Abstract
The minipig has been used as a non-rodent species in nonclinical toxicology studies, but little is known about amyloid A (AA) amyloidosis in this species. Among domestic pigs, reports of AA amyloidosis have been limited to animals with mutations in the N-terminal residue of serum AA (SAA), which is thought to be a primary etiological factor. In this study, we histologically examined 26 microminipigs aged 0.6 to 10 years and observed amyloid deposition in one 0.6-year-old and six 5-year-old or older microminipigs. The amyloid deposits were identified as AA based on mass spectrometry (MS) and immunohistochemistry (IHC). The 0.6-year-old microminipig showed severe deposition in the renal cortex and spleen, whereas 5-year-old or older animals had severe deposition in the renal medulla. MS and IHC detected serum amyloid P-component (SAP) in amyloid deposits in older animals but not in a 0.6-year-old animals. Based on the proteomic analysis and gene sequencing, amino acid mutations of SAA, previously found in domestic pigs, were not involved in the pathogenesis of AA amyloidosis in microminipigs. This study demonstrates that microminipigs with wild-type SAA develop AA amyloidosis and presents the possibility that differences in the environment surrounding amyloid, such as SAP, may influence differences in the pathological phenotype.
Keywords
Introduction
The minipig has been used as a non-rodent species in nonclinical toxicology studies, but little is known about amyloid A (AA) amyloidosis in this species. Amyloid A amyloidosis is caused by the aggregation of amyloid fibrils derived from serum AA (SAA) in the systemic tissues. 21 Although elevated SAA levels in circulation due to infection or chronic inflammation are prerequisites for the development of AA amyloidosis, it is not uncommon for the precursor disease to be absent. Amyloid A amyloidosis is an extremely rare disease in pigs, and amyloid deposition is categorized as “uncommon” or “not observed, but potentially relevant” change in minipigs used in toxicity studies, 19 even though it is observed in livestock, such as cattle,11,22 hens, 12 ducks, 20 and quails. 14 Niewold et al 16 analyzed the N-terminal residues of AA peptides extracted from an AA amyloidosis-affected domestic pig and found two amino acid deletions. Kamiie et al 4 showed that this mutant AA peptide forms amyloid fibrils in vitro, suggesting that this unique SAA sequence may play an essential role in developing AA amyloidosis in pigs. These previous studies suggest that the development of AA amyloidosis in pigs requires the SAA mutation and may otherwise be AA amyloidosis-resistant. Meanwhile, these previous studies analyzed pigs younger than two years of age, and AA amyloidosis in older pigs has not been searched for to date.
The microminipig is the smallest laboratory pig, weighing less than 10 kg at 6 months of age and less than 80 cm in length, developed by crossbreeding pot-bellied pigs and another type of minipig by Fuji Micra Inc. 5 The genome sequences of microminipigs and domestic pigs are very similar and have been used as a compact alternative model for domestic pigs. 9 Except for differences in blood coagulation characteristics, the main hematological and biochemical parameters of the microminipigs are similar to those in Göttingen and Yucatan minipigs, 7 and they also develop similar age-related changes. 6 We previously analyzed 27 microminipigs aged 3 to 10 years and found amyloid deposition in the liver or kidney in 4 microminipigs older than eight. 6 However, the amyloid precursor proteins of amyloidosis in those microminipigs were not identified. In addition to previous amyloidosis cases, we found a new case of systemic amyloidosis in a 0.6-year-old microminipig. Therefore, in the present study, 26 microminipigs aged 0.6 to 10 years, including the 25 microminipigs used in the previous study, 6 were analyzed again to identify the amyloid precursor protein and to compare the pathogenesis of amyloidosis in 0.6-year-old and older microminipigs. Elucidating the mechanism of AA amyloidosis in minipigs will help pathologists better understand the characteristics of the species used in toxicology studies.
