Abstract
Whether biliary proliferative lesions in nonclinical species are predictive of potential hepatotoxicity in humans depends, at least in part, on the nature and severity of such changes in the nonclinical species. We reviewed published literature (clinical and nonclinical) and experimental data from rat toxicology studies conducted by GlaxoSmithKline and the National Institute of Environmental Health Sciences’ National Toxicology Program in an effort to better characterize the relative risk of hepatobiliary effects in humans. Available evidence supports the interpretation that minimal “typical” appearing bile duct hyperplasia limited to the portal triads may be considered non-adverse in the rat and is of little to no concern to humans. The toxicological relevance of mild to moderate “typical” hyperplasia is less certain, and may be considered adverse in the rat and potentially pose a risk for humans, particularly if accompanied by evidence of hepatobiliary injury or functional compromise. In addition, any proliferative lesion that includes atypical or dysplastic epithelial changes, oval cell proliferation, and/or significant extension beyond the portal tracts is considered more ominous and may be considered adverse in the rat.
Introduction
Drug-induced liver injury is common in rodent toxicity studies, and the biliary tree can be affected solely or in combination with hepatocellular injury. Interpretation of these effects is the first step in characterizing potential risks to humans. Determining to what extent a rodent finding translates—will occur and/or be problematic in the clinical setting—is often complicated and requires acquisition, integration, and interpretation of all relevant data about the specific compound and any other information/data that may impact the overall interpretation. Determination of translatability is often an evolving process in which increased confidence and/or accuracy is gained as the body of scientific data increases. At one extreme, with no information other than a histological finding in a rodent species, to protect human health one may assume the finding is translatable into humans. The other end of the spectrum is direct correlation of nonclinical and clinical data for the compound in question or use of historical data for class or chemical series mediated effects to demonstrate positive or negative associations to provide a more informed assessment of translatability. Currently, there is little published information indicating how proliferative lesions of the biliary tree in rats may translate to the clinical setting.
While risk/benefit must always be considered, minimal non-adverse test article–related findings in rodent toxicology studies are generally considered to pose little to no risk to humans, while adverse findings in animals are considered to more likely represent risk of harmful effects in humans. Adverse is herein defined as “a biochemical, morphological or physiological change (in response to a stimulus) that either singly or in combination adversely affects the performance of the whole organism or reduces the organism’s ability to respond to an additional environmental challenge” (Lewis et al. 2002). Assessing adversity in toxicology species requires integration and interpretation of all pertinent information/data and confidence in the assessment increases with increased information. Critical to assessment of adversity of biliary proliferation in the rat is whether the proliferation has the potential for functional alterations (e.g., cholestasis) and/or progression to more severe proliferative lesions (including neoplasia) and/or fibrosis. We reviewed published literature (clinical and nonclinical) as well as data from rat studies conducted by GlaxoSmithKline (GSK) and the National Institute of Environmental Health Sciences’ National Toxicology Program (NTP) in an effort to further characterize adversity and/or translatability of biliary proliferative lesions in rat toxicology studies. We also reviewed the significance of biliary proliferative lesions in humans. There are many important aspects of assessing potential human health risks (e.g., therapeutic margin, mechanism of action, drug indication, and target product profile) that are outside of the scope of this article.
Results/Discussion
Hepatobiliary Similarities between Species
Despite species differences in the hepatobiliary system, sufficient metabolic, functional, and morphologic similarities exist to suggest that chemically induced biliary proliferations in the rat may be relevant to humans. Unlike humans, rats do not have a gallbladder. Rats respond differently to some toxicants such as α-naphthylisothiocyanate (ANIT), a compound commonly used to study cholestasis and bile duct injury in laboratory animals and other chemicals (Zimmerman 1999b). Notwithstanding the differences, the fundamental processes of biliary epithelial injury and repair are similar for rodents and humans as supported by similar responses to a number of chemicals (Zimmerman 1999a,b) and pathologies such as bile duct ligation/obstruction which produces cholestasis, bile duct proliferation, and eventually fibrosis and cirrhosis in both species (Kountouras et al. 1984). Cholangiopathies, including drug-induced cholestasis, cause significant morbidity and mortality in humans (Bjornsson and Jonasson 2013), and are a major reason for transplantation (LeSage et al. 2001). Cholangiocarcinoma is a lethal disease, afflicting 3,000 individuals in the United States and many more globally (Chun-Nan et al. 2004).
Nomenclature
Since biliary proliferation in rats is potentially relevant to humans, it is important to know if there is any degree/severity that may be considered non-adverse in the rat and therefore predict little to no hepatotoxicity risk to humans. To address this requires clarification of the categories of biliary proliferation. The “International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) for Lesions in Rats and Mice” guidance document for the liver includes bile duct hyperplasia, oval cell hyperplasia, and cholangiofibrosis as diagnoses for nonneoplastic proliferative lesions of the biliary tree, each graded (1–4) for severity (Thoolen et al. 2010). Some have subcategorized bile duct hyperplasia as “typical” and “atypical” (LeSage et al. 2001; Sirica 1995), while others (Roskams et al. 2004) discourage use of these subcategories. “Typical” hyperplasia is described as an increase in the number of intrahepatic bile ducts with a well-defined lumen, confined to the portal area and thought to represent proliferation of preexisting ductules. “Atypical” hyperplasia is characterized by irregular proliferation extending into periportal and parenchymal regions, occasionally forming anastomosing cords with adjacent hepatocytes and is thought to arise from proliferation of preexisting ductules and expansion of the hepatic progenitor cells (LeSage et al. 2001; Priester et al. 2010). INHAND nomenclature encompasses the morphological spectrum of bile duct hyperplasia through the use of severity grades and/or qualifiers as appropriate. LeSage et al. defined oval cell proliferation, considered a potential precursor to cancer, as disorganized tubular structures with poorly defined lumens. These categories provide a morphological framework for assessing potential adversity; however, the degree to which they reflect discrete pathogeneses is uncertain, and may reflect a continuum that is dose and/or time dependent. Intuitively, the presence of atypical and/or dysplastic epithelium, significant extension of a proliferative response into the hepatic parenchyma, or proliferation of a more primitive cell type (oval cell) reflects a stronger direct stimulus and/or reparative response to significant hepatobiliary injury, and if present in the rat may be considered adverse. Cholangiofibrosis is a morphological entity that appears to only occur in the rat; its significance for humans is therefore uncertain.
Bile Duct Hyperplasia (Typical)
“Typical” hyperplasia is rarely observed in humans. It occurs during the acute phase of complete bile duct obstruction and in early phases of chronic cholestatic liver diseases in association with “atypical” proliferation (Alvaro et al. 2007). In one report in humans, bile duct hyperplasia similar to the “typical” form described in rats (Sonzogni et al. 2004) was considered likely nonprogressive in these patients. In this report, routine examination of liver biopsy specimens revealed minimal idiopathic ductular “proliferation” characterized by an increase in the number of biliary structures within and around portal tracts due to proliferation of preexisting bile ductules or to ductular metaplasia of periportal hepatocytes. Total bilirubin (TB), alkaline phosphatase (ALP), gamma-glutamyl transpeptidase (GGT), and alanine aminotransferase (ALT) values in serum were elevated in 25, 19, 81, and 69% of the patients, respectively, and had been for years. A comprehensive evaluation of these patients revealed no other cause for the increases in clinical pathology parameters. In contrast to “ductular reaction,” which is commonly observed in response to different forms of liver damage, the increased number of periportal and intralobular biliary structures in these patients’ livers was not accompanied by significant fibrosis or inflammation. With ductular reaction due to chronic obstructive cholangiopathies, ductules retain features of immaturity, such as positivity for neural cell adhesion molecule (NCAM), a marker for immature atypical ductules, and negativity for epithelial membrane antigen (EMA), a marker for mature, differentiated cholangiocytes. In primary sclerosing cholangitis (PSC), which may be a precursor to cholangiocarcinoma, there is strong NCAM expression, even at early stages. The idiopathic ductular proliferations in the human subjects in the Sonzogni study were EMA-positive and NCAM-negative suggesting that this isolated ductular hyperplasia may represent a regenerative response to a mild form of occult biliary damage, which occurred without significant inflammation. In contrast, ductular reaction may represent a reparative response to more severe forms of chronic liver injury. After 2 to 5 years follow-up, the subjects with idiopathic ductular hyperplasia remained clinically asymptomatic with persistently increased, but stable clinical pathology parameters. The histologic picture was considered benign, and with a long history of relatively stable changes, the assessment was that the condition is likely nonprogressive (Sonzogni et al. 2004).
