related to the dosing procedure, particularly as the range of potential procedure-related findings is often not captured within the limited numbers of concurrent control animals on a nonrodent toxicity study. Many of the early procedure-related findings observed by ophthalmoscopic examination (such as pinpoint acute retinal hemorrhages) have resolved completely by necropsy, and therefore have no histopathologic correlate. However, procedure-related RPE findings may still be observed in life and histopathologically at the end of the study and may appear morphologically like TA-related effects. In these cases, the location (usually at the inferior bleb margin) and time course (arising within a few days of dosing and stabilizing or resolving with time) may aid in their differentiation as procedural vs TA-related effects.
Inflammation/infiltration
Ocular inflammation is a common sequel to intraocular administration of AAV-based gene therapy vectors. This finding is observed both nonclinically and clinically, and associations have been shown with both dose (where increases in dose raise the incidence and severity) and ROA (where intravitreal injections elicit more inflammation compared to subretinal and suprachoroidal delivery).14,46 Product quality aspects, including the numbers of empty capsids in a given dose or the presence of contaminants (such as endotoxin or host-cell DNA), may also contribute to the extent of this finding. 44
In patients, inflammation associated with ocular gene therapies (termed “gene therapy-associated uveitis” [GTAU]) is observed, which often triggers routine prophylaxis with systemic and/or topical corticosteroids. This inflammation may be clinically significant, leading to deterioration of visual acuity, and may be associated with gradual loss of therapeutic efficacy (after initial functional improvements). 11
Nonclinically, inflammation after the ocular administration of AAV-based vectors is common and often biologically consequential.10,14 The inflammation may substantially impact animal welfare, causing pain and visual impairment that may necessitate early euthanasia for humane reasons. From the toxicology and pathology perspectives, ocular inflammation itself may produce noteworthy secondary degenerative changes in the surrounding tissue. Such inflammatory lesions may potentially impact the ability to identify primary TA-related toxicities. Moreover, immune-mediated destruction of transduced cells over time may decrease expression and therefore exposure to the transgene product.
Leukocyte infiltrates or inflammation are the histopathologic manifestations of the ocular inflammation that occurs in life. In the globally recognized terminology set forth by the INHAND (International Harmonization of Nomenclature and Diagnostic Criteria for Lesions) initiative, “infiltrates” are leukocyte accumulations that do not damage the involved organ parenchyma whereas “inflammation” is characterized by leukocyte influx that injures the parenchyma. In the eye, these two diagnoses are distinguished from one another by the presence of associated effects such as edema, congestion, hemorrhage, degeneration, and/or necrosis accompanying inflammation. 39 Traditionally, in-life assessments as part of ophthalmologic evaluation, particularly indirect ophthalmoscopy and slit lamp biomicroscopy, have been considered to be more sensitive than histopathologic evaluation in identifying and scoring ocular inflammation as the in-life methods not only permit evaluation of the globe as a whole but are also amenable to longitudinal monitoring of any structural findings.2,21 Predominantly affecting the uvea (specifically the iris and/or ciliary body anteriorly and the choroid posteriorly), leukocyte infiltrates are often comprised of mononuclear cells (primarily lymphocytes and macrophages), although in some cases mixed cell lesions may also include neutrophils and possibly a few multinucleated giant cells. Inflammation can be extensive and associated with substantial bystander effects, particularly when located in the choroid, where it may disrupt Bruch’s membrane (located between the retina and the choroid), the RPE, and the outer retina (Figure 4).
Inflammation associated with ocular administration of AAV-based gene therapy vectors is often extensive and typically is most substantial in the uvea (iris, ciliary body, and choroid). Choroidal inflammation may disrupt the overlying retina, thus leading to degeneration of the retinal pigmented epithelium (RPE) and/or overlying retina. (A) Mononuclear cell infiltrates, ciliary body (asterisk); (B) mononuclear cell inflammation in the choroid and retina, disrupting RPE (arrows) and cuffing retinal vessels (arrowhead) (compare to control retina in Figure 1); (C) mononuclear cell inflammation in the choroid (asterisk) with intact overlying RPE. Nonhuman primate.
Multifocal, perivascular leukocyte cuffing of retinal vessels may also be identified, similar in appearance to that commonly observed in the nonclinical development of intravitreal biologics. 15 In some cases, this finding is correlated with systemic anti-drug antibodies (ADA) against non-native (typically human-derived) transgene products and may suggest a contribution of an adaptive humoral immune response to the process. Depending on relative sequence homology, the nonclinical ADA response to the human-origin transgene product may not be predictive of ADA formation in humans, so ocular inflammation secondary to this xenogeneic antigen may be exacerbated in nonclinical species when compared to patients. 38
Although co-medications are avoided whenever possible during nonclinical toxicity studies, prophylactic immunosuppression (IS) regimens often may be incorporated during nonclinical safety assessment of AAV-based ocular gene therapies. The twin rationale for their inclusion is that nonclinical study endpoints may be compromised in their absence, and ocular gene therapy patients typically receive topical and/or systemic corticosteroids to facilitate transduction and maintain longer-term expression. Accordingly, prophylactic IS should be considered nonclinically for a proportion of groups as an aid to prevent/control ocular inflammation, and may be needed across all groups, particularly for chronic toxicity studies, as inflammation can greatly impact the ability to identify potentially translatable, irreversible lesions (Figure 5). Nonetheless, in risk assessment for retina-targeted gene therapy TAs, ocular inflammation—while a potentially severe sequel—is both a known and monitorable risk and a potentially reversible finding,
Impact of immunosuppression (IS) on histopathologic findings produced by a subretinal administration of an AAV-based ocular gene therapy vector. (A) No prophylactic IS. There is significant choroidal inflammation that obscures the retinal pigmented epithelium (RPE) and outer retina (asterisk). (B) Use of prophylactic IS (subconjunctival triamcinolone at the time of subretinal dosing along with intramuscular methylprednisolone [40 mg] once weekly) leads to substantially less inflammation while RPE degeneration is still observed, suggesting a direct toxic effect of the test article on RPE. Nonhuman primate.
Due to logistics around frequent administration of topical corticosteroid treatments to large cohorts of animals (particularly NHPs) and the relative resistance of some nonclinical species to corticosteroids, adaptation of typical clinical protocols for prophylactic IS is usually not appropriate for direct nonclinical implementation. That said, corticosteroid protocols in the nonclinical setting have some advantages in terms of interpreting toxicity data, such as producing a well-characterized spectrum of clinical pathology and histopathology findings that can readily be differentiated from TA-related effects. In practice, local and systemic IS treatments can effectively be undertaken in the NHP by administering subconjunctival triamcinolone at the time of dosing followed by intramuscular methylprednisolone weekly. This regimen is usually very well tolerated in nonclinical species.
Retinal pigment epithelium, pigment alteration
Changes in pigment distribution are a common response of RPE cells to insult or stress. Findings can vary from decreased or increased pigment quantity through aberrant pigment character such as intracytoplasmic melanin “clumping” rather than the normally uniform distribution of fine brown-black pigment granules. 9 As noted above, this finding can also be observed as a procedure-related finding following subretinal injection, but it is additionally seen as a TA-related effect with both subretinal, and to a lesser extent suprachoroidal, ROA.