Materials and Methods
Animal Information and Histological Analysis
Twenty-six microminipigs (13 females and 13 males) aged 0.6 and 10.7 years were analyzed in this study. The animals were born and raised in Fuji Micra Inc. or Shizuoka Prefectural Research Institute of Animal Industry Swine & Poultry Research Center. Animal information, macroscopic and microscopic findings are shown in Table S1. Animal 2 to 26 are animals with spontaneous age-related lesions investigated in the previous study. 6 Tissue processing from necropsy to paraffin-embedding was described in the previous study. 6 In animals 1, 17, 18, 21, and 26, in which amyloid deposition had already been confirmed, systemic organs were examined, as shown in Figure 1. In other animals, the liver, kidneys, spleen, and thyroid/adrenal glands, which are amyloid deposition-preferential organs, 6 were examined to revalidate the presence of amyloid deposition. Formalin-fixed paraffin wax-embedded (FFPE) tissues were cut into 3 µm and subjected to hematoxylin and eosin staining, Congo red staining, and immunohistochemistry (IHC). This study was approved by the institutional review board and conducted in accordance with humane animal care measures as previously described. 6
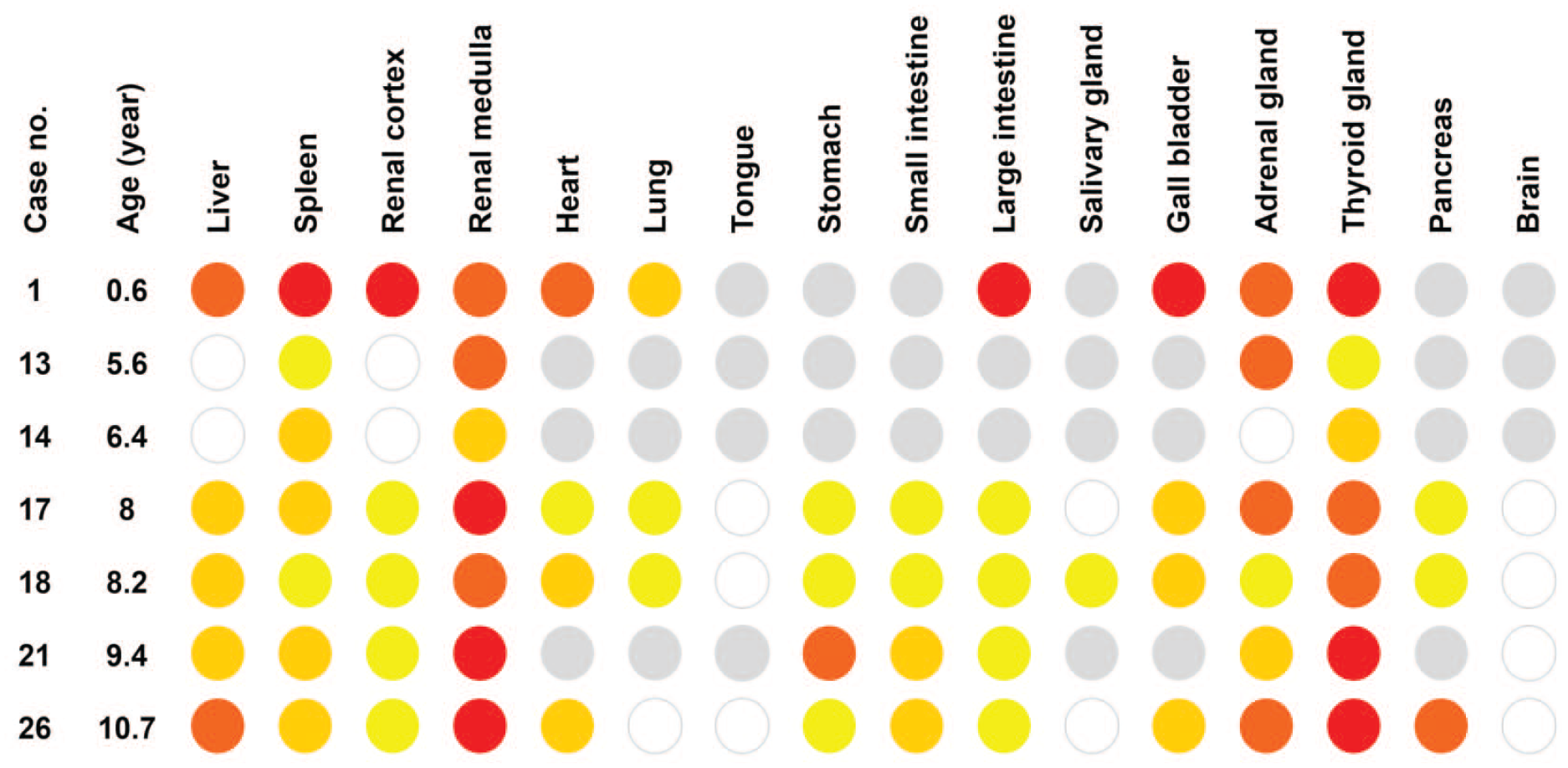
Distribution of amyloid deposition in amyloid-positive microminipigs. The intensity of amyloid deposits was shown in the following colors: white, no deposition; yellow, deposition in few vessels; light orange, deposition in several vessels; dark orange, deposition in almost all vessels and perivascular areas; red, severe deposition in vessels and interstitial tissues; gray, not tested.
A heatmap was prepared to show the intensity and distribution of systemic amyloid deposition. The colors were assigned according to the degree of amyloid deposition as follows: white, no deposition; yellow, deposition in a few vessels; light orange, deposition in several vessels; dark orange, deposition in almost all vessels and perivascular areas; red, severe deposition in vessels and interstitial tissues.
Tissue Microdissection and Proteomic Analysis
Amyloid deposits were collected from FFPE specimens of the kidneys (animals 1, 17, 18, 21, 26) and a spleen (animal 1). Tissue microdissection, sample processing, tryptic digestion, dithiothreitol reduction, and liquid chromatography-tandem mass spectrometry (LC-MS/MS) of the digested peptides were performed as described previously. 13 The resulting MS/MS data were collated with the theoretical fragmentation patterns of tryptic peptide sequences of porcine proteins registered in the UniProt database using the Mascot Server (Matrix Science Inc., Boston, MA, USA). “semiTrypsin” was used as the in silico proteolytic enzyme. After an error-tolerant search, oxidation of methionine, deamidation of asparagine and glutamine, pyroglutamylation of glutamine and glutamic acid, and formylation and methylation of lysine were selected as the in silico variable modifications. Statistically significant proteins and peptides (P < .05, score > 20) were extracted using Mascot’s probability-based scoring algorithm. 18
To verify which SAA region was detected, the MS/MS data were again collated with a home-grown database (Table S2). This database contains porcine SAA sequences from UniProt, and the SAA N-terminus sequence (QSFVGEAYEGAKDMWR) detected in AA amyloidosis pigs in previous studies.4,16 A heat map was created based on each peptide’s detection frequency.