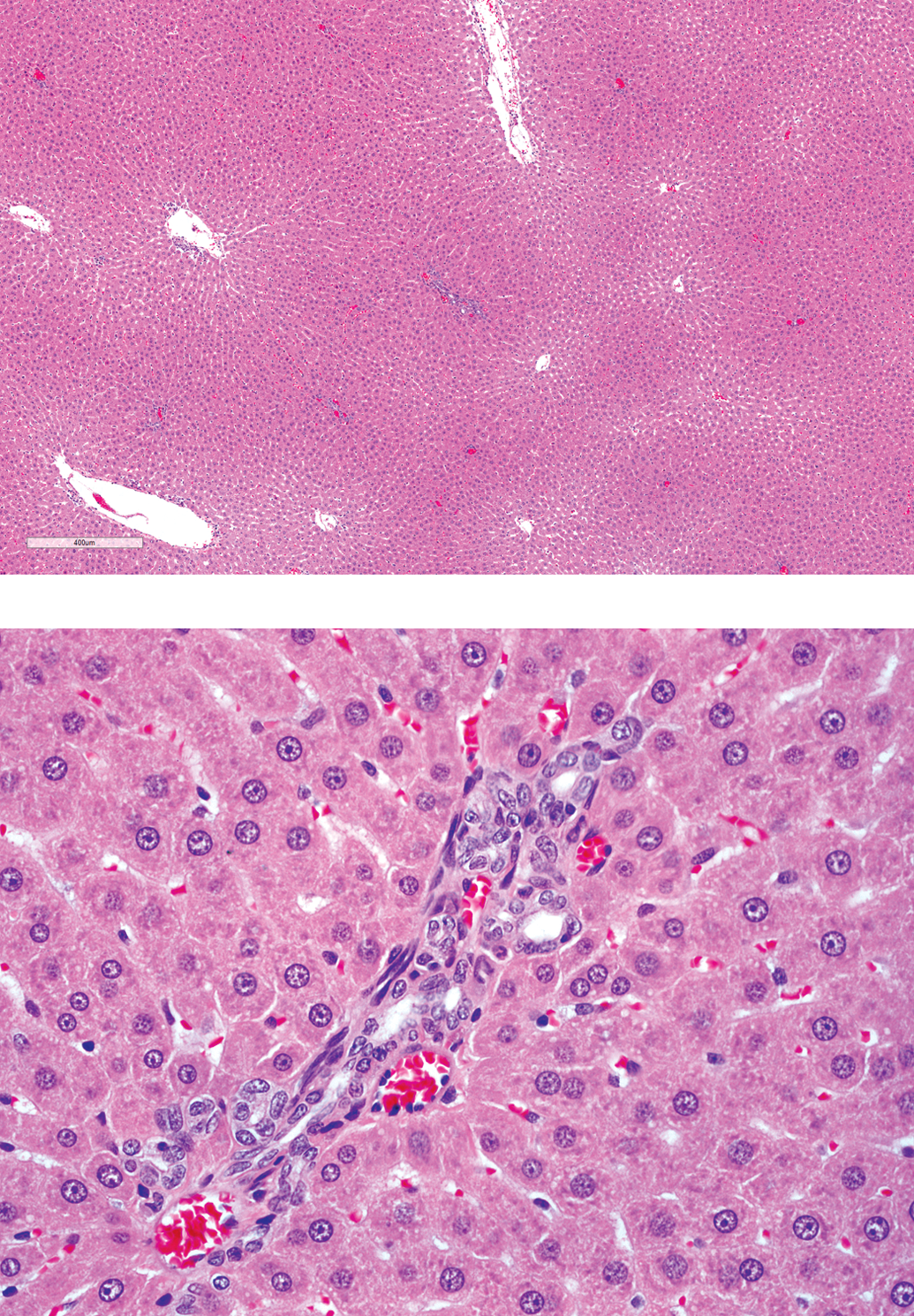
In laboratory animals, typical hyperplasia can be elicited by several experimental approaches including bile duct ligation and chronic feeding of ANIT (LeSage et al. 2001). Bile duct hyperplasia limited to and/or mildly expanding the portal region also occurs commonly as rats age (Stewart et al. 1980; Greenblatt 1982). In the F344 rat, this spontaneous change is often mild and frequently associated with mild fibrosis and sometimes inflammation (Figure 1). There is no evidence that it causes significant alteration of hepatic function, nor progresses to cancer (Eustis et al. 1990). In fact, while bile duct hyperplasia is considered a common background finding, the incidences of spontaneous cholangioma or cholangiocarcinoma in male and female F344 and Crl:CD (Sprague-Dawley [SD]) rats are rare. Of the 1,928 control male and 1,928 control female F344 rats, there were no cholangiocarcinomas and only one female with a cholangioma (Haseman et al. 1990). Similarly, in 2,146 control male and 2,344 control female SD (Crl:CD) rats, there were no cholangiocarcinomas and 8 females (0.34%) had bile duct adenoma (cholangioma; Giknis and Clifford 2004).
Control male F344 rat from a National Toxicology Program 2-year carcinogenicity study of butyl benzyl phthalate. Spontaneous bile duct hyperplasia (“duplication of bile ducts”) with minimal fibrosis and periductular inflammatory cell infiltrate (H&E; 20×). H&E = hematoxylin and eosin.
In addition to the lack of or low progression of spontaneous bile duct hyperplasia to neoplasia, chemical induction of neoplasms of the biliary tree is also uncommon in rodents. This is particularly true for chemically induced biliary neoplasms with morphologies resembling spontaneous biliary neoplasms. Slightly more common is chemical induction of an unusual spectrum of proliferative lesions generally classified as cholangiofibrosis, cholangiofibroma, and/or cholangiocarcinoma. Particularly in the older literature, a distinction between this spectrum and the biliary neoplasms that can occur in untreated rats is not always clear, and both spectrums may be observed in a single study. Most investigators consider ductular proliferation as reparative and not due to direct carcinogenic effects of the agent on the bile duct system. Initial bile duct proliferation is not considered to represent pre-neoplasia and may be reversible after withdrawal of the agent (Schauer and Kunze 1976). Based on rodent carcinogenicity studies conducted by the NTP, chemically induced hepatocellular neoplasms are relatively common while neoplasms of the biliary tree are very uncommon and the latter generally only occur in the presence of the former. When chemical associated biliary neoplasms do occur, they are often associated with significant liver toxicity (Bannasch et al. 1985). Therefore, serum or tissue markers of hepatocellular injury may serve as a sentinel for potential untoward biliary epithelial proliferations.
Within the NTP, over 575 compounds have been tested for carcinogenicity with a total of 46 positive (clear evidence or some evidence of carcinogenic activity) in the liver of male rats and 49 in female rats (NTP web site 2011). The vast majority of these studies were conducted in the F344 rat; however, 1/46 and 8/49 positive studies for males and females, respectively, were part of a group of 9 studies conducted in Harlan SD rats (8 of these conducted only in females). Relative to biliary neoplasms, in NTP studies of 1-amino-2,4-dibromoanthraquinone (ADBAQ; NTP Report 383 [NTP 1996]), methyleugenol (NTP report 491 [NTP 2000]), and p-cresidine (report 142 [National Cancer Institute 1979]) conducted in the F344 rat, very low numbers of neoplasms considered mixed (hepatocellular and biliary—hepatocholangiocarcinomas or hepatocholangiomas) were diagnosed in males and females (ADBAQ and methyleugenol) or males only (p-cresidine). In all three studies, benign and malignant hepatocellular neoplasms also occurred. The presence of both is also reported in other studies noted to cause cholangiocarcinoma in rats (methapyrilene, Ohshima et al. 1984; thioacetamide, Chun-Nan et al. 2004; and chloroform, Reuber 1979). However, in the studies of coumarin in which cholangiofibrosis and cholangiocarcinoma were observed, hepatocellular tumors did not occur (Evans et al. 1989). Neither tumor type was increased in the F344 rat in a 2-year study of coumarin conducted by the NTP (NTP report 422 [NTP 1993c]). Low numbers of cholangiocarcinomas were present in F344 male rats in the study of ortho-nitrotoluene (NTP report 504 [NTP 2002]) and in males and females in the study of polybrominated biphenyl mixture (firemaster FF-1; NTP report 244 [NTP 1983]).
