Abstract
Introduction
Painful peripheral neuropathy is a debilitating disorder, characterized by a reduced quality of life, which can lead to neuropathic pain in a subset of patients. 1 Small fiber neuropathy (SFN), a disorder characterized pathologically by damage to the smallest axons of the peripheral nervous system, occurs as an accompaniment of metabolic disorders, such as diabetes, exposure to toxins, and cancer chemotherapy. 2 Chemotherapy induced peripheral neuropathy (CIPN) is a critical side effect in patients receiving antitumor agents, such as bortezomib, antitubulin, and oxaliplatin.3,4 In most CIPN patients, pain and other sensory abnormalities appear first in the territories dominated by the longest nerve fibers, which leads to the term “stocking-glove” neuropathy. 2 Despite a large numbers of studies have study the mechanisms of CIPN, there is still no approved drug for treatment or prevention of CIPN.
Oxaliplatin is commonly used platinum-based chemotherapy drug for colorectal cancer, which has better efficacy and lower toxicity than cisplatin and carboplatin.5,6 Oxaliplatin-induced peripheral neuropathy (OIPN) can be divided into acute and chronic types. The main clinical features of acute OIPN were cold sensitive sensory symptoms and neuropathic pain in limbs. In addition to the above symptoms, chronic OIPN also produces autonomic nerve dysfunction. 6 Previous studies showed different mechanisms of OIPN, which might be related with its neurotoxicity through increasing the expression of NR2B containing N-methyl-d-aspartate receptor (NMDAR) and phosphorylation of calcium/calmodulin-dependent protein kinase II (CaMKII) in the spinal cord,7,8 degeneration of myelinated fibers in sciatic nerve, and shrinkage of dorsal root ganglion (DRG) neurons,9,10 inducing oxidative stress and inflammation in the spinal cord. 11
Recently, astragaloside IV (AS-IV), a saponin extracted from the root of Huangqi (Astragalus membranaceus (Fisch.) Bunge), has shown protective effects on diabetic peripheral neuropathy.12,13 In addition, Mannelli and his colleagues have found astragali radix extracts showed protective effects against oxaliplatin induced lipid peroxidation to reduce neurotoxicity in vitro. 14 For hydrogen peroxide (H2O2) induced retinal ganglion cells (RGC) injury, AS-IV treatment decreased the reactive oxygen species levels and protected RGC from oxidative stress injury. 15 However, the role of AS-IV on the OPIN remains unclear.
In this research, we hypothesized that AS-IV played neuroprotective effects on OIPN. A rat peripheral neuropathy model induced by repeated and prolonged administration of oxaliplatin was established to assess the AS-IV treatments on changes of behaviors, sciatic nerve, and DRG.
Material and methods
Animals and ethics
Forty female and male Sprague Dawley rats, weighting 200–220g, were used for this study, which were obtained from Pengyue Experimental Animal Breeding Co,Ltd (SYXK(Lu) 20180030, Jinan, China). Animals were housed in temperature (23 ± 2)°C, humidity (55 ± 5)%, 12-h light/dark, with free food and water.
All animal experiments were performed with the guidelines of National Institute of Health for the Care and Protection of Laboratory Animals (NIH, 1996). This study was examined and approved by the Yantaishan Hospital Animal Protection and Use Committee (Approval No. 20210017).
Drug administration
Oxaliplatin (4 mg/kg, 200,120AM, Jiangsu Hengrui Pharmaceutical Co, Ltd, China) dissolved with 5% glucose, was injected intraperitoneally twice a week for 4 weeks to establish an OIPN rat model. 16 The injection time was 1, 2, 8, 9, 15, 16, 22, and 23 days. As a control, rats were injected with equal volume of 5% glucose at the same time.