Immunohistochemistry
For IHC, anti-human SAA antibody (Cloud-Clone, polyclonal rabbit, dilution 1:200), anti-canine apolipoprotein E (ApoE) antibody (Cloud-Clone, polyclonal rabbit, dilution 1:50), anti-human apolipoprotein A-IV (ApoA-IV) antibody (Cloud-Clone, polyclonal rabbit, dilution 1:200), anti-human serum amyloid P-component (SAP) antibody (Cloud-Clone, polyclonal rabbit, dilution 1:50) were used for the primary antibody. For antigen retrieval, specimens were autoclaved in deionized water at 121°C for 10 minutes prior to reaction with the primary antibodies. Horseradish peroxidase-labeled polymer anti-rabbit immunoglobulin antibody (Dako, CA, USA) was used as a secondary antibody. A diaminobenzidine-4HCl substrate kit (Liquid DAB + Substrate Chromogen System, Dako) and hematoxylin were used for color development and counterstaining, respectively. For the negative control, the primary antibody was omitted. Positive controls were not used.
Gene Sequencing
Gene sequencing was performed to determine the SAA2 gene sequence in amyloidosis-positive microminipig. One-step reverse transcription-polymerase chain reaction (RT-PCR) was performed using a PrimeScript One-Step RT-PCR Kit (Takara Bio, Shiga, Japan). Primer sequences (5′-3′) designed using Primer Blast in NCBI based on the mRNA sequences of Sus scrofa familiaris SAA2 (accession: XM_005661095.3; encoding protein ID in NCBI: XP_005661152.2) are as follows: forward, CCTGACCCTCTGTTGTCATTTC; reverse, GGCATGGAAGTACTTGTCTGAA. Since fresh samples were disposed of in all microminipigs in this study, total RNA was extracted from FFPE tissue sections using the innuPREP FFPE total RNA Kit (Analytik Jena). RNA extraction was performed for all amyloidosis-affected microminipigs, but RNA of sufficient quality could be extracted in only animal 17. RT-PCR was performed as follows: reverse transcription at 50°C for 30 minutes, followed by 40 cycles of 94°C for 30 seconds, 57°C for 30 seconds, and 72°C for 1 minute, followed by a final extension at 72°C for 3 minutes. The product size was analyzed using agarose gel electrophoresis, and target DNA fragments were purified from the gel using NucleoSpin Gel and PCR Clean-up (MACHEREY-NAGEL, Düren, Germany). The sequence of the purified DNA was obtained using an outsourcing service from Takara Bio Inc. Pairwise sequence alignment of sequenced results from animal 17, and the mRNA sequences of Sus scrofa familiaris SAA2 (accession: XM_005661095.3) were analyzed using EMBOSS Needle. 15 The amino acid sequence was translated from the sequenced codons using EMBOSS Transeq. 8
Results
Histological Analysis
Histologically, amyloid deposition was observed in 7 of 26 animals. Four amyloid-positive animals, animals 17, 18, 21, and 26, were the same as in the previous report. 6 Animals 13 and 14 were those for which amyloid was not detected in the previous report 6 but was found to be amyloid-positive in the current study. Animal 1 was a newly examined animal in the current study. The age of the amyloid-positive microminipigs were 0.6, 5.6, 6.4, 8, 8.2, 9.4, and 10.7 years. Amyloid-positive microminipigs, except animals 1 and 13, had defined underlying diseases such as lung abscesses (Table S1, S3).
The distribution of amyloid deposition in the seven amyloid-positive microminipigs is shown in Figure 1. Histological amyloid deposition pattern in each organ was as follows: liver, Glisson’s sheath, and space of Disse; spleen, around central arteries. In animal 1, also in the red pulp; kidney, glomeruli, and interstitial tissue around small vessels and tubules; adrenal gland, mainly in the cortex, interstitial tissue surrounding vessels; thyroid gland, vessels between the follicles and in the perivascular areas; digestive tract, submucosal vessels and the connective tissue between the crypts; other organs, vessel walls, and perivascular area.
Animal 1, at age 0.6 years, showed severe deposition in the liver, spleen, and renal cortex (Figure 2). In contrast, in animals 13, 14, 17, 18, 21, and 26, aged 5 years and older, hepatic and splenic amyloid deposition were milder than in animal 1. In addition, these microminipigs showed none or mild renal cortical amyloid deposits and severe deposition in the renal medulla.