Significant liver changes involving hepatocytes and biliary epithelium (toxic hepatopathy) occurred in an NTP study of furan in male and female F344N rats (NTP report 402 [NTP 1993b]; Maronpot et al. 1991); in 6 studies of dioxin and dioxin-like compounds (2,3,7,8-tetrachlorodibenzo-p-dioxin [TCDD]—NTP report 521 [NTP 2006b]; 3,3′4,4′5-pentachlorobiphenyl [PCB] 126—NTP report 520 [NTP 2006a]; PCB 118—NTP report 559 [NTP 2010b]; PCB 153—NTP report 529 [NTP 2006e]; 2,3,4,7, 8-pentachlorodibenzofuran [PeCDF]—NTP report 525 [NTP 2006c]; and 3,3′,4,4′-tetrachloroazobenzene [TCAB]—NTP report 558 [NTP 2010a]), and in 3 studies of mixtures of these compounds (TCDD + PCB 126 + PeCDF—NTP report 526 [NTP 2006d]; PCB 126 + PCB 118—NTP report 531 [NTP 2006g]; and PCB-126 + PCB 153—NTP report 530 [NTP 2006f]), all conducted only in female Harlan SD rats except TCAB conducted in males and females. All but PCB 153 were positive for cholangiocarcinomas, and unlike the above positive studies, generally had robust increases in frequency of cholangiocarcinomas. Most studies also had increases in hepatocellular adenomas with occasional hepatocellular carcinoma, hepatocholangioma, and/or cholangioma. In these studies, cholangiocarcinomas were clearly associated with the spectrum involving cholangiofibrosis and there was oval cell hyperplasia as well.
In nonclinical toxicity studies, there are instances whereby test article–induced bile duct hyperplasia is limited to the portal/periportal region with “typical” appearing epithelium. Minimal to mild bile duct hyperplasia and chronic active inflammation (portal) were observed in male and female rats given 1- or 5-mg/kg/day GW677954, a peroxisome proliferator-activated receptor (PPAR) α, γ, and δ ligand (GSK proprietary data) for 3 months and bile duct hyperplasia in rats (primarily males) given 0.5- or 2-mg/kg/day GW677954 for 6 months. Figures 2 and 3 represent the most advanced lesion that was identified in these studies, but are not representative of the appearance of most portal tracts or the livers as a whole as a significant portion of the liver parenchyma, including portal tracts, was normal in affected animals. Figures 4 and 5 are representative of the overall change. Although the biliary changes did not reverse after 4 weeks of recovery in the 3-month study, there was evidence of complete reversal after an 8-week recovery period in the 6-month study. Modest increases (up to 2.4× and 1.69× control means in the 3- and 6-month studies, respectively) in serum ALP were accompanied by slight decreases in other indicators of cholestasis including TB, total bile acids (TBA), and 5′-nucleotidase in the 6-month study with GGT being similar to controls. Furthermore, bile duct hyperplasia did not progress between 3 and 6 months, as the incidence and severity were similar between the two studies in the face of overlapping systemic exposures. Additional evidence of reversibility of biliary hyperplasia was observed in another GSK study in which GW695634G (GSK proprietary data) was administered orally for 6 months with minimal to moderate biliary hyperplasia associated with granulomatous inflammation and gold/brown crystals (determined to be crystallized compound). The changes associated with the biliary tract persisted at the 4- and 13-week recovery periods; however, they were reduced in severity at 13 weeks. Despite this evidence of reversibility, because of the severity and associated granulomatous inflammation, biliary epithelial hyperplasia was considered adverse in that study (GW695634G). Because of the lack of progression with continued treatment and the absence of cholestasis, the minimal to mild “typical” bile duct hyperplasia was considered non-adverse in the 6-month study of GW677954. Although not a strong consideration for determining a lesion to be non-adverse, the bile duct hyperplasia in this study was also reversible.
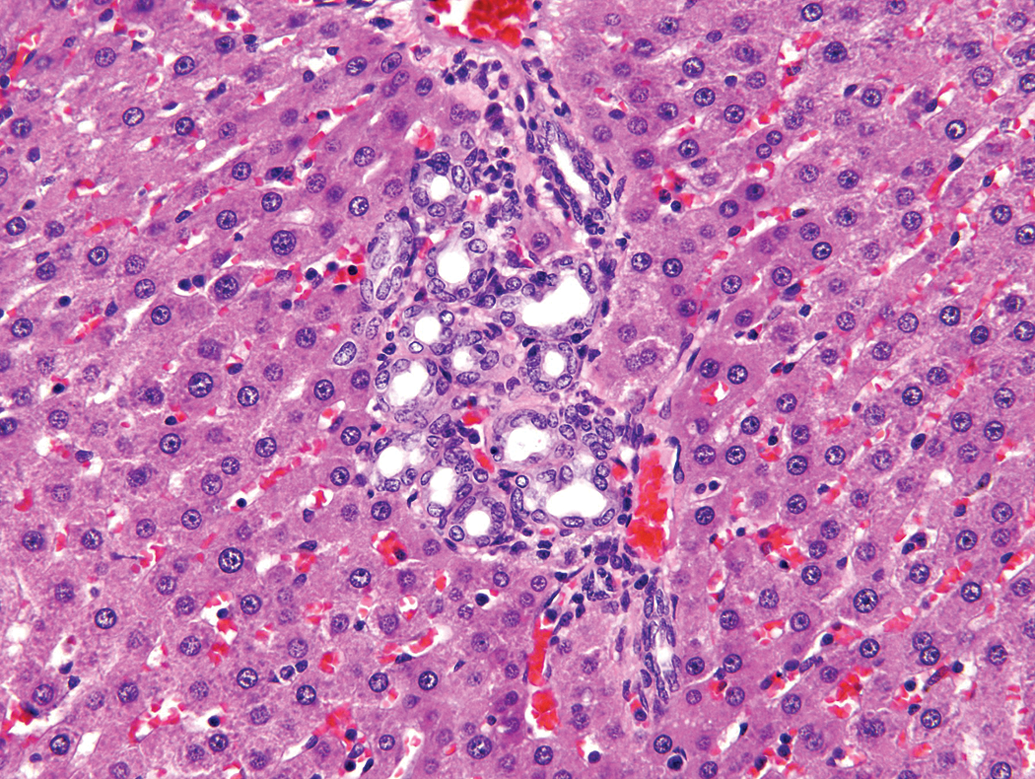
Male SD rat given 0.5-mg/kg/day GW677954 for 6 months. Note: The “duplication of bile ducts” and similarity to Figure 1 (20×).
Male SD rat given 2-mg/kg/day GW677954 for 6 months. This lesion has more associated fibrous connective tissue and inflammatory cell infitrate (H&E; 20×). H&E = hematoxylin and eosin.
