Abstract
Abnormal findings in the biliary tree are frequently encountered in response to acute and chronic exposures to various compounds. The more common findings are described here in an overview of previous publications such as the INHAND Proliferative and Nonproliferative Lesions of the Rodent Liver and the Liver-Nonneoplastic Lesion Atlas NTP with comments regarding current considerations. This was presented at the 2023 Annual Meeting of the Society of Toxicologic Pathology. Histologic descriptions and some discussions regarding the pathogenesis of the various categories of non-neoplastic lesions in the biliary tree are presented. Discussions regarding the use of the term oval cell versus ductular reaction and the potentially neoplastic nature of cholangiofibrosis are presented in some detail.
Introduction
The biliary tree conducts bile from the modified membranes of the hepatocyte that form the borders of the canaliculus to the duodenum through a series of enlarging ducts. The lining epithelium of the biliary tree begins at the Canals of Herring, the site where the canaliculi terminate, and the epithelial lining begins. There is considerable functional and phenotypic heterogeneity in the biliary tree which underlies the modification of bile, the secretory product of the liver, along the route to the duodenum. Additional modifications take place in the gallbladder of species that have a gallbladder. The major constituents of bile are bilirubin/biliverdin, bile acids, phospholipids, cholesterol, electrolytes, and water. In addition, bile contains metabolites of both endogenous and exogenous substances. Damage to various levels of the biliary tree can be driven by the effects of these metabolites as well as a variety of endogenous substances and inflammatory mediators. This manuscript will provide an overview of the histologic appearance and pathogenesis of classic types of injury to the biliary tree. For more detail and more extensive references, please refer to the INHAND Proliferative and Nonproliferative Lesions of the Rat and Mouse Hepatobiliary System 32 and the Liver-Nonneoplastic Lesion Atlas—National Toxicology Program 21 from which this manuscript was primarily derived.
Biliary Cysts
Biliary cysts can be unilocular or multilocular and are characterized as spaces lined with a single layer of well-differentiated biliary epithelium that may be flattened (Figures 1-3). They contain clear to pale yellow fluid. Biliary cysts can arise from congenital ductal plate developmental disorders or may be found as an age-related change, resulting from dilation of ducts.19,25,26 Larger cysts can compress adjacent parenchyma. Adjacent parenchyma may also be inflamed and contain fibrosis and bile duct proliferation. Ductal plate developmental disorders leading to cyst formation are recognized in some lineages of Sprague-Dawley rats, hamsters, and dogs and are often combined with renal cysts.19,25,26 Cysts arising from congenital disorders are typically diffusely distributed throughout the liver and may be smaller than individual biliary cysts.
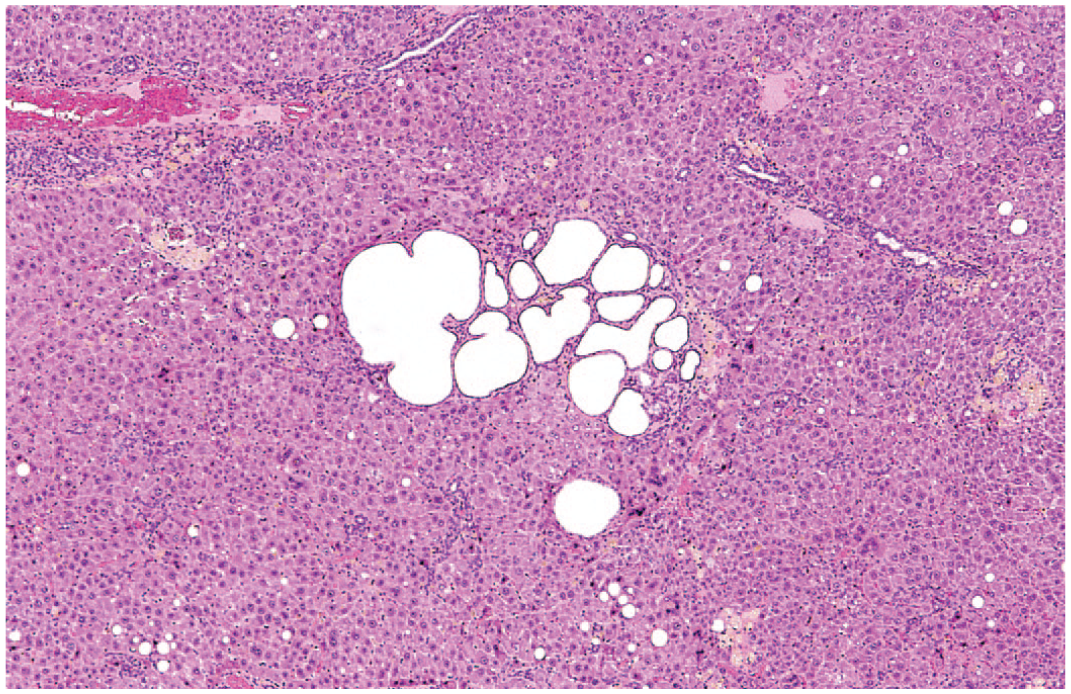
Rat liver. Biliary cyst. A small collection of cysts in a periportal region. Reprinted with permission from Sage Publications. 32
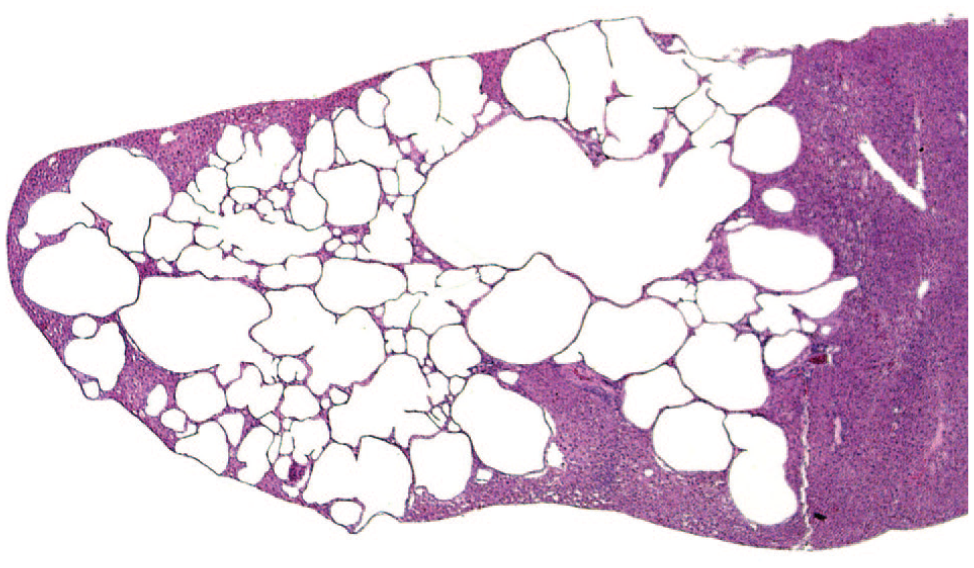
Rat liver. Biliary cyst. Substantial involvement of a liver lobe with a multilocular cystic structure lined with well-differentiated biliary epithelium. Reprinted with permission from Sage Publications. 32
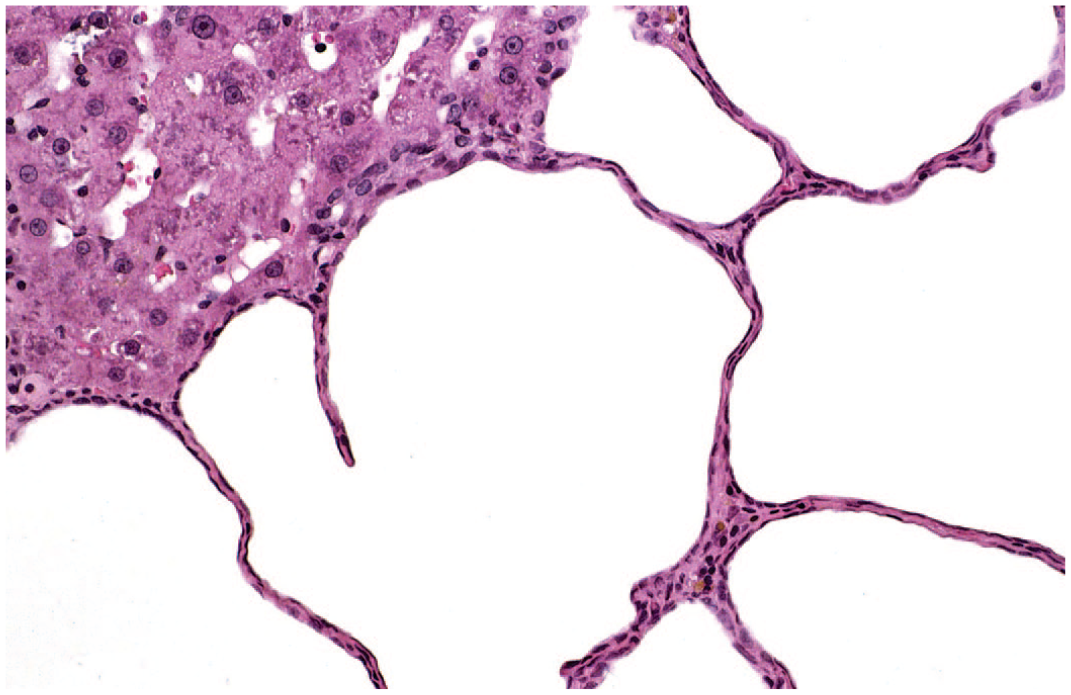
Rat liver. Biliary cyst. A higher magnification of a biliary cyst lined with flattened biliary epithelium. Reprinted with permission from Sage Publications. 32
Canalicular Cholestasis
Canalicular cholestasis is identified histologically as elongated pale green-brown plugs of bile within bile canaliculi (Figure 4). Bland cholestasis refers to evidence of canalicular plugs only without injury to the bile ducts or hepatocytes. Bile pigment, bilirubin in particular, is identified histochemically as a green pigment when stained via the Hall’s method and is best appreciated in frozen sections as the loss of bile pigment can occur in fixative. Bile plugs, particularly later in the process of cholestasis, will be phagocytosed by Kupffer cells following rupture of bile canaliculi. Bile can, on occasion, appear as fine granular pigment within the hepatocyte cytoplasm, but this finding is more frequent in primates, including humans than in rodents, and almost never occurs in dogs, where it occurs only in instances of profound cholestasis. Overall, rodents only infrequently develop canalicular cholestasis following exposure to xenobiotics, and this finding is more common in dogs and primates.