In life, RPE pigment alteration may be seen as mottling of the RPE by indirect ophthalmoscopy. In general, TA-related pigment changes tend to arise at least 2 weeks after treatment, so they may be differentiated from procedure-related findings based on the time course; a dose-response relationship is frequently observed with respect to incidence and severity. Similar morphological changes are reported in patients treated with subretinal gene therapies.13,27
Retinal pigment epithelium, degeneration
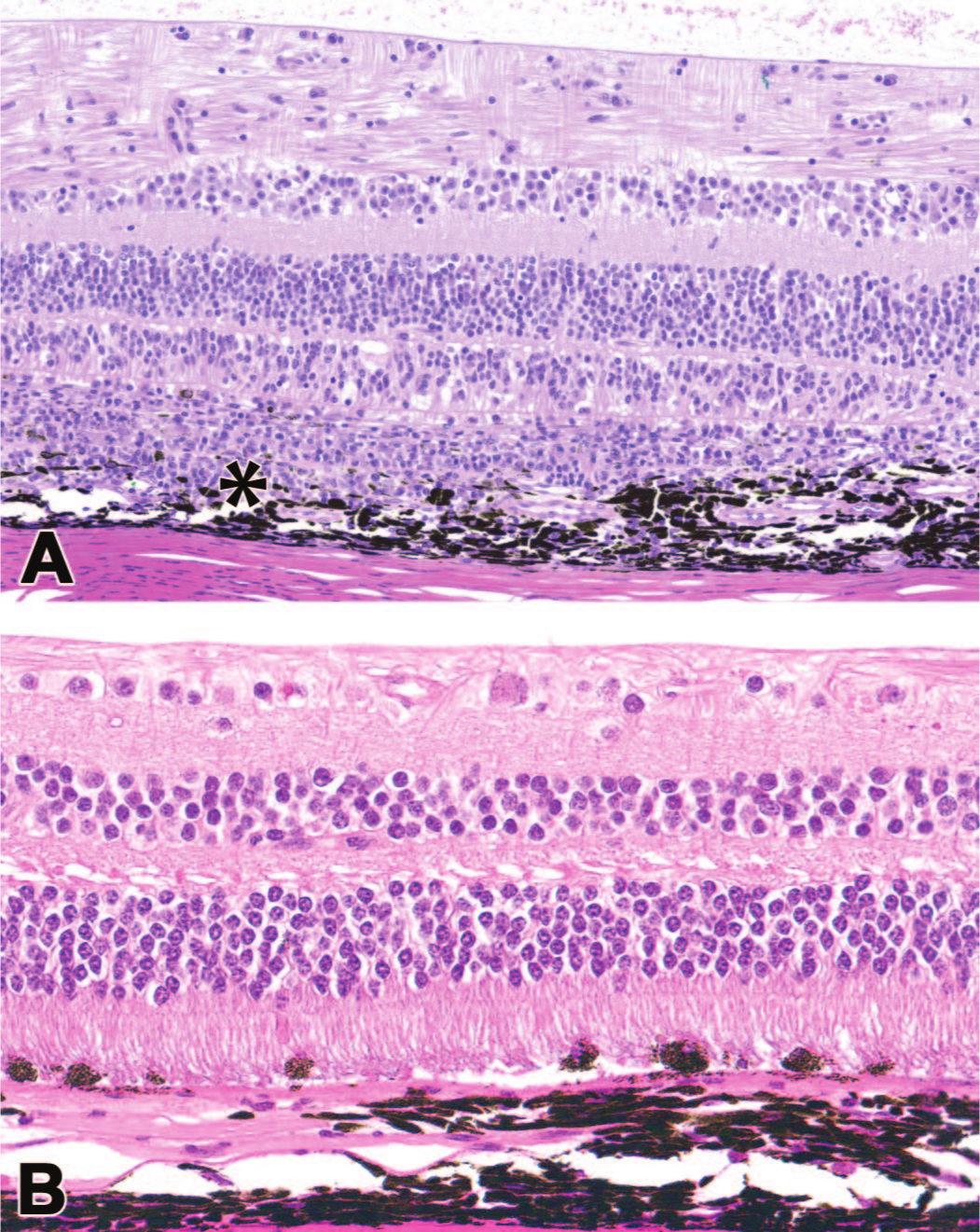
Degeneration of the RPE is a dose-related finding observed commonly with subretinal and rarely with suprachoroidal ROA. It is characterized histologically by attenuation of RPE cells, loss of apical microvilli, and changes in pigmentation. In some cases, pigmented cells (typically interpreted as detached RPE cells) are present in the outer retina (Figure 6).
Retinal pigmented epithelium (RPE) degeneration observed with subretinal (SR) administration of AAV-based gene therapy vectors is evident as a series of structural effects associated with disruption of the retina: RPE interface (enclosed in blue box). Compared to the RPE of a vehicle control animal (A), SR injection of an AAV agent (B) leads to variable RPE attenuation, pigment alteration, and RPE detachment with localized degeneration in the retinal photoreceptor layer. During life, degeneration of RPE is characterized by optical coherence tomography (OCT) as loss of normal lamination in the outer retina (D, blue box) with hyper-reflective foci (D, arrows) compared to the pretest baseline appearance (C), and by indirect ophthalmoscopy as retinal mottling (altered pigmentation [E, white circle]), all of which correlate with the microscopic presence of RPE degeneration and detached, depolarized RPE cells (F, arrows). Nonhuman primate. (Image E provided courtesy of Paul Miller and Hugh Waber.)
The RPE degeneration does not appear to be a species-specific effect and has been observed and/or reported in the literature in the rat, mouse, minipig, and NHP (Figure 7). 48 The RPE degeneration is often associated with high vector transduction and high expression of transgene product. It is independent of capsid serotype or transgene, and there are data to suggest it can be mitigated by decreasing transgene expression in RPE cells. 48 However, the contributions of the innate and/or adaptive immune responses in driving this effect are not clear.
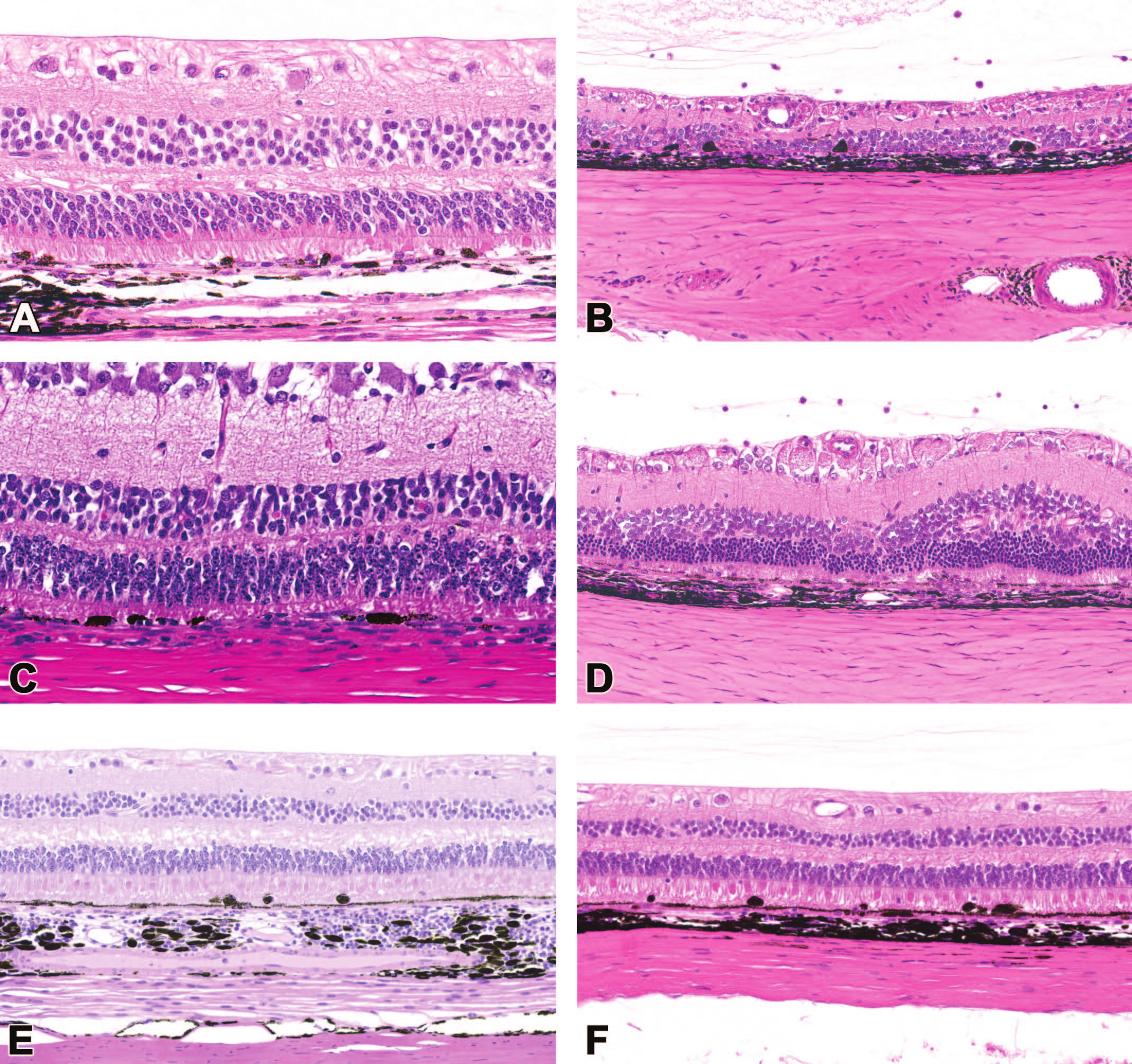
Dose-dependent retinal pigment epithelium (RPE) degeneration is observed across species, with various capsids, transgenes, and both secreted and intracellular proteins introduced by AAV-based retinal gene therapies possessing strong ubiquitous promotors. (A) AAV serotype 8 (AAV8), secreted transgene product, nonhuman primate (NHP); (B) AAV8, intracellular transgene product, rat; (C) AAV8, intracellular transgene product, minipig; (D) AAV9, intracellular transgene product, rat; (E) AAV2, secreted transgene product, NHP; (F) variant AAV, secreted transgene product, NHP.