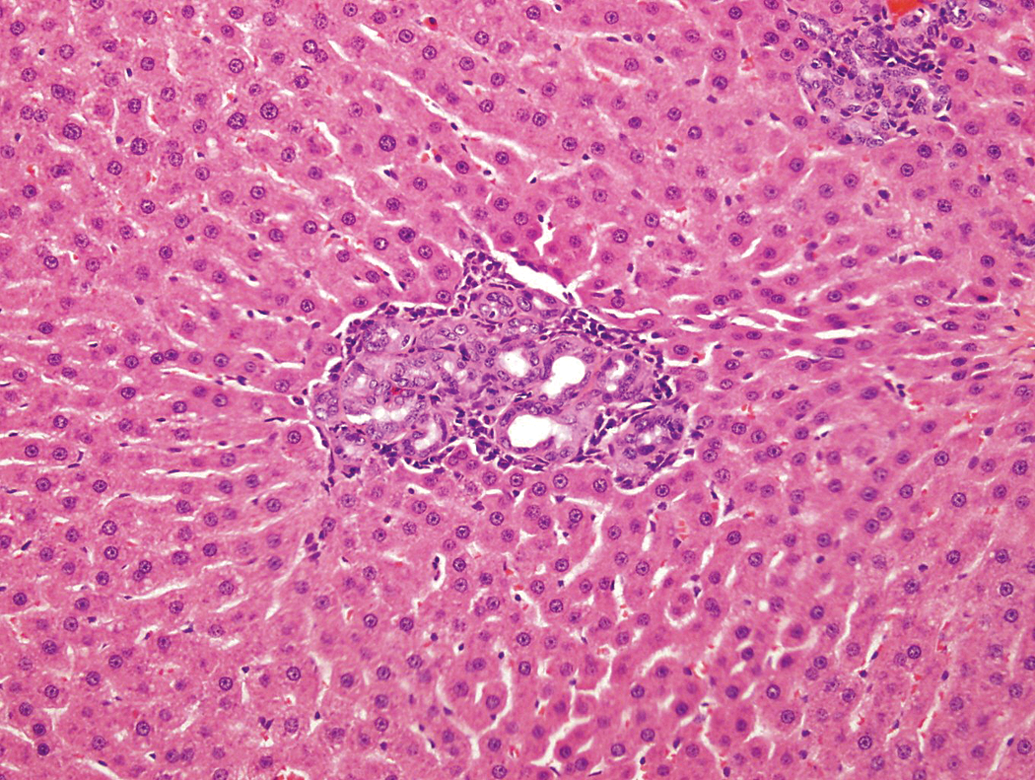
Male SD rat given 2-mg/kg/day GW677954 for 6 months. This minimal change or no change at all in many portal triads is typical for the study (H&E; 4× and 20×). H&E = hematoxylin and eosin.
Two-year carcinogenicity studies of GW677954 were conducted in SD rats and CD-1 mice, and it is clear that in the rat, bile duct hyperplasia did not progress (incidence or severity) beyond what was observed at 3 and 6 months. In the rat carcinogenicity study (0, 1, 5, 12, and 20/12 mg/kg/day; reduced survival at 12 and 20/12 mg/kg/day), the incidences of bile duct hyperplasia/fibrosis in males (69/125 [55%], 30/66 [45%], 42/68 [62%], 56/74 [76%], 45/77 [58%]) and females (20/125 [16%], 13/66 [20%], 18/67 [27%], 20/72 [28%], 15/77 [19%]) were interpreted as increased in animals given ≥5 mg/kg/day. Awareness of bile duct hyperplasia in early studies and reduced survival in higher dose groups impacted the conclusion that this finding was test article related in this carcinogenicity study. There was a modestly higher incidence of hepatocellular adenoma and carcinoma in males given 20/12 mg/kg/day (7/77; 9%) compared to controls (3/125; 2%), with no neoplasms of biliary origin (the hepatocellular tumors were an expected outcome as a result of peroxisome proliferator-activated receptor alpha [PPARα] activation). These data confirm that with this compound, minimal bile duct hyperplasia in the rat does not progress and likely does not represent a significant risk in humans. Additionally, there was no evidence of hepatic (cholestatic or otherwise) effects in the clinical trials of this compound.
In the 3-month mouse study of GW677954 at 0, 3, 10, 30, and 100 mg/kg/day (data for the high-dose group excluded due to early deaths), there was bile duct hyperplasia at 10 (1/11) and 30 mg/kg/day (11/12; 2.5 severity). There was significant hepatotoxicity at ≥10 mg/kg/day. In the 2-year carcinogenicity study at doses of 0, 1, 3, 10, and 15 mg/kg/day, there was an increased incidence of mostly minimal to mild bile duct hyperplasia in males (0/150, 3/90, 3/90, 22/90, and 43/90) and females (0/150, 0/90, 0/90, 18/90, and 31/90), and biliary cysts in males (2/150, 3/90, 8/90/ 23/90, and 38/90; 9 graded marked at ≥3 mg/kg/day) and females (0/150, 2/90, 6/90, 30/90, and 33/90; 8 graded marked at ≥10 mg/kg/day). Dose-related increased incidences of hepatocellular adenomas and/or carcinomas occurred in all treated groups of mice, but there were no increases in neoplasms of biliary origin. As in the rat, the hepatocelluar tumors observed in the mice were an expected outcome of chronic PPARα activation.
Bile Duct Hyperplasia (Atypical)
In humans, “atypical” hyperplasia is observed in a number of conditions (many progressive) to include PSC, primary biliary cirrhosis, after massive hepatic necrosis, focal nodular hyperplasia, chronic cholestatic disease, alcoholic liver diseases, and long-standing hepatic biliary obstruction (Priester et al. 2010; Svegliati-Baroni et al. 2008). Intra- and extrahepatic bile duct carcinomas represent a rare but significant complication of long-standing ulcerative colitis and are commonly preceded by sclerosing cholangitis and epithelial dysplasia (Anthony 2002). The biliary epithelium in patients with pancreaticobiliary maljunction demonstrates chronic injury and repeated regeneration that progresses to papillary hyperplasia and metaplasia or dysplasia, eventually resulting in carcinoma (Seki et al. 2005).
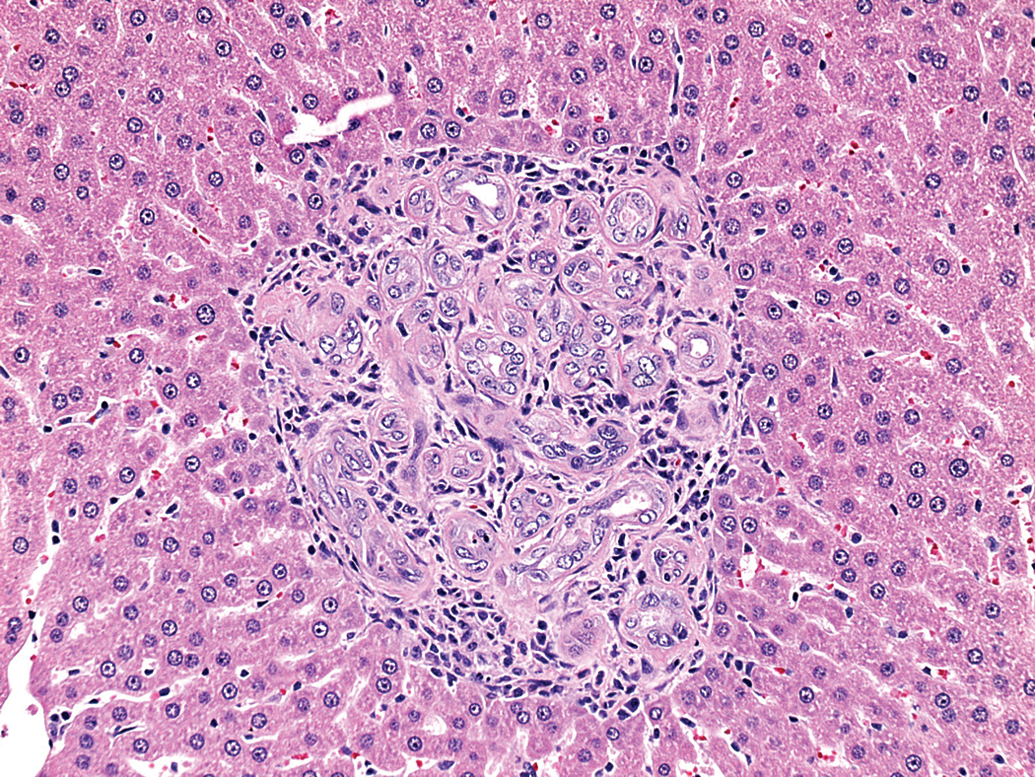
In laboratory animals, some biliary epithelial hyperplasia may appear atypical (Figure 6) and/or may occur outside the portal tract (Figure 7), as was observed in a 2-day study of thioacetamide and a 90-day study of 1,1,2,2-tetrachloroethane conducted by the NTP, and a 7-day and a 4-day study conducted by GSK. The carcinogenicity of thioacetamide is well established, and histologic atypia (biliary dysplasia) was observed by the 9th week in a study of rats exposed to 300-mg/l thioacetamide (Chun-Nan et al. 2004). Induced hyperplasia generally limited to the portal tract occurred in GSK’s 7-day study of GSK220405C (GSK proprietary data). In that study, minimal to moderate hypertrophy/hyperplasia, cytoplasmic vacuolation, and/or necrosis of the biliary epithelium were diagnosed. This was a more severe overall response than observed in the study of GW677954, and considered adverse based on severity alone; however, the component epithelium in some livers may be considered “atypical” (Figure 6).