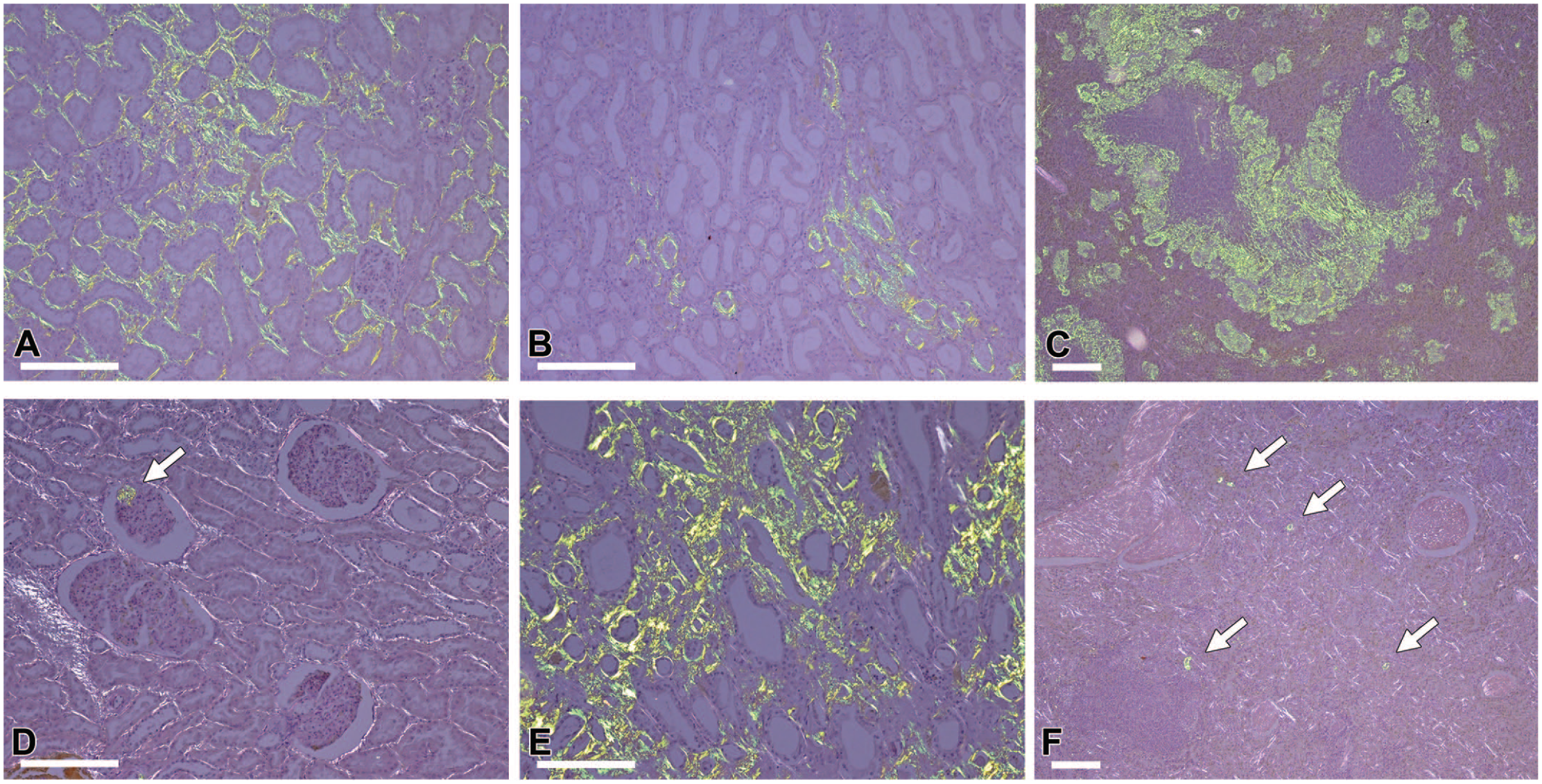
Histological features of amyloid deposits in young and older microminipigs. Congo red stain under polarized light. (A-C) Amyloid deposits in animal 1. In the 0.6-year-old microminipig, amyloid deposits were severe in the renal cortex (A) and spleen (C) and moderate in the renal medulla (B). (D-F) Amyloid deposits in animal 26. Amyloid deposits were severe in the renal medulla (E) but mild in the renal cortex (D) and spleen (F). Arrows indicate amyloid deposits. Bars, 200 µm.
Proteomic Analysis
Table 1 shows representative proteins detected by MS. SAA2 was detected in all microminipigs with a high level. Serum amyloid P-component and ApoE, known as amyloid signature proteins,10,17 were detected from amyloid deposits in animals 17, 18, 21, and 26 but were either not detected or at low levels in animal 1. Trypsinogen was detected in all samples but was removed from the table because it was thought to be derived from the trypsin used in sample processing.
Representative proteins detected by mass spectrometry of amyloid deposits and summary of immunohistochemistry results.