Mouse liver. Canalicular cholestasis. Reprinted with permission from Sage Publications. 32
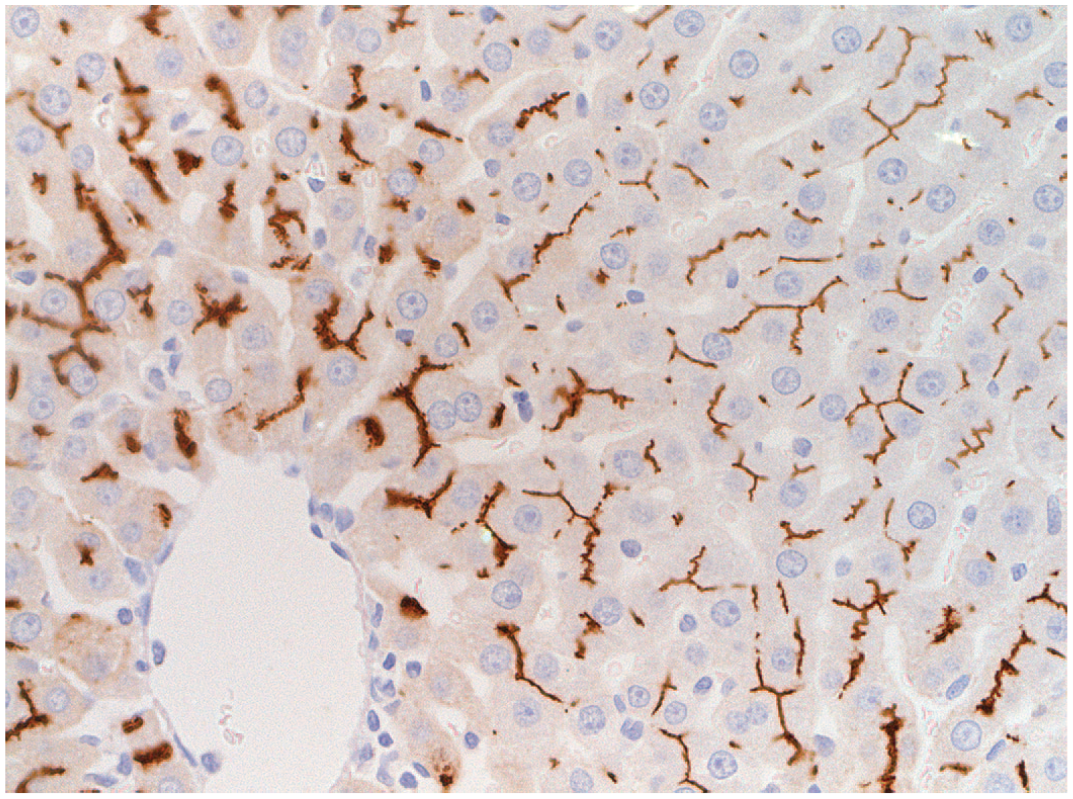
Rat liver. Imunohistochemical stain of the transporter molecule MRP2 from a rat.
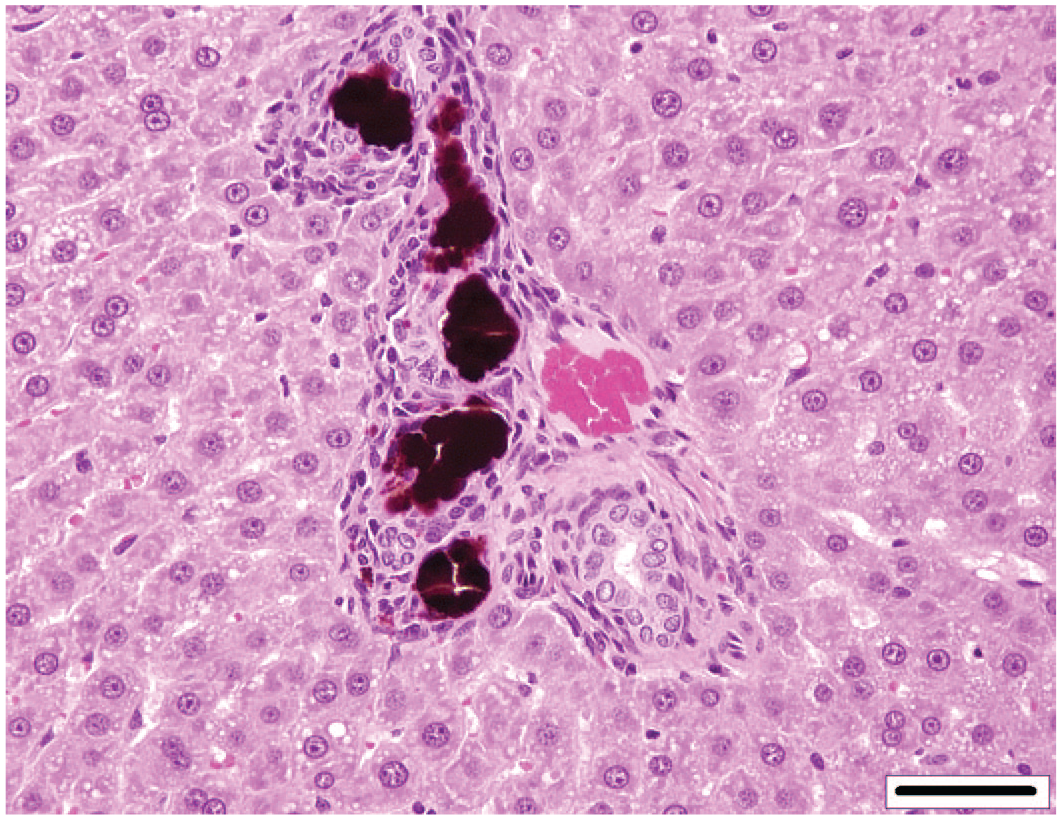
Rat liver. Porphryin deposits within the biliary tree. Reprinted with permission from the American Chemistry Council. 37
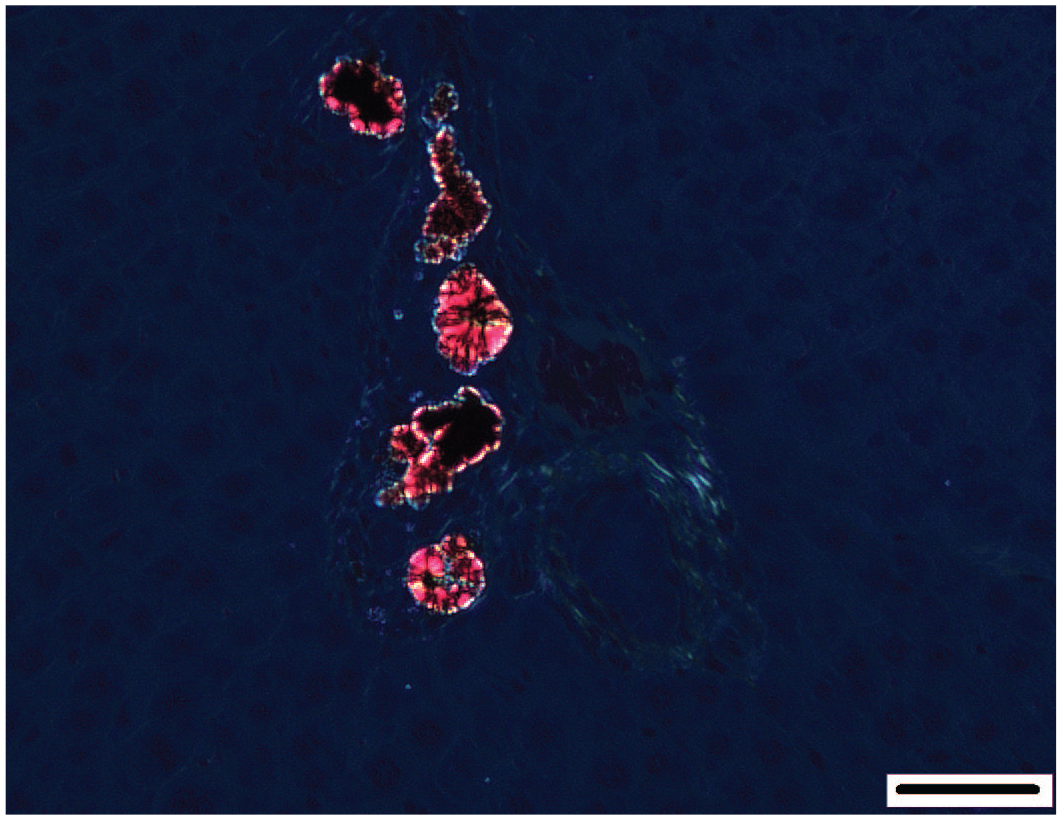
Rat liver. Birefringence of porphyrin under polarized light. Reprinted with permission from the American Chemistry Council. 37
Mouse liver. Hyalinosis of hyperplastic bile ducts with crystal formation and peribiliary inflammatory infiltrate. Reprinted with permission from Sage Publications. 32
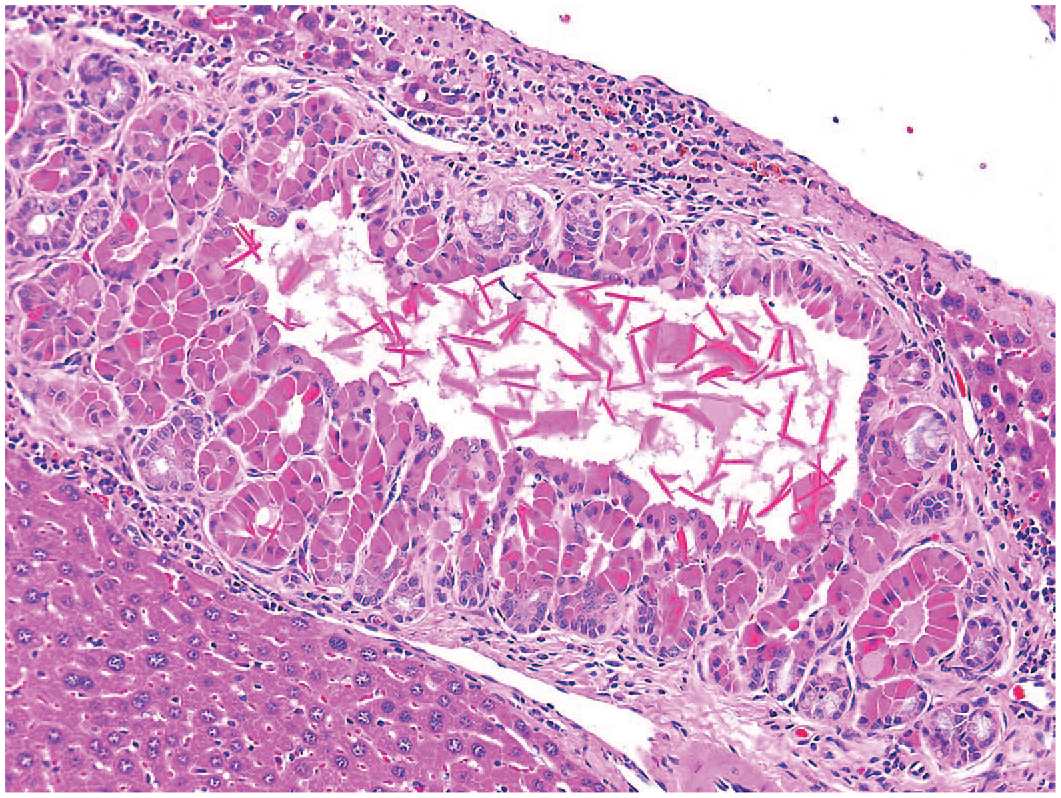
Hyalinosis of hyperplastic bile ducts with crystal formation and peribiliary inflammatory infiltrate. Reprinted with permission from Sage Publications. 32
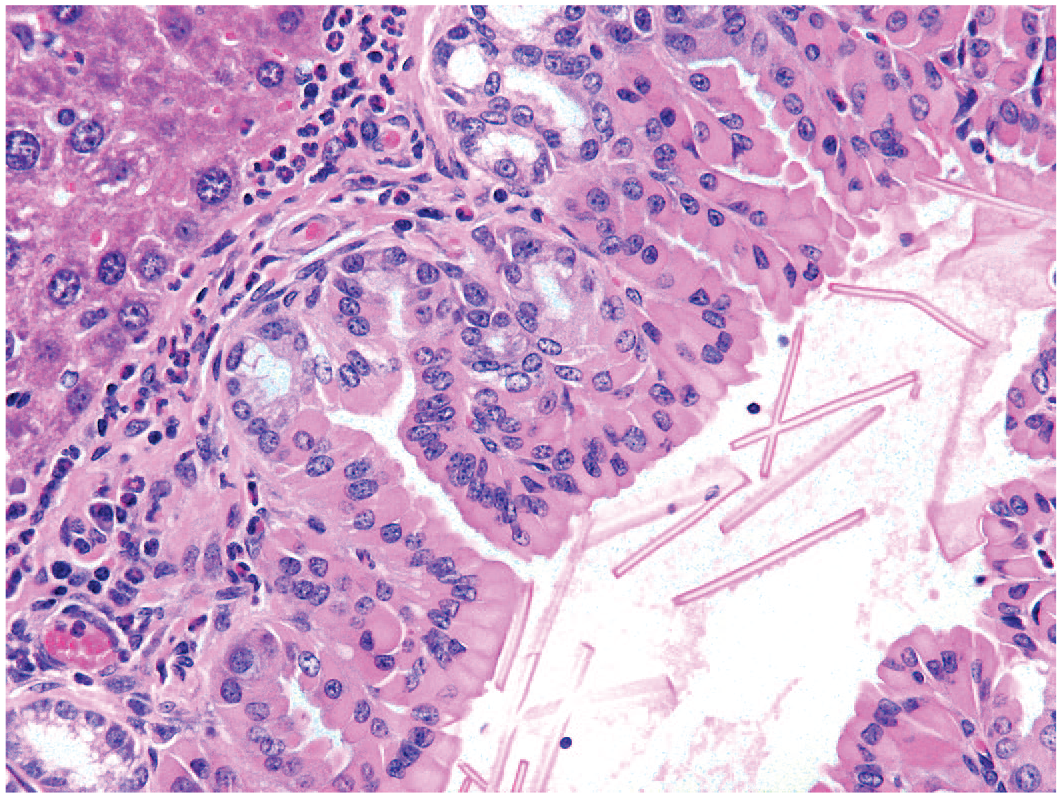
Mouse liver. Higher magnification of hyalinosis of hyperplastic bile ducts with elongated eosinophilic crystal formation and peribiliary inflammatory infiltrate. Reprinted with permission from Sage Publications. 32
The process of bile formation at the level of the canaliculus is complex. Canaliculi are composed of modified hepatocyte membranes from two apposing hepatocytes with tight junctions and gap junctions sealing the outer margins. Pericanalicular actin-myosin filaments surround the canaliculi and can contract to assist in the movement of bile toward the portal tract bile ducts. These membranes are studded with a variety of molecular transporters, such as the bile salt export pump, which modify bile by adding various constituents from the hepatocyte cytoplasm along with a transcytotic vesicular pathway15,34 (Figure 5). Various substances can disrupt the molecular transporters. Examples include endotoxin, leading to the cholestasis of sepsis (infection), gastrointestinal metabolites, and various compounds such as antibiotics, estrogens, and others. 13 Genetic predisposition to form abnormal bile and consequential obstruction are factors as well. 16 Hepatocytes, in turn, also have a variety of transporters to move bile constituents from the plasma into the hepatocyte cytoplasm. Functional disturbance of these transporters can also lead to canalicular cholestasis.
