Abstract
Recombinant adeno-associated virus (AAV)-mediated degeneration of sensory neurons in the dorsal root ganglia (DRG) and trigeminal ganglia (TG) has been observed in non-human primates (NHPs) following intravenous (IV) and intrathecal (IT) delivery. Administration of recombinant AAV encoding a human protein transgene via a single intra-cisterna magna (ICM) injection in New Zealand white rabbits resulted in histopathology changes very similar to NHPs: mononuclear cell infiltration, degeneration/necrosis of sensory neurons, and nerve fiber degeneration of sensory tracts in the spinal cord and of multiple nerves. AAV-associated clinical signs and incidence/severity of histologic findings indicated that rabbits were equally or more sensitive than NHPs to sensory neuron damage. Another study using human and rabbit transgene constructs of the same protein demonstrated comparable changes suggesting that the effects are not an immune response to the non-self protein transgene. Rabbit has not been characterized as a species for general toxicity testing of AAV gene therapies, but these studies suggest that it may be an alternative model to investigate mechanisms of AAV-mediated neurotoxicity and test novel AAV designs mitigating these adverse effects.
Introduction
Recombinant adeno-associated virus (AAV)-based gene therapies have become a major area of emphasis in drug discovery and development with the promise of delivering lifetime efficacy to patients with a single dose. Recombinant AAVs (rAAVs) are particularly appealing for pharmaceutical applications because of their lack of replication, low potential for chromosomal integration, and large range of viral tropism. 11
Like other modalities, as the field has matured, recent insights have uncovered possible modality-related adverse effects. One of the potential toxicities identified following either intravenous (IV) or intrathecal (IT) administration of AAV is degeneration of sensory neurons in the spinal sensory ganglia (dorsal root ganglia [DRG]) and trigeminal ganglia [TG], in non-human primates (NHPs), pigs, and rodents.1,2,4,10 Recent publications describing the DRG degeneration in NHPs highlight hallmark characteristics, including sensory neuron degeneration/necrosis with mononuclear cell infiltration and nerve fiber degeneration of sensory tracts in the spinal cord and of multiple nerves,1,3,4,10 manifesting anywhere from 2 to 4 weeks after IV and central nervous system (CNS) administration of AAV. This effect is reproducible using different AAV capsid serotypes with different transgene products, suggesting there is an inherent characteristic of the AAV vector and transgene expression that is driving this finding. 4 Translatability to humans is not firmly established. However, reports of sensory neuropathies in clinical trials8,9 and DRG degeneration of similar phenotype observed in post-mortem autopsy tissues from clinical trial participants 12 suggest that this effect can occur in humans though these clinical cases are not definitive evidence of clinical translatability as they could be confounded by disease progression in these patients. While NHPs have proven to be a good model to study AAV-mediated DRG toxicity, mechanistic investigations are hindered by the ethical responsibilities and technical difficulties of utilizing this test species.
In this report, we demonstrate that the New Zealand white rabbits dosed with a proprietary AAV encoding a human (non-self) protein transgene (AAV-HuTg) or rabbit (self) protein transgene (AAV-RbTg) exhibit DRG toxicity with similar pathological characteristics and timeframe to NHPs. These findings indicate that rabbits can recapitulate the pathological phenotype observed in NHPs, and that this effect does not appear to be related to immune recognition of a non-self transgene protein, consistent with observations in NHPs by other investigators. 4
Methods
Rabbit Studies
Two separate studies were conducted. In both studies, male New Zealand white rabbits (4-6 months of age, N = 3 per group, Envigo) in a laterally recumbent position were dosed via single intra-cisterna magna (ICM) bolus injection, confirmed by fluoroscopic imaging. In the first study (Study 1), rabbits were injected with vehicle (artificial cerebrospinal fluid [CSF]) or AAV-HuTg at 1 × 1013 genome copy (GC)/animal or 3 × 1013 GC/animal. In the second study (Study 2), rabbits were injected with vehicle (artificial CSF), AAV-HuTg at 1 × 1013 GC or AAV-RbTg at ~8 × 1011 GC/animal (actual dose based on titer 7.4 × 1011 or 8.9 × 1011), 3 × 1012 GC/animal (actual dose based on titer of 2.4 × 1012), or 1 × 1013 GC/animal (actual dose based on titer of 1.2 × 1013 or 8.4 × 1012 GC/animal). Animals are referred to based on target dose throughout this article for simplicity. Both studies used a dose volume of 0.3 ml, with a 4-week post-injection in-life observation period. Doses were selected based on extrapolation from doses used in NHP studies with the AAV-HuTg construct corrected for brain weight in rabbits (10 g) compared with NHP (70 g) as estimated from the historical database maintained at the facility where the studies were conducted. The dose spread was selected to balance the expected transduction compared with what was known in NHP with the desire to best detect DRG degeneration, if present.
For both studies, parameters and end points evaluated included mortality and morbidity, clinical signs, body weights, food consumption, serum clinical chemistry (just prior to euthanasia), CSF cell count and clinical chemistry, gross pathology, organ weights (recorded on animals surviving until scheduled euthanasia), in situ hybridization (ISH, for cargo RNA expression), immunohistochemistry (IHC, for cargo protein expression), biodistribution (real-time quantitative polymerase chain reaction [qPCR] for vector DNA), serum neurofilament levels, microscopic pathology, and transmission electron microscopy (TEM only in Study 2) examinations. The care and use of the animals on the study were conducted in accordance with the guidelines of the US National Research Council and the Canadian Council on Animal Care. 7 All rabbits were housed in set-ups as described, 13 provided with toys and food enrichment, and fed 120 g per day of a certified laboratory rabbit diet and provided water ad libitum. At the time of necropsy, all animals were administered a sedative, Ketamine HCl for injection, xylazine and glycopyrrolate by intramuscular injection and euthanized by exsanguination via incision of the axillary or femoral arteries.
For both studies, all tissues were fixed in neutral-buffered formalin for histological processing. A select tissue list, including the administration site, brain, spinal cord, DRG with dorsal nerve roots, ventral nerve roots, sympathetic chain ganglia (C3, T6, L3 level), TG with trigeminal nerve (unilateral/right in Study 1; bilateral in Study 2), median nerve (bilateral), sciatic nerve (bilateral), kidney, liver, and gross lesions were processed, embedded in paraffin, sectioned, mounted on glass slides, stained with hematoxylin and eosin (H&E), and evaluated microscopically from all scheduled euthanasia animals, while a full tissue list was evaluated microscopically for early euthanized animals. Cervical, thoracic, lumbar spinal cord was collected with DRGs with meninges. For the spinal cord, DRG, and dorsal and ventral nerve roots, two to three samples were microscopically evaluated per cervical (C2, C3, and/or C6), thoracic (T2, T6, and/or T8), and lumbar (L1, L3, L4, and/or L5-L6) regions. The severity grade of microscopic findings recorded in the incidence/severity table for each of these regions was based on the most severely affected sample per region. The details for severity grade in each sample (e.g., C2, C3, and/or C6) within the region (e.g., cervical) were recorded in the individual animal data. In addition to the standard H&E staining, the cervical, thoracic, and lumbar DRGs were stained using GFAP (glial fibrillary acidic protein) and GLUL (glutamine synthetase GS-6) IHC to better identify the satellite glial cells. The IHC grading was based on the amount of signal per cell (signal intensity) and the number of immunopositive cells. The grading scheme generally followed Boos et al, 2021 (https://pubmed.ncbi.nlm.nih.gov/34307520/) and consisted of the following grades: non-altered (grade 0), minimal (grade 1), mild (grade 2), and moderate (grade 3). Cervical and lumbar DRGs for transmission electron microscopic evaluation were fixed in McDowell-Trumps fixative (4.0% formaldehyde-1.0% glutaraldehyde in phosphate buffer), processed into resin blocks, thick sectioned at approximately 1 micron and stained with toluidine blue. Images were captured using a JEOL JEM-1400+ with an AMT XR16MP digital camera. Select animals (N = 1; with most severe microscopic findings in AAV-dosed groups) were evaluated by TEM from vehicle, AAV-HuTg (1 × 1013 GC) and AAV-RbTg (1 × 1013 GC) groups in Study 2 only. Histopathological and TEM evaluations were performed by the American College of Veterinary Pathologists (ACVP) board-certified veterinary pathologists experienced in toxicologic pathology.
Vector Design and Production
Human and rabbit protein-expressing AAV cis plasmids were generated by insertion of the respective open reading frames (ORFs) under the control of a CAGG promoter and completed by addition of the rabbit globin poly adenylation (pA) signal in the 3′ region. AAV vectors were produced by Packgene (Worcester, MA) by triple transfection in HEK293 cells and purified by iodixanol gradient. Residual iodixanol was removed by affinity chromatography using Poros™ CaptureSelect™ AAV resin (Thermo Fisher Scientific, Waltham, MA) according to the manufacturer’s guidelines. AAV-containing fractions were concentrated and dialyzed to a proprietary formulation using Amicon centrifuge devices (Millipore, Burlington, MA).
DNA Extraction and Real-Time Quantitative Polymerase Chain Reaction for Biodistribution Analysis
Tissues were homogenized and subjected to tissue lysis, digestion and DNA extraction procedures using the DNeasy 96 Blood & Tissue Kit (Qiagen, Germany). Male New Zealand white rabbit genomic DNA was used as tissue-type matrix (Zyagen, San Diego, CA). Genomic DNA samples were analyzed by quantitative polymerase chain reaction (qPCR; TaqMan Universal PCR Master Mix, no AmpErase UNG, Applied Biosystems) using custom TaqMan primers and probes that target the rabbit RPP30, ribonuclease P protein subunit p30, housekeeping gene (IDT) and the AAV transgene (Thermo Fisher) in single-plex. RPP30 (Forward: 5′-CCATGTGAGAGACCTGGAATAAG-3′; Reverse: 5′-GAGAAGGTGAGGCAGAGAAAG-3′; Probe: 5′-/5HEX/AG CTC CAG C/ZEN/C TTT GTG GTC ATC TG/3IABkFQ/-3′) and the information of transgene primers and probes is proprietary to Biogen. The cycling conditions were 10 minutes at 95°C, 40 cycles of 15 seconds at 95°C, and 1 minute at 60°C. Samples at Northern Biomolecular Services for Study 2 were evaluated as follows: DNA was analyzed by qPCR using custom Taqman primers and probes as above at (Taqman Multiplex PCR Master Mix, Thermo Fisher Scientific, 4461882). The cycling conditions were 20 seconds at 95°C, and 40 cycles of 1 second at 95°C and 20 seconds at 60°C.
The copy numbers of the viral DNA and quantity of gDNA were interpolated from a standard curve. The units reported from this analysis are viral GCs/diploid GC numbers of rbRPP30 (dg).
RNA Extraction, cDNA Synthesis, and RT-qPCR Analysis for Transgene mRNA Expression
Brain (motor cortex, frontal cortex, cerebellum, and brain stem) and liver were collected and stored frozen at −60°C to −90°C until tissue homogenization. Each tissue was homogenized and lysed in Precellys tubes containing QIAzol lysis reagent (QIAGEN, 79306). Automated RNA extraction was performed using the QIAsymphony. Total RNA samples were reverse transcribed into cDNA using the Superscript IV VILO reverse transcription kit (Invitrogen, 11756500). Reverse transcription was performed using 500 ng of total RNA under the following conditions: 25°C for 10 minutes, 50°C for 10 minutes, 85°C for 5 minutes and held at 4°C.