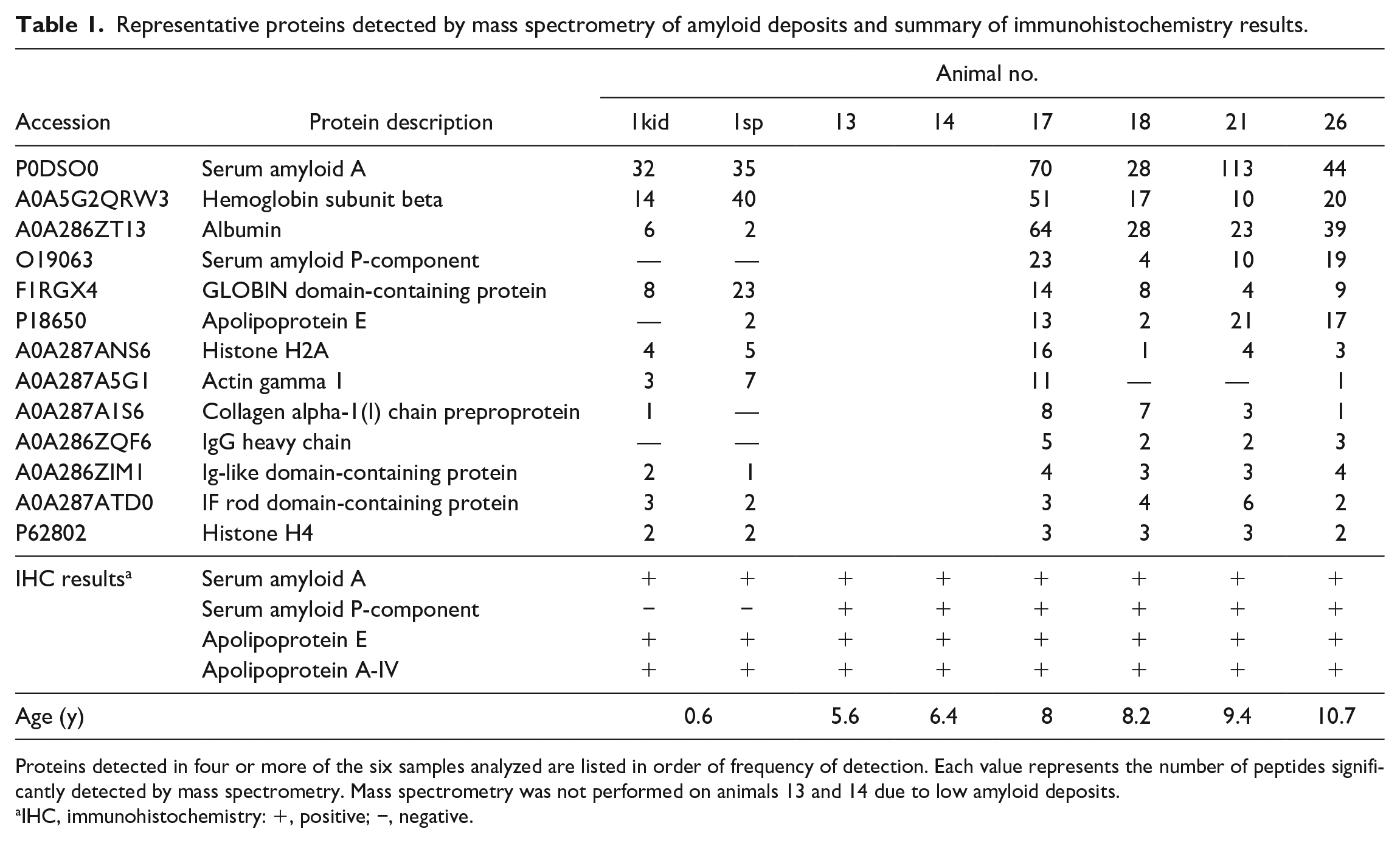
Proteins detected in four or more of the six samples analyzed are listed in order of frequency of detection. Each value represents the number of peptides significantly detected by mass spectrometry. Mass spectrometry was not performed on animals 13 and 14 due to low amyloid deposits.
IHC, immunohistochemistry: +, positive; −, negative.
In all microminipigs, signal peptides (Met1-Ser18) of SAA2 were not detected. On the other hand, Glu19-Lys36 and Gly57-Lys90 were detected frequently (Figure 3). There is no apparent difference in the detected peptides between 0.6-year-old and older microminipigs.
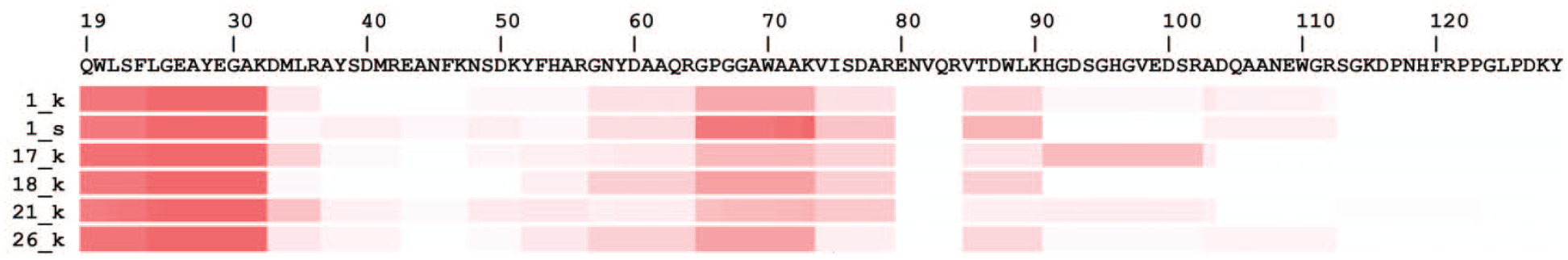
Mapping of AA peptides detected by mass spectrometry. Sheer numbers on the left indicate animal numbers. “k” and “s” stand for samples derived from the kidney and spleen, respectively. The heatmap shows the detection frequency of SAA-derived peptides by LC-MS/MS. Frequency is described as white (low) to red (high). Regions with high frequency are considered to be amino acid sequences with strong involvement in amyloid formation. AA indicates amyloid A; LC-MS/MS, liquid chromatography-tandem mass spectrometry; SAA, serum AA.