Canalicular cholestasis leads to significant ultrastructural modifications. The connections that seal off the canaliculi are disrupted and allow movement of bile constituents, and bile plugs, into the vascular system at the level of the sinusoids. The pericanalicular actin-myosin bundles that normally propel bile can be disturbed, which limits the movement of bile leading to stasis. The transcytotic vesicular pathway can also be disrupted as well.
Canalicular cholestasis can be confused with stain artifact or acid hematin deposits, but these are rarely localized in the canalicular region only. Other substances or pigments such as lipofuscin, typically an age-related by-product of oxidation of various substances, can accumulate in a pericanalicular locale. Various xenobiotics and oxidant agents, such as peroxisome proliferator- activated receptor alpha (PPAR alpha) agonists, can increase the formation of lipofuscin. 20 Basophilic mineral deposits are also possible.27,31 Disturbances of bile flow at any level of the biliary tree can lead to canalicular cholestasis. Inflammation, necrosis, or obstruction of the duct system can cause canalicular cholestasis, particularly during the acute phase of injury, as well.
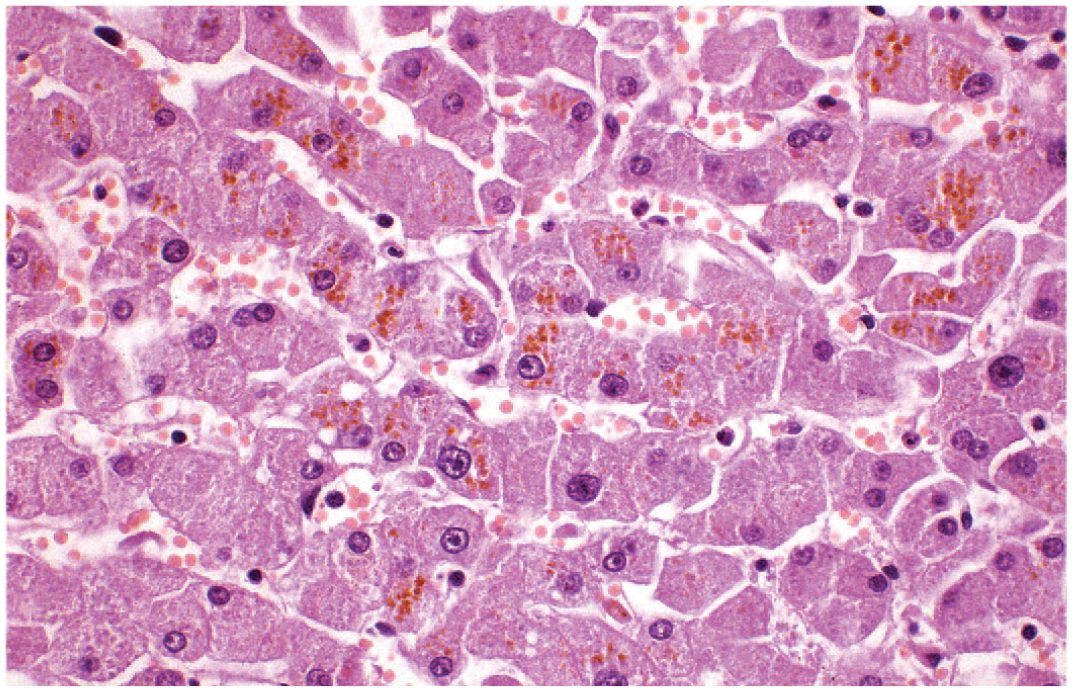
Porphyrin pigment, a precursor in the synthesis of heme protein, can be observed following disruption of the normal heme metabolism causing a buildup of porphyrin metabolites such as uroporphyrinogen and protoporphyrin, resulting in porphyrin deposition in the liver of rodents. 30 These porphyrins are identified as dark brown pigment in canaliculi, or bile ducts, and, on occasion, hepatocytes. The birefringence of porphyrin appears to be associated with bilamellar components within the pigment, and a typical Maltese Cross is observed under polarized light, and they are activated and show a red fluorescence, under ultraviolet light 31 (Figures 6 and 7). Griseofulvin is one compound that leads to disruption of the mitochondrial enzyme ferrochelatase in mice and dogs, although other substances can also disrupt heme metabolism.14,31,36 Accumulation of porphyrins in mouse liver may lead to cell damage and necrotic and inflammatory processes. 14 Protoporphyrin pigment in the liver of rats and mice is mainly found in the bile ducts and leads to bile duct proliferation and portal inflammation, but it can also be found within hepatocytes, Kupffer cells, and portal macrophages. 11 The presence of this pigment can also lead to bile duct proliferation, periportal inflammation, liver fibrosis, and cirrhosis.8,11 Porphyrin may also occur mixed with other material within the canaliculi. Other crystalline substances can be found within canaliculi or bile ducts. One such substance is chitinase-like proteins, Chi3l3, which forms birefringent eosinophilic rhomboid or needle-like crystals within canaliculi, bile ducts, and the gallbladder of some mice 35 (Figures 8-10). Cholesterol crystals may occur in association with hyperlipidemia. 8 Crystal formation may develop in the presence of or be the cause of inflammatory and/or proliferative bile duct changes and fibrosis in mice. Spontaneous occurrence is possible. 17
Cholangitis
Cholangitis refers to inflammation of bile ducts. Inflammatory cells should be present between biliary epithelial cells or within ducts to use this term, and it should not be confused with inflammatory cells simply within the portal tract connective tissue. Infiltrates may develop as “clean up” following biliary epithelial necrosis, and this should be differentiated from active inflammation of the structures. Choledochitis refers to inflammation of the intrahepatic ducts, and cholecystitis refers to the gallbladder specifically. Inflammation can vary in degree and type but does not differ from typical inflammatory patterns.
Infiltration, Peribiliary
Inflammatory infiltrates, typically mononuclear, in the connective tissue that surrounds the bile ducts, do not enter the ducts or between biliary epithelial cells. A few to many portal tracts may be affected. Fibrosis may be present along with proliferation of bile ducts. This is often an aging change in rodents.
Biliary Epithelial Necrosis
Necrosis of the biliary epithelium can occur when injurious primary substances or metabolites are present within the lumen of the bile ducts. Several “tool” compounds such as methylenedianiline and alpha-napthyl isothiocyanate (ANIT) are recognized for their ability to cause acute lytic necrosis of biliary epithelium 18 (Figure 11). There is functional heterogeneity of the biliary epithelium leading to modification of bile at distinct levels of the biliary tree. 7 Because of this, injury may also occur at distinct levels of the biliary tree. ANIT is significantly less injurious to hepatocytes and canaliculi than to the biliary epithelium of interlobular bile ducts. It has been suggested that the change in pH of the bile in the ducts disrupts the conjugation of the ANIT-metabolite leading to damage to the lining biliary epithelial cells. 3
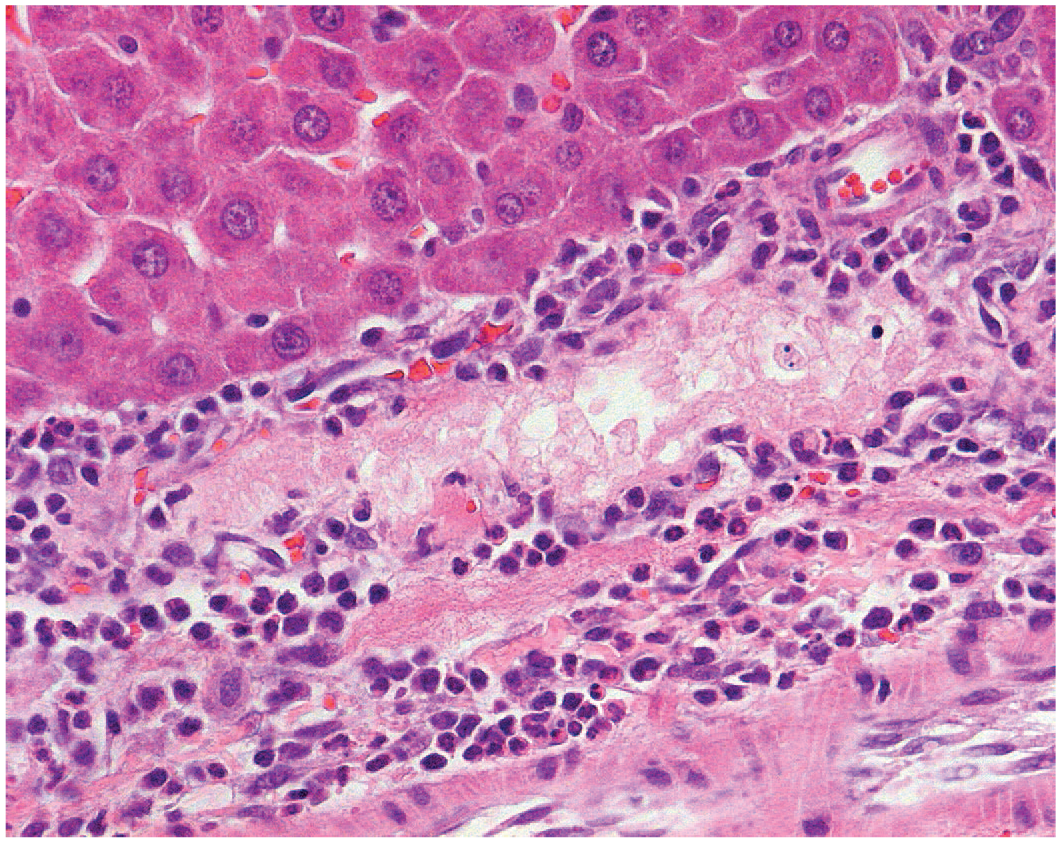
Rat liver. Acute necrosis of the biliary epithelium 24 hours following alpha-naphthyl isothiocyanate–treatment in a rat. Reprinted with permission from Sage Publications. 38
The bile ducts have an arterial rete that surrounds the interlobular and larger bile ducts. Disruption of this blood flow can lead to acute necrosis of the biliary epithelium. This change is recognized in human liver transplant patients and has the potential to occur in rodents with xenobiotic-induced vascular injury.