Samples were analyzed by Taqman qPCR (Taqman Multiplex PCR Master Mix, Applied Biosystems, 4461884) using custom duplexed primers and probes targeting the human and rabbit AAV transgenes, and rabbit RPP30 housekeeping gene (Thermo Fisher, Oc06741068_m1). The cycling conditions were 20 seconds at 95°C, and 40 cycles of 1 seconds at 95°C and 20 seconds at 60°C. RNA expression is reported as fold change of transgene expression normalized to rabbit RPP30 using ΔCT method.
ISH for Transgene mRNA Expression
Transgene expression was detected in histologic sections using red chromogenic RNAscope assays (Advanced Cell Diagnostics). Assays were run on a Leica BOND RX autostainer according to the manufacturer’s instructions, including the use of rabbit Ppib and dapB quality control assays. A custom antisense 16zz pair probe set was used to detect codon-optimized sense HuTg transcripts and an antisense 14zz pair probe set was used to detect sense RbTg transcripts. In assay optimization experiments, RNAse digestion performed on DRG tissue sections from rabbits dosed with AAV-HuTg eliminated cytoplasmic RNA reactivity and most nuclear RNA reactivity, but sparse multifocal nuclear dots, consistent with episomal vector reactivity, persisted. In spinal cord sections, ISH for transgene expression was duplexed with CHAT IHC. Whole slide images (WSIs) were generated using a 3D-Histech Panoramic P250 and were analyzed using Visiopharm software (v. 20.11.0.8401). DRG and somatic motor neurons, inclusive of cytoplasmic and nuclear compartments, were segmented using artificial intelligence-based algorithms trained on over 1000 neurons of each type. Custom algorithms were designed to segment transgene hybridization signal in individual segmented neurons. Percent neuronal transduction was calculated in HuTg experiments as the fraction of neurons with transgene reactivity over all segmented DRG neurons. Single neuron reactivity densities were calculated as the fraction of neuronal area (cytoplasmic and nuclear compartments) reactive for ISH signal over the entire neuronal area, inclusive of the limitation that vector reactivity persisting after RNAse digestion comprised a very minor component of nuclear reactivity. Data were plotted using Graphpad Prism.
Serum Neurofilament Heavy Enzyme-Linked Immunosorbent Assay
Whole blood was collected into serum separator tubes at baseline, 12 hours, and 2, 5, 9, 15, and 29 days following ICM injection of vehicle or AAV administration. Samples were centrifuged at 1400 × g for 10 minutes at 4°C and then stored at −80°C until use. Serum samples were then evaluated by enzyme-linked immunosorbent assay (ELISA) for neurofilament heavy (NF-H) (BioTechne, SPCKA-PS-000519). Serum samples were diluted 1:1 in equivalent volume of the provided sample diluent and 50 µl was loaded per well on a 72-well cartridge.
Results
In Study 1, the high dose of AAV-HuTg (3 × 1013 GC) was not tolerated. All three high-dose animals receiving the AAV-HuTg (3 × 1013 GC) were euthanized early (between Days 12 and 26) in the interests of animal welfare due to decreased appetite, activity, body weight loss, and/or general poor clinical condition. Body weight increased slightly in one animal (+7%) and was decreased in the remaining two animals (−9% to −22%) with a corresponding decrease in food consumption (−2% to −96%). One low-dose (1 × 1013 GC) animal was euthanized early on Day 18 due to decreased activity that did not improve with supportive care. Additional clinical observations related to AAV-HuTg vector administration included, but were not limited to, reduced or abnormal fecal output, tongue erosions/ulcerations, abnormal/irregular respiratory rate (starting from Day 10), limited use of hindlimbs and abnormal gait (starting from Day 12), incoordination (Day 24) and dehydration.
In Study 2, all animals at all dose levels (8 × 1011, 3 × 1012, and 1 × 1013 GC) of AAV-RbTg and at 1 × 1013 GC of AAV-HuTg survived to the end of the planned study in-life period. Clinical observations related to AAV-HuTg and/or AAV-RbTg included, but were not restricted to, limited usage of hindlimbs, reduced fecal output, transient abnormal gait, thinness, reduced muscle tone, and incoordination. Body weight decreases correlated with reduced food consumption, which was more prominent in AAV-HuTg compared with AAV-RbTg group at the same dose level (1 × 1013 GC) (Table 1).
Changes in mean (n = 3/group) food consumption (FC) and body weight (BW) from Study 2.
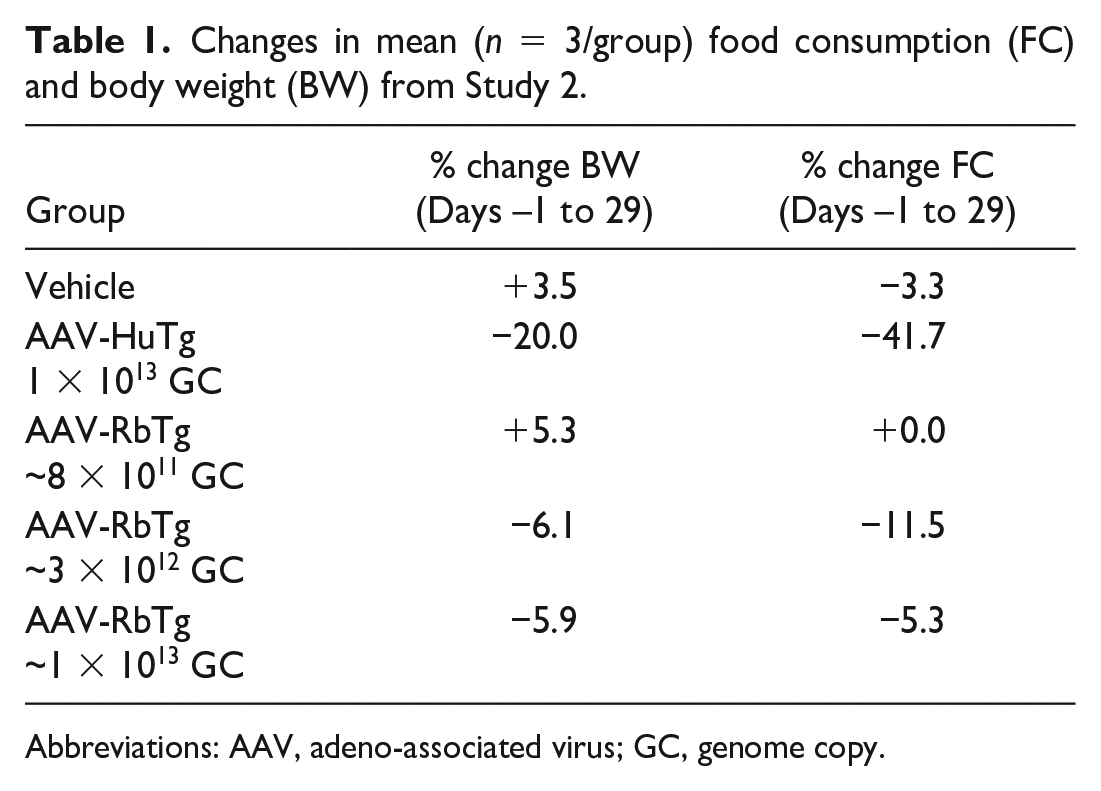
Abbreviations: AAV, adeno-associated virus; GC, genome copy.
Clinical chemistry changes apparent in pre-terminally euthanized rabbits in Study 1 receiving the AAV-HuTg were generally similar in each animal. These serum chemistry alterations were dominated by changes consistent with inflammatory responses to AAV treatment, and included decreased albumin and albumin/globulin ratio, increased globulin, and/or decreased serum iron. Additional serum chemistry changes were considered secondary to declining clinical conditions of the animals and were generally indicative of dehydration/decreased glomerular filtration rate (increased urea nitrogen and/or creatinine), loss of protein (decreased total protein), stress (increased glucose), and prolonged recumbency-related acute muscular injury (increased creatine kinase activity). Increases in total nucleated cell counts (15-23 cells/µl) were observed in the CSF of pre-terminally euthanized animals receiving the AAV-HuTg on the day of their unscheduled euthanasia compared with pre-study and control animals’ results (≤ 7 cells/µl). In addition, minimally to moderately increased CSF total protein (0.39-0.96 g/l) was observed in all pre-terminal animals receiving the AAV-HuTg when compared with both pre-study and control animals’ results (generally ≤ 0.23 g/l). Combined increases in CSF total nucleated cell counts and total protein were suggestive of an AAV-HuTg-related inflammatory process. At scheduled termination of animals receiving the AAV-HuTg, there was a moderate increase in CSF-nucleated cell count (58 cells/µl) predominantly due to increased number of lymphocytes and fewer macrophages. There were no remarkable CSF clinical chemistry changes in AAV-HuTg-dosed animals that survived to scheduled euthanasia. Clinical chemistry changes in animals receiving the AAV-RbTg were limited to increases in CSF total nucleated cell count and protein in only one animal at the highest dose level tested.
Administration of AAV-HuTg vector in both Studies 1 and 2 resulted in microscopic changes in the brain (predominantly brain stem/adjacent to injection site), spinal cord (predominantly dorsal funiculi and ventral horn), DRG and dorsal nerve roots, TG and trigeminal nerve, and sciatic and median nerves in all animals treated with AAV-HuTg and all time points (scheduled and unscheduled necropsies) (Table 2—Study 1; Table 3—Study 2). In Study 1, the brain (Figure 1), changes affected multiple nuclei in the midbrain, pons, and/or medulla oblongata and included minimal to moderate neuronal central chromatolysis and minimal neuronal degeneration/necrosis with minimal to mild gliosis and satellitosis. These findings were accompanied by minimal to mild nerve fiber degeneration, characterized by dilated axonal sheaths with or without phagocytic macrophages (gitter cells), located in close proximity to the injection site (cisterna magna), and usually affecting the spinal trigeminal tract and/or rubrospinal tract. Mononuclear cell infiltration, consisting predominantly of lymphocytes and fewer macrophages, were minimal to mild in severity and present in multiple brain sections and locations. These infiltrates were most often noted in the meninges and choroid plexus, or less frequently in the parenchyma within the Virchow-Robin (perivascular) space. In the spinal cord (Figure 2), minimal to moderate nerve fiber degeneration of the dorsal funiculi (always bilateral) was present at all levels (cervical, thoracic, and lumbar) with the highest severity in the lumbar segments. It was characterized by dilated axonal sheaths with or without phagocytic macrophages (gitter cells), with a dose-related trend in severity. Nerve fiber degeneration was also occasionally observed in ventral and/or lateral funiculi of the cervical and thoracic spinal cord segments. In the gray matter ventral horn, minimal gliosis and/or neuronal central chromatolysis was observed in both AAV-dosed groups (Study 1, Table 2). In the DRG with dorsal nerve root, minimal to moderate neuronal cell body central chromatolysis and degeneration/necrosis accompanied by minimal to marked mononuclear cell infiltration (Figure 3A-C) and dorsal nerve root nerve fiber degeneration were present at all spinal cord levels. Satellite glial cell proliferation often formed whorls around affected neurons or nests replacing lost neurons (nodules of Nageotte). The severity of DRG microscopic findings was dose dependent, with the highest severity score recorded in the lumbar segments. Findings in the DRG correlated well with nerve fiber degeneration of the dorsal funiculi in the spinal cord (Figure 4). In the TG and nerve (Figure 5A and 5B), minimal to moderate neuronal cell body central chromatolysis and degeneration/necrosis accompanied by minimal to marked mononuclear cell infiltration and trigeminal nerve fiber degeneration were present in both AAV-dosed groups with dose-dependent increase in severity and incidence. There was also satellite glial cell proliferation in TG that often formed whorls around affected neurons or nests replacing lost neurons in the high-dose group. In the sciatic nerve, mild to marked, bilateral nerve fiber degeneration accompanied by increased cellularity of Schwann cells was observed bilaterally in both AAV-dosed groups at similar incidence and severity (Figure 6A and 6B and Study 1, Table 2). In addition, mild to moderate, bilateral nerve fiber degeneration was noted in the median nerves.