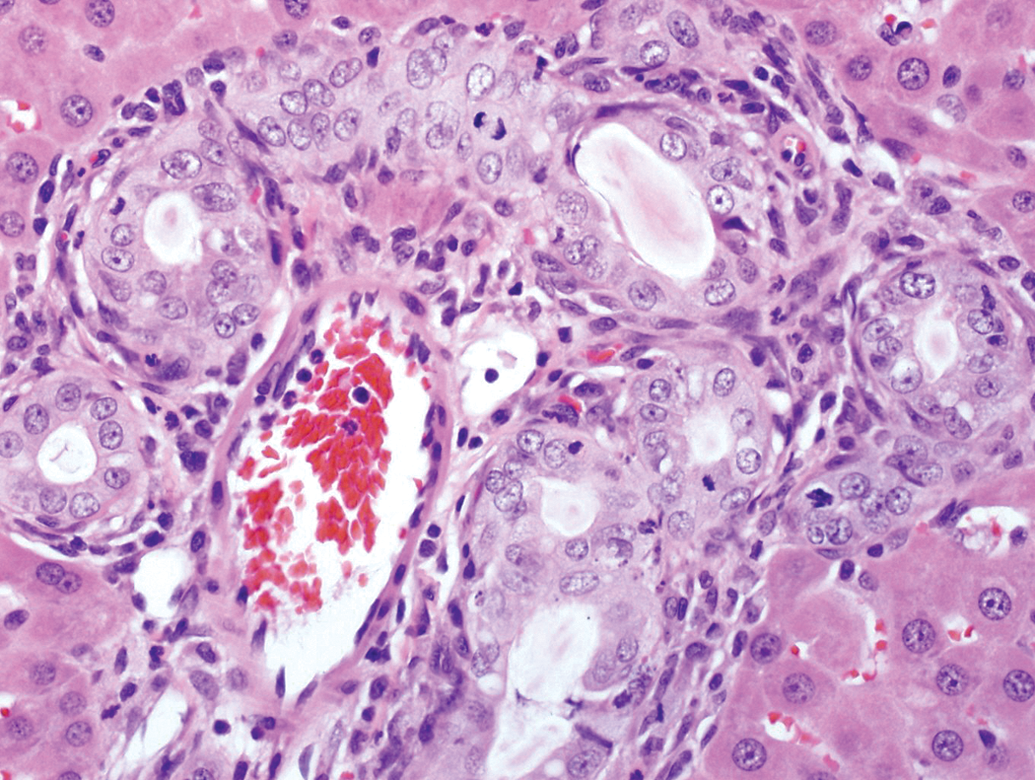
Male SD rat given 425-mg/kg/day GSK220405C for 7 days. Note: The increased profiles of bile ducts, hypertrophic biliary epithelium, atypia, mitotic figures, and inflammatory cell infiltrate (H&E; 40×). H&E = hematoxylin and eosin.
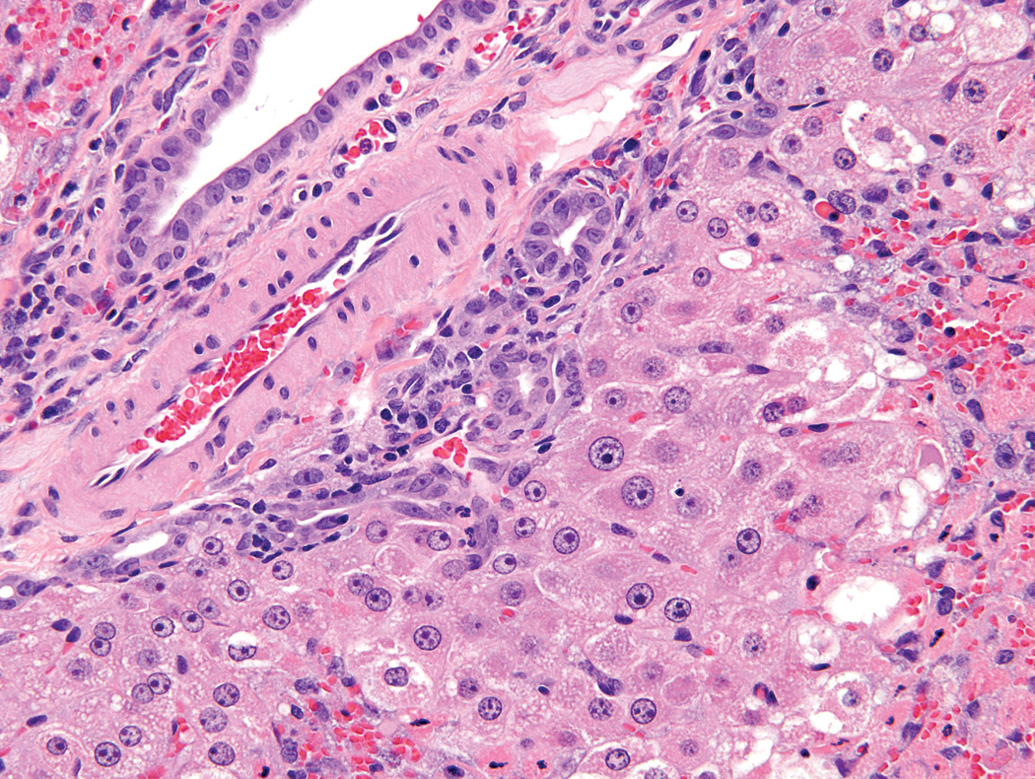
Male F344 rat from a National Toxicology Program study 48 hr after receiving a single dose of 150-mg/kg thioacetamide. Note: The slightly enlarged, basophilic, occasional spindled epithelium with slight extension into the adjacent hepatic parenchyma (H&E, 40×). H&E = hematoxylin and eosin.
Oval Cell Hyperplasia
There is considerable literature regarding types of stem cells of the liver; however, it is clear that small, oval shaped cells, generally referred to as oval cells are important. Oval cells are considered facultative stem cell precursors that arise from the canals of Herring and can differentiate along cholangiocellular or hepatocellular pathways (Sell and Dunsford 1989; Gennero et al. 2010). Oval cells are present in humans and are increased significantly in patients with nonalcoholic fatty liver disease or alcoholic liver disease, and cirrhosis is not required for accumulation of oval cells. Oval cells may proliferate in response to significant injury to cholangiocytes and/or hepatocytes or due to inhibition of replication of mature hepatocytes (Roskams et al. 2003). These authors suggest that the activation of progenitor cells (oval cells) during fatty liver disease may increase the risk of hepatocellular cancer, similar to that observed in the Solt–Farber model of hepatocarcinogenesis. This model has also been shown to result in extensive oval cell proliferation (Sell and Dunsford 1989) in the rat.
Oval cell hyperplasia (Figure 8), intestinal metaplasia (Figure 9), and/or cholangiofibrosis (Figure 10) are lesions that may be associated with significant liver damage, and at the severities shown here would be considered adverse in the rat. Cholangiofibrosis, a potential precursor to biliary neoplasia has not been observed in untreated control F344 rats in NTP studies (Eustis et al. 1990). Bannasch et al. (1985) report that the sequence of cellular changes leading to cholangiocellular tumors in rats has been investigated, and that these tumors develop after the application of high doses of carcinogens (e.g., N-nitrosomorpholine) that produced pronounced necrotic alterations of the liver parenchyma. Hepatocellular necrosis was also reported in the study of coumarin (Evans et al. 1989). However, hepatocellular necrosis is not reported in all studies in which oval cell proliferation and cholangiocarcinoma are observed. Although very early time points were not evaluated, hepatocellular necrosis was not observed in the group of studies conducted by the NTP on dioxins and dioxin-like compounds (NTP reports 520, 521, 525, 526, 529, 530, 531, 558, and 559) and was not mentioned (Oshima, 1984) or was minimal (GSK databases) or periportal only at a high dose (Craig et al. 2006) in the studies of methapyrilene. In a GSK oral study of dioxin administered for only 4 days but at doses (1 mg/kg/day, 7.5 mg/kg/day, and 15 mg/kg/day in Labrafil) higher than the highest dose (100 ng/kg/day) used in the NTP studies, minimal to mild hepatocyte hypertrophy and vacuolation were the predominant changes observed in treated animals (GSK databases). N-acetyl-2-aminofluorene is an inhibitor of hepatocellular replication and a potent inducer of oval cell hyperplasia, which may occur without obvious necrosis (Sell and Dunsford 1989; Tatematsu et al. 1984).