Proliferative Issues in Biliary Epithelium
Ductular reaction is defined as a reaction of ductular phenotype, possibly but not necessarily of ductular origin, in acute and chronic liver disease. Ductular reaction may arise from (1) proliferation of pre-existing cholangiocytes; (2) progenitor cells (local and/or circulating cells probably bone marrow-derived); (3) rarely, biliary metaplasia of hepatocytes. 23 One form of ductular reaction observed histologically in liver specimens is termed bile duct proliferation or hyperplasia and is commonly identified in biliary injury or cholestasis.
Ductular reaction may play a significant role in hepatic regeneration and is associated with liver fibrosis and damage. Signaling pathways could differ depending on the specific liver injury or animal models used in the study. However, further studies are needed to elucidate detailed mechanisms and the functional roles in liver diseases.
Roskams et al 23 published a review of nomenclature of the human biliary tree, and these authors have recommended the discontinuation of the term “oval cell” in human liver, recognizing that their proposed terminology was the equivalent of the term “oval cell” in rodents (Table 1). Adoption of the proposed human terminology would enhance the understanding of a universal process across species and should be embraced. The use of the terms “typical” and “atypical” duct proliferation may retain some usefulness for regulatory purposes given the association with the potential for the development of neoplasia in bioassays.
Suggested terms for descriptions of human tissues, with definitions and terms to be discontinued.*

Modified from Roskams et al. Hepatology 2004,39:1739.
Bile Duct Hyperplasia
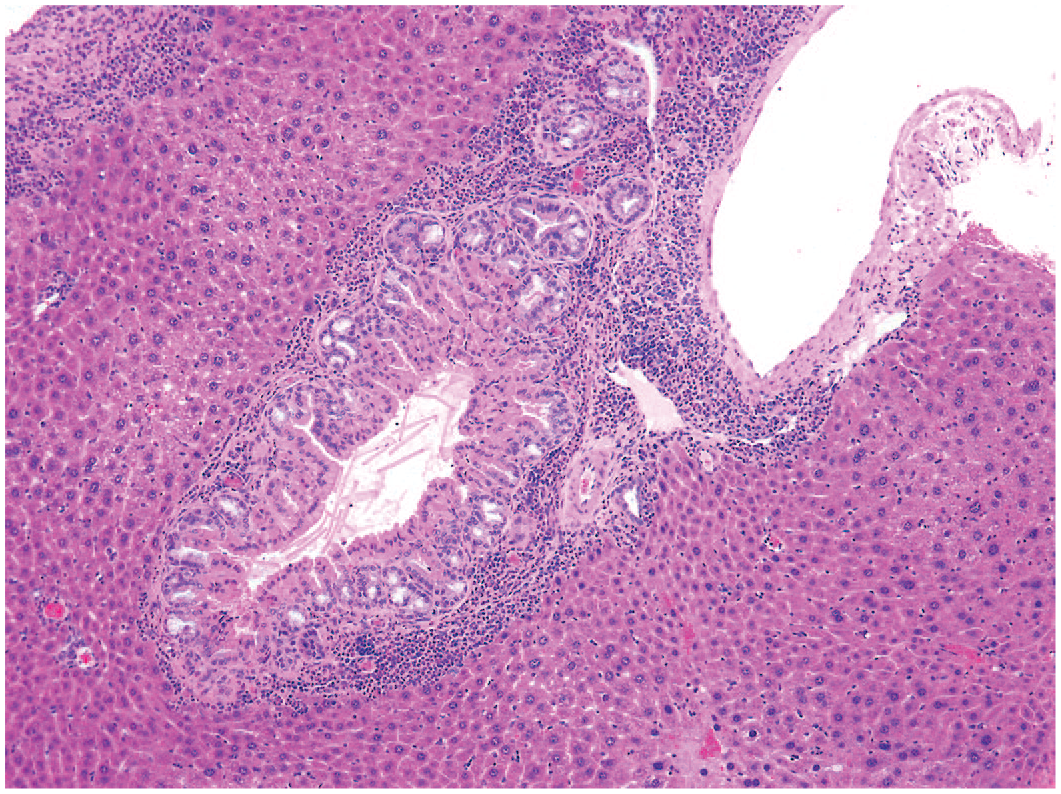
Bile duct hyperplasia, a form of ductular reaction, can arise from multiple causes (Figures 12 and 13). “Typical” bile duct hyperplasia may occur as a spontaneous age-related finding in older rats and, to a lesser extent, in mice. 9 This form is characterized by well-differentiated biliary epithelium and mild periductal fibrosis that is confined to the portal tract area. Proliferated ducts are typically smaller than interlobular bile ducts and may only involve a proportion of portal tracts (Figure 12). Dilation of a subset of these bile ducts may occur, and in mice, mucous metaplasia can be found. There is little significance attached to this finding. Other causes of bile duct hyperplasia include bile duct obstruction, damage from injurious compounds and a response to hepatic inflammation.
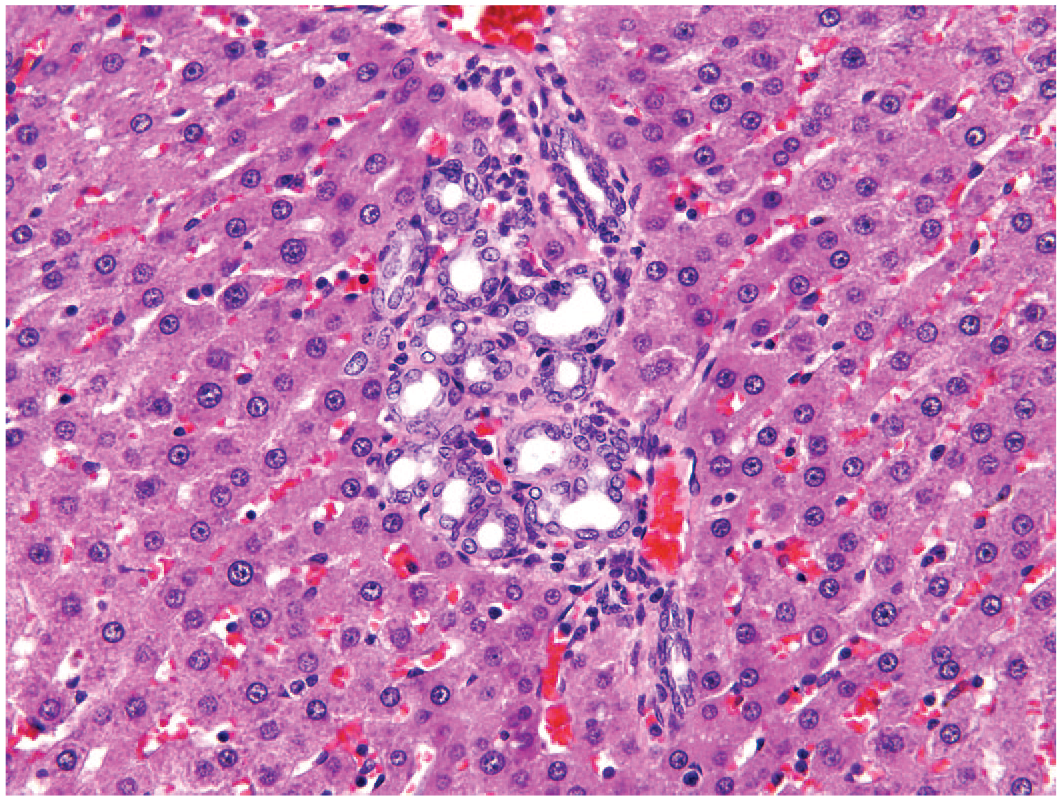
Rat liver. Spontaneous bile duct hyperplasia with minimal fibrosis and periductular inflammatory cell infiltration. Reprinted with permission from Sage Publications. 39
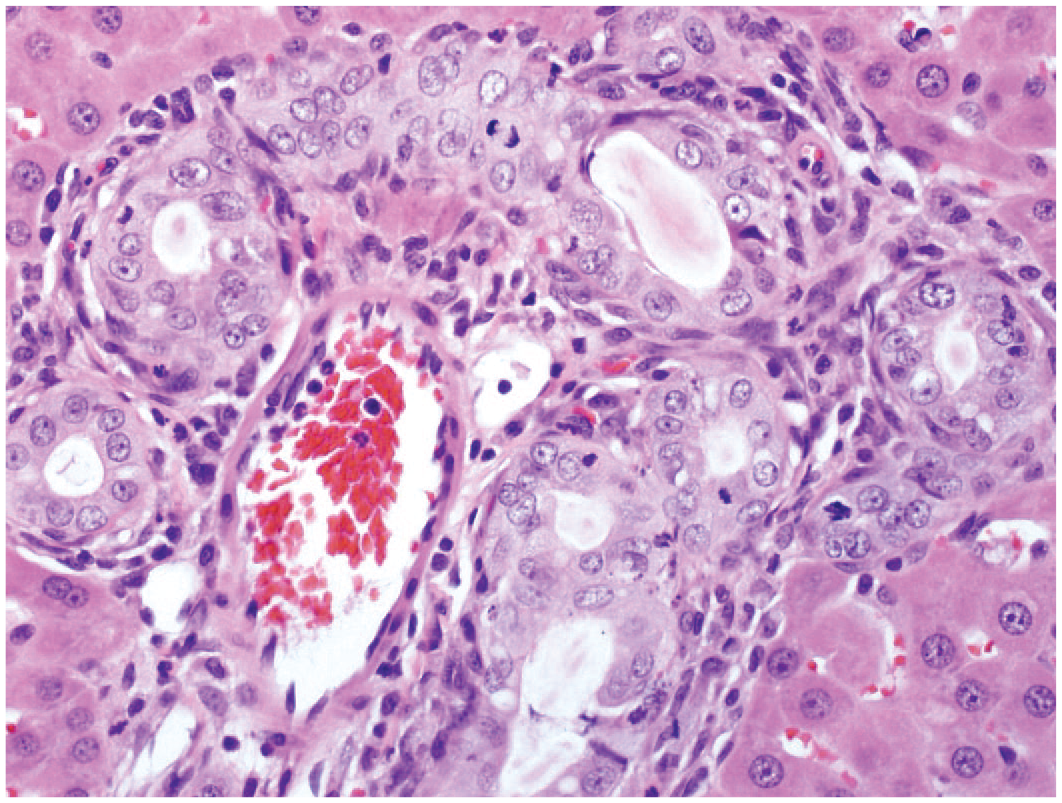
Rat liver. “Atypical” biliary hyperplasia with increased profiles of bile ducts, hypertrophic biliary epithelium, atypical, mitotic figures, and inflammatory cell infiltrate. Reprinted with permission from Sage Publications. 39