AS-IV (CAS:84,687-43-4,cat no, S31401,Yuanye, Shanghai) dissolved with 0.1% dimethyl sulfoxide and diluted with saline to working concentrations, was orally treated with OIPN rats once a day for 4 weeks at the beginning of oxaliplatin administration.17,18
Animal groups
All rats were randomly divided into five groups (8 each group), including control group, OIPN model (model) group, low dose of AS-IV (AS-IV-low, 5 mg/kg) group, medium dose of AS-IV (AS-IV-medium, 10 mg/kg) group, high dose of AS-IV (AS-IV-high, 20 mg/kg) group. In the control group, rats were injected with the same volume of 5% glucose twice a week for 4 weeks. In the model group, rats were also orally treated with the same volume of 0.1% dimethyl sulfoxide once a day for 4 weeks at the beginning of oxaliplatin administration.
Assessment of mechanical and cold allodynia
For each animal, mechanical allodynia was assessed before the beginning the treatment time, and after treatment 1, 2, 3 and 4 weeks by von Frey test. A series of Von Frey hairs from 1.0 to 50 g within 20 s were used to stimulate the hind paw of the rats. The hind paw having a quick licking or withdrawal on the stimulus for about 5 s is considered a positive response. In the absence of response, a 30 s cut-off time was used to prevent tissue damage. The applied force (g) was recorded as mechanical withdrawal threshold with mean thresholds.
After mechanical allodynia for 2 h, the cold allodynia of each animal was measured by acetone test. The evaluation time was before the beginning the treatment time, and after treatment 1, 2, 3, and 4 weeks. A droplet of acetone was felled on the plantar surface of the hind paw of each rat through metallic mesh floor. The amount of time (seconds), rats spent licking, cowering, or shaking the acetone treated hind paw, was observed during the following 60 s. Acetone was applied twice with an interval of 5 min. The average time of each animal was used for analysis.
Enzyme linked immunosorbent assay (ELISA)
At the end of treatment, the rats were euthanized with 3% pentobarbital sodium (150 mg/kg) intraperitoneally. Then, venous plasma was collected and put into sodium citrate vacuum vein tube. The blood serum was collected at 4°C, 2500 g for 15min. The levels of nerve growth factor (NGF, 2007M, MEIMIAN, Jiangsu, China), tumor necrosis factor-α (TNF-α, MM-0180R2, MEIMIAN, Jiangsu, China) and interleukin-6 (IL-6, MM-0190R2, MEIMIAN, Jiangsu, China) were obtained by an enzyme reader (version 352, Labsystems Multiskan MS).
Immunofluorescence
At the end of treatment, the spinal cord was obtained from each rat for immunofluorescence. The samples were fixed with 4% paraformaldehyde (C11100022, Macklin, Shanghai, China) for 24 h, then embedded with paraffin. The sections (3 μm) were placed in a 0.1 M sodium citrate buffer solution for 30 min at 95°C. Then, the sections were blocked with 1% bovine serum albumin and 0.1% TritonTM X-100 for 60 min. The ionized calcium binding adapter molecule 1 (Iba-1) antibody (1:300, #171985, Cell Signaling Technology) was added to incubate for overnight at 4°C. The secondary antibody anti-rabbit IgG HRP-linked antibody (1:1000, #7074, Cell Signaling Technology) was incubated for 2 h in the dark. Then, DAPI was used to stain for 10 min in the dark. The results were observed by laser confocal microscopy (LSM800, Zeiss, Germany) and photographed. Fluorescence intensity was analyzed by ImageJ software (version six; National Institutes of Health).
Hematoxylin eosin (H&E) staining
At the end of treatment period, the sciatic nerve was obtained from each rat for H&E staining. The sections (3 μm) were dewaxed with xylene, and hydrated with graded ethanol (ethanol, 5 min; 95% ethanol, 2 min; 80% ethanol, 2 min; 70% ethanol, 2 min). After that, the hematoxylin staining (Jiangyuan Industry, Wuxi, batch No: 190,704) was carried out for 15 min. After differentiation for 30 s, the sections were soaked at 50°C for 5 min. Then, the sections were stained with eosin (20170309, Solarbio) for 40 s. After dehydration with ethanol and purification with xylene, the sections were observed under a light microscope (DM1000 LED, Leica, Germany).