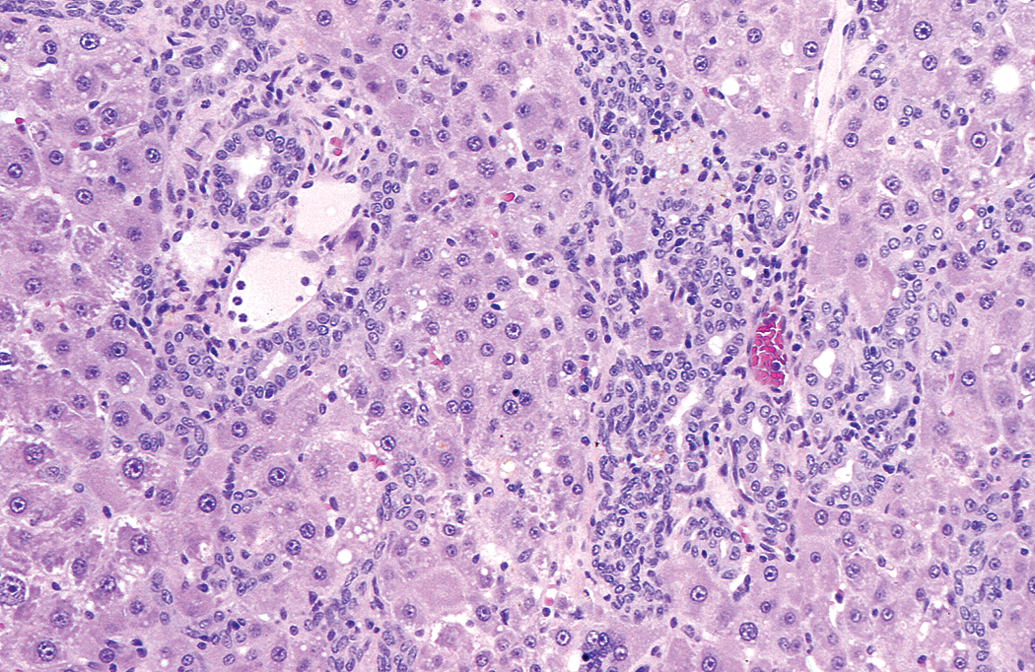
Female Harlan SD rat from a National Toxicology Program 2-year carcinogenicity study of PCB-126. Note: A slight increase in bile ducts and proliferation of spindled (oval) cells (H&E, 20×). H&E = hematoxylin and eosin.
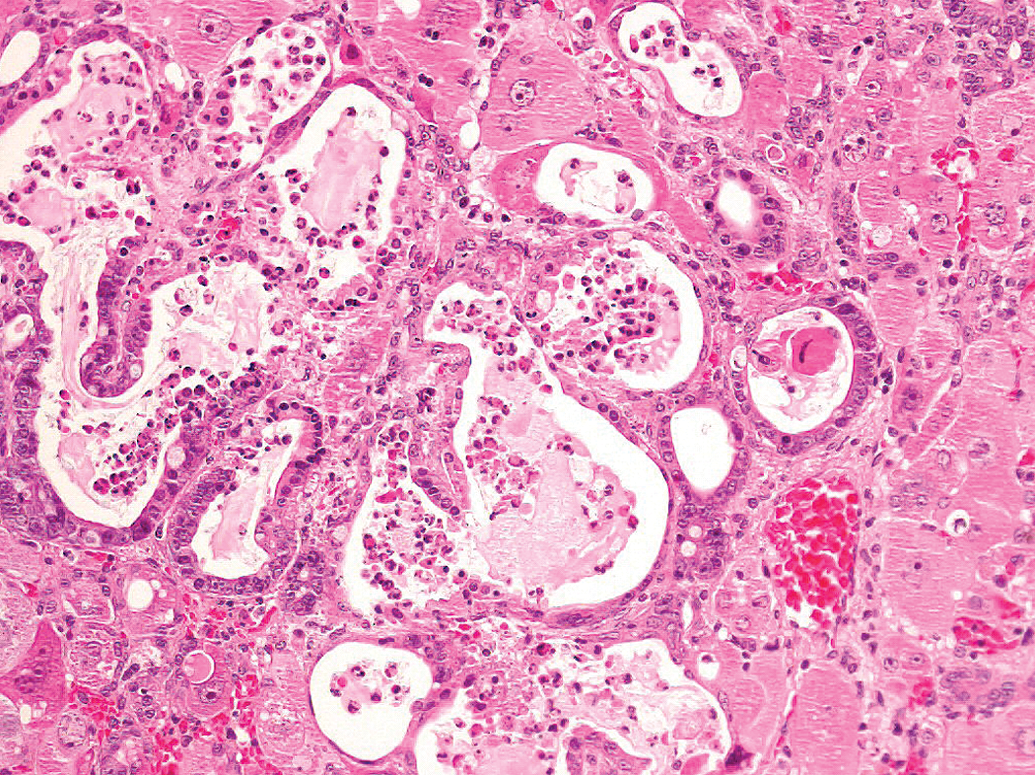
Male SD rat given GSK619437 for 4 days. Note: The tall cuboidal to columnar epithelium lining some ductules/glandular structures (intestinal metaplasia) and the luminal debris (H&E, 20×). H&E = hematoxylin and eosin.
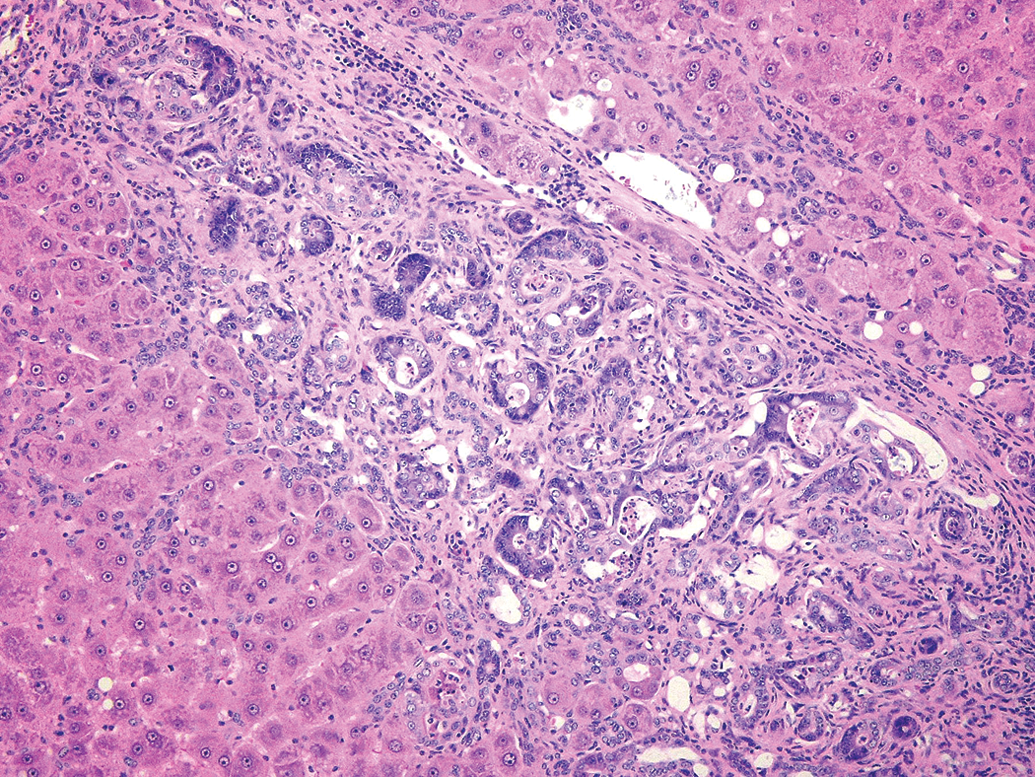
Female Harlan SD rat from a National Toxicology Program 2-year carcinogenicity study of PCB-126. Note: The ductular/glandular structures within fibrous connective tissue (cholangiofibrosis). Some glandular structures are lined with tall columnar epithelium (intestinal metaplasia) (H&E, 16×). H&E = hematoxylin and eosin.