Incidence and severity for histopathologic findings from Study 1.

Abbreviations: AAV, adeno-associated virus; GC, genome copy.
Numbers in parentheses represent the number of animals with the finding.
Nerve fiber degeneration noted in longitudinal section of spinal cord, thus affected funiculi not specified.
Incidence and severity for histopathologic findings from Study 2.

Abbreviations: AAV, adeno-associated virus; GC, genome copy; GFAP, glial fibrillary acidic protein; GLUL, glutamine synthetase GS-6.
Animal No. 3101 received 8.91 × 1011 GC.
Animal No. 5102 received 8.43 × 1012 GC.
Numbers in parentheses represent the number of animals with the finding.
Nerve fiber degeneration noted in longitudinal section of spinal cord.
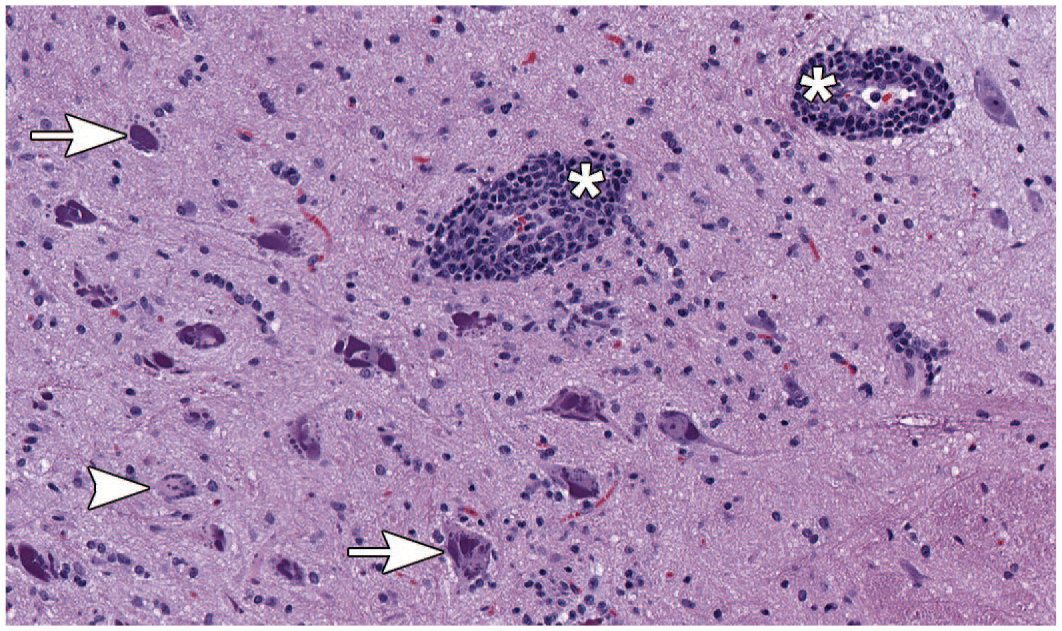
Microscopic image representative of changes observed in the brain (medulla oblongata). Animal dosed with 3 × 1013 GC/animal of AAV-HuTg with neuronal central chromatolysis (arrowhead) and purple granular material accumulation (arrow) accompanied by perivascular mononuclear cell cuffs (asterisks) in multiple nuclei of medulla oblongata (H&E staining; original objective 20X). AAV indicates adeno-associated virus; GC, genome copy.
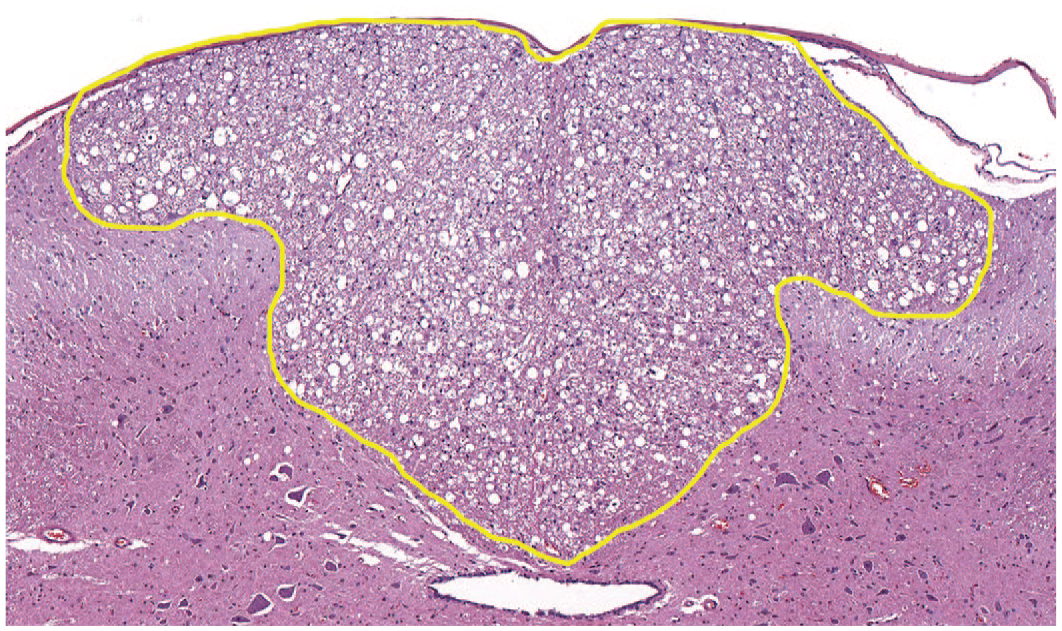
Microscopic image representative of changes observed in the spinal cord. Lumbar spinal cord (L3) cross-section of animal dosed with 3 × 1013 GC/animal of AAV-HuTg with marked, bilateral nerve fiber degeneration in the dorsal funiculi (outlined area) (H&E staining; original objective 7.7X). AAV indicates adeno-associated virus; GC, genome copy.
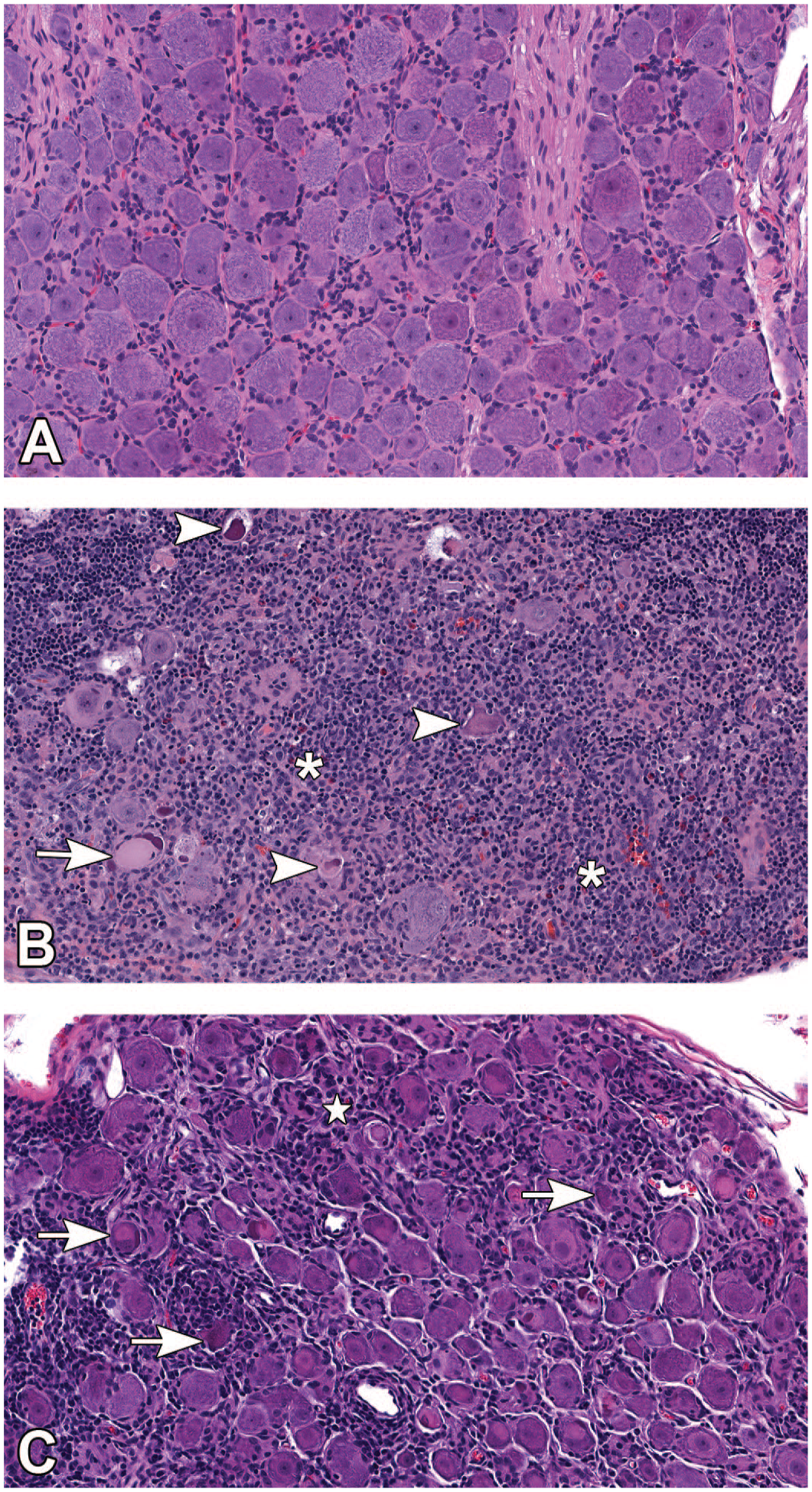
Microscopic images representative of changes observed in the DRG. (A) DRG from a control animal dosed with vehicle (H&E staining; original objective 20X). (B) Lumbar DRG (L3) of animal dosed with 3 × 1013 GC/animal of AAV-HuTg with moderate neuronal cell body degeneration/necrosis (arrowhead), mild neuronal central chromatolysis (arrow) and marked mononuclear cell infiltration/inflammation (asterisk) (H&E staining; original objective 20X). (C) Cervical DRG (C3) of animal dosed with 3 × 1013 GC/animal of AAV-HuTg. Moderate neuronal cell body degeneration/necrosis with neuronophagia (arrow), moderate mononuclear cell infiltration, and satellite glial cell proliferation (star) (H&E staining; original objective 20X). AAV indicates adeno-associated virus; DRG, dorsal root ganglia; GC, genome copy.