In life by indirect ophthalmoscopy, RPE degeneration may be seen as RPE mottling similar to that observed with RPE pigment alteration. However, optical coherence tomography (OCT) reveals loss of differentiation of the outer retinal layers (ellipsoid zone), disruption of the outer retina, and hyper-reflective foci in the outer retina (thought to represent degenerating RPE cells/pigmented cells in the outer retina) (Figure 6). These findings tend to arise at least a month after dosing (in some cases much later) and progressively spread away from the bleb area.
As the RPE is required for retinal function, RPE degeneration may progress to outer retinal or even global retinal atrophy (Figure 8). In rodents, degeneration of the overlying retina appears to happen rapidly, but in the NHP overlying retinal morphology may be normal despite significant changes to the subjacent RPE. Functional manifestations, particularly in the multifocal electroretinogram (mfERG), may be seen prior to morphologic effects in the overlying retina.
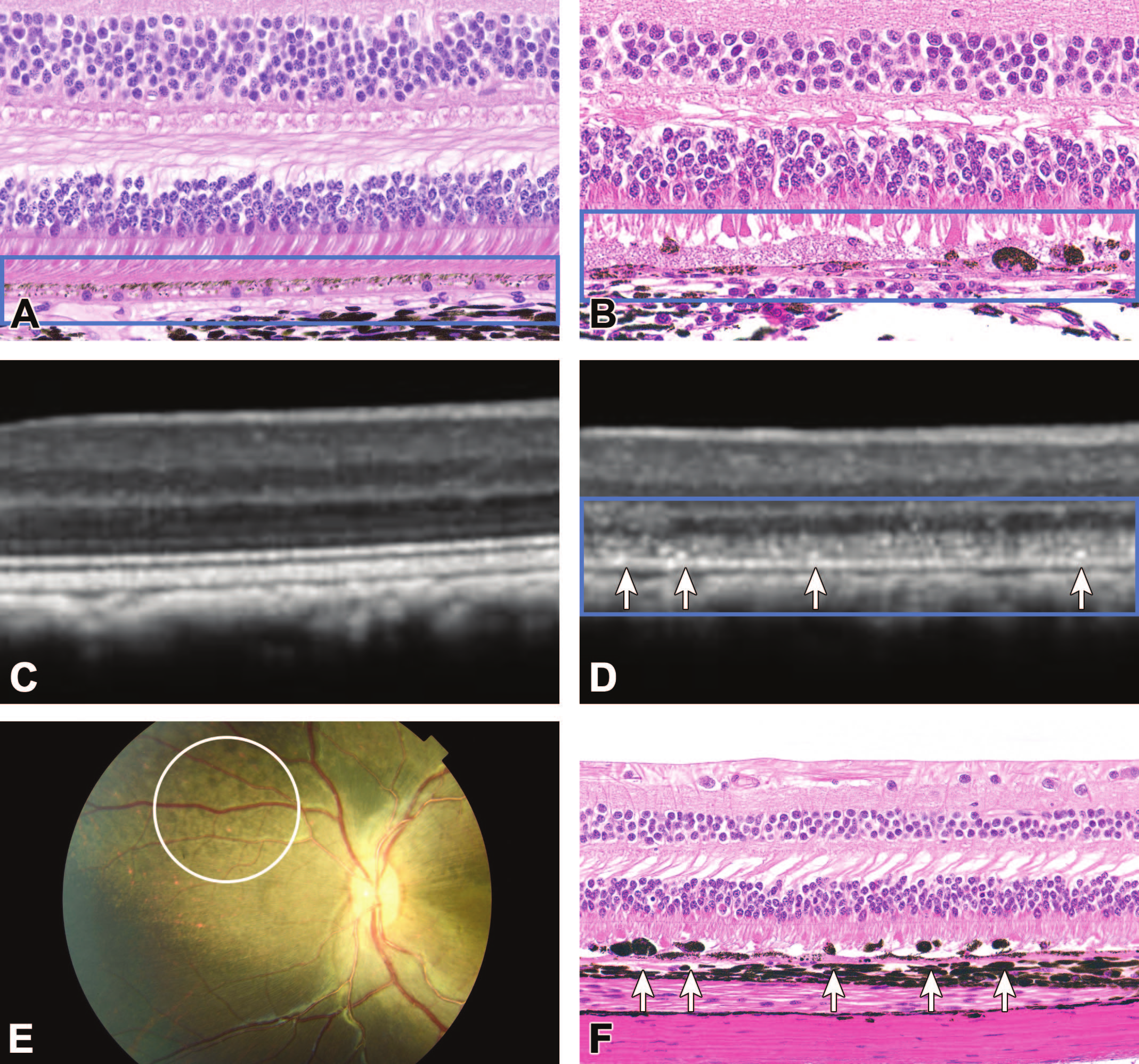
Retinal degeneration and atrophy localized at and around the bleb area (administration site) are common sequelae to subretinal administration of AAV-based gene therapy vectors. Compared to a control retina (Figure 1), affected retinas are thinner (A), have attenuated or missing layers (A, B, C), and/or exhibit disorganized cell organization in one or more nuclear layers (B, C) and/or the photoreceptor layer (B, C). In rare cases, the retinal pigmented epithelium (RPE) may be degenerate (C), or retina and subjacent RPE may be entirely absent (D). Altered retinal thickness and loss of lamination may be demonstrated during life using optical coherence tomography (F, compared to the pretreatment baseline structure in E). Nonhuman primate. Abbreviations: PR, photoreceptor layer; ONL, outer nuclear layer; INL, inner nuclear layer; GCL, ganglion cell layer.
Findings like those observed in nonclinical species have begun to be reported in the literature in patients treated by subretinal administration of AAV-based gene therapy vectors. Atrophy of the RPE and retina has specifically been reported as post-marketing findings in patients treated with Luxturna.7,19,41 In some macular degeneration patients treated with subretinal RGX-314 injections, pigmentary changes (in the macula and associated with decreases in vision), ellipsoid zone changes (by OCT), and, in some cases, RPE clumping and retinal thinning have been observed 6 to 12 months after dosing. 13 Interestingly, the RPE morphologic changes and their progression as observed in nonclinical studies of ocular AAV therapies are similar both during life and by microscopy to those described for geographic atrophy in human patients. 19
Retina, degeneration and atrophy
Retinal degeneration and atrophy are frequent findings in nonclinical studies of ocular AAV agents, particularly those with subretinal ROA. These lesions are commonly characterized by variable attenuation or loss of retinal layers with cell fragmentation and increased intercellular spaces (Figure 8). Retinal degeneration and atrophy may be primary findings, but they occur more commonly secondary to primary RPE degeneration and/or RPE or retinal inflammation. Retinal degeneration and atrophy also may be a procedural effect, arising secondary to the retinal detachment that develops following subretinal injection. Procedure-related retinal degeneration is more common in the rabbit and the rodent but may be seen in any species if persistence of the injection site bleb is prolonged.