Drug-induced liver injury in rodent toxicity studies can affect the biliary tree solely or in combination with hepatocellular injury. The interpretation of the significance of rat bile duct hyperplasia for humans is not straightforward because bile duct hyperplasia is an uncommon finding in humans in response to drug exposure. A review of the relationship between rodent bile duct hyperplasia in Sprague-Dawley rats and cancer risk suggests three categories of bile duct hyperplasia. 9 These include minimal “typical”-appearing bile duct hyperplasia that is limited to the portal triads, which can be considered nonadverse in the rat and is of little to no concern to humans. Mild-to-moderate “typical” hyperplasia may pose a health risk to humans, but the interpretation is not clear. The concurrent presence of hepatobiliary injury or functional compromise raises concern. In laboratory animals, “atypical” biliary hyperplasia can be characterized by hypertrophic or pleomorphic lining biliary epithelium, increased mitotic figures with proliferation of ducts that may occur outside the portal tract (Figure 13). Atypical or dysplastic epithelial changes, oval-cell proliferation, and/or significant extension beyond the portal tracts are considered more ominous and may be considered adverse in the rat, as these changes have been observed in response to exposure to known carcinogens.
Ductular Reaction Oval-Cell Proliferation
One form of ductular reaction has been termed oval-cell proliferation or bile ductule cell hyperplasia. This form of ductular reaction involves bipotential progenitor cells of the liver (Figures 14-15). 24 These bipotential progenitor cells are found within the periportal space associated with canals of Herring and have the potential to develop into mature hepatocytes or biliary epithelium. This form of ductular reaction is initially evident histologically at the margins of the portal tract. Ductular reaction is characterized by a single or double row of oval-to-round basophilic cells along sinusoids in linear arrays. The cells may form thin cords or small ductules with or without evident lumens. Bipotential progenitor cells are usually uniform in size and shape and may be fusiform. The cytoplasm is basophilic and scant, and nuclei are round to oval. Immunohistochemical staining or other techniques reveal both hepatocellular and biliary markers. In humans and dogs, cytokeratin 19 and 7 may be positive, and the cells are also positive for hepatocellular markers, such as α-fetoprotein and breast cancer resistance protein (BCRP)/ATP-binding cassette transporter G2 (ABCG2). Ductular reaction is a very rare spontaneous lesion in rats and dogs 12 but can be seen in model systems of chronic degenerative/regenerative liver disease such as that produced in rats by chronic, low-dose carbon tetrachloride 28 or 2-acetylaminoflouride 28 (Figures 14 and 15). In mice ductular reaction/oval-cell hyperplasia can be induced by chronic infection and inflammation caused by Helicobacter hepaticus and Helicobacter bilis. 6 In conditions of significant hepatocellular injury or biliary injury induced by various xenobiotics, including carcinogens or dietary manipulations, often in a background environment that inhibits proliferation of mature cells, these bipotential cells proliferate. There is often a close relation to the portal tract, although more scattered ductular reaction forming groups of proliferating cells can be seen diffusely throughout the liver following xenobiotic-induced hepatic injury. 4 Ductular reaction/oval-cell hyperplasia can be associated with a high incidence of hepatocellular neoplasms and may play a significant role in hepatocarcinogenesis. Some studies support the view that bipotential progenitor cells forming a ductular reaction/oval cells may participate in the lineage of hepatocellular and cholangiocellular carcinomas. 28
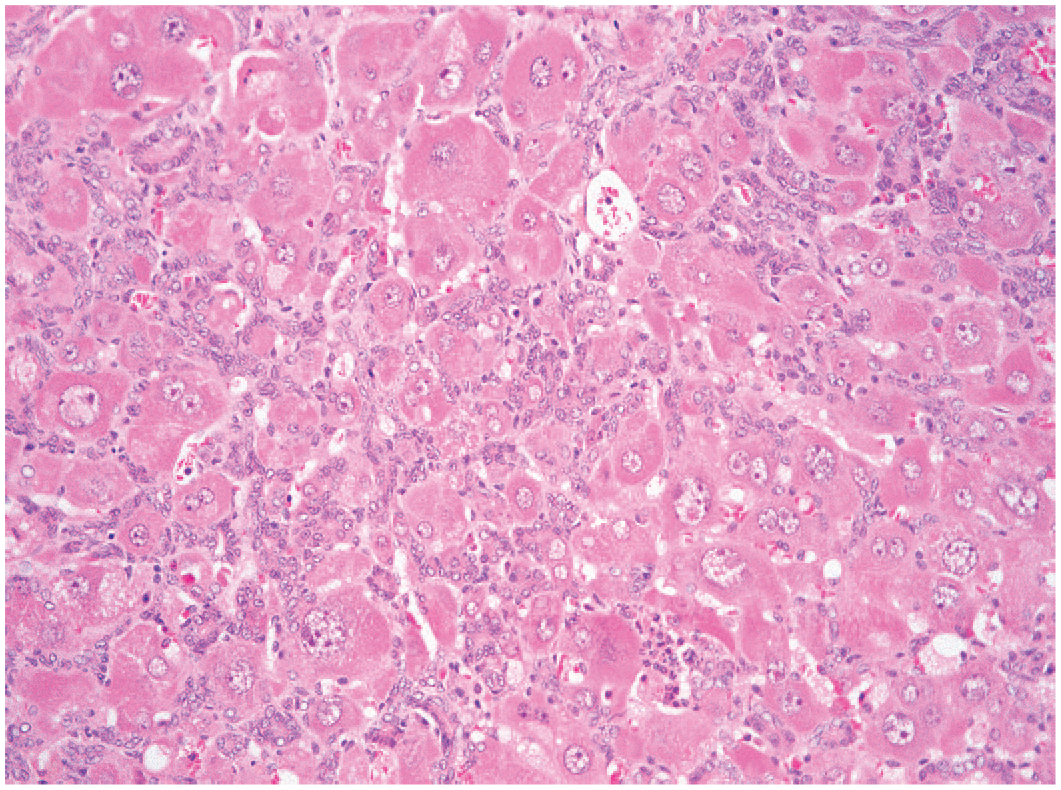
Rat liver. Ductular reaction of bipotential progenitor cells (oval-cell hyperplasia) following chronic carbon tetrachloride exposure.