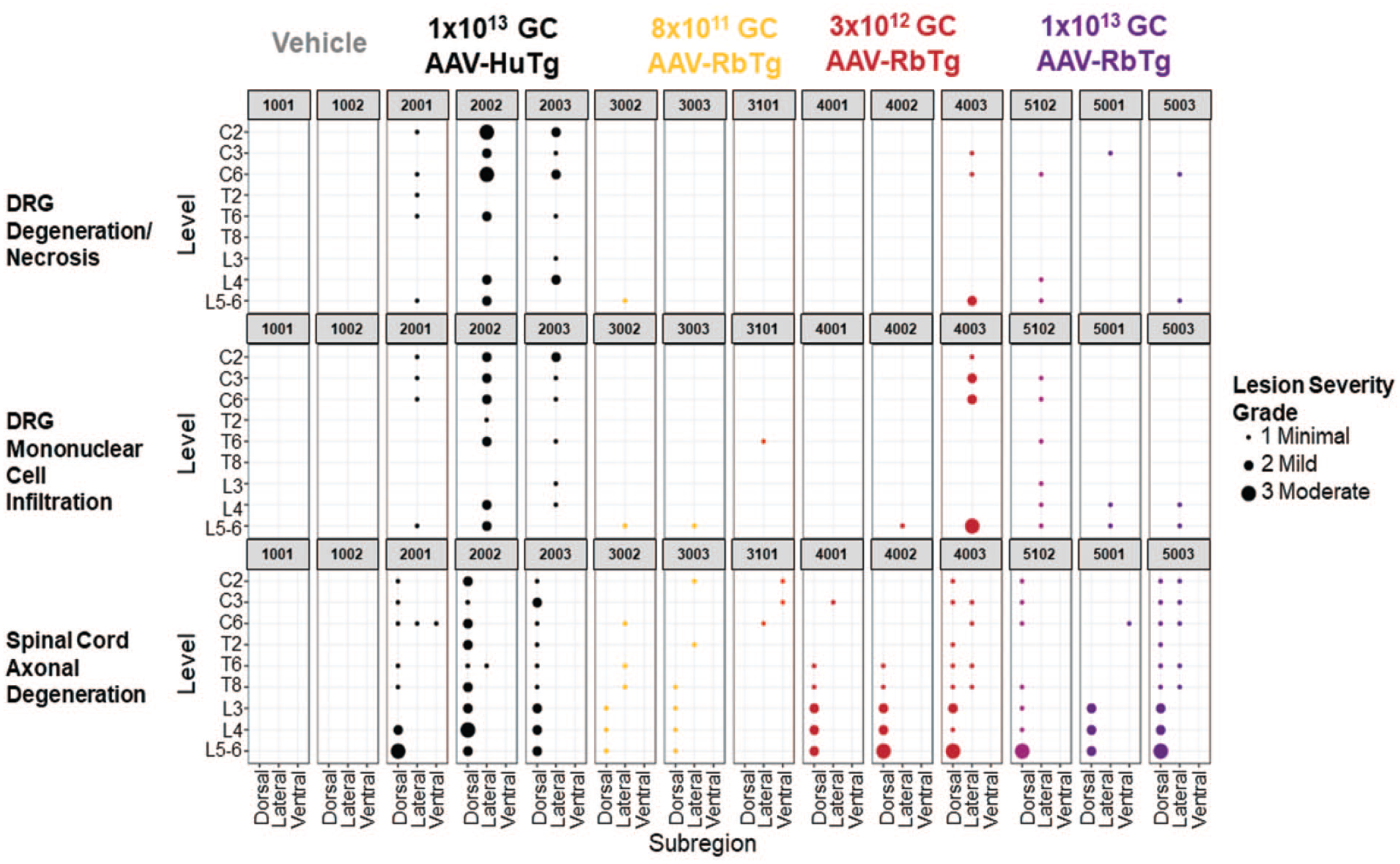
Study 2 Bubble Plot: DRG and spinal cord histopathology summarized across subregions of the spinal cord levels evaluated. Histopathology parameters presented in this bubble plot include DRG, neuron degeneration/necrosis; DRG, mononuclear cell infiltration; spinal cord, nerve fiber degeneration. Each bubble represents a lesion at the indicated location in each subject tested. The size of the bubble represents the lesion severity. AAV indicates adeno-associated virus; C, cervical; DRG, dorsal root ganglia; GC, genome copy; L, lumbar; T, thoracic.
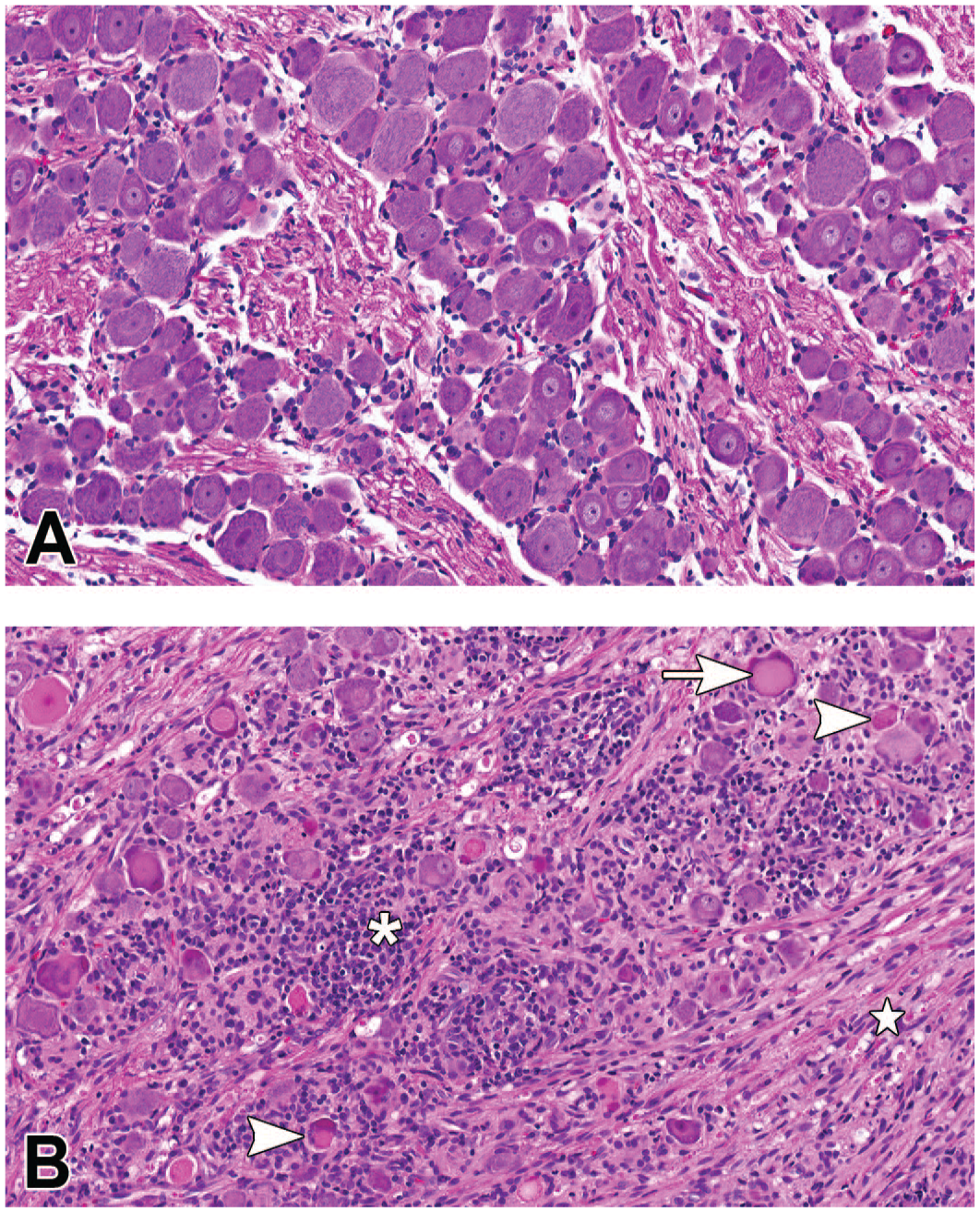
Microscopic images representative of changes observed in the trigeminal ganglia. (A) Trigeminal ganglia from a control animal dosed with vehicle (H&E staining; original objective 20X). (B) Trigeminal ganglia of animal dosed with 3 × 1013 GC/animal of AAV-HuTg with moderate mononuclear cell infiltration/inflammation (asterisk), neuronal cell body degeneration/necrosis (arrowhead), neuronal central chromatolysis (arrow) and nerve fiber degeneration of the trigeminal nerve (star) (H&E staining; original objective 20X). AAV indicates adeno-associated virus; GC, genome copy.