Bannasch (1984) states that under experimental conditions, the changes in the bile duct epithelia have been shown to start with a ductular (oval) cell proliferation, which may progress to cholangiofibrotic areas, benign cystic cholangiomas, cholangiofibromas, and eventually cholangiocarcinomas. It is commonly agreed that one of the earliest morphologically detectable changes in liver carcinogenesis induced by chemicals or by radiation is the proliferation of small epithelial cells (oval cells) in the periportal areas (Schauer and Kunze 1976), although some data (Paku et al. 2005) suggest a stem cell precursor to oval cells proliferates first. Oval cells appear in most, although not all, models of chemically induced hepatocarcinogenesis (Fausto 1990). Oval cell proliferation may occur as the result of massive or chronic liver injury (Housset 2000) but also may be associated with the inhibition of hepatocyte proliferation (Tatematsu et al. 1984; Roskams et al. 2003). The spectrum of oval cell hyperplasia, cholangiofibrosis, and cholangiocarcinoma referred to by Bannasch was described in NTP’s dioxin (NTP reports 520, 521, 525, 526, 530, 531, 558, and 559) and furan studies (NTP report 402 [NTP 1993b]; Maronpot et al. 1991). Although not an exhaustive search, this spectrum is also described in other studies (methapyrilene, Ohshima et al. 1984; thioacetamide, Praet and Roels 1989; chloroform, Jamison et al. 1996; and coumarin, Evans et al. 1989) in which chemical induction of cholangiocarcinomas are reported. Some of these compounds appear to be genotoxic while others do not.
Bile duct, but not oval cell, proliferation is generally mentioned when ANIT-induced lesions are discussed (Greaves 2000; Zimmerman 1999b). This chemical is referred to as a noncarcinogen (Dunsford et al. 1984; Greaves 2000); however, an International Agency for Research on Cancer listing of agents studied or under study for carcinogenicity (Vol. 16, 1994) did not list ANIT as having been tested. In a GSK oral study of ANIT administered for 4 days at doses of 10, 30, and 100 mg/kg/day in PEG 400, mild to moderate biliary hyperplasia and oval cell hyperplasia were diagnosed (GSK databases).
While evidence suggests that oval cell proliferation may be a harbinger for cholangiocarcinoma in the rat, this is most useful in nonclinical study evaluation if it or an associated/precursor lesion can be observed at the lowest doses in which cholangiocarcinomas would potentially occur, and at an early time point, preferably within 4 weeks. We assessed available study data from six chemicals known to cause cholangiocarcinoma for this correlation.
Thioacetamide administered at 300 mg/l in drinking water resulted in cholangiofibrosis and cholangiocarcinoma (Chun-Nan et al. 2004). In this study, animals were examined weekly, and the first histologic evidence of biliary toxicity, bile ductular proliferation with atypia (biliary dysplasia) occurred at week 9 with 50% of examined animals affected. In a short-term NTP study, animals were administered single doses of 15, 50, or 150-mg/kg thioacetamide with histological evaluations at 6-, 24-, and 48-hr time points. Minimal hepatocellular necrosis was seen as early as 6 hr, and was marked at 48 hr, particularly in the 150-mg/kg group. Minimal biliary epithelial hyperplasia was first observed at 48 hr in the 50-mg/kg and 150-mg/kg groups (Houle 2005). The biliary epithelium was deeply basophilic (Figure 7) and streaming in some areas with abundant mitotic figures. Oval cell hyperplasia was not observed at this early time point. In a GSK oral study of thioacetamide administered for 4 days at doses of 10 mg/kg/day, 25 mg/kg/day, and 50 mg/kg/day in 1% methylcellulose, biliary hyperplasia was observed in 2/5 animals exposed to 25-mg/kg/day thioacetamide. To what extent the hepatic necrosis contributed to the biliary epithelial hyperplasia in the NTP study is not known however, in a GSK study of dimethylformamide, there was marked to severe liver necrosis in rats given 1,000 mg/kg/day for 4 days with no apparent affect on the biliary epithelium.
As with thioacetamide, evaluation of study data for methapyrilene hydrochloride (Ohshima et al. 1984, GSK databases), furan (Maronpot et al. 1991; Elmore and Sirica 1992; GSK databases), coumarin (Evans et al. 1989; GSK databases), chloroform (Reuber 1979; Jamison et al. 1996; GSK databases), and TCDD (NTP report 521; GSK databases) was often hampered by a lack of toxicity data at comparable doses or relevant times. Therefore, whether oval cell hyperplasia would always be observed within 4 weeks at the lowest dose inducing cholangiocarcinoma was not determined from evaluation of these six chemicals. However, with the possible exception of dioxin, it is likely that an adverse liver effect would have been identified within 4 weeks.
The spectrum of lesions generally classified as cholangiofibrosis, cholangiofibroma, and cholangiocarcinoma in the rat is a morphological continuum, and identifying specific criteria for separation of the various categories is difficult and controversial with biological information lacking or conflicting (Bannasch et al. 1985; Maronpot et al. 1991; Elmore and Sirica 1992; Jamison et al. 1996; Evans et al. 1989). It is clear that the lesions progress with time, and some studies have demonstrated progression in the absence of continued treatment as well as transplantability (Maronpot et al. 1991; Elmore and Sirica 1993). Although large numbers of cholangiocarcinomas are diagnosed in some studies, distant metastasis is rare (NTP reports 520 and 521; Maronpot et al. 1991; Ohshima et al. 1984; Evans et al. 1989). Jamison et al. (1996) suggest that at least in some instances these lesions may reflect intestinal metaplasia rather than neoplasia. The INHAND guidance document indicates that most of these lesions should be diagnosed as cholangiofibrosis rather than cholangiocarcinoma. It is unclear whether this spectrum of lesions may occur in humans.
While there is no known correlate of cholangiofibrosis in humans, von Meyenburg complexes are small (<0.5 cm in diameter) grayish white or green lesions usually scattered in both lobes. Microscopically, the lesions are discrete, round to irregular in shape, and typically paraportal in location. The constituent ducts are embedded in collagenous stroma and are often round, but may be irregular, and have a slightly dilated lumen. They are lined by low columnar or cuboidal epithelium and contain pink amorphous material that may be bile stained, or actual bile. Cholangiofibrosis in rats is a rounded to irregular lesion with rounded to irregular glands within a collagenous stroma and a multifocal scattered distribution. It differs in that the component cells are generally taller (columnar) often with intestinal metaplasia, and lumens often filled with cellular debris. Cholangiocarcinomas have been reported in association with von Meyenburg complexes (Ishak and Sharp 2002).
In humans, there are many risk factors for intrahepatic cholangiocarcinoma including PSC, parasitic infections, choledochal cysts, hepatolithiasis, and exposure to exogenous carcinogens such as thorotrast (thorium dioxide; de Groen et al. 1999; Chun-Nan et al. 2004). Chun-Nan et al. suggest that the spectrum of lesions (cholangiofibrosis to cholangiocarcinoma) induced by the administration of thioacetamide to rats represents an animal model for progression of PSC to cholangiocarcinoma in humans. In rats exposed to thioacetamide, there was upregulation of the proto-oncogenes c-met and c-erbB-2 in dysplastic and neoplastic glands and cholangiocarcinomas compared with normal ductules in the livers. These tyrosine kinase receptors may also be overexpressed in cholangiocarcinomas in humans. Mutations in other oncogenes as well as tumor-suppressor genes have been described in specimens of biliary tract tumors from humans to include K-ras, c-myc, c-neu, p53, and bcl-2.