In life, retinal degeneration and atrophy may be observed longitudinally by indirect ophthalmoscopy, but they can frequently be more objectively quantified, and the specific retinal layers that have been impacted can be more effectively assessed, with OCT (Figure 8). Retinal degeneration, thinning, and/or atrophy have been reported nonclinically with CPK850 and in patients treated with voretigene neparvovec or RGX-314.7,13,19,29,41
Fibrosis, subretinal
Subretinal fibrosis (expansion of the subretinal space by connective tissue) is seen occasionally with subretinal AAV-based gene therapies, more commonly in rodents than in NHPs. This may be observed in the rodent or NHP as a focal, minimal, procedure-related change. Unlike procedure-related subretinal fibrosis, which is usually focal, minimal, and related to the retinotomy site (the point at which the dosing cannula penetrates the retina during a subretinal injection), TA-related subretinal fibrosis is typically more extensive, progressive, and occurs in association with degeneration or loss of RPE cells (Figure 9). The connective tissue is thought to be produced by a mesenchymal cell population that may originate from RPE via a process of epithelial-mesenchymal transformation (EMT). 28 Support for this hypothesis is provided by the obvious morphological effects in the RPE and the known role for EMT of RPE in the pathogenesis of subretinal fibrosis in diseases such as age-related macular degeneration (AMD).
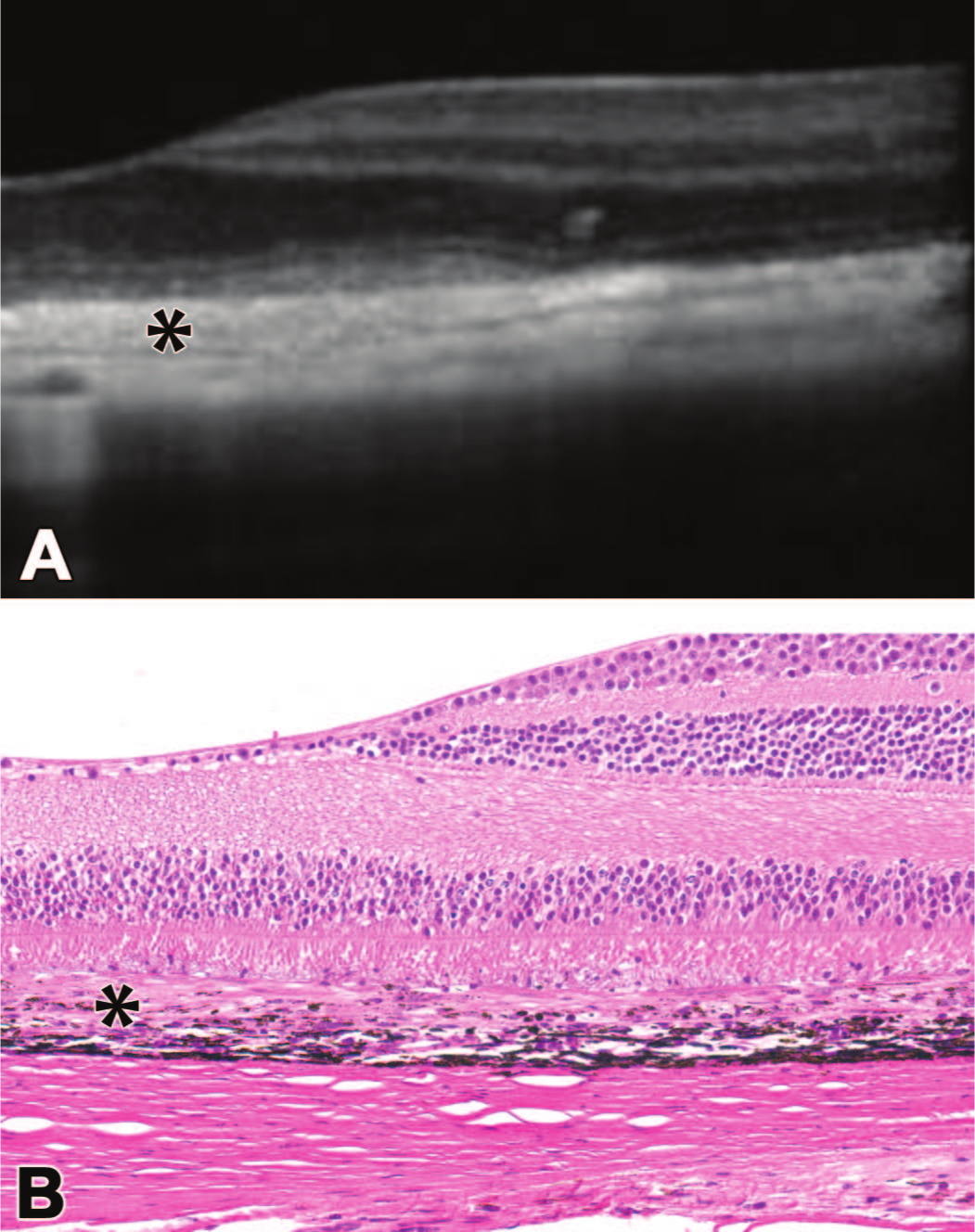
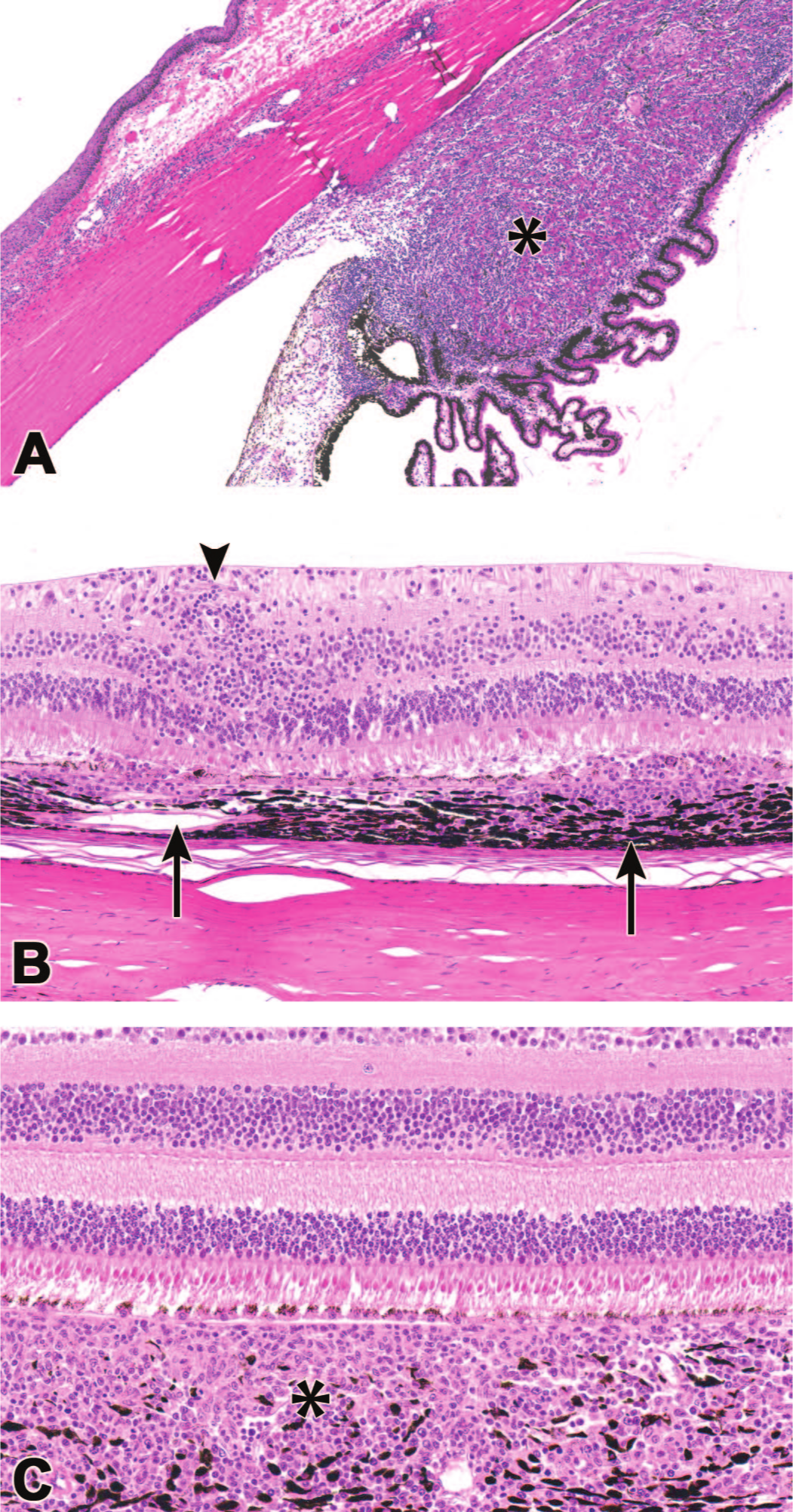
Subretinal fibrosis (asterisks) is a chronic change that may follow subretinal administration of an AAV-based gene therapy vector. At 15 weeks post-injection, the finding is present as increased subretinal hyper-reflectivity with loss of normal retinal lamination during life by optical coherence tomography (A), which correlates with subretinal deposition of connective tissue with proliferation of mesenchymal cells associated with degeneration/loss of the overlying retinal pigmented epithelium (B). Nonhuman primate.