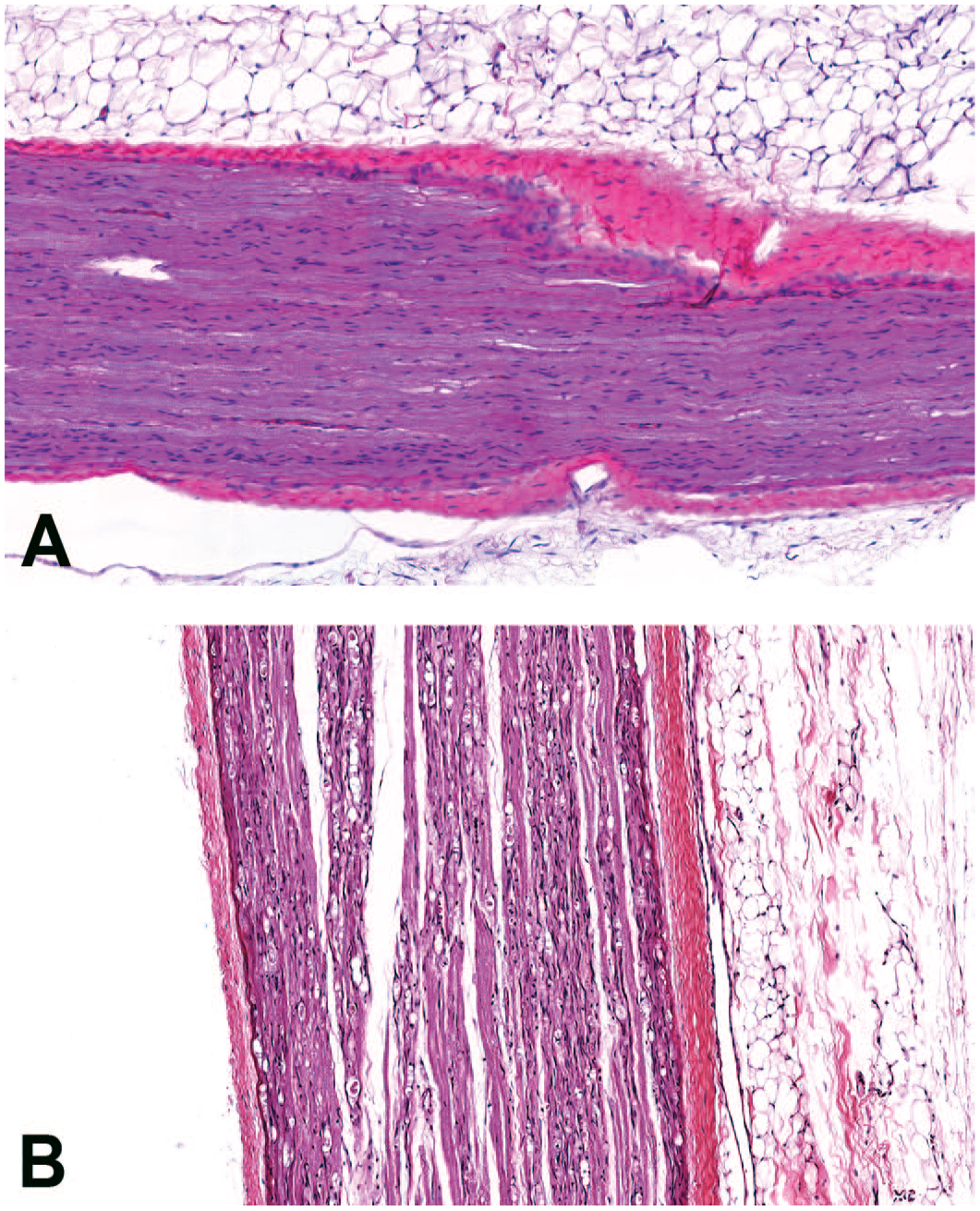
Microscopic image representative of changes observed in the sciatic nerve. (A) Sciatic nerve from a control animal dosed with vehicle (H&E staining; original objective 10X). (B) Sciatic nerve of animal dosed with 3 × 1013 GC/animal of AAV-HuTg with marked, bilateral nerve fiber degeneration (H&E staining; original objective 10X). AAV indicates adeno-associated virus; GC, genome copy.
In Study 2, administration of AAV-RbTg resulted in microscopic changes in the brain (predominantly medulla oblongata), spinal cord (predominantly dorsal funiculi), DRG and dorsal nerve roots, TG and trigeminal nerve, and sciatic and median nerves (Study 2, Table 3). Findings in the medulla oblongata consisted of minimal nerve fiber degeneration affecting the rubrospinal tract. In addition, there was minimal perivascular mononuclear cell infiltration, neuropil pallor suggestive of degeneration, and gliosis adjacent to the ICM injection site. In the spinal cord, minimal to moderate nerve fiber degeneration of the dorsal funiculi (always bilateral) was present at all levels (cervical, thoracic, and lumbar) with the highest severity in lumbar region and a dose dependent trend in severity (Figure 4). Nerve fiber degeneration was also occasionally observed in ventral and/or lateral funiculi without obvious dose relationship. Microscopic changes in the DRG and dorsal nerve roots (cervical, thoracic, and lumbar) were observed at all dose levels and consisted of minimal to mild neuronal degeneration/necrosis and minimal decrease in neuronal cellularity accompanied by minimal to mild mononuclear cell infiltration and moderate dorsal nerve root nerve fiber degeneration. Findings in the DRG correlated well with nerve fiber degeneration of the dorsal funiculi in the spinal cord (Figure 4). TG findings consisted of minimal to mild neuronal degeneration/necrosis with minimal decrease in neuronal cellularity and minimal to moderate mononuclear cell infiltration. These findings were present at all dose levels with a dose-dependent incidence and severity (Study 2, Table 3). In the trigeminal, median, and sciatic nerves, there was nerve fiber degeneration with increased Schwann cell cellularity at all dose levels. The incidence and severity of findings in the trigeminal and sciatic nerves was dose dependent (Study 2, Table 3).
In addition to the routine H&E stain, IHC staining against GFAP and GLUL, 13 both known markers for satellite glial cells, were performed on all DRG for all animals in Study 2 only to better distinguish satellite glial cells from mononuclear cell infiltrates. GLUL IHC signal intensity in the immunopositive satellite glial cells appeared more consistent across all groups including controls when compared with the GFAP IHC signal intensity; therefore, only GLUL IHC was used to assess AAV-related increased cellularity of these cells (Table 3). Minimal increase in satellite glial cell cellularity was present in one or multiple DRG samples in rabbits administered AAV-HuTg at 1 × 1013 GC/animal and AAV-RbTg at all dose levels. This increase in satellite glial cell cellularity was characterized by the presence of satellite glial cells piling up and surrounding affected neuronal cell bodies. GFAP IHC signal intensity in the immunopositive satellite glial cells showed much more variation, that is, increased signal intensity (which often coincided with enlarged cytoplasm) in AAV-HuTg- or AAV-RbTg-dosed groups compared with controls, suggesting activation of satellite glial cells by treatment.
TEM was conducted in Study 2 only on cervical and lumbar DRGs from animals dosed with vehicle, AAV-HuTg (1 × 1013 GC) and AAV-RbTg (1 × 1013 GC). Ultrastructural findings observed in the DRG sensory neurons included more densely packed cytoplasmic structures compared with concurrent vehicle control consistent with light microscopic presentation of shrunken neurons; mitochondrial swelling; dilated endoplasmic reticulum; and/or nucleolus fragmentation. Increased numbers of autophagolysosomes (i.e., lamellar bodies) in the sensory neuron cell bodies and axons were also observed in animals dosed with either AAV-HuTg or AAV-RbTg compared with the control animal (Figure 7A-D). All of these ultrastructural findings were consistent with sensory neuron degeneration/necrosis. Additional ultrastructural findings observed included dilatation of the endoplasmic reticulum in Schwann cells or satellite glial cells.
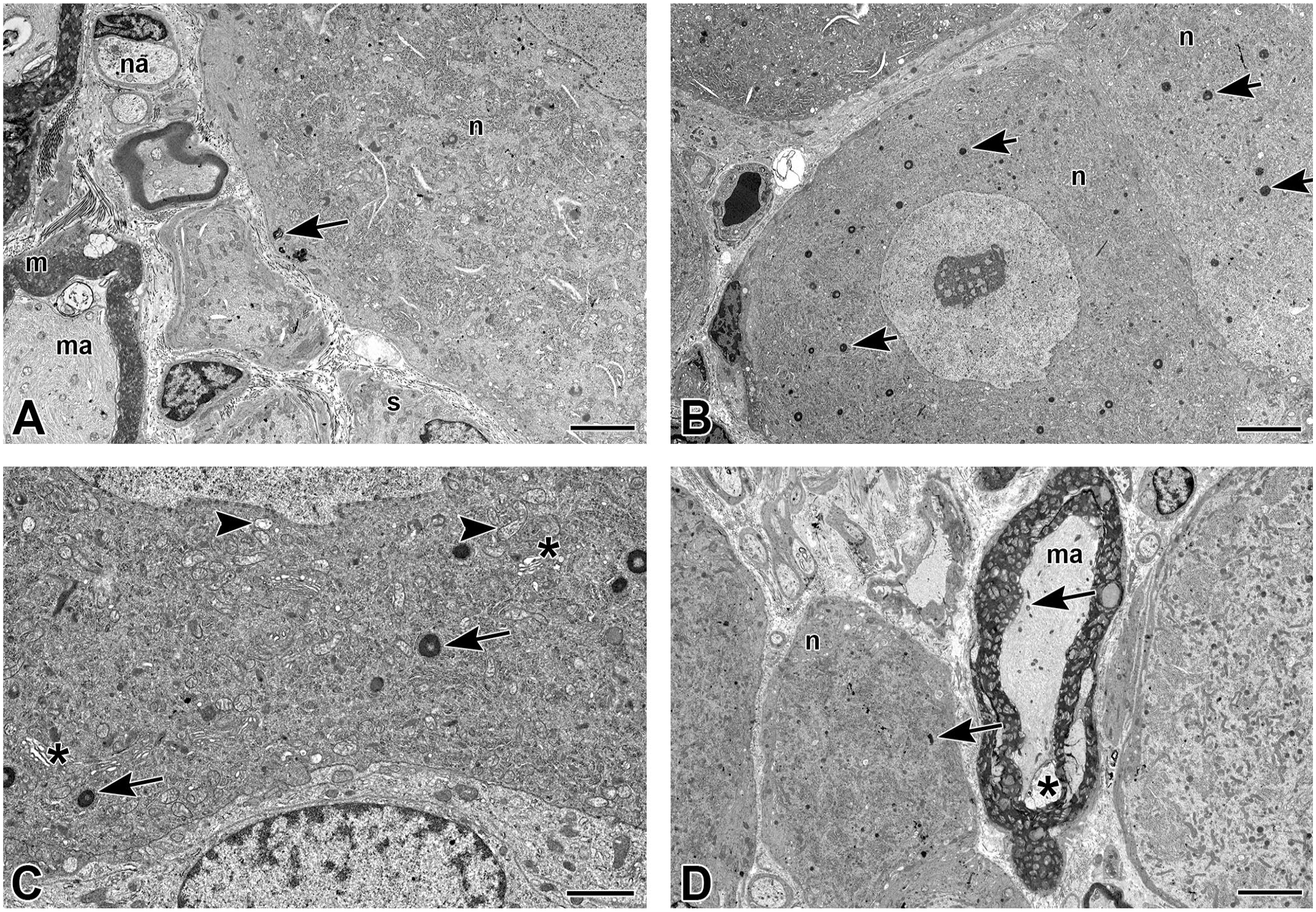
Transmission electron microscopy images representative of changes in the DRG. (A) DRG (L3) from a control animal. Sensory neuron and axons. The cytoplasmic organelles were without obvious changes; there was a single focal intracytoplasmic lamellar body (arrow) in a sensory neuron (n). ma = myelinated axon; m = myelin; na = nonmyelinated axon; s = satellite glial cell. (Direct Mag: 6320X; scale bar = 4 µm). (B) DRG (L3) of animal dosed with 1 × 1013 GC/animal of AAV-HuTg. Sensory neurons (n) with more densely packed cytoplasmic structures than in the control animal DRG, consistent with light microscopic presentation of shrunken neurons. In addition, numerous lamellar bodies (arrow) were observed in the cytoplasm of these sensory neurons. (Direct Mag: 4210X; scale bar = 6 µm). (C) DRG (L3) of animal dosed with 1 × 1013 GC/animal of AAV-HuTg. Sensory neuron with densely packed cytoplasmic structures. Numerous mitochondria (arrowhead) were swollen with peripherally placed to disintegrating cristae, membrane remnants, and/or occasional dense bodies. Endoplasmic reticulum (asterisk) was dilated to vesiculated. Intracytoplasmic, compact, lamellar bodies (arrow) were variable in size and ranged from a few to numerous. (Direct Mag: 13,100X; scale bar = 2 µm). (D) DRG (C6) of animal dosed with 1 × 1013 GC/animal of AAV encoding the AAV-RbTg. Scattered sensory neurons (n) with densely packed cytoplasmic structures and numerous lamellar bodies (arrow) in the neuronal cytoplasm and axons (ma). Artifact of tissue fixation/processing was noted in the myelin sheath (asterisk). (Direct Mag: 4210X; scale bar = 6 µm). AAV indicates adeno-associated virus; DRG, dorsal root ganglia; GC, genome copy.