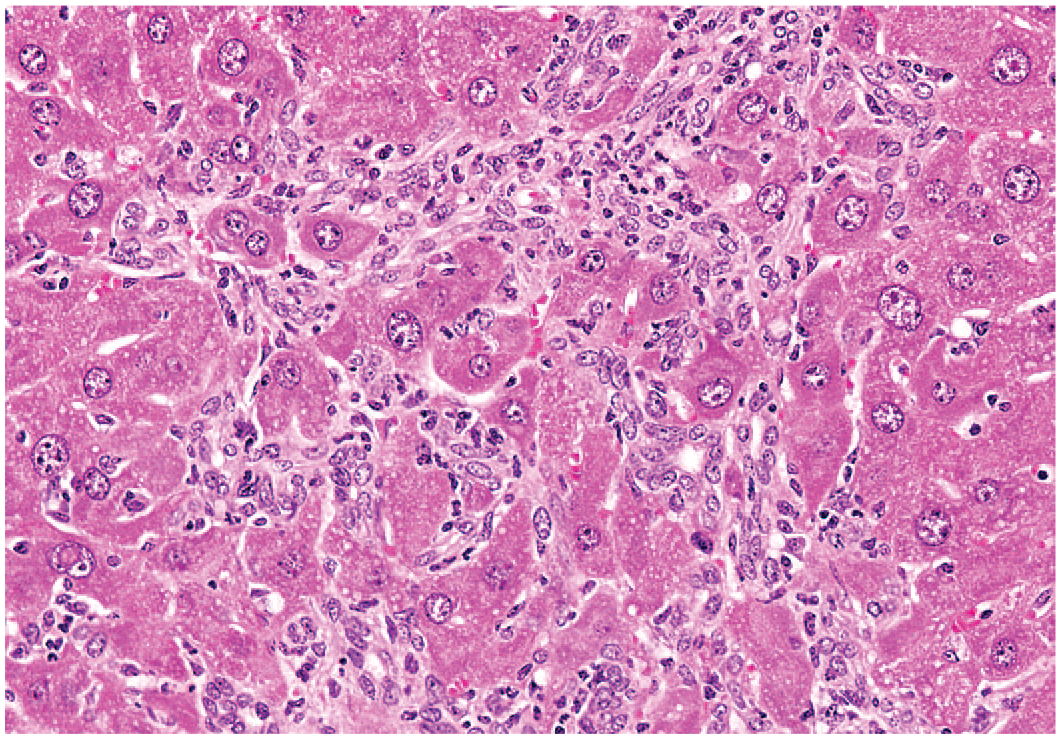
Mouse liver: Ductular reaction of bipotential progenitor cells (oval-cell hyperplasia). Reprinted with permission from Sage Publications. 32
Cholangiofibrosis
Cholangiofibrosis is a chronic inflammatory condition of the biliary tract that is characterized by partially lined cystic glands filled with varying amounts of mucus in a spectrum of progressive lesions starting as a ductal reaction (oval-cell proliferation) following pronounced hepatic parenchymal necrosis and hepatocyte hypertrophy (Figures 16 and 17). The masses start out as multiple, scattered fibrosis that expands in to bridging tracks of liver forming multinodular hepatic small to large masses (Figure 16) that expand and compress (with atrophy) surrounding tissues. The bile ducts become ulcerated, cystic, and partially lined by epithelium (Figure 17). Cysts were also filled with varying amounts of mucus (from minimal to abundant), proteinaceous fluid, cell debris, inflammatory cell infiltrates, and cuboidal to columnar basophilic cells well-differentiated to dysplastic ductal and intestinal epithelial cells including Goblet and Paneth cells, characteristic of intestinal metaplasia. Cholangioadenomas do not appear to be a part of this condition. Cholangiofibrosis developed following exposure to a small number of hepatotoxic xenobiotics including coumarin, dioxins, furans, and related chemicals.5,19 The lesions have occurred in treated rats, in studies including dioxins and polychlorinated biphenyls. Cholangiofibrosis is reported rarely in mice. 33
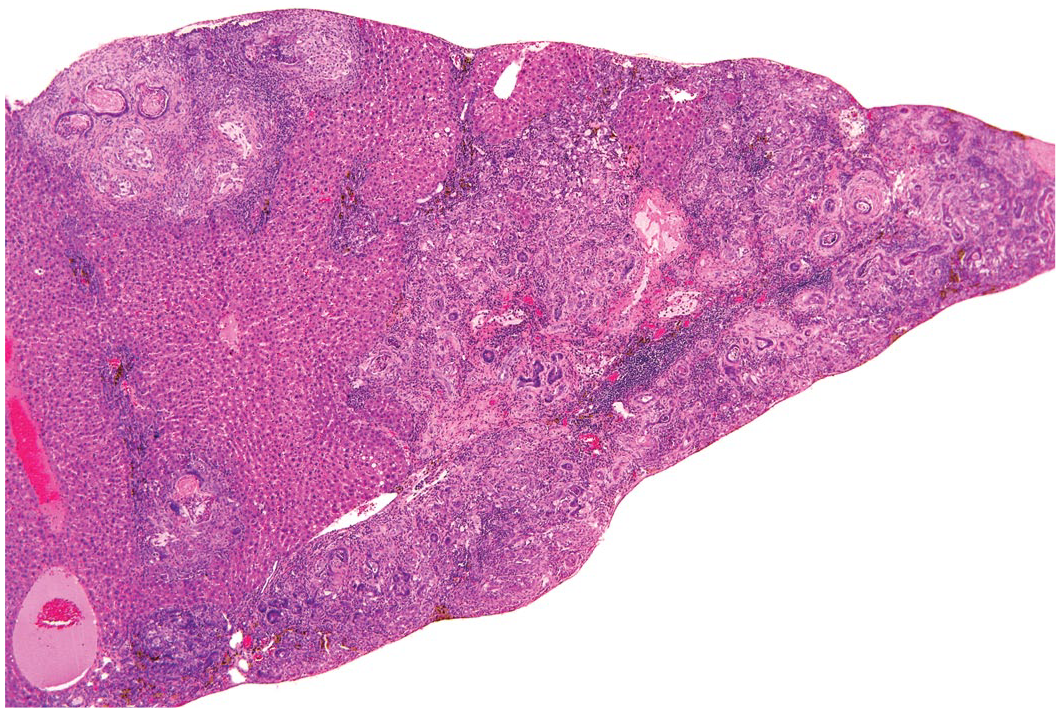
Rat liver: Cholangiofibrosis. Reprinted with permission from Sage Publications. 32
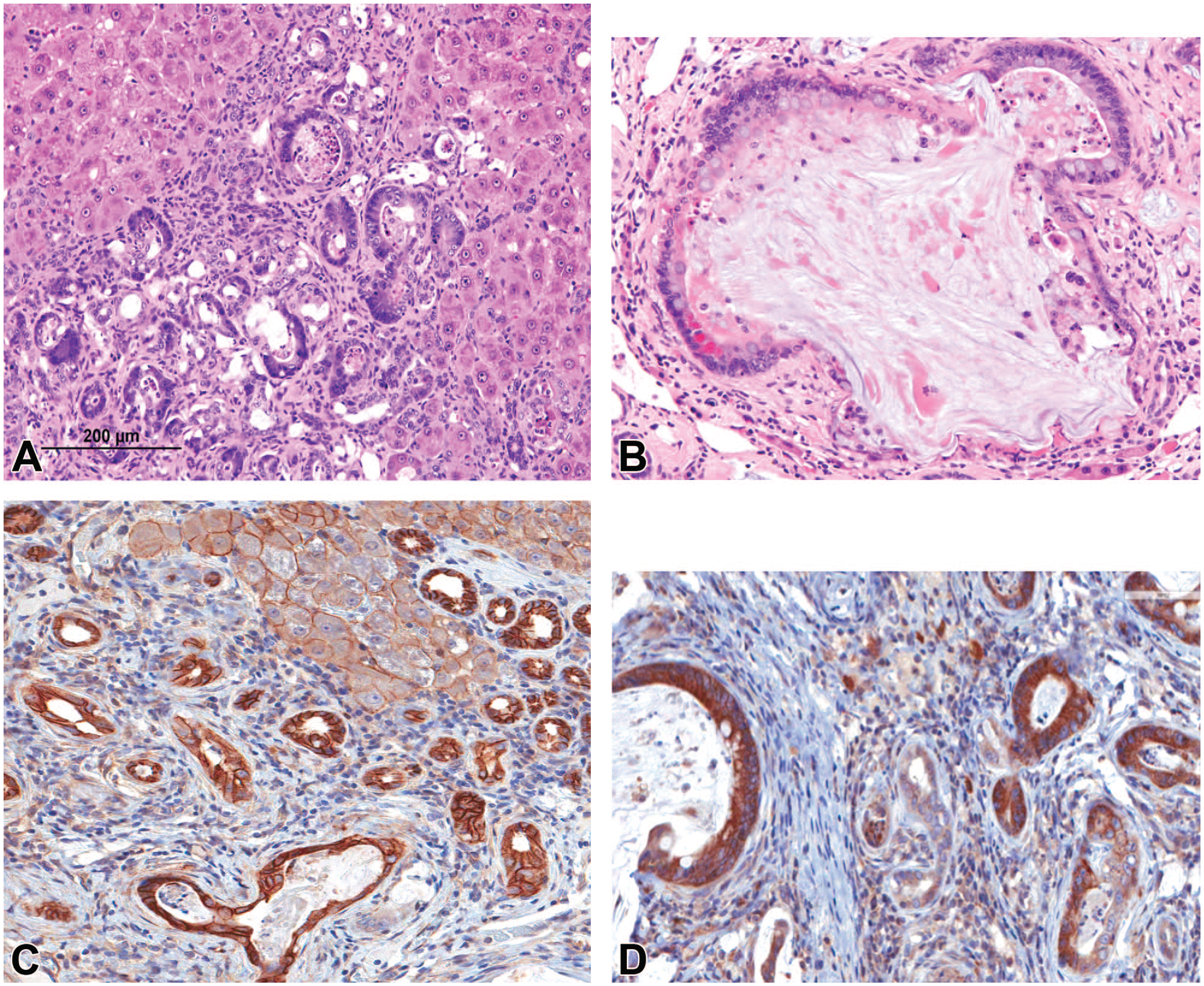
Rat liver: Features of cholangiofibrosis in the F344/N rat from a chronic furan study (Panels A-D. In panel A there is bridging fibrosis with invasive bile ducts epithelium demonstrating frequent intestinal metaplasia (comprised of Goblet cells, cuboidal to columnar epithelium, and Paneth cells) surrounded by hypertrophic and hyperplastic hepatocytes compressing adjacent fibrous tissue and parenchyma that is infiltrated by inflammatory cells. Panel A reprinted with permission from Sage Publications. 21 Non-neoplastic Lesion Atlas: Hepatobiliary (2023). Panel B shows a relatively large typical biliary cyst partially lined by intestinal metaplastic cells and filled with a hypocellular proteinaceous fluid with necrotic cell debris. Immunohistochemical results for beta-catenin (panel C) ERB-2 (NEU) (panel D) are indicative of a potential role of oncoproteins during the evolution and transformation of cholangiofibrosis to cholangiocarcinoma.
Degeneration of the component epithelial cells and interstitial fibrosis are hallmarks of the lesion. Remnant, crescent-shaped ducts may be present, and regions of fibrosis occur centrally in larger lesions. Foci of cholangiofibrosis vary from small, isolated forms to much larger lesions affecting large regions within the liver lobe, typically without disturbing the lobular margins, although in some instances, the lesions can protrude from the capsular outline. Fibrotic scars may be the residuum of these lesions. Cholangiofibrosis may resemble cholangiocarcinoma and may often be differentiated by the latter’s more prominent biliary epithelial changes (e.g., multiple layers or piling up of epithelial cells growth as a solid tumor and pleomorphism), as well as epithelial cell atypia and increased mitotic activity.
Controversy persists regarding the nature of these lesions with disagreement regarding the malignant potential of cholangiofibrosis. There is no doubt that hepatotoxic chemicals, such as furan, can produce cholangiocarcinomas, in addition to cholangiofibrosis, but the question that has challenged toxicopathologists for the last 50+ years regards whether or not cholangiofibrosis is a precursor lesion (preneoplastic) for the development of carcinoma, most especially cholangiocarcinoma, or whether the two lesions can develop independently. 2 While cholangiofibrosis, induced by certain genotoxic liver carcinogenic chemicals, has been reported to progress to cholangiocarcinoma with malignant features,1,29 unequivocal metastases have not been confirmed in most cases, and whether this phenomenon can be extrapolated to chemicals generally remains an open and undetermined question. The difficulty in unequivocally concluding that the cholangiofibrosis can progress to cholangiocarcinoma is complicated by the overlapping histological features of the two entities, together with the frequent inability to differentially diagnose the two. Undoubtedly some chemicals, such as furan, which induce cholangiofibrosis at early timepoints of dosing, show the presence of cholangiocarcinoma at later timepoints, while others such as coumarin fail to show the evolution of cholangiocarcinoma despite lengthy periods of exposure. Whether one develops into, or is a precursor for the other, could be identified if the presence of key mutations could be proved to be present in cholangiofibrosis prior to the development of cholangiocarcinoma.