In life, subretinal fibrosis is observed as a yellow-white subretinal opacity by indirect ophthalmoscopy. In contrast by OCT, this finding appears as a hyper-reflective proliferation at the level of the RPE (Figure 9).
Iris, posterior epithelium, degeneration
Degeneration of the posterior iris epithelium has been observed with some intravitreal injections of AAV-based gene therapies in the NHP. Usually occurring one to two weeks after dosing (although observed at time points up to 4 weeks), the pathogenesis is uncertain.
Histologically, the finding is characterized by rounding and detachment of epithelial cells with attenuation of the remaining epithelium and, in some cases, exposure of the denuded basement membrane and edema of the iris stroma (Figure 10). In the NHP iris, these cells are intensely pigmented, so intracellular features of degeneration may be difficult to observe. If extensive, affected animals may develop posterior synechiae (ie, adhesions between the posterior iris surface and lens) or even iris bombe (ie, anterior bulging of the iris due to full-circumference posterior synechiae leading to fluid buildup between the iris and lens). In life, changes in iris pigmentation (pigment dispersion) and pigmented cells in the aqueous (cell score/color) can be observed on slit lamp biomicroscopy.
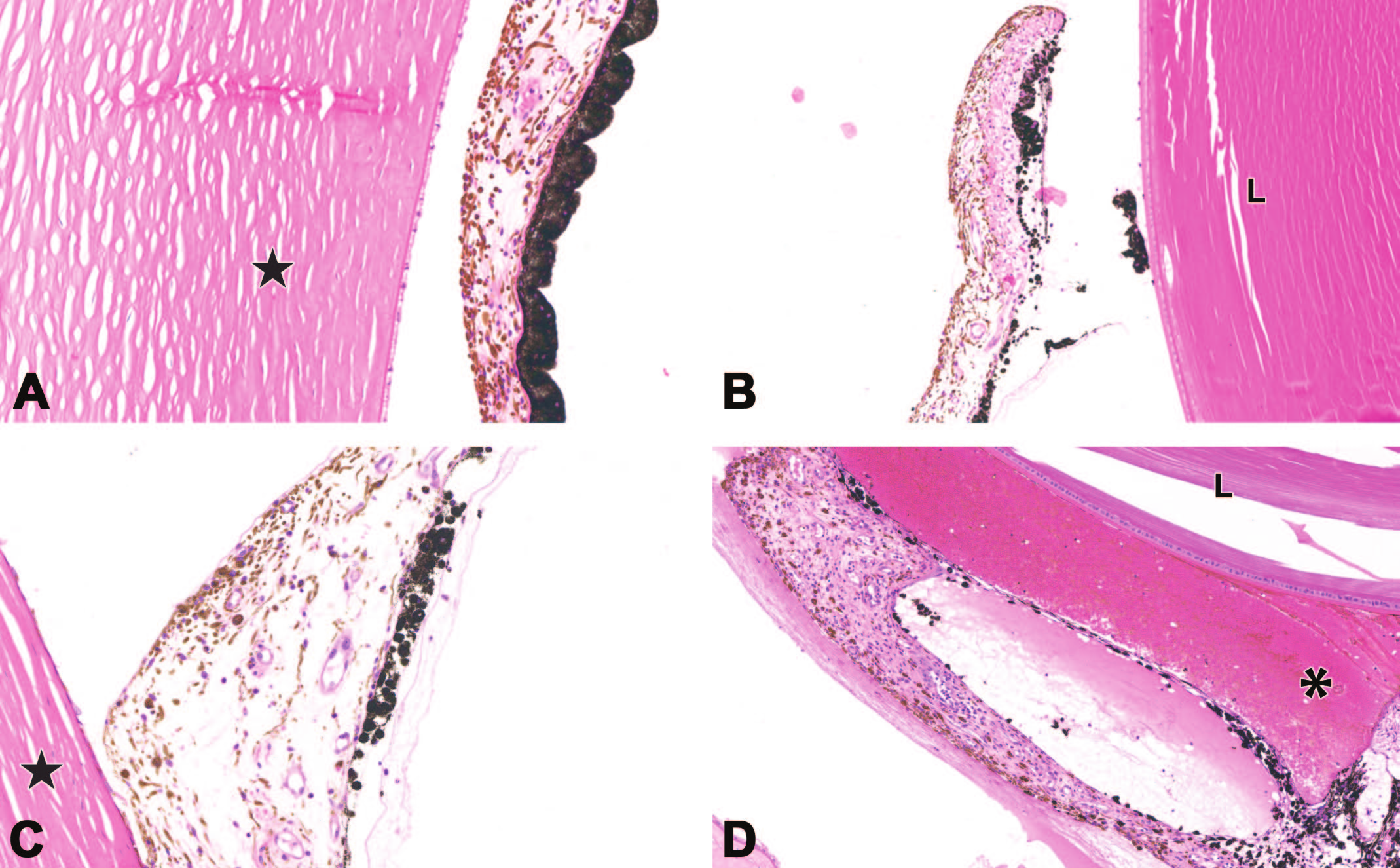
Degeneration of the posterior iris epithelium may develop following ocular administration of an AAV-based gene therapy vector. Relative to the control structure (A), the iris of an AAV-treated eye may exhibit attenuation (B, C) or loss (B) of the posterior epithelium, sometimes in conjunction with degeneration of the ciliary body epithelium (asterisk, D). Synechiae (a secondary finding) are indicated by attachment of the iris or pigmented epithelial cells to either the cornea (anterior synechiae) or lens (posterior synechiae). The star in (A) and (C) identifies the cornea, while the “L” in (B) and (D) denotes the lens. Nonhuman primate.
As with RPE degeneration, ciliary body and posterior iris epithelial degeneration appear to be often associated with high vector transduction at these locations following intravitreal ROA. However, the involvement of innate and/or adaptive immune response or exogenous protein expression in the degeneration of anterior segment structures is not clear.
Iris or ciliary body, pigmented cells, stroma
Pigmented cells within the iris stroma represent a chronic change that may be seen after some intravitreal administrations of AAV-based ocular gene therapies in the NHP. In the iris, these pigmented cells must be differentiated from the normal iris plications, which are commonly seen along the iris margin. These pigmented stromal cells are rounded, heavily pigment-laden, and labeled immunohistochemically with the macrophage marker CD68 (Figure 11). This finding is considered to be a possible sequel to iris/ciliary body epithelial degeneration, with subsequent removal by a macrophage population of pigment released from degenerate cells.