Finally, liver microscopic findings were observed in Study 2 but not in Study 1 animals. Liver findings consisted of minimal periportal mixed cell infiltration (1/3 animals dosed with AAV-HuTg at 1.104 × 1013 GC/animal and 1/3 animals dosed with AAV-RbTg at 8.43 × 1012 GC/animal), minimal periportal fibrosis (1/3 animals dosed with AAV-RbTg at 8.43 × 1012 GC/animal) and minimal periportal pigmented macrophages (1/3 animals dosed with AAV-HuTg at 1.104 × 1013 GC/animal and 2/3 animals dosed with AAV-RbTg at 1.15 × 1013 GC/animal). There was no evidence of hepatocellular injury.
Vector Genome Biodistribution of AAV Encoding Human Transgene
Motor cortex, brain stem (Levels 8, 9, and 10), spinal cord (C4, T10, and L2), DRG (C4, T10, and L2), and liver (right and left) of New Zealand white rabbits after ICM administration of AAV-HuTg (1 × 1013 and 3 × 1013 GC) from Study 1 were collected for viral vector genome biodistribution evaluation. Dose-response in viral genome concentration was observed in all spinal cord regions, while this effect was less clear in other collected tissues including DRGs (Figure 8A and 8B) though it is acknowledged that some animals were euthanized early. In DRGs, AAV-HuTg viral genome concentration ranged from ~1/10th (high-dose group) to ~1/2 levels (low-dose group) of that observed in the spinal cord regions, but it was equal or higher (> 1.07 GC/DG) than the levels observed in the liver and motor cortex samples (Figure 8B).
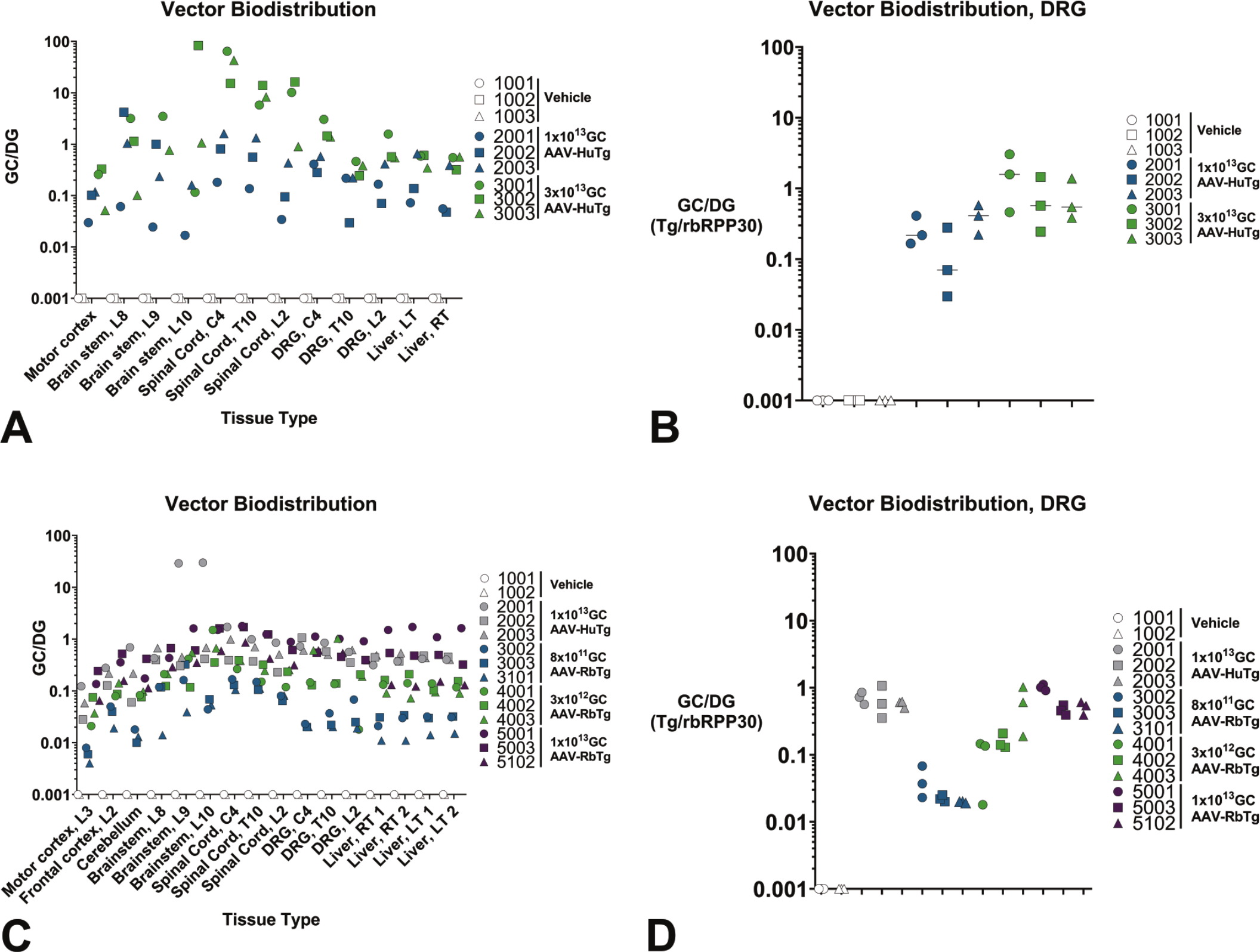
Vector genomic copies per diploid genomes. Vector genome copies per diploid genomes in central and peripheral nervous system tissues, and peripheral tissues at terminal time points in Study 1 (A and B) ranging from 12 to 29 days post-administration of AAV-HuTg at 1 × 1013 (animals 2001-2003) or 3 × 1013 GC (animals 3001-3003), or Study 2 (C and D) at Day 29 post-administration of either AAV-HuTg at 1 × 1013 GC (animals 2001-2003) or AAV-RbTg at ~8 × 1011 (animals 3002, 3003, 3101), 3 × 1012 (animals 4001-4003), or 1 × 1013 GC doses (animals 5001, 5003, 5102). Each symbol represents an individual sample from an individual animal and colors indicate separate AAV transgene and/or doses. AAV indicates adeno-associated virus; GC, genome copy, DG, diploid genome.
Vector Genome Biodistribution and Bulk Tissue Transgene RNA Expression of AAV Encoding Rabbit Transgene
Motor cortex (L3), frontal cortex (L2), cerebellum, brain stem (L8, L9, and L10), spinal cord (C4, T10, and L2), DRG (C4, T10, and L2), and liver (2 samples from the right and left lobes) of New Zealand white rabbits after ICM administration of AAV-HuTg (1 × 1013 GC) or AAV-RbTg (~8 × 1011, ~3 × 1012, or ~1 × 1013 GC) from Study 2 were collected for viral vector genome biodistribution evaluation. A dose-response was observed in all tissues collected, including in the DRGs (Figure 8D). AAV-HuTg (1 × 1013 GC) and AAV-RbTg (1 × 1013 GC) demonstrated similar biodistribution, ranging from 0.028 to 1.064 GC/DG (with the exception of two brain stem samples from animal 2001) in the AAV-HuTg (1 × 1013 GC) group and from 0.136 to 1.455 GC/DG in the AAV-RbTg (1 × 1013 GC) group. Motor cortex (L3), frontal cortex (L2), cerebellum, brain stem (L8, L9, and L10) and two liver samples from the right and left lobes were collected for transgene expression analysis. AAV-RbTg expression was dose-dependent in all tissues tested (Figure 8C and 8D). RNA expression levels in CNS tissue ranged from 59 fold to 1.9 × 107 fold, with the highest RNA expression detected in the two brain stem samples with the highest vector genome biodistribution. RNA expression in the peripheral organs (i.e., liver) was lower than in the CNS, ranging from 1 fold to ~2000 fold expression when compared with vehicle controls.
Transgene RNA Expression in DRG and Spinal Cord Cells of AAV Encoding Human Transgene
In Study 1 histologic sections of cervical, thoracic and lumbar DRGs (Figure 9A), rabbit DRG neurons, but not satellite glial cells, were transduced with AAV (expressed HuTg RNA). Percent neuron transduction was 97% and 99% in the 1 × 1013 GC and 3 × 1013 GC dose groups, respectively (Figure 9B). Frequency histograms of transgene reactivity densities in DRG neurons were similar in all spinal regions (Figure 9C). In the spinal cord, a modest dose-related trend in somatic motor neuron transduction was observed, estimated at 84% in the 1 × 1013 groups and 91% in the 3 × 1013 group. Percent motor neuron transduction trended lower in lumbar spinal cord compared with cervical and thoracic spinal cord. A modest right shift in mean motor neuron transgene HuTg reactivity density was observed in the 3 × 1013 dose group compared with the 1 × 1013 dose group (data not shown). This information was not generated for Study 2.