Myelin staining
At the end of treatment period, the sciatic nerve was obtained from each rat for myelin staining. The embedded sections were dewaxed, and stained with myelin staining (G1030, Servicebio@, Wuhan, China) for 4 h at 65°C. After washing, the sections were differentiated to colorless in the background. After washing, the sections were dehydrated with ethanol and cleared with xylene. Neutral gum was used to seal the pieces. The results were observed under a light microscope and analyzed by ImageJ software.
Nissl staining
At the end of treatment period, the L4-L5 dorsal root ganglia (DRG) were obtained from each rat and used for Nissl staining. The samples were fixed with 4% paraformaldehyde, embedded with paraffin, and cut into 3 μm. The dewaxing and hydration were carried out according to the H&E method. Nissl staining solution (Toluidine blue method, 870,102, Chroma) was used to stain at 55°C for 30 min. After washing with distilled water, the sections were differentiated with 95% ethanol, dehydrated with ethanol, and purified with xylene. After sealing with neutral gum, the sections were observed under a light microscope (DM1000 LED, Leica, Germany).
Statistical analysis
SPSS 20.0 statistical analysis software (IBM, Chicago, IL, USA) was implemented to process the data, and all results were expressed as the mean ± standard deviation (‾X ± SD). One-way analysis of variance was exerted for data analysis among multiple groups, followed by Tukey’s test for post hoc analysis. p < 0.05 indicated that the difference was statistically significant.
Results
Changes of body weight in rats
The changes of body weight in rats. Compared with control group, *p < 0.05, **p < 0.01; Compared with model group, #p < 0.05, ##p < 0.01; Compare with AS-IV-low group, &p < 0.05, &&p < 0.01; Compare with AS-IV-medium group, %p < 0.05, %%p < 0.01.