The mutant SAA N-terminal sequence QSFVGEAYEGAK (Figure 4A) reported in previous studies4,16 was not detected in the present samples. In other words, the SAA N-terminal peptides detected in all samples were derived from wild-type SAA (Figures 4B, 4C).
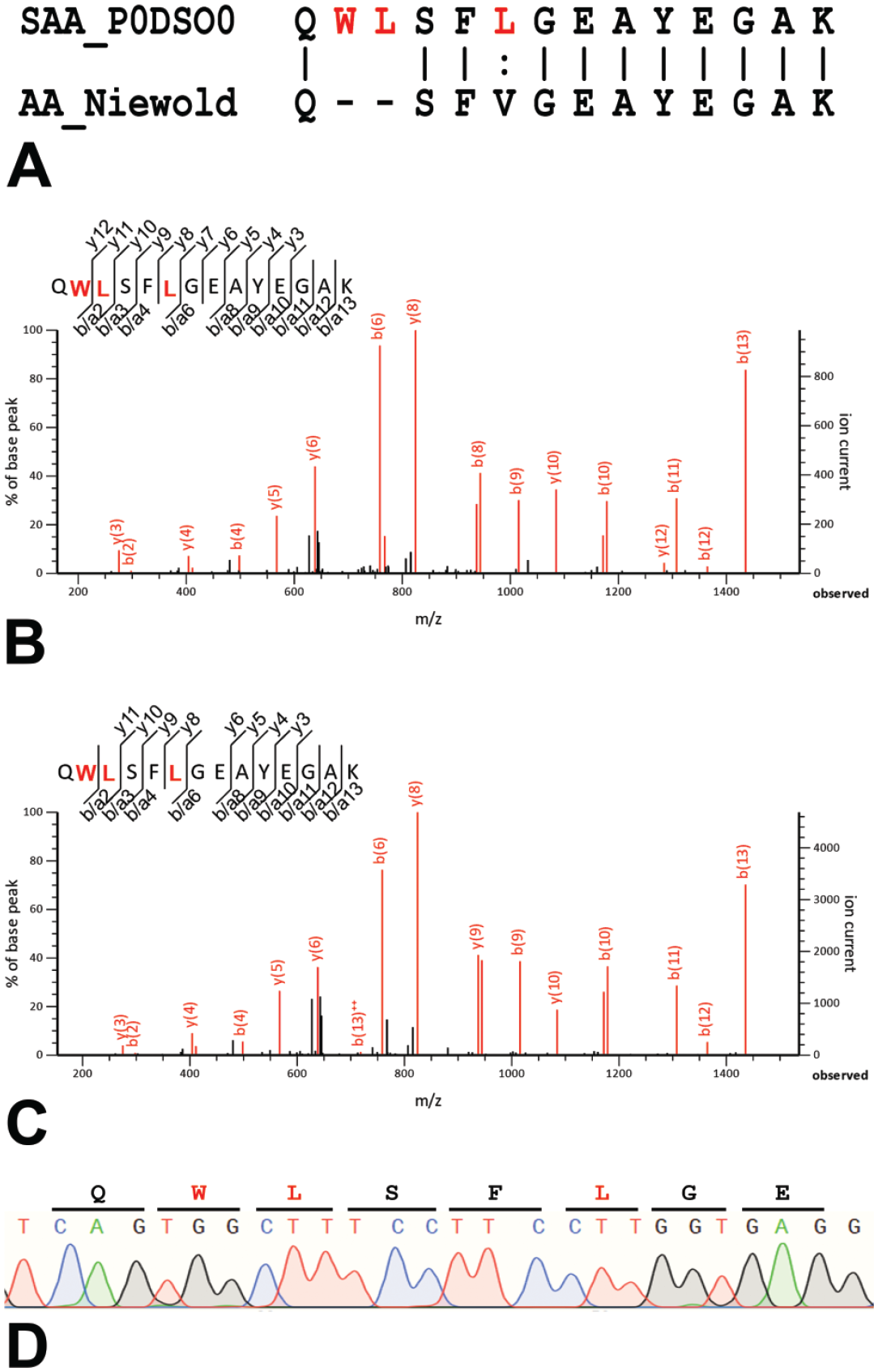
N-terminal sequence of SAA in microminipigs. (A) Pairwise alignment of wild-type SAA (Swiss-Prot accession: P0DSO0) and amyloid-forming mutant SAA. 16 Vertical bars (|) indicate fully conserved residues. Colon (:) indicates conserved residues with strongly similar properties. Hyphens (-) indicate gaps. (B, C) Mass spectrum of N-terminal peptides of SAA detected from renal amyloid deposits in animals 1 (B) and 26 (C). (D) The partial DNA sequence of SAA in animal 17. There was no mutation identified. SAA indicates serum AA.
Immunohistochemistry
In all seven amyloid-positive microminipigs, amyloid deposits were positive for SAA (Figures 5A, 5E). In these microminipigs, AA amyloid deposits in systemic organs led to the definitive diagnosis of AA amyloidosis. Amyloid deposits were also positive for ApoE (Figures 5C, 5G) and ApoA4 (Figures 5D, 5H) in both 0.6-year-old and older microminipigs. In contrast, SAP was positive in older microminipigs (Figure 5F) but negative in a 0.6-year-old microminipig (Figure 5B).