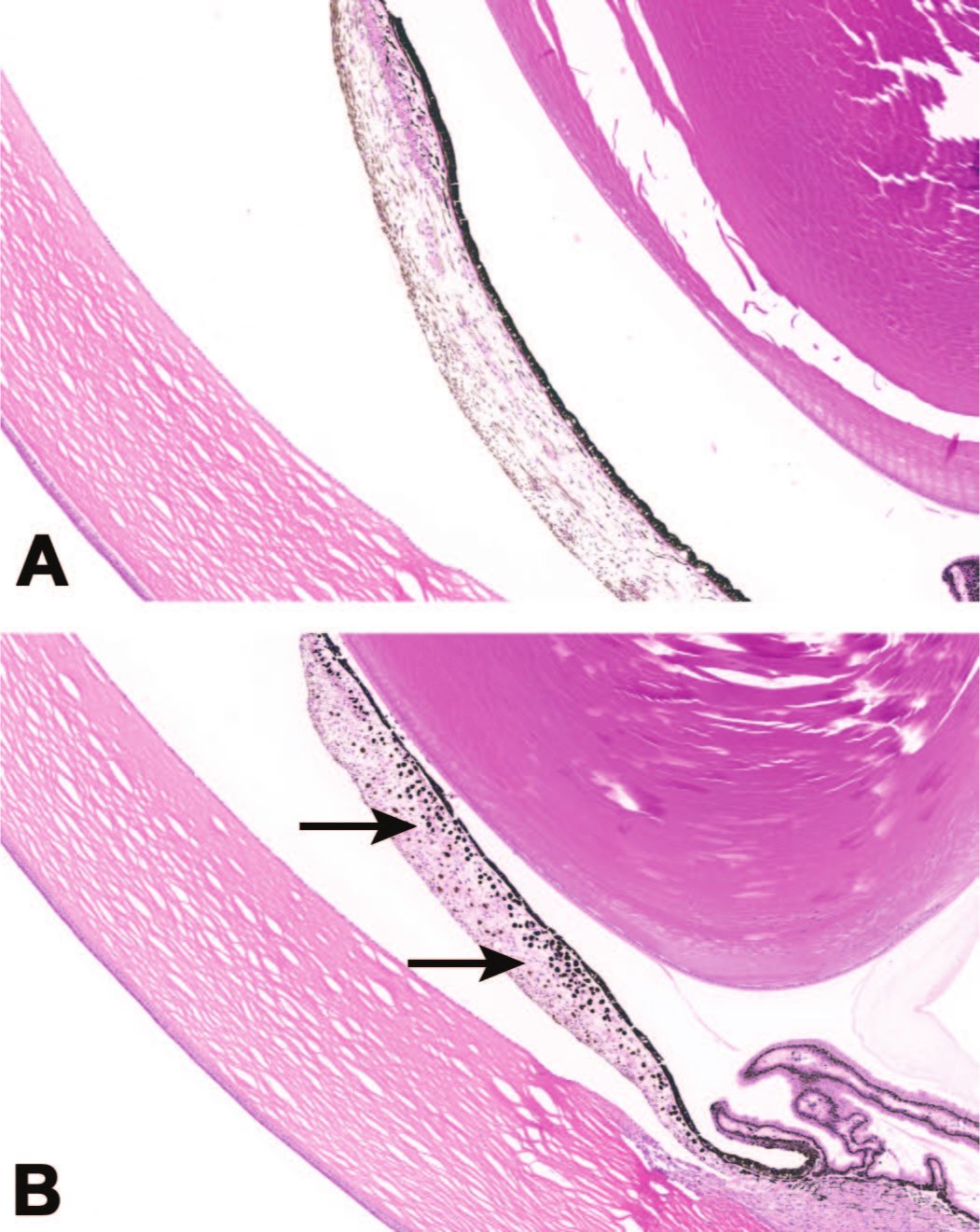
Increased pigmented cells in the iris stroma (arrows) may occur as a sequel to earlier degeneration of the posterior iris epithelium following ocular administration of an AAV-based gene therapy vector. (A) Control and (B) intravitreal injection of AAV. Nonhuman primate.
During life, altered iris pigmentation may be observed by slit lamp biomicroscopy as a clinical correlate for substantial microscopic lesions.
Ciliary body, degeneration and atrophy
Ciliary body degeneration as an acute change with ciliary body atrophy as a potential chronic sequel has been observed in NHPs given intravitreal injections of AAV-based ocular gene therapy vectors. The extent and severity of these findings vary considerably. An effect may be evident only on the ciliary body epithelium, where the appearance is similar to that of degeneration of the posterior iris epithelium. More substantial lesions may affect both the ciliary body epithelium and the ciliary processes, leading to loss of the ciliary processes as well as epithelial degeneration. In the most severe cases, the epithelium, ciliary processes, and the underlying muscle of the ciliary body are all impacted (Figure 12).
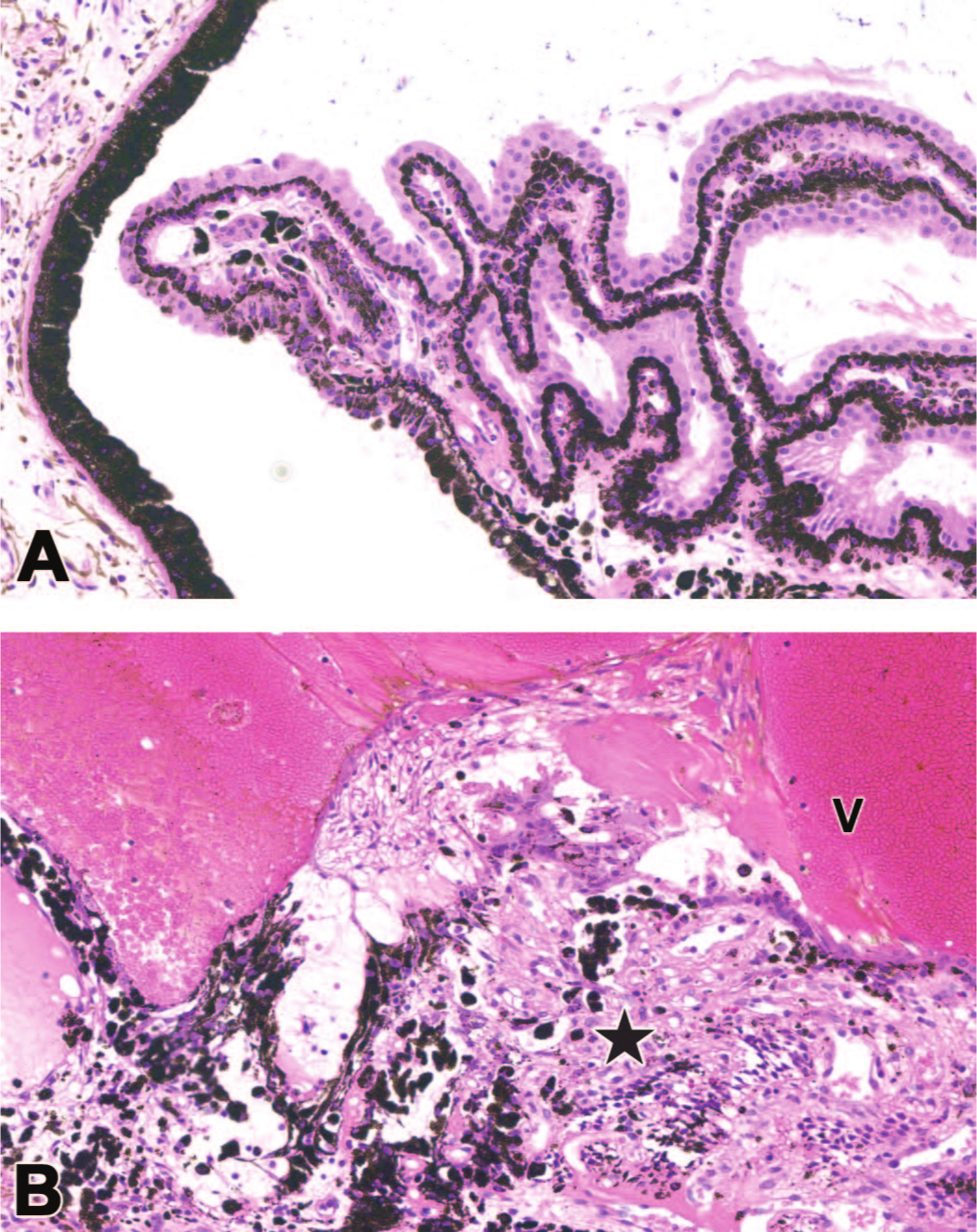
Degeneration of the ciliary body epithelium and loss of ciliary processes may develop following ocular administration of an AAV-based gene therapy vector. Compared to a control eye (A), intravitreal AAV injection may induce degeneration and detachment (cell displacement into the vitreous) of the ciliary body epithelium, expansion of the ciliary body stroma, and/or fusion of the ciliary processes (B [star]). Substantial hemorrhage is evident as a field of intact erythrocytes within the vitreous (V). NHP.
Ciliary body and posterior iris epithelial degeneration are often associated with high vector transduction associated with intravitreal ROA. However, the involvement of innate and/or adaptive immune response or exogenous protein expression in the degeneration of anterior segment elements is not clear.