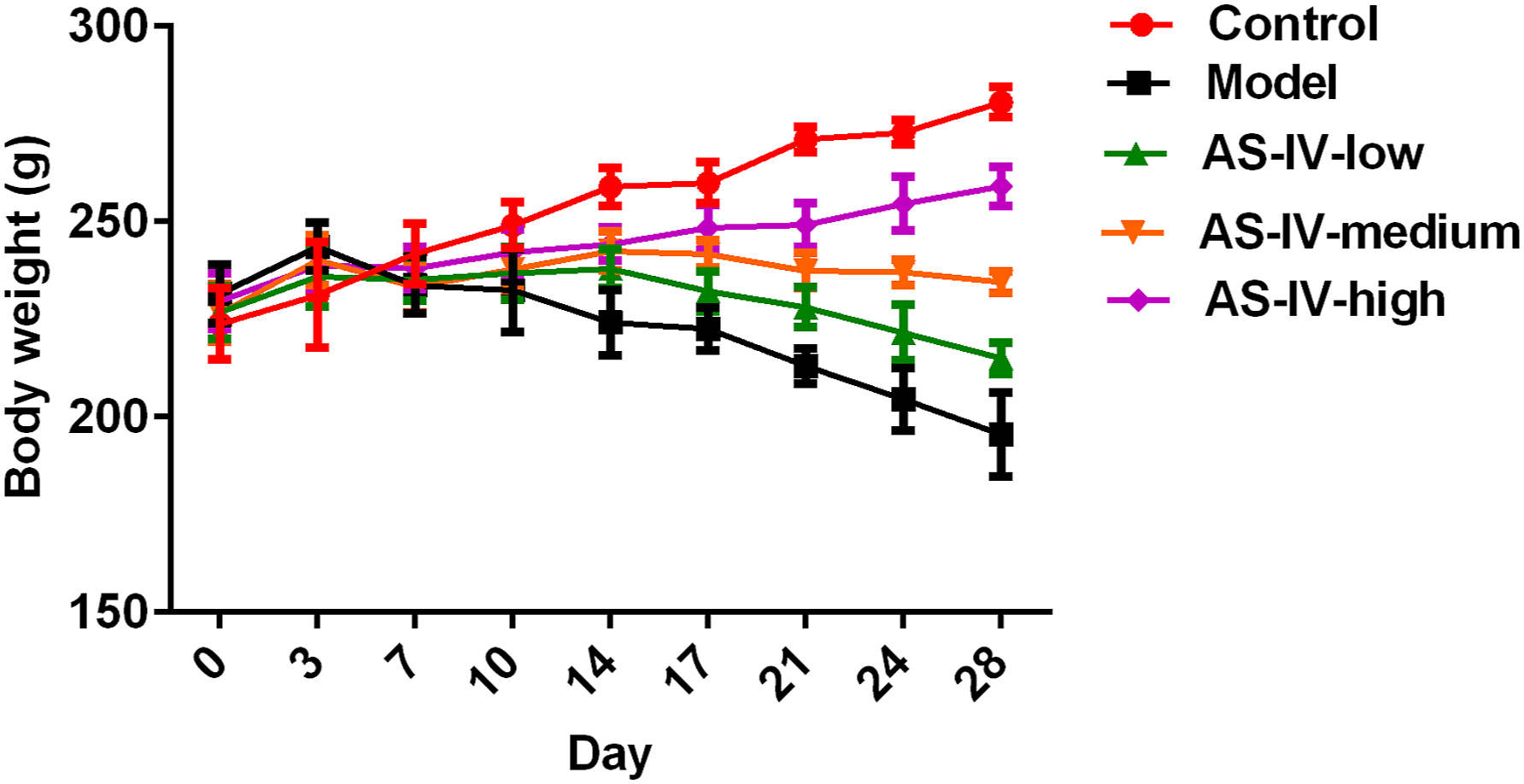
Effect of astragaloside IV (AS-IV) on body weight in rats. The statistical significances among groups were showed in Table 1.
AS-IV administration reduced oxaliplatin-induced mechanical and cold allodynia
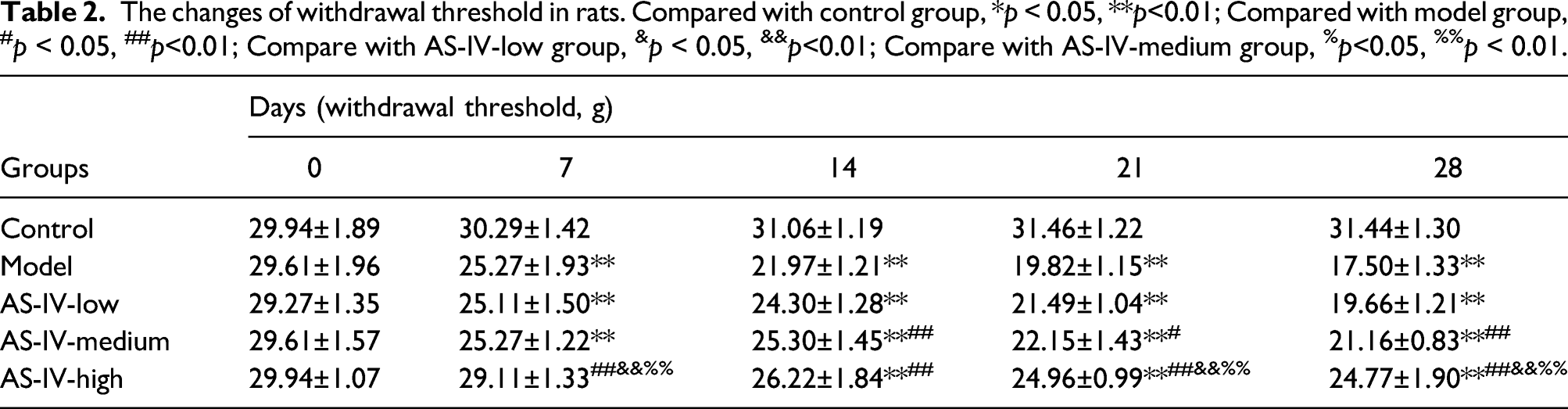
At beginning (0 days), there was no significant difference in the mechanical and cold allodynia among groups (Figure 2, Table 2 and Table 3). After 7 days of OIPN model, the mean withdrawal threshold (Table 2, Figure 2(a)) were decreased, and the amount time of rats spent licking, cowering, or shaking the acetone treated hind paw (Table 3, Figure 2(b)) were notably increased when compared with the control group (p < 0.01). Unfortunately, 5 mg/kg AS-IV administration did not ameliorate the oxaliplatin-induced mechanical and cold allodynia all the time. After 7 days of 20 mg/kg AS-IV treatment, mechanical and cold allodynia showed an effective recovery compared to the model group (p < 0.05). Lasting 14 days of 10 mg/kg AS-IV treatment, the mechanical and cold allodynia began to significantly improve compared with the model group (p < 0.05). After 21 days, we found that the analgesic effects in the AS-IV high group were better than the AS-IV medium group (Figure 2, p < 0.05). AS-IV administration reduced oxaliplatin-induced mechanical and cold allodynia. (a) The changes of withdrawal threshold; (b) The changes of time of rats spent licking, cowering, or shaking the acetone treated hind paw. Compared with control group, **p < 0.01; Compared with model group, #p < 0.05,##p < 0.01; Compare with AS-IV-low group, &&p< 0.01; Compare with AS-IV-medium group, %p < 0.05, %%p < 0.01. The changes of withdrawal threshold in rats. Compared with control group, *p < 0.05, **p<0.01; Compared with model group, #p < 0.05, ##p<0.01; Compare with AS-IV-low group, &p < 0.05, &&p<0.01; Compare with