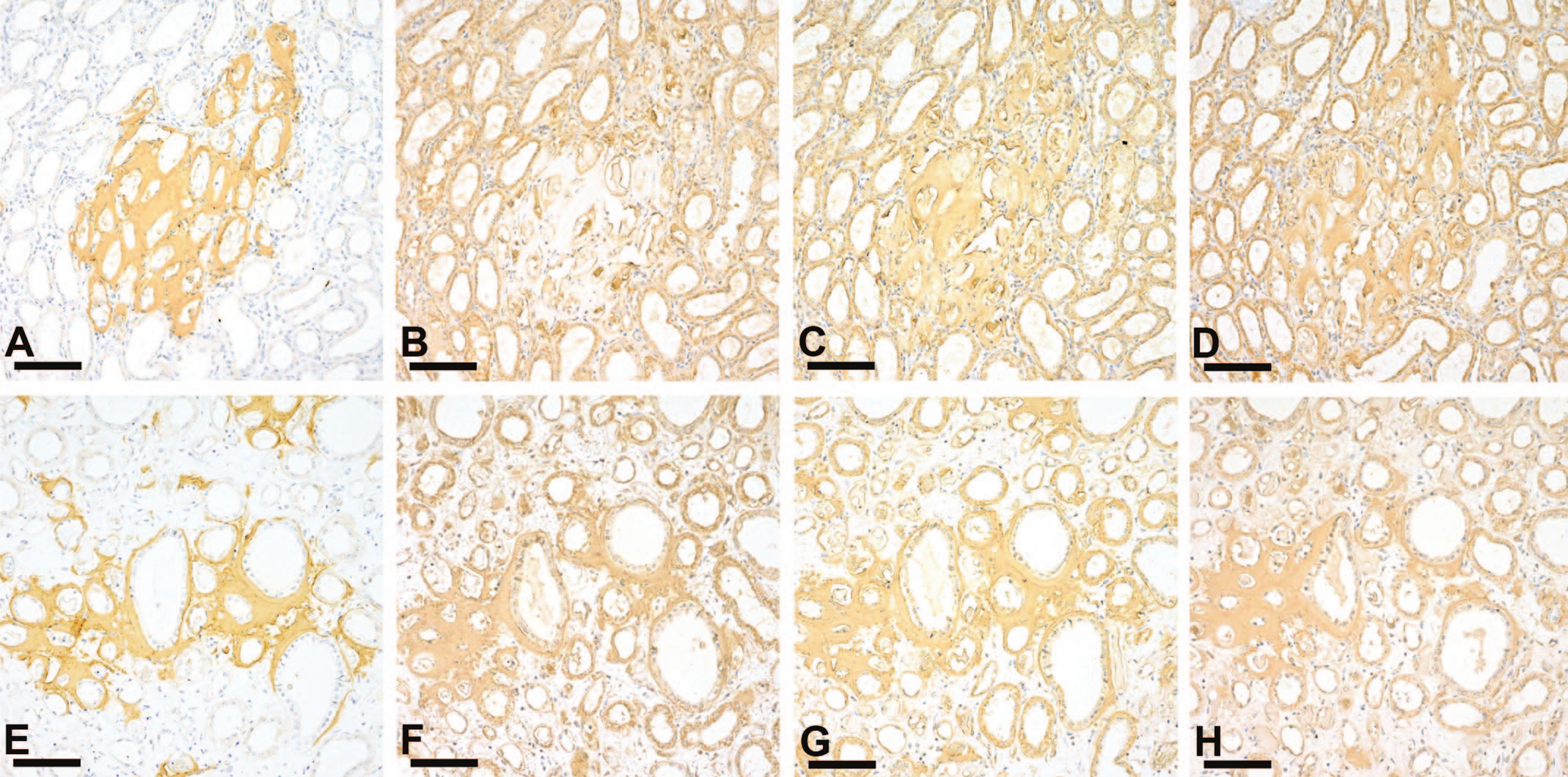
Immunohistochemistry of amyloid deposits in young and older microminipigs. (A-H) Immunohistochemistry of serial sections of amyloid deposits in the renal medulla of animals 1 (A-D) and 18 (E-H). In case 1, 0.6-year-old microminipig, amyloid deposits were positive for SAA (A), ApoE (C), and AopA4 (D) but negative for SAP (B). In animal 18, 8-year-old microminipig, amyloid deposits were positive for SAA (E), SAP (F), ApoE (G), and ApoA4 (H). Bars, 100 µm. ApoE indicates anti-canine apolipoprotein E; SAP, serum amyloid P; SAA, serum AA.
Gene Sequencing
As a result of gene sequencing of animal 17, N-terminal sequence QWLSFLGEAYEGAK in SAA was determined (Figure 4D), which matches with wild-type SAA sequence in pigs (UniProt accession: P0DSO0).