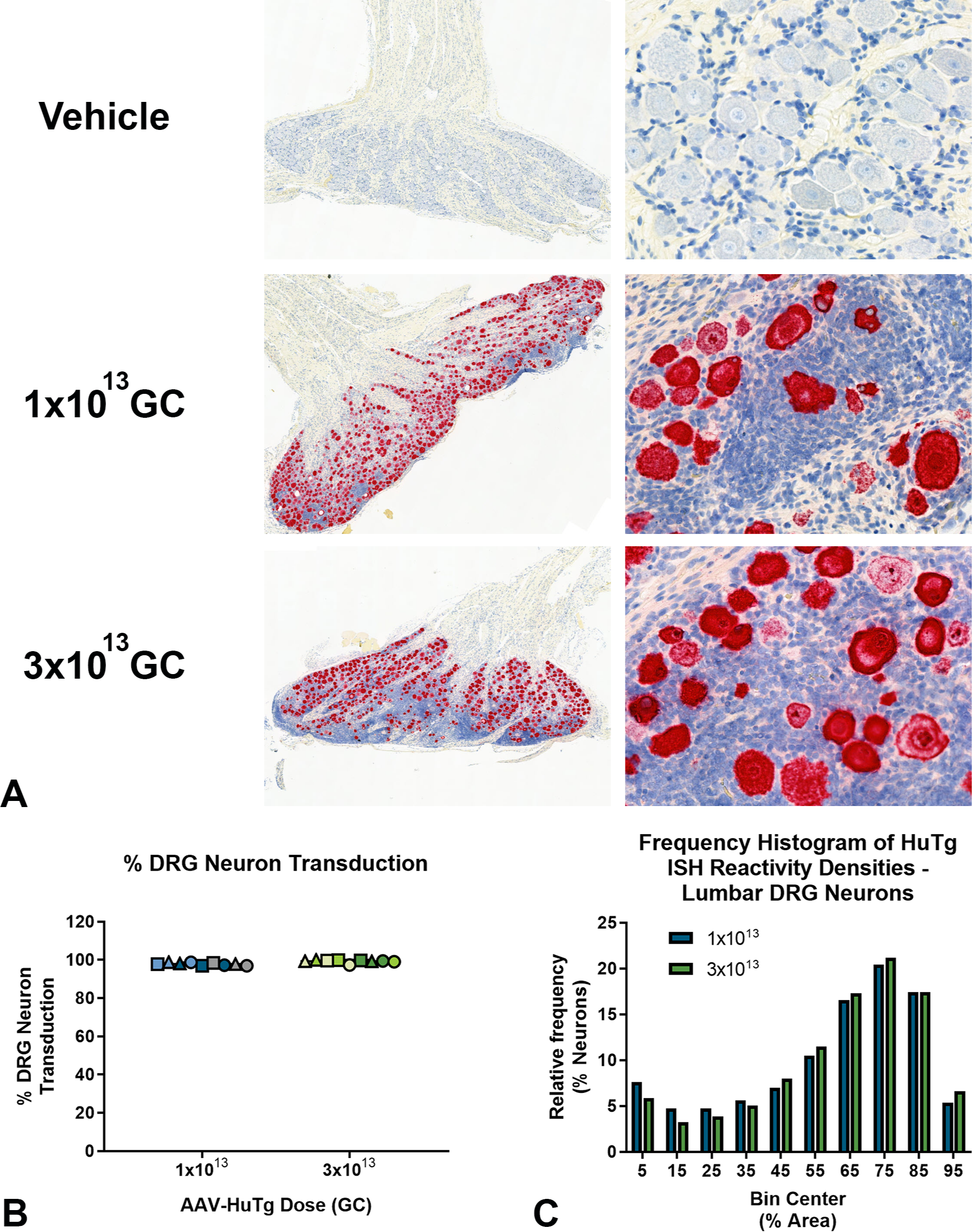
Human transgene ISH reactivity in DRG. (A) From Study 1, Top—Representative image from a reference animal lumbar DRG showing no reactivity with the HuTg ISH probes. Middle—Representative DRG image from an animal dosed with 1 × 1013 GC showing abundant DRG neurons that are densely reactive with the HuTg ISH probes among proliferations of mononuclear cells. Bottom—Representative DRG image from an animal dosed with 3 × 1013 GC showing abundant DRG neurons that are densely reactive with the HuTg ISH probes among more confluent proliferations of mononuclear cells compared with the middle panel. Original image magnifications: left column, 2X; right column, 40X. (B) From Study 1, bar plot demonstrating 97% and 99% DRG neuron transduction in the 1 × 1013 and 3 × 1013 dose groups, respectively. Lumbar DRGs are denoted by circles, thoracic DRGs by triangles and cervical DRGs by squares. Data points from spinal regions of individual animal donors are denoted by matching colors. (C). From Study 1, frequency histogram of individual lumbar DRG neuron HuTg reactivity densities are similar between 1 × 1013 and 3 × 1013 GC dose groups. Overall, the findings indicate no differences between AAV-transgene transduction among residual DRG neurons in the 1 × 1013 and 3 × 1013 GC/dose groups. AAV indicates adeno-associated virus; DRG, dorsal root ganglia; GC, genome copy; ISH, in situ hybridization.
Transgene RNA Expression in DRG Neurons of AAV Encoding Rabbit Transgene
For Study 2, due to cross-reactivity of the rabbit transgene probe with endogenous mRNA, we did not estimate % DRG neuron transduction by AAV-RbTg. Rather, we compared the mean RbTg probe reactivity density in DRG neurons (Figure 10A), expressed as % cell area reactive for RbTg reactivity, across treatment groups. We found increases in mean DRG neuron RbTg reactivity density, estimated at 4, 33 and 63%, for the 8 × 1011, 3 × 1012, and 1 × 1013 GC dose groups, respectively. Compared with rabbits dosed with AAV-HuTg at 1 × 1013 GC, mean DRG neuron Tg reactivity densities were lower in the groups administered AAV-RbTg at 8 × 1011 GC (P < .0001) and 3 × 1012 GC (P < .001), but was not significantly lower in rabbits administered AAV-RbTg at 1 × 1013 GC (P = .19) (Figure 10B).
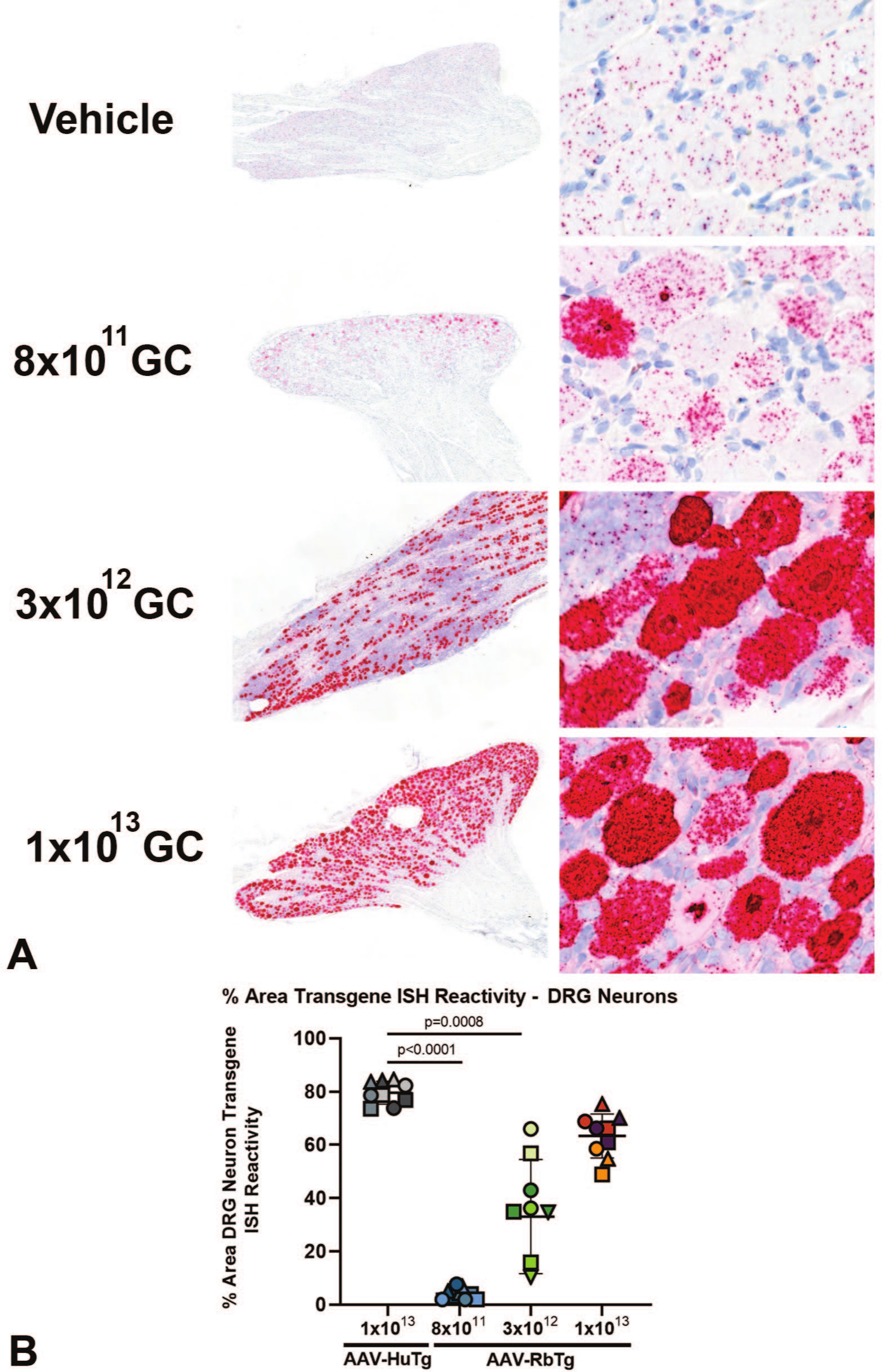
Rabbit transgene ISH reactivity in DRG. (A) From Study 2, Top—Representative image from a reference animal lumbar DRG showing DRG neurons with uniform endogenous reactivity with the RbTg probe. Other images—Representative DRG images from animals dosed with 8 × 1011, 3 × 1012, and 1 × 1013 GC showing DRG neurons with dose-related increases in RbTg ISH probe reactivity among increased numbers of mononuclear cells. Original image magnifications: left column, 2X; right column, 40X. (B) From Study 2, bar plot demonstrating dose dependent DRG neuron RbTg ISH probe reactivity in the AAV-RbTg dose groups, respectively, compared with HuTg ISH probe reactivity in the AAV-HuTg dose group. Lumbar DRGs are denoted by circles, thoracic DRGs by triangles and cervical DRGs by squares. Data points from spinal regions of individual animal donors are denoted by matching colors. AAV indicates adeno-associated virus; DRG, dorsal root ganglia; GC, genome copy; ISH, in situ hybridization.
Evaluation of Serum NF-H
Serum from Study 2 was collected from all subjects at baseline, 12 hours, and 2, 5, 9, 15, and 29 days post-ICM injection of vehicle or AAV administration with AAV-HuTg (1 × 1013 GC) or AAV-RbTg (8 × 1011, 3 × 1012, and 1 × 1013 GC) to evaluate NF-H levels, as a biomarker of neuronal damage mediated by AAV dosing. All ICM-dosed groups showed an increase in serum NF-H compared with their baseline level, which may be reflective of damage caused by the ICM injection. For AAV-dosed subjects, serum NF-H showed a dose-dependent increase in both magnitude and kinetics (Figure 11). Elevations of serum NF-H above vehicle controls were first detected at Day 9 post-AAV administration and were most evident in the AAV-HuTg (1 × 1013 GC)-dosed group (with serum neurofilament 12,711 ± 6340 pg/ml in AAV-HuTg-dosed vs 1286 ± 352 pg/ml in vehicle-dosed controls). Serum NF-H increased over time in all AAV-dosed groups, peaking on Day 29. Given that the NF-H ELISA is designed against human NF-H, these values may be underestimates in rabbit. Nonetheless, the kinetics of NF-H elevation in rabbit are similar to that observed in NHPs following intra-CSF AAV administration and suggest that this may also serve as a biomarker of AAV-mediated toxicity in rabbits.