AS-IV-medium group, %p<0.05, %%p < 0.01. The changes of amount time of rats spent licking, cowering, or shaking the acetone treated hind paw in each group. Compared with control group,**p<0.01; Compared with model group, #p < 0.05, ##p<0.01; Compare with AS-IV-low group, &&p < 0.01; Compare with AS-IV-medium group, %p < 0.05.

AS-IV administration increased the levels of NGF and decreased the inflammation in OIPN model rats
At the end of treatment, the levels of NGF (Figure 3(a)), TNF-α (Figure 3(b)) and IL-6 (Figure 3(c)) in serum were measured using ELISA. The expression of Iba-1 in anterior horn of spinal cord was measured using immunofluorescence (Figure 3(d)). The data showed that the levels of NGF were clearly decreased, but the levels of TNF-α and IL-6 were obviously increased after rats were treated with oxaliplatin (p < 0.01). Additionally, the secretions of NGF were significantly increased, and the levels of TNF-α and IL-6were decreased, after rats were treated with different concentrations of AS-IV (p < 0.01). The mean gray value of Iba-1 was also increased after treated with oxaliplatin compared with control group (p < 0.01). After treatment with different concentrations of AS-IV, the mean gray value of Iba-1 was decreased. At the same time, the effectiveness of AS-IV treatment was increased with its increase of concentration. AS-IV administration increased the levels of NGF, decreased the inflammation in OIPN model rats. (a) The levels of NGF in serum; (b) The levels of TNF-α in serum; (c) The levels of IL-6 in serum; (d) The activation of Iba-1 in anterior horn of spinal cord (red represented Iba-1, blue represented DAPI). Compared with control group, *p < 0.05,**p < 0.01; Compared with model group, #p < 0.05,##p < 0.01; Compare with AS-IV-low group, &p < 0.05, &&p < 0.01; Compare with AS-IV-medium group, %p < 0.05, %%p < 0.01.