Discussion
Previous studies have reported that in pigs affected with AA amyloidosis,4,16 the N-terminal sequence of SAA is QSFVGEAYEGAK (Figure 4A), missing two amino acids (leucine and serine) from the wild-type N-terminal sequence QWLSFLGEAYEGAK; this unique sequence has been thought to be necessary for pathogenesis. In this study, however, LC/MS/MS and genetic analyses revealed the QWLSFLGEAYEGAK sequence, and QSFVGEAYEGAK was not detected (Figures 4B-4D), indicating that mutant SAA is not necessarily required for the development of AA amyloidosis in microminipigs. In the present study, AA amyloid deposition was found in 6 of 16 (38%) of the microminipigs aged 5 years or older, and the deposition was more severe in older animals. In many animal species, AA amyloidosis develops in mature to elderly animals,1,2,22 suggesting that aging also plays a vital role in developing AA amyloidosis in microminipigs. Unlike dairy cattle, which are raised for long periods and often develop amyloidosis,11,22 the typical meat pig is shipped at approximately 150 to 180 days, and the minipigs are also subjected to toxicologic study at approximately this age. One of the reasons for the low incidence of amyloidosis in pigs may be that most pigs are slaughtered or euthanized before they reach the age of onset of the disease.
Because the precursor protein, SAA, is an inflammatory protein, AA amyloidosis is commonly secondary to inflammatory diseases, except in some idiopathic or familial conditions.4,12,14 In this study, inflammation was histologically observed in amyloidosis-affected animals. The development of AA amyloidosis secondary to inflammatory conditions seems to be common in microminipigs, as well as other animals. On the other hand, not all animals with inflammation showed amyloid deposition, suggesting that there may be other etiologies besides inflammation and aging.
Interestingly, renal and splenic amyloid deposition patterns differed between the 0.6-year-old and older microminipigs, suggesting that the mechanism of pathogenesis may differ between young and older animals. In this study, we specifically focused on the changes in SAA protein to determine the reason for this difference. There were no apparent differences in the detected peptide sequence of SAA in 0.6-year-old and older microminipigs, suggesting that the differences in amyloid deposition patterns between young and older microminipigs are not due to differences in amino acid mutation or amyloidogenic regions in SAA. On the other hand, MS and immunohistochemistry revealed that SAP co-deposits in amyloid deposits in older animals but not in the 0.6-year-old microminipig. In humans, SAP is defined as an amyloid signature protein that co-localizes with amyloid and is closely associated with various amyloidosis pathologies. 17 However, SAP has not been detected in canine or feline amyloid,3,10 and the role of SAP in animal amyloidosis is unknown. Interestingly, in this study, the co-localization of SAP in amyloid lesions differed between the 0.6-year-old and older pigs. Differences in the environment surrounding the AA protein, such as SAP, rather than the AA protein itself, may be responsible for the differences in pathogenesis between young and older animals. It should be noted, however, that this study examined only one 0.6-year-old microminipig as a young animal affected with AA amyloidosis. More affected animals need to be analyzed to scrutinize the differences in pathogenesis between young and aged animals.
Of the six amyloidosis-affected microminipigs over 5 years of age, two animals were males aged 5.6 and 6.4 years old, and four were females over 8 years old. However, because of the skewed sex ratio investigated in the different age bands (males predominate in the 3-6 age group and females in the 7-10 age group), it is difficult to mention sex differences in the development of AA amyloidosis.
The present study revealed that mutant SAA was not a prerequisite in developing AA amyloidosis in microminipigs and that there was a difference in the SAP co-localization in the amyloid lesions between young and older animals. Since understanding the characteristics of the species used in toxicology studies is essential for pathologists, further studies are needed to clarify the pathogenesis of AA amyloidosis in microminipigs and to understand if there is any difference in pathological mechanisms among minipig strains and domestic pigs.
Supplemental Material
sj-xls-1-tpx-10.1177_01926233231204019 – Supplemental material for Pathological Characterization of Spontaneous AA Amyloidosis in Microminipigs
Supplemental material, sj-xls-1-tpx-10.1177_01926233231204019 for Pathological Characterization of Spontaneous AA Amyloidosis in Microminipigs by Misaki Inoue, Shinya Miyazaki, Natsumi Kobayashi, Akihisa Kangawa and Tomoaki Murakami in Toxicologic Pathology
Supplemental Material
sj-xls-2-tpx-10.1177_01926233231204019 – Supplemental material for Pathological Characterization of Spontaneous AA Amyloidosis in Microminipigs
Supplemental material, sj-xls-2-tpx-10.1177_01926233231204019 for Pathological Characterization of Spontaneous AA Amyloidosis in Microminipigs by Misaki Inoue, Shinya Miyazaki, Natsumi Kobayashi, Akihisa Kangawa and Tomoaki Murakami in Toxicologic Pathology
Supplemental Material
sj-xls-3-tpx-10.1177_01926233231204019 – Supplemental material for Pathological Characterization of Spontaneous AA Amyloidosis in Microminipigs
Supplemental material, sj-xls-3-tpx-10.1177_01926233231204019 for Pathological Characterization of Spontaneous AA Amyloidosis in Microminipigs by Misaki Inoue, Shinya Miyazaki, Natsumi Kobayashi, Akihisa Kangawa and Tomoaki Murakami in Toxicologic Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by JSPS KAKENHI (grant nos 22K19242 and 23H02380), the Program on Open Innovation Platform with Enterprises, Research Institute and Academia (OPERA) from JST, and the Smart-Core-Facility Promotion Organization at the Tokyo University of Agriculture and Technology.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