A Stop-Exposure Evaluation, carried out as part of the NTP evaluation of the potential carcinogenicity of coumarin, showed that rats receiving 100 mg/kg coumarin in corn oil by gavage for 9 months showed liver lesions at this time, which subsequently regressed completely following a 15-month recovery period when the rats received corn oil only. While chemical-related hepatic lesions were seen at both the 9- and 15-month interim evaluations, the incidences and severities of these lesions, following the recovery period, were like controls. Thus, the hepatic lesions produced by 9 or 15 months of exposure were reversible. Coumarin was subsequently classified as a carcinogen in rats based on an increased incidence of kidney tumors and in male B6C3F1 mice based on an increase in alveolar/bronchiolar neoplasms and in female mice based on an increase in lung neoplasms and hepatocellular adenomas. 22
In contrast to the data with coumarin, the National Toxicology Program (NTP) study with the chemical intermediate, furan, in the F344 rat and B6C3F1 mouse showed the presence of cholangiofibrosis at 13 weeks in both species and a very high incidence of cholangiocarcinoma at 2 years in rats, with an increased incidence of hepatocellular neoplasms in mice at the same time period. 22 Cholangiofibrosis was not present in the liver of rats or mice at the 2-year time interval. Cholangiocarcinoma was also present, at an incidence of 100%, in male rats at the 9- and 15-month timepoints in a concurrent “stop-exposure” study with furan. A commentary added to the Discussion and Conclusions section in the NTP report stated that “An important result of the stop-exposure study is the finding that the benign-appearing lesions of the bile duct epithelium present after 13 weeks of furan exposure (including cholangiofibrosis) frequently progressed into cholangiocarcinoma.” While this may well be true, it is not possible to conclude, from the report data, that the cholangiofibrosis present in the rat livers at the 13-week timepoint progressed to cholangiocarcinomas. 10
An alternative perspective suggests that cholangiofibrosis should be considered preneoplastic because cholangiocarcinoma can frequently be seen arising from within cholangiofibrosis, and it resembles the morphology of cholangiofibrosis, but with more prominent biliary epithelial changes (e.g., multiple layers or piling up of epithelial cells, pleomorphism, and/or florid invasion typify cholangiocarcinomas). Occasional suspect metastasis has been reported.1,29 The morphological data support malignant invasive growth as the basis for the diagnosis of cholangiocarcinoma. Supportive immunohistochemical (IHC) evidence of altered cancer gene expression levels by IHC and transcriptomics profile evaluation further demonstrate alterations are acquired in a progression toward neoplastic growth (Malarkey-personal observation). Extensive local invasion into hepatic tissue, evidence of increased mitotic activity, and evidence of tumors arising from biliary epithelium and intestinal metaplasia (cuboidal, columnar, and Paneth cells) all are supportive of the progression of neoplastic cells.
The morphological data support malignant invasive growth as the basis for the diagnosis of cholangiocarcinoma. Chronic exposure to agents that cause cholangiofibrosis lead to an increased risk of developing cholangiocarcinoma. Supportive immunohistochemical evidence of altered cancer gene expression and transcriptomics profile evaluation further demonstrate alterations that are acquired in the progressing behaviors. Alterations in some common cancer genes found in human and rat liver cancers such as beta-catenin and erythroblastic oncogene B-2 (ERB-2) (NEU) are all altered in different cells of the biliary system (Figure 17).
Conclusion
In conclusion, the biliary tree is subject to congenital developmental issues that, on rare occasions, can be confused with treatment-related responses or that may make particular strains, or individuals, unsuitable for study. These abnormalities need to be recognized and assessed to prevent interpretative issues. There are a series of acquired lesions in the biliary tree affecting the smallest branches as well as the larger ducts. Different compounds/drugs/chemicals, and different circumstances, can affect the constellation of findings. Standardized terminology, considering contemporary pathobiology, will enable pathologists, and other scientists, to better understand the effects of novel compounds and assess potential safety concerns,
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