AS-IV administration improved the oxaliplatin-induced sciatic nerve injury
At the end of treatment period, the pathologic changes of sciatic nerve in each group were observed using H&E staining (Figure 4(a)), and the changes of myelin sheath in each group were observed using myelin staining (Figure 4(b)). In the control group, the axons and myelin sheath of most nerve fibers were intact. We found oxaliplatin injection destroyed the structure of nerve fibers, which made axons (red arrows) disappearance and myelin sheath (black arrows) deformation. After administration of different concentrations of AS-IV, the damage degrees of sciatic nerve fibers were improved with the increase of AS-IV dose. Especially, the axons and myelin sheath were clearly observed in the AS-IV high group compared with the model group. AS-IV administration improved the oxaliplatin-induced sciatic nerve injury. (A) H&E staining; (B) Myelin staining. Scale bar = 50 μm. The black arrows represented myelin sheath. The red arrows represented axon. Compared with control group, *p < 0.05, **p < 0.01; Compared with model group, #p < 0.05, ##p < 0.01; Compare with AS-IV-low group, &p < 0.05.
AS-IV administration improved the oxaliplatin-induced DRG injury
The morphological alteration of DRG in each group was observed at the end of treatment period using Nissl staining (Figure 5). After oxaliplatin injection, some DRG cells appeared vacuolar degeneration (black arrows). Compared with the control group, the numbers of degenerative DRG cells were significantly increased in the other groups (p < 0.01). After 10 mg/kg or 20 mg/kg AS-IV treatment, the numbers of degenerative DRG cells were clearly decreased when compared to the model group (p < 0.05). Especially, there were significant differences among the three AS-IV treatment groups. The damaged DRG cells were ameliorated in the AS-IV high group when compared with the other AS-IV treatment groups (p < 0.05). AS-IV administration improved the oxaliplatin-induced DRG injury. The black arrows represented the vacuolar degeneration of DRG cells. The numbers of degenerative DRG cells were counted in the graph (Scale bar = 50 μm). Compared with control group, *p < 0.05, **p < 0.01; Compared with model group, #p < 0.05, ##p<0.01; Compare with AS-IV-low group, &p < 0.05, &&p < 0.01; Compare with AS-IV-medium group, %p<0.05, %%p < 0.01.
Discussion
Oxaliplatin, in combination with 5-fluoruracil and folinic acid, is the stand for care for advanced colorectal cancer and gastrointestinal cancer. 19 Diagnostic advances and effective drugs such as oxaliplatin have allowed patients with cancer to increase the survival. Therefore, chronic or “toxic effects” (i.e., lasting more than 12 months or occurring 12 months after the end of chemotherapy) have become an urgent problem as a consequence of their effect on the quality of life of survivors.20,21 Among these toxicities, the CIPN is one of the main dose-limiting toxicities for oxaliplatin. In this study, we used AS-IV to treat the oxaliplatin-induced CIPN rats for accessing its feasibility. The results showed that the body weights of rats increased, and the mechanical and cold allodynia were improved with the increase of AS-IV concentrations, suggesting that AS-IV treatment could relieve the OIPN.
In order to further confirm that AS-IV could improve the OIPN, we measured the levels of NGF, TNF-α, and IL-6 in serum of rats using ELISA, and observed the activation of glial cells in the anterior horn of spinal cord using immunofluorescence. It is known that NGF is associated with Alzheimer’s disease and intractable pain, which could combine the tropomyosin receptor kinase (Trk) A. The NGF/TrkA signaling supports the survival and maintenance of NGF-dependent neurons in the peripheral nervous system.22, 23 In neuropathic pain of various etiologies, including OIPN, abnormal communication between neurons and glial could arise from the increasing of neuro-immune activation, e.g. the production of glial derived inflammatory cytokines. 24 In our study, AS-IV administration significantly inhibited the activation of Iba-1, companied the increase of NGF and decrease of TNF-α and IL-6 in OIPN rats, indicating AS-IV has anti-inflammatory and neuronal repair functions.