Biliary Hyperperplasia with Concomitant Hepatocellular Damage
Hepatocellular effects may result in minimal to mild biliary hyperplasia being considered adverse due to the aggregate liver effect. Often, responses in the biliary tract have been considered separate from those involving hepatocytes, and it has been proposed that neoplasms of hepatocytes and biliary epithelium should not be combined for assessment of liver tumor response (McConnell et al. 1986). While there are instances whereby the hepatocyte or biliary epithelium is thought to be the primary (e.g., ANIT) or only target, as more is learned about the regeneration/repair of the liver, it is apparent that often the hepatocyte and biliary epithelium are affected, and both along with intermediate and/or progenitor cells replicate/respond to injury; for example, ANIT is now known to secondarily injure hepatocytes as well (Zimmerrman 1999b and GSK databases). Not only do hepatocytes and biliary epithelium have the ability to replicate and repair damaged liver in the rat, oval cells thought to arise from the canals of Herring may also be involved in a reparative/regenerative response, and may differentiate toward hepatocytes and/or biliary epithelium (Sell and Dunsford 1989). Hepatocytes and epithelial cells of the intrahepatic system of bile ducts originate from primitive hepatocytes (hepatoblasts) and it is likely that in adult livers, cells located very near to the canals of Herring constitute a compartment of facultative progenitor cells which have the ability to give rise to both differentiated hepatocytes and bile duct epithelium. Morphological evidence of this mixed response includes liver tumors classified as hepatocholangioma and hepatocholangiocarcinoma. Also, within NTP’s study of PCB-126, glandular structures present in severely damaged liver, and assumed to represent regeneration, were lined by cells resembling hepatocytes, biliary epithelium, and/or a combination of both (Figure 11); similar lesions have been described by Sirica (1995) as “neocholangiolar” proliferations. Therefore, morphological separation of a hepatocyte response from a biliary epithelium response may not be clear cut in some instances, nor necessarily appropriate.
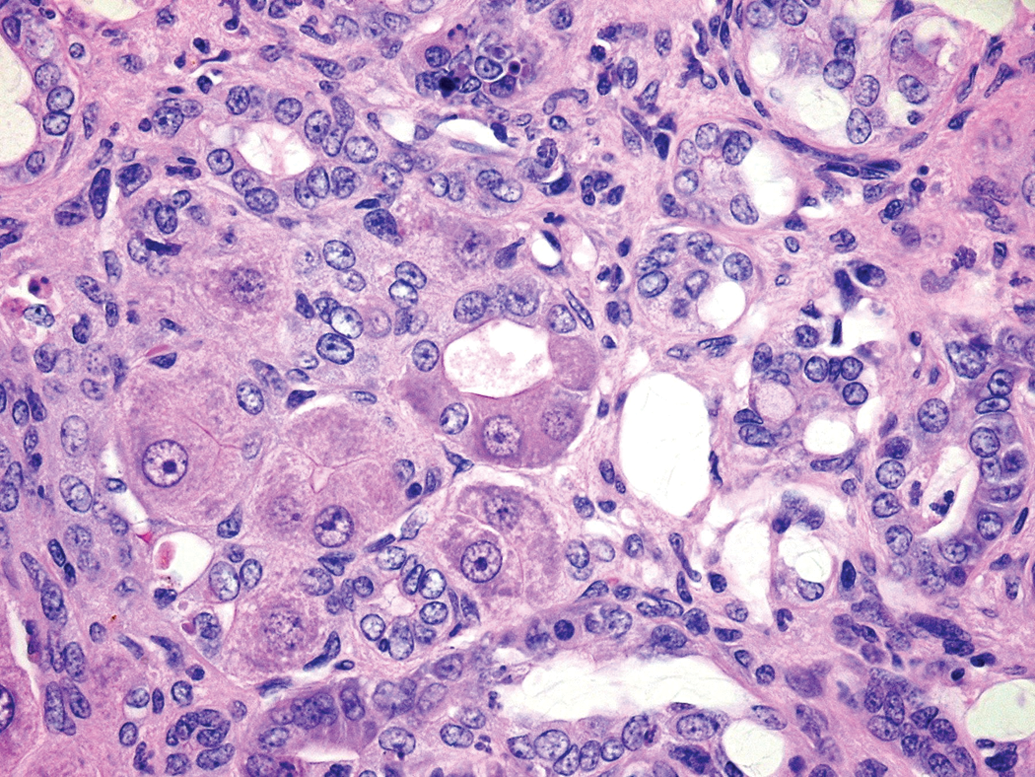
Female Harlan SD rat from a National Toxicology Program 2-year carcinogenicity study of PCB-126. Note: The ductular/glandular structures lined by cells resembling hepatocytes, biliary epithelium, or both (H&E, 60×). H&E = hematoxylin and eosin.
Biomarkers
While circulating biomarkers of liver injury involving hepatocellular damage (e.g., transaminase activities) and cholestasis (e.g., TB, ALP, and TBA) are available for preclinical (Boone et al.) and clinical monitoring, there remains a paucity of acceptable biomarkers of minimal to mild biliary epithelium hyperplasia, although this gap is being addressed by the Critical Path Predictive Safety Testing Consortium and other public–private partnerships. An internal review of data from ∼3,200 rats with hepatotoxicity in GSK studies demonstrated that biliary damage (including biliary hyperplasia, degeneration/regeneration, and/or necrosis) in the absence of other types of liver injury was uncommon (n = 126). In addition, there were no consistent correlations between serum ALT, aspartate aminotransferase, ALP, and GGT activities, TB or TBA, and biliary hyperplasia in these animals (Ennulat et al. 2010). In a study of 1,235 liver biopsies from patients with abnormal clinical chemistry markers (Sonzogni et al. 2004), idiopathic bile duct proliferation occurred in only 16 specimens. The chemistry abnormalities in these patients included mildly increased GGT (81%), ALT (69%), TB (25%), and/or ALP (19%). Gamma-glutamyl transferase is present in high concentrations in human and rat biliary epithelium, and while serum activity may increase as a result of biliary necrosis (Leonard et al. 1984), it is less reliable as a marker of biliary epithelial hyperplasia in the rat due to its short circulating serum half-life. Elevated serum activities of other enzymes, such as ALP and 5′nucleotidase, have been associated with biliary diseases; however, they are also increased with hepatocellular injury and with a variety of non-hepatic disorders. Because biliary epithelium comprises less than 5% of the liver parenchyma, the challenges of accurately identifying aberrations in this small cell population via a peripheral blood sample have led an increasing number of investigators to explore bile constituents as alternatives (Lankisch et al. 2011).
Summary
Whether biliary proliferative lesions in the rat are adverse and potentially predictive of hepatotoxicity in humans is dependent on the nature and severity of the morphologic change in the rat. This is best addressed with direct correlation between nonclinical and clinical data; however, these data are not sufficiently complete for development of definitive conclusions. Based on the evaluation of existing data and grouping proliferations into four broad categories (typical and atypical biliary epithelial hyperplasia, oval cell hyperplasia, and cholangiofibrosis), we conclude that minimal “typical” (e.g., duplication of bile ducts) biliary epithelial hyperplasia alone or with very minimal associated inflammatory cell infiltrate and/or hepatocellular changes may be considered non-adverse in the rat in nonclinical safety studies. Information/data supporting this conclusion includes (a) low occurrence of typical biliary epithelial hyperplasia in humans where the only reported cases were nonprogressive; (b) occurrence in rats as a common spontaneous change that has no apparent functional impact on the liver, and without progression to neoplasia; (c) relatively low incidence of chemically induced neoplasms of the biliary tract in rodent chronic studies; and (d) nonclinical toxicology study data demonstrating that chemically induced minimal biliary hyperplasia in the rat does not progress with time, is reversible, and does not affect functionality (e.g., cholestasis). The biological relevance of mild to moderate “typical” hyperplasia is less certain, and may be considered adverse in the rat, particularly if accompanied by other evidence of hepatobiliary injury or functional compromise. Further, any proliferative lesion with atypical or dysplastic epithelial changes, oval cell proliferation, and/or significant extension beyond the portal tracts appears to be more ominous, and therefore may be considered adverse in the rat. It is likely that sufficient correlative nonclinical and clinical data exist across pharmaceutical companies, and combined data sets could be invaluable in confirming or modifying these conclusions.
Footnotes
Authors’ Note
All studies were conducted after review by the GSK Institutional Animal Care and Use Committee and in accordance with the GSK Policy on the Care, Welfare, and Treatment of Laboratory Animals.
Acknowledgement
The authors gratefully acknowledge The Critical Path Institute's Predictive Safety Testing Consortium for its support in enabling discussions and debates that helped shape the thinking, and inspire the effort.
Abbreviations
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