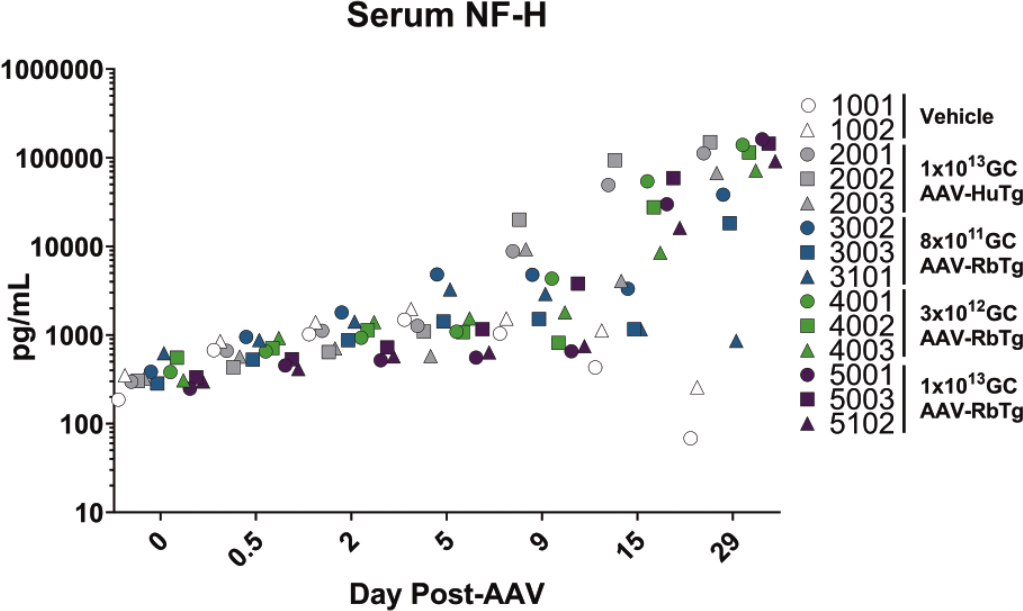
Serum neurofilament heavy levels. Measured in vehicle (white symbols) or AAV-dosed subjects at Days 0, 0.5, 2, 5, 9, 15, and 29 post-AAV administration of AAV-HuTg (1 × 1013 GC; animals 2001-2003; gray symbols) or AAV-RbTg (8 × 1011 (animals 3002, 3003, and 3101) 3 × 1012 (animals 4001-4003), or 1 × 1013 GC (animals 5001, 5003, and 5102); green, blue, and purple symbols, respectively). AAV indicates adeno-associated virus; GC, genome copy.
Discussion
In this investigation, adult male New Zealand white rabbits were dosed via a single ICM injection of AAV at doses comparable with those used in NHP that demonstrate DRG degeneration when scaled for brain weight. A dose of 3 × 1013GC/animal with an AAV harboring a transgene for a human protein was not tolerated in rabbits. Based on brain weight, this would equate to about a 2 × 1014GC/animal dose in NHPs that, while high for this species, has been demonstrated in previous studies can be tolerated based on clinical signs in NHPs, 1 though it varies depending on the transgene cargo. No immune suppression regimens were tested in these studies. In rabbits, human and rabbit transgenes were tolerated at 1 × 1013GC/animal (equivalent to about 5-7 × 1013GC/animal in NHPs). In the interest of animal welfare, a dose level higher than 1 × 1013 GC/animal of the AAV-RbTg was not conducted. Histopathologic features, incidence and severity, lack of tolerability and presence of clinical signs indicate that rabbits are at least equally, if not more, sensitive to AAV-mediated DRG and TG toxicity as NHP macaques.
Both the rabbit and human proteins demonstrated DRG toxicity in the form of neuronal degeneration/necrosis accompanied by mononuclear cell infiltration in the DRGs and nerve fiber degeneration in the spinal cord and nerves (dorsal nerve root and peripheral nerves). AAV-HuTg resulted in greater incidence and severity of these lesions versus AAV-RbTg when administered at an equivalent dose of 1 × 1013 GC. This difference may be attributed to (1) slightly increased expression of the human transgene versus rabbit transgene (Figures 8 and 9), (2) codon optimization that may result in protein misfolding and endoplasmic reticulum stress, and/or (3) an immune response to the non-self human transgene. However, we acknowledge more work is needed in this area to disentangle these possibilities.
Interestingly, the cervical and lumbar regions tended to exhibit the most severe findings while the thoracic was largely less affected in both species. The cause of this distribution of the lesions is unclear though it has been observed with AAVs administered by both the ICM (as in these studies) and IT lumbar routes of injection. 4 Morphologically, the presentation of the DRG toxicity in rabbits is very similar to that observed in rhesus and cynomolgus NHPs. 4 The hallmark characteristics in common between both species include neuronal cell body degeneration/necrosis accompanied by mononuclear cell infiltration and nerve fiber degeneration. Furthermore, as has been observed in NHPs, serum NF-H is elevated in rabbits following AAV administration in a time- and dose-dependent manner, with elevations occurring as early as Day 9 and increasing until the endpoint at Day 29 (Figure 11). Finally, brain stem findings consisting of neuronal degeneration/necrosis accompanied by gliosis and nerve fiber degeneration are observed in both rabbits and monkeys. The location of these findings in the immediate proximity to the injection site (i.e., cisterna magna) suggests multifactorial etiology, including inadvertent direct injection of the test article into the brain stem, high exposure to the test article from the cisterna magna, and/or lesion extension from the TG/nerve. Other preclinical species, most notably the rat and the minipig, also can replicate this finding though the temporal nature and the clinical signs vary greatly between all species1,4,10 suggesting that each species may have utility in different situations.
Some of the main differences observed between NHPs and rabbits dosed with AAV gene therapy is the higher severity of lesions observed microscopically in the rabbit TG/nerve compared with that of NHPs at comparable dose levels and the more severe clinical signs observed in rabbits compared with NHPs. This microscopic finding may have led to reduced sensation of oral cavity and tongue with subsequent inadvertent injury of the tongue and inability to eat; clinical signs that were observed in rabbits but not in NHPs. In addition, satellite glial cells are only minimally transduced in rabbit (Figure 7A-D). It is unclear whether this difference in cell tropism in the rabbit is due to differences in receptor expression or in other cellular mechanisms required for successful transduction (i.e., endosomal escape, nuclear trafficking) in rabbit satellite glial cells. Unfortunately, these studies were not designed to address these differences and require further investigation. However, the lack of transduction of rabbit satellite glial cells suggests that they may not be a major driver of the lesions observed, at least in rabbits. Finally, despite similar levels of vector genome biodistribution in the DRGs and liver, there was lower transgene expression in the liver than the DRGs in the rabbit model (Figures 8A-D and 12). In contrast, transgene expression in the liver is typically comparable or higher than in DRGs in NHPs when using a ubiquitous promoter, even when vectors are delivered via ICM/IT. This can result in liver injury in NHPs, as indicated by elevated liver enzymes that correspond to pathology. Consistent with a recent report that transgene expression is required for AAV-mediated hepatotoxicity in NHP, 5 lower transgene expression in the liver in rabbit may have mitigated this toxicity.
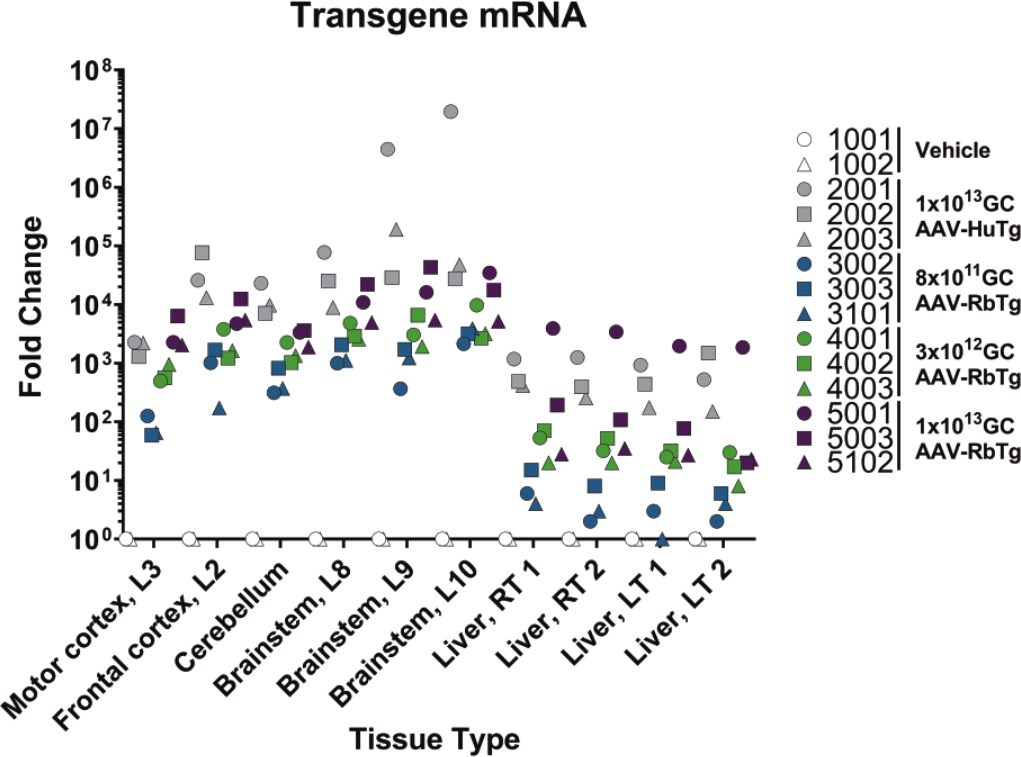
Fold change RNA expression in central nervous system and peripheral organs. Fold change RNA expression in central nervous system and peripheral organs at Day 29 in Study 2 following either AAV-HuTg at 1 × 1013 GC (animals 2001-2003) or AAV-RbTg at ~8 × 1011 (animals 3002, 3003, and 3101) 3 × 1012 (animals 4001-4003), or 1 × 1013 GC doses (animals 5001, 5003, and 5102). Determined by qPCR performed with human and rabbit transgene-specific probes. Transgene expression was normalized to rabbit RPPP30. Each symbol represents an individual subject and colors indicate separate AAV transgene and/or doses. AAV indicates adeno-associated virus; GC, genome copy; RNA, ribonucleic acid; qPCR, quantitative polymerase chain reaction.
An exploration of the mechanism behind AAV-mediated DRG toxicity in rabbits is left for subsequent investigation. Previous publications have begun to uncover this relationship1,4-6 suggesting a potential link to cargo expression but the finer details of this mechanism remain open for continued work.
Herein, we report that the rabbit exhibits DRG toxicity with very similar pathological characteristics to NHPs indicating rabbits can be an alternative in vivo model for this lesion caused by AAVs. Similar to NHPs, rabbits dosed with an AAV human or rabbit protein transgene exhibited sensory neuron degeneration/necrosis with mononuclear cell infiltration and nerve fiber degeneration across multiple nervous tissues (e.g., the DRG, TG, brain, spinal cord, nerves). These results indicate that rabbits may provide another avenue to investigate mechanisms of AAV-mediated DRG toxicity and screen novel AAV technologies mitigating these adverse effects as the gene therapy field continues to grow and evolve.
Footnotes
Acknowledgements
The authors would like to thank Walter Bobrowski for helping with image and figure formatting for this work.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors were employed by Biogen (except for Claudine Tremblay) at the time of their contribution to this work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Biogen.
Ethical Approval
The care and use of the animals on these studies were conducted in accordance with the guidelines of the U.S. National Research Council Canadian Council on Animal Care and at AAALAC-accredited facilities.