Furthermore, we observed the pathologic changes of sciatic nerve by H&E staining, and examined the morphological alteration of DRG through Nissl staining. According to previous reports, AS-IV could attenuate chronic constriction injury (CCI) induced neuropathic pain through regulation adenosine triphosphate-sensitive potassium (KATP)-JNK signaling, 18 and downregulation of the expressions of a series of proteins involved in mediating neuropathic pain in the DRG. 25 It has reported that AS-IV reduced the neuroinflammation and improved the Nissl bodies in the spinal cord of oxaliplatin-induced rats. 26 However, the effects of AS-IV on oxaliplatin-induced sciatic nerve and DRG injury are not observed. In our study, our data found that oxaliplatin could induce sciatic nerve and DRG injury, and AS-IV repaired the damaged sciatic nerve and degenerative DRG cells, to a certain extent, suggesting that the repairing function of neurons for AS-IV in OIPN. The damage induced by oxaliplatin was evident in this study, which is differently showed in other works where only occurrence of multinucleated neurons and the nucleolar eccentricity were caused in the DRG, 27 as regards myelin sheet was not seem interested, 28 suggesting that a higher damage induced by oxaliplatin in the model of this study. Considering the Km factor 37 for the conversion of animal doses to the Human Equivalent Dose, 29 the daily repeated administration of 4 mg/kg oxaliplatin performed in the rats allows to obtain a dosage of corresponding to 23.98 mg/m2. Usually 85 mg/m2 for each administration of oxaliplatin in the clinical practice, 29 the model used for the present research was consistent with the clinical practice.
However, we only preliminary studied the effects of AS-IV on OIPN in rats, more experiments are needed to study the mechanisms of AS-IV on OIPN. For example, AS-IV could inhibit mitochondrial-dependent apoptosis of the DRG in diabetic peripheral neuropathy rats through modulation of sirtuin/p53 pathway. 12 Whether sirtuin/p53 pathway is participated in the OIPN, and whether AS-IV is improved the OIPN via regulation Sirtuin/p53 pathway are unclear. Oxaliplatin induces oxidative stress in the spinal cord to explain a pathway by which it exerts the neuropathic pain. 11 On this regard, antioxidants molecules such as vitamin E or trolox will perform to provide if the AS-IV effects is due to antioxidant properties. Additionally, the efficacy of AS-IV on OIPN was only observed at the end of treatment period, the efficacy of AS-IV at different time need to evaluate.
In this study, there was no any power calculation was done for estimation of sample size in the methods, which is a limitation of this study. However, the pain relieving efficacy of AS-IV is not full, also the high dosage does not completely revert the oxaliplatin-induced pain, on the contrary some extracts (with the phyto complex) reaches a compete reversion, 30 suggesting the relevance to the extract instead of a single component.
Conclusions
Our research confirmed that AS-IV could recover the damaged sciatic nerve and degenerative DRG cells and reduce the mechanical and cold allodynia in OIPN rats, providing a potential treatment drug for oxaliplatin neurotoxicity.
Footnotes
Authors’ contributions
YL and SL carried out the experimental work and the data collection and interpretation.
YL and AH participated in the design and coordination of experimental work, and acquisition of data.
YL, SL and LD participated in the data collection, analysis of data and preparation of the manuscript.
All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Project of Yantai Science and Technology (2020MSGY086), the Key Research and Development Project of Shandong Province (2019GSF108245), and Yantai Municipal Science and Technology Project (2020YD033).
Ethical approval
This study was approved by the Yantaishan Hospital Animal Protection and Use Committee (Approval No. 20210017)*. (2) Animal welfare. The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