The ciliary body is nearly impossible to visualize during standard in-life evaluations. Degeneration of the epithelium, as with posterior epithelial degeneration of the iris, may yield release of pigmented cells into the aqueous humor that can be visualized by slit lamp biomicroscopy. Changes in intraocular pressure (IOP) measured by tonometry may also be observed as the ciliary body plays a key role in aqueous fluid dynamics, including the production of aqueous humor (which is essential in maintaining IOP).9,22 Ciliary body atrophy will lead to failure of aqueous production and eventually decreased IOP (ie, hypotony), which can be a vision-threatening event. 35
Given the propensity of ocular AAVs to induce inflammation, chronic inflammation affecting the ciliary body (“uveitis”) may be a cause of ciliary body atrophy and resultant hypotony. 35 As such, control of intraocular inflammation is particularly important to distinguish between what is potentially a primary effect of the initial AAV-based therapy and what could be secondary to long-standing inflammation.
In a phase I clinical trial in diabetic macular edema (DME) patients, ocular administration of ixoberogene soroparvovec (Ixo-vec, previously called ADVM-022) resulted in rapid, clinically relevant decreases in IOP that were refractory to steroids. This clinical change occurred at 16 to 36 weeks after treatment with the high AAV dose. 1
Extraocular Findings
Ocular administration of AAV-based gene therapies usually produces few systemic findings, but histopathologic findings have been observed in a limited number of extraocular tissues. Commonly affected sites are physically connected to the eye, including the extraocular muscles (EOM), ciliary regional autonomic (ciliary) ganglia, and central visual pathways (ie, visual domains of the brain). Therefore, collection and evaluation of these tissues are important to completely assess the range of potential toxicities in ocular AAV studies, and the tissues (or sub-anatomic regions) detailed below should be included for microscopic evaluation.
Extraocular muscles
Both leukocyte infiltrates and overt inflammation have been observed in the EOM of NHP treated by various ocular ROA with AAV-based gene therapy vectors. Infiltrates usually occur as isolated perivascular foci of mononuclear cells (lymphocytes and macrophages). Inflammation typically is more extensive, may be comprised of mononuclear cells only or mixed cells (lymphocytes, macrophages, and variable numbers of neutrophils and sometimes other leukocyte classes), and is localized around vessels and/or penetrating into and among myofibers (Figure 13). The associated myofibers may be degenerating or regenerating or both depending on the time course. Extensive lesions may efface large amounts of the muscle. The specific EOM does not appear to be driven by dose location, with the various rectus muscles (lateral, medial, dorsal [superior], and ventral [inferior]) appearing to be randomly affected. Occasionally, leukocyte infiltrates and/or overt inflammation have also been recorded extending into the EOM of the eyelid (eg, levator palpebrae and orbicularis oculi). In rare instances, effects in the EOM are accompanied by infiltrates into the oculomotor nerve (cranial nerve III), extending both peripherally and centrally along the nerve trunk. The precise pathogenesis of EOM effects—as primary myotoxicity or secondary to immunogenicity directed against the transgene product—remains uncertain. Utilization of AAV capsids that do not transduce muscle cells eliminates these findings.
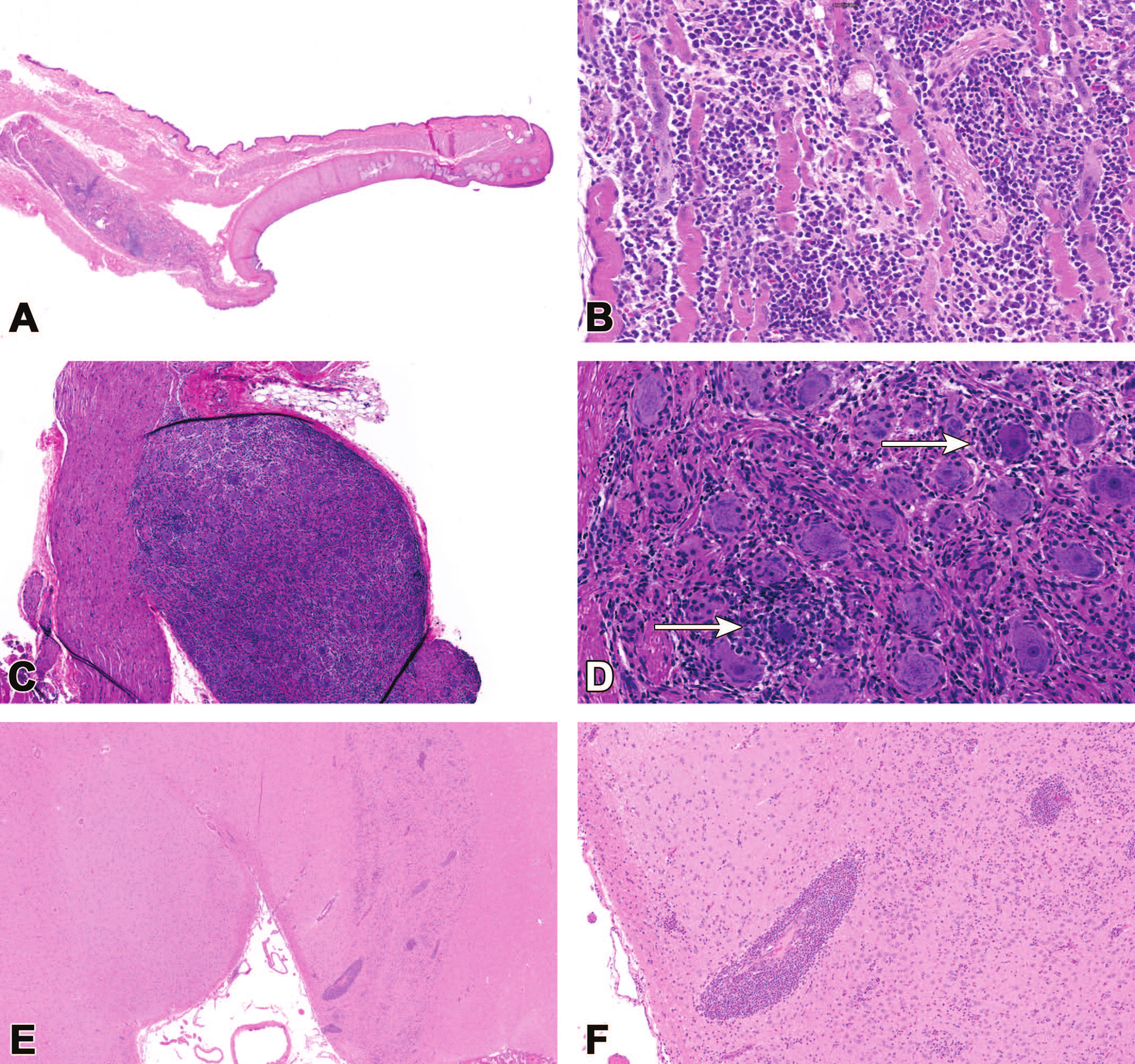
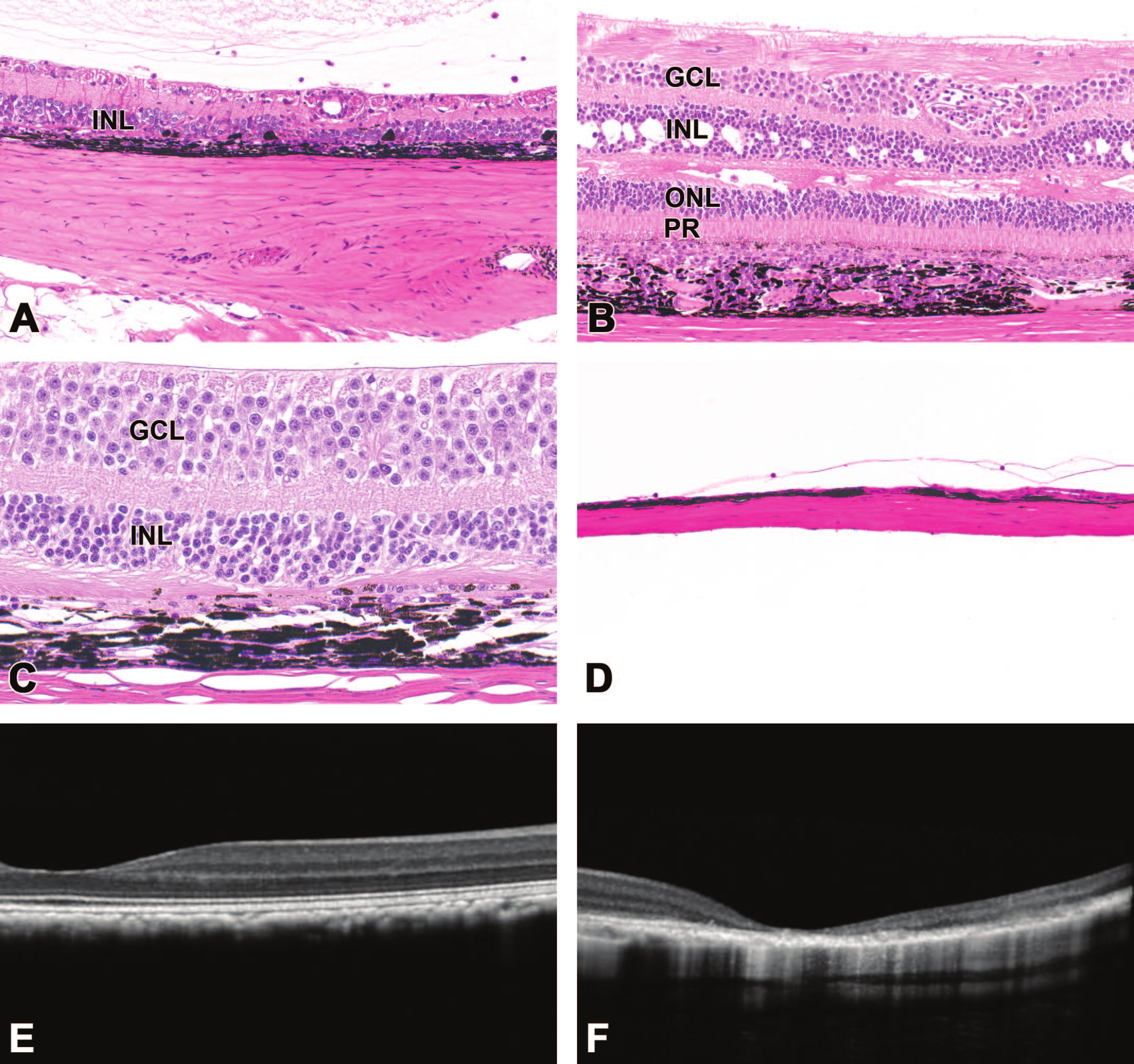
Extraocular findings are sometimes observed in tissues adjacent to or linked directly to the eye following ocular administration of AAV-based gene therapy vectors. (A, B) Mixed cell inflammation with myofiber degeneration and regeneration in the extraocular muscles of the eyelid related to suprachoroidal AAV injection. (C, D) Mononuclear cell inflammation (increased inter-neuronal cellularity) with sporadic neuronal degeneration and necrosis (arrows) and modest neuronal loss (indicated by increased stromal prominence), ciliary ganglion. (E, F) Mononuclear cell infiltration (visible as multifocal perivascular leukocyte cuffs) and gliosis (evident as variably increased cellularity within the regional parenchyma) in the lateral geniculate nucleus (LGN, a visual processing center in the brain) following subretinal AAV administration. Nonhuman primate.
Ganglia
Toxicities of the dorsal root ganglia (DRG) and trigeminal ganglia have been reported as a consistent consequence of CNS (eg, intrathecal) or systemic administration (eg, intramuscular, intravenous) of AAV-based gene therapy vectors.3,4,12,23,45 To date, ganglionic toxicities have not been observed consistently with ocular administration of AAVs. However, TA-related findings have been seen sporadically in the ciliary ganglia, which are parasympathetic ganglia located near the apex of the orbit between the optic nerve and the lateral rectus muscle. 5 Histopathologic changes in affected ciliary ganglia are similar to those reported in DRG, including mononuclear cell infiltrates or inflammation and sometimes neuronal degeneration and necrosis (Figure 13).
Central visual pathways
Toxicities impacting vision may occur in the extraocular (central) visual system, as well as within the eye itself. The central visual pathways include the optic nerves (cranial nerve II, essentially a CNS extension), optic chiasm, and within the brain, the bilaterally symmetrical optic tracts, lateral geniculate nuclei (LGN), superior (dorsal) colliculi, and the primary and secondary visual cortices. Accordingly, all these visual pathway structures should be collected and evaluated for all nonclinical ocular toxicity studies.8,33
In general, AAV-related findings in the central visual system following ocular administration of AAV agents are particularly localized between the eye and the LGN. Commonly, these findings are observed as mononuclear cell infiltrates forming small perivascular cuffs; in exceptional cases, such infiltrates may be numerous and larger. Less commonly, infiltrating leukocytes may extend into the surrounding neuropil, where they are occasionally accompanied by gliosis (increased glial cell numbers and sizes within the brain parenchyma; Figure 13). The LGN serves as the initial central synapse of the retinal ganglion cell (RGC) axons, and as such findings in the central visual system to the level of the LGN may potentially occur secondary to anterograde transport of transgene product along the normal axonal transport routes of RGC axons.25,49
Adversity of Nonclinical Findings Related to Ocular Adeno-Associated Virus Administration
A principal interpretation expected for nonclinical toxicity data is whether a finding is adverse or nonadverse. In current practice, adversity may be interpreted in several ways. The “best practice” recommendations of the STP hold that adversity means that the finding causes ‘harm’ to the animal,” 26 while another perspective is that certain changes—including retinal degeneration—are “inherently adverse.” 36
Vision is a primary sense used by humans, so substantial effects to the eye or visual processing centers—findings that are considered to impact vision—are usually viewed as adverse. Nonetheless, in our experience, limited intraocular findings associated with ocular injection of an AAV-based gene therapy (eg, localized retinal or RPE degeneration) can still support advancing AAV-based TA for ocular administration into the clinic, given an appropriate benefit:risk for the patient population to be treated and the ability to easily and longitudinally monitor the eye in patients. Interestingly, as ocular inflammation is a known liability both nonclinically and clinically and because patients are proactively immunosuppressed, ocular inflammation itself does not necessarily preclude progression into clinical testing. From a pathologist’s perspective, what is key is that pathology reports provide detailed descriptions with rational interpretations of the main nonclinical findings so that clinical trial physicians and regulatory reviewers have the necessary information to underpin their patient treatment, risk assessment, and regulatory decision-making.
Conclusions
The AAV-based retinal gene therapies are a rapidly evolving field, both clinically and nonclinically. These products hold remarkable promise in addressing inherited retinal diseases that were once considered untreatable and offer groundbreaking treatment approaches for acquired ocular conditions.
The AAV-based retinal gene therapies commonly cause ocular inflammation, which is generally more pronounced with intravitreal ROA, and in some cases is accompanied by secondary degeneration and atrophy of the retina and/or RPE. With more standard utilization of prophylactic immunosuppressive protocols during nonclinical toxicity studies for AAV-based therapies, a number of toxicities have emerged as AAV-related class effects. In the eye, many of these findings were previously obscured by inflammation and/or frequently dismissed as bystander damage. However, what appear to be primary degenerative findings are also observed in the RPE, retina, iris, and ciliary body with these therapies. We now understand that many of these degenerative changes, particularly in the RPE, iris, and ciliary body, are dose-related and associated with high vector transduction and transgene product expression. The precise pathogeneses, including contributions of the innate and/or adaptive immune responses to degeneration, still remain unclear.
Extraocular structures (eg, the EOMs, local ganglia, and the central visual pathways) have also been identified as occasional albeit unintended target tissues following ocular administration of AAV-based therapies. As such, they should be considered for collection and evaluation in these studies.6,33 Recent papers on optimal tissue collection and trimming practices and preferred terminology for diagnosing ocular findings should help pathologists generate more consistent and reproducible data sets for ocular toxicity studies of AAV-based agents in the future.8,39
With our improved understanding, ophthalmological and histopathological investigations in the nonclinical setting going forward will require more detailed evaluation and correlation to define and understand the principal types, severities, pathogeneses, mechanisms, and potential relevance for translation to patients before rational risk assessments can be completed.
Footnotes
Acknowledgements
The authors gratefully thank Jayson Chen and Rebecca Atkinson-Dell for their study management, monitoring, and toxicology expertise; Paul Miller and Ryan Boyd for their invaluable past discussions around in-life correlates of ocular histopathology findings; and Paul Miller and Hugh Wabers for the fundus image included in ![]() .
.
Author Contributions
The authors are solely responsible for the contents and crafting of this paper. Dr HSB prepared the initial draft, and all authors contributed to editing the content.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
